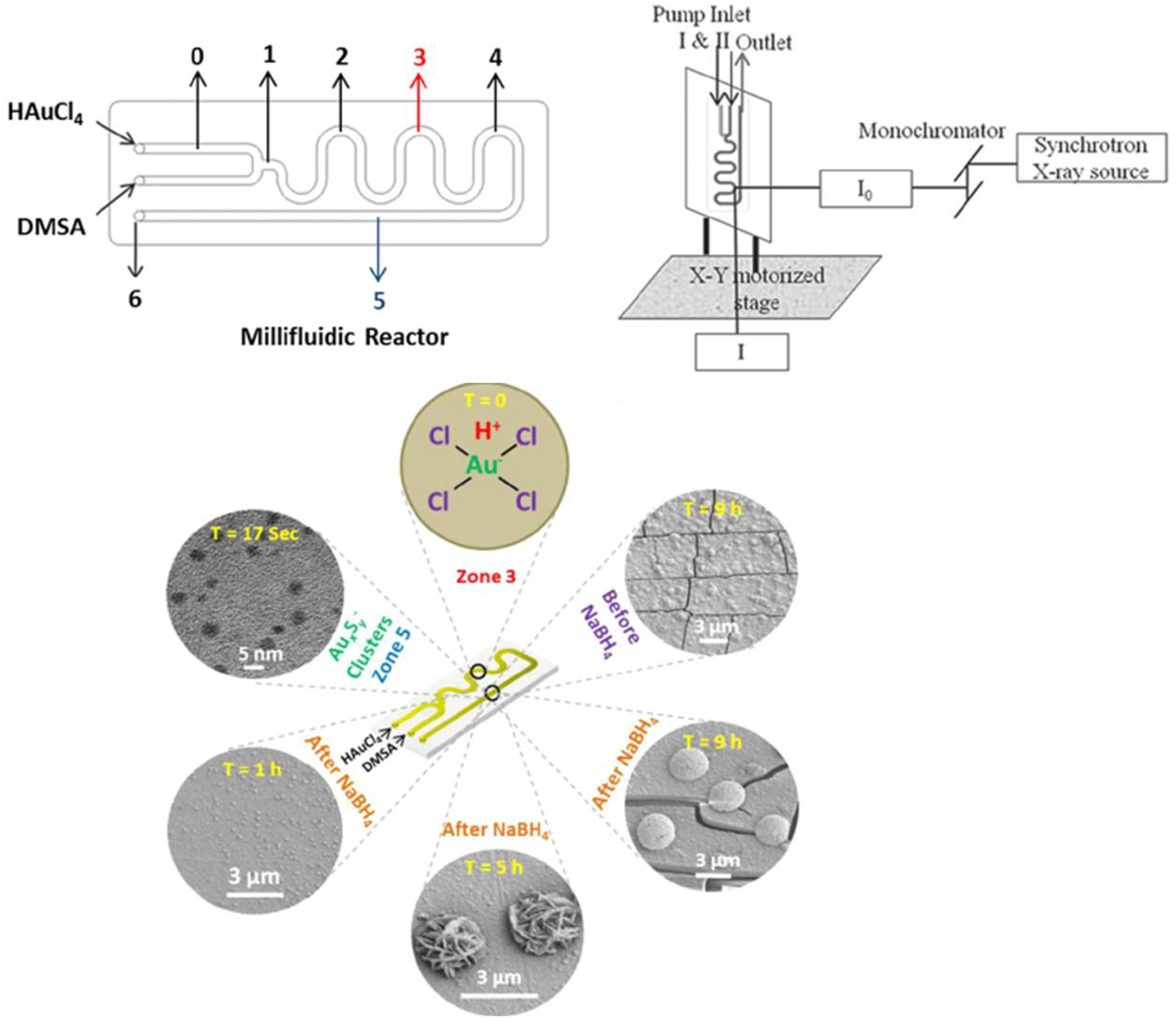
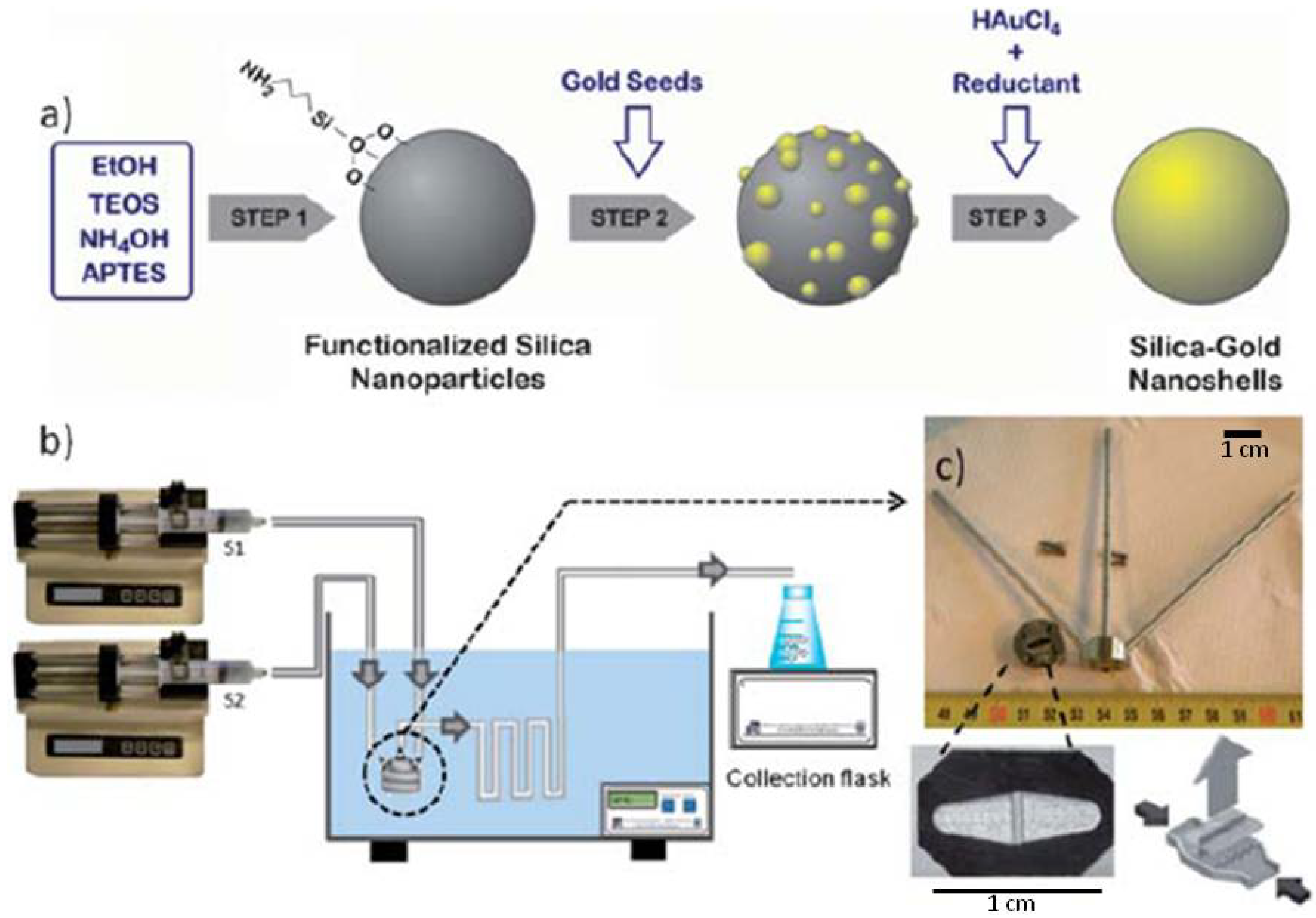
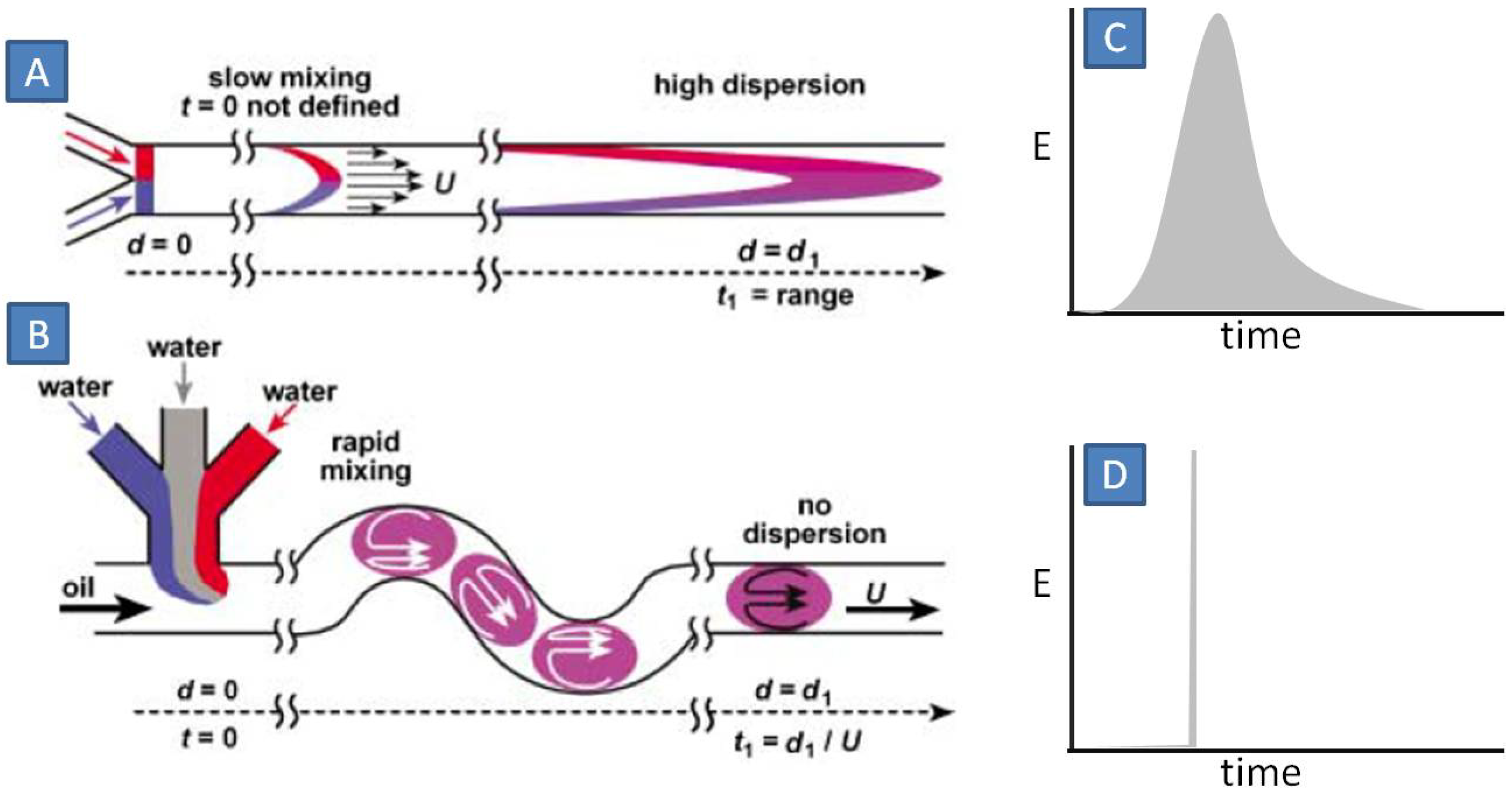
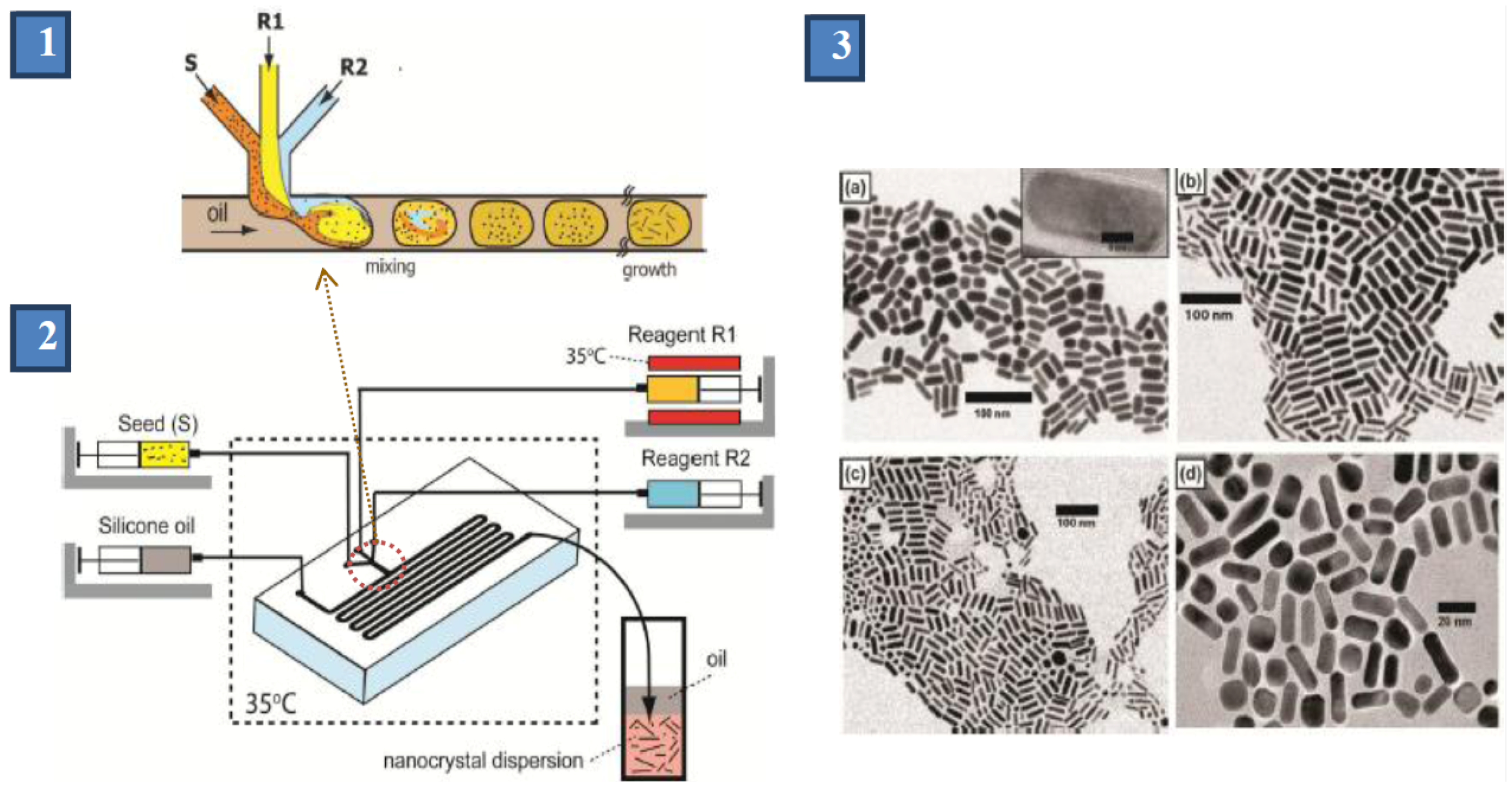
1. Introduction: Gold Nanomaterials—Syntheses and Applications
Gold is one of the most important elements in the human civilization. In early human societies, gold was a symbol of power and wealth, and was used as ornaments for both living and the dead. Gold has been, ever since the very dawn of commerce, a reliable and universal means of financial transactions and reserve. Hence, the desire and dream of “transmuting anything into gold” paved the way for alchemy, which is the predecessor of modern Chemistry. Rather unknowingly, non-bulk form of gold had also been a part of humanity from the time of antiquity. Colloidal gold dispersed in glass or ceramics produce hues of red or green, which were used to decorate and color Roman cage-cups, or diatretum. One famous example is the Lycurgus cup, dating from the 4th century A.D., made from nanogold-infested glass, now preserved in the British Museum in London.
Although widely used throughout the history for decorative purposes and as medicines [
1,
2], the nature of colloidal gold was investigated only sporadically [
3]: In 1676, a German chemist, Johann Kunckels, inferred that non-bulk gold exists in infinitesimal state that is not visible to human eyes. It was Michael Faraday who pioneered the first systematic synthesis of gold nanoparticles using phosphorous-based reducing agents. He is widely credited for insightful and for the “first” scientific discussion on size dependent optical properties and their coagulation behavior of colloidal gold [
4].
Rapid growth in interests about colloidal gold was sparked by the availability of electron microscopes that enabled direct visualization of the nanoparticles [
5,
6]. Over the past two decades, tremendous effort was dedicated to synthesize and optimize a wide class of GNPs possessing a broad spectrum of geometry, size, chemistry and functionality. This paved the way for applications of GNPs in heterogeneous catalysis, bio-imaging, medicines, optics, analytical sciences, sensing,
etc. [
7,
8,
9,
10,
11,
12,
13,
14,
15,
16,
17]. Because of widespread applications of GNPs in different branches of sciences and technologies, need for establishing reliable supply chains for such materials is becoming ever more important, hence the involved chemical processes are of utmost importance.
