IF-WS2/Nanostructured Carbon Hybrids Generation and Their Characterization
Abstract
:1. Introduction
2. Results and Discussion
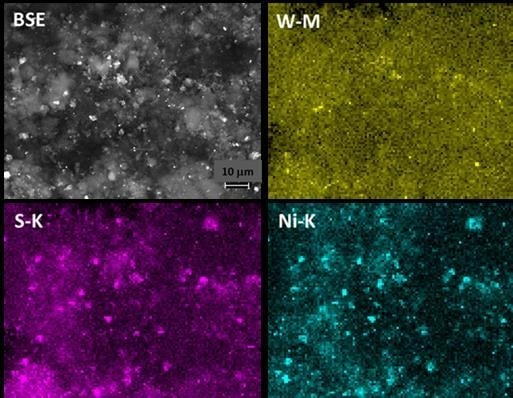
2.1. Carbon Nanofiber/IF-WS2 Hybrids


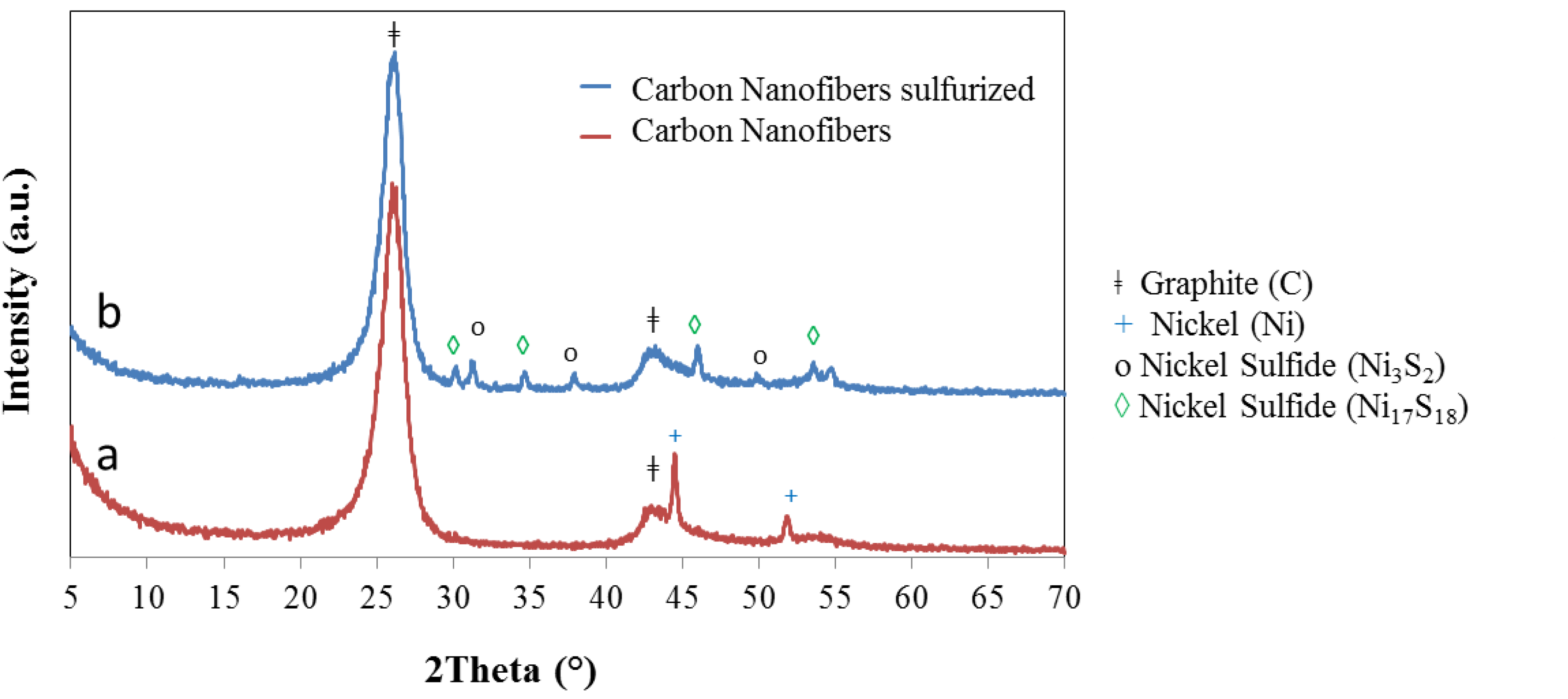
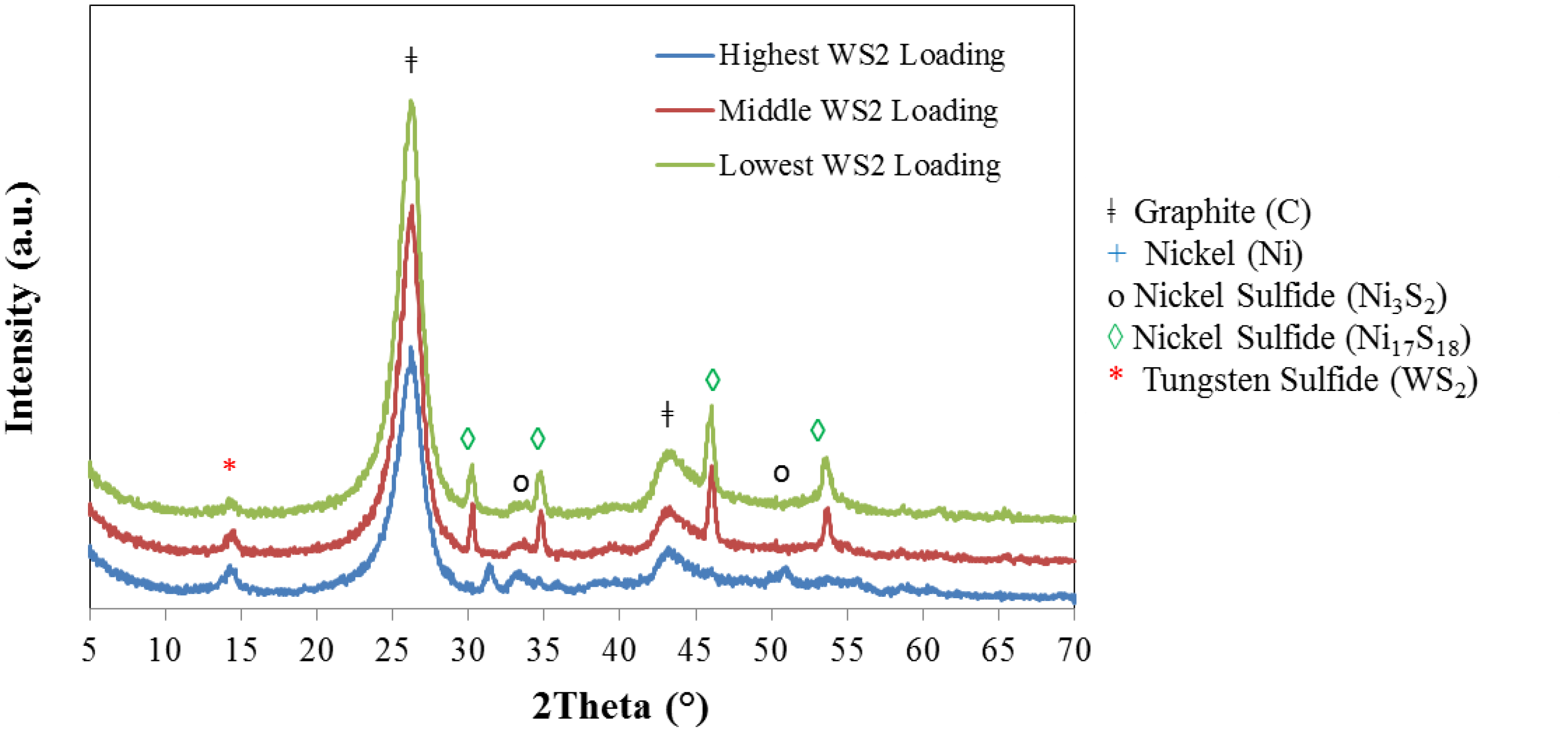

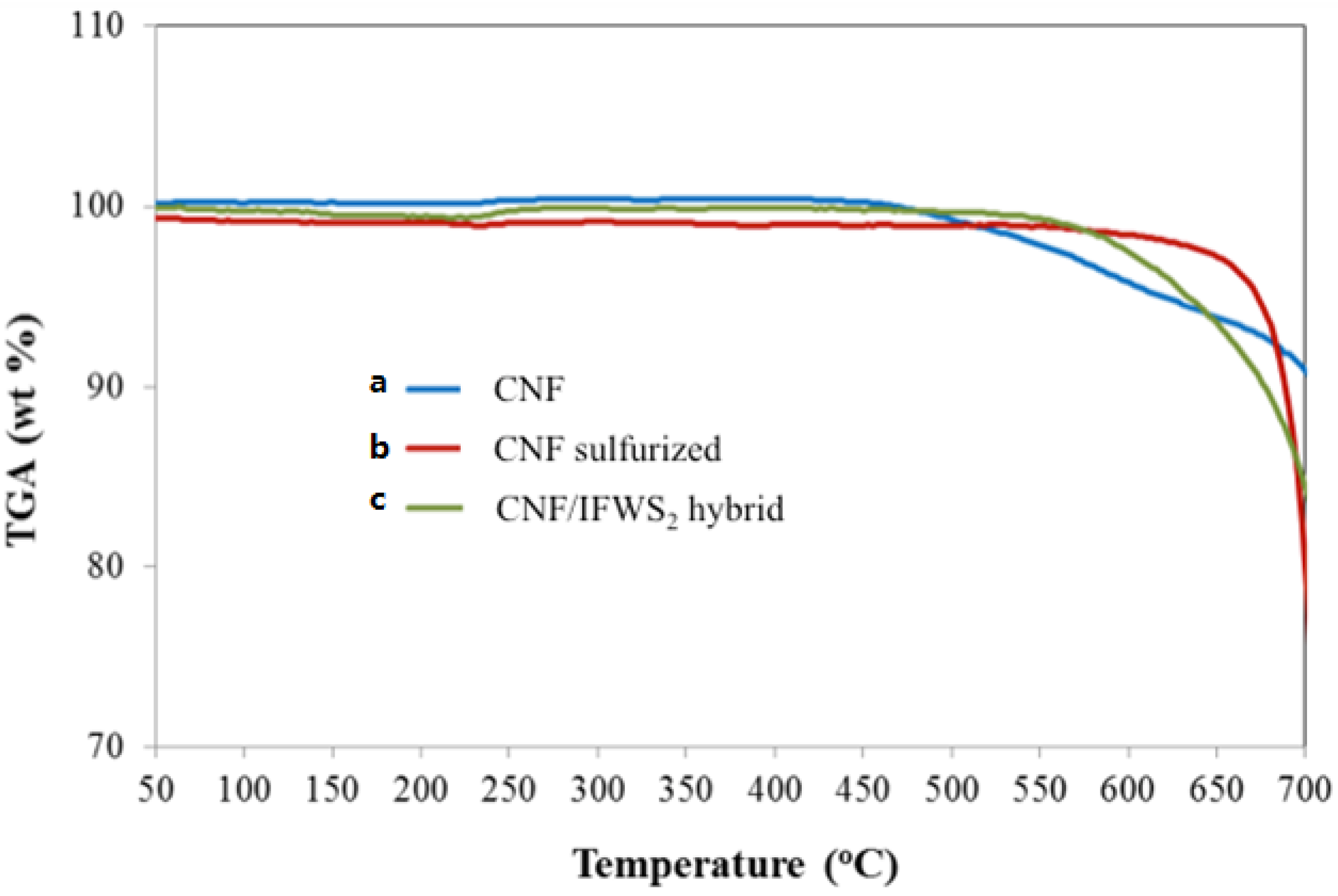
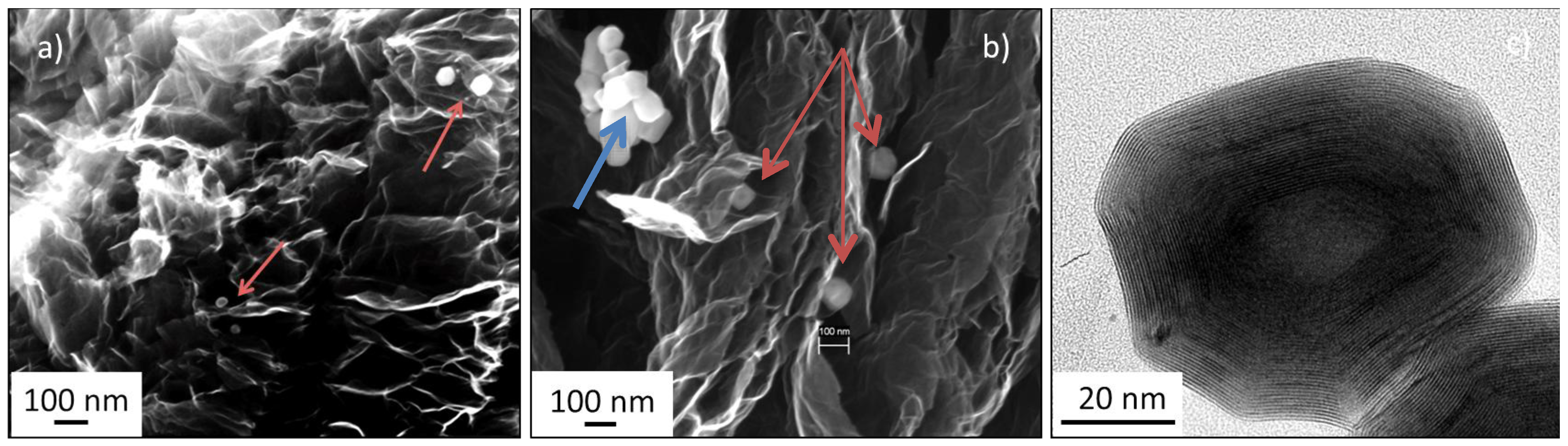
2.2. IF-WS2/Graphene Hybrids
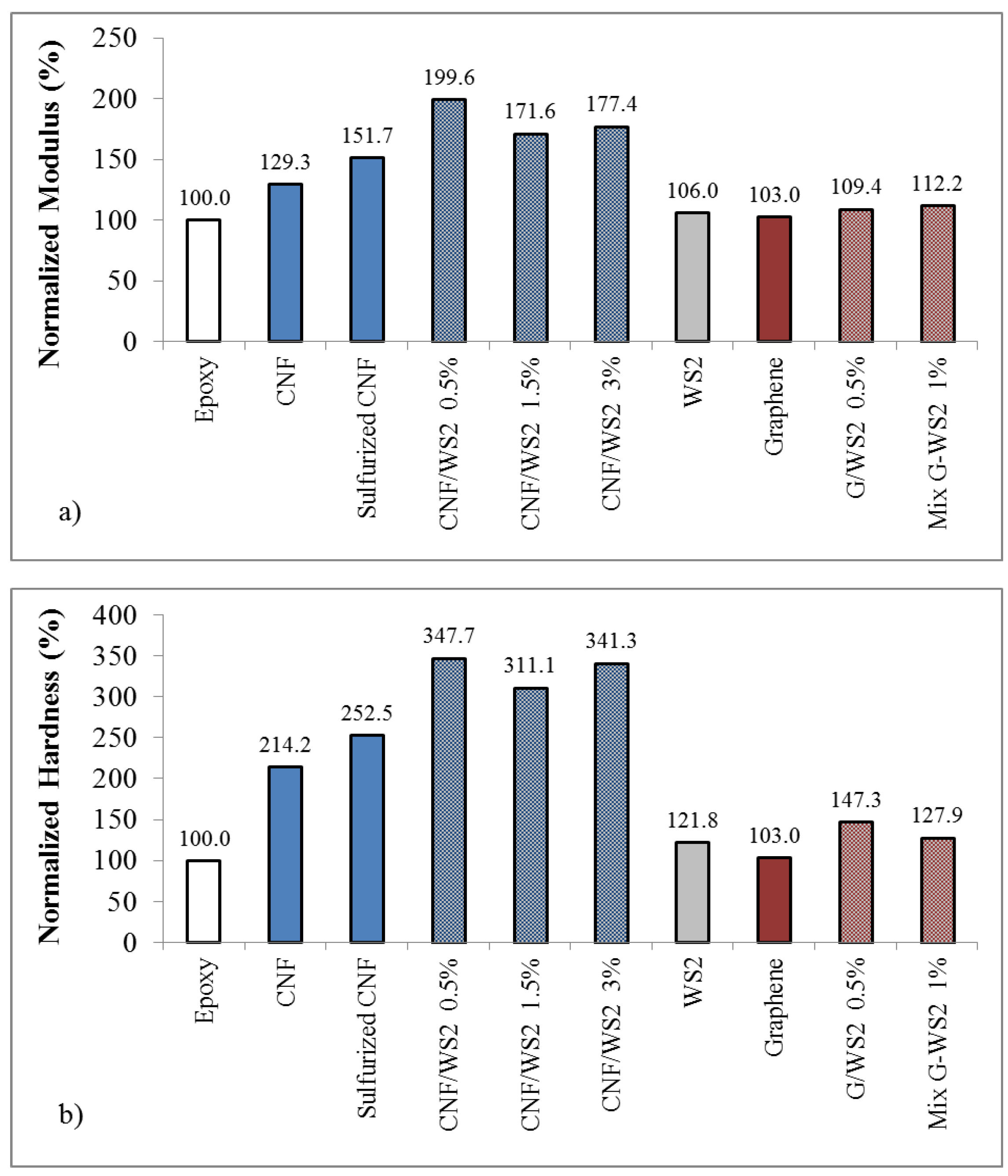
2.3. Epoxy Composites

3. Experimental Section
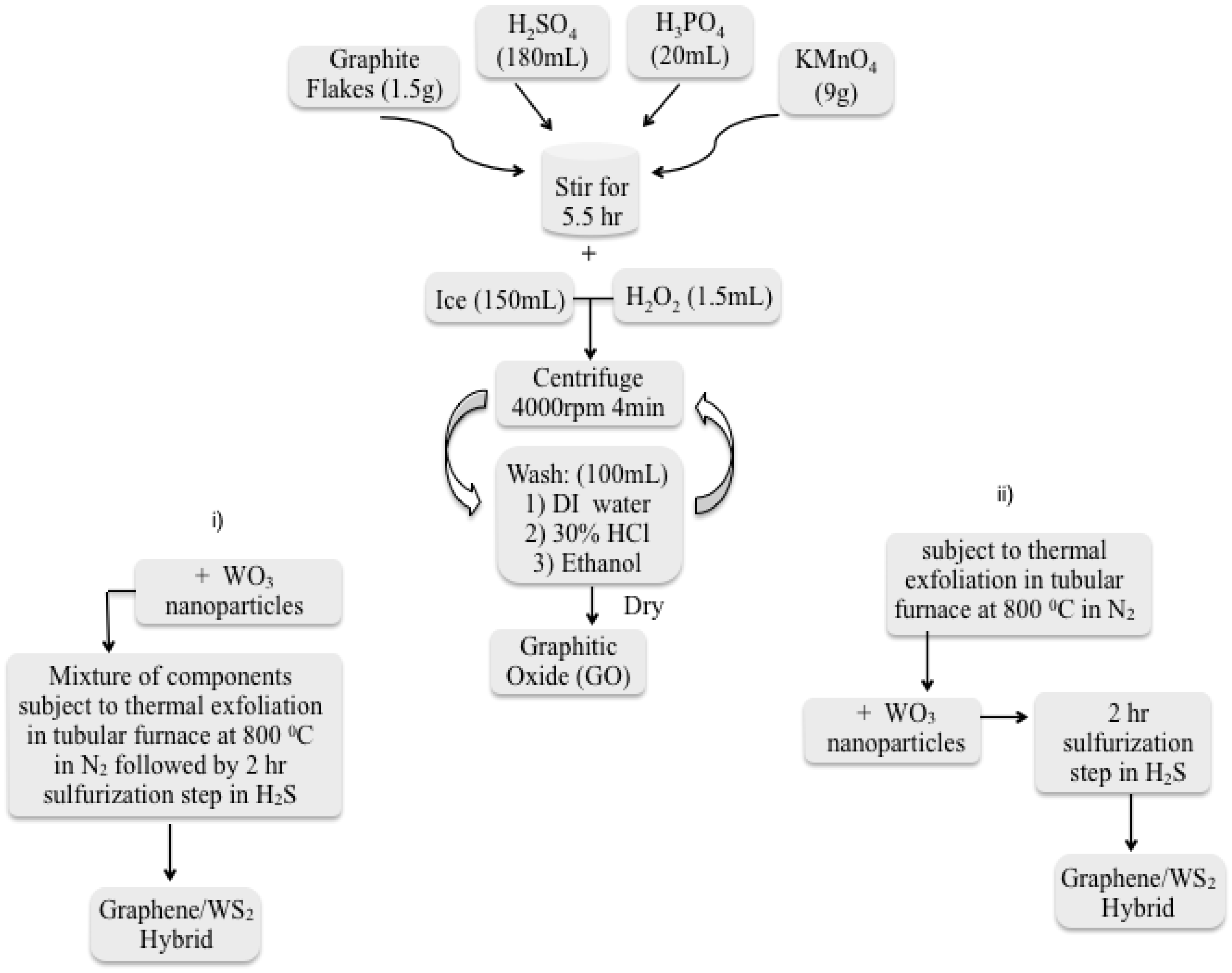
3.1. Carbon Nanofiber Hybrids/IF-WS2

3.2. IF-WS2/Graphene Hybrids
3.3. Epoxy Composites
| Sample ID (IF-WS2 nominal composition – filler only) | ICP value CNF | ICP value Ni | ICP value IF-WS2 |
| CNF | 96.8% | 3.2% | |
| CNF/IF-WS2 (3%) | 94% | 3% | 3% |
| CNF/IF-WS2 (1.5%) | 94.6% | 3.2% | 1.6% |
| CNF/IF-WS2 (0.5%) | 96.4% | 2.9% | 0.7% |
| IF-WS2 (100%) | 100% | ||
| Sample ID (IFWS2 nominal composition-filler only) | Graphene | IF-WS2 | |
| Graphene | 100% | ||
| G/IF-WS2 0.5% | 99.5% | 0.5% | |
| Physical Mix G/IF-WS2 1% | 99% | 1% |

3.4. Characterization Methods
| Depth limit | 2000 nm |
| Strain rate during loading | 0.08/s |
| Maximum allowable drift rate | 0.05 nm/s |
| Peak hold time | 10 s |
| Assumed Poisson’s ratio | 0.40 |
| % to unload | 90 |
| % Unload in stiffness calculation | 50 |
| Flat punch diameter | 50 µm |
| Assumed Poisson’s ratio | 0.40 |
| Pre-compression depth | 2 µm |
| Oscillation amplitude | 50 nm |
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Margulis, L.; Tenne, R.; Iijima, S. Nucleation of WS2 fullerenes at room temperature. Microsc. Microanal. Microstruct. 1996, 7, 87–89. [Google Scholar] [CrossRef]
- Rapoport, L.; Bilik, Y.; Feldman, Y.; Homyonfer, M.; Cohen, S.; Tenne, R. Hollow Nanoparticles of WS2 as potential solid-state lubricants. Nature 1997, 387, 791–793. [Google Scholar] [CrossRef]
- Eidelman, O.; Friedman, H.; Tenne, R. Metallic films with fullerene-Like WS2 (MoS2) nanoparticles: Self-lubricating coatings with potential applications fullerene-like nanoparticles. In Technological Innovations in Sensing and Detection of Chemical, Biological, Radiological, Nuclear Threats and Ecological Terrorism; Springer: Dordrecht, Netherlands, 2012; pp. 59–67. [Google Scholar]
