Azospirillum brasilense Enhances Recycling of Fish Effluent to Support Growth of Tomato Seedlings
Abstract
:1. Introduction
2. Materials and Methods
2.1. Seed and Inoculum Preparation, and Inoculation
2.2. Growing Condition and Treatments
2.3. Measurements and Analyses
2.4. IAA Quantification
2.5. Peroxidase Activity
3. Results and Discussion
3.1. Effects on Seedling Growth
| A. brasilense Strains | Leaf Number | Plant Height (cm) | Stem Diameter (mm) | Leaf Area (cm2/Plant) | Root Length (cm) | Fresh Weight (g/Plant) | Dry Weight (mg/Plant) |
|---|---|---|---|---|---|---|---|
| Control | 3.07 c | 11.43 b | 1.78 b | 24.12 c | 60 b | 0.50 c | 36 c |
| Sp7 | 3.34 a | 12.75 a | 2.04 a | 28.05 b | 100 a | 0.76 a | 64 a |
| Sp7-S | 3.15 b,c | 12.07 b | 1.80 b | 29.88 a,b | 97 a | 0.59 b | 48 b |
| Sp245 | 3.29 a,b | 11.57 b | 1.86 b | 30.31 a | 104 a | 0.65 b | 47 b |
| LSD (5%) | 0.14 | 0.65 | 0.13 | 1.99 | 12 | 0.09 | 8 |

3.2. Effects on Metabolic Activities
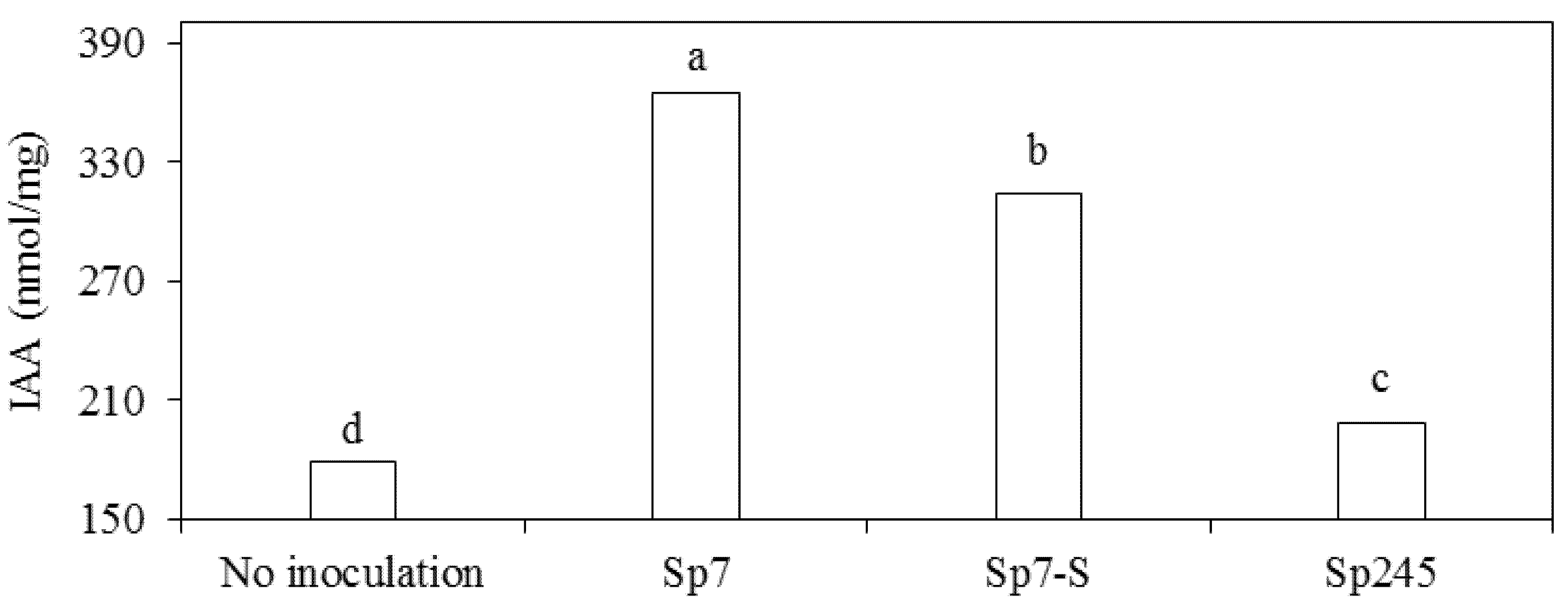
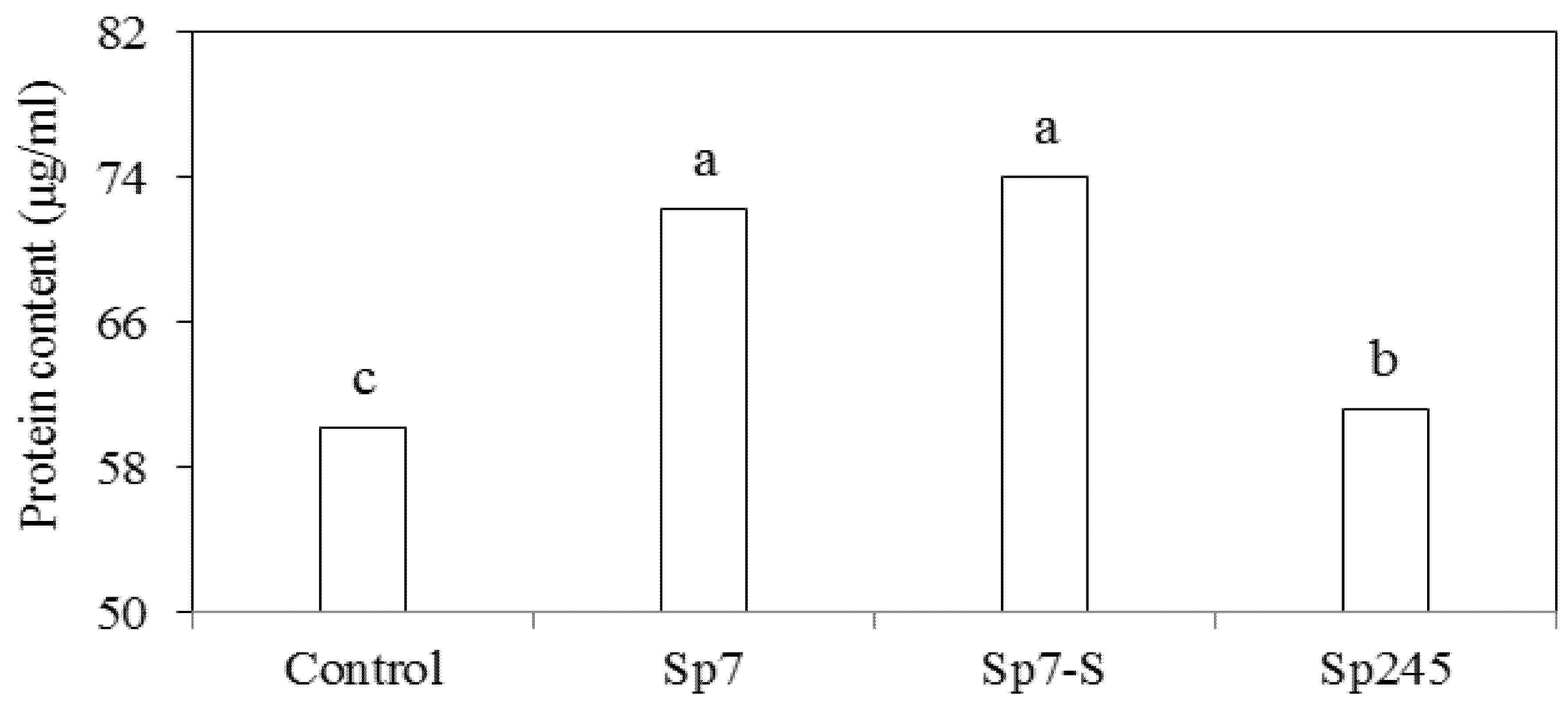
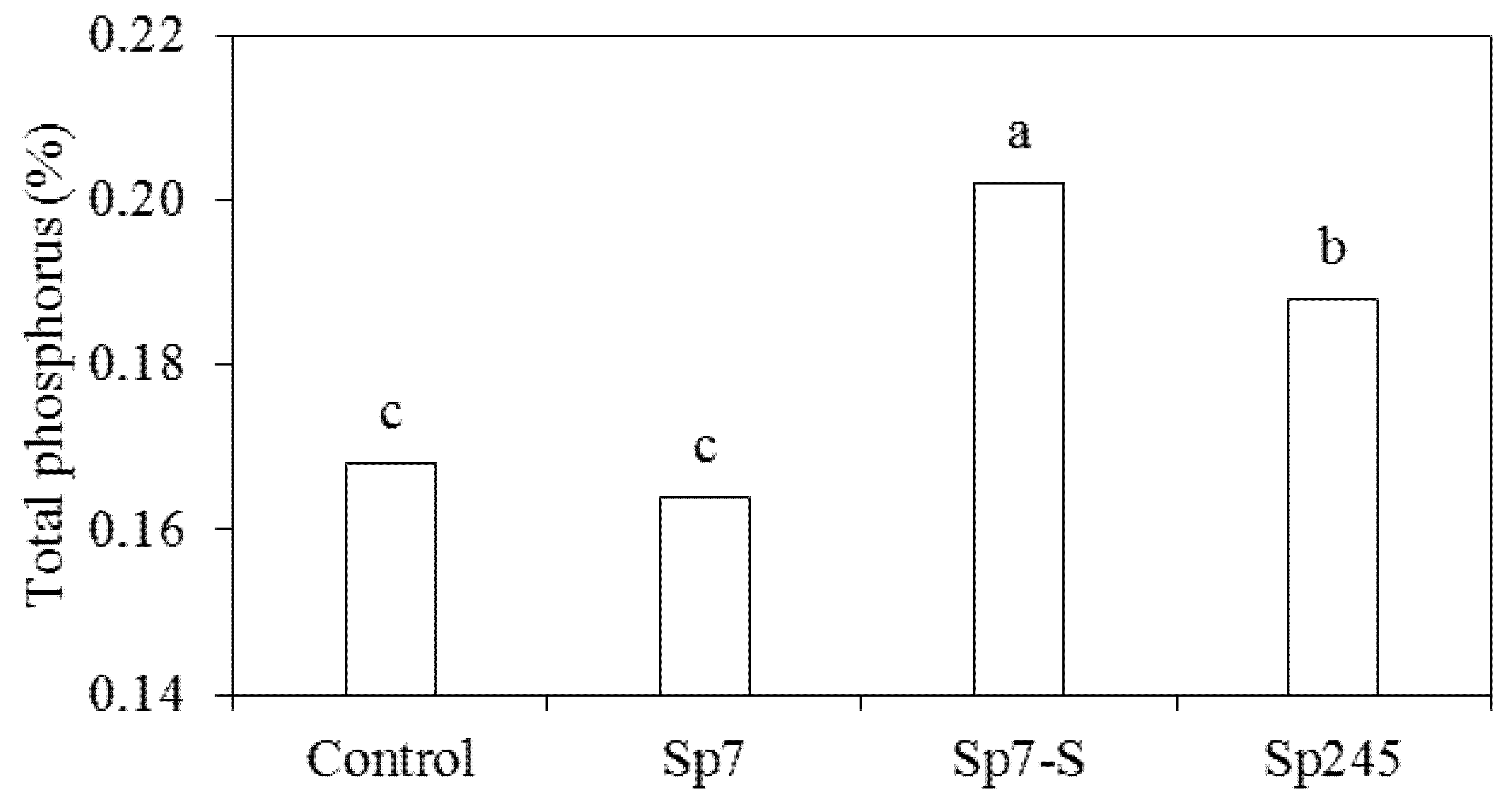
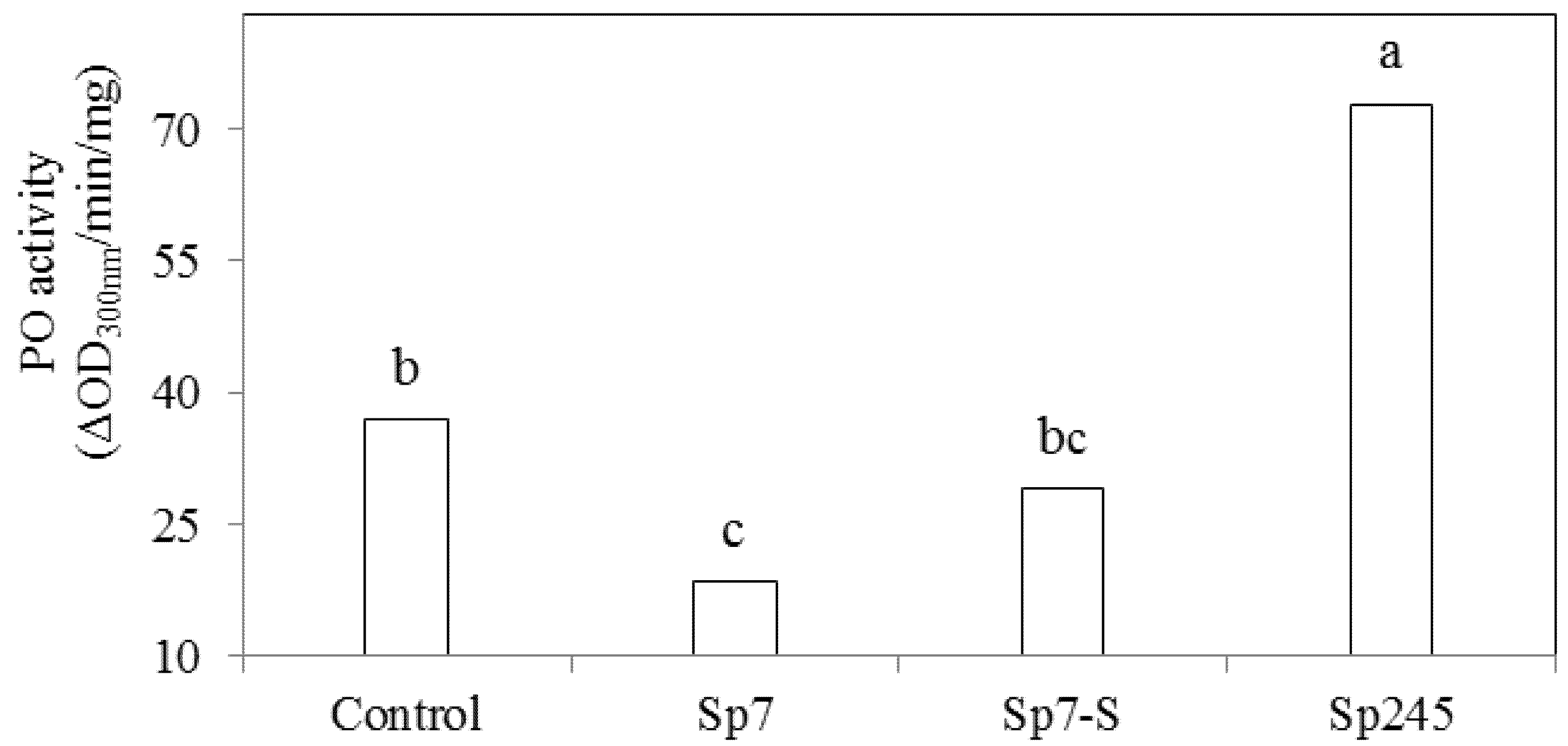
4. Conclusions
Acknowledgement
Author Contributions
Conflict of interest
References
- Rakocy, J.E.; Bailey, D.S.; Shultz, R.C.; Danaher, J.J. Preliminary evaluation of organic waste from two aquaculture systems as a source of inorganic nutrients for hydroponics. Acta Hort. 2007, 742, 201–208. [Google Scholar]