4. Synthesis of Colloidal Gold Nanoparticles in Microreactors
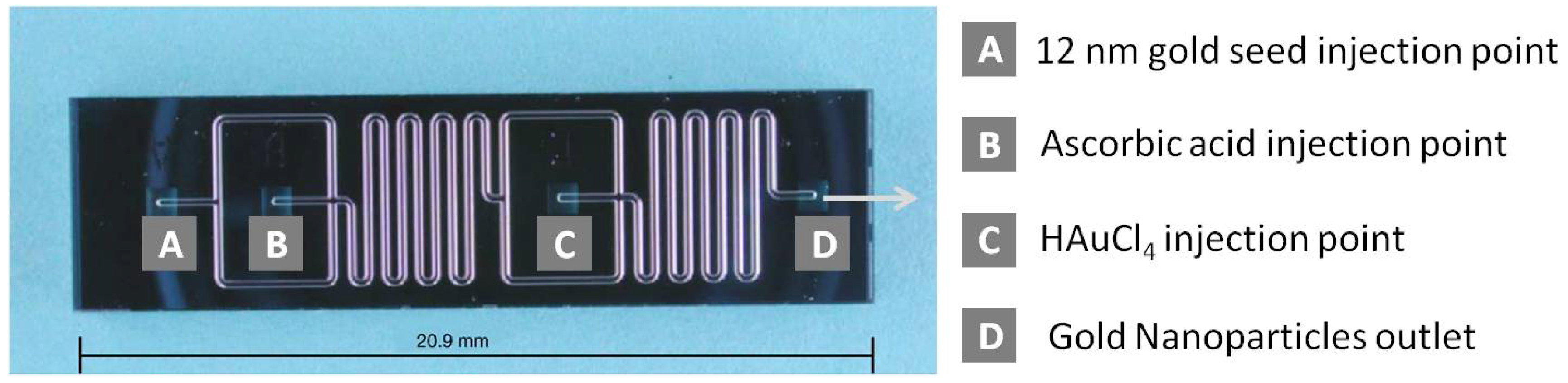
Wagner
et al. were the first to report synthesis of gold nanoparticles in a microreactor [
27]. They used a microfluidic chip fabricated from Pyrex glass by wet etching method. The chip consisted of two micromixers and a residence time unit (
Figure 1). In the first micromixer, a solution of ascorbic acid, acting as a reducing agent, was mixed with preformed gold seeds of 12 nm in diameter. This mixture was guided through a serpentine channel to complete mixing. Then, reduction of the gold ions onto the good seeds started in the second micromixer with a volume of 2.3 µL where a solution of chloroauric acid was added to the flow to keep the total flow rate in the range of 5–50 µL/min. The reaction mixture was collected from the reactor outlet via a PTFE tube and analyzed by spectroscopic and microscopic methods.
The channel walls were negatively charged at the pH of 10, used in all experiments. When citrate ions were used as capping agent, the surface of gold nanoparticles was also negatively charged. Therefore electrostatic repulsion prevented seed deposition onto the inner channel walls. However, when the seeds were grown in the presence of ascorbic acid, significant gold deposition on the walls was observed. This undesirable deposition was reduced by increasing the seed to gold ion ratio in the solution. At the 1:1 molar ratio, gold particles with a size of 24 nm were obtained, albeit with poor monodispersity. The size was further controlled by varying the respective flow rates of the reactants and/or the reducing agents.
Figure 1.
Microfluidic device for GNP synthesis. Adapted from Ref. [
27] (with permission from Elsevier Ltd.
Figure 1.
Microfluidic device for GNP synthesis. Adapted from Ref. [
27] (with permission from Elsevier Ltd.
In the absence of seeds in the synthesis with ascorbic acid, both high pH of the reaction mixture and highly hydrophobic surface of the inner reactor walls prevented the nanoparticle deposition onto the inner walls of a pyrex/silicone microreactor [
28]. A principle of split-and-recombine mixing was utilized in this microreactor. The hydrophobic surface was created by modification with perfluorosilane and polyvinylpyrrolidone (PVP) was used as capping agent. As the flow rate increased, the mean diameter of the gold nanoparticles decreased, while the total number of nanoparticles increased. It has been shown that there exists a range of flow rates, where axial dispersion effects can be neglected leading to improved control over mixing. The obtained gold nanoparticles showed better distribution, which was twice as narrow in comparison to those obtained in the conventional reactor and in the microreactor with a substantial degree of backmixing observed at low flow rates. In a follow up work, controlled accumulation of the discrete nanoparticles into larger clusters of micrometer size was demonstrated in the channels of 180 × 200 µm
2 made in a glass chip [
29]. In this reactor, a solution of reducing agents (ascorbic acid and iron(II) sulfate) was mixed with polyvinyl alcohol in the first mixer. Then, a sodium metasilicate solution was introduced in the second mixer and gold ions were injected in the third mixer. Reduction of gold ions into colloidal gold in the presence of polymer and electrolytes facilitated the clustering of gold nanoparticles into larger clusters. Small GNPs with a diameter of 3–5 nm were obtained when sodium borohydride was used as reducing agent [
30]. The size can further be fine-tuned by controlling the concentration (via dilution by introducing diluents water stream) of the gold ions and borohydride. Thus, the authors were able to control the cluster size between 3 nm up to micrometer size by varying the respective flow rates and concentration.
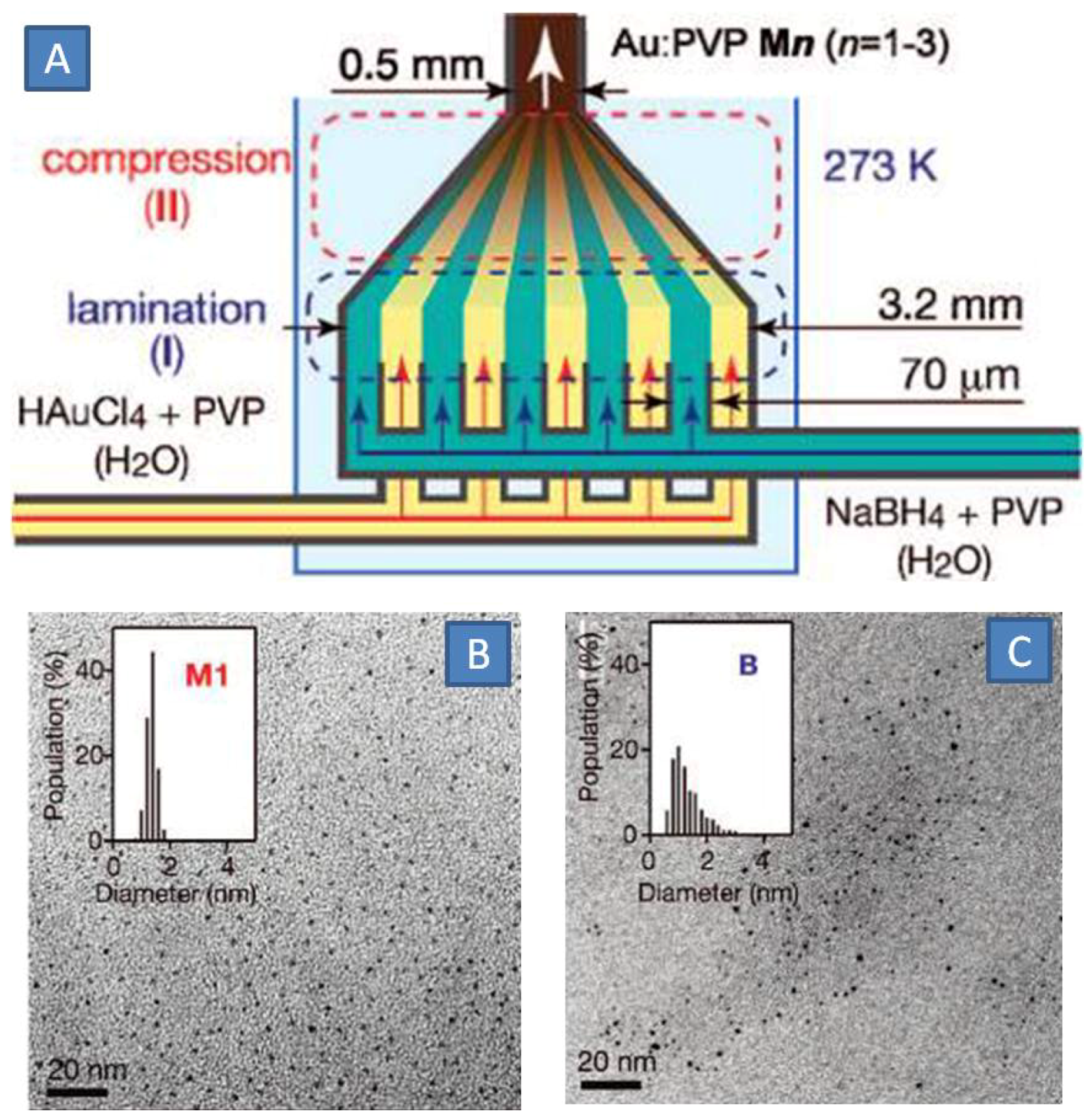
Ultra-small gold clusters with a diameter of 1 nm are an important class of gold nanomaterials with regards to their high catalytic activity in many organic reactions [
31]. Synthesis of such gold clusters is a difficult task in conventional stirred reactors due to pure control of mixing. Deviations of the local concentration of reactants, particularly strong reducing agents like sodium borohydride, influence the kinetics of the cluster formation and are detrimental to the size distribution. Tsunoyama
et al. presented a microfluidic method to produce ultra-small PVP-stabilized gold nanoparticles using a SIMM-V2 micromixer (IMM,
Figure 2A) [
32]. In this micromixer, the total flow of HAuCl
4 gold precursor, PVP and an aqueous solution of sodium borohydride was split in a multitude of parallel substreams with a thickness below 100 µm. This reduced characteristic diffusion time from several minutes to a few seconds. Moreover, it was speculated that microbubbles, which were formed due to the decomposition of borohydride, assisted in breaking the lamellar structure into smaller fragments, further accelerating the mass-transport. Sub 2 nm gold nanoparticles were produced with a monodispersity of 14% from a 10 mM Au precursor solution at a relatively low PVP:Au molar ratio of 40:1
Figure 2B). It should be noted that larger and polydisperse PVP-protected Au clusters were produced in a conventional reactor, when similar concentrations of reactants were used (
Figure 2C). The activity of Au clusters in oxidation of 4-hydroxybenzyl alcohol was 50% greater than that with Au clusters produced by classical batch protocol demonstrating the advantages of monodispersed Au nanoparticles in structure sensitive reactions.
