Journal Description
Vaccines
Vaccines
is an international, peer-reviewed, open access journal published monthly online by MDPI. The American Society for Virology (ASV) is affiliated with Vaccines and their members receive a discount on the article processing charges.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), PubMed, PMC, Embase, CAPlus / SciFinder, and other databases.
- Journal Rank: JCR - Q1 (Immunology) / CiteScore - Q1 (Pharmacology (medical))
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 19.2 days after submission; acceptance to publication is undertaken in 2.9 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Impact Factor:
7.8 (2022);
5-Year Impact Factor:
7.4 (2022)
Latest Articles
Graduate and Health Professional Student Knowledge, Attitudes, Beliefs, and Behavior Related to Human Papillomavirus and Human Papillomavirus Vaccination: A Scoping Review of the Literature
Vaccines 2024, 12(5), 507; https://doi.org/10.3390/vaccines12050507 (registering DOI) - 07 May 2024
Abstract
Human papillomavirus (HPV) is a common sexually transmitted infection. Despite a safe and effective vaccine, uptake continues to be suboptimal. Recently, focus has moved to college campuses in an effort to increase vaccination rates. Little is known about the extent of efforts to
[...] Read more.
Human papillomavirus (HPV) is a common sexually transmitted infection. Despite a safe and effective vaccine, uptake continues to be suboptimal. Recently, focus has moved to college campuses in an effort to increase vaccination rates. Little is known about the extent of efforts to reach graduate students on college campuses in the United States and the vaccination rates within this subpopulation. This scoping review assessed the literature on knowledge, attitudes, beliefs, and behaviors about HPV and HPV vaccination among graduate and post-baccalaureate professional students in the United States. This review also aims to identify areas for further research to improve institutions’ abilities to create health programming to increase HPV awareness and HPV vaccination coverage on their campuses. Publications focusing on knowledge, attitudes, beliefs, and behaviors about HPV and HPV vaccination in post-baccalaureate students were included. The systematic review of PubMed, CINAHL, and Embase identified 2562 articles, and 56 articles met all inclusion criteria and were included in this scoping review. A majority of the reviewed studies investigated some combination of knowledge, attitudes, behaviors, and beliefs about HPV and the HPV vaccine in students in professional programs such as medicine. Study design approaches were primarily cross-sectional, utilizing web-based survey distribution methods. HPV vaccination status and HPV screening behaviors were primarily measured through participant self-report. There is limited research investigating post-baccalaureate student knowledge, attitudes, beliefs, and behaviors about HPV and HPV vaccination. There is a need for researchers to further investigate the needs of graduate students to create informative and effective HPV programming.
Full article
(This article belongs to the Special Issue Vaccine Strategies for HPV-Related Cancers)
►
Show Figures
Open AccessBrief Report
Antibody Response against SARS-CoV-2 after mRNA Vaccine in a Cohort of Hospital Healthy Workers Followed for 17 Months
by
Domenico Tripodi, Roberto Dominici, Davide Sacco, Claudia Pozzobon, Simona Spiti, Rosanna Falbo, Paolo Brambilla, Paolo Mascagni and Valerio Leoni
Vaccines 2024, 12(5), 506; https://doi.org/10.3390/vaccines12050506 (registering DOI) - 07 May 2024
Abstract
The assessment of antibody response to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is of critical importance to verify the protective efficacy of available vaccines. Hospital healthcare workers play an essential role in the care and treatment of patients and were particularly at
[...] Read more.
The assessment of antibody response to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is of critical importance to verify the protective efficacy of available vaccines. Hospital healthcare workers play an essential role in the care and treatment of patients and were particularly at risk of contracting the SARS-CoV-2 infection during the pandemic. The vaccination protocol introduced in our hospital protected the workers and contributed to the containment of the infection’ s spread and transmission, although a reduction in vaccine efficacy against symptomatic and breakthrough infections in vaccinated individuals was observed over time. Here, we present the results of a longitudinal and prospective analysis of the anti-SARS-CoV-2 antibodies at multiple time points over a 17-month period to determine how circulating antibody levels change over time following natural infection and vaccination for SARS-CoV-2 before (T0–T4) and after the spread of the omicron variant (T5–T6), analyzing the antibody response of 232 healthy workers at the Pio XI hospital in Desio. A General Estimating Equation model indicated a significant association of the antibody response with time intervals and hospital area, independent of age and sex. Specifically, a similar pattern of antibody response was observed between the surgery and administrative departments, and a different pattern with higher peaks of average antibody response was observed in the emergency and medical departments. Furthermore, using a logistic model, we found no differences in contracting SARS-CoV-2 after the third dose based on the hospital department. Finally, analysis of antibody distribution following the spread of the omicron variant, subdividing the cohort of positive individuals into centiles, highlighted a cut-off of 550 BAU/mL and showed that subjects with antibodies below this are more susceptible to infection than those with a concentration above the established cut-off value.
Full article
(This article belongs to the Section COVID-19 Vaccines and Vaccination)
Open AccessArticle
Antigenic Characterization of Circulating and Emerging SARS-CoV-2 Variants in the U.S. throughout the Delta to Omicron Waves
by
Han Di, Elizabeth A. Pusch, Joyce Jones, Nicholas A. Kovacs, Norman Hassell, Mili Sheth, Kelly Sabrina Lynn, Matthew W. Keller, Malania M. Wilson, Lisa M. Keong, Dan Cui, So Hee Park, Reina Chau, Kristine A. Lacek, Jimma D. Liddell, Marie K. Kirby, Genyan Yang, Monique Johnson, Sharmi Thor, Natosha Zanders, Chenchen Feng, Diya Surie, Jennifer DeCuir, Sandra N. Lester, Lydia Atherton, Heather Hicks, Azaibi Tamin, Jennifer L. Harcourt, Melissa M. Coughlin, Wesley H. Self, Jillian P. Rhoads, Kevin W. Gibbs, David N. Hager, Nathan I. Shapiro, Matthew C. Exline, Adam S. Lauring, Benjamin Rambo-Martin, Clinton R. Paden, Rebecca J. Kondor, Justin S. Lee, John R. Barnes, Natalie J. Thornburg, Bin Zhou, David E. Wentworth and Charles Todd Davisadd
Show full author list
remove
Hide full author list
Vaccines 2024, 12(5), 505; https://doi.org/10.3390/vaccines12050505 - 07 May 2024
Abstract
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has evolved into numerous lineages with unique spike mutations and caused multiple epidemics domestically and globally. Although COVID-19 vaccines are available, new variants with the capacity for immune evasion continue to emerge. To understand and characterize
[...] Read more.
