Cd, Pb and Hg Biomonitoring in Fish of the Mediterranean Region and Risk Estimations on Fish Consumption
Abstract
:1. Introduction
| Metal | JECFA | EFSA | EC | ||
|---|---|---|---|---|---|
| TDI (µg/kg bw) | PTWI (μg/kg bw) | PTWI (μg/kg bw) | MRLs (mg/g ww) | MRLs (mg/g ww) | |
| Hg | NA * | NA * | 4 | 0.5 (1.0 **) | 0.5 (1.0 **) |
| MeHg | 0.23 | 1.6 | 1.6 | 1.3 | NA * |
| Cd | NA * | 7 (2003) | 2.5 | 0.05 (0.1–0.3 **) | 0.050–0.30 *** |
| Pb | NA * | 25 | 25 | 0.3 | 0.30 *** |
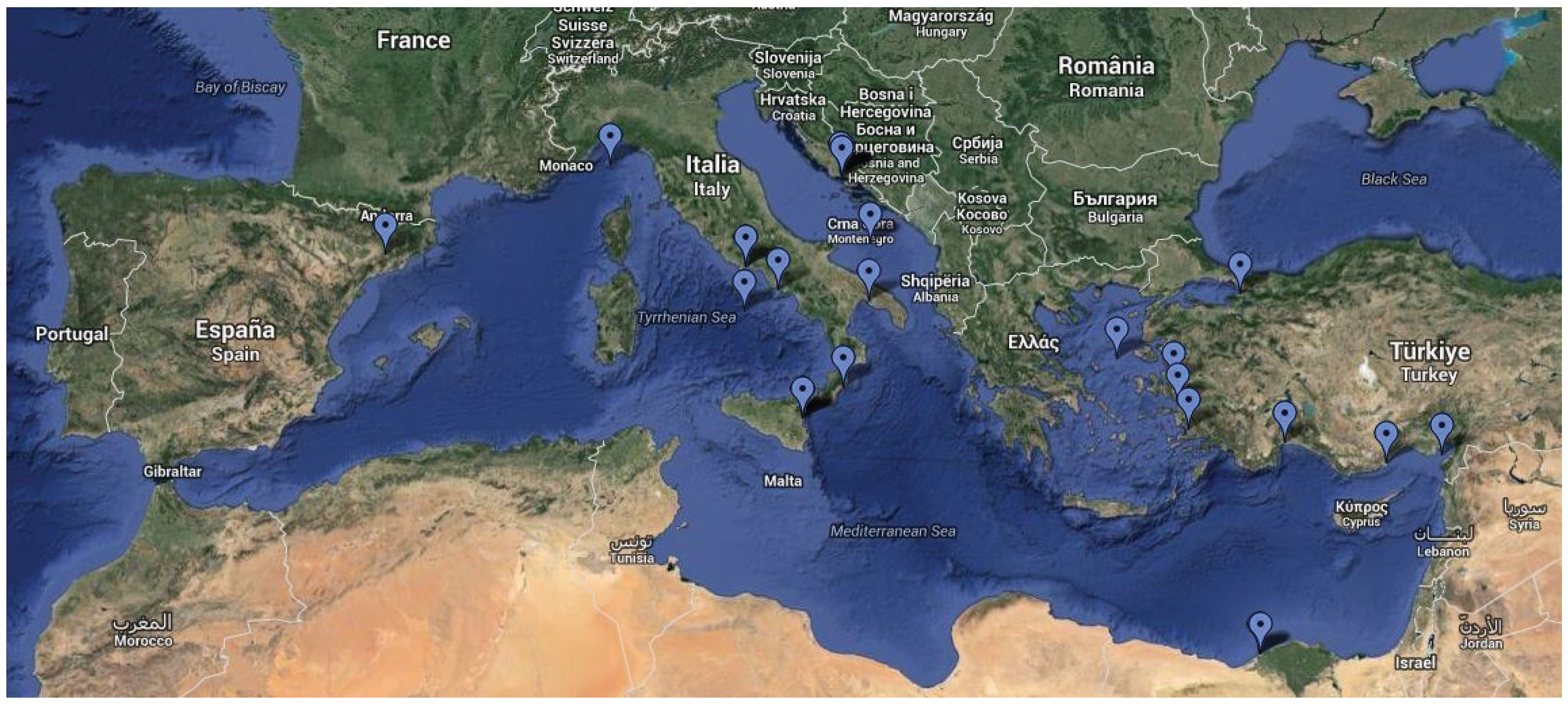
2. Study Selection and Methods
2.1. Criteria for the Selection of the Studies
- a)
- The literature collection regarding biomonitoring studies was based on the following criteria: At least one of the three metals (Cd, Pb, Hg) was studied,
- b)
- The samples collected for each study included sea fish samples and only data on sea fish samples were recorded and listed for the review, excluding data on other species i.e., fresh water fish, crustaceans etc.
- c)
- The sampling was conducted in the broader area of the Mediterranean Sea (Figure 1),
- d)
- The publication year of each study was between 2000 and 2012.
- (a)
- Sampling was not conducted according to seasons or for a period longer than 2 years,
- (b)
- Analysis of the fish samples was not done by age or sex groups.
- (a)
- Human intakes (EDI/EWI) of at least one of the metals (Cd, Pb and Hg) were estimated through the consumption of sea fish species and/or
- (b)
- Risk of human intake, expressed as a hazard index, was estimated by using human intake data (either obtained from Country’s Institutions or estimated experimentally, depending on the study) and metal levels determined in fish tissue, in the respective studies.
- (c)
- Studies performed on fish bought from supermarkets, were not considered.
