Formation and Oriented Aggregation of Tabular Hexagonal Silver Particles
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Chemical Synthesis
Tabular Hexagonal
2.3. Characterization
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Klabunde, K.J. Nanoscale Materials in Chemistry; Wiley: New York, NY, USA, 2001; Volume 2, ISBN 9780470222706. [Google Scholar]
- Goia, D.V.; Matijević, E. Preparation of monodispersed metal particles. New J. Chem. 1998, 22, 1203–1215. [Google Scholar] [CrossRef]
- Sondi, I.; Goia, D.V.; Matijević, E. Preparation of highly concentrated stable dispersions of uniform silver nanoparticles. J. Colloid Interface Sci. 2003, 260, 75–81. [Google Scholar] [CrossRef]
- Kamat, P.V.; Flumiani, M.; Dawson, A. Metal-metal and metal-semiconductor composite nanoclusters. Colloids Surf. A Physicochem. Eng. Asp. 2002, 202, 269–279. [Google Scholar] [CrossRef]
- Liz-Marzán, L.M.; Giersig, M.; Mulvaney, P. Synthesis of Nanosized Gold−Silica Core−Shell Particles. Langmuir 1996, 12, 4329–4335. [Google Scholar] [CrossRef]
- Lee, M.H.; Oh, S.G.; Suh, K.D.; Kim, D.G.; Sohn, D. Preparation of silver nanoparticles in hexagonal phase formed by nonionic Triton X-100 surfactant. Colloids Surfaces A Physicochem. Eng. Asp. 2002, 210, 49–60. [Google Scholar] [CrossRef]
- Velikov, K.; Zegers, G.; van Blaaderen, A. Synthesis and characterization of large colloidal silver particles. Langmuir 2003, 19, 1384–1389. [Google Scholar] [CrossRef]