- Nichols, M.; Savidov, N. Aquaponics: A nutrient and water efficient production system. Acta Hort. 2011, 947, 129–132. [Google Scholar]
- Graber, A.; Junge, R. Aquaponic systems: Nutrient recycling from fish wastewater by vegetable production. Desalination 2009, 246, 147–156. [Google Scholar] [CrossRef]
- Tyson, R.V.; Treadwell, D.D.; Simonne, E.H. Opportunities and challenges to sustainability in aquaponic systems. Horttechnology 2011, 21, 6–13. [Google Scholar]
- Dubrovsky, J.G.; Puente, M.E.; Bashan, Y. Arabidopsis thaliana as a model system for the study of the effect of inoculation by azospirillum brasilense sp245 on root hair growth. Soil Biol. Biochem. 1994, 26, 1657–1664. [Google Scholar] [CrossRef]
- Saharan, B.; Nehra, V. Plant growth promoting rhizobacteria: A critical review. Life Sci. 2011, 21, 1–30. [Google Scholar]
- Okon, Y.; Labandera-Gonzalez, C.A. Agronomic applications of Azospirillum: An evaluation of 20 years worldwide field inoculation. Soil Biol. Biochem. 1994, 26, 1591–1601. [Google Scholar] [CrossRef]
- Bashan, Y.; Holguin, G. Azospirillum—Plant relationships: Environmental and physiological advances (1990–1996). Can. J. Microbiol. 1997, 43, 103–121. [Google Scholar] [CrossRef]
- Okon, Y. Azospirillum as a potential inoculant for agriculture. Trend. Biotechnol. 1985, 3, 223–228. [Google Scholar] [CrossRef]
- Bashan, Y.; de-Bashan, L.E. How the plant growth-promoting bacterium Azospirillum promotes plant growth: A critical assessment. Adv. Agro. 2010, 108, 77–136. [Google Scholar]