2.2. Data Process on Metal Levels
2.3. Risk Assessment Methods
| Ref NO. | Muscle | Liver | Gonads | Other | Analytical Technique | Limit of Detection/(Min) (ppm) | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cd | Pb | Hg | Cd | Pb | Hg | ||||||||||||||
| [29] a | + | + | - | - | GF-AAS | GF-AAS | CV-AAS | 0.002 | 0.04 | 0.02 | |||||||||
| [30] a | - | + | - | + | GF-AAS | - | - | NR | - | - | |||||||||
| [31] b | - | + | + | + | GF-AAS | - | - | 1 | - | - | |||||||||
| [32] a | + | + | - | - | GF-AAS | GF-AAS | - | 0.0041 | 0.049 | - | |||||||||
| [33] a | + | + | + | + | ICP-OES | ICP-OES | - | 1 | 1 | - | |||||||||
| [5] a | + | - | - | - | AAS | AAS | - | NR | NR | - | |||||||||
| [34] a | + | + | - | - | ICP-OES | ICP-OES | - | 0.01 | 0.04 | - | |||||||||
| [17] b | + | - | - | - | GF-AAS | GF-AAS | - | (<0.01) | (<0.02) | - | |||||||||
| [35] a | + | - | - | - | GF-AAS | GF-AAS | - | 0.0003 | 0.005 | - | |||||||||
| [36] a | + | + | - | - | ICP-OES | ICP-OES | - | NR | NR | - | |||||||||
| [37] b | + | - | - | - | FAAS | FAAS | - | 0.08 | 0.19 | - | |||||||||
| [2] a | + | - | - | - | GF-AAS | GF-AAS | HG-AAS | NR | NR | NR | |||||||||
| [38] a | + | - | - | - | ICP-OES | ICP-OES | - | NR | NR | - | |||||||||
| [7] a | + | - | - | - | GF-AAS | GF-AAS | CV-AAS | 0.17 | 0.05 | ND | |||||||||
| [13] b | + | - | - | - | AAS | - | - | NR | - | - | |||||||||
| [39] a | + | + | - | - | ICP-OES | ICP-OES | - | 0.02 | 0.33 | - | |||||||||
| [40] b | + | - | - | - | ICP-OES | ICP-OES | - | 1 | 4.2 | - | |||||||||
| [41] b | + | + | - | + | FAAS | FAAS | - | 28 | 28 | - | |||||||||
| [14] a | + | - | + | + | - | ICP-OES | - | - | NR | - | |||||||||
| [42] a | + | - | - | - | GF-AAS | GF-AAS | CV-AAS | 0.4 | 5 | 0.4 | |||||||||
| [43] a | + | + | + | - | GF-AAS | GF-AAS | CV-AAS | 0.1 | 0.1 | 0.5 | |||||||||
| [44] a | + | + | - | - | GF-AAS | GF-AAS | CV-AAS | 0.05 | 0.03 | 0.03 | |||||||||
| [45] a | + | - | - | - | GF-AAS | GF-AAS | GF-AAS | NR | NR | NR | |||||||||
| [46] a | NS | NS | NS | NS | DPSAV | DPSAV | - | NR | NR | - | |||||||||
| [47] a | + | - | - | - | ET-AAS | ET-AAS | CV-AAS | 0.1 | 0.9 | 3.3 | |||||||||
| [48] b | + | - | - | - | - | - | AMA | NR | 0.324 | 0.2 | |||||||||
| [15] a | + | - | - | - | GF-AAS | GF-AAS | CV-AAS | 0.0001 | 0.0001 | 0.0005 | |||||||||
| [49] b | + | - | - | - | - | - | ICP-OES | - | - | 0.1 | |||||||||
| [50] a | + | + | - | - | ICP-OES | ICP-OES | - | −0.01 | NR | - | |||||||||
| [51] a | + | + | - | + | ICP-OES | ICP-OES | - | NR | NR | - | |||||||||
| [52] a | + | - | - | - | - | - | CV-AAS | - | - | NR | |||||||||
| [53] a | + | + | + | + | ICP-OES | ICP-OES | - | 0.1 | 0.19 | - | |||||||||
| [54] b | + | - | - | - | - | - | AAS | - | - | 7 | |||||||||
| [55] a | + | - | - | - | - | - | CV-AAS | - | - | 0.04 | |||||||||
| [56] b | + | - | - | - | FF-AAS | FF-AAS | CV-HGUnit | 100 | 100 | 50 | |||||||||
| [57] b | + | + | - | + | FAAS | FAAS | - | NR | NR | 0.04 | |||||||||
| [58] b | + | - | - | - | GF-AAS | GF-AAS | - | 0.45 | 0.33 | NR | |||||||||
| [13] b | + | - | - | - | AAS | AAS | - | 0.16 | 0.88 | - | |||||||||
| [59] a | + | - | - | - | GF-AAS | GF-AAS | MHS | 1 | 0.005 | 0.003 | |||||||||
| [60] a | + | + | + | + | GF-AAS | GF-AAS | CV-AAS | 0.0001 | 0.0001 | 0.00005 | |||||||||
| [8] a | + | - | - | - | GF-AAS | GF-AAS | CV-AAS | 0.0001 | 0.0001 | 0.00005 | |||||||||
| [61] b | + | - | - | - | GF-AAS | - | AFS | NR | - | NR | |||||||||
| [62] b | + | - | - | - | - | - | AuCV-AAS | - | - | 6 | |||||||||
| [63] a | + | - | - | - | - | - | CV-AAS | - | - | 0.16 | |||||||||
| [64] b | + | - | - | - | FAAS | FAAS | - | 20 | 100 | - | |||||||||
| Ref No | Sampling Site | Fish | Tissue | Mean (μg/g) | ||
|---|---|---|---|---|---|---|
| Cd | Pb | Hg | ||||
| [29] | Ionian Sea, IT | Xiphias gladius | Muscle | 0.005 | 0.05 | 0.07 |
| Thunnus thynnus | Muscle | 0.16 | 0.09 | 0.19 | ||
| Xiphias gladius | Liver | 0.02 | 0.1 | 0.2 | ||
| Thunnus thynnus | Liver | 1.5 | 0.21 | 0.39 | ||
| [32] | E.Adriatic Sea, CR | Merluccius merluccius | Muscle | NE | NE | NE |
| Merluccius merluccius | Liver | NE | NE | NE | ||
| Mullus barbatus | Muscle | NE | NE | NE | ||
| Mullus barbatus | Liver | NE | NE | NE | ||
| [33] | Paradeniz Lagoon, TU | Liza saliens | Muscle | 0.48 | 0.52 | NE |
| Liza saliens | Liver | 0.63 | 0.82 | NE | ||
| Liza saliens | Gill | 0.48 | 0.73 | NE | ||
| Liza saliens | Gonad | 0.49 | 0.51 | NE | ||
