3.1. Model Calibration
Analysis of the concentration gradients over time during the laboratory processing of the passive samplers revealed that the most hydrophobic compounds under investigation had very little to no desorption from the PDMS sorbent layer of the SPME PDMS fibers. For the 559/486 µm and 1060/1000 SPME PDMS fiber, no measurable desorption was observed for compounds with a log
Kow (log
Kow values for PAHs (SPARC estimates) [
15]) of 4.7 or higher, 5.29 or higher, 6.58 or higher, for ambient temperatures of 4 °C, 20 °C, and 25 °C, respectively. For the 230/210 µm SPME PDMS fiber, there was substantial change from the initial concentration sorbed to the PDMS layer over the duration of the experiment for all three temperature treatments. Generally, the PAHs of potential concern for substantial loss from the SPME PDMS fibers included naphthalene (log
Kow = 3.41), acenaphthene (log
Kow = 4.06), fluorene (log
Kow = 4.2), anthracene (log
Kow = 4.69), and phenanthrene (log
Kow = 4.74). The concentration
versus time behavior for these compounds suggests a relatively rapid (exponential) loss initially followed by a slowing of evaporation after a long time, likely associated with increasing importance of internal mass transfer resistances on evaporation. Our goal is to model losses to ensure that any such losses are small (e.g., less than 10%) for a sampling event either due to sample processing times or environmental factors like tides. Thus the model and the analysis was focused on the initial exponential loss phase.