- Rapoport, L.; Fleischer, N.; Tenne, R. Applications of WS2 (MoS2) inorganic nanotubes and fullerene-like nanoparticles for solid lubrication and for structural nanocomposites. J. Mater. Chem. 2005, 15, 1782–1788. [Google Scholar] [CrossRef]
- Drummond, C.; Alcantar, N.; Israelachvili, J.; Tenne, R.; Golan, Y. Microtribology and friction-induced material transfer in WS2 nanoparticle additives. Adv. Funct. Mater. 2001, 11, 348–354. [Google Scholar] [CrossRef]
- Rothschild, A.; Cohen, S.; Tenne, R. WS2 nanotubes as tips in scanning probe microscopy. Appl. Phys. Lett. 1999, 75, 4025–4027. [Google Scholar] [CrossRef]
- Dominko, R.; Arčon, D.; Mrzel, A.; Zorko, A.; Cevc, P.; Venturini, P.; Gaberscek, M.; Remskar, M.; Mihailovic, D. Dichalcogenide nanotube electrodes for li-ion batteries. Adv. Mater. 2002, 14, 1531–1534. [Google Scholar] [CrossRef]
- Cheng, F.Y.; Chen, J.; Gou, X.L. MoS2/Ni Nanocomposites as catalysts for hydrodesulfurization of thiophene and thiophene derivatives. Adv. Mater. 2006, 18, 2561–2564. [Google Scholar] [CrossRef]
- Wang, Y.; Shi, Z.; Huang, Y.; Ma, Y.; Wang, C.; Chen, M.; Chen, Y. Supercapacitor devices based on graphene materials. J. Phys. Chem. 2009, 113, 13103–13107. [Google Scholar]
- Zhu, Y.; Sekine, T.; Brigatti, K.; Firth, S.; Tenne, R.; Rosentsveig, R.; Kroto, H.; Walton, D. Shock-wave resistance of WS2 nanotubes. J. Am. Chem. Soc. 2003, 125, 1329–1333. [Google Scholar]
- Zhu, Y.; Sekine, T.; Li, Y.; Wang, W.; Fay, M.; Edwards, H.; Brown, P.; Fleischer, N.; Tenne, R. WS2 and MoS2 Inorganic fullerenes—super shock absorbers at very high pressures. Adv. Mater. 2005, 17, 1500–1503. [Google Scholar] [CrossRef]
- Zhu, Y.; Sekine, T.; Li, Y.; Fay, M.; Zhao, Y.; Poa, C.; Wang, W.; Roe, M.; Brown, P.; Fleischer, N.; et al. Shock-absorbing and failure mechanisms of WS2 and MoS2 nanoparticles with fullerene-like structures under shock wave pressure. J. Am. Chem. Soc. 2005, 127, 16263–16272. [Google Scholar] [CrossRef]
