Impact of Caterpillar Increased Feeding Rates on Reduction of Bt Susceptibility
Abstract
:1. Introduction
2. Results
2.1. Temperature Elicited Reduced Food Consumption Increased C. virescens Bt Susceptibility
2.2. Temperature Elicited Reduced Food Consumption Rate Increased H. zea Bt Susceptibility
2.3. Development of Methodology to Identify Naturally Occurring Slow and Fast Feeding H. zea Neonates
2.4. Impact of Feeding Rate at a Constant Temperature on Susceptibility to MVPII Cry1Ac
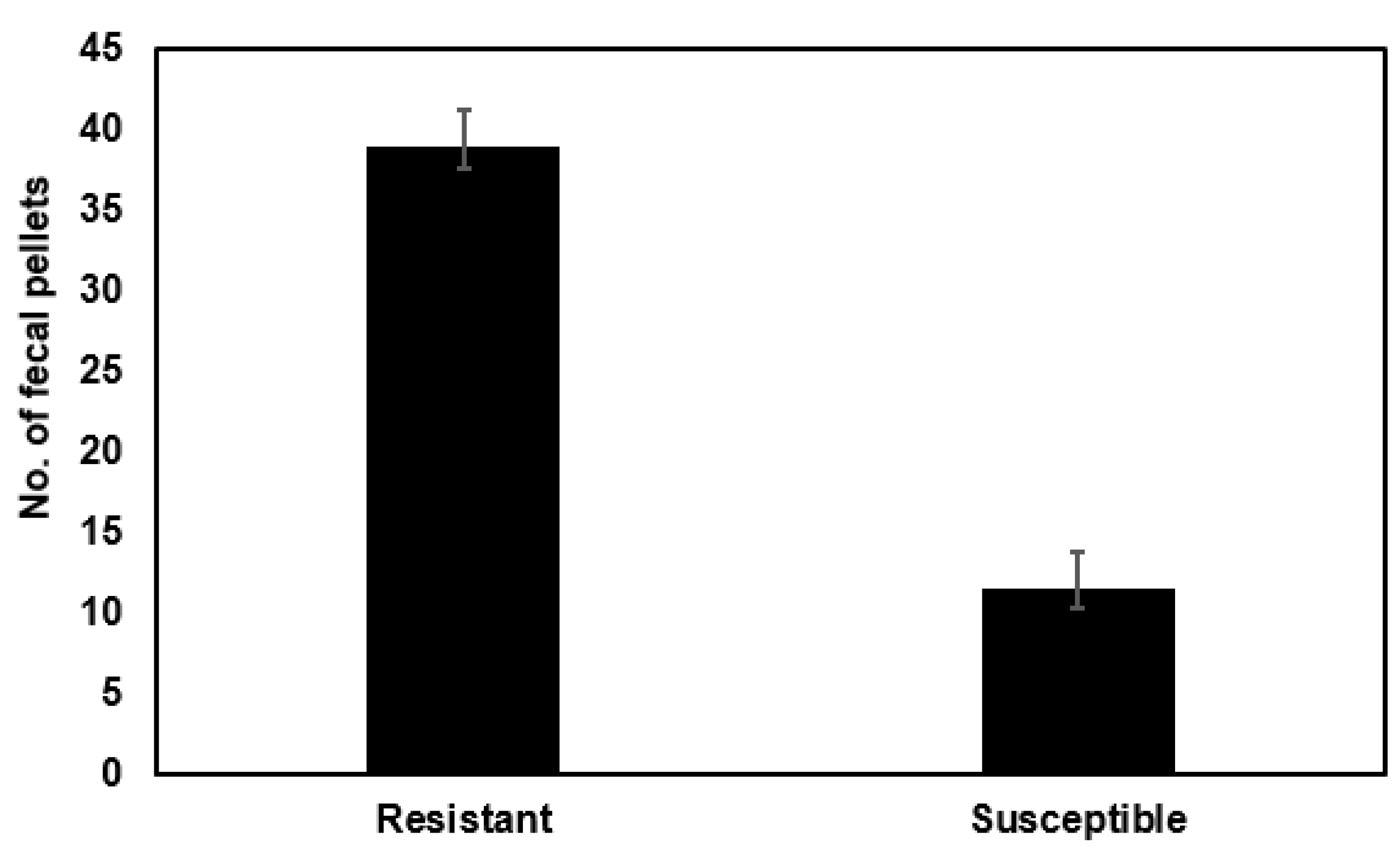
2.5. Differences in Feeding Rates between Bt Susceptible and Bt Resistant Caterpillar Strains on Non-Bt Artificial Diet
3. Discussion
4. Materials and Methods
4.1. Insects
4.2. Feeding Rate and Bt Susceptibility in Caterpillars Reared at Different Temperatures
4.3. Bollworm MVPII Cry1Ac Bt Toxin Susceptibility in Slow and Fast Feeders
4.4. Fecal Production Rate for Bt Susceptible versus Resistant Caterpillars
4.5. Data Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Christou, P.; Capell, T.; Kohli, A.; Gatehouse, J.A.; Gatehouse, A.M. Recent developments and future prospects in insect pest control in transgenic crops. Trends Plant Sci. 2006, 11, 302–308. [Google Scholar] [CrossRef] [PubMed]
- James, C. Global status of commercialized Biotech/GM crops. In ISAAA Brief No. 49; ISAAA: Ithaca, NY, USA, 2014. [Google Scholar]
- Yang, F.; Kerns, D.L.; Little, N.S.; Santiago González, J.C.; Tabashnik, B.E. Early warning of resistance to Bt toxin Vip3Aa in Helicoverpa zea. Toxins 2021, 13, 618. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Wei, J.; Ni, X.; Zhang, J.; Jurat-Fuentes, J.L.; Fabrick, J.A.; Carrière, Y.; Tabashnik, B.E.; Li, X. Decreased Cry1Ac activation by midgut proteases associated with Cry1Ac resistance in Helicoverpa zea. Pest Manag. Sci. 2018, 75, 1099–1106. [Google Scholar] [CrossRef] [PubMed]
- Gassmann, A.J.; Petzold-Maxwell, J.L.; Keweshan, R.S.; Dunbar, M.W. Field-evolved resistance to Bt maize by western corn rootworm. PLoS ONE 2011, 6, e22629. [Google Scholar] [CrossRef] [Green Version]
- Dively, G.P.; Venugopal, P.D.; Finkenbinder, C. Field-evolved resistance in corn earworm to Cry proteins expressed by transgenic sweet corn. PLoS ONE 2016, 11, e0169115. [Google Scholar] [CrossRef] [Green Version]
- Zhao, J.-Z.; Cao, J.; Li, Y.; Collins, H.L.; Roush, R.T.; Earle, E.D.; Shelton, A.M. Transgenic plants expressing two Bacillus thuringiensis toxins delay insect resistance evolution. Nat. Biotechnol. 2003, 21, 1493–1497. [Google Scholar] [CrossRef]
