Reducing Capacity, Chlorogenic Acid Content and Biological Activity in a Collection of Scarlet (Solanum aethiopicum) and Gboma (S. macrocarpon) Eggplants
Abstract
:1. Introduction
2. Results and Discussion
2.1. Variation among Accessions
| Trait | Sum of Squares (%) | Mean | Minimum | Maximum | SE | CVP (%) | CVG (%) | H2 | |
|---|---|---|---|---|---|---|---|---|---|
| Accession | Residual | ||||||||
| Scarlet eggplant (n = 56) | |||||||||
| Total reducing capacity (equivalents of CGA; g·kg−1) | 87.55 *** | 12.45 | 7.45 | 3.83 | 16.92 | 0.62 | 28.61 | 21.79 | 0.58 |
| Chlorogenic acid (g·kg−1) | 82.76 *** | 17.24 | 1.51 | 0.21 | 4.47 | 0.19 | 39.92 | 27.69 | 0.48 |
| Chlorogenic acid peak area (%) | 41.56 *** | 58.44 | 78.62 | 50.3 | 95.3 | 2.82 | 10.06 | 6.07 | 0.36 |
| Gboma eggplant (n = 8) | |||||||||
| Total reducing capacity (equivalents of CGA; g·kg−1) | 84.34 *** | 15.66 | 11.16 | 7.15 | 22.69 | 1.09 | 32.37 | 23.82 | 0.54 |
| Chlorogenic acid (g·kg−1) | 94.89 *** | 5.11 | 1.66 | 0.48 | 4.87 | 0.15 | 46.26 | 41.58 | 0.81 |
| Chlorogenic acid peak area (%) | 73.65 *** | 26.35 | 60.87 | 50.4 | 71.5 | 2.03 | 9.41 | 5.73 | 0.37 |
2.2. Differences between Groups
| Group | n | Total Reducing Capacity (Equivalents of CGA; g·kg−1) z | Chlorogenic Acid (g·kg−1) z | Chlorogenic Acid Peak Area (%) z | |||
|---|---|---|---|---|---|---|---|
| Mean | Range | Mean | Range | Mean | Range | ||
| Scarlet eggplant (n = 56) | |||||||
| Aculeatum | 5 | 7.39 c | 6.02–8.64 | 1.28 c | 0.70–2.16 | 79.4 a | 76.5–82.3 |
| aethiopicum-anguivi | 6 | 8.01 c | 6.23–11.55 | 2.25 bc | 1.17–4.47 | 78.7 a | 67.6–87.0 |
| Gilo | 34 | 6.02 c | 3.83–16.45 | 1.46 c | 0.21–3.69 | 79.6 a | 50.3–93.3 |
| Kumba | 9 | 12.86 b | 9.41–16.92 | 0.99 c | 0.23–1.55 | 72.5 ab | 58.8–82.9 |
| Shum | 2 | 5.87 c | 3.86–7.87 | 3.03 b | 2.21–3.83 | 87.6 a | 86.0–89.2 |
| Gboma eggplant (n = 8) | |||||||
| S. dasyphyllum | 1 | 22.69 a | – | 4.87 a | – | 50.4 c | – |
| S. macrocarpon | 7 | 9.51 bc | 7.15–16.03 | 1.20 c | 0.48–1.98 | 62.4 bc | 54.3–71.5 |
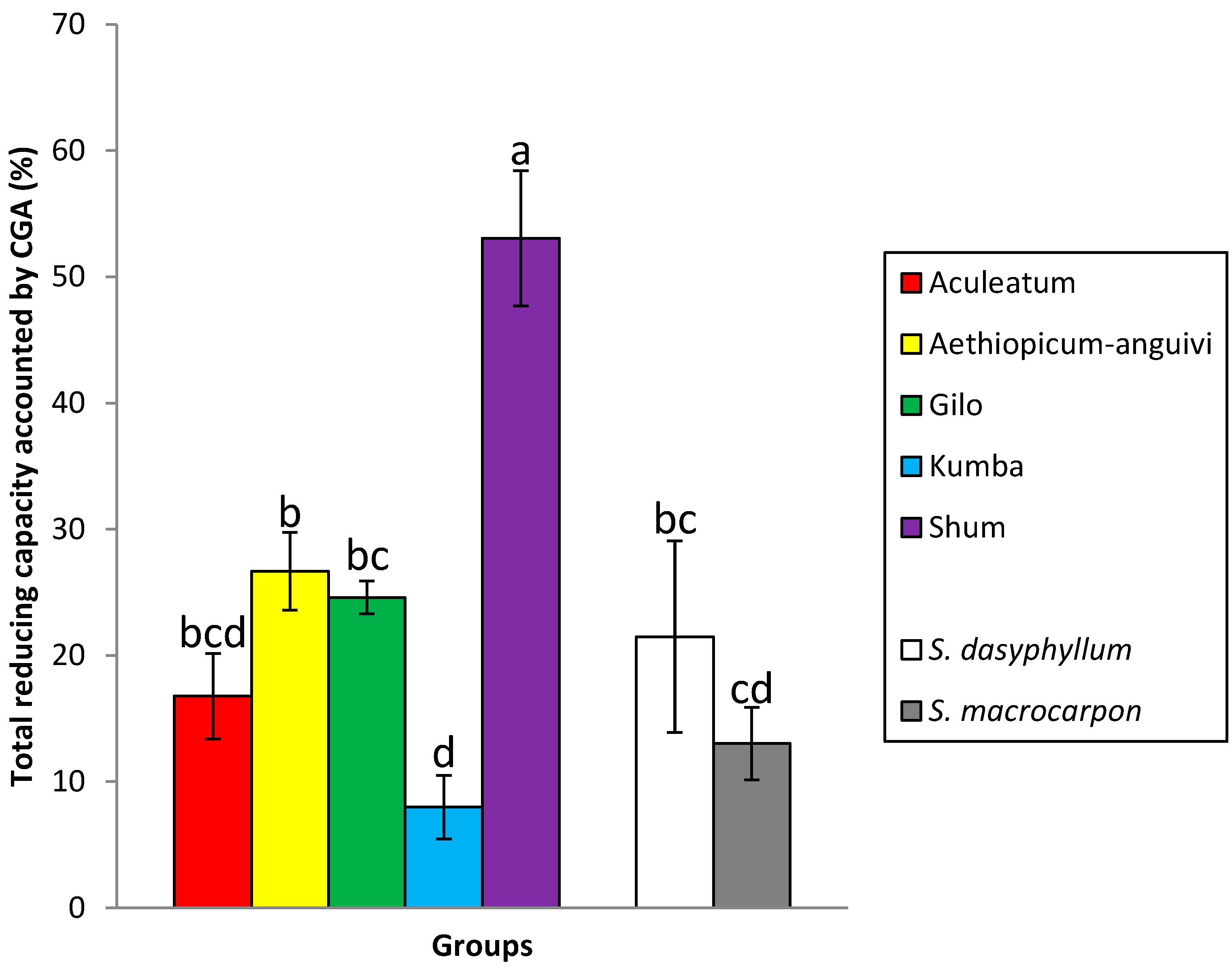
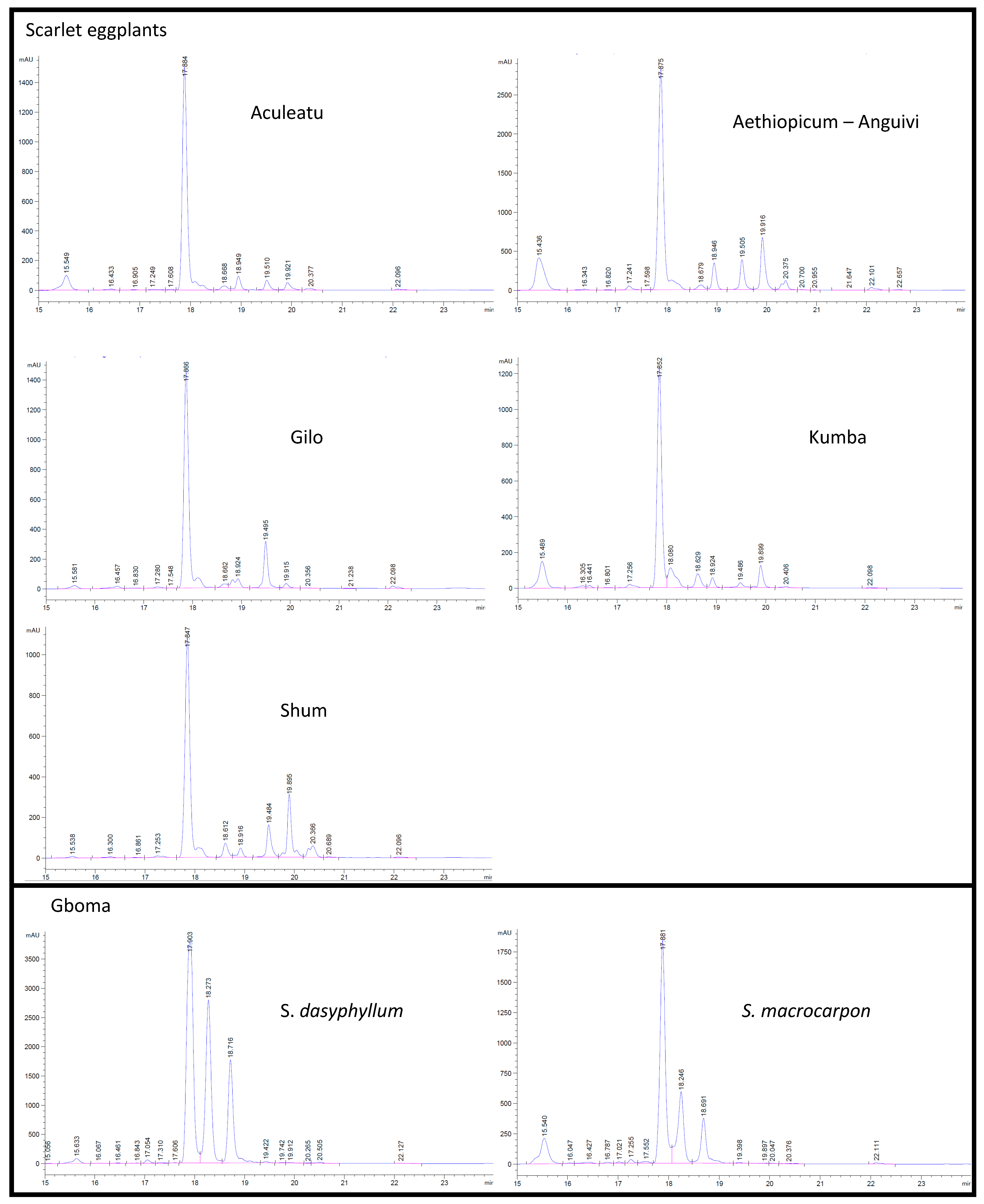
2.3. Relationship between Total Reducing Capacity and Chlorogenic Acid Content
| Group | n | Coef. Correlation | Coef. Determination (%) | F-Ratio | Prob. F |
|---|---|---|---|---|---|
| All accessions | 64 | 0.370 | 13.7 | 9.83 | 0.0026 |
| Scarlet eggplant | 56 | 0.197 | 3.9 | 2.18 | 0.1453 |