- Volkova, E.I.; Jones, I.A.; Brooks, R.; Zhu, Y.; Bichoutskaia, E. Meso-scale Modelling of Shock Wave Propagation in a SiC/Al nanocomposite reinforced with WS2-inorganic fullerene nanoparticles. Compos. Struct. 2013, 96, 601–605. [Google Scholar] [CrossRef]
- Tehrani, M.; Luhrs, C.C.; Al-Haik, M.S.; Trevino, J.; Zea, H. Synthesis of WS2 nanostructures from the reaction of WO3 with CS2 and mechanical characterization of WS2 nanotube composites. Nanotechnology 2011, 22, 285714. [Google Scholar] [CrossRef]
- Naffakh, M.; Diez-Pascual, A.M.; Marco, C.; Ellis, G.J.; Gomez-Fatou, M.A. Opportunities and challenges in the use of inorganic fullerene-like Nanoparticles to Produce Advanced Polymer Nanocomposites. Prog. Polym. Sci. 2013, 38, 1163–1231. [Google Scholar] [CrossRef]
- Naffakh, M.; Diez-Pascual, A.M.; Gomez-Fatou, M.A. New Hybrid nanocomposites containing carbon nanotubes, inorganic fullerene-like WS2 nanoparticles and poly(ether ether ketone) (PEEK). J. Mater. Chem. 2011, 21, 7425–7433. [Google Scholar] [CrossRef]
- Naffakh, M.; Diez-Pascual, A.M.; Marco, C.; Gomez, M.A.; Jimenez, I. Novel melt-processable poly (ether ether ketone) (PEEK)/inorganic fullerene-like WS2 nanoparticles for critical applications. J. Phys. Chem. B 2010, 114, 11444–11453. [Google Scholar] [CrossRef]
- Diez-Pascual, A.M.; Naffakh, M.; Marco, C.; Ellis, G. Rheological and tribological properties of carbon nanotube/thermoplastic nanocomposites incorporating inorganic fullerene-Like WS2 Nanoparticles. J. Phys. Chem. 2012, 116, 7959–7969. [Google Scholar] [CrossRef]
- Diez-Pascual, A.M.; Naffakh, M. Mechanical and thermal behaviour of isotactic polypropylene reinforced with inorganic fullerene-like WS2 nanoparticles: Effect of Filler Loading and Temperature. Mater. Chem. Phys. 2013, 141, 979–989. [Google Scholar] [CrossRef]
- Ma, T.; Zhang, T.; Gao, P.G.; Zhang, J.C. Synthesis and properties of ultrahigh molecular weight polyethylene/WS2 nanoparticle fiber for bullet-proof materials. Chin. Sci. Bull. 2013, 58, 945–948. [Google Scholar] [CrossRef]
- Shneider, M.; Dodiuk, H.; Tenne, R.; Kenig, S. Nanoinduced morphology and enhanced properties of epoxy containing tungsten disulfide nanoparticles. Polym. Eng. Sci. 2013, 53, 2624–2632. [Google Scholar] [CrossRef]
