Journal Description
Drugs and Drug Candidates
Drugs and Drug Candidates
is an international, peer-reviewed, open access journal on drug discovery, development, and knowledge, published quarterly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 22.4 days after submission; acceptance to publication is undertaken in 6.6 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: APC discount vouchers, optional signed peer review, and reviewer names published annually in the journal.
- Drugs and Drug Candidates is a companion journal of Pharmaceuticals.
Latest Articles
Essential Oil of Psidium glaziovianum Kiaersk Alleviates the Effects of Complete Freund’s Adjuvant (CFA)-Induced Arthritis by Regulating Inflammation and Oxidative Stress
Drugs Drug Candidates 2024, 3(2), 380-390; https://doi.org/10.3390/ddc3020023 - 07 May 2024
Abstract
Rheumatoid arthritis (RA) is a chronic and debilitating condition that affects a significant number of individuals worldwide. Unfortunately, the currently available therapeutic approaches often yield unsatisfactory results and may be accompanied by harmful side effects. A medicinal plant called Psidium glaziovianum Kiaersk has
[...] Read more.
Rheumatoid arthritis (RA) is a chronic and debilitating condition that affects a significant number of individuals worldwide. Unfortunately, the currently available therapeutic approaches often yield unsatisfactory results and may be accompanied by harmful side effects. A medicinal plant called Psidium glaziovianum Kiaersk has potential benefits in the treatment of this condition due to its anti-inflammatory and analgesic properties. In this study, our objective was to investigate the potential therapeutic effects of P. glaziovianum essential oil (PgEO) in alleviating arthritis symptoms in mice induced by Complete Freund’s Adjuvant (CFA). The effect of P. glaziovianum essential oil was evaluated in mice with Complete Freund’s Adjuvant (CFA)-induced arthritis. Edema sizes, macroscopic and radiographic images, cytokine levels, and oxidative stress were evaluated. Administration of PgEO at dosages of 50 and 100 mg/kg effectively prevented CFA-induced osteoarticular changes in arthritic mice, resulting in a significant reduction in joint damage. Additionally, the PgEO treatment exhibited the ability to minimize edema, a common symptom associated with arthritis. Furthermore, PgEO can modulate the levels of pro-inflammatory cytokines and oxidative stress, both of which play crucial roles in the progression of the disease. In conclusion, our study suggests that PgEO holds great potential as a natural therapeutic agent for rheumatoid arthritis.
Full article
(This article belongs to the Section Preclinical Research)
►
Show Figures
Open AccessArticle
Preclinical Testing of Chronic ICA-1S Exposure: A Potent Protein Kinase C-ι Inhibitor as a Potential Carcinoma Therapeutic
by
Christopher A. Apostolatos, Wishrawana S. Ratnayake, Sloan Breedy, Jacqueline Kai Chin Chuah, James Alastair Miller, Daniele Zink, Marie Bourgeois and Mildred Acevedo-Duncan
Drugs Drug Candidates 2024, 3(2), 368-379; https://doi.org/10.3390/ddc3020022 - 07 May 2024
Abstract
Protein kinase C-iota (PKC-ι) is an oncogene overexpressed in many cancer cells including prostate, breast, ovarian, melanoma, and glioma cells. Previous in vitro studies have shown that 5-amino-1-((1R,2S,3R,4R)-2-3-dihydroxy-4-(hydroxymethyl)cyclopentyl)-1H-imidazole-4-carboxamide (ICA-1S), a PKC-ι-specific inhibitor, has low toxicity in both acute and sub-acute mouse model toxicological
[...] Read more.
Protein kinase C-iota (PKC-ι) is an oncogene overexpressed in many cancer cells including prostate, breast, ovarian, melanoma, and glioma cells. Previous in vitro studies have shown that 5-amino-1-((1R,2S,3R,4R)-2-3-dihydroxy-4-(hydroxymethyl)cyclopentyl)-1H-imidazole-4-carboxamide (ICA-1S), a PKC-ι-specific inhibitor, has low toxicity in both acute and sub-acute mouse model toxicological testing and is an effective therapeutic against several cancer cell lines showing significant reductions in tumor growth when treating athymic nude mice with xenografted carcinoma cell lines. To further assess ICA-1S as a possible therapeutic agent, chronic mouse model toxicological testing was performed in vivo to provide inferences concerning the long-term effects and possible health hazards from repeated exposure over a substantial part of the animal’s lifespan. Subjects survived well after 30, 60, and 90 days of doses ranging from 50 mg/kg to 100 mg/kg. Heart, liver, kidney, and brain tissues were then analyzed for accumulations of ICA-1S including the measured assessment of aspartate transaminase (AST), alkaline phosphatase (ALK-P), gamma-glutamyl transferase (GGT), troponin, and C-reactive protein (CRP) serum levels to assess organ function. Predictive in vitro/in silico methods were used to predict compound-induced direct hepatocyte toxicity or renal proximal tubular cell (PTC) toxicity in humans based on the high-content imaging (HCI) of compound-treated cells in combination with phenotypic profiling. In conclusion, ICA-1S shows low toxicity in both acute and chronic toxicology studies, and shows promise as a potential therapeutic.
Full article
(This article belongs to the Section Preclinical Research)
►▼
Show Figures
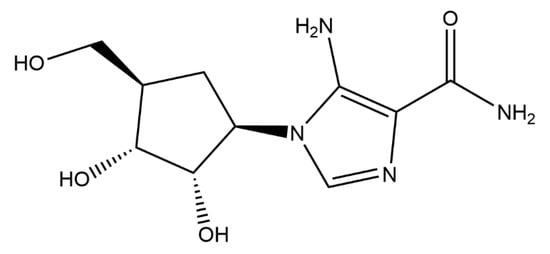
Figure 1
Open AccessArticle
Toxicity and Teratogenic Potential of Piplartine from Piper tuberculatum Jacq. during Embryonic Development in Mice (Mus musculus)
by
Giulliano Rezende Silva, Lívia Thaís Gontijo Miranda, Shirley Aline da Costa Arteaga da Silva, Laise Rodrigues de Andrade, Natanael Carvalho de Souza, Bruno Silva Sá, Elivaldo Ribeiro de Santana, Andreanne Gomes Vasconcelos, Daniel Carneiro Moreira, Aline Pic-Taylor, Alessandra Durazzo, Massimo Lucarini, Lydia Fumiko Yamaguchi, Massuo Jorge Kato, Amilcar Sabino Damazo, Daniel Dias Rufino Arcanjo, José Roberto de Souza de Almeida Leite and José Eduardo Baroneza
Drugs Drug Candidates 2024, 3(2), 353-367; https://doi.org/10.3390/ddc3020021 - 30 Apr 2024
Abstract
Piplartine, also known as piperlongumine, is a natural and biologically active amide alkaloid found in various Piper species within the Piperaceae family. It possesses numerous beneficial properties that can be leveraged in the development of nanotechnological and pharmaceutical products. However, information on the
[...] Read more.