- Gafny, R.; Okon, Y.; Kapulnik, Y.; Fischer, M. Adsorption of Azospirillum brasilense to corn roots. Soil Biol. Biochem. 1986, 18, 69–75. [Google Scholar] [CrossRef]
- Jain, D.K.; Patriquin, D.G. Root hair deformation, bacterial attachment, and plant growth in wheat-Azospirillum associations. Appl. Environ. Microbiol. 1984, 48, 1208–1213. [Google Scholar] [PubMed]
- Arsène, F.; Katupitiya, S.; Kennedy, I.R.; Elmerich, C. Use of LacZ-fusions to study the expression of nif genes of Azospirillum brasilense in association with plants. MPMI-Mol. Plant Microb. Interact. 1994, 7, 748–757. [Google Scholar] [CrossRef]
- Kennedy, I.R.; Tchan, Y.-T. Biological nitrogen fixation in non-leguminous field crops: Recent advances. In Biological Nitrogen Fixation for Sustainable Agriculture; Springer: Amsterdam, the Netherlands, 1992; pp. 93–118. [Google Scholar]
- Baldani, V.L.D.; Alvarez, M.A.B.; Baldani, J.; Döbereiner, J. Establishment of inoculated Azospirillum spp. in the rhizosphere and in roots of field grown wheat and sorghum. Plant Soil 1986, 90, 35–46. [Google Scholar] [CrossRef]
- Schloter, M.; Hartmann, A. Endophytic and surface colonization of wheat roots (Triticum aestivum) by different Azospirillum brasilense strains studied with strain-specific monoclonal antibodies. Symbiosis 1998, 25, 159–179. [Google Scholar]
- Vessey, J.K. Plant growth promoting rhizobacteria as biofertilizers. Plant Soil 2003, 255, 571–586. [Google Scholar] [CrossRef]
