vQRS Based on Hybrids of CNT with PMMA-POSS and PS-POSS Copolymers to Reach the Sub-PPM Detection of Ammonia and Formaldehyde at Room Temperature Despite Moisture
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Sensor’s Fabrication and Chemo-Resistive Characterization
3. Results and Discussion
3.1. vQRS Morphological Characterization
3.2. vQRS Chemo-Resistive Characterization
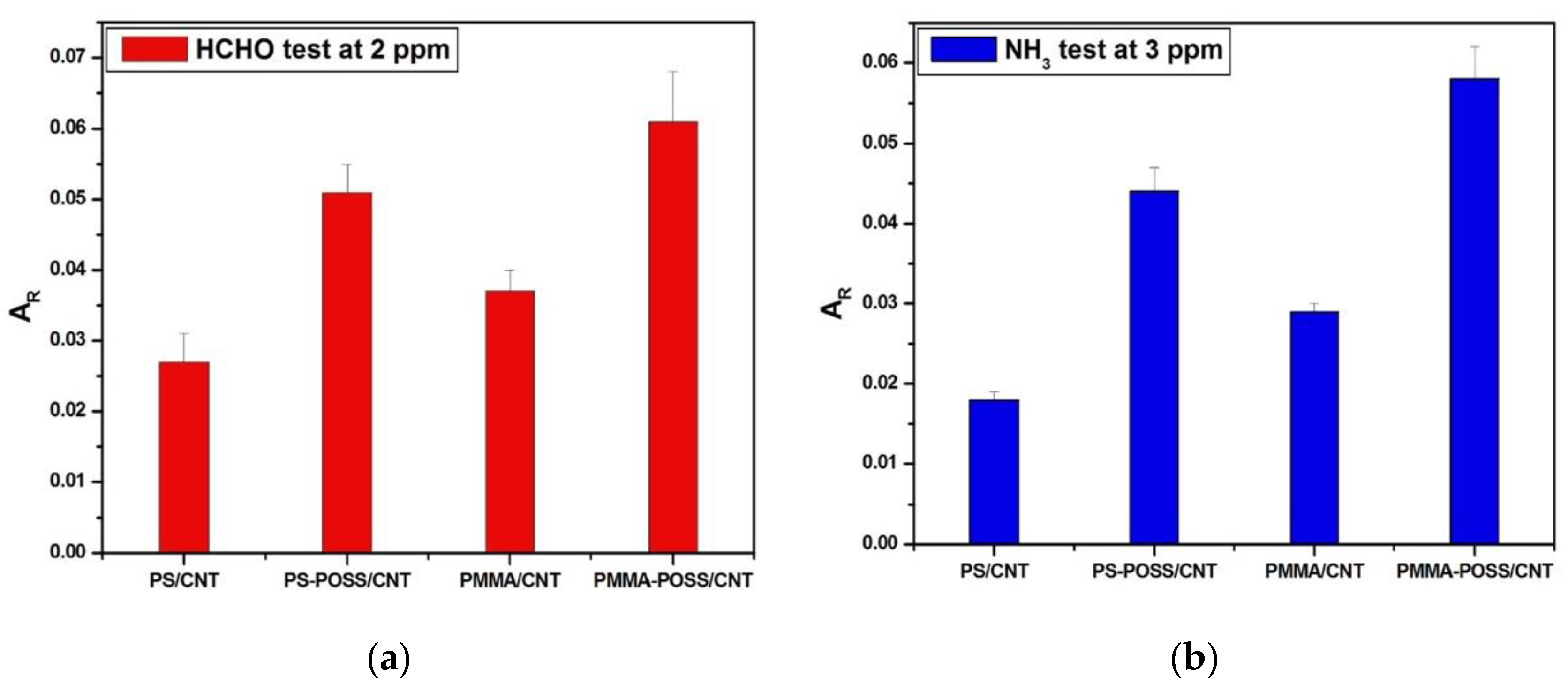
3.2.1. Effect of POSS on the Chemo-Resistive Response?
3.2.2. Principle of CH2O and NH3 Detection with vQRS
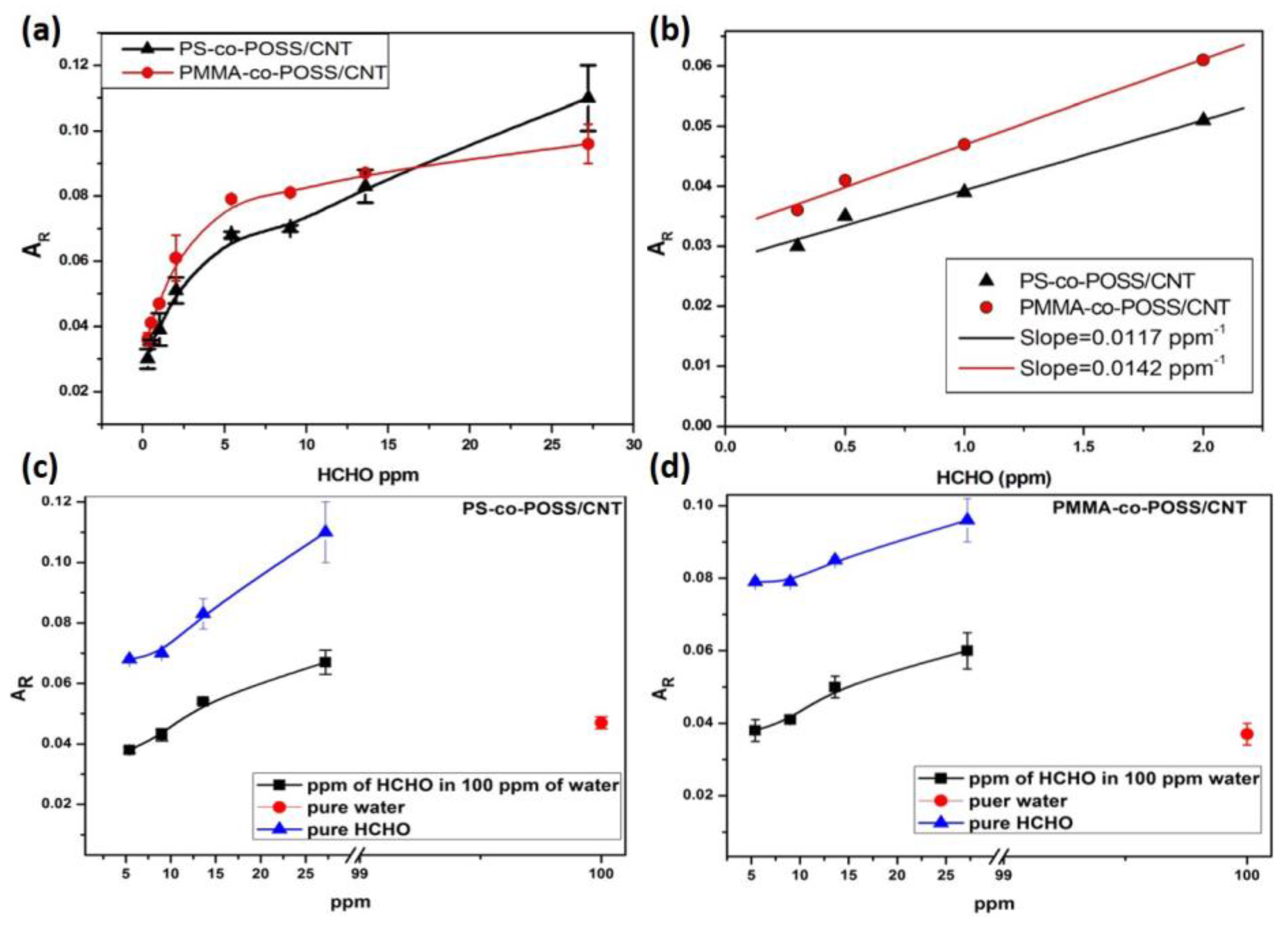
3.2.3. Formaldehyde Sensing with PS-co-POSS/CNT and PMMA-co-POSS/CNT
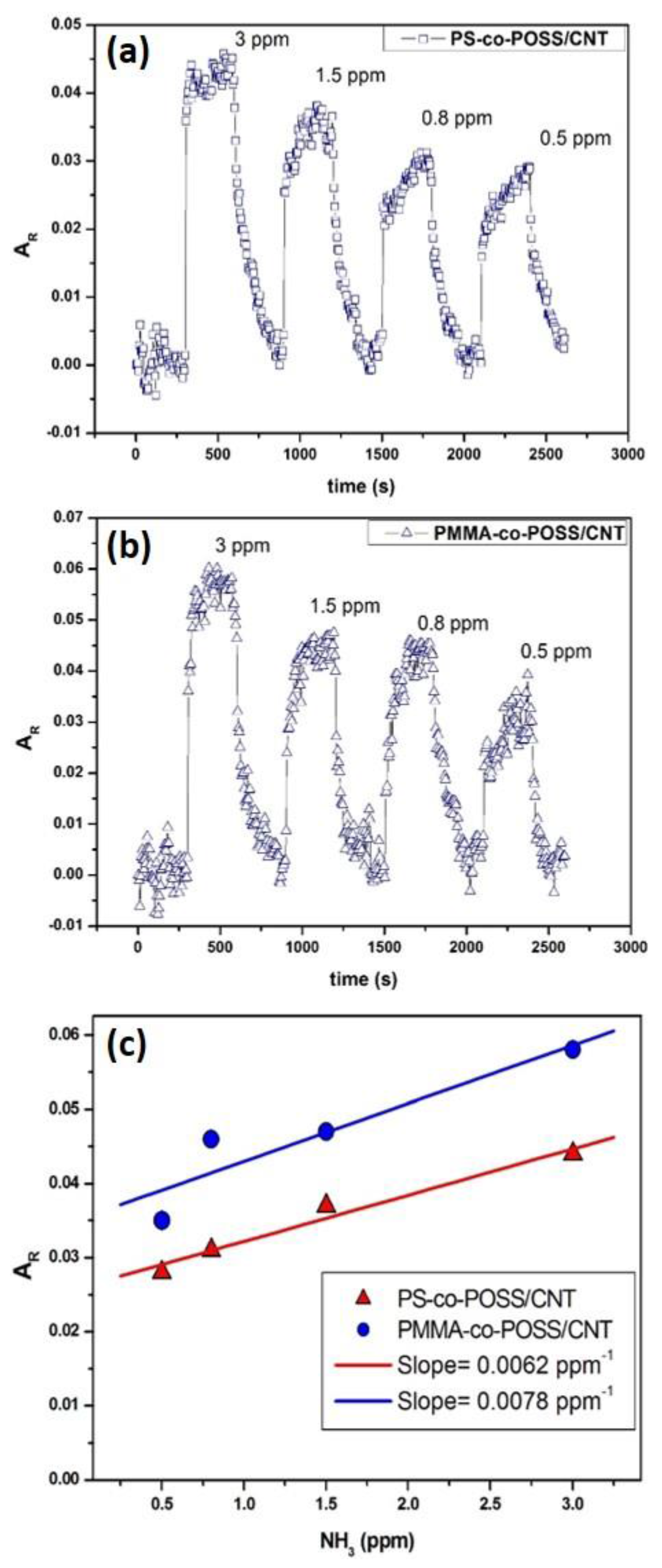
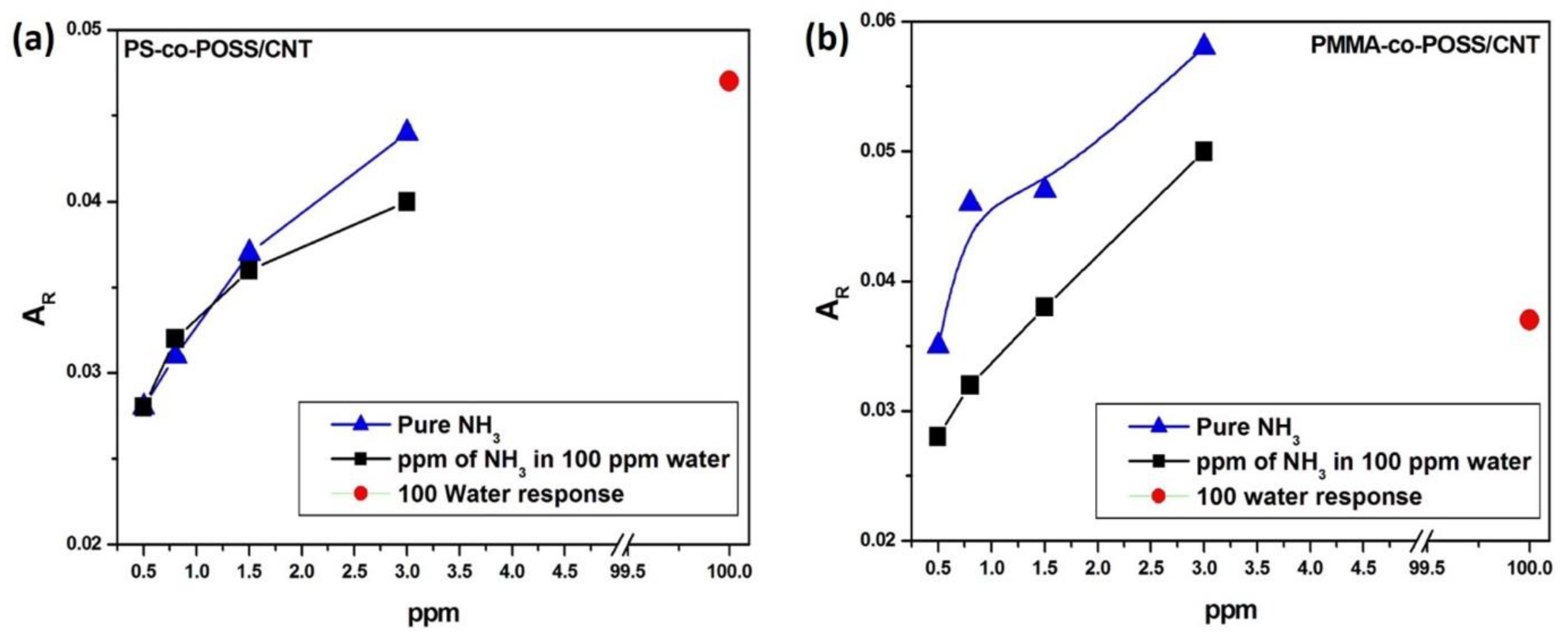
3.2.4. Ammonia Sensing with PS-co-POSS/CNT and PMMA-co-POSS/CNT
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
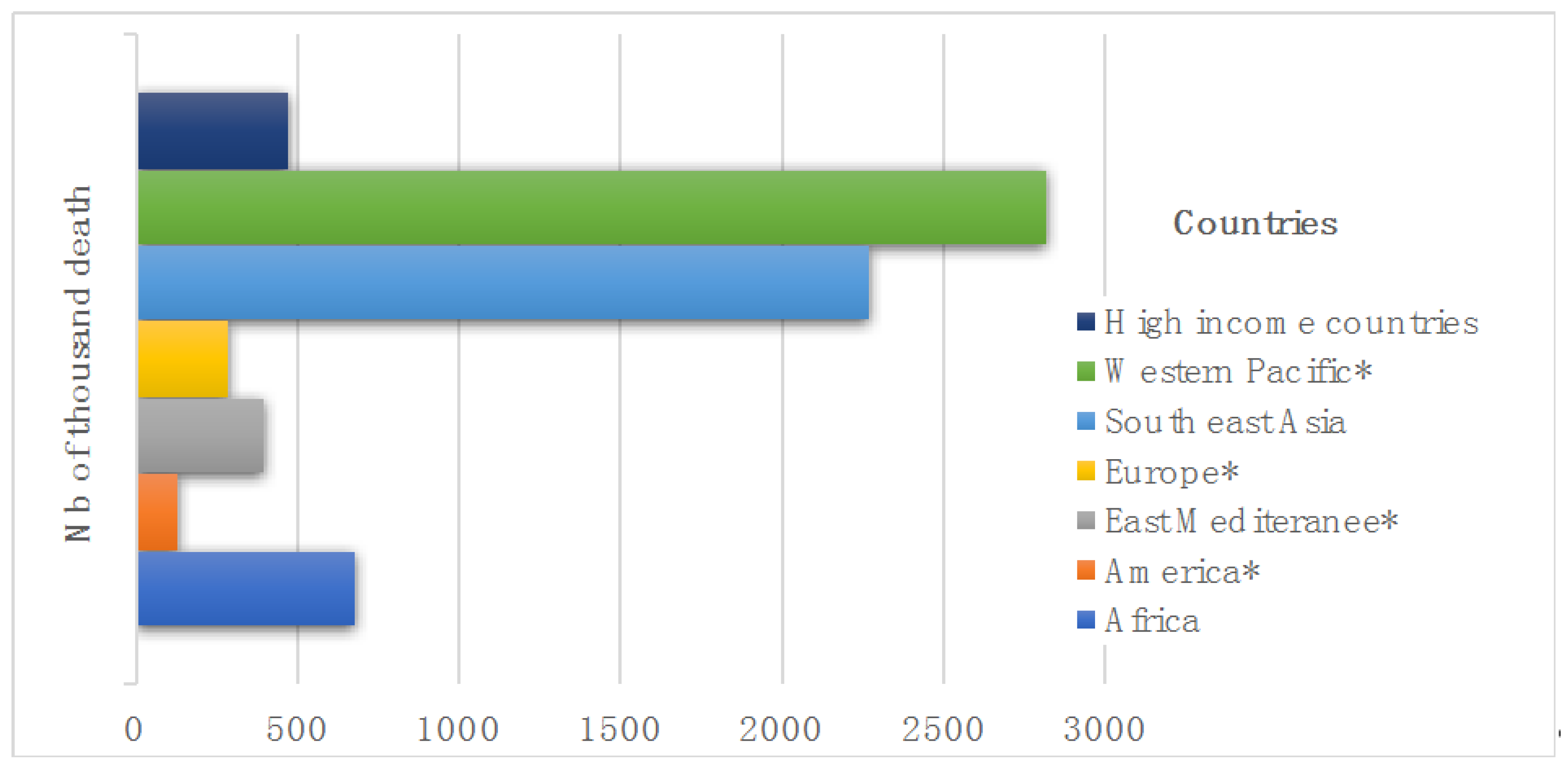
- Lindmeier, C.; Osseiran, N.; Chriscaden, K. An Estimated 12.6 Million Deaths Each Year are Attributable to Unhealthy Environments; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- Cancer Today: Population Fact Sheets, World Estimated Incidence and Mortality of Cancer, Fact Sheet. 2012. Available online: http://globocan.iarc.fr/Pages/fact_sheets_population.aspx (accessed on 11 July 2017).
- Broza, Y.Y.; Kremer, R.; Tisch, U.; Gevorkyan, A.; Shiban, A.; Best, L.A. A nanomaterial-based breath test for short-term follow-up after lung tumor resection. Nanomed. Nanotechnol. Biol. Med. 2013, 9, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Broza, Y.Y.; Zuri, L.; Haick, H. Combined volatolomics for monitoring of human body chemistry. Sci. Rep. 2014, 4, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Hashim, D.; Boffetta, P. Occupational and environmental exposures and cancers in developing countries. Ann. Glob. Health 2014, 80, 393–411. [Google Scholar] [CrossRef] [PubMed]