Figure 2.
(
A) Multilamination and mixing of gold precursors (Au ions and PVP) with reducing agent (sodium borohydride and PVP); (
B,
C) TEM photographs of produced GNPs with size distribution (insets) for microfluidic and batch processes, respectively. Adapted from Ref. [
32] (with permission from American Chemical Society).
Figure 2.
(
A) Multilamination and mixing of gold precursors (Au ions and PVP) with reducing agent (sodium borohydride and PVP); (
B,
C) TEM photographs of produced GNPs with size distribution (insets) for microfluidic and batch processes, respectively. Adapted from Ref. [
32] (with permission from American Chemical Society).
The SIMM-V2 micromixer in combination with a microreactor was employed to examine the dependence of the particles size on the reactant flow rates [
33]. The authors used HAuCl
4 as gold source, while ascorbic acid and PVP were used as reducing agent and surfactant, respectively. They obtained a set of GNPs with different sizes in the range from 1.7 to 33 nm by varying the flow rate from 7.5 to 0.1 mL/min. The characteristic half time of ascorbic acid mediated reduction of gold ions was found to be 31 ms. Still, this is much shorter as compared to the characteristic diffusion time of at least several seconds. As the diffusion time approaches the characteristic reaction time, it enables to decouple the nucleation and growth steps, which in turn provides better degree of control over the particle size via variation of the residence time. The authors did not observe deposition of on the inner metal walls of the micromixer at high flow rates.
5. Effect of pH
Ishizaka
et al. adjusted the pH of a gold precursor solution with addition of an aqueous sodium hydroxide solution in a Teflon mixer connected to a capillary [
34]. This solution was then mixed with glucose acting as reducing agent. As the hydroxide flow rate decreased, the pH of the resulting solution decreased too yielding non-spherical GNPs as detected by a blue shift in the light absorption (
Figure 3). At a pH of 6.9, star-like GNPs were obtained solely in the microreactor, while in their batch system this morphology could not be obtained at the same reaction conditions.
Figure 3.
Upper panel: Microfluidic pH Control of Gold solution with NaOH and then online reduction with glucose. Lower panel: GNP samples of wide UV absorption characteristics (depending on shape); TEM photograph of star-like GNP (while line at the bottom left of TEM photo corresponds to scale bar, in addition to the bottom right side, unaltered from the original image). Adapted from Ref. [
34] (with permission from Elsevier Ltd.).
Figure 3.
Upper panel: Microfluidic pH Control of Gold solution with NaOH and then online reduction with glucose. Lower panel: GNP samples of wide UV absorption characteristics (depending on shape); TEM photograph of star-like GNP (while line at the bottom left of TEM photo corresponds to scale bar, in addition to the bottom right side, unaltered from the original image). Adapted from Ref. [
34] (with permission from Elsevier Ltd.).
It is known that the reducing potential of ascorbic acid depends on the pH of the solution. At pH below 4.1, ascorbic acid exists in the protonated form, AscH
2. As the pH increases, it transforms into ascorbate, AscH
−, while at pH above 11.6 it is completely deprotonated to form, Asc
2−. Such speciation of ascorbic acid at different pH alters its reducing power. The control of the initial pH of the ascorbic acid solution increased the isolation of spherical GNPs, with high monodispersity [
35]. Au nanoparticles with average diameter of 18, 10 and 7 nm, with narrow relative polydispersity indices of 0.4, 0.3 and 0.2 were obtained from a HAuCl
4/ascorbic acid mixture at a pH of 10.2, 10.7 and 11.1, other parameters being the same. The initial pH was adjusted with a sodium hydroxide solution. A microfluidic mixer was operated at Reynolds number of 2000, enabling intense mixing between ascorbic acid and gold precursors.
6. Transient Operation
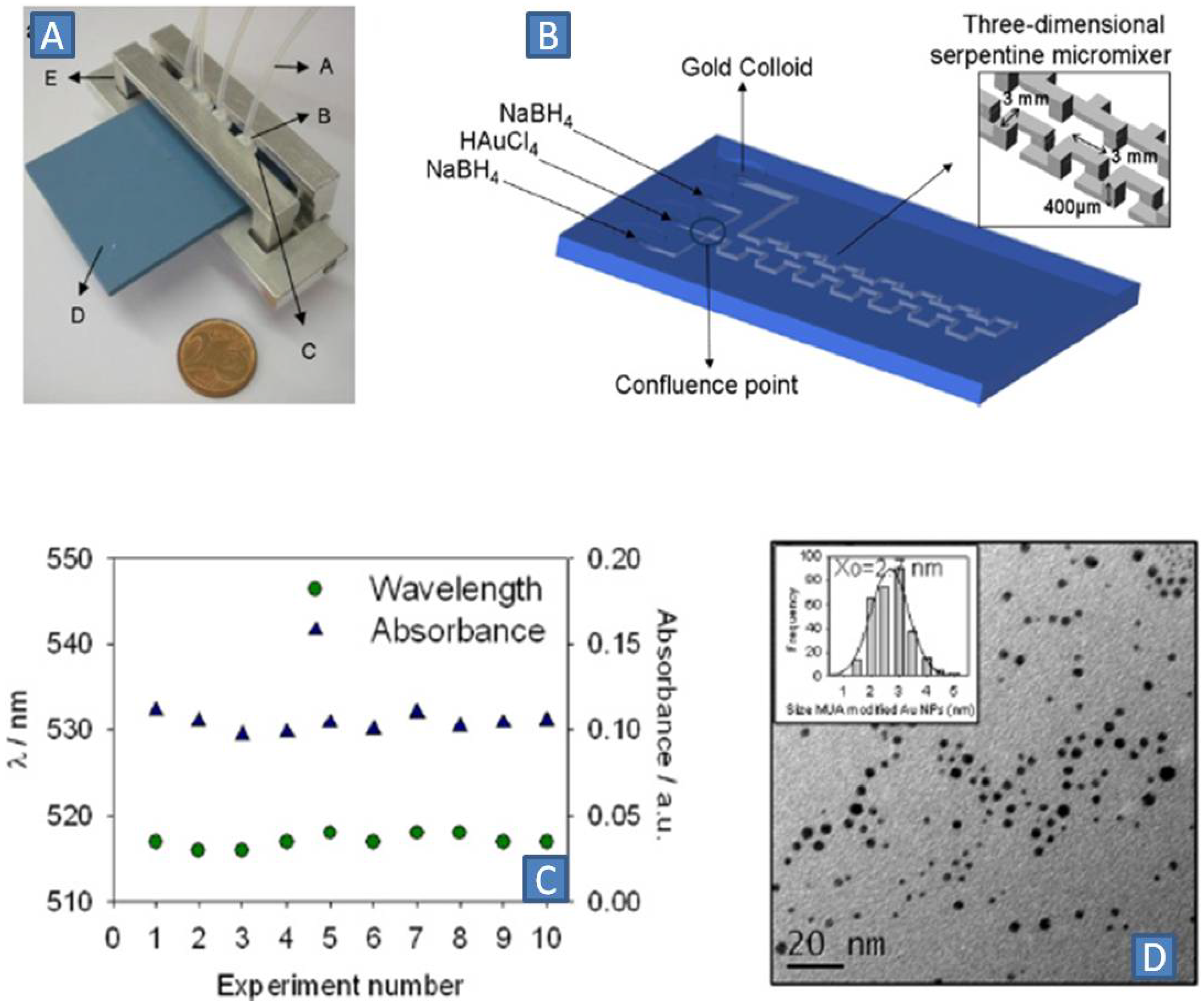
A pulsed dosage of 0.5 µL of a 1 mM Au(III) solution (every 2 s) to a continuous flow of a 1.5 mM sodium borohydride solution was proposed by Pedro
et al. [
36]. The two solutions were mixed in a flow-focusing T-junction connected to a three-dimensional serpentine channel network (
Figure 4A,B). This enabled controlled and homogeneous formation of the gold seed/nuclei due to the creation of supersaturated zones in each dosed volume of gold ions. The entire microreactor was made by low-temperature co-fired ceramic technology (LTCC). Highly reproducible 2.7 nm GNPs with high monodispersity were obtained in at least 10 consecutive runs (
Figure 4C,D).
Figure 4.
(
A) Microfluidic device made by low-temperature co-fired ceramic technology LTCC; (
B) Micromixer and 3-D serpentine residence time unit enabling efficient mixing; (
C) Reproducibility between several runs of experiment; (
D) TEM photograph: Size-distribution of GNPs in the form of histogram is provided in the inset; mean average diameter is 2.7 nm for the Mercaptoundecanoic acid MUA modified gold nanoparticles. Adapted from Ref. [
36] (with permission from IOP Publishing Ltd.).
Figure 4.
(
A) Microfluidic device made by low-temperature co-fired ceramic technology LTCC; (
B) Micromixer and 3-D serpentine residence time unit enabling efficient mixing; (
C) Reproducibility between several runs of experiment; (
D) TEM photograph: Size-distribution of GNPs in the form of histogram is provided in the inset; mean average diameter is 2.7 nm for the Mercaptoundecanoic acid MUA modified gold nanoparticles. Adapted from Ref. [
36] (with permission from IOP Publishing Ltd.).