- Bashan, Y.; Holguin, G.; Lifshitz, R. Isolation and characterization of plant growth-promoting rhizobacteria. In Methods in Plant Molecular Biology and Biotechnology; Glick, B.R., Thompson, J.E., Eds.; CRC Press: Boca Raton, FL, USA, 1993; pp. 331–345. [Google Scholar]
- Patten, C.L.; Glick, B.R. Role of Pseudomonas putida indoleacetic acid in development of the host plant root system. Appl. Environ. Microbiol. 2002, 68, 3795–3801. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.B., Jr. Laboratory Guide for Conducting Soil Tests and Plant Analysis; CRC Press: Boca Raton, FL, USA, 2001. [Google Scholar]
- Gamalero, E.; Berta, G.; Massa, N.; Glick, B.R.; Lingua, G. Synergistic interactions between the ACC deaminase-producing bacterium Pseudomonas putida UW4 and the AM fungus Gigaspora rosea positively affect cucumber plant growth. FEMS Microbiol. Ecol. 2008, 64, 459–467. [Google Scholar] [CrossRef] [PubMed]
- Ribaudo, C.; Krumpholz, E.; Cassán, F.; Bottini, R.; Cantore, M.; Curá, J. Azospirillum sp. promotes root hair development in tomato plants through a mechanism that involves ethylene. J. Plant Growth Regulat. 2006, 25, 175–185. [Google Scholar] [CrossRef]
- Ben-Shalom, N.; Ardi, R.; Pinto, R.; Aki, C.; Fallik, E. Controlling gray mould caused by Botrytis cinerea in cucumber plants by means of chitosan. Crop. Protect. 2003, 22, 285–290. [Google Scholar] [CrossRef]