- Shneider, M.; Rapoport, L.; Moshkovich, A.; Dodiuk, H.; Kenig, S.; Tenne, R.; Zak, A. Tribological performance of the epoxy-based composite reinforced by WS2 fullerene-like nanoparticles and nanotubes. Phys. Status Solidi A 2013, 210, 2298–2306. [Google Scholar] [CrossRef]
- Zohar, E.; Baruch, S.; Shneider, M.; Dodiuk, H.; Kenig, S.; Tenne, R.; Wagner, H.D. The Effect of WS2 nanotubes on the properties of epoxy-based nanocomposites. J. Adhes. Sci. Technol. 2011, 25, 1603–1617. [Google Scholar] [CrossRef]
- Shneider, M.; Dodiuk, H.; Kenig, S.; Tenne, R. The Effect of Tungsten Sulfide Fullerene-Like Nanoparticles on the toughness of epoxy adhesives. J. Adhes. Sci. Technol. 2010, 24, 1083–1095. [Google Scholar] [CrossRef]
- Lalwani, G.; Kwaczala, A.T.; Kanakia, S.; Patel, S.C.; Judex, S.; Sitharaman, B. Fabrication and characterization of three-dimensional macroscopic all-carbon scaffolds. Carbon 2013, 53, 90–100. [Google Scholar] [CrossRef]
- Franklin, N.; Dai, H. An enhanced CVD approach to extensive nanotube networks with directionality. Adv. Mater. 2000, 12, 890–894. [Google Scholar] [CrossRef]
- Calvo, M.; Garcia, R.; Arenillas, A.; Suarez, I.; Moinelo, S. Carbon foams from coals. A preliminary study. Fuel 2005, 84, 2184–2189. [Google Scholar] [CrossRef]
- Calvo, M.; Garcia, R.; Moinelo, S.R. Carbon foams from different coals. Energy Fuels 2008, 22, 3376–3383. [Google Scholar] [CrossRef]
- Chen, C.; Kennel, E.; Stiller, A.; Stansberry, P.; Zondlo, J. Carbon foam derived from various precursors. Carbon 2006, 44, 1535–1543. [Google Scholar] [CrossRef]
- Chakrapani, N.; Wei, B.; Carrillo, A.; Ajayan, P.; Kane, R. Capillarity-driven assembly of two-dimensional cellular carbon nanotube foams. Proc. Natl. Acad. Sci. USA 2004, 101, 4009–4012. [Google Scholar] [CrossRef]
- Chakrapani, N.; Wei, B.; Carrillo, A.; Ajayan, P.; Kane, R. Capillarity-driven assembly of cellular carbon nanotube foams. Abstr. Pap. Am. Chem. Soc. 2004, 228, U481–U481. [Google Scholar]
- Gallego, N.; Klett, J. Carbon Foams for Thermal Management. Carbon 2003, 41, 1461–1466. [Google Scholar] [CrossRef]