- Tabashnik, B.E.; Cushing, N.L.; Finson, N.; Johnson, M.W. Field development of resistance to Bacillus thuringiensis in diamondback moth (Lepidoptera: Plutellidae). J. Econ. Entomol. 1990, 83, 1671–1676. [Google Scholar] [CrossRef]
- Tabashnik, B.E.; Gassmann, A.J.; Crowder, D.W.; Carrière, Y. Insect resistance to Bt crops: Evidence versus theory. Nat. Biotechnol. 2008, 26, 199–202. [Google Scholar] [CrossRef] [PubMed]
- Tabashnik, B.E.; Van Rensburg, J.; Carrière, Y. Field-evolved insect resistance to Bt crops: Definition, theory, and data. J. Econ. Entomol. 2009, 102, 2011–2025. [Google Scholar] [CrossRef] [PubMed]
- Tabashnik, B.E. Evolution of resistance to Bacillus thuringiensis. Annual Rev Entomol 1994, 39, 47–79. [Google Scholar] [CrossRef]
- Gould, F.; Martinez-Ramirez, A.; Anderson, A.; Ferre, J.; Silva, F.J.; Moar, W.J. Broad-spectrum resistance to Bacillus thuringiensis toxins in Heliothis virescens. Proc. Natl. Acad. Sci. USA 1992, 89, 7986–7990. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gould, F.; Anderson, A.; Reynolds, A.; Bumgarner, L.; Moar, W. Selection and genetic analysis of a Heliothis virescens (Lepidoptera: Noctuidae) strain with high levels of resistance to Bacillus thuringiensis toxins. J. Econ. Entomol. 1995, 88, 1545–1559. [Google Scholar] [CrossRef]
- Bauer, L.S. Resistance: A threat to the insecticidal crystal proteins of Bacillus thuringiensis. Fla. Entomol. 1995, 78, 414–443. [Google Scholar] [CrossRef]
- Moar, W.; Roush, R.; Shelton, A.; Ferré, J.; MacIntosh, S.; Leonard, B.R.; Abel, C. Field-evolved resistance to Bt toxins. Nat. Biotechnol. 2008, 26, 1072–1074. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.; Qureshi, J.A.; Meagher, R.L., Jr.; Reisig, D.D.; Head, G.P.; Andow, D.A.; Ni, X.; Kerns, D.; Buntin, G.D.; Niu, Y.; et al. Cry1F resistance in fall armyworm Spodoptera frugiperda: Single gene versus pyramided Bt maize. PLoS ONE 2014, 9, e112958. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Rensburg, J.B.J. First report of field resistance by the stem borer, Busseola fusca (Fuller) to Bt-transgenic maize. S. Afr. J. Plant Soil 2007, 24, 147–151. [Google Scholar] [CrossRef] [Green Version]