- Shelud’Ko, A.V.; Shirokov, A.A.; Sokolova, M.K.; Sokolov, O.I.; Petrova, L.P.; Matora, L.Y.; Katsy, E.I. Wheat root colonization by Azospirillum brasilense strains with different motility. Microbiology 2010, 79, 688–695. [Google Scholar] [CrossRef]
- Dobbelaere, S.; Croonenborghs, A.; Thys, A.; Ptacek, D.; Vanderleyden, J.; Dutto, P.; Labandera-Gonzalez, C.; Caballero-Mellado, J.; Aguirre, J.F.; Kapulnik, Y. Responses of agronomically important crops to inoculation with Azospirillum Funct. Plant. Biol. 2001, 28, 871–879. [Google Scholar]
- Jones, J.B. Hydroponics: A Practical Guide for the Soilless Grower, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2005; Volume 1. [Google Scholar]
- Olsen, C. The significance of concentration for the rate of ion absorption by higher plants in water culture. Physiol. Plant. 1950, 3, 152–164. [Google Scholar] [CrossRef]
- Bashan, Y.; Singh, M.; Levanony, H. Contribution of Azospirillum brasilense cd to growth of tomato seedlings is not through nitrogen fixation. J. Bot. 1989, 67, 2429–2434. [Google Scholar]
- Gholami, A.; Shahsavani, S.; Nezarat, S. The effect of plant growth promoting rhizobacteria (PGPR) on germination, seedling growth and yield of maize. World Acad. Sci. Engineer. Technol. 2009, 37, 2070–3740. [Google Scholar]
- Dobbelaere, S.; Croonenborghs, A.; Thys, A.; Ptacek, D.; Okon, Y.; Vanderleyden, J. Effect of inoculation with wild type Azospirillum brasilense and A. Irakense strains on development and nitrogen uptake of spring wheat and grain maize. Biol. Fertility Soils 2002, 36, 284–297. [Google Scholar] [CrossRef]
