Quality Assessment of Platelet-Rich Fibrin-Like Matrix Prepared from Whole Blood Samples after Extended Storage
Abstract
:1. Introduction
2. Experimental Section
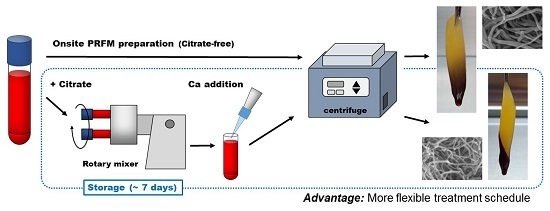
2.1. Blood Collection, Preservation, and Platelet-Rich Fibrin–Like Matrix (PRFM) Preparation
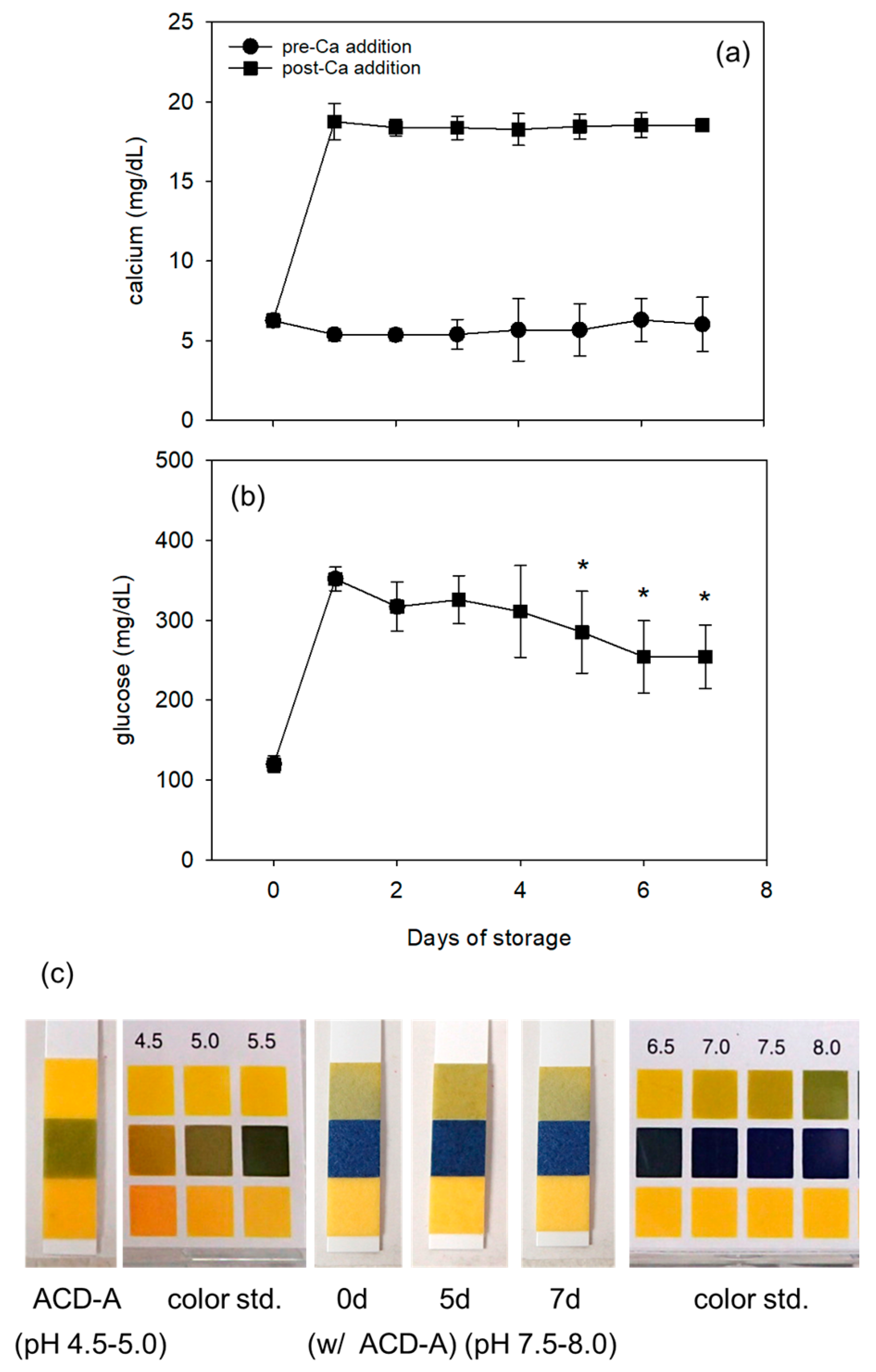
2.2. Measurement of Glucose and Ca2+ Levels and pH
2.3. Quantification of a Growth Factor by an Enzyme-Linked Immunosorbent Assay (ELISA)
2.4. Determination of Blood Cell Counts
2.5. Flow-Cytometric (FCM) Analyses
2.6. Scanning Electron Microscopy
2.7. Evaluation of Platelet Surface Antigen Expression by an Immunofluorescence Assay
2.8. Statistical Analysis
3. Results
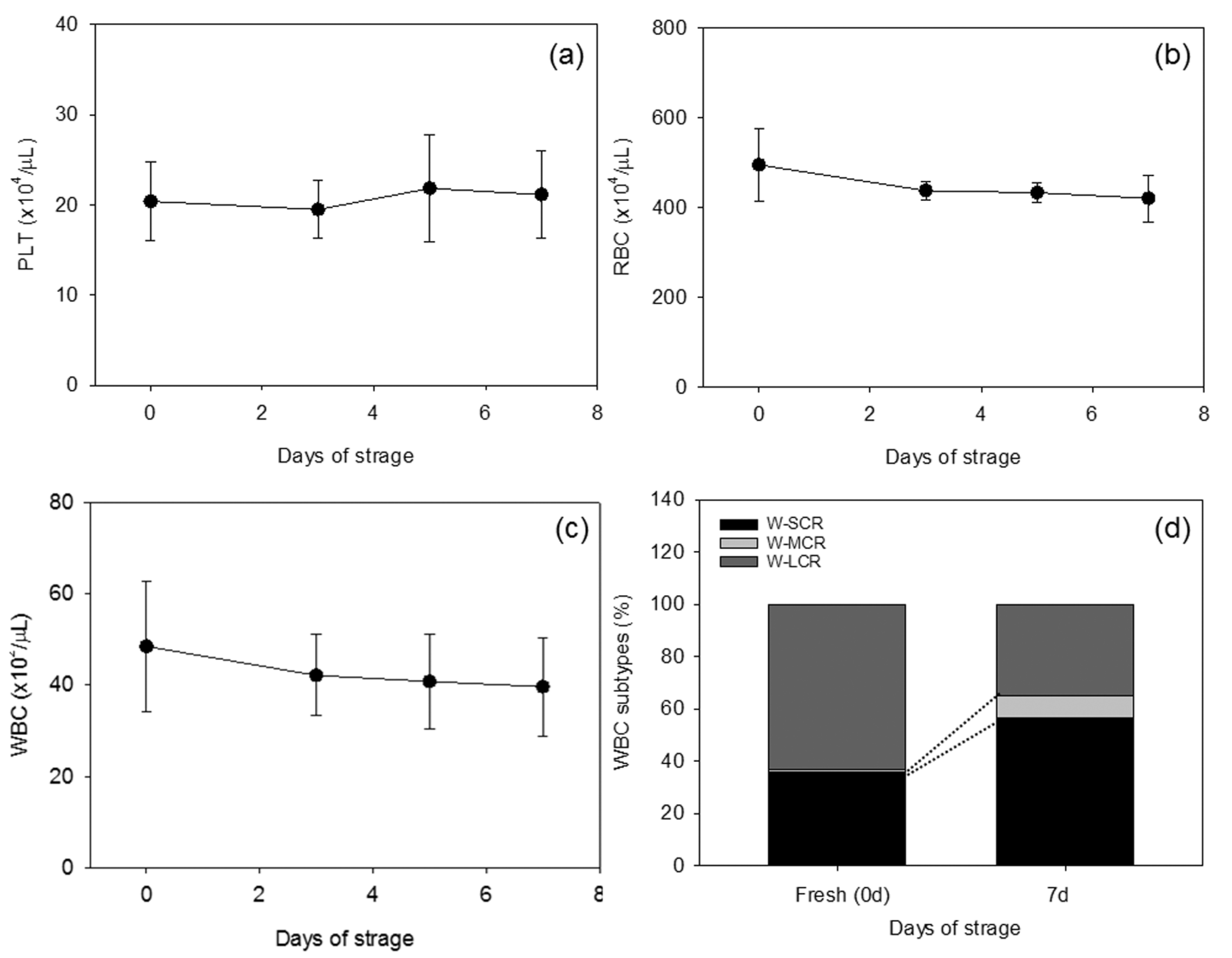
3.1. Time-Dependent Changes in The Characteristics of Whole Blood Samples
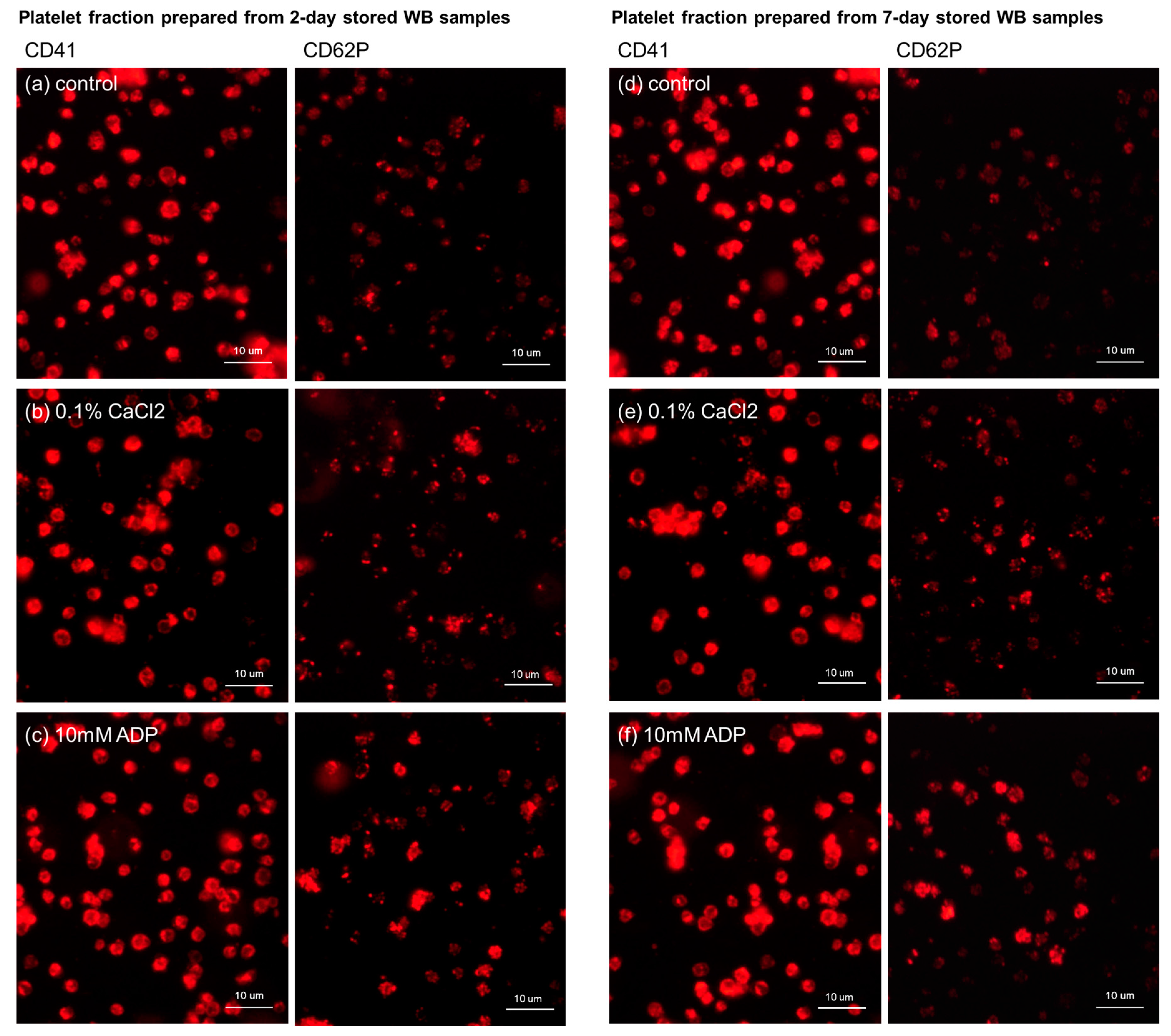
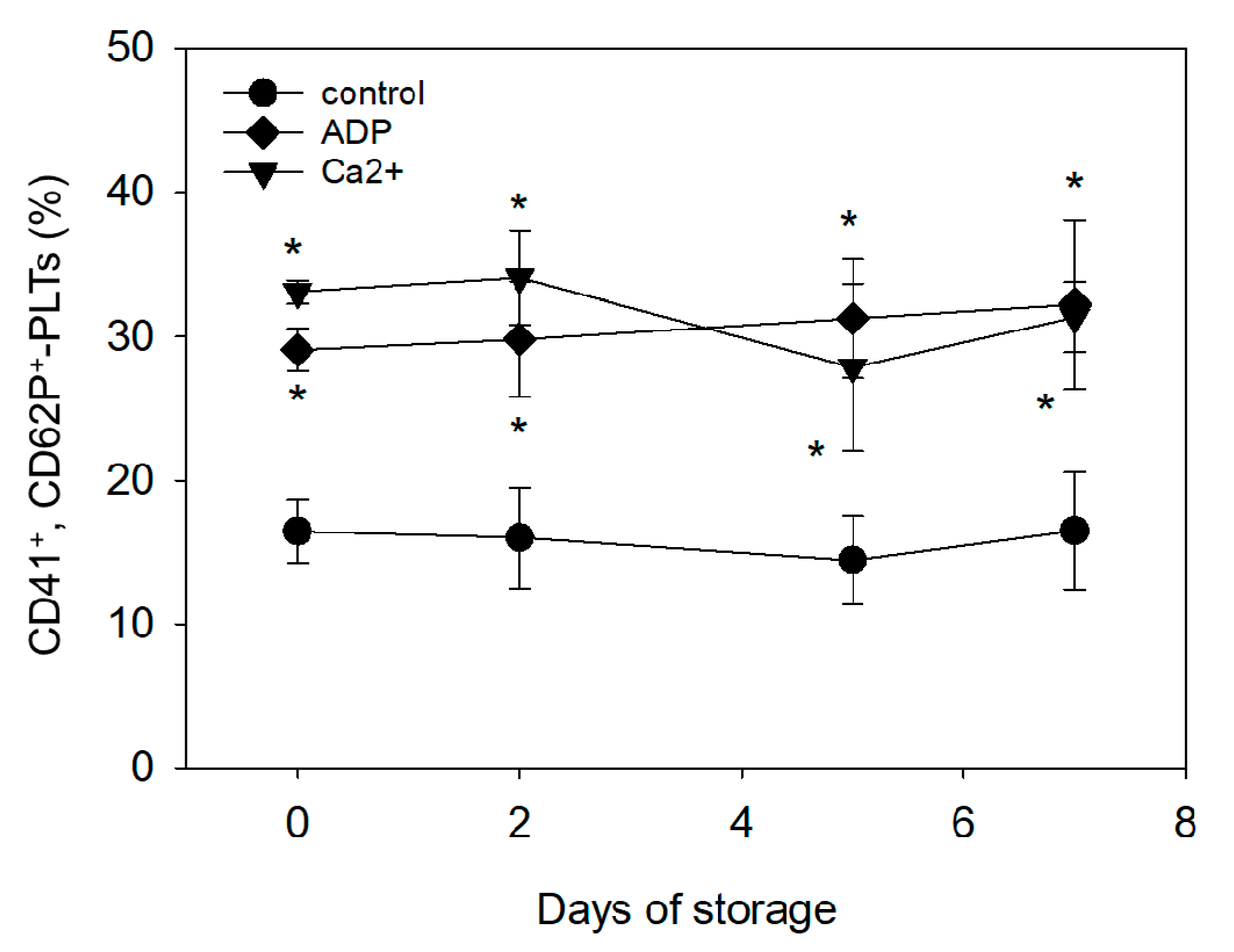
3.2. Time-Dependent Changes in the Quality of The Resultant PRFM Samples
4. Discussion