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has evolved into numerous lineages with unique spike mutations and caused multiple epidemics domestically and globally. Although COVID-19 vaccines are available, new variants with the capacity for immune evasion continue to emerge. To understand and characterize the evolution of circulating SARS-CoV-2 variants in the U.S., the Centers for Disease Control and Prevention (CDC) initiated the National SARS-CoV-2 Strain Surveillance (NS3) program and has received thousands of SARS-CoV-2 clinical specimens from across the nation as part of a genotype to phenotype characterization process. Focus reduction neutralization with various antisera was used to antigenically characterize 143 SARS-CoV-2 Delta, Mu and Omicron subvariants from selected clinical specimens received between May 2021 and February 2023, representing a total of 59 unique spike protein sequences. BA.4/5 subvariants BU.1, BQ.1.1, CR.1.1, CQ.2 and BA.4/5 + D420N + K444T; BA.2.75 subvariants BM.4.1.1, BA.2.75.2, CV.1; and recombinant Omicron variants XBF, XBB.1, XBB.1.5 showed the greatest escape from neutralizing antibodies when analyzed against post third-dose original monovalent vaccinee sera. Post fourth-dose bivalent vaccinee sera provided better protection against those subvariants, but substantial reductions in neutralization titers were still observed, especially among BA.4/5 subvariants with both an N-terminal domain (NTD) deletion and receptor binding domain (RBD) substitutions K444M + N460K and recombinant Omicron variants. This analysis demonstrated a framework for long-term systematic genotype to antigenic characterization of circulating and emerging SARS-CoV-2 variants in the U.S., which is critical to assessing their potential impact on the effectiveness of current vaccines and antigen recommendations for future updates.
Full article
(This article belongs to the Special Issue SARS-CoV-2 Variants: Unraveling Vaccines and Immune Responses)
Open AccessArticle
Human Papilloma Virus Vaccination and Cervical Screening in the Italian Regions: An Overview of the Current State of the Art
by
Angela Bechini, Claudia Cosma, Giulia Di Pisa, Alice Fanfani, Giulia Ionita, Davide Liedl, Carla Lunetta, Linda Martorella, Silvia Mele, Lorenzo Stacchini, Gabriele Vaccaro, Ornella Zuccaro, Stefano Valente, Gian Marco Prandi, Paolo Bonanni and Sara Boccalini
Vaccines 2024, 12(5), 504; https://doi.org/10.3390/vaccines12050504 - 07 May 2024
Abstract
Human Papilloma Virus (HPV) infection and HPV-related cancers can be prevented through vaccinations and mass cervical screening programmes. The Ministry of Health in Italy provides recommendations on primary and secondary prevention of HPV-related diseases, but the 19 Italian regions and 2 autonomous provinces
[...] Read more.
Human Papilloma Virus (HPV) infection and HPV-related cancers can be prevented through vaccinations and mass cervical screening programmes. The Ministry of Health in Italy provides recommendations on primary and secondary prevention of HPV-related diseases, but the 19 Italian regions and 2 autonomous provinces have organisational and decision-making autonomy, with differences in the strategies for offering prevention. The aim of this study is to describe the HPV vaccination and cervical screening offered in all Italian regions. Regional official documents up until 31 December 2021 were first identified. Subsequently, primary and secondary prevention experts from each region were interviewed to validate the previously collected data. The National Immunisation Plan (NIP) 2017–2019 recommends HPV vaccination from the age of 11 for both sexes, with a coverage target of 95%. HPV vaccination is offered free of charge or co-payment. All regions have screening programmes for cervical cancer, using PAP or HPV-DNA tests every three to five years. All regions have an electronic registry for vaccination and screening status. All regions have developed awareness-raising campaigns. It is important to harmonise regional policies with the implementation of information systems integration. The collected data could enhance both regional and national public health initiatives, bolstering the resilience of vaccination programs.
Full article
(This article belongs to the Special Issue Vaccine Strategies for HPV-Related Cancers)
Open AccessArticle
Association between COVID-19 Booster Vaccination and COVID-19 Outcomes among U.S. Adults
by
Kimberly H. Nguyen, Cheyenne McChesney, Ruchi Patel, Robert A. Bednarczyk, Lavanya Vasudevan and Laura Corlin
Vaccines 2024, 12(5), 503; https://doi.org/10.3390/vaccines12050503 - 07 May 2024
Abstract
Understanding the association between booster vaccination and COVID-19 outcomes can help strengthen post-pandemic messaging and strategies to increase vaccination and reduce severe and long-term consequences of COVID-19. Using the Household Pulse Survey data collected from U.S. adults from 9 December 2022 to 13
[...] Read more.
Understanding the association between booster vaccination and COVID-19 outcomes can help strengthen post-pandemic messaging and strategies to increase vaccination and reduce severe and long-term consequences of COVID-19. Using the Household Pulse Survey data collected from U.S. adults from 9 December 2022 to 13 February 2023 (n = 214,768), this study assessed the relationship between COVID-19 booster vaccination and COVID-19 outcomes (testing positive for COVID-19, moderate/severe COVID-19, and long COVID). Disparities were found in COVID-19 outcomes (e.g., testing positive for COVID-19, moderate/severe COVID-19, and long COVID) by sociodemographic characteristics, region of residence, food insecurity status, mental health status, disability status, and housing type. Receipt of a COVID-19 booster vaccination was negatively associated with testing positive for COVID-19 (aOR = 0.75, 95%CI: 0.72,0.79), having moderate/severe COVID-19 (aOR = 0.92, 95%CI: 0.88, 0.97), or having long COVID (aOR = 0.86 (0.80, 0.91)). Even among those who tested positive for COVID-19, those who received the booster vaccine were less likely to have moderate/severe COVID-19 and less likely to have long COVID. Communicating the benefits of COVID-19 booster vaccination, integrating vaccination in patient visits, and reducing access barriers can increase vaccination uptake and confidence for all individuals and protect them against the severe negative outcomes of COVID-19.
Full article
(This article belongs to the Special Issue Vaccines and Vaccinations in the Pandemic Period)
►▼
Show Figures

Figure 1
Open AccessArticle
Trends in Intention to Take the Second Booster COVID-19 Vaccination and Associated Factors in China: Serial Cross-Sectional Surveys
by
Lingyu Kong, Xu Wang, Ziying Yang, Yihan Tang, Zhiwei Wang, Yu Ma, Jinghua Li, Zhoubin Zhang and Jing Gu
Vaccines 2024, 12(5), 502; https://doi.org/10.3390/vaccines12050502 - 07 May 2024
Abstract
Background: The escalating complexity of the COVID-19 epidemic underscores the need for heightened attention to booster vaccinations. This study aims to examine the changing trend in the public’s intention to receive the second COVID-19 booster vaccination over time and the associated factors following
[...] Read more.
Background: The escalating complexity of the COVID-19 epidemic underscores the need for heightened attention to booster vaccinations. This study aims to examine the changing trend in the public’s intention to receive the second COVID-19 booster vaccination over time and the associated factors following the COVID-19 policy optimization in China. Method: Eight cross-sectional surveys utilizing SMS questionnaire links were conducted in Guangzhou, China, from December 2022 to April 2023. The Mann–Kendall test was employed to analyze the trend in intentions to receive the second booster vaccination across the survey time. Adjusted and multivariate logistic analyses were used to analyze the factors associated with vaccination intention. Parallel analyses were performed for two subgroups with different COVID-19 infection statuses. Results: A total of 9860 respondents were surveyed in the eight rounds, of which 8048 completed the first booster vaccination and were included in the analysis. The overall COVID-19 infection rate was 60.0% (4832/8048), while the overall vaccination intention was 72.2% (5810/8048) among respondents. The vaccination intention exhibited a significant declining trend over time, decreasing from 81.5% in December 2022 to 52.2% in April 2023. An adjusted logistic regression analysis revealed that anxiety and depression were negatively associated with an intention to receive the second booster vaccination, while COVID-19-related preventive behaviors and a high engagement in COVID-19-related information were positively associated with an intention to receive the second booster vaccination. A subgroup analysis revealed that the association between psychological and behavioral characteristics and vaccination intention remained relatively stable among individuals with different histories of COVID-19 infections. Conclusion: There was a significant decline in the intention to receive the second booster vaccination following the optimization of the COVID policy in China. Our findings emphasize the urgency of the second booster vaccination and provide a foundation for the development of tailored interventions to enhance and sustain vaccination intention among the public.