| Mugil cephalus | Muscle | 0.49 | 0.63 | NE | ||
| Mugil cephalus | Liver | 0.62 | 0.88 | NE | ||
| Mugil cephalus | Gill | 0.52 | 0.54 | NE | ||
| Mugil cephalus | Gonad | 0.5 | 0.57 | NE | ||
| Dicentrarchus labrax | Muscle | 0.67 | 0.67 | NE | ||
| Dicentrarchus labrax | Liver | 0.74 | 0.74 | NE | ||
| Dicentrarchus labrax | Gill | 0.75 | 0.75 | NE | ||
| Dicentrarchus labrax | Gonad | 1.25 | 1.25 | NE | ||
| Sparus aurata | Muscle | 0.2 | 0.2 | NE | ||
| Sparus aurata | Liver | 0.27 | 0.27 | NE | ||
| Sparus aurata | Gill | 0.26 | 0.26 | NE | ||
| Sparus aurata | Gonad | NE | NE | NE | ||
| [37] | Iskenderun Bay, TU | Saurida undosquamis | Muscle | 1.31 | 3.474 | NE |
| Mullus barbatus | Muscle | 0.831 | 1.808 | NE | ||
| Sparus aurata | Muscle | 1.341 | 2.314 | NE | ||
| [17] | Gullk Bay, TU | Dicentrarchus labrax | Muscle | <0.01 | <0.02 | NE |
| [13] | Eastern Harbour and El-Mex Bay, EG | Saurida undosquamis | Muscle | 1.79 | NE | NE |
| Lithognathus mormyrus | Muscle | 1.62 | NE | NE | ||
| Sphyraena sphyraena | Muscle | 1.83 | NE | NE | ||
| Siganus rivulatus | Muscle | 2.82 | NE | NE | ||
| [40] | Iskenderun Bay, TU | Merlangius merlangus | Muscle | 1.685 | 0.426 | NE |
| Engraulis encrasicholus | Muscle | 0.183 | 0.055 | NE | ||
| Mullus barbatus | Muscle | 0.494 | 0.559 | NE | ||
| [42] | Adriatic Sea, CR | Engraulis encrasicholus | Muscle | 0.002 | 0.01 | 0.04 |
| Scomber japonicus | Muscle | 0.006 | 0.01 | 0.08 | ||
| Mullus surmuletus | Muscle | 0.002 | 0.02 | 0.06 | ||
| Spicara smaris | Muscle | 0.003 | 0.02 | 0.08 | ||
| [44] | Adriatic and Ionian seas, IT | Fish | Muscle | 0.05 | 0.08 | 0.85 |
| Fish | Liver | 0.28 | 0.47 | 0.57 | ||
| [49] | Central Adriatic Sea, IT | Mullus barbatus | Homogenized | NE | NE | 0.48 |
| Merluccius merluccius | Homogenized | NE | NE | 0.59 | ||
| Micromesistius poutassou | Homogenized | NE | NE | 0.38 | ||
| Scomber scombrus | Homogenized | NE | NE | 0.36 | ||
| [51] | Iskenderun Bay TU | Triglia lucerna | Liver | 0.24 | 2.48 | NE |
| Triglia lucerna | Skin | 0.12 | 1.81 | NE | ||
| Triglia lucerna | Muscle | 0.01 | 0.14 | NE | ||
| Lophius budegassa | Liver | 0.26 | 1.77 | NE | ||
| Lophius budegassa | Skin | 0.09 | 1.69 | NE | ||
| Lophius budegassa | Muscle | 0.02 | 0.17 | NE | ||
| Solea lascaris | Liver | 0.39 | 2.98 | NE | ||
| Solea lascaris | Skin | 0.08 | 2.09 | NE | ||
| Solea lascaris | Muscle | 0.04 | 0.39 | NE | ||
| [53] | Yelkoma Lagoon, TU | Sparus aurata | Muscle | 0.3 | 0.45 | NE |
| Dicentrarchus labrax | Muscle | 0.1 | 0.19 | NE | ||
| Liza carinata | Muscle | 0.47 | 0.47 | NE | ||
| Sparus aurata | Liver | 0.56 | 0.59 | NE | ||
| Dicentrarchus labrax | Liver | 0.16 | 0.52 | NE | ||
| Liza carinata | Liver | 0.58 | 0.71 | NE | ||
| Sparus aurata | Gonad | 0.62 | 0.43 | NE | ||
| Dicentrarchus labrax | Gonad | 0.13 | 0.42 | NE | ||
| Liza carinata | Gonad | 0.49 | 0.68 | NE | ||
| Sparus aurata | Gill | 0.43 | 0.44 | NE | ||
| Dicentrarchus labrax | Gill | 0.14 | 0.54 | NE | ||
| Liza carinata | Gill | 0.62 | 0.41 | NE | ||
| [54] | Gulf of Lions, FR | Mullus barbatus | Muscle | NE | NE | 1.11 |
| Mullus surmuletus | Muscle | NE | NE | 0.92 | ||
| [55] | Ionian and Adriatic Seas, IT | M. merluccius (Ionian sea) | Muscle | NE | NE | 0.09 |
| M.merluccius (Adriatic sea) | Muscle | NE | NE | 0.18 | ||
| Mullus barbatus | Muscle | NE | NE | 0.4 | ||
| Mullus barbatus | Muscle | NE | NE | 0.49 | ||
| [57] | N.E.Mediterranean Sea, TU | Sparus auratus | Muscle | 0.37 | 5.54 | NE |
| Atherina hepsetus | Muscle | 0.37 | 6.12 | NE | ||
| Trigla cuculus | Muscle | 0.66 | 5.32 | NE | ||
| Sardina pilchardus | Muscle | 0.79 | 4.27 | NE | ||
| Scomberesox saurus | Muscle | 0.55 | 5.57 | NE | ||
| Sparus aurata | Liver | 0.96 | 8.87 | NE | ||
| Atherina hepsetus | Liver | 1.17 | 41.24 | NE | ||
| Trigla cuculus | Liver | 1.64 | 12.59 | NE | ||
| Sardina pilchardus | Liver | 4.5 | 23.01 | NE | ||
| Scomberesox saurus | Liver | 2.99 | 39.43 | NE | ||
| Sparus auratus | Gill | 1.79 | 13.31 | NE | ||
| Atherina hepsetus | Gill | 1.85 | 12.37 | NE | ||
| Trigla cuculus | Gill | 2.08 | 8.95 | NE | ||
| Sardina pilchardus | Gill | 2.25 | 12.81 | NE | ||
| Scomberesox saurus | Gill | 1.56 | 8.99 | NE | ||
| [58] | Aegean Sea, TU | Sparus aurata | NS | 0.5 | 0.62 | NE |
| Scorpaena porcus | NS | 0.8 | 0.66 | NE | ||
| [62] | Ligurian Sea, IT | Farmed Sparus aurata | Muscle | NE | NE | 0.12 |