| Aculeatum | 5 | 0.904 | 81.6 | 13.34 | 0.0354 |
| aethiopicum-anguivi | 6 | 0.901 | 81.1 | 17.17 | 0.0143 |
| Gilo | 34 | 0.675 | 45.5 | 26.72 | <0.0001 |
| Kumba | 9 | −0.179 | 3.2 | 0.23 | 0.6451 |
| Shum a | 2 | – | – | – | – |
| Gboma eggplant | 8 | 0.893 | 79.7 | 23.59 | 0.0028 |
| S. dasyphylluma | 1 | – | – | – | – |
| S. macrocarpon | 7 | 0.499 | 24.9 | 1.65 | 0.2548 |
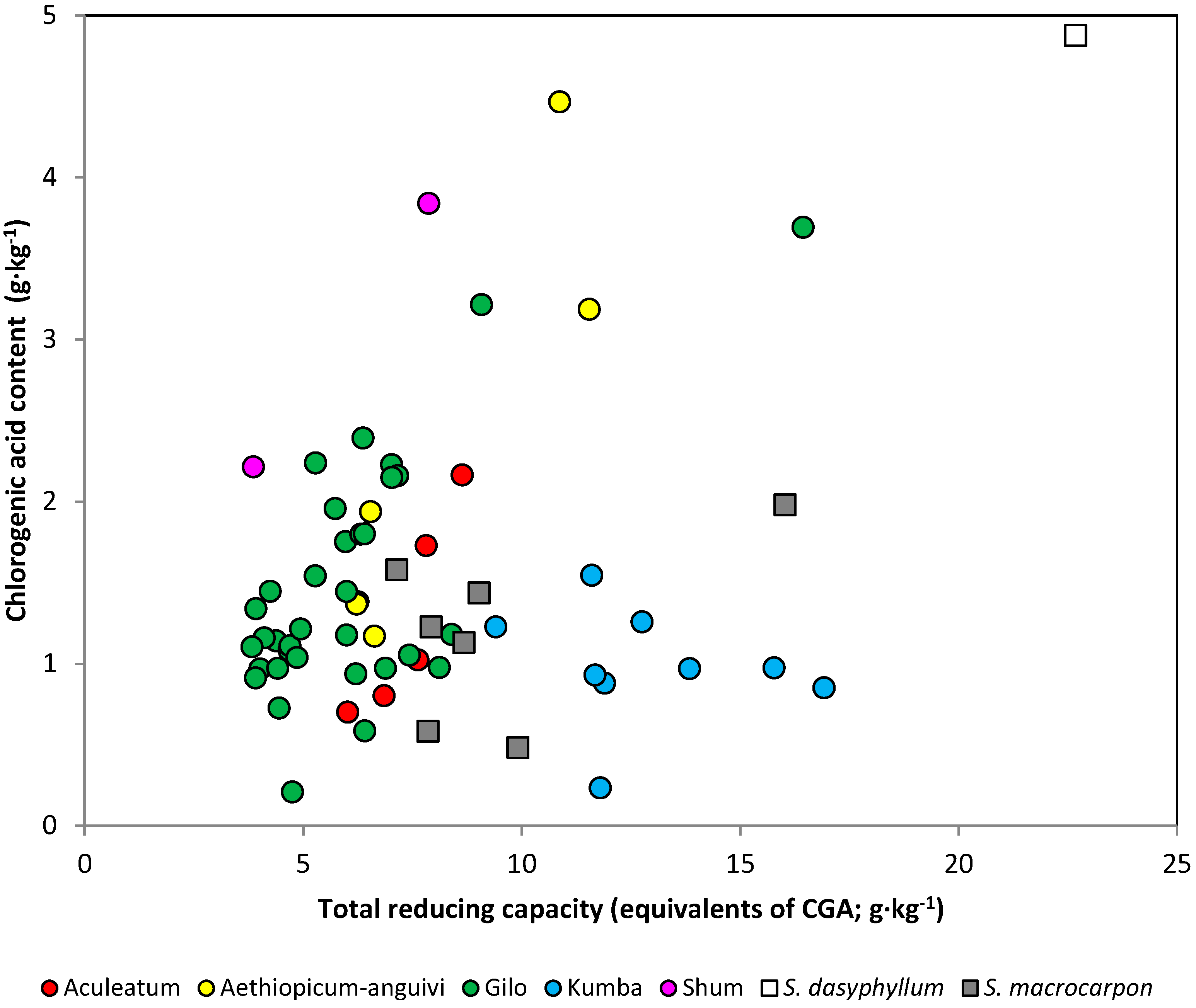
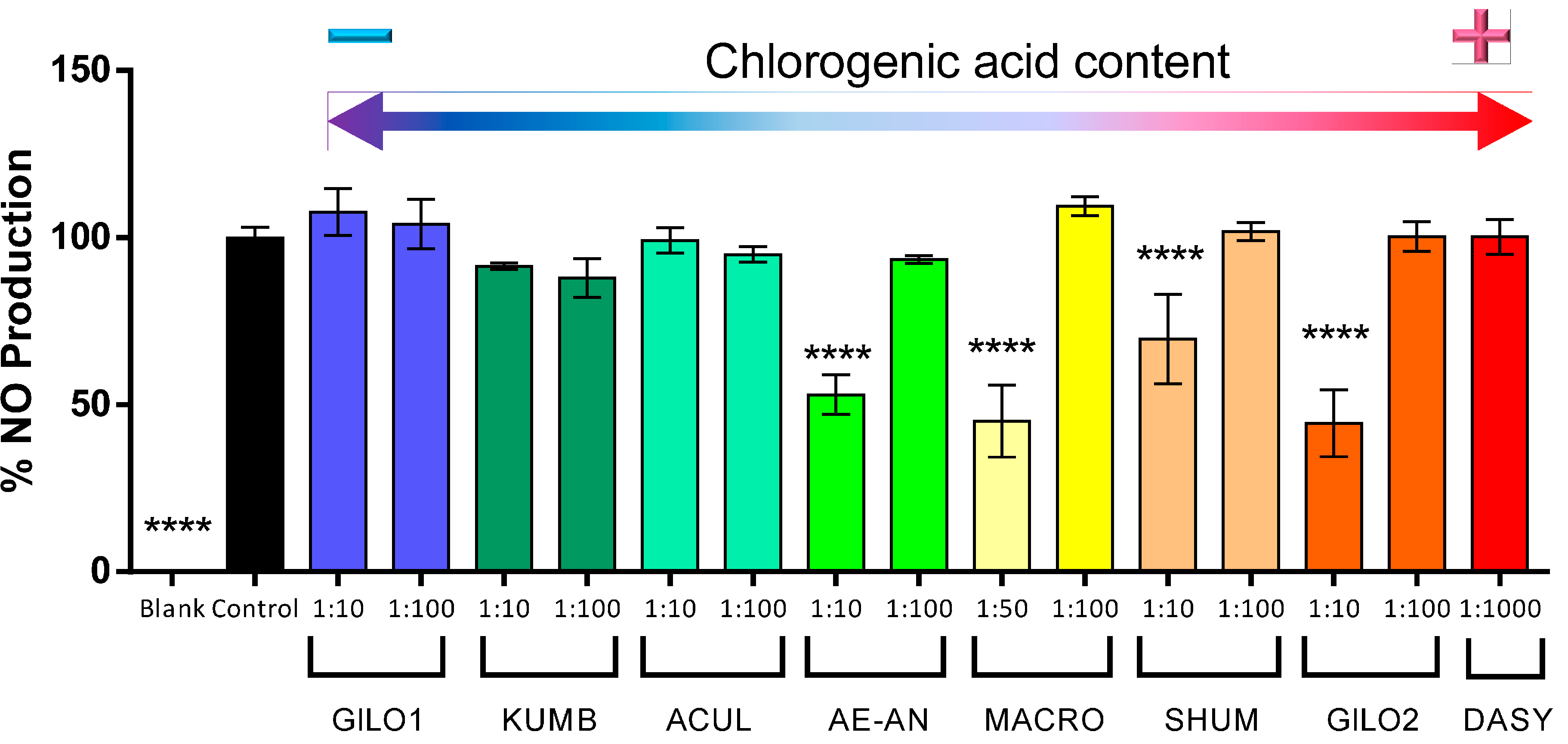
2.4. Biological Activity
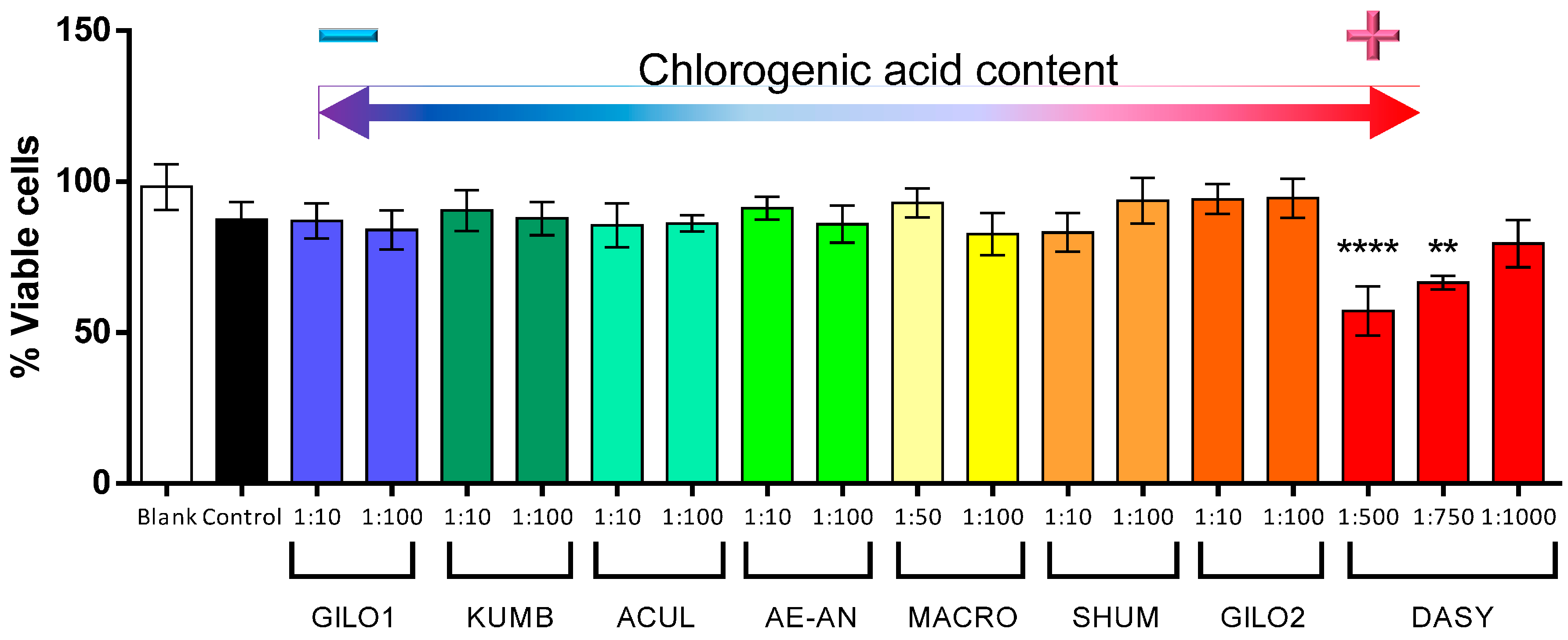
3. Experimental Section
3.1. Plant Material
| Group | n | Type | Common Use | Fruit Weight (g) | Fruit Shape | Fruit Diameter (cm) | Fruit Grooves | Fruit Locules | Calyx Prickliness |
|---|---|---|---|---|---|---|---|---|---|
| Scarlet eggplant (n = 56) | |||||||||
| Aculeatum | 5 | Cultivated | Ornamental | 20–40 | Flattened | 3–8 | Many | 4–10 | Very high |
| aethiopicum-anguivi | 6 | Weedy a | Medicinal | 3–8 | Ellipsoid | 1–3 | None to few | 2–3 | Absent to low |
| Gilo | 34 | Cultivated | Food (fruits) | 10–70 | Subspherical to ellipsoid | 3–8 | None to few | 2–6 | Absent to low |
| Kumba | 9 | Cultivated | Food (fruits and leaves) | 50–350 | Flattened | 5–12 | Very many | 10–20 | None |
| Shum | 2 | Cultivated | Food (leaves) | 2–6 | Round | 2–3 | None to few | 2–4 | None |
| Gboma eggplant (n = 8) | |||||||||
| S. dasyphyllum | 1 | Wild | Medicinal | 15–30 | Subspherical | 3–5 | None | 2–5 | Very high |
| S. macrocarpon | 7 | Cultivated | Food (fruits and leaves) | 50–150 | Subspherical | 5–9 | None | 4–6 | Absent to low |
3.2. Chemical Analysis
3.2.1. Total Reducing Capacity
3.2.2. Chlorogenic Acid and Other Free Phenolic Acids
3.3. Biological Assays
3.3.1. Cell Cultures
3.3.2. Preparation of Extracts for Biological Assays
3.3.3. Cell Viability Assay
3.3.4. Nitrite Determination
| Group | Code | CGA Content (g/kg) | CGA Rank |
|---|---|---|---|
| Scarlet eggplant | |||
| Aculeatum | ACUL | 1.02 | 6 |
| aethiopicum-anguivi | AE-AN | 1.40 | 5 |
| Gilo | GILO1 | 0.21 | 8 |
| Gilo | GILO2 | 3.69 | 2 |
| Kumba | KUMB | 0.23 | 7 |
| Shum | SHUM | 2.21 | 3 |
| Gboma eggplant | |||
| S. dasyphyllum | DASY | 4.87 | 1 |
| S. macrocarpon | MACR | 1.98 | 4 |
3.4. Data Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Diamanti, J.; Battino, M.; Mezzetti, B. Breeding for Fruit nutritional and nutraceutical quality. In Breeding for Fruit Quality; Jenks, M.A., Bebeli, P., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2011; pp. 61–80. [Google Scholar]