Piplartine, also known as piperlongumine, is a natural and biologically active amide alkaloid found in various Piper species within the Piperaceae family. It possesses numerous beneficial properties that can be leveraged in the development of nanotechnological and pharmaceutical products. However, information on the effects of piplartine on mammalian embryonic development is scarce. This study aims to assess the general toxicity and teratogenic potential of piplartine during the embryonic development of mice. Pregnant mice received daily treatments of 25, 50, or 100 mg/kg of piplartine via gavage from the sixth day of gestation (implantation) to the eighteenth. On the eighteenth day, the mice were euthanized, and whole organs, blood samples (for hematological and biochemical analyses), and bone marrow cells (for DNA fragmentation and cell cycle assays) were collected. The uterus was examined for implantation sites and embryo resorptions. Additionally, fetuses were collected to assess for fetal anomalies. Piplartine did not result in maternal or embryo-fetal toxicity, induce fetal anomalies, cause hematological and biochemical alterations, or lead to DNA fragmentation. The oral administration of piplartine is safe and does not exhibit toxicity or teratogenic effects in mice. This finding opens avenues for the development of piplartine-based biotechnological products for therapeutic interventions in disease treatment.
Full article
(This article belongs to the Section Preclinical Research)
►▼
Show Figures
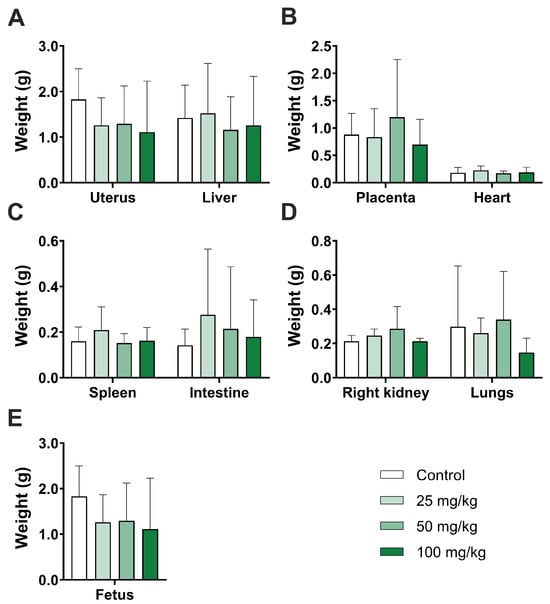
Figure 1
Open AccessArticle
Relaxant Activity of 4H-Pyran and 1,6-Dihydropyridine Derivatives on Isolated Rat Trachea
by
Samuel Estrada-Soto, Soledad Alemán-Pantitlán, Emmanuel Gaona-Tovar, Fernando Hernández-Borja, Yolanda Alcaraz, Rafael Villalobos-Molina and Miguel A. Vázquez
Drugs Drug Candidates 2024, 3(2), 342-352; https://doi.org/10.3390/ddc3020020 - 11 Apr 2024
Abstract
Derivatives of 4H-pyrans and 1,6-dihydropyridines have generated considerable attention due to their interesting biological and therapeutic values. Their pharmacological activities include vasorelaxant, anticarcinogenic, antimicrobial, and antioxidant activities. Thus, the aim of the current work is to determine the relaxant effect of synthesized 4H-pyran
[...] Read more.
Derivatives of 4H-pyrans and 1,6-dihydropyridines have generated considerable attention due to their interesting biological and therapeutic values. Their pharmacological activities include vasorelaxant, anticarcinogenic, antimicrobial, and antioxidant activities. Thus, the aim of the current work is to determine the relaxant effect of synthesized 4H-pyran and 1,6-dihydropyridine derivatives with potential anti-asthmatic properties on the smooth muscle airway, with a possible Ca2+-channel blockade as a mechanism of action due to their analogy with 1,4-dihidropyridines. 4H-pyrans and 1,6-dihydropyridines were achieved using multicomponent reactions by microwave and conventional heating. Also, test samples were evaluated ex vivo to determine their relaxant effect on isolated rat tracheal rings pre-contracted with carbachol. All compounds evaluated showed a significant relaxant effect on carbachol-induced contraction in tracheal rat rings. Compounds 4b, 4e, 7a, and 8d were the most potent from the entire series and were also more potent than theophylline, used as a positive control. In conclusion, in the current work some relaxant compounds of the airway smooth muscle with potential to be developed as anti-asthmatic drugs were obtained.