- Sun, Y. Shape-Controlled Synthesis of Gold and Silver Nanoparticles. Science 2002, 298, 2176–2179. [Google Scholar] [CrossRef] [PubMed]
- Blatchford, C.G.; Campbell, J.R.; Creighton, J.A. Plasma resonance—Enhanced raman scattering by absorbates on gold colloids: The effects of aggregation. Surf. Sci. 1982, 120, 435–455. [Google Scholar] [CrossRef]
- De, S.; Pal, A.; Jana, N.R.; Pal, T. Anion effect in linear silver nanoparticle aggregation as evidenced by efficient fluorescence quenching and SERS enhancement. J. Photochem. Photobiol. A Chem. 2000, 131, 111–123. [Google Scholar] [CrossRef]
- Linnert, T.; Mulvaney, P.; Henglein, A. Surface chemistry of colloidal silver: Surface plasmon damping by chemisorbed iodide, hydrosulfide (SH-), and phenylthiolate. J. Phys. Chem. 1993, 97, 679–682. [Google Scholar] [CrossRef]
- Cui, Y.; Wei, Q.; Park, H.; Lieber, C.M. Nanowire Nanosensors for Highly Sensitive and Selective Detection of Biological and Chemical Species. Science 2001, 293, 1289–1292. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Lieber, C.M. Functional Nanosccale Electronic Devices Assemble Using Silicon Nanowire Building Blocks. Science 2001, 291, 851–853. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Xia, Y. Large-scale synthesis of uniform silver nanowires through a soft, self-seeding, polyol process. Adv. Mater. 2002, 14, 833–837. [Google Scholar] [CrossRef]