- Plazas, M.; Andújar, I.; Vilanova, S.; Hurtado, M.; Gramazio, P.; Herraiz, F.J.; Prohens, J. Breeding for chlorogenic acid content in eggplant: Interest and prospects. Notulae Botanicae Horti Agrobotanici Cluj-Napoca 2013, 41, 26–35. [Google Scholar]
- Soobrattee, M.A.; Neergheen, V.S.; Luximon-Ramma, A.; Aruoma, O.I.; Bahorun, T. Phenolics as potential antioxidant therapeutic agents: Mechanism and action. Mutat. Res. 2005, 579, 200–213. [Google Scholar]
- Fresco, P.; Borges, F.; Diniz, C.; Marques, M.P.M. New insights on the anticancer properties of dietary polyphenols. Med. Res. Rev. 2006, 16, 747–766. [Google Scholar]
- Dai, J.; Mumper, R.J. Plant phenolics: Extraction, analysis and their antioxidant and anticancer properties. Molecules 2010, 15, 7313–7352. [Google Scholar]
- Sato, Y.; Itakasi, S.; Kurokawa, T.; Ogura, J.; Kobayashi, M.; Hirano, T.; Sugawara, M.; Iseki, K. In vitro and in vivo antioxidant properties of chlorogenic and caffeic acid. Int. J. Pharm. 2011, 403, 136–138. [Google Scholar]
- Surh, Y.J. Cancer chemoprevention with dietary phytochemicals. Nat. Rev. Cancer 2003, 3, 768–780. [Google Scholar]
- Virgili, F.; Marino, M. Regulation of cellular signals from nutritional molecules: A specific role for phytochemicals, beyond antioxidant activity. Free Radic. Biol. Med. 2008, 45, 1205–1206. [Google Scholar]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, J. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radic. Biol. Med. 1996, 20, 933–956. [Google Scholar]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar]
- Alarcón-Flores, M.I.; Romero-González, R.; Martínez Vidal, J.L.; Egea González, F.J.; Garrido Frenich, A. Monitoring of phytochemicals in fresh and fresh-cut vegetables: A comparison. Food Chem. 2014, 142, 392–399. [Google Scholar]
- Suzuki, A.; Yamamoto, N.; Jokura, H.; Yamamoto, M.; Fujii, A.; Tomikitsu, I.; Saito, I. Chlorogenic acid attenuates hypertension and improves endothelial function in spontaneously hypertensive rats. J. Hypertens. 2006, 24, 1065–1073. [Google Scholar]
- Cho, A.S.; Jeon, S.M.; Kim, M.J.; Yeo, J.; Seo, K.I.; Choi, M.S.; Lee, M.K. Chlorogenic acid exhibits anti-obesity property and improves lipid metabolism in high-fat diet-induced-obese mice. Food Chem. Toxicol. 2010, 48, 937–943. [Google Scholar]