Full article
(This article belongs to the Section Medicinal Chemistry and Preliminary Screening)
►▼
Show Figures
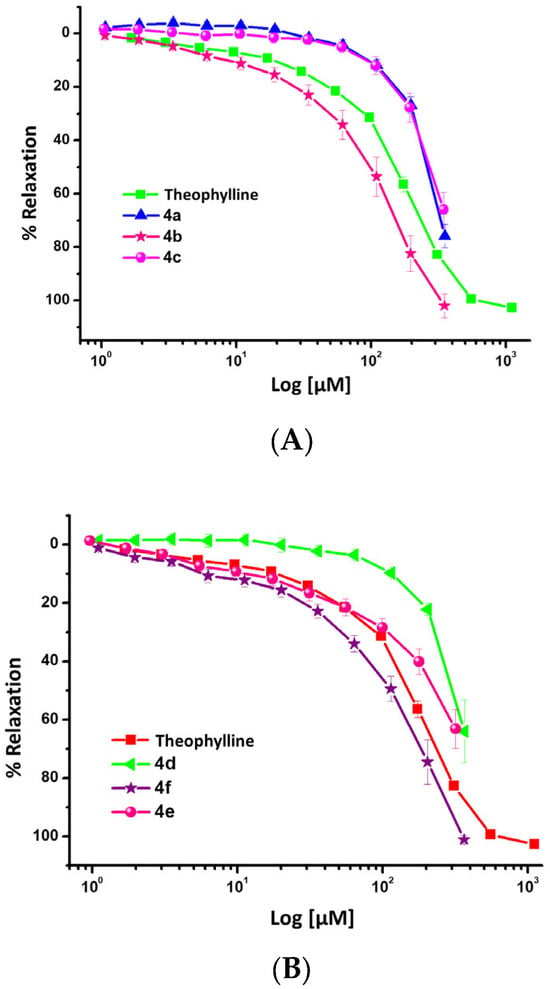
Figure 1
Open AccessReview
Sebetralstat: A Rapidly Acting Oral Plasma Kallikrein Inhibitor for the On-Demand Treatment of Hereditary Angioedema
by
Edward P. Feener, Rebecca L. Davie, Nivetha Murugesan, Stephen J. Pethen, Sally L. Hampton, Michael D. Smith, Paul K. Audhya and Chris M. Yea
Drugs Drug Candidates 2024, 3(2), 328-341; https://doi.org/10.3390/ddc3020019 - 07 Apr 2024
Abstract
Sebetralstat is a novel, potent, and selective oral plasma kallikrein inhibitor drug candidate in clinical development for the on-demand treatment of hereditary angioedema (HAE). Upon binding, sebetralstat induces a conformational change in the active site of plasma kallikrein, which contributes to its high
[...] Read more.
Sebetralstat is a novel, potent, and selective oral plasma kallikrein inhibitor drug candidate in clinical development for the on-demand treatment of hereditary angioedema (HAE). Upon binding, sebetralstat induces a conformational change in the active site of plasma kallikrein, which contributes to its high potency (Ki 3 nM) and selectivity (>1500 fold) against other serine proteases. Its physiochemical properties promote both rapid dissolution in the stomach and rapid absorption in the upper intestine that contribute to its fast and efficient absorption. A single oral administration of sebetralstat rapidly provides near-complete inhibition of plasma kallikrein and blockade of high-molecular-weight kininogen cleavage as early as 15 min, which drives its clinical efficacy. In a phase 2 clinical trial, sebetralstat significantly reduced the time to beginning of symptom relief (p < 0.0001) with median times of 1.6 h (95% CI: 1.5–3.0) with sebetralstat versus 9.0 h (4.0–17.2) with placebo. KONFIDENT (NCT05259917) is a phase 3 clinical trial assessing the on-demand use of sebetralstat for HAE. If successful, this trial could support the approval of sebetralstat as the first noninvasive, on-demand treatment option to rapidly halt HAE attacks and provide fast symptom relief.
Full article
(This article belongs to the Special Issue Drugs of the Kallikrein-Kinin System)
►▼
Show Figures
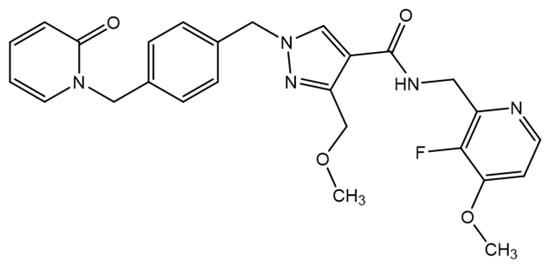
Figure 1
Open AccessFeature PaperReview
Zilucoplan: A Newly Approved Macrocyclic Peptide for Treatment of Anti-Acetylcholine Receptor Positive Myasthenia Gravis
by
Lia Costa and Carla Fernandes
Drugs Drug Candidates 2024, 3(2), 311-327; https://doi.org/10.3390/ddc3020018 - 27 Mar 2024
Abstract
Zilucoplan is a synthetic macrocyclic peptide approved by the Food and Drug Administration (FDA), in October 2023, for the treatment of generalized myasthenia gravis. It is considered as an orphan drug that causes the inhibition of terminal complement cascade activation with a dual
[...] Read more.
Zilucoplan is a synthetic macrocyclic peptide approved by the Food and Drug Administration (FDA), in October 2023, for the treatment of generalized myasthenia gravis. It is considered as an orphan drug that causes the inhibition of terminal complement cascade activation with a dual mechanism of action preventing the formation of the membrane attack complex (MAC) and the destruction of the neuromuscular junction. This drug has been demonstrated to be able to treat the generalized myasthenia gravis without significant adverse effects, with good efficacy, safety, and tolerability profile. Zilucoplan is not only innovative and promising in the therapeutics of generalized myasthenia gravis, but it could also be beneficial for the treatment of other diseases as well as a model for synthesis of analogues to improve pharmacological profile.
Full article
(This article belongs to the Section Marketed Drugs)
►▼
Show Figures
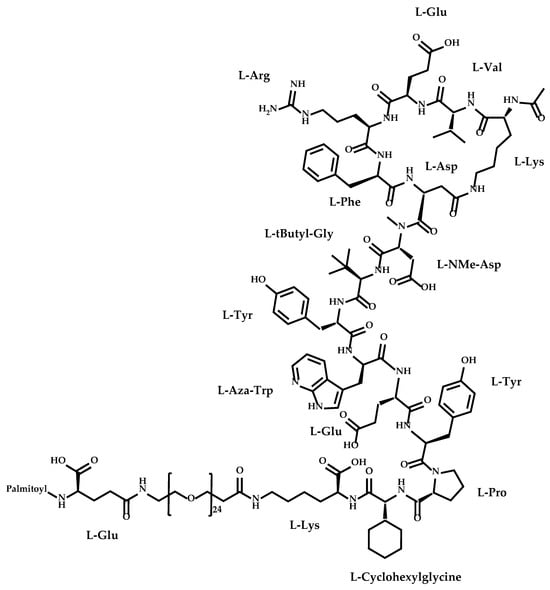
Figure 1
Open AccessArticle
DFT Calculations, Pro-Apoptotic Effects, and Anti-Infective Investigations of Alkaloids Isolated from the Stem Bark Extract of Enantia chlorantha
by
Vincent O. Imieje, Ahmed A. Zaki, Mansour A. E. Bashar, Islam Rady, Mohamed A. M. El-Tabakh, Mohamed A. E. Abd El-Aziz, Eman. S. Abou-Amra, Shahd Yasser, Ibraheem M. M. Gobaara, Mohammed A. S. Abourehab, Reham M. Samra, Hussein A. El-Naggar and Abiodun Falodun
Drugs Drug Candidates 2024, 3(1), 291-310; https://doi.org/10.3390/ddc3010017 - 07 Mar 2024
Abstract
Fractionation of the stem bark of Enantia chlorantha Oliv yields three alkaloids, palmatine (1), jatrorrhizine (2), columbamine (3), and β-Sitosterol (4). In this investigation, density functional theory (DFT) calculations were carried out to evaluate
[...] Read more.