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Kawase, T. Platelet-rich plasma and its derivatives as promising bioactive materials for regenerative medicine: Basic principles and concepts underlying recent advances. Odontology 2015, 103, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Bai, M.Y.; Wang, C.W.; Wang, J.Y.; Lin, M.F.; Chan, W.P. Three-dimensional structure and cytokine distribution of platelet-rich fibrin. Clinics 2017, 72, 116–124. [Google Scholar] [CrossRef]
- Marrelli, M.; Tatullo, M. Influence of PRF in the healing of bone and gingival tissues. Clinical and histological evaluations. Eur. Rev. Med. Pharmacol. Sci. 2013, 17, 1958–1962. [Google Scholar] [PubMed]
- Kawase, T.; Watanabe, T.; Okuda, K. Platelet-rich plasma and its derived platelet concentrates: What dentists involved in cell-based regenerative therapy should know (in Japanese). Nihon Shishubyo. Gakkaikaishi. 2017, 59, 68–72. [Google Scholar] [CrossRef]
- Isobe, K.; Suzuki, M.; Watanabe, T.; Kitamura, Y.; Suzuki, T.; Kawabata, H.; Nakamura, M.; Okudera, T.; Okudera, H.; Uematsu, K.; et al. Platelet-rich fibrin prepared from stored whole-blood samples. Int. J. Implant. Dent. 2017, 3, 6. [Google Scholar] [CrossRef] [PubMed]
- Kreuger, A.L.; Caram-Deelder, C.; Jacobse, J.; Kerkhoffs, J.L.; van der Bom, J.G.; Middelburg, R.A. Effect of storage time of platelet products on clinical outcomes after transfusion: A systematic review and meta-analyses. Vox Sang. 2017, 112, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Isobe, K.; Watanebe, T.; Kawabata, H.; Kitamura, Y.; Okudera, T.; Okudera, H.; Uematsu, K.; Okuda, K.; Nakata, K.; Tanaka, T.; et al. Mechanical and degradation properties of advanced platelet-rich fibrin (A-PRF), concentrated growth factors (CGF), and platelet-poor plasma-derived fibrin (PPTF). Int. J. Implant. Dent. 2017, 3, 17. [Google Scholar] [CrossRef] [PubMed]