- Ahn, E.H.; Kim, D.W.; Shin, M.J.; Kwon, S.W.; Kim, Y.N.; Kim, D.S.; Lim, S.S.; Kim, J.; Park, J.; Eum, W.S.; et al. Chlorogenic acid improves neuroprotective effect of PEP-1-ribosomal protein S3 against ischemic insult. Exp. Neurobiol. 2011, 20, 169–175. [Google Scholar]
- Burgos-Morón, E.; Calderón-Montaño, J.M.; Orta, M.L.; Pastor, N.; Pérez-Guerrero, C.; Austin, C.; Mateos, S.; López-Lázaro, M. The coffee constituent chlorogenic acid induces cellular DNA damage and formation of topoisomerase I- and II-DNA complexes in cells. J. Agric. Food Chem. 2012, 60, 7384–7391. [Google Scholar]
- Coman, C.; Rugină, O.D.; Socaciu, C. Plants and natural compounds with antidiabetic action. Notulae Botanicae Horti Agrobotanici Cluj-Napoca 2012, 40, 314–325. [Google Scholar]
- Zhao, Y.; Wang, J.; Ballevre, O.; Luo, H.; Zhang, W. Antihypertensive effects and mechanisms of chlorogenic acid. Hypertens. Res. 2012, 35, 370–374. [Google Scholar]
- Dos Santos, M.D.; Almedia, M.C.; Lopes, N.P.; de Souza, G.E. Evaluation of the anti-imflammatory, analgesic and antipyrectic activities of the natural polyphenol chlorogenic acid. Biol. Pharm. Bull. 2006, 29, 2236–2240. [Google Scholar]
- Stommel, J.R.; Whitaker, B.D. Phenolic acid content and composition of eggplant fruit in a germplasm core subset. J. Am. Soc. Hortic. Sci. 2003, 128, 704–710. [Google Scholar]
- Whitaker, B.D.; Stommel, J.R. Distribution of hydroxycinnamic acid conjugates in fruit of commercial eggplant (Solanum melongena L.) cultivars. J. Agric. Food Chem. 2003, 51, 3448–3454. [Google Scholar]
- Prohens, J.; Rodríguez-Burruezo, A.; Raigón, M.D.; Nuez, F. Total phenolic concentration and browning susceptibility in a collection of different varietal types and hybrids of eggplant: Implications for breeding for higher nutritional quality and reduced browning. J. Am. Soc. Hortic. Sci. 2007, 132, 638–646. [Google Scholar]
- Prohens, J.; Whitaker, B.D.; Plazas, M.; Vilanova, S.; Hurtado, M.; Blasco, M.; Gramazio, P.; Stommel, J.R. Genetic diversity in morphological characters and phenolic acids content resulting from an interspecific cross between eggplant, Solanum melongena, and its wild ancestor (S. incanum). Ann. Appl. Biol. 2013, 162, 242–257. [Google Scholar]
- Schippers, R.R. African Indigenous Vegetables: An Overview of the Cultivated Species; Natural Resources Institute: Chatham, UK, 2000; pp. 147–176. [Google Scholar]