Fractionation of the stem bark of Enantia chlorantha Oliv yields three alkaloids, palmatine (1), jatrorrhizine (2), columbamine (3), and β-Sitosterol (4). In this investigation, density functional theory (DFT) calculations were carried out to evaluate the electronic structure and properties of 1–4 by DFT-B3LYP/6-31G level of theory using Gaussian 09 software. The highest occupied molecular orbital (HOMO), lowest unoccupied molecular orbital (LUMO), HOMO-LUMO energy difference (band gap), hardness (η), softness (S), dipole moment (μ), electronegativity (χ), hydrophobicity (logP), topological surface area (TPSA), and energy gap (Eg) were calculated. The in vitro cytotoxicity of the compounds was investigated against MCF-7 and HCT116 cancer cell lines using Wi-38 cells as a control. The compounds inhibited the proliferation of the MCF-7 and HCT116 cell lines and induced apoptosis via upregulation of caspase-3, Bax, PARP cleavage, and downregulation of Bcl-2. DFT analyses revealed that compounds 1 and 3 have smaller energy gaps, 0.072 and 0.071eV, respectively, with the highest dipole moments; hence, these compounds are more chemically reactive and exhibit better modulation of caspase-3 enzyme and inhibitory activities of the MCF-3 and HCT116 cell lines. The antimicrobial and antiparasitic evaluation of 1–4 showed moderate efficacy against the bacterial strains and moderate antiparasitic activity against Cichlidogyrus tilapia.
Full article
(This article belongs to the Section Drug Candidates from Natural Sources)
►▼
Show Figures
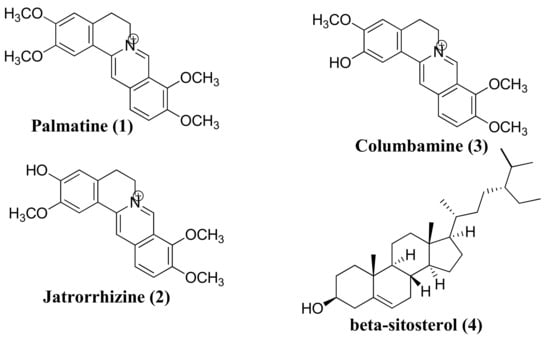
Figure 1
Open AccessArticle
Development of Paclitaxel Proliposomal Dry Powder Inhaler (PTX-PLM-DPI) by Freeze-Drying Method for Lung Cancer
by
Chinmoyee Borah, Trideep Saikia, Alakesh Bharali, Madhuchandra Lahan, Nikhil Biswas and Bhanu P Sahu
Drugs Drug Candidates 2024, 3(1), 275-290; https://doi.org/10.3390/ddc3010016 - 05 Mar 2024
Abstract
Despite various efforts, a successful selective delivery system for chemotherapeutic agents for lung cancer is still lacking. Dry powder inhaler (DPI) systems based on proliposomes (PLMs) could be a potential system for the efficient delivery of paclitaxel to lungs. PLM-based DPI prepared with
[...] Read more.
Despite various efforts, a successful selective delivery system for chemotherapeutic agents for lung cancer is still lacking. Dry powder inhaler (DPI) systems based on proliposomes (PLMs) could be a potential system for the efficient delivery of paclitaxel to lungs. PLM-based DPI prepared with a freeze-drying method can therefore be an alternative. Paclitaxel-loaded PLM-based DPI (PTX-PLM-DPI) powders were prepared using the method of thin film deposition on a carrier followed by freeze drying. These were prepared using soya phosphatidylcholine (SPC) and cholesterol as the lipids and mannitol as the carrier. The reconstituted liposomes were evaluated in terms of size, morphology, drug entrapment, release and cytotoxicity. The DPI powders were evaluated for their flow property, surface topography, dose uniformity and in vitro lung deposition. Stable and free-flowing PTX-PLM-DPI powder was obtained that could be reconstituted into homogenous liposomal vesicles < 200 nm as confirmed by TEM and SEM studies. The liposomes showed drug entrapment of 92.64 ± 1.4% and diffusion-controlled release of up to 28% in 24 h. These liposomes showed better dose-dependent cytotoxicity in A549 cells in comparison to paclitaxel suspension with IC50 values of 46 ± 0.87 ng/mL and 154.9 ± 3.64 ng/mL, respectively. In vitro lung deposition studies of the PTX-PLM-DPI showed sufficient deposition with the fine particle fraction (FPF) of 50.86 ± 2.8% of particles with an aerodynamic diameter less than 5 µ. Hence, it canbe concluded that PLM-based DPI prepared by freeze drying can be a promising, stable, safe and free-flowing system for the enhanced lung delivery of paclitaxel.
Full article
(This article belongs to the Section Preclinical Research)
►▼
Show Figures

Figure 1
Open AccessArticle
Anti-Shigellosis Activity and Mechanisms of Action of Extracts from Diospyros gilletii Stem Bark
by
Audrey Carrel Nguelo Talla, Eugénie Aimée Madiesse Kemgne, Vincent Ngouana, Bijou-Lafortune Noumboue Kouamou, Listone Monelle Nzeye Ngameni, Brice Rostan Pinlap, Yanick Kevin Dongmo Melogmo, Branly-Natalien Nguena-Dongue, Boniface Pone Kamdem, Paul Keilah Lunga and Fabrice Fekam Boyom
Drugs Drug Candidates 2024, 3(1), 256-274; https://doi.org/10.3390/ddc3010015 - 01 Mar 2024
Abstract
Shigellosis is a pathological condition that affects the digestive system and possibly causes diarrhoea. Shigella species, which are responsible for this disease, are highly contagious and spread through contaminated food and water. The increasing development of resistance by Shigella species necessitates the urgent
[...] Read more.
