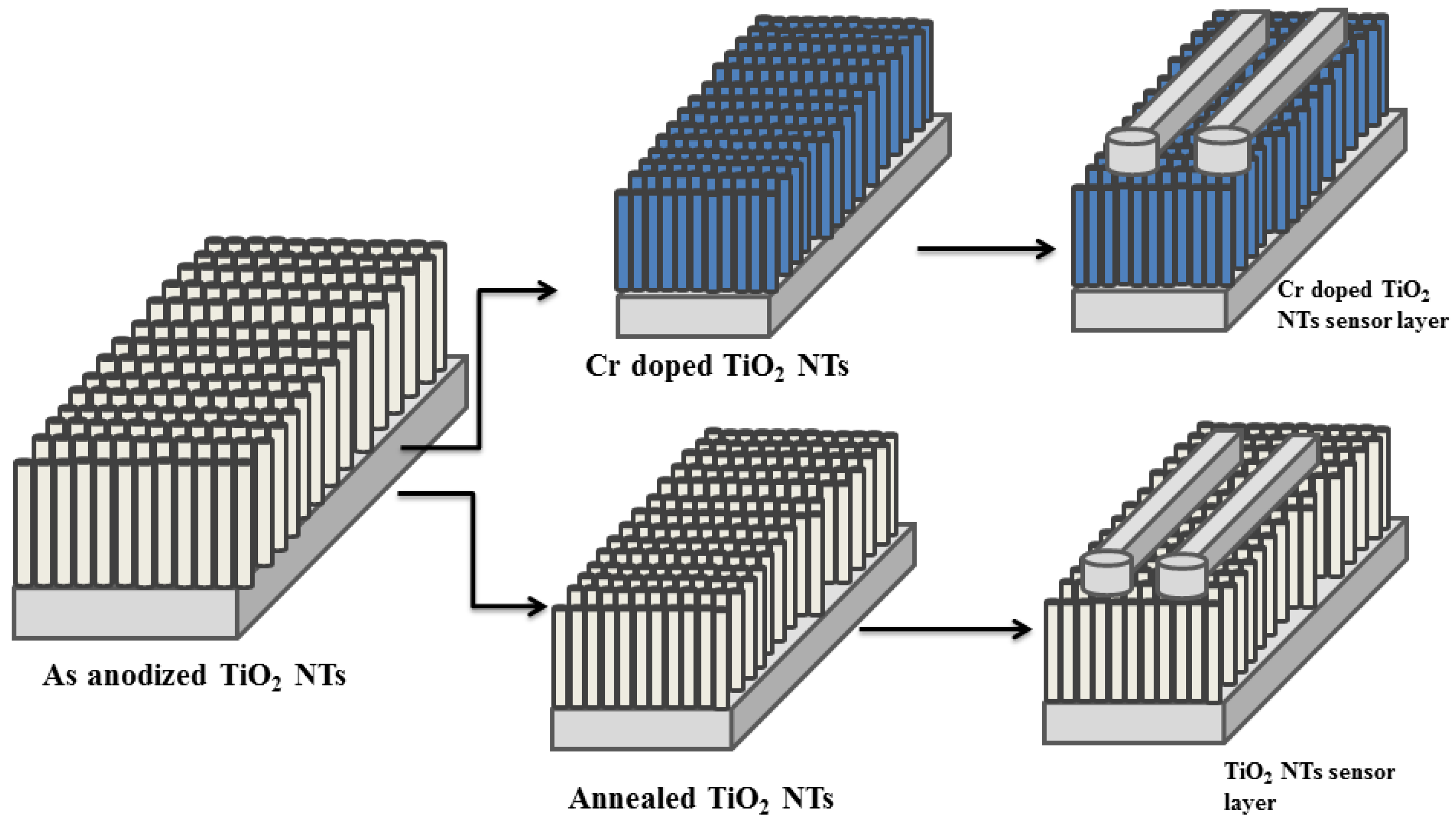
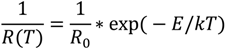3.3.1. Effect of Thickness on Gas Sensing Behavior
In order to understand the sensing mechanism of TiO
2 NTs, DC-response and AC-impedance measurements have been carried out for TiO
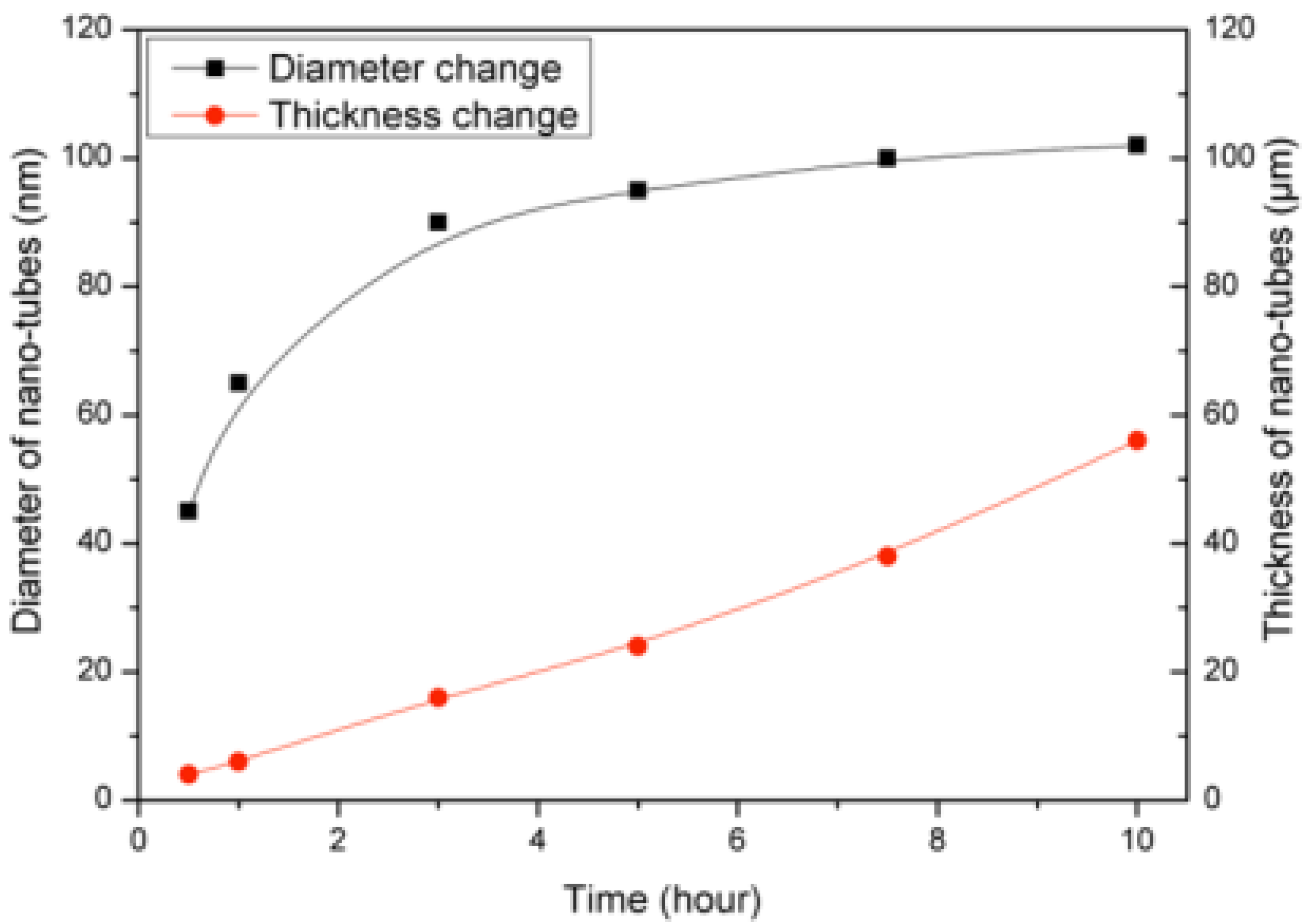
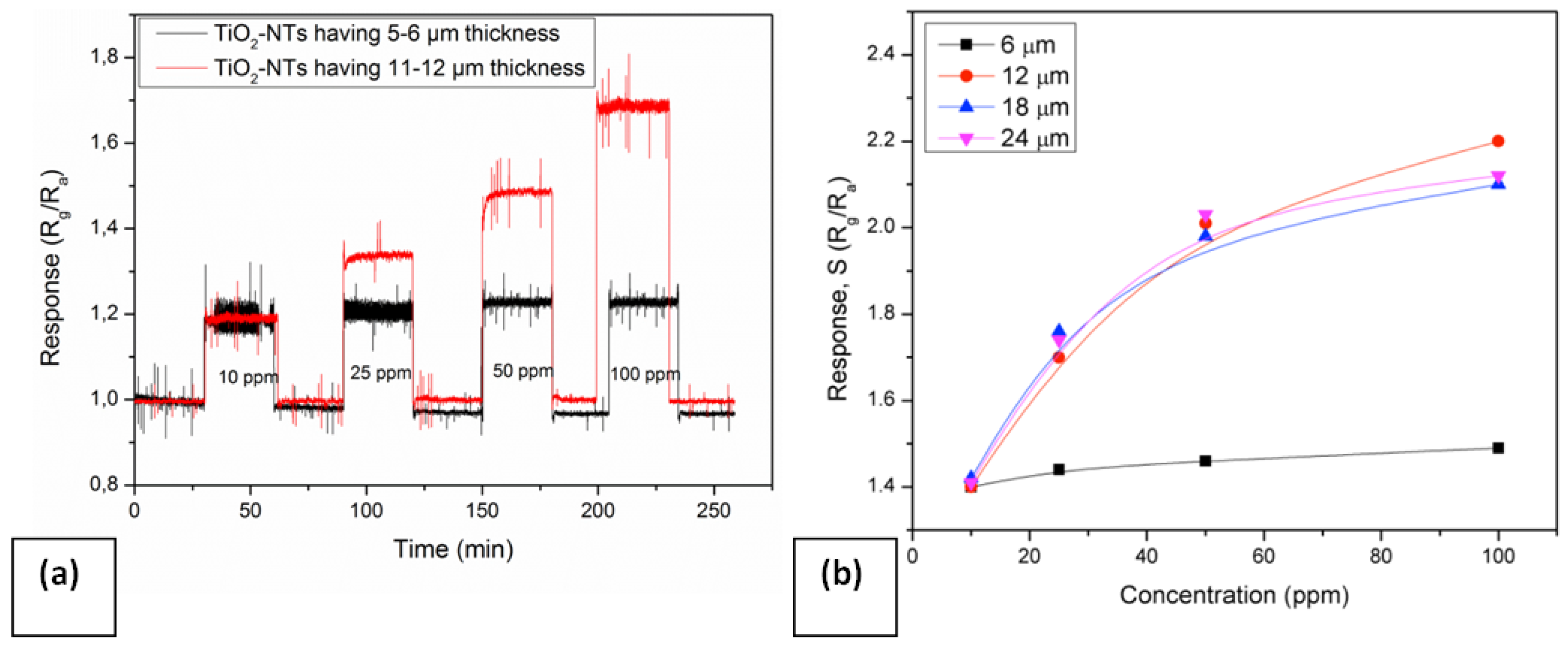
2 NTs having different thickness. The variation of sensor response with two different thicknesses of TiO
2 NT layers (6 to 12 µm) are given in
Figure 9a. TiO