- Lester, R.N. Taxonomy of scarlet eggplants, Solanum aethiopicum L. Acta Hortic. 1986, 182, 125–132. [Google Scholar]
- Lester, R.N.; Hakiza, J.J.H.; Stavropoulos, N.; Teixeira, M.M. Variation patterns in the African scarlet eggplant, Solanum aethiopicum L. In Infraspecific Classification of Wild and Cultivated Plant; Styles, B.T., Ed.; Clarendon Press: Oxford, UK, 1986; pp. 283–307. [Google Scholar]
- Bukenya, Z.R.; Carasco, J.F. Biosystematic study of Solanum macrocarpon-S. dasyphyllum complex in Uganda and relations with S. linnaeanum. East Afr. Agric. For. J. 1994, 59, 187–204. [Google Scholar]
- Lester, R.N.; Daunay, M.C. Diversity of African vegetable Solanum species and its implications for a better understanding of plant domestication. Schriften zu Genetischen Ressourcen 2003, 22, 137–152. [Google Scholar]
- Polignano, G.; Uggenti, P.; Bisignano, V.; della Gatta, C. Genetic divergence analysis in eggplant (Solanum melongena L.) and allied species. Genet. Resour. Crop Ev. 2010, 57, 171–181. [Google Scholar]
- Sunseri, F.; Polignano, G.B.; Alba, V.; Lotti, C.; Bisignano, V.; Mennella, G.; D’Alessandro, A.; Bacchi, M.; Riccardi, P.; Fiore, M.C.; et al. Genetic diversity and characterization of African eggplant germplasm collection. Afr. J. Plant Sci. 2010, 4, 231–241. [Google Scholar]
- Plazas, M.; Andújar, I.; Vilanova, S.; Gramazio, P.; Herraiz, F.J.; Prohens, J. Conventional and phenomics characterization provides insight into the diversity and relationships of hypervariable scarlet (Solanum aethiopicum L.) and gboma (S. macrocarpon L.) eggplant complexes. Front. Plant Sci. 2014, 5, 318. [Google Scholar]
- Lester, R.N.; Niakan, L. Origin and domestication of the scarlet eggplant, Solanum aethiopicum, from S anguivi in Africa. In Solanaceae: Biology and Systematics; Styles, B.T., Ed.; Clarendon Press: Oxford, UK, 1986; pp. 433–456. [Google Scholar]
- Daunay, M.C.; Lester, R.N. The use of wild species for the genetic improvement of brinjal-eggplant (Solanum melongena) and tomato (Lycopersicon esculentum). In Solanaceae III: Taxonomy, Chemistry, Evolution; Hawkes, J.G., Lester, R.N., Nee, M., Estrada, N., Eds.; Royal Botanic Gardens: Kew, UK, 1991; pp. 389–412. [Google Scholar]
- Oyelana, O.A.; Ugborogho, R.E. Phenotypic variation of F1 and F2 populations from three species of Solanum L. (Solanaceae). Afr. J. Biotechnol. 2008, 7, 2359–2367. [Google Scholar]