Full article
(This article belongs to the Section COVID-19 Vaccines and Vaccination)
►▼
Show Figures

Figure 1
Open AccessArticle
Factors Associated with COVID-19 Vaccination Hesitancy and Most Frequently Vaccinated Status in a Japanese Population-Based Sample
by
Aya Shirama, Andrew Stickley and Tomiki Sumiyoshi
Vaccines 2024, 12(5), 501; https://doi.org/10.3390/vaccines12050501 - 07 May 2024
Abstract
While many countries across the world have had difficulty in providing continuous coronavirus disease (COVID-19) vaccine boosters, in Japan, the number of COVID-19 vaccinations has increased rapidly in the past two years to the point where the vaccination booster numbers are now at
[...] Read more.
While many countries across the world have had difficulty in providing continuous coronavirus disease (COVID-19) vaccine boosters, in Japan, the number of COVID-19 vaccinations has increased rapidly in the past two years to the point where the vaccination booster numbers are now at the highest level in the world (by March 2023). Against this backdrop, this study aimed to determine the characteristics of individuals who either refused to be vaccinated or who were vaccinated multiple (five) times during this period. We analyzed data from a sample of 3710 Japanese adults that were collected in a nationwide, cross-sectional Internet survey that was undertaken in March 2023. Several demographics (e.g., age, education level, marital status, job status), medical conditions, daily smoking, and mental health/psychological factors (i.e., perceived stress, anxiety symptoms, loneliness) were associated with not having been vaccinated. Although the most frequently vaccinated status was also associated with some demographics (e.g., age, employment status), medical conditions, and daily smoking, other predictors such as having a COVID-19 infection history were unique to this outcome. Moreover, age-stratified analyses showed that depression (OR: 2.05, 95%CI: 1.08–3.89) and anxiety (OR: 3.87, 95%CI: 1.80–8.31) symptoms were associated with higher odds of being unvaccinated while loneliness was associated with lower odds for the most frequently vaccinated status (OR: 0.72, 95%CI: 0.54–0.96) among older adults (aged ≥ 60 years). The findings of this study indicate that many of the same factors are associated with vaccine hesitancy and being vaccinated multiple times among adults in Japan but that among older individuals, worse mental/psychological health problems may be important for vaccine hesitancy/infrequent vaccine uptake in an age range where the majority of individuals (57.7%) had been vaccinated five times.
Full article
(This article belongs to the Special Issue COVID-19 Vaccine Acceptance and Uptake: Insights from Behavioural and Social Sciences)
Open AccessSystematic Review
Efficacy of Respiratory Syncytial Virus Vaccination to Prevent Lower Respiratory Tract Illness in Older Adults: A Systematic Review and Meta-Analysis of Randomized Controlled Trials
by
Matteo Riccò, Antonio Cascio, Silvia Corrado, Marco Bottazzoli, Federico Marchesi, Renata Gili, Pasquale Gianluca Giuri, Davide Gori and Paolo Manzoni
Vaccines 2024, 12(5), 500; https://doi.org/10.3390/vaccines12050500 - 05 May 2024
Abstract
A systematic review and meta-analysis was designed in order to ascertain the effectiveness of respiratory syncytial virus (RSV) vaccination in preventing lower respiratory tract diseases (LRTD) in older adults (age ≥ 60 years). Studies reporting on randomized controlled trials (RCTs) were searched for
[...] Read more.
A systematic review and meta-analysis was designed in order to ascertain the effectiveness of respiratory syncytial virus (RSV) vaccination in preventing lower respiratory tract diseases (LRTD) in older adults (age ≥ 60 years). Studies reporting on randomized controlled trials (RCTs) were searched for in three databases (PubMed, Embase, and Scopus) and the preprint repository medRxiv until 31 March 2024. A total of nine studies were eventually included, two of which were conference proceedings. Our analysis included five RCTs on five RSV vaccines (RSVpreF, RSVPreF3, Ad26.RSV.preF, MEDI7510, and mRNA-1345). The meta-analysis documented a pooled vaccine efficacy of 81.38% (95% confidence interval (95% CI) 70.94 to 88.06) for prevention of LRTD with three or more signs/symptoms during the first RSV season after the delivery of the vaccine. Follow-up data were available for RSVPreF3 (2 RSV seasons), RSVpreF (mid-term estimates of second RSV season), and mRNA-1345 (12 months after the delivery of the primer), with a pooled VE of 61.15% (95% CI 45.29 to 72.40). After the first season, the overall risk for developing RSV-related LRTD was therefore substantially increased (risk ratio (RR) 4.326, 95% CI 2.415; 7.748). However, all estimates were affected by substantial heterogeneity, as suggested by the 95% CI of I2 statistics, which could be explained by inconsistencies in the design of the parent studies, particularly when dealing with case definition. In conclusion, adult RSV vaccination was quite effective in preventing LRTD in older adults, but the overall efficacy rapidly decreased in the second season after the delivery of the vaccine. Because of the heterogenous design of the parent studies, further analyses are required before tailoring specific public health interventions.
Full article
(This article belongs to the Special Issue Recent Developments in Vaccines against Respiratory Pathogens)
►▼
Show Figures

Figure 1
Open AccessArticle
Understanding Reasons for Vaccination Hesitancy and Implementing Effective Countermeasures: An Online Survey of Individuals Unvaccinated against COVID-19
by
Yurie Kobashi, Makoto Yoshida, Hiroaki Saito, Hiroki Yoshimura, Saori Nonaka, Chika Yamamoto, Tianchen Zhao and Masaharu Tsubokura
Vaccines 2024, 12(5), 499; https://doi.org/10.3390/vaccines12050499 - 05 May 2024
Abstract
This online survey of unvaccinated people living in Japan aimed to identify the reasons for declining vaccination and to develop effective countermeasures. We conducted a hierarchical class analysis to classify participants, examine factors influencing their classification, and provide the information they needed about
[...] Read more.