- Kaur, S.; Ajayan, P.M.; Kane, R.S. Design and characterization of three-dimensional carbon nanotube foams. J. Phys. Chem. B 2006, 110, 21377–21380. [Google Scholar] [CrossRef]
- Dai, L.; Patil, A.; Gong, X.; Guo, Z.; Liu, L.; Liu, Y.; Zhu, D. Aligned nanotubes. Chemphyschem 2003, 4, 1150–1169. [Google Scholar] [CrossRef]
- Atwater, M.A.; Mousavi, A.K.; Leseman, Z.C.; Phillips, J. Direct synthesis and characterization of a nonwoven structure comprised of carbon nanofibers. Carbon 2013, 57, 363–370. [Google Scholar] [CrossRef]
- Safdari, M.; Al-Haik, M.S. Optimization of stress wave propagation in a multilayered elastic/viscoelastic hybrid composite based on carbon fibers/carbon nanotubes. Polym. Compos. 2012, 33, 196–206. [Google Scholar] [CrossRef]
- Khalid, W.; Ali, M.S.M.; Dahmardeh, M.; Choi, Y.; Yaghoobi, P.; Nojeh, A.; Takahata, K. High-aspect-ratio, free-form patterning of carbon nanotube forests using micro-electro-discharge machining. Diamond Relat. Mater. 2010, 19, 1405–1410. [Google Scholar] [CrossRef]
- Qiu, L.; Liu, J.Z.; Chang, S.L.Y.; Wu, Y.; Li, D. Biomimetic superelastic graphene-based cellular monoliths. Nat. Commun. 2012, 3, 1241. [Google Scholar] [CrossRef]
- Geim, A.K.; Novoselov, K.S. The rise of graphene. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef]
- Stankovich, S.; Dikin, D.A.; Dommett, G.H.B.; Kohlhaas, K.M.; Zimney, E.J.; Stach, E.A.; Piner, R.D.; Nguyen, S.T.; Ruoff, R.S. Graphene-based composite materials. Nature 2006, 442, 282–286. [Google Scholar] [CrossRef]
- Naffakh, M.; Diez-Pascual, A.M.; Marco, C.; Ellis, G. Morphology and thermal properties of novel poly (phenylene sulfide) hybrid nanocomposites based on single-walled carbon nanotubes and inorganic fullerene-like WS2 nanoparticles. J. Mater. Chem. 2012, 22, 1418–1425. [Google Scholar] [CrossRef]
- Diez-Pascual, A.M.; Naffakh, M.; Gomez-Fatou, M.A. Mechanical and electrical properties of novel poly(ether ether ketone)/carbon nanotube/inorganic fullerene-like WS2 hybrid nanocomposites: Experimental measurements and theoretical predictions. Mater. Chem. Phys. 2011, 130, 126–133. [Google Scholar] [CrossRef]