- Mount, G.H.; Rumburg, B.; Havig, J.; Lamb, B.; Westberg, H.; Yonge, D. Measurement of atmospheric ammonia at a dairy using differential optical absorption spectroscopy in the mid-ultraviolet. Atmos. Environ. 2002, 36, 1799–1810. [Google Scholar] [CrossRef]
- Pijolat, C.; Pupier, C.; Sauvan, M.; Tournier, G.; Lalauze, R. Gas detection for automotive pollution control. Sens. Actuators B Chem. 1999, 59, 195–202. [Google Scholar] [CrossRef]
- Kohl, D. Function and applications of gas sensors. J. Phys. D Appl. Phys. 2001, 34, R125. [Google Scholar] [CrossRef]
- Ampuero, S.; Bosset, J.O. The electronic nose applied to dairy products: A review. Sens. Actuators B Chem. 2003, 94, 1–12. [Google Scholar] [CrossRef]
- Greenstein, G.R. The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals (14th edition). Ref. Rev. 2006, 21, 40. [Google Scholar]
- Zakrzewska, K. Mixed oxides as gas sensors. Thin Solid Films 2001, 391, 229–238. [Google Scholar] [CrossRef]
- Wojkiewicz, J.L.; Bliznyuk, V.N.; Carquigny, S.; Elkamchi, N.; Redon, N.; Lasri, T. Nanostructured polyaniline-based composites for ppb range ammonia sensing. Sens. Actuators B Chem. 2011, 160, 1394–1403. [Google Scholar] [CrossRef]
- Peeters, R.; Berden, G.; Apituley, A.; Meijer, G. Open-path trace gas detection of ammonia based on cavity-enhanced absorption spectroscopy. Appl. Phys. B 2000, 71, 231–236. [Google Scholar] [CrossRef]
- Tiggelaar, R.M.; Veenstra, T.T.; Sanders, R.G.P.; Berenschot, E.; Gardeniers, H.; Elwenspoek, M. Analysis systems for the detection of ammonia based on micromachined components modular hybrid versus monolithic integrated approach. Sens. Actuators B Chem. 2003, 92, 25–36. [Google Scholar] [CrossRef]