This online survey of unvaccinated people living in Japan aimed to identify the reasons for declining vaccination and to develop effective countermeasures. We conducted a hierarchical class analysis to classify participants, examine factors influencing their classification, and provide the information they needed about coronavirus disease 2019 (COVID-19) and trusted sources of COVID-19 information for each group. A total of 262 participants were classified into three groups: Group 1 with no specific reason (28 participants, 10.69%); Group 2 with clear concerns about trust in the vaccine (85 participants, 32.44%), and Group 3 with attitudinal barriers, such as distrust of the vaccine and complacency towards COVID-19, and structural barriers, such as vaccination appointments (149 participants, 56.87%). For each group, females tended to be classified in Group 2 more than Group 1 (Odds ratio (OR) [95% confidential intervals (95%CI)] = 1.64 (0.63 to 2.66), p = 0.001) and in Group 3 more than Group 1 (OR [95%CI] = 1.16 (0.19 to 2.12), p = 0.019). The information that the participants wanted to know about COVID-19 was different among each group (Safety: p < 0.001, Efficacy: p < 0.001, Genetic effects: p < 0.001). Those who did not receive the COVID-19 vaccine also had lower influenza vaccination coverage (8.02%). Additionally, 38 participants (14.50%) were subject to social disadvantages because they had not received the COVID-19 vaccine. Countermeasures should be carefully tailored according to the target population, reasons for hesitancy, and specific context. The findings of this study may help develop individualized countermeasures to address vaccine hesitancy.
Full article
(This article belongs to the Special Issue COVID-19 Vaccination with Public Health Perspective: Prevention and Control Strategies)
►▼
Show Figures
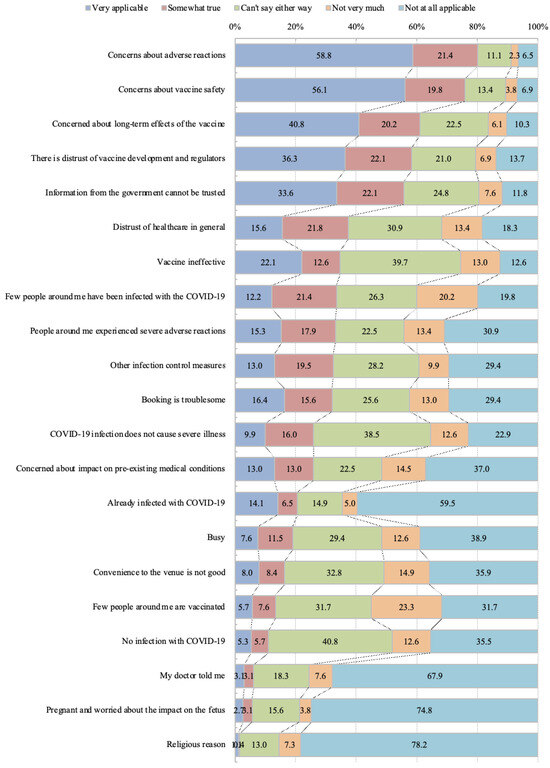
Figure 1
Open AccessReview
Neoantigen Identification and Dendritic Cell-Based Vaccines for Lung Cancer Immunotherapy
by
Komal Kumari, Amarnath Singh, Archana Chaudhary, Rakesh Kumar Singh, Asheesh Shanker, Vinay Kumar and Rizwanul Haque
Vaccines 2024, 12(5), 498; https://doi.org/10.3390/vaccines12050498 - 05 May 2024
Abstract
Immunotherapies can treat many cancers, including difficult-to-treat cases such as lung cancer. Due to its tolerability, long-lasting therapeutic responses, and efficacy in a wide spectrum of patients, immunotherapy can also help to treat lung cancer, which has few treatment choices. Tumor-specific antigens (TSAs)
[...] Read more.
Immunotherapies can treat many cancers, including difficult-to-treat cases such as lung cancer. Due to its tolerability, long-lasting therapeutic responses, and efficacy in a wide spectrum of patients, immunotherapy can also help to treat lung cancer, which has few treatment choices. Tumor-specific antigens (TSAs) for cancer vaccinations and T-cell therapies are difficult to discover. Neoantigens (NeoAgs) from genetic mutations, irregular RNA splicing, protein changes, or viral genetic sequences in tumor cells provide a solution. NeoAgs, unlike TSAs, are non-self and can cause an immunological response. Next-generation sequencing (NGS) and bioinformatics can swiftly detect and forecast tumor-specific NeoAgs. Highly immunogenic NeoAgs provide personalized or generalized cancer immunotherapies. Dendritic cells (DCs), which originate and regulate T-cell responses, are widely studied potential immunotherapeutic therapies for lung cancer and other cancers. DC vaccines are stable, reliable, and safe in clinical trials. The purpose of this article is to evaluate the current status, limitations, and prospective clinical applications of DC vaccines, as well as the identification and selection of major histocompatibility complex (MHC) class I and II genes for NeoAgs. Our goal is to explain DC biology and activate DC manipulation to help researchers create extremely potent cancer vaccines for patients.
Full article
(This article belongs to the Section Clinical Immunology)
►▼
Show Figures

Figure 1
Open AccessArticle
Safety and Immunogenicity of an Accelerated Ebola Vaccination Schedule in People with and without Human Immunodeficiency Virus: A Randomized Clinical Trial
by
Julie A. Ake, Kristopher Paolino, Jack N. Hutter, Susan Biggs Cicatelli, Leigh Anne Eller, Michael A. Eller, Margaret C. Costanzo, Dominic Paquin-Proulx, Merlin L. Robb, Chi L. Tran, Lalaine Anova, Linda L. Jagodzinski, Lucy A. Ward, Nicole Kilgore, Janice Rusnak, Callie Bounds, Christopher S. Badorrek, Jay W. Hooper, Steven A. Kwilas, Ine Ilsbroux, Dickson Nkafu Anumendem, Auguste Gaddah, Georgi Shukarev, Viki Bockstal, Kerstin Luhn, Macaya Douoguih and Cynthia Robinsonadd
Show full author list
remove
Hide full author list
Vaccines 2024, 12(5), 497; https://doi.org/10.3390/vaccines12050497 - 04 May 2024
Abstract
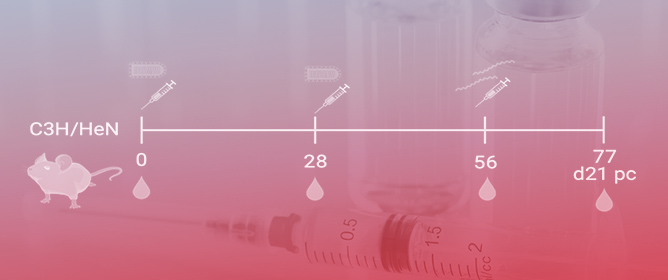
The safety and immunogenicity of the two-dose Ebola vaccine regimen MVA-BN-Filo, Ad26.ZEBOV, 14 days apart, was evaluated in people without HIV (PWOH) and living with HIV (PLWH). In this observer-blind, placebo-controlled, phase 2 trial, healthy adults were randomized (4:1) to receive MVA-BN-Filo (dose
[...] Read more.