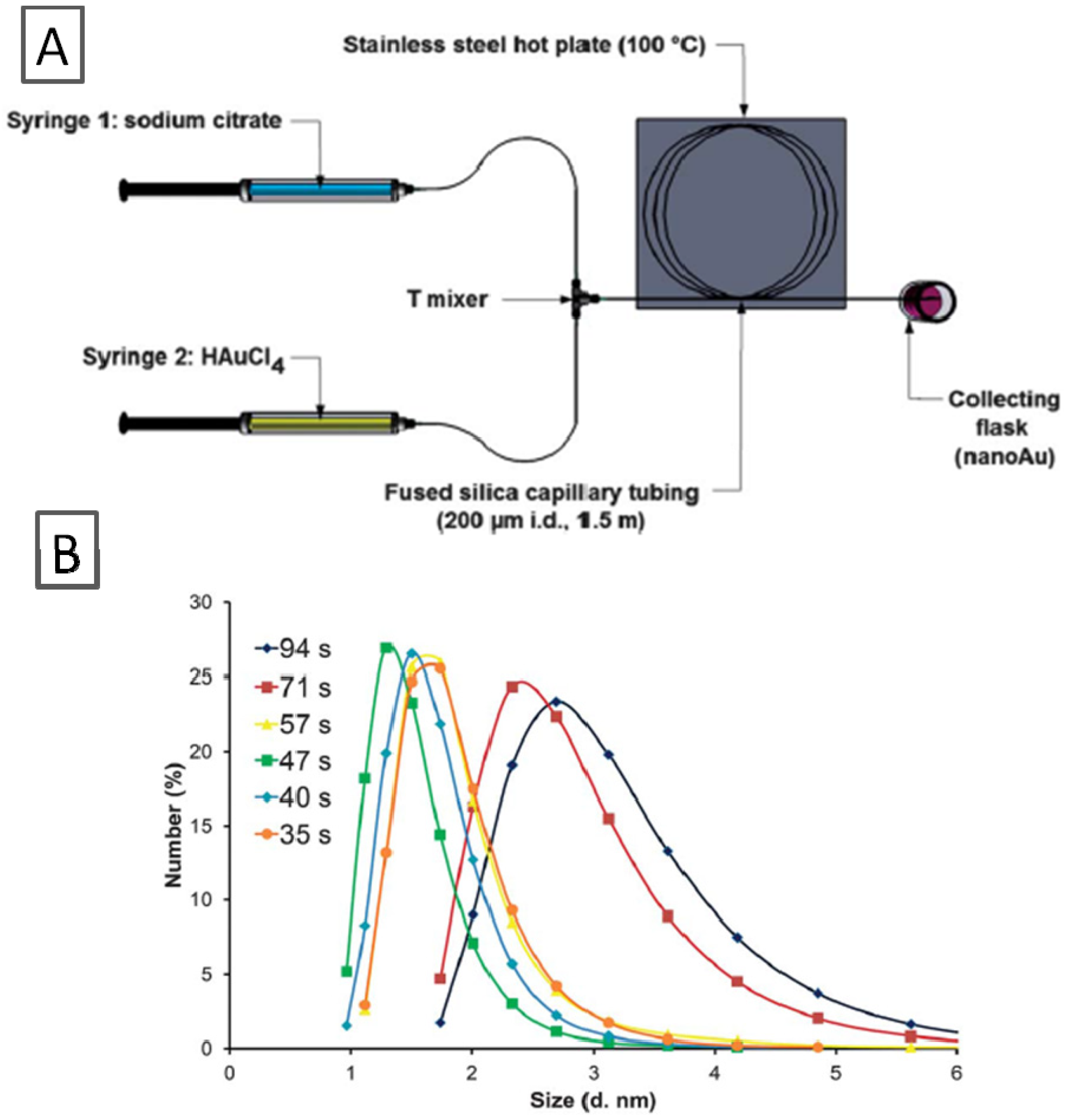
Ultra-small Au nanoparticles can also be obtained by rapid heating and quench in a microreactor system [
37]. In this method, the Au ions are reduced with a sodium citrate solution at elevated temperature, just below the boiling point of the solvent, yielding a multitude of nuclei that needs to be immediately cooled to arrest further growth. A T-mixer was connected to a 1.5 m fused silica capillary with a diameter of 200 µm maintained at 100 °C (
Figure 5A). The outlet flow was rapidly cooled to quench the reaction. With a residence time of 35 s, gold nanoparticles with an average diameter of 2 nm were produced at a citrate: Au molar ratio of 3.5 (Au 5.4 mM), while the synthesis in batch gave a polydispersed mixture of gold nanoparticles under similar reaction conditions. Different particle sizes in the range of 1.5–3.0 nm were obtained by varying the residence time in the microreactor (
Figure 5B). Thus, fast heating enabled to obtain ultra-small GNPs even with classical synthesis methods.
Figure 5.
(
A) T-mixer and Fused Silica capillary microreactor; (
B) GNP Size variation and size distribution by tuning residence time. Adapted from Ref. [
37] (with permission from Royal Society of Chemistry).
Figure 5.
(
A) T-mixer and Fused Silica capillary microreactor; (
B) GNP Size variation and size distribution by tuning residence time. Adapted from Ref. [
37] (with permission from Royal Society of Chemistry).
7. Surfactant Stabilized Gold Nanoparticles
While many reports are devoted towards preparing water-dispersible GNPs colloids in microfluidic reactors, less attention was given to organically-capped hydrophobic gold nanoparticles which are also an important class of GNPs. Sugie
et al. reported reduction of HAuCl
4 in tetrahydrofuran (THF) solvent inside a Y-mixer/PTFE microreactor assembly maintained at constant temperature (25–60 °C) using organosilanes (RSiH) as reducing agents and alkyl thiol (RSH) as capping agent [
38]. The effect of the residence time and temperatures was studied. As the residence time increased from 12 to 94 min, the size of the GNPs increased from 4.3 to 8.7 nm. The nature of the reducing agent did not alter the size of GNPs when the flow rate and temperature were kept constant. A benchmark experiment in the batch mode at 60 °C produced insoluble precipitates manifesting uncontrolled growth.
11. Au Nanorods
Gold nanorods (GNRs) is another class of gold nanoparticles, which show surface plasmon behavior [
8]. In particular, they can support a longitudinal surface plasmon, which results in suspensions of them having a strong extinction peak in the upper visible or near-infrared parts of the spectrum. The position of this peak can be readily tuned by controlling the shape of the rods. In addition, the surface of the nanorods can be functionalized by a very wide variety of molecules. This has led to interest in their use as selective biomarkers in biodiagnostics or for selective targeting in photothermal therapeutics [
46,
47,
52,
53]. Typical gold nanorods are 30–50 nm long and 5–10 nm wide. The plasmon of nanorods splits into two modes of surface electron oscillation: (1) along the long axis, longitudinal (light absorption beyond 600 nm up to near infrared region) and (2) along the short axis which is typically in the range of spherical gold nanoparticles
i.e., 520–530 nm). Longitudinal plasmon can be tuned by controlling the anisotropic growth of gold seeds along the longer axis.
Typically, chemistry of GNRs formation involves templated growth of small gold seeds (1–3 nm) with a growth solution: a mixture of gold salt, ascorbic acid reductant, templating agent CTAB and a small amount of silver nitrate in water. Boleininger
et al. reported the first GNRs synthesis in a PVC microreactor connected to a three-way valve and a 7-port manifold (Upchurch Scientific) [
52]. The authors were able to produce GNRs with different aspect ratio by changing the ratio between the preformed gold seeds solution and the growth solution. In-situ monitoring of the GNRs formation was performed with an optical probe in the downstream part of the microreactor. The effect of the reaction temperature and reactant concentrations was systematically studied. Since preformed seeds for GNRs are not stable over a period of several hours, only freshly prepared seeds can serve as reliable core materials for their growth into monodisperse GNRs. In this way, Bullen
et al. modified this synthesis by employing a sequentially rotating tube processor (RTP) connected to a microfluidic chip to perform sequential operations of seed formation and growth of seeds into GNRs (
Figure 10) [
53]. A stable continuous operation for 19 days was demonstrated, which would be hampered had they used presynthesized seeds. Spectral data for all the samples collected over 19 days of operation demonstrate the robustness of the system to produce high quality of gold nanorods having reproducible quality and least by-products (spherical or irregular-shaped gold nanoparticles) as evident from the TEM picture.
Figure 10.
(
A) Injection of reagents into Rotating Tube processor (RTP) and Residence Time Unit fitted with an on-line UV monitoring system; (
B) UV-Vis Spectrum of Gold Nanorods produced over 19 days; (
C) TEM photographs of the produced GNRs. Adapted from Ref. [
53] (with permission from Royal society of Chemistry).
Figure 10.
(
A) Injection of reagents into Rotating Tube processor (RTP) and Residence Time Unit fitted with an on-line UV monitoring system; (
B) UV-Vis Spectrum of Gold Nanorods produced over 19 days; (
C) TEM photographs of the produced GNRs. Adapted from Ref. [
53] (with permission from Royal society of Chemistry).