- Winquist, F.; Spetz, A.; Lundström, I.; Danielsson, B. Determination of ammonia in air and aqueous samples with a gas-sensitive semiconductor capacitor. Anal. Chim. Acta 1984, 164, 127–138. [Google Scholar] [CrossRef]
- Xu, C.N.; Miura, N.; Ishida, Y.; Matsuda, K.; Yamazoe, N. Selective detection of NH3 over NO in combustion exhausts by using Au and MoO3 doubly promoted WO3 element. Sens. Actuators B Chem. 2000, 65, 163–165. [Google Scholar] [CrossRef]
- Cantalini, C.; Valentini, L.; Armentano, I.; Lozzi, L.; Kenny, J.M.; Santucci, S. Sensitivity to NO2 and cross-sensitivity analysis to NH3, ethanol and humidity of carbon nanotubes thin film prepared by PECVD. Sens. Actuators B Chem. 2003, 95, 195–202. [Google Scholar] [CrossRef]
- Arab, M.; Berger, F.; Picaud, F.; Ramseyer, C.; Glory, J.; Mayne-L’Hermite, M. Direct growth of the multi-walled carbon nanotubes as a tool to detect ammonia at room temperature. Chem. Phys. Lett. 2006, 433, 175–181. [Google Scholar] [CrossRef]
- Battie, Y.; Ducloux, O.; Thobois, P.; Dorval, N.; Lauret, J.S.; Attal-Trétout, B. Gas sensors based on thick films of semi-conducting single walled carbon nanotubes. Carbon N. Y. 2011, 49, 3544–3552. [Google Scholar] [CrossRef]
- Feller, J.F.; Gatt, N.; Kumar, B.; Castro, M. Selectivity of chemoresistive sensors made of chemically functionalized carbon nanotube random networks for volatile organic compounds (VOC). ChemoSensors 2014, 2, 26–40. [Google Scholar] [CrossRef]
- Kumar, B.; Castro, M.; Feller, J.F. Quantum resistive vapour sensors made of polymer coated carbon nanotubes random networks for biomarkers detection. Chem. Sens. 2013, 3, 1–7. [Google Scholar]
- Li, Y.; Wang, H.; Yang, M. n-Type gas sensing characteristics of chemically modified multi-walled carbon nanotubes and PMMA composite. Sens. Actuators B Chem. 2007, 121, 496–500. [Google Scholar] [CrossRef]