- Gassmann, A.J.; Petzold-Maxwell, J.L.; Clifton, E.H.; Dunbar, M.W.; Hoffmann, A.M.; Ingber, D.A.; Keweshan, R.S. Field-evolved resistance by western corn rootworm to multiple Bacillus thuringiensis toxins in transgenic maize. Proc. Natl. Acad. Sci. USA 2014, 111, 5141–5146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tabashnik, B.E.; Fabrick, J.A.; Unnithan, G.C.; Yelich, A.J.; Masson, L.; Zhang, J.; Bravo, A.; Soberón, M. Efficacy of genetically modified Bt toxins alone and in combinations against pink bollworm resistant to Cry1Ac and Cry2Ab. PLoS ONE 2013, 8, e80496. [Google Scholar] [CrossRef]
- Tabashnik, B.E.; Zhang, M.; Fabrick, J.A.; Wu, Y.; Gao, M.; Huang, F.; Wei, J.; Zhang, J.; Yelich, A.; Unnithan, G.C.; et al. Dual mode of action of Bt proteins: Protoxin efficacy against resistant insects. Sci. Rep. 2015, 5, 15107. [Google Scholar] [CrossRef] [Green Version]
- Storer, N.P.; Babcock, J.M.; Schlenz, M.; Meade, T.; Thompson, G.D.; Bing, J.W.; Huckaba, R.M. Discovery and characterization of field resistance to Bt maize: Spodoptera frugiperda (Lepidoptera: Noctuidae) in Puerto Rico. J. Econ. Entomol. 2010, 103, 1031–1038. [Google Scholar] [CrossRef]
- Yu, W.; Lin, S.; Dimase, M.; Niu, Y.; Brown, S.; Head, G.P.; Price, P.A.; Reay-Jones, F.P.F.; Cook, D.; Reisig, D.; et al. Extended investigation of field-evolved resistance of the corn earworm Helicoverpa zea (Lepidoptera: Noctuidae) to Bacillus thuringiensis Cry1A.105 and Cry2Ab2 proteins in the southeastern United States. J. Invert. Pathol. 2021, 183, 107560. [Google Scholar] [CrossRef]
- Yang, F.; Santiago González, J.C.; Sword, G.A.; Kerns, D.L. Genetic basis of resistance to the Vip3Aa Bt protein in Helicoverpa zea. Pest Manag. Sci. 2020, 77, 1530–1535. [Google Scholar] [CrossRef] [PubMed]
- Soberón, M.; Pardo, L.; Muñóz-Garay, C.; Sánchez, J.; Gómez, I.; Porta, H.; Bravo, A. Pore formation by Cry toxins. In Proteins Membrane Binding and Pore Formation; Advances in Experimental Medicine and Biology; Springer: New York, NY, USA, 2010; pp. 127–142. [Google Scholar] [CrossRef]
- Pardo-López, L.; Soberón, M.; Bravo, A. Bacillus thuringiensis insecticidal three-domain Cry toxins: Mode of action, insect resistance and consequences for crop protection. FEMS Microbiol. Rev. 2013, 37, 3–22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jurat-Fuentes, J.L.; Karumbaiah, L.; Jakka, S.R.K.; Ning, C.; Liu, C.; Wu, K.; Jackson, J.; Gould, F.; Blanco, C.; Portilla, M.; et al. Reduced levels of membrane-bound alkaline phosphatase are common to lepidopteran strains resistant to Cry toxins from Bacillus thuringiensis. PLoS ONE 2011, 6, e17606. [Google Scholar] [CrossRef] [Green Version]