- Diez-Pascual, A.M.; Naffakh, M. Inorganic Nanoparticle-Modified Poly(Phenylene Sulphide)/Carbon Fiber Laminates: Thermomechanical Behaviour. Materials 2013, 6, 3171–3193. [Google Scholar] [CrossRef]
- Diez-Pascual, A.M.; Naffakh, M. Tuning the properties of carbon fiber-reinforced poly (phenylene sulphide) laminates via incorporation of inorganic nanoparticles. Polymer 2012, 53, 2369–2378. [Google Scholar] [CrossRef]
- Diez-Pascual, A.M.; Naffakh, M.; Marco, C.; Ellis, G. Mechanical and electrical properties of carbon nanotube/poly(phenylene sulphide) composites incorporating polyetherimide and inorganic fullerene-like nanoparticles. Compos. Part A 2012, 43, 603–612. [Google Scholar] [CrossRef]
- Luhrs, C.C.; Daskam, C.D.; Gonzalez, E.; Phillips, J. Fabrication of a Low Density Carbon Fiber Foam and Its Characterization as a Strain Gauge. Materials submitted for publication. 2014. [Google Scholar]
- Moberg, M. Carbon Fiber and Tungsten Disulfide Nanoscale Architectures for Armor Applications. MS Thesis, Naval Postgraduate School, June 2012. [Google Scholar]
- Feldman, Y.; Frey, G.; Homyonfer, M.; Lyakhovitskaya, V.; Margulis, L.; Cohen, H.; Hodes, G.; Hutchison, J.; Tenne, R. Bulk synthesis of inorganic fullerene-like MS2 (M = Mo, W) from the respective trioxides and the reaction mechanism. J. Am. Chem. Soc. 1996, 118, 5362–5367. [Google Scholar] [CrossRef]
- Tenne, R.; Homyonfer, M.; Feldman, Y. Nanoparticles of layered compounds with hollow cage structures (inorganic fullerene-like structures). Chem. Mater. 1998, 3225–3238. [Google Scholar]
- Tenne, R.; Remskar, M.; Enyashin, A.; Seifert, G. Inorganic nanotubes and fullerene-like structures (IF). Carbon Nanotubes 2008, 111, 631–671. [Google Scholar]
- Bar-Sadan, M.; Kaplan-Ashiri, I.; Tenne, R. Inorganic fullerenes and nanotubes: Wealth of materials and morphologies. Eur. Phys. J.-Spec. Top. 2007, 149, 71–101. [Google Scholar] [CrossRef]
- Wiesel, I.; Arbel, H.; Albu-Yaron, A.; Popovitz-Biro, R.; Gordon, J.M.; Feuermann, D.; Tenne, R. Synthesis of WS2 and MoS2 Fullerene-Like Nanoparticles from Solid Precursors. Nano Res. 2009, 2, 416–424. [Google Scholar] [CrossRef]
- Bastide, S.; Borra, J.; Duphil, D.; Levy-Clement, C. Synthesis of inorganic fullerenes and nanoboxes of MoS2 and WS2 by spray pyrolysis. Abstr. Pap. Am. Chem. Soc. 2004, 228, U482–U482. [Google Scholar]