- Kumar, B.; Feller, J.F.; Castro, M.; Lu, J. Conductive bio-Polymer nano-Composites (CPC): Chitosan-carbon nanotube transducers assembled via spray layer-by-layer for volatile organic compound sensing. Talanta 2010, 81, 908–915. [Google Scholar] [CrossRef] [PubMed]
- Kumar, B.; Castro, M.; Feller, J.F. Poly(lactic acid)–multiwall carbon nanotube conductive biopolymer nanocomposite vapour sensors. Sens. Actuators B Chem. 2012, 161, 621–628. [Google Scholar] [CrossRef]
- Huyen, D.N.; Tung, N.T.; Vinh, T.D.; Thien, N.D. Synergistic effects in the gas sensitivity of polypyrrole/single wall carbon nanotube composites. Sensors 2012, 12, 7965–7974. [Google Scholar] [CrossRef] [PubMed]
- Van Hieu, N.; Dung, N.Q.; Tam, P.D.; Trung, T.; Chien, N.D. Thin film polypyrrole/SWCNTs nanocomposites-based NH3 sensor operated at room temperature. Sens. Actuators B Chem. 2009, 140, 500–507. [Google Scholar] [CrossRef]
- Setka, M.; Drbohlavova, J.; Hubalek, J. Nanostructured polypyrrole-based ammonia and volatile organic compound sensors. Sensors 2017, 17, 562. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Hussain, S.; Singh, S.; Islam, S.S. MWCNT-conducting polymer composite based ammonia gas sensors: A new approach for complete recovery process. Sens. Actuators B Chem. 2014, 194, 213–219. [Google Scholar] [CrossRef]
- Huotari, J.; Lappalainen, J.; Eriksson, J.; Bjorklund, R.; Heinonen, E.; Miinalainen, I. Synthesis of nanostructured solid-state phases of V7O16 and V2O5 compounds for ppb-level detection of ammonia. J. Alloy. Compd. 2016, 675, 433–440. [Google Scholar] [CrossRef]
- Krivetskiy, V.; Malkov, I.; Garshev, A.; Mordvinova, N.; Lebedev, O.I.; Dolenko, S. Chemically modified nanocrystalline SnO2-based materials for nitrogen-containing gases detection using gas sensor array. J. Alloy. Compd. 2017, 691, 514–523. [Google Scholar] [CrossRef]