Shigellosis is a pathological condition that affects the digestive system and possibly causes diarrhoea. Shigella species, which are responsible for this disease, are highly contagious and spread through contaminated food and water. The increasing development of resistance by Shigella species necessitates the urgent need to search for new therapies against diarrhoea-causing shigellosis. The scientific validation of medicinal plants, such as Diospyros gilletii, which is used for the traditional treatment of diarrhoeal conditions is worthwhile. The present study aims to investigate the antibacterial activity of extracts from D. gilletii against selected Shigella species. Extracts from D. gilletii stem bark were prepared by maceration using various solvents. The antibacterial activity of D. gilletii extracts was evaluated in Shigella dysenteriae, S. flexneri, S. boydii, and S. sonnei using a microdilution method, whereas a cytotoxicity test was performed on Vero and Raw cells using resazurin-based colorimetric assays. Bacterial membrane-permeability studies were evaluated using propidium iodide (PI)- and 1-N-phenyl-naphthylamine (NPN)-uptake assays, whereas inhibition and eradication tests on bacterial biofilms were carried out by spectrophotometry. As a result, methanol, ethanol and hydroethanol (water: ethanol; 30:70, v/v) extracts of D. gilletii inhibited the growth of S. boydii, S. flexneri and S. sonnei, with minimum inhibitory concentration (MIC) values ranging from 125 to 500 µg/mL, without toxicity to Vero and Raw cells. Time-kill kinetics revealed bactericidal orientation at 2 MIC and 4 MIC and a bacteriostatic outcome at 1/2 MIC. The mechanistic basis of antibacterial action revealed that D. gilletii extracts inhibited and eradicated Shigella biofilms and promoted the accumulation of NPN and PI within the inner and outer membranes of bacteria to increase membrane permeability, thereby causing membrane damage. This novel contribution toward the antibacterial mechanisms of action of D. gilletii extracts against Shigella species substantiates the use of this plant in the traditional treatment of infectious diarrhoea.
Full article
(This article belongs to the Section Drug Candidates from Natural Sources)
►▼
Show Figures

Figure 1
Open AccessArticle
Susceptibility Patterns in Staphylococcus and Klebsiella Causing Nosocomial Infections upon Treatment with E-Anethole-Rich Essential Oil from Clausena anisata
by
François Nguimatsia, Evariste Josué Momo, Paul Keilah Lunga, Virginia Lum Tamanji, Boniface Pone Kamdem and Pierre Michel Jazet Dongmo
Drugs Drug Candidates 2024, 3(1), 244-255; https://doi.org/10.3390/ddc3010014 - 01 Mar 2024
Abstract
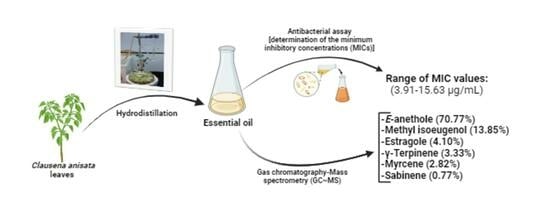
High rates of resistance to antibiotics are associated with healthcare-related infections, thus demonstrating the urgent need for effective antimicrobials against these maladies. The present study aims to determine the chemical composition of essential oil (EO) from Clausena anisata leaves and evaluate their antibacterial
[...] Read more.
High rates of resistance to antibiotics are associated with healthcare-related infections, thus demonstrating the urgent need for effective antimicrobials against these maladies. The present study aims to determine the chemical composition of essential oil (EO) from Clausena anisata leaves and evaluate their antibacterial activity against selected nosocomial bacteria. To this end, one kilogram (1 kg) of fresh leaves of C. anisata was washed and boiled with 500 mL of distilled water for 2−4 h using a Clevenger apparatus. The oil was then collected in an Erlenmeyer, dried using anhydrous sodium sulfate, bottled in a tinted glass bottle and refrigerated at 4 °C before analysis. Next, the as-prepared oil was analyzed using gas chromatography-mass spectrometry (GC-MS). The essential oil was further tested against a panel of selected nosocomial bacteria, including Staphylococcus and Klebsiella species, among others, by microdilution using a resazurin assay to determine the minimum inhibitory and minimum bactericidal concentrations (MICs and MBCs, respectively). As a result, 0.77% of EO was extracted from fresh leaves of C. anisata. The GC-MS analysis revealed that the as-prepared essential oil contained E-anethole (70.77%), methyl isoeugenol (13.85%), estragole (4.10%), γ-terpinene (3.33%), myrcene (2.82%) and sabinene (0.77%), with E-anethole being the major constituent. Twenty-two compounds were identified in the EO of C. anisata leaves through gas chromatography. Upon antibacterial testing against selected nosocomial pathogens, the E-anethole-rich essential oil exhibited MIC and MBC values ranging from 3.91 to 125 µg/mL and 7.81 to 125 µg/mL, respectively, indicative of a bactericidal orientation of the plant’s essential oil (MIC/MBC ratio < 4). This novel contribution highlights the scientific validation of the use of C. anisata leaves in the traditional treatment of various infectious diseases. However, toxicity and pharmacokinetic studies, mechanistic bases of the antibacterial action, and in vivo antibacterial experiments of the E-anethole-rich EO of C. anisata should be investigated to successfully use this plant in the treatment of infectious diseases.
Full article
(This article belongs to the Section Drug Candidates from Natural Sources)
►▼
Show Figures
Graphical abstract
Open AccessConference Report
31st Annual GP2A Medicinal Chemistry Conference
by
Nicolas Primas, Caroline Castera-Ducros, Romain Paoli-Lombardo, Christophe Curti, Fanny Mathias, Pascal Rathelot, Pascal Marchand and Patrice Vanelle
Drugs Drug Candidates 2024, 3(1), 209-243; https://doi.org/10.3390/ddc3010013 - 22 Feb 2024
Abstract
The Group for the Promotion of Pharmaceutical Chemistry in Academia (GP2A) held its 31st annual conference in August 2023 at the Faculty of Pharmacy of Aix-Marseille University, Marseille, France. There were 8 keynote presentations, 10 early career researcher oral presentations and
[...] Read more.