- Gahan, L.J.; Pauchet, Y.; Vogel, H.; Heckel, D.G. An ABC transporter mutation is correlated with insect resistance to Bacillus thuringiensis Cry1Ac toxin. PLoS Genet 2010, 6, e1001248. [Google Scholar] [CrossRef] [Green Version]
- Atsumi, S.; Miyamoto, K.; Yamamoto, K.; Narukawa, J.; Kawai, S.; Sezutsu, H.; Kobayashi, I.; Uchino, K.; Tamura, T.; Mita, K.; et al. Single amino acid mutation in an ATP-binding cassette transporter gene causes resistance to Bt toxin Cry1Ab in the silkworm, Bombyx mori. Proc. Natl. Acad. Sci. USA 2012, 109, E1591–E1598. [Google Scholar] [CrossRef] [Green Version]
- Jin, L.; Wang, J.; Guan, F.; Zhang, J.; Yu, S.; Liu, S.; Xue, Y.; Li, L.; Wu, S.; Wang, X.; et al. Dominant point mutation in a tetraspanin gene associated with field-evolved resistance of cotton bollworm to transgenic Bt cotton. Proc. Natl. Acad. Sci. USA 2018, 115, 11760–11765. [Google Scholar] [CrossRef] [Green Version]
- Jurat-Fuentes, J.L.; Heckel, D.G.; Ferré, J. Mechanisms of resistance to insecticidal proteins from Bacillus thuringiensis. Ann. Rev. Entomol. 2021, 66, 121–140. [Google Scholar] [CrossRef]
- Lawrie, R.D.; Mitchell Iii, R.D.; Deguenon, J.M.; Ponnusamy, L.; Reisig, D.; Pozo-Valdivia, A.D.; Kurtz, R.W.; Roe, R.M. Multiple known mechanisms and a possible role of an enhanced immune system in Bt-resistance in a field population of the bollworm, Helicoverpa zea: Differences in gene expression with RNAseq. Int. J. Mol. Sci. 2020, 21, 6528. [Google Scholar] [CrossRef]
- Caccia, S.; Moar, W.J.; Chandrashekhar, J.; Oppert, C.; Anilkumar, K.J.; Jurat-Fuentes, J.L.; Ferré, J. Association of Cry1Ac toxin resistance in Helicoverpa zea (Boddie) with increased alkaline phosphatase levels in the midgut lumen. Appl. Envrion. Microbiol. 2012, 78, 5690–5698. [Google Scholar] [CrossRef] [Green Version]
- Fritz, M.L.; Nunziata, S.O.; Guo, R.; Tabashnik, B.E.; Carrière, Y. Mutations in a novel cadherin gene associated with Bt resistance in Helicoverpa zea. G3 (Bethesda, Md.) 2020, 10, 1563–1574. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perera, O.P.; Little, N.S.; Abdelgaffar, H.; Jurat-Fuentes, J.L.; Reddy, G.V.P. Genetic knockouts indicate that the ABCC2 protein in the Bollworm Helicoverpa zea is not a major receptor for the Cry1Ac insecticidal protein. Genes 2021, 12, 1522. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Zhang, M.; Liang, G.; Li, X. Alkaline phosphatase 2 is a functional receptor of Cry1Ac but not Cry2Ab in Helicoverpa zea. Insect. Mol. Biol. 2019, 28, 372–379. [Google Scholar] [CrossRef] [PubMed]