- Dalavi, D.S.; Harale, N.S.; Mulla, I.S.; Rao, V.K.; Patil, V.B.; Kim, I.Y. Nanoporous network of nickel oxide for ammonia gas detection. Mater. Lett. 2015, 146, 103–107. [Google Scholar] [CrossRef]
- Kwon, O.S.; Hong, J.Y.; Park, S.J.; Jang, Y.; Jang, J. Resistive gas sensors based on precisely size-controlled polypyrrole nanoparticles: Effects of particle size and deposition method. J. Phys. Chem. C 2010, 114, 18874–18879. [Google Scholar] [CrossRef]
- Xiang, C.; Jiang, D.; Zou, Y.; Chu, H.; Qiu, S.; Zhang, H. Ammonia sensor based on polypyrrole-graphene nanocomposite decorated with titania nanoparticles. Ceram. Int. 2015, 41, 6432–6438. [Google Scholar] [CrossRef]
- Korpan, Y.I.; Gonchar, M.V.; Sibirny, A.A.; Martelet, C.; El’skaya, A.V.; Gibson, T.D. Development of highly selective and stable potentiometric sensors for formaldehyde determination. Biosens. Bioelectron. 2000, 15, 77–83. [Google Scholar] [CrossRef]
- Kawamura, K.; Kerman, K.; Fujihara, M.; Nagatani, N.; Hashiba, T.; Tamiya, E. Development of a novel hand-held formaldehyde gas sensor for the rapid detection of sick building syndrome. Sens. Actuators B Chem. 2005, 105, 495–501. [Google Scholar] [CrossRef]
- Que, Z.; Furuno, T.; Katoh, S.; Nishino, Y. Evaluation of three test methods in determination of formaldehyde emission from particleboard bonded with different mole ratio in the urea-formaldehyde resin. Build. Environ. 2007, 42, 1242–1249. [Google Scholar] [CrossRef]
- An, J.Y.; Kim, S.; Kim, H.J.; Seo, J. Emission behavior of formaldehyde and TVOC from engineered flooring in under heating and air circulation systems. Build. Environ. 2010, 45, 1826–1833. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Air Quality Guidelines for Europe. Available online: http://www.euro.who.int/en/publications/abstracts/air-quality-guidelines-for-europe (accessed on 11 July 2017).