The safety and immunogenicity of the two-dose Ebola vaccine regimen MVA-BN-Filo, Ad26.ZEBOV, 14 days apart, was evaluated in people without HIV (PWOH) and living with HIV (PLWH). In this observer-blind, placebo-controlled, phase 2 trial, healthy adults were randomized (4:1) to receive MVA-BN-Filo (dose 1) and Ad26.ZEBOV (dose 2), or two doses of saline/placebo, administered intramuscularly 14 days apart. The primary endpoints were safety (adverse events (AEs)) and immunogenicity (Ebola virus (EBOV) glycoprotein-specific binding antibody responses). Among 75 participants (n = 50 PWOH; n = 25 PLWH), 37% were female, the mean age was 44 years, and 56% were Black/African American. AEs were generally mild/moderate, with no vaccine-related serious AEs. At 21 days post-dose 2, EBOV glycoprotein-specific binding antibody responder rates were 100% among PWOH and 95% among PLWH; geometric mean antibody concentrations were 6286 EU/mL (n = 36) and 2005 EU/mL (n = 19), respectively. A total of 45 neutralizing and other functional antibody responses were frequently observed. Ebola-specific CD4+ and CD8+ T-cell responses were polyfunctional and durable to at least 12 months post-dose 2. The regimen was well tolerated and generated robust, durable immune responses in PWOH and PLWH. Findings support continued evaluation of accelerated vaccine schedules for rapid deployment in populations at immediate risk. Trial registration: NCT02598388 (submitted 14 November 2015).
Full article
(This article belongs to the Special Issue Clinical Strategies to Improve Efficacy, Effectiveness, and Safety of Vaccination in Humans)
►▼
Show Figures
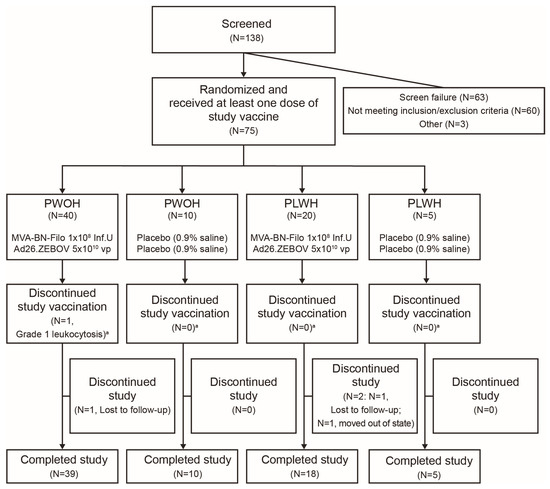
Figure 1
Open AccessArticle
Vaccine Effectiveness against GP-Attended Symptomatic COVID-19 and Hybrid Immunity among Adults in Hungary during the 2022–2023 Respiratory Season Dominated by Different SARS-CoV-2 Omicron Subvariants
by
Judit Krisztina Horváth, Gergő Túri, Katalin Krisztalovics, Katalin Kristóf and Beatrix Oroszi
Vaccines 2024, 12(5), 496; https://doi.org/10.3390/vaccines12050496 - 04 May 2024
Abstract
►▼
Show Figures
Hungary provides the opportunity to evaluate the effectiveness of COVID-19 vaccination in a setting where naturally acquired immunity and hybrid immunity are likely to play a greater role due to suboptimal vaccination coverage. Methods: A test-negative study was conducted during the 2022–2023 respiratory
[...] Read more.
Hungary provides the opportunity to evaluate the effectiveness of COVID-19 vaccination in a setting where naturally acquired immunity and hybrid immunity are likely to play a greater role due to suboptimal vaccination coverage. Methods: A test-negative study was conducted during the 2022–2023 respiratory season at the primary care level to determine the effectiveness of at least one COVID-19 booster dose in preventing medically attended symptomatic RT-PCR-confirmed SARS-CoV-2 infection in adults. Unvaccinated patients were used as a reference group. Results: A total of 247 cases and 1073 controls were included in the analysis. CVE was 56.8% (95% CI: 11.9–78.8%) in the population aged 60 years and older and 2.3% (95% CI: −50.0–36.3%) in the younger adults against COVID-19 caused by Omicron subvariants, mainly BA.5, BQ.1, and XBB.1. Self-reported COVID-19 in the 60–365 days prior to the current illness did not confer protection against reinfection without vaccination, but together with booster vaccination, it reduced the risk of COVID-19 by 63.0% (95% CI: −28.0–89.3%) and 87.6% (95% CI: 26.4–97.9%) among the 18–59 and 60+ age groups, respectively. Conclusions: CVE against COVID-19 was moderately high in the 60+ age groups. Because of the benefit of hybrid immunity, persons with previous SARS-CoV-2 infection should still be considered for vaccination campaigns.
Full article
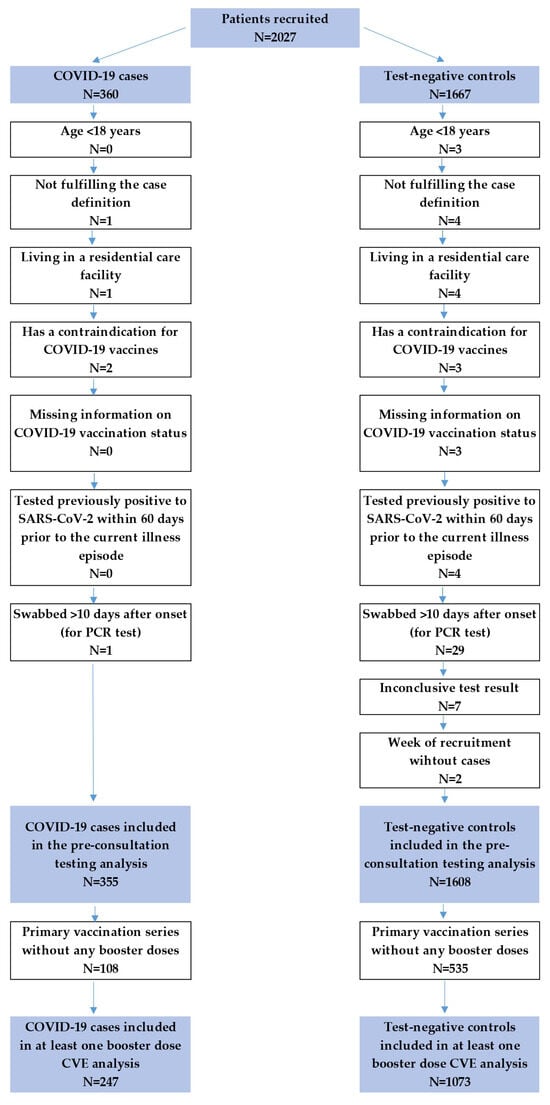
Figure 1
Open AccessArticle
Sociodemographic Trends and Correlation between Parental Hesitancy towards Pediatric COVID-19 Vaccines and Routine Childhood Immunizations in the United States: 2021–2022 National Immunization Survey—Child COVID Module
by
Olufunto A. Olusanya, Nina B. Masters, Fan Zhang, David E. Sugerman, Rosalind J. Carter, Debora Weiss and James A. Singleton
Vaccines 2024, 12(5), 495; https://doi.org/10.3390/vaccines12050495 - 03 May 2024
Abstract
Multiple factors may influence parental vaccine hesitancy towards pediatric COVID-19 vaccines and routine childhood immunizations (RCIs). Using the United States National Immunization Survey—Child COVID Module data collected from parents/guardians of children aged 5–11 years, this cross-sectional study (1) identified the trends and prevalence
[...] Read more.