Vapor pressure, Henry’s Law Coefficient, and temperature relationships were available for the PAHs of most concern in this study and dimensionless Henry’s Law Coefficients ((mol/m
3)
air/(mol/m
3)
water) at ambient temperatures of 4 °C, 20 °C, and 25 °C are compiled in
Table 1 [
16]. The estimated log
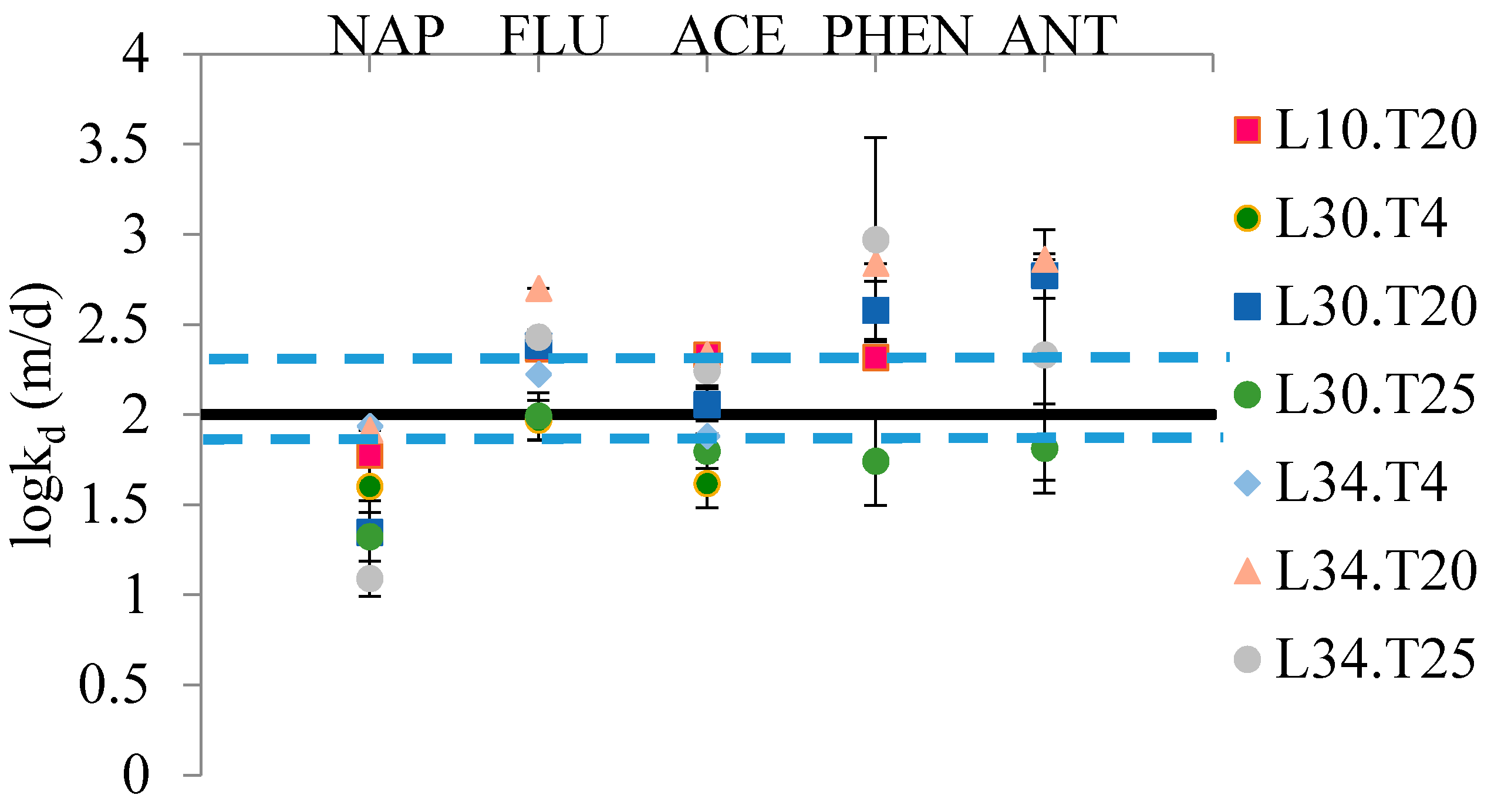
kd (in m/day) consistent with the observed loss over time for these five compounds averaged approximately 2 ± 0.2 (average ± 95% confidence interval on the mean) with no significant dependence upon compound, sampler thickness or temperature as shown in
Figure 1. This is for the relatively static environment in a lab or protected area in the field likely to be used for sample processing and the variability likely represents variations associated with air motion in the static but uncontrolled environment. Naphthalene and the thinnest PDMS layer
kd values were more variable due to the relatively rapid loss of this compound making measurements difficult and the greater influence of internal mass transfer resistances.
Table 1.
logKfw, (log(μg/L)PDMS/(μ/L)water), logH (log (mol/m3)air/(mol/m3)water), and logH/Kfw values for select polynuclear aromatic hydrocarbons (PAHs) at the temperatures of interest.
Table 1.