- Hadas, R.; Okon, Y. Effect of Azospirillum brasilense inoculation on root morphology and respiration in tomato seedlings. Biol. Fert. Soil. 1987, 5, 241–247. [Google Scholar] [CrossRef]
- El-Katatny, M.H. Enzyme production and nitrogen fixation by free, immobilized and coimmobilized inoculants of Trichoderma harzianum and Azospirillum brasilense and their possible mole in growth promotion of tomato. Food Technol. Biotechnol. 2010, 48, 161–174. [Google Scholar]
- Vikram, A.; Hamzehzarghani, H.; Alagawadi, A.R.; Krishnaraj, P.U.; Chandrashekar, B.S. Production of plant growth promoting substances by phosphate solubilizing bacteria isolated from vertisols. J. Plant. Sci. 2007, 2, 326–333. [Google Scholar]
- Khalid, A.; Arshad, M.; Zahir, Z.A. Screening plant growth-promoting rhizobacteria for improving growth and yield of wheat. J. Appl. Microbiol. 2004, 96, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Miransari, M. Plant growth promoting rhizobacteria. J. Plant Nutr. 2014, 37, 2227–2235. [Google Scholar] [CrossRef]
- Saubidet, M.I.; Fatta, N.; Barneix, A.J. The effect of inoculation with Azospirillum brasilense on growth and nitrogen utilization by wheat plants. Plant Soil 2002, 245, 215–222. [Google Scholar] [CrossRef]
- Fages, J. Azospirillum inoculants and field experiments. In Azospirillum-Plant Associations; Okon, Y., Ed.; CRC Press: Boca Raton, FL, USA, 1994; pp. 87–109. [Google Scholar]