In the “seedless” method, a gold precursor solution (HAuCl4, CTAB, acetylacetone) and a reactant mixture (CTAB, pH 10 carbonate buffer and AgNO3) were fed into the RTP (30 cm × 6 cm, rotating at 1000 rpm). The centrifugal force of the RTP generated a dynamic thin film (300 µm) on the inner wall of the reactor facilitating the Au-seed formation in 30 s. These seeds subsequently grew to nanorods of 24.2 nm × 6.6 nm in the microfluidic chip. Typically, the control of shape and size of anisotropic gold nanoparticles is accomplished by adjusting the concentration of shape modulating agents: silver and CTAB capping agents. By varying the silver concentrations in the second feed, the authors were able to control the aspect ratio of the produced rods. Although, such capping agents enable synthesis of high quality GNRs, both Ag and CTAB exhibit in vivo cytotoxicity, limiting their direct use in biological systems.
In an attempt to create a CTAB and Ag-free variant of anisotropic GNRs synthesis, Sebastian
et al. presented a Y-mixer connected to a 760 µm i.d.; internal diameter PTFE microreactor for continuous flow synthesis of biocompatible GNRs using amino acid (Lysine) instead of CTAB [
54]. The presence of two amino groups in Lysine molecule serves a dual purpose of capping and bridging, which is essential for anisotropic growth of gold particles. A premixed solution of gold salt, potassium carbonate and Lysine was contacted for 10 min with sodium borohydride solution in the microreactor placed in an ultrasonic bath (
Figure 11). GNRs are obtained with high monodispersity with microreactor (
Figure 11B,C,E) while GNPs with irregular shapes and polydispersity were obtained in benchmark experiments in a batch reactor (
Figure 11A,D). Fast mixing between the reactants is attributed to such pronounced difference between conventional and flow reactors. The produced GNRs were biocompatible and exhibited strong absorption in the near-infrared range. These GNRs were successfully used in photothermal optical coherence tomography (OCT) of human breast tissue.
Figure 11.
Upper panel: Schematic of Microfluidic setup for CATB and Ag-free Gold Nanorods synthesis. Lower panel: (
A) Irregular-shaped Gold Nanoparticles from Batch experiments; (
B) Gold nanorods from microfluidic experiment; (
C) Absorption spectra of GNP produced in batch and continuous microfluidic device; (
D) Size distribution of GNPs from batch reactor; (
E) Size distribution of GNRs from microfluidic device. Adapted from Ref. [
54] (with permission from Royal society of Chemistry).
Figure 11.
Upper panel: Schematic of Microfluidic setup for CATB and Ag-free Gold Nanorods synthesis. Lower panel: (
A) Irregular-shaped Gold Nanoparticles from Batch experiments; (
B) Gold nanorods from microfluidic experiment; (
C) Absorption spectra of GNP produced in batch and continuous microfluidic device; (
D) Size distribution of GNPs from batch reactor; (
E) Size distribution of GNRs from microfluidic device. Adapted from Ref. [
54] (with permission from Royal society of Chemistry).
So far, we considered syntheses under single-phase conditions, via controlled mixing and reactions between two or more miscible reagent solutions. This protocol is simple and easily adaptable to different microfluidic set-ups. However, there are several drawbacks associated with single-phase materials synthesis: (1) due to axial diffusion in laminar flow, there exists wide distribution in the residence time (
Figure 12A,C), which contributes to polydisperse and intractable mixture of several products and by products [
55,
56]; (2) mixing occurs solely by diffusion, which requires micromachining of additional structural elements (e.g., pin-fin or split-and-recombine) with lithographic techniques to induce convective flux to assist mixing (Hydrodynamics and reaction studies in a layered herringbone channel [
57]; (3) deposition of gold nanoparticles occurs onto the inner reactor walls. The deposit serves as nucleation sites hence depleting feed stock in an unproductive way. The reactor becomes unusable at longer reaction times, which impedes the utility of the reactor for long-term use. Passivation of the channel surface with hydrophobic functional groups was proposed as a possible remedy; however, such methods are not generic for all materials used for microfluidic device fabrication. Specifically, while PDMS is the most widely used materials for microfluidic reactor fabrication, unfortunately PDMS is unsuitable for such chemical passivation.
Figure 12.
Cartoons for liquid flow pattern and velocity dispersion in liquid elements for (
A) one-phase laminar flow, (
B) water-in-oil droplet flow. Residence time distribution in (
C) laminar flow and (
D) droplet flow regimes. Adapted from Ref. [
55] (with permission from Wiley-VCH).
Figure 12.
Cartoons for liquid flow pattern and velocity dispersion in liquid elements for (
A) one-phase laminar flow, (
B) water-in-oil droplet flow. Residence time distribution in (
C) laminar flow and (
D) droplet flow regimes. Adapted from Ref. [
55] (with permission from Wiley-VCH).
13. Three Phase Segmented Flow
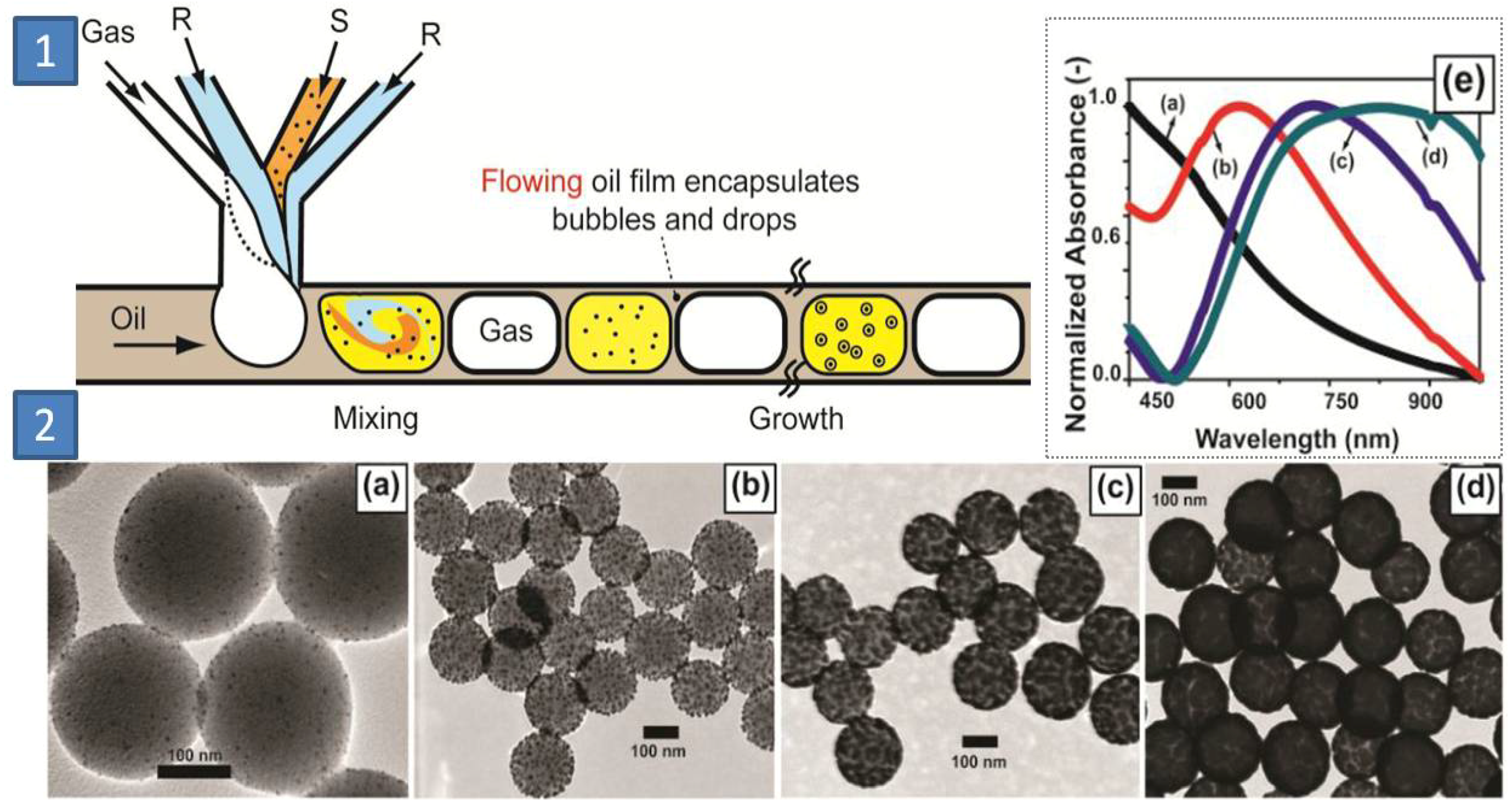
Three phase segmented flow of inert gas, reactant containing aqueous droplets dispersed in a continuous oil phase was employed inside a microchannel by Duraiswamy
et al. to produce SiO
2@Au core-shell materials with variable plasmonic behavior (600–900 nm in absorption spectrum) [
65] (
Figure 16). In this approach, inlet clogging due to particle deposition was avoided and droplet coalescence was considerably reduced. The gold coverage onto the silica particles was controlled by the inlet flow rates. Moreover, uncontrolled formation of free gold outside the surface of seeded-silica particles was largely avoided, which enabled pure batches of core-shell materials of varying plasmonic signatures.
Figure 16.