logKfw, (log(μg/L)PDMS/(μ/L)water), logH (log (mol/m3)air/(mol/m3)water), and logH/Kfw values for select polynuclear aromatic hydrocarbons (PAHs) at the temperatures of interest.
| Compound | logKfw1 | logH (277 K) | logH/Kfw (277 K) | logH (293 K) | logH/Kfw (293 K) | logH (298 K) | logH/Kfw (298 K) |
|---|
| Naphthalene | 2.95 | −2.38 2 | −5.3 | 1.86 3 | −4.8 | −1.76 3 | −4.7 |
| Acenaphthene | 3.42 | −2.81 4 | −6.2 | −2.35 4 | −5.8 | −2.28 4 | −5.7 |
| Fluorene | 3.52 | −3.05 4 | −6.6 | −2.61 4 | −6.1 | −2.39 4 | −5.9 |
| Anthracene | 3.77 | −3.26 4 | −7.1 | −2.84 4 | −6.7 | −2.63 4 | −6.5 |
| Phenanthrene | 3.79 | −3.39 4 | −7.3 | −2.96 4 | −6.9 | −2.75 4 | −6.7 |
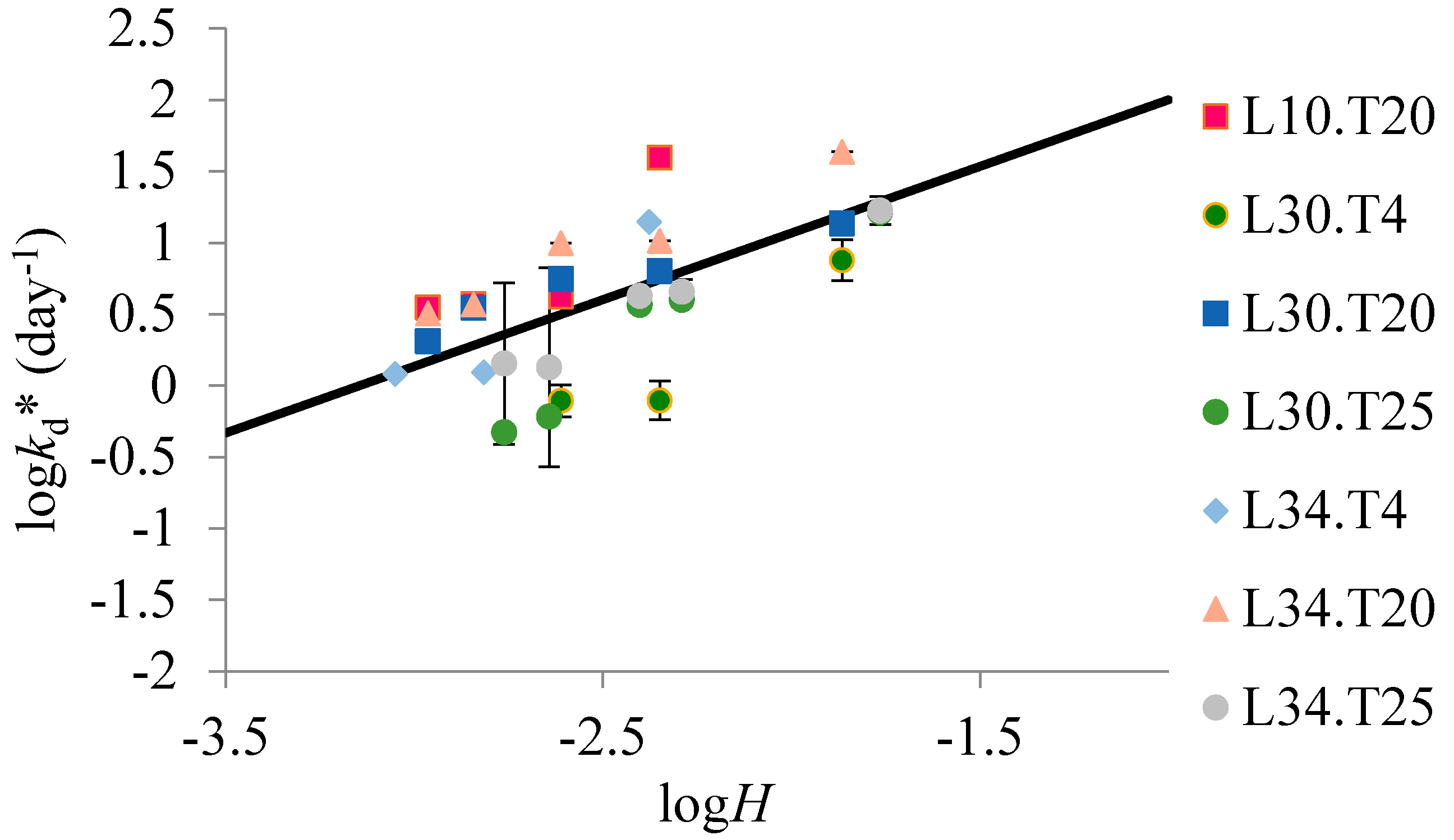
Defining
suggests a linear relationship between
and
H if
kd is constant (
Figure 2). The slope of
and log(
H) is approximately one, which lends support to the model. High
H compounds will evaporate more rapidly and H can be minimized by reducing the temperature. The variability seen in
Figure 1 and
Figure 2 could be attributed to the different air flow conditions of the temperature controlled environmental chambers, as indicated previously. Keeping the samplers at a lower temperature, and thereby reducing the Henry’s constant, reduces evaporation. Based upon the experimental results, the half-life of naphthalene on a 230/210 µm SPME PDMS fiber can be increased tenfold to 0.2 h by storing the sampler at 4 °C rather than 25 °C.