Multiple factors may influence parental vaccine hesitancy towards pediatric COVID-19 vaccines and routine childhood immunizations (RCIs). Using the United States National Immunization Survey—Child COVID Module data collected from parents/guardians of children aged 5–11 years, this cross-sectional study (1) identified the trends and prevalence estimates of parental hesitancy towards pediatric COVID-19 vaccines and RCIs, (2) examined the relationship between hesitancy towards pediatric COVID-19 vaccines and RCIs, and (3) assessed trends in parental hesitancy towards RCIs by sociodemographic characteristics and behavioral and social drivers of COVID-19 vaccination. From November 2021 to July 2022, 54,329 parents or guardians were interviewed. During this 9-month period, the proportion of parents hesitant about pediatric COVID-19 vaccines increased by 15.8 percentage points (24.8% to 40.6%). Additionally, the proportion of parents who reported RCIs hesitancy increased by 4.7 percentage points from November 2021 to May 2022 but returned to baseline by July 2022. Over nine months, parents’ concerns about pediatric COVID-19 infections declined; however, parents were increasingly worried about pediatric COVID-19 vaccine safety and overall importance. Furthermore, pediatric COVID-19 vaccine hesitancy was more prevalent among parents of children who were White (43.2%) versus Black (29.3%) or Hispanic (26.9%) and those residing in rural (51.3%) compared to urban (28.9%) areas. In contrast, RCIs hesitancy was higher among parents of children who were Black (32.0%) versus Hispanic (24.5%) or White (23.6%). Pediatric COVID-19 vaccine hesitancy was 2–6 times as prevalent among parents who were RCIs hesitant compared to those who were RCIs non-hesitant. This positive correlation between parental hesitancy towards pediatric COVID-19 vaccines and RCIs was observed for all demographic and psychosocial factors for unadjusted and adjusted prevalence ratios. Parent–provider interactions should increase vaccine confidence, shape social norms, and facilitate behavior change to promote pediatric vaccination rates.
Full article
(This article belongs to the Special Issue Inequality in Immunization 2024)
►▼
Show Figures

Figure 1
Open AccessSystematic Review
Serological Correlates of Protection Induced by COVID-19 Vaccination in the Working Age Population: A Systematic Review and Meta-Analysis
by
Alborz Rahmani, Alfredo Montecucco, Luca Priano, Lucia Mandolini, Guglielmo Dini and Paolo Durando
Vaccines 2024, 12(5), 494; https://doi.org/10.3390/vaccines12050494 - 03 May 2024
Abstract
COVID-19 vaccines represent effective public health measures in contrasting the pandemic worldwide. However, protection at the individual-level, which is of crucial importance from an occupational health perspective, is commonly assessed by a serological correlate of protection (CoP) for SARS-CoV-2, which has not yet
[...] Read more.
COVID-19 vaccines represent effective public health measures in contrasting the pandemic worldwide. However, protection at the individual-level, which is of crucial importance from an occupational health perspective, is commonly assessed by a serological correlate of protection (CoP) for SARS-CoV-2, which has not yet been determined. The emergence of variants of concern (VOCs) that have shown high rates of breakthrough infections has further complicated the understanding of immune protection against infection. To define a potential serological correlate of protection induced by the COVID-19 vaccination, a systematic review and meta-analysis was performed to summarize the evidence concerning the binding antibody concentration corresponding to a protective effect. Eighteen and four studies were included in the qualitative and quantitative analyses, respectively. The protection against infection was shown for anti-receptor-binding domain (RBD) titers ranging from 154 to 168.2 binding antibody units (BAU)/mL during the pre-Omicron period, while ranging from 1235 to 3035 BAU/mL in the Omicron period. Pooling the results from the studies concerning anti-RBD and anti-Spike antibody titer, we found a mean of 1341.5 BAU/mL and 1400.1 BAU/mL, respectively. These findings suggest that although a fixed serological threshold corresponding to protection against different SARS-CoV-2 variants is not yet definable, higher binding antibody concentrations are associated with increased protective effects.
Full article
(This article belongs to the Special Issue Prevention of SARS-CoV-2 Infection and COVID-19: Safety, Immunogenicity and Effectiveness of Available Vaccines)
►▼
Show Figures

Figure 1
Open AccessArticle
The Equal Neutralizing Effectiveness of BNT162b2, ChAdOx1 nCoV-19, and Sputnik V Vaccines in the Palestinian Population
by
Alexia Damour, Muriel Faure, Nicolas Landrein, Jessica Ragues, Narda Ardah, Haneen Dhaidel, Marie-Edith Lafon, Harald Wodrich and Walid Basha
Vaccines 2024, 12(5), 493; https://doi.org/10.3390/vaccines12050493 - 03 May 2024
Abstract
Since the beginning of the COVID-19 pandemic, different viral vector-based and mRNA vaccines directed against the SARS-CoV-2 “S” spike glycoprotein have been developed and have shown a good profile in terms of safety and efficacy. Nevertheless, an unbiased comparison of vaccination efficiency, including
[...] Read more.
Since the beginning of the COVID-19 pandemic, different viral vector-based and mRNA vaccines directed against the SARS-CoV-2 “S” spike glycoprotein have been developed and have shown a good profile in terms of safety and efficacy. Nevertheless, an unbiased comparison of vaccination efficiency, including post-vaccination neutralizing activity, between the different vaccines remains largely unavailable. This study aimed to compare the efficacy of one mRNA (BNT162b2) and two non-replicating adenoviral vector vaccines (ChAdOx1 nCoV-19 and Sputnik V) in a cohort of 1120 vaccinated Palestinian individuals who received vaccines on an availability basis and which displayed a unique diversity of genetic characteristics. We assessed the level of anti-S antibodies and further determined the antibody neutralizing activity in 261 of those individuals vaccinated with BNT162b2a (121), ChAdOx1 (72) or Sputnik V (68). Our results showed no significant difference in the distribution of serum-neutralizing activity or S-antibody serum levels for the three groups of vaccines, proving equivalence in efficacy for the three vaccines under real-life conditions. In addition, none of the eight demographic parameters tested had an influence on vaccination efficacy. Regardless of the vaccine type, the vaccination campaign ultimately played a pivotal role in significantly reducing the morbidity and mortality associated with COVID-19 in Palestine.
Full article
(This article belongs to the Special Issue Advances in COVID-19 Vaccines and Neutralizing Antibody)
►▼
Show Figures

Figure 1
Open AccessArticle
Pharmacokinetic and Environmental Risk Assessment of Prime-2-CoV, a Non-Replicating Orf Virus-Based Vaccine against SARS-CoV-2
by
Carina Metz, Verena Haug, Melanie Müller and Ralf Amann
Vaccines 2024, 12(5), 492; https://doi.org/10.3390/vaccines12050492 - 02 May 2024
Abstract
Viral vector vaccines represent a substantial advancement in immunization technology, offering numerous benefits over traditional vaccine modalities. The Orf virus (ORFV) strain D1701-VrV is a particularly promising candidate for vaccine development due to its distinctive attributes, such as a good safety profile, the
[...] Read more.