- Zink, N.; Pansiot, J.; Kieffer, J.; Therese, H.A.; Panthofer, M.; Rocker, F.; Kolb, U.; Tremel, W. Selective synthesis of hollow and filled fullerene-like (IF) WS2 nanoparticles via metal-organic chemical vapor deposition. Chem. Mater. 2007, 6391–6400. [Google Scholar]
- Brooks, D.; Douthwaite, R.; Brydson, R.; Calvert, C.; Measures, M.; Watson, A. Synthesis of inorganic fullerene (MS2, M = Zr, Hf and W) phases using H2S and N2/H2 microwave-induced plasmas. Nanotechnology 2006, 17, 1245–1250. [Google Scholar] [CrossRef]
- Tenne, R. Inorganic nanotubes and fullerene-like nanoparticles. Nat. Nanotechnol. 2006, 1, 103–111. [Google Scholar] [CrossRef]
- Luhrs, C.C.; Garcia, D.; Tehrani, M.; Al-Haik, M.; Taha, M.R.; Phillips, J. Generation of carbon nanofilaments on carbon fibers at 550 degrees C. Carbon 2009, 47, 3071–3078. [Google Scholar] [CrossRef]
- Pierson, H.O. Handbook of Carbon, Graphite, Diamond, and Fullerenes; Noyes Publications: Park Ridge, NJ, USA, 1993. [Google Scholar]
- Fleet, M.E. Crystal-Structure of Heazlewoodite, and Metallic Bonds in Sulfide Minerals. Am. Mineral. 1977, 62, 341–345. [Google Scholar]
- Parise, J.B. Structure of Hazelwoodite (Ni3S2). Acta Crystallogr. Sect. B 1980, 36, 1179–1180. [Google Scholar] [CrossRef]
- Villars, P.; Cenzual, K. Landolt-Bornstein—Group III Condensed Matter; Springer: Berlin, Germany, 2010. [Google Scholar]
- Xu, F.; Almeida, T.P.; Chang, H.; Xia, Y.; Wears, M.L.; Zhu, Y. Multi-walled carbon/IF-WS2 nanoparticles with improved thermal properties. Nanoscale 2013, 5, 10504–10510. [Google Scholar] [CrossRef]
- Schuffenhauer, C.; Wildermuth, G.; Felsche, J.; Tenne, R. How stable are inorganic fullerene-like particles. Thermal analysis (STA) of inorganic fullerene-like NbS2, MoS2, and WS2 in oxidizing and inert atmospheres in comparison with the bulk material. Phys. Chem. Chem. Phys. 2004, 6, 3991–4002. [Google Scholar] [CrossRef]
- Chang, L.; Yang, H.; Fu, W.; Yang, N.; Chen, J.; Li, M.; Zou, G.; Li, J. Synthesis and thermal stability of W/WS2 inorganic fullerene-like nanoparticles with core-shell structure. Mater. Res. Bull. 2006, 41, 1242–1248. [Google Scholar] [CrossRef]