- Masuki, H.; Okudera, T.; Watanabe, T.; Suzuki, M.; Nishiyama, K.; Okudera, H.; Nakata, K.; Uematsu, K.; Su, C.Y.; Kawase, T. Growth factor and pro-inflammatory cytokine contents in PRP, plasma rich in growth factors (PRGF), advanced-platelet-rich fibrin (A-PRF) and concentrated growth factors (CGF). Int. J. Implant. Dent. 2016, 2, 19. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Isobe, K.; Suzuki, T.; Kawabata, H.; Nakamura, M.; Tsukioka, T.; Okudera, T.; Okudera, H.; Uematsu, K.; Okuda, K.; et al. An Evaluation of the Accuracy of the Subtraction Method Used for Determining Platelet Counts in Advanced Platelet-Rich Fibrin and Concentrated Growth Factor Preparations. Dent. J. 2017, 5, 7. [Google Scholar] [CrossRef]
- Rodella, L.F.; Favero, G.; Boninsegna, R.; Buffoli, B.; Labanca, M.; Scari, G.; Sacco, L.; Batani, T.; Rezzani, R. Growth factors, CD34 positive cells, and fibrin network analysis in concentrated growth factors fraction. Microsc. Res. Tech. 2011, 74, 772–777. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Kawase, T.; Horimizu, M.; Okuda, K.; Wolff, L.F.; Yoshie, H. A proposed protocol for the standardized preparation of PRF membranes for clinical use. Biologicals 2012, 40, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Kawase, T.; Okuda, K.; Wolff, L.F.; Yoshie, H. In vitro immunological and biological evaluations of the angiogenic potential of platelet-rich fibrin preparations: a standardized comparison with PRP preparations. Int. J. Implant. Dent. 2015, 1, 31. [Google Scholar]
- Nishiyama, K.; Okudera, T.; Watanabe, T.; Isobe, K.; Suzuki, M.; Masuki, H.; Okudera, H.; Uematsu, K.; Nakata, K.; Kawase, T. Basic characteristics of plasma rich in growth factors (PRGF): Blood cell components and biological effects. Clin. Exp. Dent. Res. 2016, 2. [Google Scholar] [CrossRef]
- Kawase, T.; Hayama, K.; Tsuchimochi, M.; Nagata, M.; Okuda, K.; Yoshie, H.; Burns, D.M.; Nakata, K. Evaluating the Safety of Somatic Periosteal Cells by Flow-Cytometric Analysis Monitoring the History of DNA Damage. Biopreserv. Biobank. 2016, 14, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Kawase, T.; Tanaka, T.; Nishimoto, T.; Okuda, K.; Nagata, M.; Burns, D.M.; Yoshie, H. Improved adhesion of human cultured periosteal sheets to a porous poly(L-lactic acid) membrane scaffold without the aid of exogenous adhesion biomolecules. J. Biomed. Mater. Res. A 2011, 98, 100–113. [Google Scholar] [CrossRef] [PubMed]
- Murugappa, S.; Kunapuli, S.P. The role of ADP receptors in platelet function. Front. Biosci. 2006, 11, 1977–1986. [Google Scholar] [CrossRef] [PubMed]
- Daugirdas, J.T.; Bernardo, A.A. Hemodialysis effect on platelet count and function and hemodialysis-associated thrombocytopenia. Kidney Int. 2012, 82, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Paniccia, R.; Priora, R.; Liotta, A.A.; Abbate, R. Platelet function tests: A comparative review. Vasc. Health Risk Manag. 2015, 11, 133–148. [Google Scholar] [CrossRef] [PubMed]
- Boold Centers of AMERICA. Platelets. Available online: http://bca.coop/products-services/blood-products/platelets/ (assessed on 4 July 2017).