- Roe, R.M.; Clifford, C.a.; Woodring, J. The effect of temperature on energy distribution during the last-larval stadium of the female house cricket, Acheta domesticus. J. Insect. Physiol. 1985, 31, 371–378. [Google Scholar] [CrossRef]
- Bailey, W.D.; Brownie, C.; Bacheler, J.S.; Gould, F.; Kennedy, G.G.; Sorenson, C.E.; Roe, R.M. Species diagnosis and Bacillus thuringiensis resistance monitoring of Heliothis virescens and Helicoverpa zea (Lepidoptera: Noctuidae) field strains from the southern United States using feeding disruption bioassays. J. Econ. Entomol. 2001, 94, 76–85. [Google Scholar] [CrossRef]
- Cabrera, A.R.; Van Kretschmar, J.; Bacheler, J.S.; Burrack, H.; Sorenson, C.E.; Roe, R.M. Resistance monitoring of Heliothis virescens to pyramided cotton varieties with a hydrateable, artificial cotton leaf bioassay. Crop. Prot. 2011, 30, 1196–1201. [Google Scholar] [CrossRef]
- Van Kretschmar, J.; Bailey, W.; Arellano, C.; Thompson, G.; Sutula, C.; Roe, R. Feeding disruption tests for monitoring the frequency of larval lepidopteran resistance to Cry1Ac, Cry1F and Cry1Ab. Crop. Prot. 2011, 30, 863–870. [Google Scholar] [CrossRef]
- Roe, R.M.; Bailey, W.D.; Gould, F.; Kennedy, G.G. Insecticide Resistance Assay. U.S. Patent No. 6,060,039, 9 May 2000. [Google Scholar]
- Roe, R.M.; Bailey, W.D.; Gould, F.; Kennedy, G.G.; Sutula, C.L. Insecticide Resistance Assay. U.S. Patent No. 6,517,856 B1, 11 February 2003. [Google Scholar]
- van Kretschmar, B.; Cabrera, A.R.; Bradley, J.R.; Roe, R.M. Novel adult feeding disruption test (FDT) to detect insecticide resistance of lepidopteran pests in cotton. Pest Manag. Sci. 2013, 69, 652–660. [Google Scholar] [CrossRef]
- Bailey, W.; Zhao, G.; Carter, L.; Gould, F.; Kennedy, G.; Roe, R. Feeding disruption bioassay for species and Bacillus thuringiensis resistance diagnosis for Heliothis virescens and Helicoverpa zea in cotton (Lepidoptera: Noctuidae). Crop. Prot. 1998, 17, 591–598. [Google Scholar] [CrossRef]
- Roe, R.; Van Kretschmar, J.; Thompson, D.; Donohue, K.; Sorenson, C.; Thompson, G.; Storer, N.; Blanco, C.; Lopez, J., Jr.; Leonard, B. Larval feeding disruption tests (FDT) for monitoring insect resistance to Cry 1Ac, Cry 1F and Cry 1Ab. In Proceedings of the Beltwide Cotton Conferences, New Orleans, LA, USA, 4–7 January 2005; pp. 1651–1661. [Google Scholar]