- Zhang, D.; Qi, L.; Yang, J.; Ma, J.; Cheng, H.; Huang, L. Wet Chemical Synthesis of Silver Nanowire Thin Films at Ambient Temperature. Chem. Mater. 2004, 16, 872–876. [Google Scholar] [CrossRef]
- Zeng, J.; Zheng, Y.; Rycenga, M.; Tao, J.; Li, Z.Y.; Zhang, Q.; Zhu, Y.; Xia, Y. Controlling the shapes of silver nanocrystals with different capping agents. J. Am. Chem. Soc. 2010, 132, 8552–8553. [Google Scholar] [CrossRef] [PubMed]
- Ozaki, M.; Kratohvil, S.; Matijević, E. Formation of monodispersed spindle-type hematite particles. J. Colloid Interface Sci. 1984, 102, 146–151. [Google Scholar] [CrossRef]
- Morales, M.P.; González-Carreño, T.; Serna, C.J. The formation of α-Fe2O3 monodispersed particles in solution. J. Mater. Res. 1992, 7, 2538–2545. [Google Scholar] [CrossRef]
- Johnson, C.J.; Dujardin, E.; Davis, S.A.; Murphy, C.J.; Mann, S. Growth and form of gold nanorods prepared by seed-mediated, surfactant-directed synthesis. J. Mater. Chem. 2002, 12, 1765–1770. [Google Scholar] [CrossRef]
- Suber, L.; Sondi, I.; Matijević, E.; Goia, D.V.D. Preparation and the mechanisms of formation of silver particles of different morphologies in homogeneous solutions. J. Colloid Interface Sci. 2005, 288, 489–495. [Google Scholar] [CrossRef] [PubMed]
- Campi, G.; Mari, A.; Amenitsch, H.; Pifferi, A.; Cannas, C.; Suber, L. Monitoring early stages of silver particle formation in a polymer solution by in situ and time resolved small angle X-ray scattering. Nanoscale 2010, 2, 2447–2455. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.L. Book Reprint Characterization of Nanophase Materials. Part. Part. Syst. Charact. 2001, 18, 142–165. [Google Scholar] [CrossRef]