| Wild Sparus aurata | Muscle | NE | NE | 0.54 | ||
| [63] | Adriatic and Ionian Sea, IT | Thunnus alalunga | Muscle | NE | NE | 1.17 |
| Thunnus thynnus | Muscle | NE | NE | 1.18 | ||
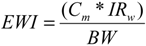
3. Biomonitoring
3.1. Analytical Methods Applied in the Studies
| Country | Demersal | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Mean (μg/g) | EWI (μg/Week/70 kg bw) | EWI/PTWI Ratio | |||||||
| Cd | Pb | Hg | Cd | Pb | Hg | Cd | Pb | Hg | |
| TU | 0.23 | 1.05 | NR | 0.12 | 0.52 | NR | 0.05 | 0.02 | NR |
| IT | 0.08 | 0.13 | 0.28 | 0.16 | 0.25 | 0.53 | 0.06 | 0.01 | 0.13 |
| EG | NR | 1.74 | NR | NR | 2.09 | NR | NR | 0.08 | NR |
| FR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| CR | 0.01 | 0.03 | 0.20 | 0.00 | 0.02 | 0.14 | 0.00 | 0.00 | 0.03 |
| Country | Pelagic | ||||||||
| Mean (μg/g) | EWI (μg/week/70 kg bw) | EWI/PTWI ratio | |||||||
| Cd | Pb | Hg | Cd | Pb | Hg | Cd | Pb | Hg | |
| TU | 0.14 | 2.03 | NR | 0.20 | 2.84 | NR | 0.08 | 0.11 | NR |
| IT | 0.16 | 0.49 | 0.64 | 0.23 | 0.68 | 0.90 | 0.09 | 0.03 | 0.22 |
| EG | 0.19 | 1.70 | NR | 0.19 | 1.70 | NR | 0.08 | 0.07 | NR |
| FR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| CR | 0.01 | 0.02 | 0.18 | 0.03 | 0.05 | 0.49 | 0.01 | 0.00 | 0.12 |
| Country | Marine | ||||||||
| Mean (μg/g) | EWI (μg/week/70 kg bw) | EWI/PTWI ratio | |||||||
| Cd | Pb | Hg | Cd | Pb | Hg | Cd | Pb | Hg | |
| TU | 0.22 | 5.66 | 0.50 | 0.13 | 3.40 | 0.30 | 0.05 | 0.14 | 0.08 |
| IT | NR | NR | 1.05 | NR | NR | 0.53 | NR | NR | 0.13 |
| EG | 0.75 | 1.46 | NR | 0.45 | 0.87 | NR | 0.18 | 0.03 | NR |
| FR | 0.04 | NR | 0.26 | 0.01 | NR | 0.08 | 0.00 | NR | 0.02 |
| CR | 0.00 | 0.02 | 0.32 | 0.00 | 0.00 | 0.10 | 0.00 | 0.00 | 0.02 |
3.2. Issues of Metal Accumulation in Fish
3.3. Cadmium, Lead and Mercury in Fish Tissues of the Mediterranean
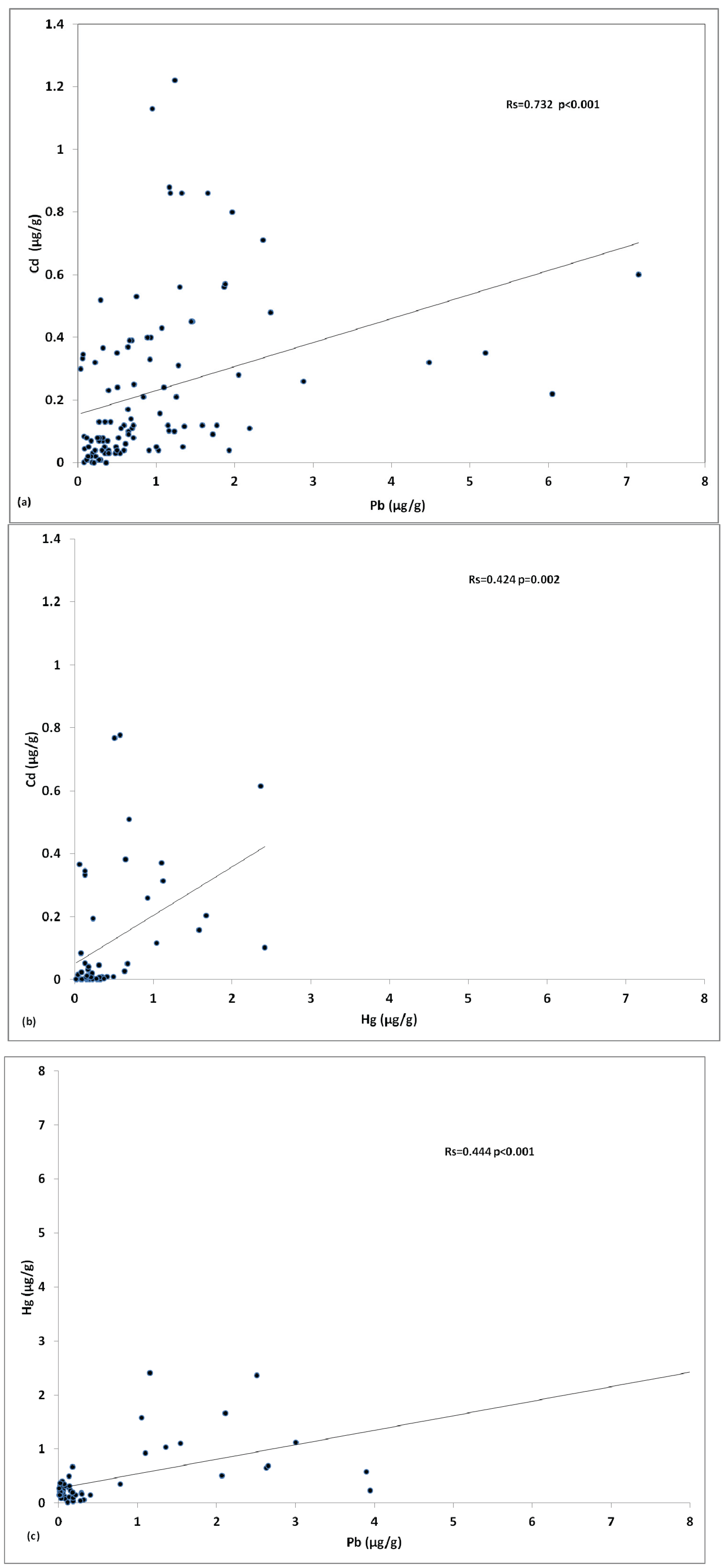
3.4. Distribution of Cadmium, Lead and Mercury amongst Fish Species
3.5. Distribution of Cadmium, Lead and Mercury amongst Fish Gonad, Liver and Muscle Tissues

4. Risk Assessment
4.1. Human Health Effects-Implications for Fish Consumption
4.2. Data from Human Dietary Intake Studies
4.3. Estimation of Risk Assessment Scenario
5. Conclusions
Abbreviations
| JECFA | The Joint FAO/WHO Expert Committee on Food Additives |
| US-EPA | The US Environmental Protection Agency |
| EFSA | European Food Safety Authority |
| US FDA | The US Food and Drug Administration |
| EC | European Commission Regulation |
| EWI | Estimated Weekly Intake (μg/week/70 kg body weight) |
| EDI | Estimated Daily Intake in μg/week/70 kg body weight |
| PTWI | Provisional Tolerable Weekly Intake PTWI (μg/week/kg body weight) |
| PTWIa | PTWI for 70 kg adult person (μg/week/70 kg body weight) |
Acknowledgments
Author Contributions
Conflicts of Interest
References
- European Environment Agency (EEA). Priority Issues in the Mediterranean Environment; EEA: Copenhagen, Denmark, 2006. [Google Scholar]
- Damiano, S.; Papetti, P.; Menesatti, P. Accumulation of heavy metals to assess the health status of swordfish in a comparative analysis of mediterranean and atlantic areas. Mar. Pollut. Bull. 2011, 62, 1920–1925. [Google Scholar] [CrossRef]
- World Health Organization. Health Risks of Heavy Metals from Long-Range Transboundary Air Pollution; WHO Regional Office for Europe: Copenhagen, Denmark, 2007. [Google Scholar]
- Alonso Castillo, M.L.; Sanchez Trujillo, I.; Vereda Alonso, E.; Garcia de Torres, A.; Cano Pavon, J.M. Bioavailability of heavy metals in water and sediments from a typical mediterranean bay (Malaga Bay, region of Andalucia, Southern Spain). Mar. Pollut. Bull. 2013, 76, 427–434. [Google Scholar] [CrossRef]
- Copat, C.; Bella, F.; Castaing, M.; Fallico, R.; Sciacca, S.; Ferrante, M. Heavy metals concentrations in fish from sicily (Mediterranean sea) and evaluation of possible health risks to consumers. Bull. Environ. Contam. Toxicol. 2012, 88, 78–83. [Google Scholar] [CrossRef]
- UNEP/FAO/IAEA. Designing of Monitoring Programs and Management of Data Concerning Chemical Contaminants in Marine Organisms. In Map Technical Reports Series No. 77; UNEP: Athens, Greece, 1993. [Google Scholar]
- Storelli, M.M.; Marcotrigiano, G.O. Bioindicator organisms: Heavy metal pollution evaluation in the Ionian sea (Mediterranean sea–Italy). Environ. Monit. Assess. 2005, 102, 159–166. [Google Scholar] [CrossRef]