Figure 1.
Experimentally determined log (kd) values for the compounds that showed the most rapid desorption for all experimental treatments. L refers to volume to area ratio of sampler and T is temperature, °C. kd is the desorption rate coefficient (m/day) associated with the air motion around the sampler. logkd is a constant equal to 2 ± 0.2 (average ± 95% confidence interval on mean, n = 31). Solid line represents average and broken line represents confidence interval on mean. NAP: naphthalene; FLU: fluorene; ACE: acenaphthene; PHEN: phenanthrene; ANT: anthracene.
Figure 1.
Experimentally determined log (kd) values for the compounds that showed the most rapid desorption for all experimental treatments. L refers to volume to area ratio of sampler and T is temperature, °C. kd is the desorption rate coefficient (m/day) associated with the air motion around the sampler. logkd is a constant equal to 2 ± 0.2 (average ± 95% confidence interval on mean, n = 31). Solid line represents average and broken line represents confidence interval on mean. NAP: naphthalene; FLU: fluorene; ACE: acenaphthene; PHEN: phenanthrene; ANT: anthracene.
Figure 2.
logH (H is dimensionless) versus logkd* for all solid phase microextraction (SPME) polydimethylsiloxane (PDMS) thickness (L10, L30, or L34) and temperature treatments (T4, T20, or T25) for naphthalene, fluorene, acenaphthalene, and anthracene. L10, L30, and L34 refer to the PDMS thickness of the 230/210 µm, 1060/1000 µm, and the 559/486 µm SPME PDMS fibers used in this experiment, respectively. T4, T20, and T25 refer to the temperatures of 4 °C, 20 °C, and 25 °C used in this experiment, respectively , where kd is the desorption rate coefficient (m/day) associated with the air motion around the sampler, H is the dimensionless Henry’s constant, L is the PDMS sorbent thickness, and Kfw is the PDMS-water partition coefficient. The solid black line represents the relationship between logH and logkd*, where logkd* = 1(±0.2)logH + 3(±0.5), r2 = 0.53. The error bars represent the standard deviation associated with each measurement.
Figure 2.
logH (H is dimensionless) versus logkd* for all solid phase microextraction (SPME) polydimethylsiloxane (PDMS) thickness (L10, L30, or L34) and temperature treatments (T4, T20, or T25) for naphthalene, fluorene, acenaphthalene, and anthracene. L10, L30, and L34 refer to the PDMS thickness of the 230/210 µm, 1060/1000 µm, and the 559/486 µm SPME PDMS fibers used in this experiment, respectively. T4, T20, and T25 refer to the temperatures of 4 °C, 20 °C, and 25 °C used in this experiment, respectively , where kd is the desorption rate coefficient (m/day) associated with the air motion around the sampler, H is the dimensionless Henry’s constant, L is the PDMS sorbent thickness, and Kfw is the PDMS-water partition coefficient. The solid black line represents the relationship between logH and logkd*, where logkd* = 1(±0.2)logH + 3(±0.5), r2 = 0.53. The error bars represent the standard deviation associated with each measurement.
The model also suggests that the sorbent dimensions will influence evaporation half-life through L, the sampler’s volume to area ratio or essentially the sorbent thickness. There was found to be little difference in half-lives between the 1060/1000 µm and 559/486 µm SPME PDMS fibers for the three temperatures of this study due to their PDMS thicknesses being similar (~30 µm). In general, there is a decrease in the desorption half-life of the compounds for all temperatures when using the 230/210 µm SPME PDMS fiber versus the 1060/1000 µm and 559/486 µm SPME PDMS fiber. For example, naphthalene, the most volatile PAH in this study, has a half-life of 0.2 h when initially sorbed to a 1060/1000 µm or 559/486 µm SPME PDMS fiber and exposed to an ambient air temperature of 25 °C compared to a half-life of 0.02 h when sorbed to a 230/210 µm SPME PDMS fiber (10 µm thick PDMS layer) at the same temperature. The very fast desorption time onto the thinner fibers suggests that naphthalene is not a viable target compound of the thinner PDMS fiber in the field. Evaporation of naphthalene and fluorene from the 230/210 fiber was generally too rapid to be accurately measured and these were excluded from subsequent analysis.