- Wallvik, J.; Akerblom, O. Platelet concentrates stored at 22 degrees C need oxygen. The significance of plastics in platelet preservation. Vox. Sang. 1983, 45, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Racz, Z.; Hasko, F. Room temperature storage of pooled platelet concentrates in gas-permeable plastic bags for five days. Haematologia 1989, 22, 89–96. [Google Scholar] [PubMed]
- Devine, D.V.; Serrano, K. The platelet storage lesion. Clin Lab Med 2010, 30, 475–487. [Google Scholar] [CrossRef] [PubMed]
- Sonker, A.; Dubey, A. Determining the Effect of Preparation and Storage: An Effort to Streamline Platelet Components as a Source of Growth Factors for Clinical Application. Transfus. Med. Hemother. 2015, 42, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Bausset, O.; Giraudo, L.; Veran, J.; Magalon, J.; Coudreuse, J.M.; Magalon, G.; Dubois, C.; Serratrice, N.; Dignat-George, F.; Sabatier, F. Formulation and storage of platelet-rich plasma homemade product. Biores. Open Access 2012, 1, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Moore, G.W.; Maloney, J.C.; Archer, R.A.; Brown, K.L.; Mayger, K.; Bromidge, E.S.; Najafi, M.F. Platelet-rich plasma for tissue regeneration can be stored at room temperature for at least five days. Br. J. Biomed. Sci. 2017, 74, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Shevkoplyas, S.S. Anaerobic storage of red blood cells. Blood Transfus. 2010, 8, 220–236. [Google Scholar] [CrossRef] [PubMed]
- Gulliksson, H. Platelet storage media. Vox Sang. 2014, 107, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Dhurat, R.; Sukesh, M. Principles and Methods of Preparation of Platelet-Rich Plasma: A Review and Author's Perspective. J. Cutan. Aesthet. Surg. 2014, 7, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Bogdanova, A.; Makhro, A.; Wang, J.; Lipp, P.; Kaestner, L. Calcium in Red Blood Cells—A Perilous Balance. Int. J. Mol. Sci. 2013, 14, 9848–9872. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, S.; Kumar, A. Leukocyte depletion for safe blood transfusion. Biotechnol. J. 2009, 4, 1140–1151. [Google Scholar] [CrossRef] [PubMed]
- How quickly do different cells in the body replace themselves? Available online: http://book.bionumbers.org/how-quickly-do-different-cells-in-the-body-replace-themselves/ (assessed on 8 July 2017).
- Van der Meer, P.F.; de Wildt-Eggen, J. The effect of whole-blood storage time on the number of white cells and platelets in whole blood and in white cell-reduced red cells. Transfusion 2006, 46, 589–594. [Google Scholar] [CrossRef] [PubMed]
- Hess, J.R. Conventional blood banking and blood component storage regulation: Opportunities for improvement. Blood Transfus. 2010, 8, s9–s15. [Google Scholar] [CrossRef] [PubMed]
- Hogman, C.F.; Gong, J.; Eriksson, L.; Hambraeus, A.; Johansson, C.S. White cells protect donor blood against bacterial contamination. Transfusion 1991, 31, 620–626. [Google Scholar] [CrossRef] [PubMed]



© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kawabata, H.; Isobe, K.; Watanabe, T.; Okudera, T.; Nakamura, M.; Suzuki, M.; Ryu, J.; Kitamura, Y.; Okudera, H.; Okuda, K.; et al. Quality Assessment of Platelet-Rich Fibrin-Like Matrix Prepared from Whole Blood Samples after Extended Storage. Biomedicines 2017, 5, 57. https://doi.org/10.3390/biomedicines5030057
Kawabata H, Isobe K, Watanabe T, Okudera T, Nakamura M, Suzuki M, Ryu J, Kitamura Y, Okudera H, Okuda K, et al. Quality Assessment of Platelet-Rich Fibrin-Like Matrix Prepared from Whole Blood Samples after Extended Storage. Biomedicines. 2017; 5(3):57. https://doi.org/10.3390/biomedicines5030057
Chicago/Turabian StyleKawabata, Hideo, Kazushige Isobe, Taisuke Watanabe, Toshimitsu Okudera, Masayuki Nakamura, Masashi Suzuki, Jietsu Ryu, Yutaka Kitamura, Hajime Okudera, Kazuhiro Okuda, and et al. 2017. "Quality Assessment of Platelet-Rich Fibrin-Like Matrix Prepared from Whole Blood Samples after Extended Storage" Biomedicines 5, no. 3: 57. https://doi.org/10.3390/biomedicines5030057