Viral vector vaccines represent a substantial advancement in immunization technology, offering numerous benefits over traditional vaccine modalities. The Orf virus (ORFV) strain D1701-VrV is a particularly promising candidate for vaccine development due to its distinctive attributes, such as a good safety profile, the ability to elicit both humoral and cellular immunity, and its favorable genetic and thermal stability. Despite ORFV’s theoretical safety advantages, such as its narrow host range and limited systemic spread post-inoculation, a critical gap persists between these theoretical benefits and the empirical evidence regarding its in vivo safety profile. This discrepancy underscores the need for comprehensive preclinical validations to bridge this knowledge gap, especially considering ORFV’s use in humans. Our research introduces Prime-2-CoV, an innovative ORFV-based vaccine candidate against COVID-19, designed to elicit a robust immune response by expressing SARS-CoV-2 Nucleocapsid and Spike proteins. Currently under clinical trials, Prime-2-CoV marks the inaugural application of ORFV in human subjects. Addressing the aforementioned safety concerns, our extensive preclinical evaluation, including an environmental risk assessment (ERA) and detailed pharmacokinetic studies in rats and immunocompromised NOG mice, demonstrates Prime-2-CoV’s favorable pharmacokinetic profile, negligible environmental impact, and minimal ERA risks. These findings not only affirm the vaccine’s safety and efficacy but also pioneer the use of ORFV-based therapeutics, highlighting its potential for wider therapeutic applications.
Full article
(This article belongs to the Special Issue Safety and Immunogenicity of the COVID-19 Vaccine)
►▼
Show Figures

Figure 1
Open AccessArticle
Induction of Superior Systemic and Mucosal Protective Immunity to SARS-CoV-2 by Nasal Administration of a VSV–ΔG–Spike Vaccine
by
Yfat Yahalom-Ronen, Sharon Melamed, Boaz Politi, Noam Erez, Hadas Tamir, Liat Bar-On, Julia Ryvkin, Dena Leshkowitz, Ofir Israeli, Shay Weiss, Amir Ben-Shmuel, Moria Barlev-Gross, Lilach Cherry Mimran, Hagit Achdout, Nir Paran and Tomer Israely
Vaccines 2024, 12(5), 491; https://doi.org/10.3390/vaccines12050491 - 01 May 2024
Abstract
The emergence of rapidly spreading variants of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) poses a major challenge to vaccines’ protective efficacy. Intramuscular (IM) vaccine administration induces short-lived immunity but does not prevent infection and transmission. New vaccination strategies are needed to extend
[...] Read more.
The emergence of rapidly spreading variants of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) poses a major challenge to vaccines’ protective efficacy. Intramuscular (IM) vaccine administration induces short-lived immunity but does not prevent infection and transmission. New vaccination strategies are needed to extend the longevity of vaccine protection, induce mucosal and systemic immunity and prevent viral transmission. The intranasal (IN) administration of the VSV–ΔG–spike vaccine candidate directly to mucosal surfaces yielded superior mucosal and systemic immunity at lower vaccine doses. Compared to IM vaccination in the K18–hACE2 model, IN vaccination preferentially induced mucosal IgA and T-cells, reduced the viral load at the site of infection, and ameliorated disease-associated brain gene expression. IN vaccination was protective even one year after administration. As most of the world population has been vaccinated by IM injection, we demonstrate the potential of a heterologous IM + IN vaccination regimen to induce mucosal immunity while maintaining systemic immunity. Furthermore, the IM + IN regimen prevented virus transmission in a golden Syrian hamster co-caging model. Taken together, we show that IN vaccination with VSV–ΔG–spike, either as a homologous IN + IN regimen or as a boost following IM vaccination, has a favorable potential over IM vaccination in inducing efficient mucosal immunity, long-term protection and preventing virus transmission.
Full article
(This article belongs to the Special Issue SARS-CoV-2 Variants, Vaccines, and Immune Responses)
Open AccessArticle
Novel Multi-Antigen Orf-Virus-Derived Vaccine Elicits Protective Anti-SARS-CoV-2 Response in Monovalent and Bivalent Formats
by
Dominique Julien Burri, Louis Renz, Melanie Mueller, Felix Pagallies, Ute Klinkhardt, Ralf Amann and Madiha Derouazi
Vaccines 2024, 12(5), 490; https://doi.org/10.3390/vaccines12050490 - 01 May 2024
Abstract
►▼
Show Figures
Prime-2-CoV_Beta is a novel Orf virus (ORFV)-based COVID-19 vaccine candidate expressing both the nucleocapsid and spike proteins of SARS-CoV-2 with the receptor-binding domain (RBD) of the Beta strain. This candidate was shown to be safe and immunogenic in a first-in-human Phase I clinical
[...] Read more.
Prime-2-CoV_Beta is a novel Orf virus (ORFV)-based COVID-19 vaccine candidate expressing both the nucleocapsid and spike proteins of SARS-CoV-2 with the receptor-binding domain (RBD) of the Beta strain. This candidate was shown to be safe and immunogenic in a first-in-human Phase I clinical trial. With the shift in the immune landscape toward the Omicron variant and the widespread vaccine- and/or infection-derived immunity, further pre-clinical research was needed to characterize Prime-2-CoV. Here, we quantified the humoral and cellular response to Prime-2-CoV_Beta in pre-immunized mice and compared the protective efficacy of mono- and bivalent variant-based Prime-2-CoV vaccine candidates in hamsters. Prime-2-CoV_Beta induced robust humoral and cellular immune responses in naïve animals but did not further boost antibody titers in the tested setting when given as repeat booster at short interval. We furthermore showed that Prime-2-CoV_Beta-based mono- and bivalent immunization strategies produced comparable immunogenicity and protection from infection. Our results highlight the potential of the Orf virus as a vaccine platform against SARS-CoV-2 and potentially other infectious viruses.
Full article

Figure 1
Open AccessArticle
Human Papillomavirus Vaccination Acceleration and Introduction in Sub-Saharan Africa: A Multi-Country Cohort Analysis
by
Gbadebo Collins Adeyanju, Tene-Alima Essoh, Annick Raissa Sidibe, Furaha Kyesi and Muyi Aina
Vaccines 2024, 12(5), 489; https://doi.org/10.3390/vaccines12050489 - 01 May 2024
Abstract
Background: Cervical cancer, caused by human papillomavirus (HPV) infection, is the second-largest cancer killer of women in low- and middle-income countries. The brunt of the global burden is borne predominantly in Sub-Saharan Africa. In 2020 alone, 70,000 of the 100,000 infected women in
[...] Read more.