The Group for the Promotion of Pharmaceutical Chemistry in Academia (GP2A) held its 31st annual conference in August 2023 at the Faculty of Pharmacy of Aix-Marseille University, Marseille, France. There were 8 keynote presentations, 10 early career researcher oral presentations and 23 poster presentations. Among them, four awards were delivered, two for best oral communications and two for the best poster presentations.
Full article
(This article belongs to the Section Medicinal Chemistry and Preliminary Screening)
►▼
Show Figures
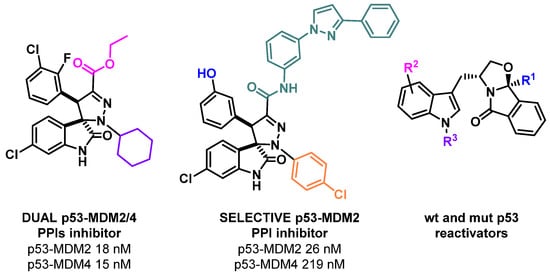
Figure 1
Open AccessCorrection
Correction: Marceau, F. Drugs of the Kallikrein–Kinin System: An Overview. Drugs Drug Candidates 2023, 2, 538–553
by
François Marceau
Drugs Drug Candidates 2024, 3(1), 208; https://doi.org/10.3390/ddc3010012 - 22 Feb 2024
Abstract
In the original publication [...]
Full article
(This article belongs to the Special Issue Drugs of the Kallikrein-Kinin System)
Open AccessReview
Plant-Derived Natural Products: A Source for Drug Discovery and Development
by
Noureddine Chaachouay and Lahcen Zidane
Drugs Drug Candidates 2024, 3(1), 184-207; https://doi.org/10.3390/ddc3010011 - 19 Feb 2024
Cited by 2
Abstract
For thousands of years, nature has been a source of medical substances, and an astounding numeral of contemporary remedies have been identified from natural origins. Plants have long been used as folk herbal medicines to treat various disorders, and their different natural products
[...] Read more.
For thousands of years, nature has been a source of medical substances, and an astounding numeral of contemporary remedies have been identified from natural origins. Plants have long been used as folk herbal medicines to treat various disorders, and their different natural products have inspired the design, discovery, and development of new drugs. With the invention of recent molecular targets based on proteins, there is a growing need for fresh chemical diversification in screening. Natural products will play a vital part in supplying this need via the continuous exploration of global biodiversity, the majority of which remains unexplored. Even though drug discovery from medicinal plants remains an important source of novel therapeutic leads, various hurdles exist, including identifying and executing suitable high-throughput screening bioassays, scaling up the supply of bioactive molecules, and acquiring plant materials. Investigating these natural resources takes multi-disciplinary, nationwide, and global partnerships in design, synthesis, discovery, and drug development techniques. This review article discusses current advancements and future approaches for discovering natural items such as health- and wellness-promoting remedies. It also summarizes strategies to unify the therapeutic use of plant-derived natural products worldwide to support future drug discoveries derived from plant sources.
Full article
(This article belongs to the Section Drug Candidates from Natural Sources)
►▼
Show Figures
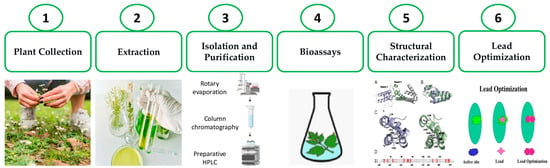
Figure 1
Open AccessArticle
Isolation and Characterization of Antimicrobial Constituent(s) from the Stem of Cissus populnea Guill. & Perr.
by
Anita Alex-Asaolu, Ahmad Uba, Umar Abubakar Birnin-Yauri and Amina Jega Yusuf
Drugs Drug Candidates 2024, 3(1), 172-183; https://doi.org/10.3390/ddc3010010 - 19 Feb 2024
Abstract
Cissus populnea Guill. & Perr. (Vitaceae) is used in traditional medicine to treat microbial infections, venereal diseases and infertility, among others. The aim of this research is to isolate and characterize the antimicrobial constituent(s) from the stem of C. populnea. The n-butanol
[...] Read more.
Cissus populnea Guill. & Perr. (Vitaceae) is used in traditional medicine to treat microbial infections, venereal diseases and infertility, among others. The aim of this research is to isolate and characterize the antimicrobial constituent(s) from the stem of C. populnea. The n-butanol fraction of C. populnea, being most active, was subjected to silica gel column chromatography, which led to the isolation of white solid and white crystalline substances coded compounds C1 and C4C5, respectively. Spectral analysis (1D and 2D-NMR) of the isolated compounds and comparison with the literature data indicated C1 to be Bis-(2-ethyloctyl)-phthalate and C4C5 to be a mixture of stigmasterol and β-sitosterol; C4C5 exhibited a zone of inhibition ranging from 24 to 29 mm against the test organisms with Candida albicans being the most sensitive organism while Trichophyton rubrum was the least sensitive organism. Of the standard drugs, ciprofloxacin had 27–37 mm while fluconazole and fulcin exhibited zones of inhibition ranging from 34 mm to 29–32 mm, respectively. The MIC and MBC/MFC values for C4C5 ranges from 12.5 to 25.0 µg/cm3 and 25.0 to 50.0 µg/cm3 against methicillin-resistant Staphylococcus aureus, Staphylococcus. aureus, Escherichia coli, Candida albicans, Trichophyton rubrum and Trichophyton mentagrophyte, respectively. In conclusion, Bis-(-(2-ethyloctyl)-phthalate) and a mixture of stigmasterol and β-sitosterol were identified for the first time from the stem of C. populnea.
Full article
(This article belongs to the Section Drug Candidates from Natural Sources)
►▼
Show Figures

Figure 1
Open AccessReview
Revolutionizing Drug Discovery: A Comprehensive Review of AI Applications
by
Rushikesh Dhudum, Ankit Ganeshpurkar and Atmaram Pawar
Drugs Drug Candidates 2024, 3(1), 148-171; https://doi.org/10.3390/ddc3010009 - 13 Feb 2024
Abstract
The drug discovery and development process is very lengthy, highly expensive, and extremely complex in nature. Considering the time and cost constraints associated with conventional drug discovery, new methods must be found to enhance the declining efficiency of traditional approaches. Artificial intelligence (AI)
[...] Read more.