- U.S. Dept Health & Human Services. Occupational Safety and Health Guideline for Formaldehyde Potential Human Carcinogen; NIOSH: Washington, DC, USA, 1988.
- Suzuki, Y.; Nakano, N.; Suzuki, K. Portable sick house syndrome gas monitoring system based on novel colorimetric reagents for the highly selective and sensitive detection of formaldehyde. Environ. Sci. Technol. 2003, 37, 5695–5700. [Google Scholar] [CrossRef] [PubMed]
- Achmann, S.; Hermann, M.; Hilbrig, F.; Jérôme, V.; Hämmerle, M.; Freitag, R. Direct detection of formaldehyde in air by a novel NAD+- and glutathione-independent formaldehyde dehydrogenase-based biosensor. Talanta 2008, 75, 786–791. [Google Scholar] [CrossRef] [PubMed]
- Lv, P.; Tang, Z.A.; Yu, J.; Zhang, F.T.; Wei, G.F.; Huang, Z.X. Study on a micro-gas sensor with SnO2-NiO sensitive film for indoor formaldehyde detection. Sens. Actuators B Chem. 2008, 132, 74–80. [Google Scholar] [CrossRef]
- Li, X.; Liu, J.; Guo, H.; Zhou, X.; Wang, C.; Sun, P. Au@In2O3 core-shell composites: A metal-semiconductor heterostructure for gas sensing applications. RSC Adv. 2015, 5, 545–551. [Google Scholar] [CrossRef]
- Barsan, N.; Koziej, D.; Weimar, U. Metal oxide-based gas sensor research: How to? Sens. Actuators B Chem. 2007, 121, 18–35. [Google Scholar] [CrossRef]
- Zhang, D.; Liu, J.; Jiang, C.; Liu, A.; Xia, B. Quantitative detection of formaldehyde and ammonia gas via metal oxide-modified graphene-based sensor array combining with neural network model. Sens. Actuators B Chem. 2017, 240, 55–65. [Google Scholar] [CrossRef]
- Zhang, D.; Jiang, C.; Li, P.; Sun, Y. Layer-by-layer self-assembly of Co3O4 nanorod-decorated MoS2 nanosheet-based nanocomposite toward high-performance ammonia detection. ACS Appl. Mater. Interfaces 2017, 9, 6462–6471. [Google Scholar] [CrossRef] [PubMed]
- Fang, F.; Bai, L.; Sun, H.; Kuang, Y.; Sun, X.; Shi, T. Hierarchically porous indium oxide nanolamellas with ten-parts-per-billion-level formaldehyde-sensing performance. Sens. Actuators B Chem. 2015, 206, 714–720. [Google Scholar] [CrossRef]
- Park, H.J.; Choi, N.J.; Kang, H.; Jung, M.Y.; Park, J.W.; Park, K.H. A ppb-level formaldehyde gas sensor based on CuO nanocubes prepared using a polyol process. Sens. Actuators B Chem. 2014, 203, 282–288. [Google Scholar] [CrossRef]
- Güntner, A.T.; Koren, V.; Chikkadi, K.; Righettoni, M.; Pratsinis, S.E. E-Nose sensing of low-ppb formaldehyde in gas mixtures at high relative humidity for breath screening of lung cancer? ACS Sens. 2016, 1, 528–535. [Google Scholar] [CrossRef]
- Nag, S.; Castro, M.; Choudhary, V.; Feller, J.F. Sulfonated poly(ether ether ketone) [SPEEK] nanocomposites based on hybrid nanocarbons for the detection and discrimination of some lung cancer VOC biomarkers. J. Mater. Chem. B Biol. Med. 2017, 5, 348–359. [Google Scholar] [CrossRef]
- Nag, S.; Sachan, A.; Castro, M.; Choudhary, V.; Feller, J.F. Spray layer-by-layer assembly of POSS functionalized CNT quantum chemo-resistive sensors with tuneable selectivity and ppm resolution to VOC biomarkers. Sens. Actuators B Chem. 2016, 222, 362–373. [Google Scholar] [CrossRef]
- Feller, J.F. Des Composites Polymères Conducteurs Aux éco-Composites Polymères, 2nd ed.; Universitaires Européennes (EUE): Sarrebruck, Germany, 2003. [Google Scholar]
- Zhang, W.; Müller, A.H.E. Architecture, self-assembly and properties of well-defined hybrid polymers based on polyhedral oligomeric silsequioxane (POSS). Prog. Polym. Sci. 2013, 38, 1121–1162. [Google Scholar] [CrossRef]
- Pielichowski, K.; Njuguna, J.; Janowski, B.; Pielichowski, J. Polyhedral oligomeric silsesquioxanes (POSS)-containing nanohybrid polymers. In Supramolecular Polymers Polymeric Betains Oligomers; Springer: Berlin/Heidelberg, Germany, 2006; pp. 225–296. [Google Scholar]
- Gnanasekaran, D.; Madhavpan, K.; Reddy, R.S.R. Developments of polyhedral oligomeric silsesquioxanes (POSS), POSS nanocomposites and their applications: A review. J. Sci. Ind. Res. 2009, 68, 437–464. (In India) [Google Scholar]