- Kucuksezgin, F.; Altay, O.; Uluturhan, E.; Kontas, A. Trace metal and organochlorine residue levels in red mullet (Mullus barbatus) from the Eastern Aegean, Turkey. Water Res. 2001, 35, 2327–2332. [Google Scholar]
- Ferreira, M.; Caetano, M.; Antunes, P.; Costa, J.; Gil, O.; Bandarra, N.; Pousao-Ferreira, P.; Vale, C.; Reis-Henriques, M.A. Assessment of contaminants and biomarkers of exposure in wild and farmed seabass. Ecotoxicol. Environ. Saf. 2010, 73, 579–588. [Google Scholar] [CrossRef]
- Creti, P.; Trinchella, F.; Scudiero, R. Heavy metal bioaccumulation and metallothionein content in tissues of the sea bream Sparus aurata from three different fish farming systems. Environ. Monit. Assess. 2010, 165, 321–329. [Google Scholar] [CrossRef]
- Shahidul Islam, M.; Tanaka, M. Impacts of pollution on coastal and marine ecosystems including coastal and marine fisheries and approach for management: A review and synthesis. Mar. Poll. Bull. 2004, 48, 624–649. [Google Scholar] [CrossRef]
- Cesar, A.; Marin, A.; Marin-Guirao, L.; Vita, R.; Lloret, J.; del Valls, T.A. Integrative ecotoxicological assessment of sediment in Portman bay (Southeast Spain). Ecotoxicol. Environ. Saf. 2009, 72, 1832–1841. [Google Scholar] [CrossRef]
- Abdallah, M.A.; Abdallah, A.M. Biomonitoring study of heavy metals in biota and sediments in the south eastern coast of Mediterranean Sea, Egypt. Environ. Monit. Assess. 2008, 146, 139–145. [Google Scholar] [CrossRef]
- Yilmaz, A.B. Levels of heavy metals (Fe, Cu, Ni, Cr, Pb, and Zn) in tissue of Mugil cephalus and Trachurus mediterraneus from Iskenderun Bay, Turkey. Environ. Res. 2003, 92, 277–281. [Google Scholar] [CrossRef]
- Kucuksezgin, F.; Kontas, A.; Uluturhan, E. Evaluations of heavy metal pollution in sediment and Mullus barbatus from the Izmir bay (Eastern Aegean) during 1997–2009. Mar. Pollut. Bull. 2011, 62, 1562–1571. [Google Scholar] [CrossRef]
- Houston, M.C. The role of mercury and cadmium heavy metals in vascular disease, hypertension, coronary heart disease, and myocardial infarction. Altern. Ther. Health Med. 2007, 13, S128–S133. [Google Scholar]
- Dalman, Ö.; Demirak, A.; Balcl, A. Determination of heavy metals (Cd, Pb) and trace elements (Cu, Zn) in sediments and fish of the Southeastern Aegean Sea (Turkey) by atomic absorption spectrometry. Food Chem. 2006, 95, 157–162. [Google Scholar]
- Fallah, A.A.; Saei-Dehkordi, S.S.; Nematollahi, A.; Jafari, T. Comparative study of heavy metal and trace element accumulation in edible tissues of farmed and wild rainbow trout (Oncorhynchus mykiss) using ICP-OES technique. Microchem. J. 2011, 98, 275–279. [Google Scholar] [CrossRef]
- Khan, M.U.; Malik, R.N.; Muhammad, S. Human health risk from heavy metal via food crops consumption with wastewater irrigation practices in Pakistan. Chemosphere 2013, 93, 2230–2238. [Google Scholar]
- European Union. Setting Maximum Levels for Certain Contaminants in Foodstuffs; Official Journal of the European Union, 2006; pp. 5–24. [Google Scholar]
- European Union. Setting Maximum Levels for Certain Contaminants in Foodstuffs; Official Journal of the European Union, 3 July 2008; pp. 6–9. [Google Scholar]
- European Food Safety Authority (EFSA). Scientific opinion of the panel on contaminants in the food chain on a request from the European commission on cadmium in food. EFSA J. 2009, 1–139. [Google Scholar]
- European Food Safety Authority (EFSA). Scientific opinion on lead in food. EFSA Panel on Contaminants in the Food Chain (CONTAM). EFSA J. 2010, 8, 147. [Google Scholar]
- European Food Safety Authority (EFSA). Scientific opinion on the risk for public health related to the presence of mercury and methylmercury in food. EFSA J. 2012, 10, 241. [Google Scholar]
- FAO/WHO. Summary and Conclusions of the Sixty-First Meeting of the Joint Fao/Who Expert Committee on Food Additives (Jecfa); World Health Organization: Rome, Italy, 2003. [Google Scholar]
- FAO/WHO. Summary and Conclusions of the Sixty-Seventh Meeting of the Joint Fao/Who Expert Committee on Food Additives (Jecfa); World Health Organization: Rome, Italy, 2006. [Google Scholar]
- FAO/WHO. Evaluation of Certain Food Additives and Contaminants: Seventy-Third Report of the Joint Fao/Who Expert Committee on Food Additives (Jecfa); World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- FAO. Available online: http://faostat.Fao.Org/ (accessed on 9 October 2013).