The half-life results suggest that when using SPME PDMS fibers with dimensions of 559/486 µm or 1060/1000 µm, compounds with
are stable (
Cf/
Cf,o ≥ 0.9) for 20 min exposure periods at an ambient air temperatures of 25 °C. Compounds with
are stable for 20 min exposure periods sorbed to a 230/210 µm SPME PDMS fiber, with an approximate 10 µm thickness of PDMS, and exposed to an ambient air temperatures of 20 °C. In general, the results suggest if rapid processing is not possible, thicker sorbent layers or lower processing temperatures should be used for volatile compounds such as naphthalene.
Figure 3 depicts the predicted
versus observed half-life times in days for the different SPME PDMS fiber thicknesses. The predicted values were calculated using
kd* values predicted from the relationship between log
H and log
kd* and Equation (5).
Figure 4 shows the modelled
Cf/
Cf,o (from Equation (4))
versus time curves for a 10 µm or 30 µm sorbent layer thickness of PDMS for sampling hydrophobic organic contaminants, exposed to air at 4 °C or 20 °C.
Table 2 provides estimates of the sampling time required to ensure 90% concentration retention of naphthalene using PDMS passive sampling sorbent of various thicknesses at 20 °C and 4 °C, respectively. If samplers are retrieved and immediately stored at low temperatures, there is an increase in the maximum time for which the fibers can be exposed without losing more than 10% of the sorbed analyte. For example, when using a 30 µm PDMS sorbent layer, the sample processing time required to minimize losses to less than 10% of the analyte sorbed to PDMS increases from approximately 2 min to 7 min. Using thicker sorbent layers is an option for increasing the retention time of the compounds sorbed to the polymer, but using thicker sorbent layers also increases the time necessary to reach equilibrium between the polymer and the sediment porewater or surface water. For example, a 1.4 cm thickness of PDMS would retain 90% of naphthalene sorbed to it for a 24 h period exposed to ambient air at 293 K, but would require lengthy absorbing time (centuries) to achieve 90% of equilibrium for porewater concentration measurement of highly hydrophobic five and six ring PAHs.
Figure 3.
Observed half-life (t1/2-Observed) values versus predicted half-life (t1/2-Predicted) values for naphthalene, fluorene, acenaphthene, phenanthrene, and anthracene desorption from SPME PDMS fibers. L30 and L34 refer to the PDMS thickness of the 1060/1000 µm, and the 559/486 µm SPME PDMS fibers used in this experiment, respectively. The observed half-lives were calculated using measured kd values in Equation (5). The predicted half-lives were calculated using the relationship determined between log(kd*) and logH (logkd* = 1(±0.2)logH + 3(±0.5), r2 = 0.53) and Equation (5). The black solid line represents a one-to-one relationship.
Figure 3.
Observed half-life (t1/2-Observed) values versus predicted half-life (t1/2-Predicted) values for naphthalene, fluorene, acenaphthene, phenanthrene, and anthracene desorption from SPME PDMS fibers. L30 and L34 refer to the PDMS thickness of the 1060/1000 µm, and the 559/486 µm SPME PDMS fibers used in this experiment, respectively. The observed half-lives were calculated using measured kd values in Equation (5). The predicted half-lives were calculated using the relationship determined between log(kd*) and logH (logkd* = 1(±0.2)logH + 3(±0.5), r2 = 0.53) and Equation (5). The black solid line represents a one-to-one relationship.
3.2. Modeling the Effect of Intertidal Drying
The model was applied to estimate losses during intertidal exposure of the surface water PDMS passive samplers in a field study. The average air temperature during the period of deployment was 7 °C and the PDMS water samplers were exposed to ambient air for up to 4 h or 8 h per day depending upon sampler location. During exposure, loss of the volatile target contaminants would limit the ability to quantify their concentration in the water. In addition, any loss of the PRCs during the exposure period would limit the ability to assess the extent of equilibrium uptake. The model developed previously was used to estimate the evaporative losses of the sixteen PAHs and the four PRCs used in the study.