- Franchini, E.; Galy, J.; Gérard, J.-F.; Tabuani, D.; Medici, A. Influence of POSS structure on the fire retardant properties of epoxy hybrid networks. Polym. Degrad. Stab. 2009, 94, 1728–1736. [Google Scholar] [CrossRef]
- Raftopoulos, K.N.; Pielichowski, K. Segmental dynamics in hybrid polymer/POSS nanomaterials. Prog. Polym. Sci. 2016, 52, 136–187. [Google Scholar] [CrossRef]
- Tanaka, K.; Adachi, S.; Chujo, Y. Structure-property relationship of octa-substituted POSS in thermal and mechanical reinforcements of conventional polymers. J. Polym. Sci. Part A Polym. Chem. 2009, 47, 5690–5697. [Google Scholar] [CrossRef]
- Li, S.; Simon, G.P.; Matisons, J.G. Morphology of blends containing high concentrations of POSS nanoparticles in different polymer matrices. Polym. Eng. Sci. 2010, 50, 991–999. [Google Scholar] [CrossRef]
- Thakur, V.K.; Ding, G.; Ma, J.; Lee, P.S.; Lu, X. Hybrid Materials and Polymer Electrolytes for Electrochromic Device Applications. Adv. Mater. 2012, 24, 4071–4096. [Google Scholar] [CrossRef] [PubMed]
- Maitra, P.; Wunder, S.L. POSS based electrolytes for rechargeable lithium batteries. Electrochem. Solide-State Lett. 2004, 7, A88. [Google Scholar] [CrossRef]
- Ghanbari, H.; Cousins, B.G.; Seifalian, A.M. A Nanocage for Nanomedicine: Polyhedral Oligomeric Silsesquioxane (POSS). Macromol. Rapid Commun. 2011, 32, 1032–1046. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Mather, P.T. POSS Polymers: Physical Properties and Biomaterials Applications. Polym. Rev. 2009, 49, 25–63. [Google Scholar] [CrossRef]
- Fina, A.; Monticelli, O.; Camino, G. POSS-based hybrids by melt/reactive blending. J. Mater. Chem. 2010, 20, 9297–9305. [Google Scholar] [CrossRef]
- Nguyen, T.-P. Polymer-based nanocomposites for organic optoelectronic devices. A review. Surf. Coat. Technol. 2011, 206, 742–752. [Google Scholar] [CrossRef]
- Wu, J.; Haddad, T.S.; Mather, P.T. Vertex Group Effects in Entangled Polystyrene—Polyhedral Oligosilsesquioxane (POSS) Copolymers. Macromolecules 2009, 42, 1142–1152. [Google Scholar] [CrossRef]
- Wu, J.; Haddad, T.S.; Kim, G.-M.; Mather, P.T. Rheological Behavior of Entangled Polystyrene—Polyhedral Oligosilsesquioxane (POSS) Copolymers. Macromolecules 2007, 40, 544–554. [Google Scholar] [CrossRef]
- Gordon, M.; Taylor, J.S. Ideal copolymers and the second-order transitions of synthetic rubbers. i. non-crystalline copolymers. J. Appl. Chem. 1952, 2, 493–500. [Google Scholar] [CrossRef]
- Ma, X.-M.; Wang, B.; Zhang, M.-X.; Min, F.-F.; He, J. Synthesis and Thermal Characterizations of Pmma Nanocomposite Functionalized by Polyhedral Oligomeric Silsesquioxane, Phosphorus. Sulfur. Silicon Relat. Elem. 2013, 188, 1819–1826. [Google Scholar] [CrossRef]
- Kotal, A.; Si, S.; Paira, T.K.; Mandal, T.K. Synthesis of semitelechelic POSS-polymethacrylate hybrids by thiol-mediated controlled radical polymerization with unusual thermal behaviors. J. Polym. Sci. Part A Polym. Chem. 2008, 46, 1111–1123. [Google Scholar] [CrossRef]
- Gao, T.; Woodka, M.D.; Brunschwig, B.S.; Lewis, N.S. Chemiresistors for array-based vapor sensing using composites of carbon black with low volatility organic molecules. Chem. Mater. 2006, 18, 5193–5202. [Google Scholar] [CrossRef]
- Deng, H.; Skipa, T.; Zhang, R.; Lellinger, D.; Bilotti, E.; Alig, I. Effect of melting and crystallization on the conductive network in conductive polymer composites. Polymer 2009, 50, 3747–3754. [Google Scholar] [CrossRef]
- Feller, J.F.; Grohens, Y. Electrical response of Poly(styrene)/carbon black conductive polymer composites (CPC) to methanol, toluene, chloroform and styrene vapors as a function of filler nature and matrix tacticity. Synth. Met. 2005, 154, 193–196. [Google Scholar] [CrossRef]