- Storelli, M.M.; Giacominelli-Stuffler, R.; Storelli, A.; Marcotrigiano, G.O. Accumulation of mercury, cadmium, lead and arsenic in swordfish and bluefin tuna from the Mediterranean Sea: A comparative study. Mar. Pollut. Bull. 2005, 50, 1004–1007. [Google Scholar] [CrossRef]
- Marijic, V.F.; Raspor, B. Age- and tissue-dependent metallothionein and cytosolic metal distribution in a native Mediterranean fish, mullus barbatus, from the Eastern Adriatic Sea. Comp. Biochem. Physiol. C 2006, 143, 382–387. [Google Scholar] [CrossRef]
- Dural, M.; Goksu, M.Z.; Ozak, A.A.; Derici, B. Bioaccumulation of some heavy metals in different tissues of Dicentrarchus labrax l, 1758, Sparus aurata l, 1758 and Mugil cephalus l, 1758 from the camlik lagoon of the eastern coast of Mediterranean (Turkey). Environ. Monit. Assess. 2006, 118, 65–74. [Google Scholar] [CrossRef]
- Gaspic, Z.K.; Zvonaric, T.; Vrgoc, N.; Odzak, N.; Baric, A. Cadmium and lead in selected tissues of two commercially important fish species from the Adriatic Sea. Water Res. 2002, 36, 5023–5028. [Google Scholar]
- Turkmen, M.; Turkmen, A.; Tepe, Y. Comparison of metals in tissues of fish from paradeniz lagoon in the coastal area of Northern East Mediterranean. Bull. Environ. Contam. Toxicol. 2011, 87, 381–385. [Google Scholar] [CrossRef]
- Tepe, Y. Metal concentrations in eight fish species from aegean and mediterranean seas. Environ. Monit. Assess. 2009, 159, 501–509. [Google Scholar] [CrossRef]
- Sepe, A.; Ciaralli, L.; Ciprotti, M.; Giordano, R.; Funari, E.; Costantini, S. Determination of cadmium, chromium, lead and vanadium in six fish species from the Adriatic Sea. Food Addit. Contam. 2003, 20, 543–552. [Google Scholar] [CrossRef]
- Türkmen, M.; Türkmen, A.; Tepe, Y.; Töre, Y.; Ateş, A. Determination of metals in fish species from Aegean and Mediterranean seas. Food Chem. 2009, 113, 233–237. [Google Scholar]
- Türkmen, A.; Türkmen, M.; Tepe, Y.; Akyurt, İ. Heavy metals in three commercially valuable fish species from İskenderun bay, Northern East Mediterranean Sea, Turkey. Food Chem. 2005, 91, 167–172. [Google Scholar] [CrossRef]
- Abernathy, C.O.; Thomas, D.J.; Calderon, R.L. Health effects and risk assessment of Arsenic. J. Nutr. 2003, 133, 1536s–1538s. [Google Scholar]
- Türkmen, M.; Türkmen, A.; Tepe, Y.; Ateş, A.; Gökkuş, K. Determination of metal contaminations in sea foods from Marmara, Aegean and Mediterranean seas: Twelve fish species. Food Chem. 2008, 108, 794–800. [Google Scholar]
- Turan, C.; Dural, M.; Oksuz, A.; Ozturk, B. Levels of heavy metals in some commercial fish species captured from the black sea and Mediterranean Coast of Turkey. Bull. Environ. Contam. Toxicol. 2009, 82, 601–604. [Google Scholar] [CrossRef]
- Cogun, H.Y.; Yuzereroglu, T.A.; Firat, O.; Gok, G.; Kargin, F. Metal concentrations in fish species from the northeast Mediterranean sea. Environ. Monit. Assess. 2006, 121, 431–438. [Google Scholar] [CrossRef]
- Bilandžić, N.; Đokić, M.; Sedak, M. Metal content determination in four fish species from the Adriatic sea. Food Chem. 2011, 124, 1005–1010. [Google Scholar] [CrossRef]
- Uluturhan, E.; Kucuksezgin, F. Heavy metal contaminants in red pandora (Pagellus erythrinus) tissues from the eastern Aegean sea, Turkey. Water Res. 2007, 41, 1185–1192. [Google Scholar]
- Marcotrigiano, G.O.; Storelli, M.M. Heavy metal, polychlorinated biphenyl and organochlorine pesticide residues in marine organisms: Risk evaluation for consumers. Vet. Res. Commun. 2003, 27, 183–195. [Google Scholar] [CrossRef]
- Papetti, P.; Rossi, G. Heavy metals in the fishery products of low lazio and the use of metallothionein as a biomarker of contamination. Environ. Monit. Assess. 2009, 159, 589–598. [Google Scholar] [CrossRef]
- Çelik, U.; Oehlenschläger, J. High contents of cadmium, lead, zinc and copper in popular fishery products sold in Turkish supermarket. Food Control. 2007, 18, 258–261. [Google Scholar]
- Juresa, D.; Blanusa, M. Mercury, arsenic, lead and cadmium in fish and shellfish from the Adriatic Sea. Food Addit. Contam. 2003, 20, 241–246. [Google Scholar] [CrossRef]
- Spada, L.; Annicchiarico, C.; Cardellicchio, N.; Giandomenico, S.; di Leo, A. Mercury and methylmercury concentrations in mediterranean seafood and surface sediments, intake evaluation and risk for consumers. Int. J. Hyg. Environ. Health 2012, 215, 418–426. [Google Scholar] [CrossRef]