The drug discovery and development process is very lengthy, highly expensive, and extremely complex in nature. Considering the time and cost constraints associated with conventional drug discovery, new methods must be found to enhance the declining efficiency of traditional approaches. Artificial intelligence (AI) has emerged as a powerful tool that harnesses anthropomorphic knowledge and provides expedited solutions to complex challenges. Advancements in AI and machine learning (ML) techniques have revolutionized their applications to drug discovery and development. This review illuminates the profound influence of AI on diverse aspects of drug discovery, encompassing drug-target identification, molecular properties, compound analysis, drug development, quality assurance, and drug toxicity assessment. ML algorithms play an important role in testing systems and can predict important aspects such as the pharmacokinetics and toxicity of drug candidates. This review not only strengthens the theoretical foundation and development of this technology, but also explores the myriad challenges and promising prospects of AI in drug discovery and development. The combination of AI and drug discovery offers a promising strategy to overcome the challenges and complexities of the pharmaceutical industry.
Full article
(This article belongs to the Section In Silico Approaches in Drug Discovery)
►▼
Show Figures
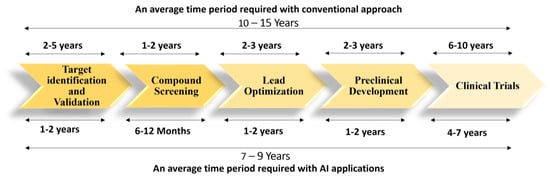
Figure 1
Open AccessReview
Beta-Thalassemia: A Pharmacological Drug-Based Treatment
by
Shrabonti Biswas, Shanita Zaman Smrity, Md. Shimul Bhuia, Fatema Akter Sonia, Mst. Asma Aktar, Raihan Chowdhury, Tawhida Islam, Muhammad Torequl Islam, Gabriel Gonçalves Alencar, Cicera Laura Roque Paulo, Ana Pavla Almeida Diniz Gurgel and Henrique Douglas Melo Coutinho
Drugs Drug Candidates 2024, 3(1), 126-147; https://doi.org/10.3390/ddc3010008 - 08 Feb 2024
Abstract
This review was performed to determine the potential of drugs that can remove or decrease the requirements for blood transfusion among beta (β)-thalassemia patients. A comprehensive literature search was conducted to identify clinical trials and studies using PubMed Central, Google Scholar, PubMed, and
[...] Read more.
This review was performed to determine the potential of drugs that can remove or decrease the requirements for blood transfusion among beta (β)-thalassemia patients. A comprehensive literature search was conducted to identify clinical trials and studies using PubMed Central, Google Scholar, PubMed, and ScienceDirect archived articles published from 1996 to November 2023. According to this review, clinical trials for a number of drugs, including luspatercept, sotatercept, mitapivat, etavopivat, hydroxyurea, rapamycin, decitabine, thalidomide, and quercetin, have been performed as part of efforts to improve the cure strategy for β-thalassemia. Of these drugs, luspatercept and sotatercept have exhibited particularly promising results and have been granted US Food and Drug Administration (FDA) approval for use in β-thalassemia patients. The mode of action for the drugs luspatercept and sotatercept involves the stimulation of hemoglobin (Hb) production or enhancement of its functionality, thereby decreasing reliance on blood transfusions and enhancing the overall quality of life. In this way, drugs like luspatercept and sotatercept present an opportunity to notably decrease the necessity for blood transfusions in β-thalassemia patients, improving their standard of living and overall prognosis. However, more research is needed to evaluate the effectiveness and safety of these drugs in the long run.
Full article
(This article belongs to the Section Clinical Research)
►▼
Show Figures

Figure 1
Open AccessReview
Nose-to Brain Delivery of Resveratrol, a Non-Invasive Method for the Treatment of Cerebral Ischemia
by
Iván Alquisiras-Burgos, Irma Gabriela González-Herrera, Sergio Alcalá-Alcalá and Penélope Aguilera
Drugs Drug Candidates 2024, 3(1), 102-125; https://doi.org/10.3390/ddc3010007 - 26 Jan 2024
Abstract
Cerebral ischemia represents a particular condition among neurological diseases due to its high frequency, high associated mortality, and the permanent disability in patients that survive it. Numerous studies in animal models have demonstrated the protective properties of resveratrol against cerebral ischemia. Resveratrol is
[...] Read more.
Cerebral ischemia represents a particular condition among neurological diseases due to its high frequency, high associated mortality, and the permanent disability in patients that survive it. Numerous studies in animal models have demonstrated the protective properties of resveratrol against cerebral ischemia. Resveratrol is a soluble molecule in polar solvents with high membrane permeability; however, it is rapidly metabolized at the liver and is also a substrate of the ATP binding cassette transporters located at the blood–brain barrier. These circumstances reduced bioavailability of resveratrol to the brain. In this review, we examined nasal resveratrol’s formulations including nanocarriers such as nanostructured lipid carriers, nanoemulsions, nanoparticles, bilosomes, cubosomal, and transferosomes that are directly transported to the brain. An intranasal administration route evades resveratrol transformation due to liver metabolism. Components of nanoformulations increased resveratrol absorption to the brain by enhancing permeation through specific approaches and also maintaining stability during storage. Both characteristics improved the delivery of resveratrol with conserved antioxidant capacity and protective properties for neurological models. Although demonstration that the nanoformulations prevents resveratrol’s blood–brain barrier retention is missing, properties of resveratrol’s nanoformulation encourage testing in clinical trials; however, regulatory approval for a novel nanocarrier in nasal drug delivery is complicated and needs approval.
Full article
(This article belongs to the Section Marketed Drugs)
►▼
Show Figures
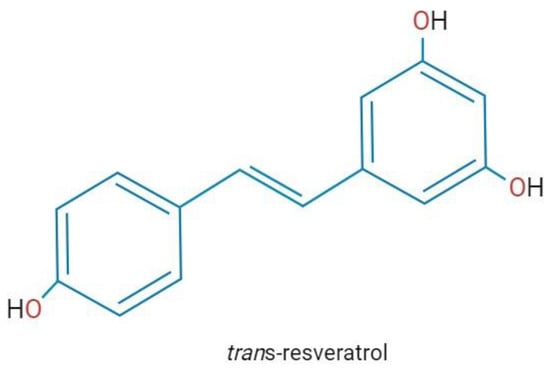
Figure 1
Open AccessReview
Opportunities and Difficulties in the Repurposing of HDAC Inhibitors as Antiparasitic Agents
by
Tapas Kumar Mohapatra, Reena Rani Nayak, Ankit Ganeshpurkar, Prashant Tiwari and Dileep Kumar
Drugs Drug Candidates 2024, 3(1), 70-101; https://doi.org/10.3390/ddc3010006 - 18 Jan 2024
Abstract
Ongoing therapy for human parasite infections has a few known drugs but with serious side effects and the problem of drug resistance, impelling us to discover novel drug candidates with newer mechanisms of action. Universally, this has boosted the research in the design
[...] Read more.