- Tripathi, S.N.; Singh, S.; Malik, R.S.; Choudhary, V. Effect of multiwalled carbon nanotubes on the properties of poly(methyl methacrylate) in PMMA/CNT nanocomposites. Macromol. Symp. 2014, 341, 75–89. [Google Scholar] [CrossRef]
- Amr, I.T.; Al-Amer, A.; Al-Harthi, S.T.P.M.; Girei, S.A.; Sougrat, R. Effect of acid treated carbon nanotubes on mechanical, rheological and thermal properties of polystyrene nanocomposites. Compos. B Eng. 2011, 42, 1554–1561. [Google Scholar] [CrossRef]
- Kumar, B.; Castro, M.; Feller, J.F. Tailoring the chemo-resistive response of self-assembled polysaccharide-CNT sensors by chain conformation at tunnel junctions. Carbon N. Y. 2012, 50, 3627–3634. [Google Scholar] [CrossRef]
- Kumar, B.; Castro, M.; Feller, J.F. Controlled conductive junction gap for chitosan-carbon nanotube quantum resistive vapour sensors. J. Mater. Chem. 2012, 22, 10656–10664. [Google Scholar] [CrossRef]
- Feller, J.F.; Castro, M.; Kumar, B. Polymer-carbon nanotube conductive nanocomposites for sensing. In Polymer Carbon Nanotube Composites: Preparation, Properties and Applications, 1st ed.; McNally, T., Pötschke, P., Eds.; Woodhead Publishing Limited: Cambridge, UK, 2011; pp. 760–803. [Google Scholar]
- Bouvrée, A.; Feller, J.F.; Castro, M.; Grohens, Y.; Rinaudo, M. Conductive Polymer nano-bioComposites (CPC): Chitosan-carbon nanoparticle a good candidate to design polar vapour sensors. Sens. Actuators B Chem. 2009, 138, 138–147. [Google Scholar] [CrossRef]
- Chatterjee, S.; Castro, M.; Feller, J.F. An e-nose made of carbon nanotube based quantum resistive sensors for the detection of eighteen polar/nonpolar VOC biomarkers of lung cancer. J. Mater. Chem. B 2013, 1, 4563. [Google Scholar] [CrossRef]
- Joulazadeh, M.; Navarchian, A.H. Ammonia detection of one-dimensional nano-structured polypyrrole/metal oxide nanocomposites sensors. Synth. Met. 2015, 210(Part B), 404–411. [Google Scholar] [CrossRef]


| Transducer | LOD | Mechanism | Op. Temp °C | Reference |
|---|---|---|---|---|
| PMMA-POSS/CNT | 0.5 ppm | Chemo-resistive | 25 | This work |
| V2O5 and V7O16 film | 0.2 ppm | Chemo-resistive | 350 | [29] |
| SnO2-Nb-Pt nanocrystaline | 10 ppm | Chemo-resistive | 355 | [30] |
| Nanoporous NiO film | 20 ppm | Chemo-resistive | 250 | [31] |
| PPy NP | 5 ppm | Chemo-resistive | 25 | [32] |
| TiO2 NP deco GR/PPy | 1 ppm | Chemo-resistive | 25 | [33] |
| Material | Tg (°C) |
|---|---|
| PS (Mn = 50,000 g·mol−1) | 100 [73] |
| PMMA (Mn = 3.9 × 104 g·mol−1) | 100.2 [74] |
| PS-1% w/w CNT | 105 [75] |
| PMMA-5% w/w CNT | 102.5 [74] |
| PS-co-POSS/2% w/w CNT | 80.7 [this work] |
| PMMA-co-POSS/2% w/w CNT | 112.7 [this work] |
| Compound | δD (MPa1/2) | δP (MPa1/2) | δH (MPa1/2) | δt (MPa1/2) | Vm (cm3·mol−1) |
|---|---|---|---|---|---|
| NH3 | 13.7 | 16.7 | 18.8 | 28.63 | 25 |
| C2HO | 12.8 | 14.4 | 15.4 | 24.66 | 36.9 |
| PS | 5.9 | 18.7 | 3.5 | 19.92 | - |
| PMMA | 10.5 | 18.8 | 5.7 | 22.27 | - |
| χ12/Ar | PS | PMMA |
|---|---|---|
| NH3 | 0.765/0.018 | 0.407/0.028 |
| CH2O | 0.334/0.026 | 0.085/0.036 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sachan, A.; Castro, M.; Choudhary, V.; Feller, J.-F. vQRS Based on Hybrids of CNT with PMMA-POSS and PS-POSS Copolymers to Reach the Sub-PPM Detection of Ammonia and Formaldehyde at Room Temperature Despite Moisture. Chemosensors 2017, 5, 22. https://doi.org/10.3390/chemosensors5030022
Sachan A, Castro M, Choudhary V, Feller J-F. vQRS Based on Hybrids of CNT with PMMA-POSS and PS-POSS Copolymers to Reach the Sub-PPM Detection of Ammonia and Formaldehyde at Room Temperature Despite Moisture. Chemosensors. 2017; 5(3):22. https://doi.org/10.3390/chemosensors5030022
Chicago/Turabian StyleSachan, Abhishek, Mickael Castro, Veena Choudhary, and Jean-François Feller. 2017. "vQRS Based on Hybrids of CNT with PMMA-POSS and PS-POSS Copolymers to Reach the Sub-PPM Detection of Ammonia and Formaldehyde at Room Temperature Despite Moisture" Chemosensors 5, no. 3: 22. https://doi.org/10.3390/chemosensors5030022