- Perugini, M.; Visciano, P.; Manera, M.; Zaccaroni, A.; Olivieri, V.; Amorena, M. Levels of total mercury in marine organisms from Adriatic Sea, Italy. Bull. Environ. Contam. Toxicol. 2009, 83, 244–248. [Google Scholar] [CrossRef]
- Turkmen, A.; Tepe, Y.; Turkmen, M. Metal levels in tissues of the European anchovy, Engraulis encrasicolus l., 1758, and picarel, spicara smaris l., 1758, from black, Marmara and Aegean Seas. Bull. Environ. Contam. Toxicol. 2008, 80, 521–525. [Google Scholar] [CrossRef]
- Yılmaz, A.B.; Sangün, M.K.; Yağlıoğlu, D.; Turan, C. Metals (major, essential to non-essential) composition of the different tissues of three demersal fish species from İskenderun Bay, Turkey. Food Chem. 2010, 123, 410–415. [Google Scholar]
- Gonul, L.T.; Kucuksezgin, F. Mercury accumulation and speciation in the muscle of red mullet (Mullus barbatus) and annular sea bream (Diplodus annularis) from Izmir bay (Eastern Aegean). Mar. Pollut. Bull. 2007, 54, 1962–1968. [Google Scholar] [CrossRef]
- Turkmen, A.; Turkmen, M.; Tepe, Y.; Cekic, M. Metals in tissues of fish from yelkoma lagoon, Northeastern Mediterranean. Environ. Monit Assess. 2010, 168, 223–230. [Google Scholar] [CrossRef]
- Harmelin-Vivien, M.; Cossa, D.; Crochet, S.; Bănaru, D.; Letourneur, Y.; Mellon-Duval, C. Difference of mercury bioaccumulation in red mullets from the North-Western Mediterranean and Black Seas. Mar. Poll. Bull. 2009, 58, 679–685. [Google Scholar] [CrossRef]
- Storelli, M.M.; Storelli, A.; Giacominelli-Stuffler, R.; Marcotrigiano, G.O. Mercury speciation in the muscle of two commercially important fish, hake (Merluccius merluccius) and striped mullet (Mullus barbatus) from the mediterranean sea: Estimated weekly intake. Food Chem. 2005, 89, 295–300. [Google Scholar]
- Aksu, A.; Balkis, N.; Taskin, O.S.; Ersan, M.S. Toxic metal (Pb, Cd, As and Hg) and organochlorine residue levels in hake (Merluccius merluccius) from the Marmara Sea, Turkey. Environ. Monit. Assess. 2011, 182, 509–521. [Google Scholar] [CrossRef]
- Canli, M.; Atli, G. The relationships between heavy metal (Cd, Cr, Cu, Fe, Pb, Zn) levels and the size of six Mediterranean fish species. Environ. Pollut. 2003, 121, 129–136. [Google Scholar] [CrossRef]
- Uluozlu, O.D.; Tuzen, M.; Mendil, D.; Soylak, M. Trace metal content in nine species of fish from the Black and Aegean Seas, Turkey. Food Chem. 2007, 104, 835–840. [Google Scholar]
- Amodio-Cocchieri, R.; Amoroso, S.; Arnese, A.; Cirillo, T.; Montuori, P.; Triassi, M. Pollution by mercury, arsenic, lead, chromium, cadmium, and polycyclic aromatic hydrocarbons of fish and mussels from the Gulf of Naples, Italy. Bull. Environ. Contam. Toxicol. 2003, 71, 551–560. [Google Scholar] [CrossRef]
- Kucuksezgin, F.; Uluturhan, E.; Kontas, A.; Altay, O. Trace metal concentrations in edible fishes from Izmir Bay, Eastern Aegean. Mar. Pollut. Bull. 2002, 44, 827–832. [Google Scholar] [CrossRef]
- Dierking, J.; Wafo, E.; Schembri, T.; Lagadec, V.; Nicolas, C.; Letourneur, Y.; Harmelin-Vivien, M. Spatial patterns in PCBs, pesticides, mercury and cadmium in the common sole in the nw mediterranean sea, and a novel use of contaminants as biomarkers. Mar. Pollut. Bull. 2009, 58, 1605–1614. [Google Scholar] [CrossRef]
- Minganti, V.; Drava, G.; De Pellegrini, R.; Siccardi, C. Trace elements in farmed and wild Gilthead seabream, Sparus aurata. Mar. Pollut. Bull. 2010, 60, 2022–2025. [Google Scholar] [CrossRef]
- Storelli, M.M.; Stuffler, R.G.; Marcotrigiano, G.O. Total and methylmercury residues in tuna-fish from the Mediterranean Sea. Food Addit. Contam. 2002, 19, 715–720. [Google Scholar] [CrossRef]
- Turkmen, A.; Turkmen, M.; Tepe, Y.; Mazlum, Y.; Oymael, S. Metal concentrations in blue crab (Callinectes sapidus) and mullet (Mugil cephalus) in Iskenderun Bay, Northern East Mediterranean, Turkey. Bull. Environ. Contam. Toxicol. 2006, 77, 186–193. [Google Scholar] [CrossRef]
- Tepe, Y.; Turkmen, M.; Turkmen, A. Assessment of heavy metals in two commercial fish species of four Turkish Seas. Environ. Monit. Assess. 2008, 146, 277–284. [Google Scholar] [CrossRef]
- Castro-González, M.I.; Méndez-Armenta, M. Heavy metals: Implications associated to fish consumption. Environ. Toxicol. Pharmacol. 2008, 26, 263–271. [Google Scholar] [CrossRef]
- Conti, G.O.; Copat, C.; Ledda, C.; Fiore, M.; Fallico, R.; Sciacca, S.; Ferrante, M. Evaluation of heavy metals and polycyclic aromatic hydrocarbons (pahs) in mullus barbatus from sicily channel and risk-based consumption limits. Bull. Environ. Contam. Toxicol. 2012, 88, 946–950. [Google Scholar] [CrossRef]