- Li, G.; Reisig, D.; Miao, J.; Gould, F.; Huang, F.; Feng, H. Frequency of Cry1F non-recessive resistance alleles in North Carolina field populations of Spodoptera frugiperda (Lepidoptera: Noctuidae). PLoS ONE 2016, 11, e0154492. [Google Scholar] [CrossRef] [Green Version]
- Yang, F.; Qureshi, J.A.; Leonard, B.R.; Head, G.P.; Niu, Y.; Huang, F. Susceptibility of Louisiana and Florida populations of Spodoptera frugiperda (Lepidoptera: Noctuidae) to pyramided Bt corn containing Genuity® VT Double Pro™ and SmartStax™ traits. Fla. Entomol. 2013, 96, 714–723. [Google Scholar] [CrossRef]
- Haygood, R.; Zorner, P.; Bradfisch, G. MVP II: An update on the encapsulated Bacillus thuringiensis (BT) bioinsecticide. In Proceedings of the the Beltwide Cotton Conferences, Nashville, TN, USA, 9–12 January 1996; pp. 902–905. [Google Scholar]
- Prosser, C.L. Temperature. In Comparastive Animal Physiology, 3rd ed.; W.B. Saunders Co.: Philadelphia, PA, USA, 1973; pp. 362–428. [Google Scholar]


| Insect | Treatment | Number of Fecal Pellets ± SEM (n) a | Percentage Mortality ± SEM (n) b | ||
|---|---|---|---|---|---|
| 30 °C | 20 °C | 30 °C | 20 °C | ||
| C. virescens | No Bt | 64.89 ± 4.25 (48)A | 27.33 ± 2.06 (48)B | 3.39 ± 2.04 (8)a | 4.69 ± 2.28 (8)a |
| MVPII | 35.35 ± 3.73 (48)A | 9.95 ± 1.36 (48)B | 35.94 ± 3.68 (8)a | 62.50 ± 3.34 (8)b | |
| H. zea | No Bt | 76.75 ± 4.91 (48)A | 33.08 ± 2.12 (48)B | 3.33 ± 3.33 (5)a | 6.60 ± 4.03 (5)a |
| MVPII | 34.44 ± 2.78 (48)A | 8.27 ± 0.59 (48)B | 52.33 ± 3.33 (5)a | 73.33 ± 3.12 (5)b | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dhammi, A.; van Krestchmar, J.B.; Zhu, J.; Ponnusamy, L.; Gould, F.; Reisig, D.; Kurtz, R.W.; Roe, R.M. Impact of Caterpillar Increased Feeding Rates on Reduction of Bt Susceptibility. Int. J. Mol. Sci. 2022, 23, 14856. https://doi.org/10.3390/ijms232314856
Dhammi A, van Krestchmar JB, Zhu J, Ponnusamy L, Gould F, Reisig D, Kurtz RW, Roe RM. Impact of Caterpillar Increased Feeding Rates on Reduction of Bt Susceptibility. International Journal of Molecular Sciences. 2022; 23(23):14856. https://doi.org/10.3390/ijms232314856
Chicago/Turabian StyleDhammi, Anirudh, Jaap B. van Krestchmar, Jiwei Zhu, Loganathan Ponnusamy, Fred Gould, Dominic Reisig, Ryan W. Kurtz, and R. Michael Roe. 2022. "Impact of Caterpillar Increased Feeding Rates on Reduction of Bt Susceptibility" International Journal of Molecular Sciences 23, no. 23: 14856. https://doi.org/10.3390/ijms232314856