Ongoing therapy for human parasite infections has a few known drugs but with serious side effects and the problem of drug resistance, impelling us to discover novel drug candidates with newer mechanisms of action. Universally, this has boosted the research in the design and development of novel medicinal agents as antiparasitic drugs with a novel mode of action. Histone deacetylase inhibitors (HDACis) are used in a vast variety of diseases due to their anti-inflammatory properties. Drug repurposing strategies have already approved HDACis as cancer therapeutics and are now under investigation for many parasitic infections. Along with the expression of the gene, histone deacetylase (HDAC) enzymes also act as a slice of great multi-subunit complexes, targeting many non-histones, changing systemic and cellular levels signaling, and producing different cell-based specified effects. Zinc (Zn2+)- and nicotinamide adenine dinucleotide (NAD+)-dependent HDACs of parasites play pivotal roles in the alteration of gene expression of parasites. Some of them are already known to be responsible for the survival of several parasites under odd circumstances; thus, targeting them for therapeutic interventions will be novel for potential antiparasitic targets. This point of view outlines the knowledge of both class-I and class-II HDACis and sirtuin inhibitors that emerged to be the key players in the treatment of human parasitic disorders like Leishmaniasis, Schistosomiasis, Malaria, Trypanosomiasis, and Toxoplasmosis. This review also focuses on repurposing opportunities and challenges in HDAC inhibitors that are preceded by their clinical development as potent new antiparasitic drugs.
Full article
(This article belongs to the Collection Anti-Parasite Drug Discovery)
►▼
Show Figures
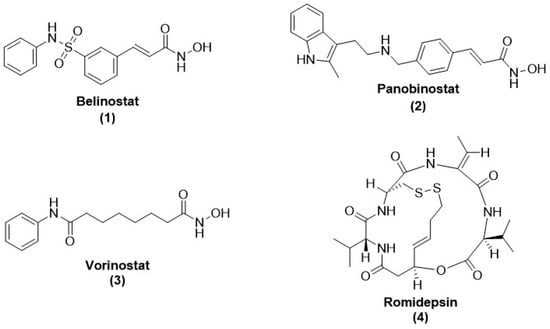
Figure 1
Open AccessReview
In Silico Therapeutic Study: The Next Frontier in the Fight against SARS-CoV-2 and Its Variants
by
Calvin R. Wei, Zarrin Basharat and Godwin C. Lang’at
Drugs Drug Candidates 2024, 3(1), 54-69; https://doi.org/10.3390/ddc3010005 - 05 Jan 2024
Abstract
COVID-19 has claimed around 7 million lives (from December 2019–November 2023) worldwide and continues to impact global health. SARS-CoV-2, the virus causing COVID-19 disease, is characterized by a high rate of mutations, which contributes to its rapid spread, virulence, and vaccine escape. While
[...] Read more.
COVID-19 has claimed around 7 million lives (from December 2019–November 2023) worldwide and continues to impact global health. SARS-CoV-2, the virus causing COVID-19 disease, is characterized by a high rate of mutations, which contributes to its rapid spread, virulence, and vaccine escape. While several vaccines have been produced to minimize the severity of the coronavirus, and diverse treatment regimens have been approved by the US FDA under Emergency Use Authorization (EUA), SARS-CoV-2 viral mutations continue to derail the efforts of scientists as the emerging variants evade the recommended therapies. Nonetheless, diverse computational models exist that offer an opportunity for the swift development of new drugs or the repurposing of old drugs. In this review, we focus on the use of various virtual screening techniques like homology modeling, molecular docking, molecular dynamics simulations, QSAR, pharmacophore modeling, etc., in repurposing SARS-CoV-2 therapeutics against major variants of SARS-CoV-2 (Alpha, Beta, Gamma, Delta, and Omicron). The results have been promising from the computer-aided drug design (CADD) studies in suggesting potential compounds for the treatment of COVID-19 variants. Hence, in silico therapeutic studies represent a transformative approach that holds great promise in advancing our fight against the ever-evolving landscape of SARS-CoV-2 and its variants.
Full article
(This article belongs to the Special Issue Fighting SARS-CoV-2 and Related Viruses)
►▼
Show Figures
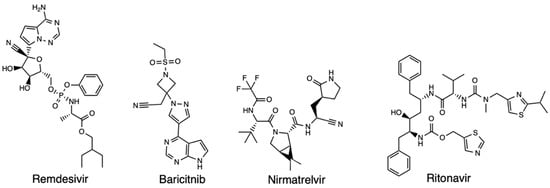
Figure 1
Open AccessEditorial
Drugs and Drug Candidates: Year in Review
by
Jean Jacques Vanden Eynde
Drugs Drug Candidates 2024, 3(1), 52-53; https://doi.org/10.3390/ddc3010004 - 05 Jan 2024
Abstract
First of all, I would like to wish you, readers, authors, referees, Editorial Board Members, and colleagues of the editorial offices, a Happy New Year filled with plenty of familial joys and professional achievements [...]
Full article
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics

Conferences
Special Issues
Special Issue in
DDC
Fighting SARS-CoV-2 and Related Viruses
Guest Editors: Jean Jacques Vanden Eynde, Annie MayenceDeadline: 30 June 2024
Special Issue in
DDC
Drugs of the Kallikrein-Kinin System
Guest Editor: François MarceauDeadline: 31 December 2024
Topical Collections
Topical Collection in
DDC
Bioinorganic Chemistry in Drug Discovery
Collection Editors: Tanja Soldatović, Snežana Jovanović-Stević
Topical Collection in
DDC
Chirality in Drugs and Drug Candidates
Collection Editors: Carla Fernandes, Maria Emília De Sousa