- Storelli, M.M. Potential human health risks from metals (Hg, Cd, and Pb) and polychlorinated biphenyls (PCBs) via seafood consumption: Estimation of target hazard quotients (THQs) and toxic equivalents (TEQs). Food Chem. Toxicol. 2008, 46, 2782–2788. [Google Scholar] [CrossRef]
- Chan, H.M.; Egeland, G.M. Fish consumption, mercury exposure, and heart diseases. Nutr. Rev. 2004, 62, 68–72. [Google Scholar] [CrossRef]
- Gochfeld, M.; Burger, J. Good fish/bad fish: A composite benefit-risk by dose curve. Neurotoxicology 2005, 26, 511–520. [Google Scholar] [CrossRef]
- Falco, G.; Llobet, J.M.; Bocio, A.; Domingo, J.L. Daily intake of arsenic, cadmium, mercury, and lead by consumption of edible marine species. J. Agric. Food Chem. 2006, 54, 6106–6112. [Google Scholar] [CrossRef]
- Gochfeld, M. Cases of mercury exposure, bioavailability, and absorption. Ecotoxicol. Environ. Saf. 2003, 56, 174–179. [Google Scholar] [CrossRef]
- Clarkson, T.W.; Magos, L.; Myers, G.J. The toxicology of mercury–current exposures and clinical manifestations. N. Engl. J. Med. 2003, 349, 1731–1737. [Google Scholar] [CrossRef]
- Goyer, R.A. Toxic and essential metal interactions. Annu. Rev. Nutr. 1997, 17, 37–50. [Google Scholar] [CrossRef]
- Wang, B.; Du, Y. Cadmium and its neurotoxic effects. Oxid Med. Cell. Longev. 2013, 2013, 898034. [Google Scholar]
- Marti-Cid, R.; Llobet, J.M.; Castell, V.; Domingo, J.L. Dietary intake of arsenic, cadmium, mercury, and lead by the population of Catalonia, Spain. Biol. Trace Elem. Res. 2008, 125, 120–132. [Google Scholar] [CrossRef]
- Vinceti, M.; Crespi, C.M.; Bonvicini, F.; Malagoli, C.; Ferrante, M.; Marmiroli, S.; Stranges, S. The need for a reassessment of the safe upper limit of selenium in drinking water. Sci. Total Environ. 2013, 443, 633–642. [Google Scholar] [CrossRef]
- Vinceti, M.; Dennert, G.; Crespi, C.M.; Zwahlen, M.; Brinkman, M.; Zeegers, M.P.; Horneber, M.; D’Amico, R.; del Giovane, C. Selenium for preventing cancer. Cochr. Database Syst. Rev. 2014, 3, CD005195. [Google Scholar]
- Zwolak, I.; Zaporowska, H. Selenium interactions and toxicity: A review. Selenium interactions and toxicity. Cell. Biol. Toxicol. 2012, 28, 31–46. [Google Scholar] [CrossRef]
- Zhang, H.; Feng, X.; Chan, H.M.; Larssen, T. New insights into traditional health risk assessments of mercury exposure: Implications of selenium. Environ. Sci. Technol. 2014, 48, 1206–1212. [Google Scholar] [CrossRef]
- Glaser, V.; Nazari, E.M.; Muller, Y.M.; Feksa, L.; Wannmacher, C.M.; Rocha, J.B.; de Bem, A.F.; Farina, M.; Latini, A. Effects of inorganic selenium administration in methylmercury-induced neurotoxicity in mouse cerebral cortex. Int. J. Dev. Neurosci. 2010, 28, 631–637. [Google Scholar] [CrossRef]
- Heinz, G.H.; Hoffman, D.J.; Klimstra, J.D.; Stebbins, K.R. A comparison of the teratogenicity of methylmercury and selenomethionine injected into bird eggs. Arch. Environ. Contam. Toxicol. 2012, 62, 519–528. [Google Scholar] [CrossRef]
- Penglase, S.; Hamre, K.; Ellingsen, S. Selenium and mercury have a synergistic negative effect on fish reproduction. Aquat. Toxicol. 2014, 149, 16–24. [Google Scholar] [CrossRef]
- Copat, C.; Vinceti, M.; D’Agati, M.G.; Arena, G.; Mauceri, V.; Grasso, A.; Fallico, R.; Sciacca, S.; Ferrante, M. Mercury and selenium intake by seafood from the Ionian Sea: A risk evaluation. Ecotoxicol. Environ. Saf. 2014, 100, 87–92. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Renieri, E.A.; Alegakis, A.K.; Kiriakakis, M.; Vinceti, M.; Ozcagli, E.; Wilks, M.F.; Tsatsakis, A.M. Cd, Pb and Hg Biomonitoring in Fish of the Mediterranean Region and Risk Estimations on Fish Consumption. Toxics 2014, 2, 417-442. https://doi.org/10.3390/toxics2030417
Renieri EA, Alegakis AK, Kiriakakis M, Vinceti M, Ozcagli E, Wilks MF, Tsatsakis AM. Cd, Pb and Hg Biomonitoring in Fish of the Mediterranean Region and Risk Estimations on Fish Consumption. Toxics. 2014; 2(3):417-442. https://doi.org/10.3390/toxics2030417
Chicago/Turabian StyleRenieri, Elisavet A., Athanasios K. Alegakis, Michalis Kiriakakis, Marco Vinceti, Eren Ozcagli, Martin F. Wilks, and Aristidis M. Tsatsakis. 2014. "Cd, Pb and Hg Biomonitoring in Fish of the Mediterranean Region and Risk Estimations on Fish Consumption" Toxics 2, no. 3: 417-442. https://doi.org/10.3390/toxics2030417