- Campi, G.; Mari, A.; Pifferi, A.; Amenitsch, H.; Fratini, M.; Suber, L. Control of silver-polymer aggregation mechanism by primary particle spatial correlations in dynamic fractal-like geometry. Nanoscale 2011, 3, 3774–3779. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Zhang, Y.; Zhang, S.; Qiu, W.; Gao, Y. Silver enhancement of gold nanoparticles for biosensing: From qualitative to quantitative. Appl. Spectrosc. Rev. 2014, 49, 121–138. [Google Scholar] [CrossRef]
- Suber, L.; Campi, G.; Pifferi, A.; Andreozzi, P.; La Mesa, C.L.; Amenitsch, H.; Cocco, R.; Plunkett, W.R. Polymer-assisted synthesis of two-dimensional silver meso-structures. J. Phys. Chem. C 2009, 113, 11198–11203. [Google Scholar] [CrossRef]
- Suber, L.; Plunkett, W.R. Formation mechanism of silver nanoparticle 1D microstructures and their hierarchical assembly into 3D superstructures. Nanoscale 2010, 2, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Linnert, T.; Mulvaney, P.; Henglein, A. Photochemistry of Colloidal Silver Particles—The Effects of N2O and Adsorbed Cn-. Berichte Bunsenges. Phys. Chem. 1991, 95, 838–841. [Google Scholar] [CrossRef]
- Fukuyo, T.; Imai, H. Morphological evolution of silver crystals produced by reduction with ascorbic acid. J. Cryst. Growth 2002, 241, 193–199. [Google Scholar] [CrossRef]