Figure 5 shows the estimated loss of the most volatile compounds and each of the PRCs from the PDMS layer during the exposure period of 4 h or 8 h. For both scenarios, naphthalene, the least hydrophobic compound of interest for this study, exhibits substantial losses. The model predicts for both scenarios losses less than 10% of the mass sorbed to the PDMS for phenanthrene and other PAHs with greater hydrophobicity. Additionally, the loss of PRCs when exposed from the surface water is negligible (<10%) and therefore kinetic uptake estimates will not be influenced. Moreover the kinetics of loss of the PRCs of different hydrophobicity were consistent as per the analysis of [
15]. If evaporative loss of the PRCs were of concern, the lighter PRCs would show greater losses than expected.
Figure 4.
Modeled Cf/Cf,o values for 10 µm and 30 µm thick PDMS passive sampling materials for NAP-blue, FLU-red, and PHEN-green exposed to ambient air at 4 °C (277 K) and 20 °C (293 K): (a) L = 10 μm, T = 293 K; (b) L = 30 µm, T = 293 K; (c) L = 10 μm, T = 277 K; (d) L = 30 μm, T = 277 K.
Figure 4.
Modeled Cf/Cf,o values for 10 µm and 30 µm thick PDMS passive sampling materials for NAP-blue, FLU-red, and PHEN-green exposed to ambient air at 4 °C (277 K) and 20 °C (293 K): (a) L = 10 μm, T = 293 K; (b) L = 30 µm, T = 293 K; (c) L = 10 μm, T = 277 K; (d) L = 30 μm, T = 277 K.
Table 2.
Estimates of the time (min) at which Cf/Cf,o = 0.9 for NAP for different thicknesses of PDMS, exposed at 277 K and 293 K using model parameters tabulated above. Estimates based on kd model fit of 102 m/d.
Table 2.
Estimates of the time (min) at which Cf/Cf,o = 0.9 for NAP for different thicknesses of PDMS, exposed at 277 K and 293 K using model parameters tabulated above. Estimates based on kd model fit of 102 m/d.
| PDMS layer thickness | NAP Cf/Cf,o = 0.9 at time (min) |
|---|
| T = 277 K | T = 293 K |
|---|
| 30 μm | 10 | 3 |
| 100 μm | 34 | 10 |
Figure 5.
(a) Model of sorption and desorption of four select COCs from the 1060/1000 SPME PDMS fiber during a low tide event lasting 4 h (tsw = 20 h); (b) model of sorption and desorption of four select COCs from SPME PDMS fiber during a low tide event lasting 8 h (tsw = 16 h); (c) desorption of the four PRCs from the SPME PDMS fiber during submersion in surface water (tsw = 20 h) and during exposure to air during a 4 h low tide event; and (d) desorption of the four PRCs from the SPME PDMS fiber during submersion in surface water (tsw = 16 h) and during exposure to air during an 8 h low tide event. Average air temperature estimated to be 7 °C during air exposure period. C/Ceq represents the fraction of steady state achieved between the porewater and the SPME PDMS fiber. CPRC/CPRC,o represents the fraction lost of PRCs from the initial concentration spiked into the SPME PDMS fiber.
Figure 5.
(a) Model of sorption and desorption of four select COCs from the 1060/1000 SPME PDMS fiber during a low tide event lasting 4 h (tsw = 20 h); (b) model of sorption and desorption of four select COCs from SPME PDMS fiber during a low tide event lasting 8 h (tsw = 16 h); (c) desorption of the four PRCs from the SPME PDMS fiber during submersion in surface water (tsw = 20 h) and during exposure to air during a 4 h low tide event; and (d) desorption of the four PRCs from the SPME PDMS fiber during submersion in surface water (tsw = 16 h) and during exposure to air during an 8 h low tide event. Average air temperature estimated to be 7 °C during air exposure period. C/Ceq represents the fraction of steady state achieved between the porewater and the SPME PDMS fiber. CPRC/CPRC,o represents the fraction lost of PRCs from the initial concentration spiked into the SPME PDMS fiber.