- Zargari, K.; Khorshidi, Y.R.; Ardakani, M.R. Growth stimulant bacteria and nitrogen fertilizer effects plant nutrient uptake in rice (Oryza sativa l). Int. J. Biosci. 2014, 4, 218–226. [Google Scholar]
- Savitsky, P.; Gazaryan, I.; Tishkov, V.; Lagrimini, L.; Ruzgas, T.; Gorton, L. Oxidation of indole-3-acetic acid by dioxygen catalysed by plant peroxidases: Specificity for the enzyme structure. Biochem. J. 1999, 340, 579–583. [Google Scholar] [CrossRef] [PubMed]
- Pereyra, C.M.; Ramella, N.A.; Pereyra, M.A.; Barassi, C.A.; Creus, C.M. Changes in cucumber hypocotyl cell wall dynamics caused by Azospirillum brasilense inoculation. Plant Physiol. Biochem. 2010, 48, 62–69. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mangmang, J.S.; Deaker, R.; Rogers, G. Azospirillum brasilense Enhances Recycling of Fish Effluent to Support Growth of Tomato Seedlings. Horticulturae 2015, 1, 14-26. https://doi.org/10.3390/horticulturae1010014
Mangmang JS, Deaker R, Rogers G. Azospirillum brasilense Enhances Recycling of Fish Effluent to Support Growth of Tomato Seedlings. Horticulturae. 2015; 1(1):14-26. https://doi.org/10.3390/horticulturae1010014
Chicago/Turabian StyleMangmang, Jonathan S., Rosalind Deaker, and Gordon Rogers. 2015. "Azospirillum brasilense Enhances Recycling of Fish Effluent to Support Growth of Tomato Seedlings" Horticulturae 1, no. 1: 14-26. https://doi.org/10.3390/horticulturae1010014