- Daunay, M.C.; Hazra, P. Eggplant. In Handbook of Vegetables; Peter, K.V., Hazra, P., Eds.; Studium Press: Houston, TX, USA, 2014; pp. 257–322. [Google Scholar]
- Rotino, G.L.; Sala, T.; Toppino, L. Eggplant. In Alien Gene Transfer in Crop Plants; Pratap, A., Kumar, J., Eds.; Springer: New York, NY, USA, 2014; Volume 2, pp. 381–409. [Google Scholar]
- Prohens, J.; Plazas, M.; Raigón, M.D.; Seguí-Simarro, J.M.; Stommel, J.R.; Vilanova, S. Characterization of interspecific hybrids and backcross generations from crosses between cultivated eggplants (Solanum melongena and S. aethiopicum) and implications for eggplant breeding. Euphytica 2012, 186, 517–538. [Google Scholar]
- Mennella, G.; Rotino, G.L.; Fibiani, M.; D’Alessandro, A.; Francese, G.; Toppino, L.; Cavallanti, F.; Acciarri, N.; Lo Scalzo, R. Characterization of health-related compounds in eggplant (Solanum melongena L.) lines derived from introgression of allied species. J. Agric. Food Chem. 2010, 58, 7597–7603. [Google Scholar]
- Cao, G.H.; Sofic, E.; Prior, R.L. Antioxidant capacity of tea and common vegetables. J. Agric. Food Chem. 1996, 44, 3426–3431. [Google Scholar]
- San José, R.; Sánchez-Mata, M.C.; Cámara, M.; Prohens, J. Eggplant fruit composition as affected by cultivation environment and genetic constitution. J. Sci. Food Agric. 2014, 94, 2774–2784. [Google Scholar]
- Hanson, P.M.; Yang, R.Y.; Tsou, C.S.S.; Ledesma, D.; Engle, L.; Lee, T.C. Diversity in eggplant (Solanum melongena) for superoxide scavenging activity, total phenolics, and ascorbic acid. J. Food Compos. Anal. 2006, 19, 594–600. [Google Scholar]
- Luthria, D.L.; Mukhopadhyay, S. Influence of sample preparation on assay of phenolic acids from eggplant. J. Agric. Food Chem. 2006, 54, 41–47. [Google Scholar]
- Mennella, G.; Lo Scalzo, R.; Fibiani, M.; D’Alessandro, A.; Francese, G.; Toppino, L.; Acciarri, N.; de Almeida, A.E.; Rotino, G.L. Chemical and bioactive quality traits during fruit ripening in eggplant (S. melongena L.) and allied species. J. Agric. Food Chem. 2012, 50, 11821–11831. [Google Scholar]
- García-Salas, P.; Gómez-Caravaca, A.M.; Morales-Soto, A.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Identification and quantification of phenolic compounds in diverse cultivars of eggplant grown in different seasons by high-performance liquid chromatography coupled to diode array detector and electro-spray-quadrupole-time of flight-mass spectrometry. Food Res. Int. 2014, 57, 114–122. [Google Scholar]