(
1) Microfluidic formation of gas-aqueous foam in continuous oil; (
2) Panel showing TEM photographs of (
a) gold-seeded silica particles, (
b) small gold island on silica, (
c) dense gold islands on silica, (
d) fully developed gold shell on silica spheres, (
e) inset showing UV-Vis absorption of the particles (
a)–(
d). Adapted from Ref. [
65] (with permission from American Chemical Society).
Figure 16.
(
1) Microfluidic formation of gas-aqueous foam in continuous oil; (
2) Panel showing TEM photographs of (
a) gold-seeded silica particles, (
b) small gold island on silica, (
c) dense gold islands on silica, (
d) fully developed gold shell on silica spheres, (
e) inset showing UV-Vis absorption of the particles (
a)–(
d). Adapted from Ref. [
65] (with permission from American Chemical Society).
In majority of work, liquid-based
i.e., aqueous solutions of reducing agents such as ascorbic acid, trisodium citrate, sodium borohydride
etc. are used in redox-based nanomaterials syntheses. Carbon monoxide or hydrogen are cleaner variants of the reducing agents and can be separated from the reaction mixture by mere exposing to ambient atmosphere. Several groups utilized gaseous reactants in nanomaterials synthesis [
66,
67,
68], including gold nanoshell synthesis [
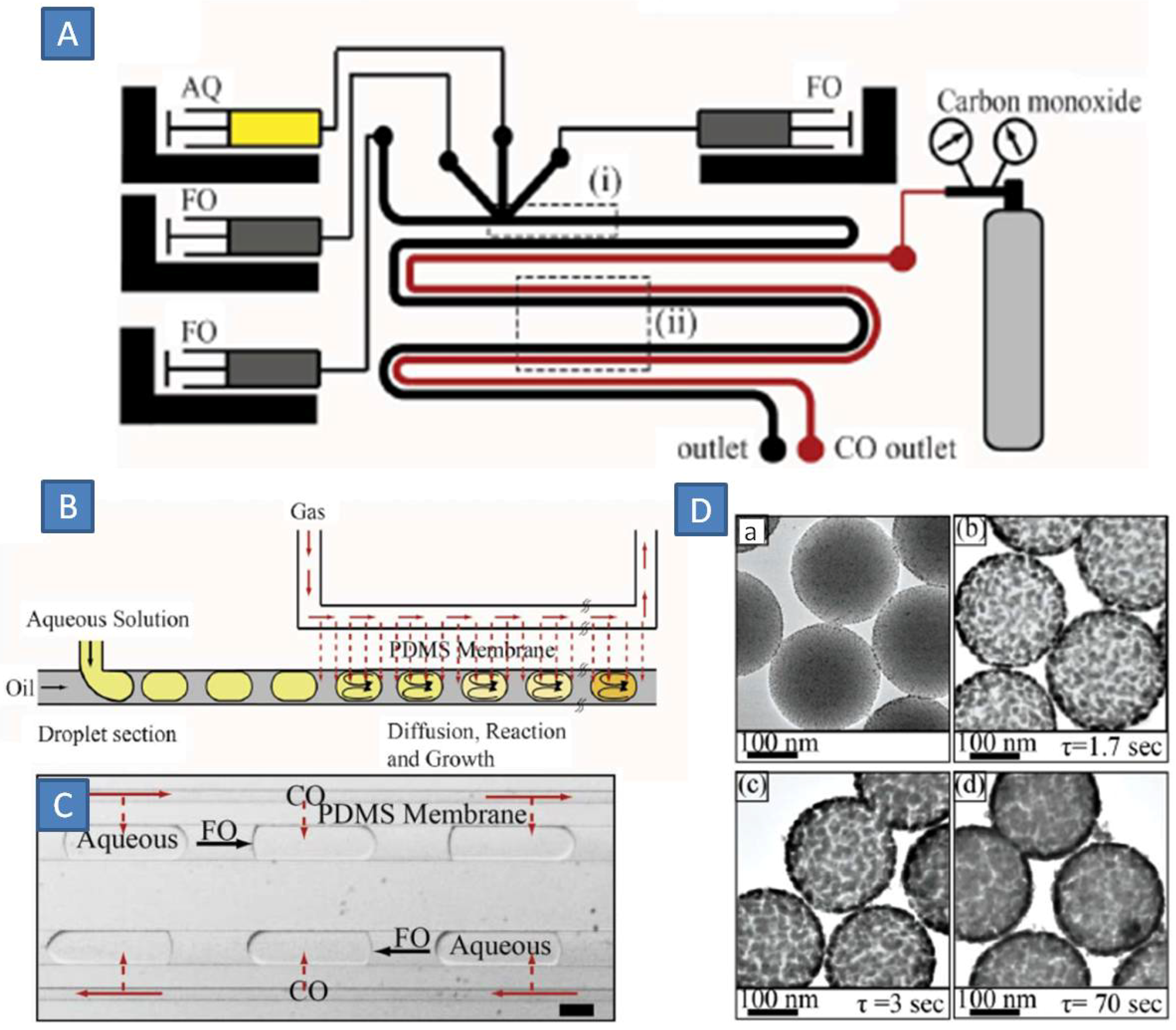
69]. As the size of GNPs depends on kinetics on nucleation and growth, any inhomogeneity during gas-liquid mass-transport would render the synthesis unreliable and irreproducible. Multiphase microchemical systems take advantage of the large interfacial areas. Carbon monoxide was used as reducing agent to produce SiO
2@Au nanomaterials in a PDMS-membrane microreactor by Rahman
et al. (
Figure 17A) [
70]. In this way, CO was dissolved inside the aqueous droplets without forming any gas bubbles. (
Figure 17B,C). Depending on the residence time of aqueous droplets (1–70 s), the flow rate of CO across the membrane was adjusted to control the rate of gold deposition onto the silica core. In this way, SiO
2@Au nanomaterials with variable gold coverage and impressively high purity (free from colloidal gold) were produced (
Figure 17D). Rapid quenching of the gold deposition was possible by eliminating gas/liquid contact in the downstream section.
Figure 17.
(
A) Microfluidic set-up for water-oil droplet formation; Schematic (
B) and microscopic photograph (
C) of gas-liquid contacting across the membrane; (
D) Core-shell formation with variable gold coverage (
a–
d). Adapted from Ref. [
70] (with permission from Royal Society of Chemistry).
Figure 17.
(
A) Microfluidic set-up for water-oil droplet formation; Schematic (
B) and microscopic photograph (
C) of gas-liquid contacting across the membrane; (
D) Core-shell formation with variable gold coverage (
a–
d). Adapted from Ref. [
70] (with permission from Royal Society of Chemistry).
14. Application of Ionic Liquids
Recently, ionic liquids (IL) are considered as an alternative reaction medium for nanoparticles synthesis, given their unique molecular nature, polarity and ability to stabilize nanoparticles via numerous modes of interactions [
71,
72,
73]. Ionic liquids may serve a dual purpose as a solvent and molecular/colloidal stabilizer, however GNPs synthesis in IL is challenging in terms of size distribution, presumably due to poor mass transport in viscous ionic liquids.
Lazarus
et al. leveraged the beneficial attributes of both ionic liquids and microreactors for the synthesis of GNPs [
74]. The authors used a flow-focusing microfluidic protocol where in the mixing zone a stream of butyl methylimidazolium tetrafluoroborate (BMIM-BF
4) was injected between the two reactant streams (HAuCl
4/1-methylimidazole and NaBH
4 in BMIM-BF
4) to allow fast mixing (
Figure 18A). As this stream arrived at a flow-focusing cross-junction, two oil streams were fed orthogonal to the IL streams. At oil flow rates below 3000 µL h
−1, the middle IL stream maintained co-parallel flow with the flanking oil streams (inset A in
Figure 18), while above this threshold value, droplets of IL were formed (Inset B in
Figure 18). Fast and homogeneous mixing enable rapid nucleation burst, and ensure monodispersity in the final fully grown GNPs. The liquid mixture was quenched at the outlet with ethanol to separate the produced GNPs and analyzed by spectroscopic and microscopic methods. The best quality GNPs in terms of polydispersity (12%), size and roundedness of the GNPs was achieved from the droplet protocol. The mean particle diameter was 4.38 ± 0.53 nm having spherical features. The roundness, defined as (4 × particle area)/(π × (major axis length)
2), below 0.85 was observed only in 15% particles. UV-vis spectra of suspensions exhibited narrow surface plasmon bands cantered at 518.5 nm, typical of non-agglomerated GNPs. With the parallel flow protocol, the GNPs were of inferior structural features compared to droplet protocol. The mean diameter was 5.65 ± 1.03 nm with a polydispersity index of 18.2%. The roundness below 0.85 was observed in 23% particles. Intriguingly, thinning of the innermost lamella (IL liquid) by increasing the oil flow rate showed tendency of the GNPs to become more monodispersed compared to the case when the inert oil phase was completely excluded. Understandably, mixing within droplets is most efficient due to intense vortex generation, while interdiffusion between liquid streams become progressively limited as the characteristic dimension of the lamella becomes larger. In all cases, the synthesis was completed in 19 s. A benchmarking experiment in the batch requires at least 1 min. The obtained nanoparticles were significantly spheroidal with 28% having a roundness less than 0.85. The mean diameter was larger than that in the flow synthesis (6.25 ± 1.29 nm; polydispersity: 20.6%).
Figure 18.