Background: Cervical cancer, caused by human papillomavirus (HPV) infection, is the second-largest cancer killer of women in low- and middle-income countries. The brunt of the global burden is borne predominantly in Sub-Saharan Africa. In 2020 alone, 70,000 of the 100,000 infected women in Africa died from it, thereby making up 21% of global cervical cancer mortality. The introduction of the HPV vaccine into the National Immunization Program was expected to change the trajectory. However, uptake of the vaccination has been poor, especially for the second dose. Only about half of the countries in Africa currently provide the vaccine. Without urgent intervention, the 2030 global cervical cancer elimination targets will be undermined. The study aims to understand the key challenges facing the HPV vaccine and to develop a roadmap to accelerate the uptake. Method: Fourteen countries were purposively included using a cohort design methodology and the investigation spanned March–July 2023. The Africa region was stratified into three focus-group discussion cohorts (Abidjan, Nairobi and Dar es Salaam), comprising pre-selected countries that have already and those about to introduce the HPV vaccine. In each country, the EPI manager, the NITAG chair or representatives and an HPV-focal researcher were selected participants. The methods involved a collaborative and knowledge-sharing format through regional and country-specific discussions, plenary discussions, and workshop-style group missions. Results: The study reached a total of 78 key stakeholders, comprising 30 participants in cohort one, 21 in cohort two and 27 in cohort three. Key outcomes included the prevalence of declining HPV2 vaccination across all countries in the region; country-specific barriers impeding uptake were identified and strategy for accelerating vaccination demand initiated, e.g., utilizing investments from COVID-19 (e.g., electronic registry and multisector coordination); individual countries developing their respective HPV vaccination recovery and acceleration roadmaps; the identification and inclusion of a zero-dose catch-up strategy into the vaccination roadmaps; support for a transition from multiple-doses to a single-dose HPV vaccine; the incorporation of implementation science research to support the decision-making process such as vaccine choices, doses and understanding behavior. Conclusion: Beyond research, the study shows the significance of scientific approaches that are not limited to understanding problems, but are also solution-oriented, e.g., development of roadmaps to overcome barriers against HPV vaccination uptake.
Full article
(This article belongs to the Special Issue Recent Research on Human Papillomavirus (HPV) Infection and Vaccination)
►▼
Show Figures
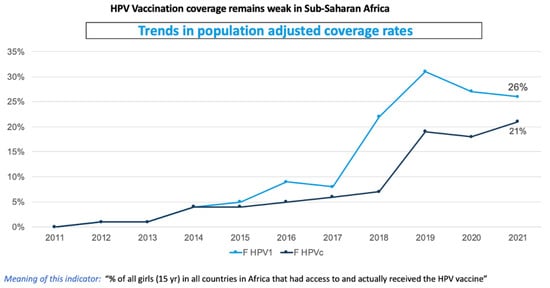
Figure 1
Open AccessArticle
Impact of the Universal Implementation of Adolescent Hepatitis B Vaccination in Spain
by
Angela Domínguez, Ana Avellón, Victoria Hernando, Núria Soldevila, Eva Borràs, Ana Martínez, Conchita Izquierdo, Núria Torner, Carles Pericas, Cristina Rius and Pere Godoy
Vaccines 2024, 12(5), 488; https://doi.org/10.3390/vaccines12050488 - 01 May 2024
Abstract
The aim of this study was to analyse the impact of the introduction of universal adolescent HBV vaccination on the incidence of acute hepatitis B virus (HBV) infections. Acute HBV cases reported to the Spanish National Epidemiological Surveillance Network between 2005 and 2021
[...] Read more.
The aim of this study was to analyse the impact of the introduction of universal adolescent HBV vaccination on the incidence of acute hepatitis B virus (HBV) infections. Acute HBV cases reported to the Spanish National Epidemiological Surveillance Network between 2005 and 2021 were included. For regions starting adolescent vaccination in 1991–1993 and in 1994–1996, HBV incidence rates were compared by calculating the incidence rate ratio (IRR) and 95% confidence interval (CI). We also analysed the 2017 Spanish national seroprevalence survey data. The overall acute HBV incidence per 100,000 persons was 1.54 in 2005 and 0.64 in 2021 (p < 0.001). The incidence in 2014–2021 was lower for regions that started adolescent vaccination in 1991–1993 rather than in 1994–1996 (IRR 0.76; 95% CI 0.72–0.83; p < 0.001). In the 20–29 age group, incidence in regions that started adolescent vaccination in 1991–1993 was also lower (IRR 0.87; 95% CI 0.77–0.98; p = 0.02 in 2005–2013 and IRR 0.71; 95% CI 0.56–0·90; p < 0.001 in 2014–2021). Anti-HBc prevalence in the 35–39 age group was lower in the regions that started vaccination earlier, although the difference was not statistically significant (p = 0.09). Acute HBV incidence decreased more in the young adult population in regions that began adolescent vaccination earlier. Maintaining high universal vaccination coverage in the first year of life and in at-risk groups is necessary to achieve HBV elimination by 2030.
Full article
(This article belongs to the Special Issue Feature Papers of Hepatitis A, B, C and E Vaccines)
►▼
Show Figures
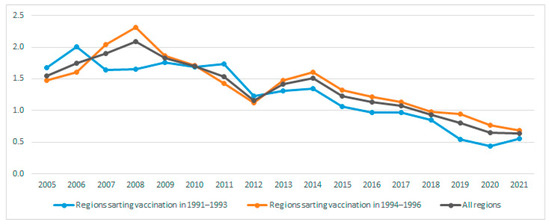
Figure 1

Journal Menu
► ▼ Journal Menu-
- Vaccines Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Diseases, IJMS, Microbiology Research, Pathogens, Vaccines
Advances in Human Pathogen Control—a 21st Century Challenge 2.0
Topic Editors: Jorge H. Leitão, Nitin Amdare, Joana R FelicianoDeadline: 30 June 2024
Topic in
Cells, Diseases, Healthcare, IJMS, Vaccines
Inflammation: The Cause of all Diseases 2.0
Topic Editors: Vasso Apostolopoulos, Jack Feehan, Vivek P. ChavdaDeadline: 31 July 2024
Topic in
Biomedicines, JCM, Pathogens, Vaccines, Viruses
Discovery and Development of Monkeypox Disease Treatments
Topic Editors: Mohd Imran, Ali A. RabaanDeadline: 31 August 2024
Topic in
Brain Sciences, Clinics and Practice, COVID, Life, Vaccines, Viruses
Multifaceted Efforts from Basic Research to Clinical Practice in Controlling COVID-19 Disease
Topic Editors: Yih-Horng Shiao, Rashi OjhaDeadline: 30 September 2024

Conferences
Special Issues
Special Issue in
Vaccines
Novel Vaccines for Infectious Pathogens
Guest Editors: Veerupaxagouda Patil, Dhruv DesaiDeadline: 20 May 2024
Special Issue in
Vaccines
COVID-19 Vaccine Acceptance and Uptake: Insights from Behavioural and Social Sciences
Guest Editor: Christian NapoliDeadline: 31 May 2024
Special Issue in
Vaccines
SARS-CoV-2 Variants, Vaccines, and Immune Responses
Guest Editor: Gianni Gori SavelliniDeadline: 15 June 2024
Special Issue in
Vaccines
Advance in Nanoparticles as Vaccine Adjuvants
Guest Editors: Eduardo Gomez-Casado, Sohrab AhmadivandDeadline: 30 June 2024
Topical Collections
Topical Collection in
Vaccines
COVID-19 Vaccine Hesitancy: Correlates and Interventions
Collection Editors: Manoj Sharma, Kavita Batra
Topical Collection in
Vaccines
Topic Advisory Panel Members’ Collection Series: Immunization and Vaccines for Infectious Diseases
Collection Editors: Shumaila Hanif, Ravinder Kumar
Topical Collection in
Vaccines
Research on Monoclonal Antibodies and Antibody Engineering
Collection Editor: Tatsuya Yamazaki