- Hvoslef, J. The crystal structure of l-ascorbic acid, ‘vitamin C’. II. The neutron diffraction analysis. Acta Crystallogr. Sect. B Struct. Crystallogr. Cryst. Chem. 1968, 24, 1431–1440. [Google Scholar] [CrossRef]


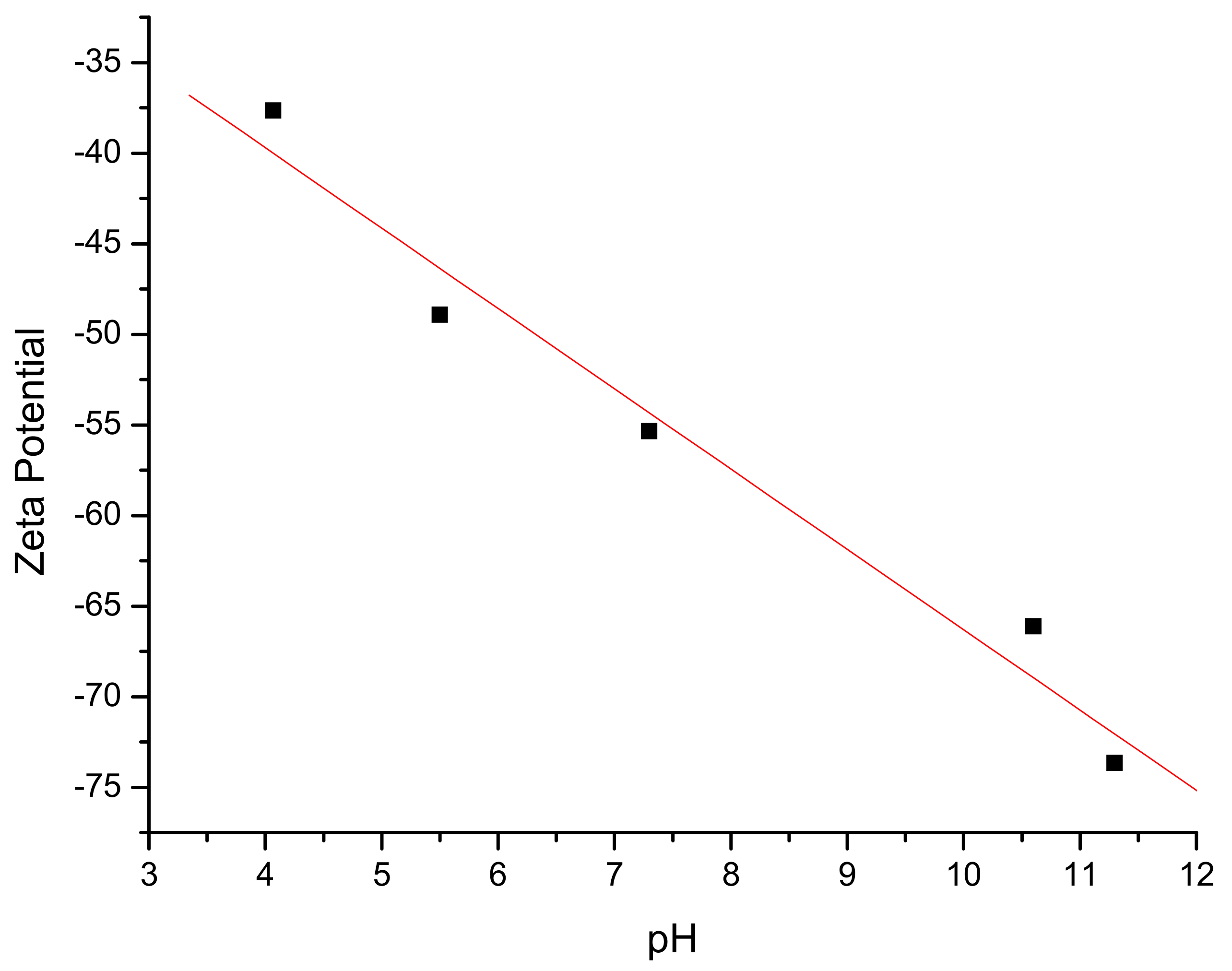

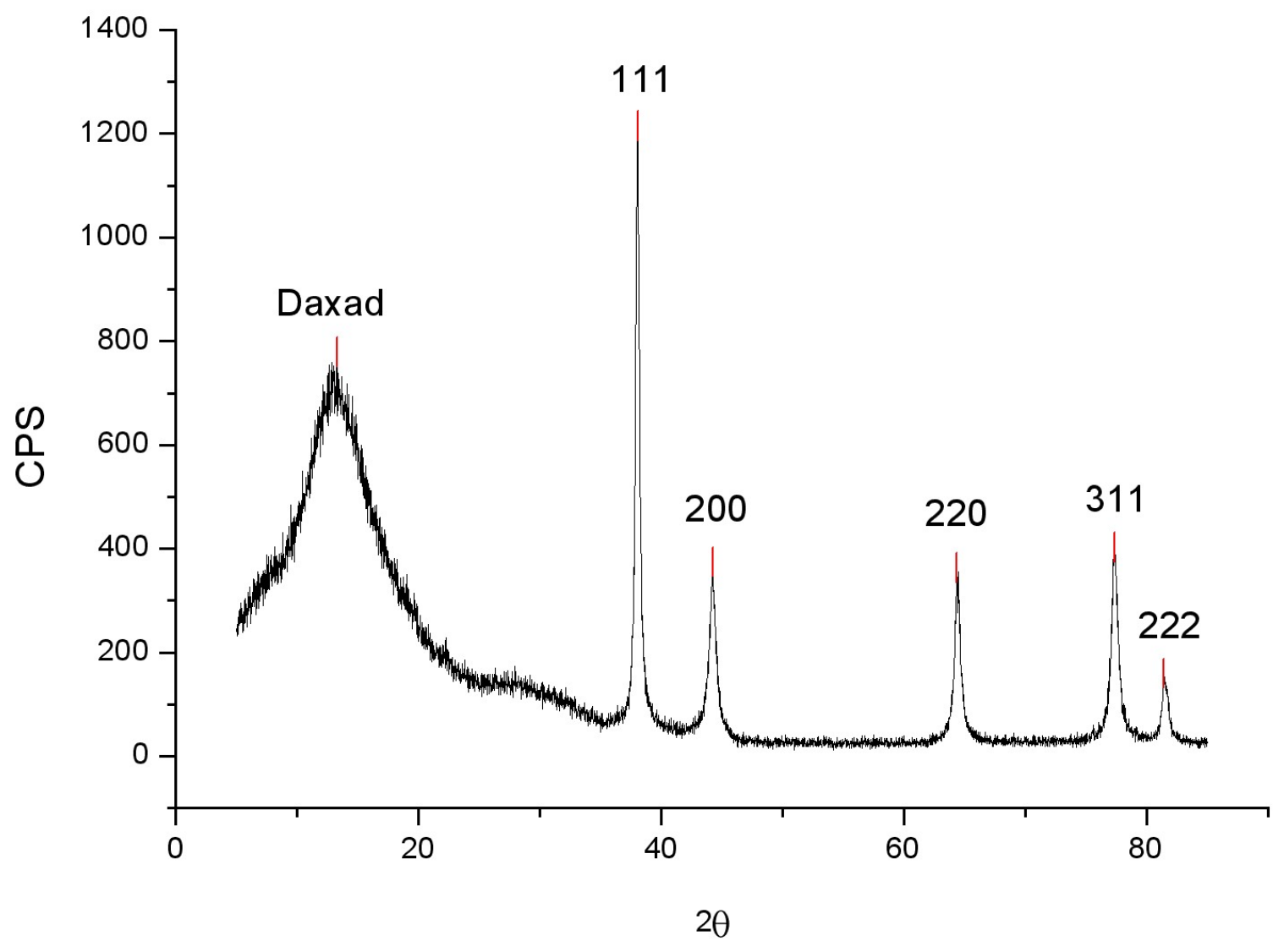


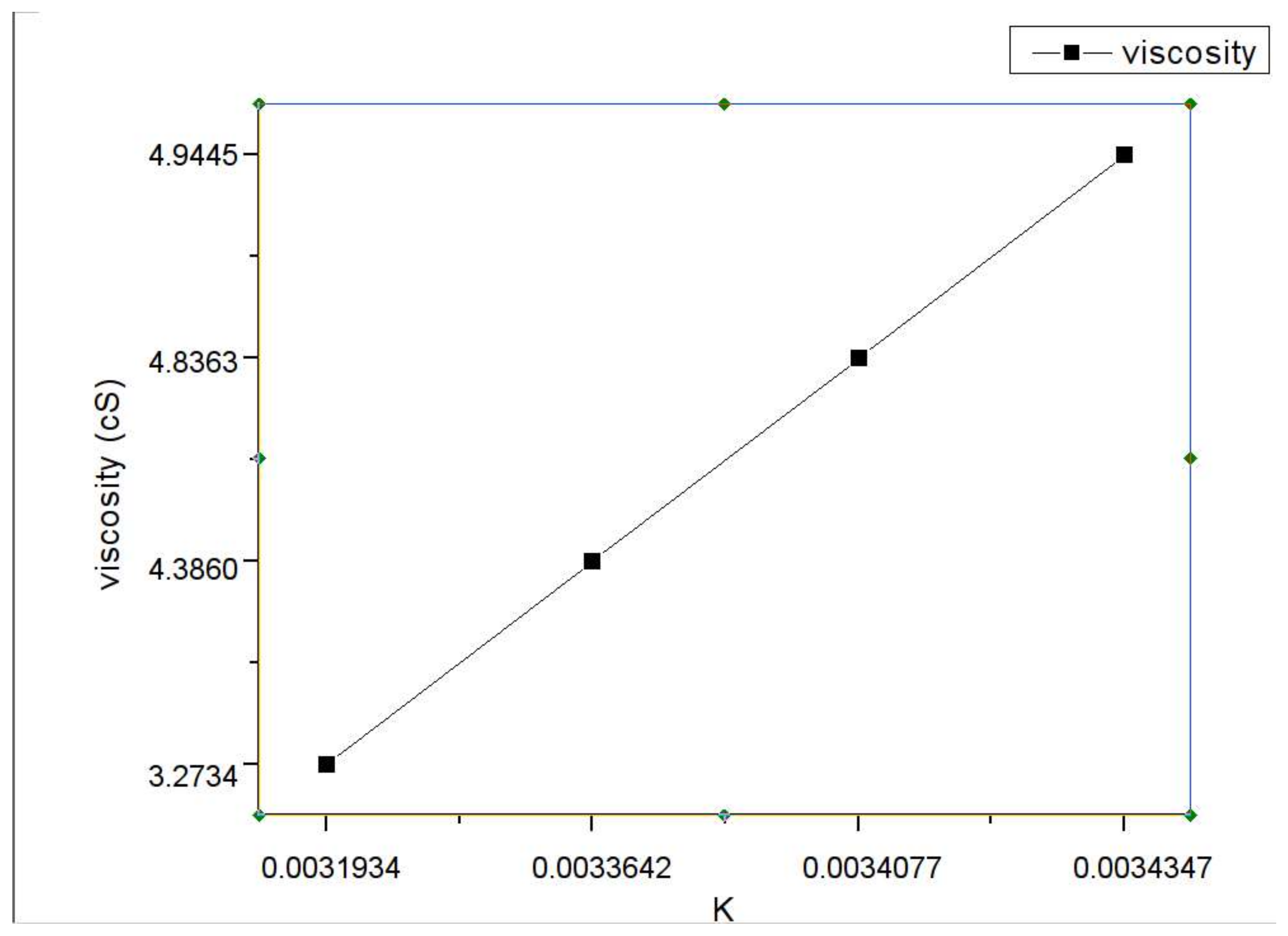

| Sample | 1 | 2 | 3 | 4 |
|---|---|---|---|---|
| T (°C) | 40 | 10 | 30 | 40 |
| t (min) | 60 | 60 | 60 | 10 |
| Ag Initial Conc. mol dm−3 | 0.2 | 0.2 | 0.2 | 0.02 |
| HNO3 mol dm−3 | 1.6 | 1.5 | 1.6 | 1.4 |
| Dispersant Initial Conc. g dm−3 | 4.9 | 4.9 | 5.4 | 5.2 |
| Dispersant/Metal Ratio | 1/4.4 | 1/4.4 | 1/3.6 | 2.4/1 |
| Asc Acid Fin. Conc. mol dm−3 | 0.18 | 0.19 | 0.19 | 0.16 |
| Vol Prior Reduction cm3 | 278 | 270 | 165 | 170 |
| Vol after Reduction cm3 | 308 | 300 | 183 | 185 |
| SEM TEM | Hexag. part 1 | Flat rods | flakesflakFl Flakes | Polydispersed particles 2 |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suber, L. Formation and Oriented Aggregation of Tabular Hexagonal Silver Particles. Condens. Matter 2018, 3, 13. https://doi.org/10.3390/condmat3020013
Suber L. Formation and Oriented Aggregation of Tabular Hexagonal Silver Particles. Condensed Matter. 2018; 3(2):13. https://doi.org/10.3390/condmat3020013
Chicago/Turabian StyleSuber, Lorenza. 2018. "Formation and Oriented Aggregation of Tabular Hexagonal Silver Particles" Condensed Matter 3, no. 2: 13. https://doi.org/10.3390/condmat3020013