- Raigón, M.D.; Prohens, J.; Muñoz-Falcón, J.E.; Nuez, F. Comparison of eggplant landraces and commercial varieties for fruit content of total phenolics, minerals, dry matter and protein. J. Food Compos. Anal. 2008, 21, 370–376. [Google Scholar]
- Wricke, G.; Weber, W. Quantitative Genetics and Selection in Plant Breeding; De Gruyter: Berlin, Germany, 1986; pp. 41–194. [Google Scholar]
- Pérez-de-Castro, A.M.; Vilanova, S.; Cañizares, J.; Pascual, L.; Blanca, J.M.; Díez, M.J.; Prohens, J.; Picó, B. Application of genomic tools in plant breeding. Curr. Genomics 2012, 13, 179–195. [Google Scholar]
- Plazas, M.; López-Gresa, M.P.; Vilanova, S.; Torres, C.; Hurtado, M.; Gramazio, P.; Andújar, I.; Herráiz, F.J.; Bellés, J.M.; Prohens, J. Diversity and relationships in key traits for functional and apparent quality in a collection of eggplant: Fruit phenolics content, antioxidant activity, polyphenol oxidase activity, and browning. J. Agric. Food Chem. 2013, 61, 8871–8879. [Google Scholar]
- Luthria, D.L. A simplified UV spectral scan method for the estimation of phenolic acids and antioxidant capacity in eggplant pulp extracts. J. Funct. Foods 2012, 4, 238–242. [Google Scholar]
- Wu, S.B.; Meyer, R.S.; Whitaker, B.D.; Litt, A.; Kennelly, E.J. A new liquid chromatography-mass spectrometry-based strategy to integrate chemistry, morphology, and evolution of eggplant (Solanum) species. J. Chromatogr. A 1314, 154–172. [Google Scholar]
- Little, T.; Hills, J. Agricultural Experimentation: Design and Analysis; Wiley: New York, NY, USA, 1978; pp. 61–194. [Google Scholar]
- Singleton, V.L.; Orthofer, R.; Lamela-Raventos, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. In Methods in Enzymology,Oxidants and Antioxidants Part A; Sies, H., Abelson, J., Simon, M., Eds.; Academic Press: San Diego, CA, USA, 1999; Volume 299, pp. 152–178. [Google Scholar]
- Everette, J.D.; Bryant, F.A.; Green, A.M.; Abbey, Y.A.; Wangila, G.W.; Walker, R.B. Thorough study of reactivity of various compound classes toward the Folin-Ciocalteu reagent. J. Agric. Food Chem. 2010, 58, 8139–8144. [Google Scholar]
- Sánchez-Rangel, J.C.; Benavides, J.; Heredia, J.B.; Cisneros-Zevallos, L.; Jacobo-Velázquez, D.A. The Folin-Ciocalteu assay revisited: Improvement of its specificity for total phenolic content determination. Anal. Methods 2013, 5, 5990–5999. [Google Scholar]