Schematic of the PDMS device (3 × 5 cm) with flow focusing geometry. channel widths and depths are 600 µm and 95 µm (flow-focusing region width: 400 µm), respectively, with a total channel volume of 7.6 µm. Inset
A: parallel flow of IL and inert Oil at Oil flow rates below 3000 µL h
−1, inset
B: IL droplet formation at Oil flow rates beyond 3000 µL h
−1. Adapted from Ref. [
74] (with permission from Royal Society of Chemistry).
Figure 18.
Schematic of the PDMS device (3 × 5 cm) with flow focusing geometry. channel widths and depths are 600 µm and 95 µm (flow-focusing region width: 400 µm), respectively, with a total channel volume of 7.6 µm. Inset
A: parallel flow of IL and inert Oil at Oil flow rates below 3000 µL h
−1, inset
B: IL droplet formation at Oil flow rates beyond 3000 µL h
−1. Adapted from Ref. [
74] (with permission from Royal Society of Chemistry).
Larazus
et al. later reported the use of ionic liquid variant of sodium borohydride, namely, 1-butyl-1-methylimidazolium borohydride (BMIM BH
4), to increase the solubility of reducing agent in butyl methylimidazolium bis-triflamide ionic liquid (BMIM NTf
2) in the synthesis of GNPs in a fluorinated PDMS channel [
75]. The dispersed phase contained the IL reducing agent and gold ions while a fluorocarbon oil was continuous phase. At low ionic liquid/oil flow ratios of 0.05–0.25, droplets were formed immediately at the junction location. In comparison to batch conditions their microfluidic method produced GNPs of smaller size and higher monodispersity.
15. Scale-Up of GNP Synthesis Using Flow-Chemistry
Only few reports emerged that tackles the scale-up issue associated with complex gold nanoparticles production. Gomez
et al. presented the first example of continuous synthesis of hollow GNPs in a flow reactor composed of T-pieces for mixing and a 1.6 mm diameter PTFE tube as residence time unit at two different scales corresponding to production rates of 0.2 and 2.0 mmol Au/min [
76]. Hollow GNPs exhibit plasmonic behavior, where the ratio of particle diameter to the thickness of the shell determines the surface plasmon resonance (SPR) peak position and their uniformity determines the SPR bandwidth. At first, a cobalt solution was mixed with sodium borohydride in the first T-junction which produced cobalt nanospheres within 180 s of reaction time (
Figure 19). This step was diffusion limited, and the reaction time was set slightly higher than the diffusive mixing time of 160 s. In the second mixer, a gold precursor was introduced which was reduced at the surface of cobalt particles which in turn was dissolved out after being oxidized. In the scale-up process, a stream of oxygen was introduced in the 3rd mixing port to facilitate this galvanic dissolution process to form completely Co free hollow GNPs.
In-situ functionalization of hollow GNPs with thiolated polyethylene glycols was demonstrated to enhance the biocompatibility. An integrated continuous UV sterilization step facilitated the on-site
in vivo use of the as-synthesized materials. Such integration of continuous production and downstream processing in a millireactor holds promises for scaling up of “ready-to-use” nanoparticles production.
Figure 19.
Microfluidic schematic of hollow gold nanoparticles in a multi-step approach with average reaction time for each step. Insets are TEM photographs of the corresponding nanoparticles formed throughout the process at different stages. Adapted from Ref. [
76] (with permission from Royal Society of Chemistry).
Figure 19.
Microfluidic schematic of hollow gold nanoparticles in a multi-step approach with average reaction time for each step. Insets are TEM photographs of the corresponding nanoparticles formed throughout the process at different stages. Adapted from Ref. [
76] (with permission from Royal Society of Chemistry).
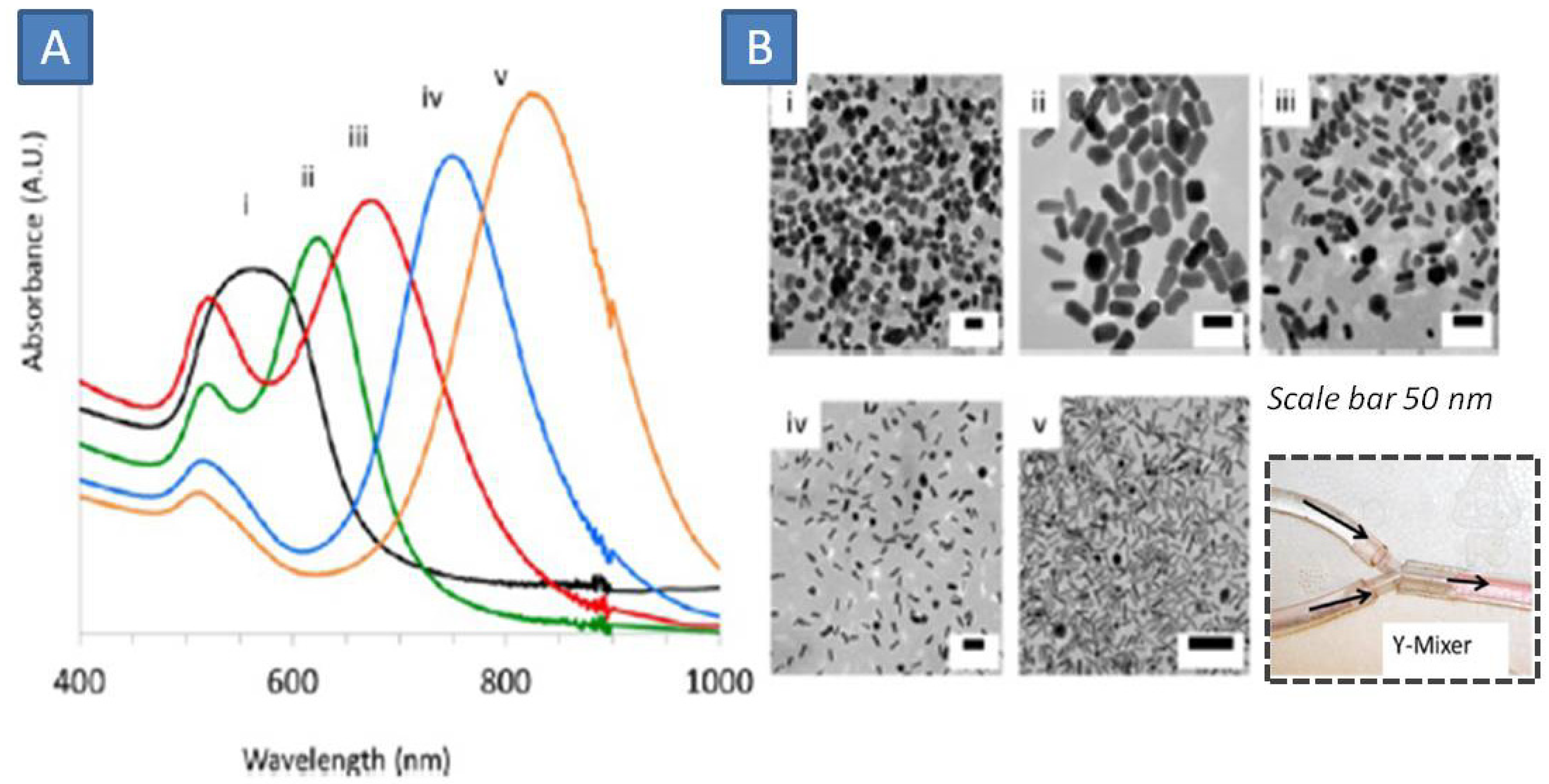
Lohse
et al. scaled up the microfluidic synthesis to produce 10 L of suspension of GNRs with aspect ratio 2 in three consecutive runs amounting to a total of 1.0 g of product [
77]. The product yield and quality showed improvement in regards to monodispersity when compared to the synthesis in a batch system. This clearly shows superiority of microfluidic synthesis over conventional bath synthesis that typically produces 50 mg of GNPs. This was possible using 5–10 fold higher concentrations of reactants in the flow system. Off-the shelf materials were used to construct the fluidic reactor, namely from TYGON polyvinyl tubing as the reactor and polyethylene connectors as mixing ports. In the first step, CTAB-stabilized spherical GNPs were sued via rapid reduction of gold with NaBH
4 was conducted in the flow reactor that produces GNPs that would be used as seed elements in the GNRs synthesis. In the second stage, these seeds were mixed with a growth solution containing (gold salt, CTAB, silver nitrate and ascorbic acid) to generate the rods of tight batch-quality (in terms of desired shape-size and avoidance of undesired by-products). By changing concentrations GNRs with different length (and aspect ratios were obtained. They produce a wide range of plasmonic bands up to near infrared region (
Figure 20).
Figure 20.
(
A) UV-Vis spectrum of GNRs of various aspect ratios; (
B) TEM Photographs of GNRs, inset showing the simple T-junction used as micromixer in this work [variable aspect ratios of the produces gold nanorods are shown in (i)-(v)]. Adapted from Ref. [
77] (with permission from American Chemical Society).
Figure 20.
(
A) UV-Vis spectrum of GNRs of various aspect ratios; (
B) TEM Photographs of GNRs, inset showing the simple T-junction used as micromixer in this work [variable aspect ratios of the produces gold nanorods are shown in (i)-(v)]. Adapted from Ref. [
77] (with permission from American Chemical Society).