- Dutta, A.; Penumadu, D.; Files, B. Nanoindentation testing for evaluating modulus and hardness of single-walled carbon nanotube-reinforced epoxy composites. J. Mater. Res. 2004, 19, 158–164. [Google Scholar] [CrossRef]
- Gromowski, K. Glass Breakage—Nickel Sulfide Inclusions; Penn State: State College, PA, USA, 2010. [Google Scholar]
- Flores, A.; Naffakh, M.; Diez-Pascual, A.M.; Ania, F.; Gomez-Fatou, M.A. Evaluating the Reinforcement of Inorganic Fullerene-like Nanoparticles in Thermoplastic Matrices by Depth-Sensing Indentation. J. Phys. Chem. C 2013, 117, 20936–20943. [Google Scholar] [CrossRef]
- Marcano, D.C.; Kosynkin, D.V.; Berlin, J.M.; Sinitskii, A.; Sun, Z.; Slesarev, A.; Alemany, L.B.; Lu, W.; Tour, J.M. Improved Synthesis of Graphene Oxide. ACS Nano 2010, 4, 4806–4814. [Google Scholar] [CrossRef]
- Oliver, W.; Pharr, G. An Improved Technique for Determining Hardness and Elastic-Modulus using Load and Displacement Sensing Indentation Experiments. J. Mater. Res. 1992, 7, 1564–1583. [Google Scholar] [CrossRef]
- Pharr, G.; Oliver, W.; Brotzen, F. On the Generality of the Relationship among Contact Stiffness, Contact Area, and Elastic-Modulus during Indentation. J. Mater. Res. 1992, 7, 613–617. [Google Scholar] [CrossRef]
- Hay, J.; Herbert, E. Measuring the Complex Modulus of Polymers by Instrumented Indentation Testing. Exp. Tech. 2013, 37, 55–61. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Luhrs, C.C.; Moberg, M.; Maxson, A.; Brewer, L.; Menon, S. IF-WS2/Nanostructured Carbon Hybrids Generation and Their Characterization. Inorganics 2014, 2, 211-232. https://doi.org/10.3390/inorganics2020211
Luhrs CC, Moberg M, Maxson A, Brewer L, Menon S. IF-WS2/Nanostructured Carbon Hybrids Generation and Their Characterization. Inorganics. 2014; 2(2):211-232. https://doi.org/10.3390/inorganics2020211
Chicago/Turabian StyleLuhrs, Claudia C., Michael Moberg, Ashley Maxson, Luke Brewer, and Sarath Menon. 2014. "IF-WS2/Nanostructured Carbon Hybrids Generation and Their Characterization" Inorganics 2, no. 2: 211-232. https://doi.org/10.3390/inorganics2020211