- Wang, J.; Mazz, G. Inhibitory Effects of anthocyanins and other phenolic compounds on nitric oxide production in LPS/IFN-γ-activated RAW 264.7 macrophages. J. Agric. Food Chem. 2002, 50, 850–857. [Google Scholar]
- Sánchez-Mata, M.C.; Yokoyama, W.E.; Hong, Y.J.; Prohens, J. α-Solasonine and α-solamargine contents of gboma (Solanum macrocarpon L.) and scarlet (Solanum aethiopicum L.) eggplants. J. Agric. Food Chem. 2010, 58, 5502–5508. [Google Scholar]
- Hwang, S.J.; Kim, Y.W.; Park, Y.; Lee, H.J.; Kim, K.W. Anti-inflammatory effects of chlorogenic acid in lipopolysaccharide-stimulated RAW 264.7 cells. Inflamm. Res. 2014, 63, 81–90. [Google Scholar]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Prior, R.L.; Wu, X.; Schaich, K. Standarized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar]
- Helmja, K.; Vaher, M.; Püssa, T.; Raudsepp, P.; Kaljurand, M. Evaluation of antioxidative capability of the tomato (Solanum lycopersicum) skin constituents by capillary electrophoresis and high-performance liquid chromatography. Electrophoresis 2008, 29, 3980–3988. [Google Scholar]
- Grisham, M.B.; Johnson, G.G.; Lancaster, J.R., Jr. Quantitation of nitrate and nitrite in extracellular fluids. Methods Enzymol. 1996, 268, 237–246. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Plazas, M.; Prohens, J.; Cuñat, A.N.; Vilanova, S.; Gramazio, P.; Herraiz, F.J.; Andújar, I. Reducing Capacity, Chlorogenic Acid Content and Biological Activity in a Collection of Scarlet (Solanum aethiopicum) and Gboma (S. macrocarpon) Eggplants. Int. J. Mol. Sci. 2014, 15, 17221-17241. https://doi.org/10.3390/ijms151017221
Plazas M, Prohens J, Cuñat AN, Vilanova S, Gramazio P, Herraiz FJ, Andújar I. Reducing Capacity, Chlorogenic Acid Content and Biological Activity in a Collection of Scarlet (Solanum aethiopicum) and Gboma (S. macrocarpon) Eggplants. International Journal of Molecular Sciences. 2014; 15(10):17221-17241. https://doi.org/10.3390/ijms151017221
Chicago/Turabian StylePlazas, Mariola, Jaime Prohens, Amparo Noelia Cuñat, Santiago Vilanova, Pietro Gramazio, Francisco Javier Herraiz, and Isabel Andújar. 2014. "Reducing Capacity, Chlorogenic Acid Content and Biological Activity in a Collection of Scarlet (Solanum aethiopicum) and Gboma (S. macrocarpon) Eggplants" International Journal of Molecular Sciences 15, no. 10: 17221-17241. https://doi.org/10.3390/ijms151017221