2 NTs having a thickness of 11–12 µm detect the gas concentration variation more selectively, while those of 5–6 µm thickness yield similar type of response toward all tested concentrations (
i.e. 10 to 100 ppm). The sensor signals, S of the nanotubular layers with thicknesses varying from 6 to 24 µm are given in
Figure 9b. The increase in the thickness of TiO
2 NTs results in an improvement in the sensor response. The sensor signal,
S does not vary with the amount of test gas when thinner TiO
2 NTs (approx. 6 μm) are used. The thickness increase from 6 to 12 μm exhibits a higher sensitivity toward NO
2 depending on the amount of testing gas. On the other hand, no more sensitivity improvement can be detected when the thickness increases above 12 μm (
i.e., 18 or 24 μm). It appears that a critical thickness (d
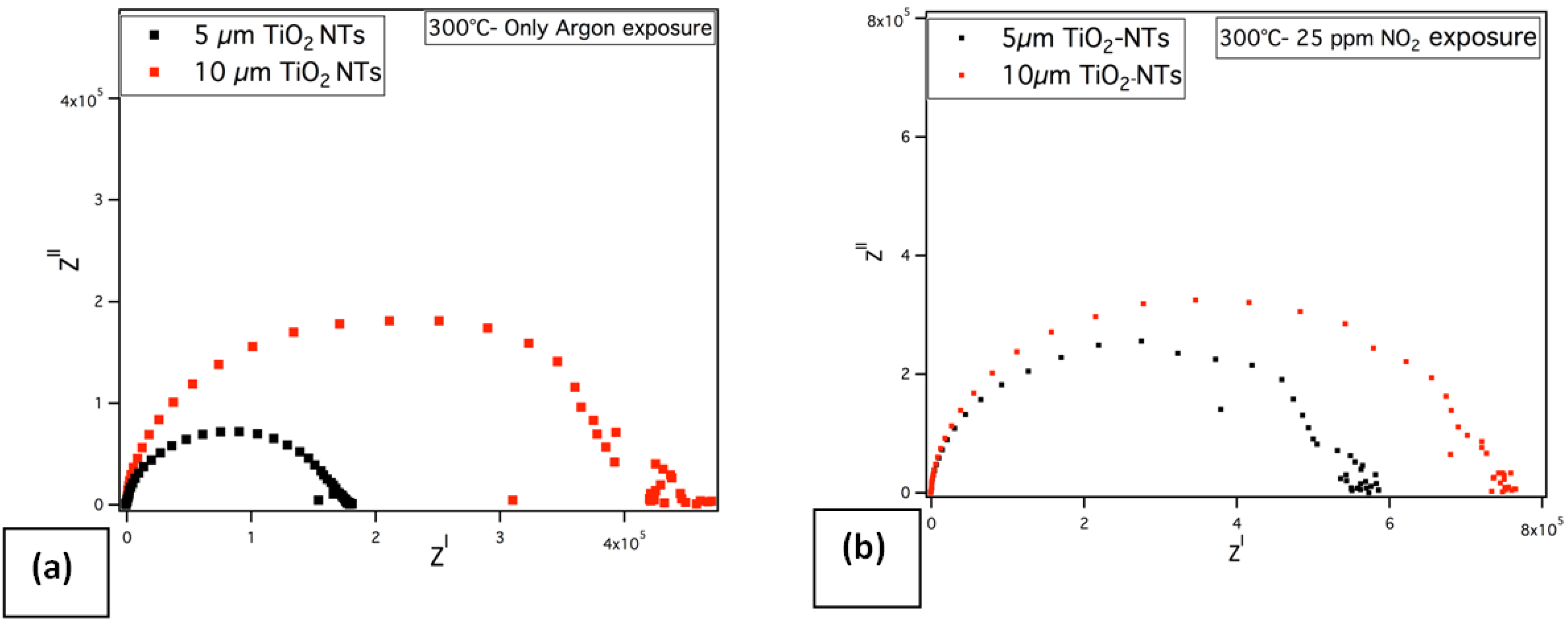
crit.) exists at about a thickness between 6 and 12 μm. The impedance measurements support also these results. The impedance measurements of TiO
2 NTs having different thicknesses have been carried out towards 25 ppm NO
2 in argon ambient gas atmosphere and in pure argon atmosphere (see
Figure 10). The impedance spectra of TiO
2 NTs show solely the typical semi-circle part at high frequencies. As the thickness of TiO
2 NTs increases, an increase in the complex resistance, Z
I has been observed.
Figure 9.
Responses of undoped TiO2 NTs for different thicknesses at 500 °C towards various NO2 concentrations (a) response versus time (b) response versus concentration.
Figure 9.
Responses of undoped TiO2 NTs for different thicknesses at 500 °C towards various NO2 concentrations (a) response versus time (b) response versus concentration.
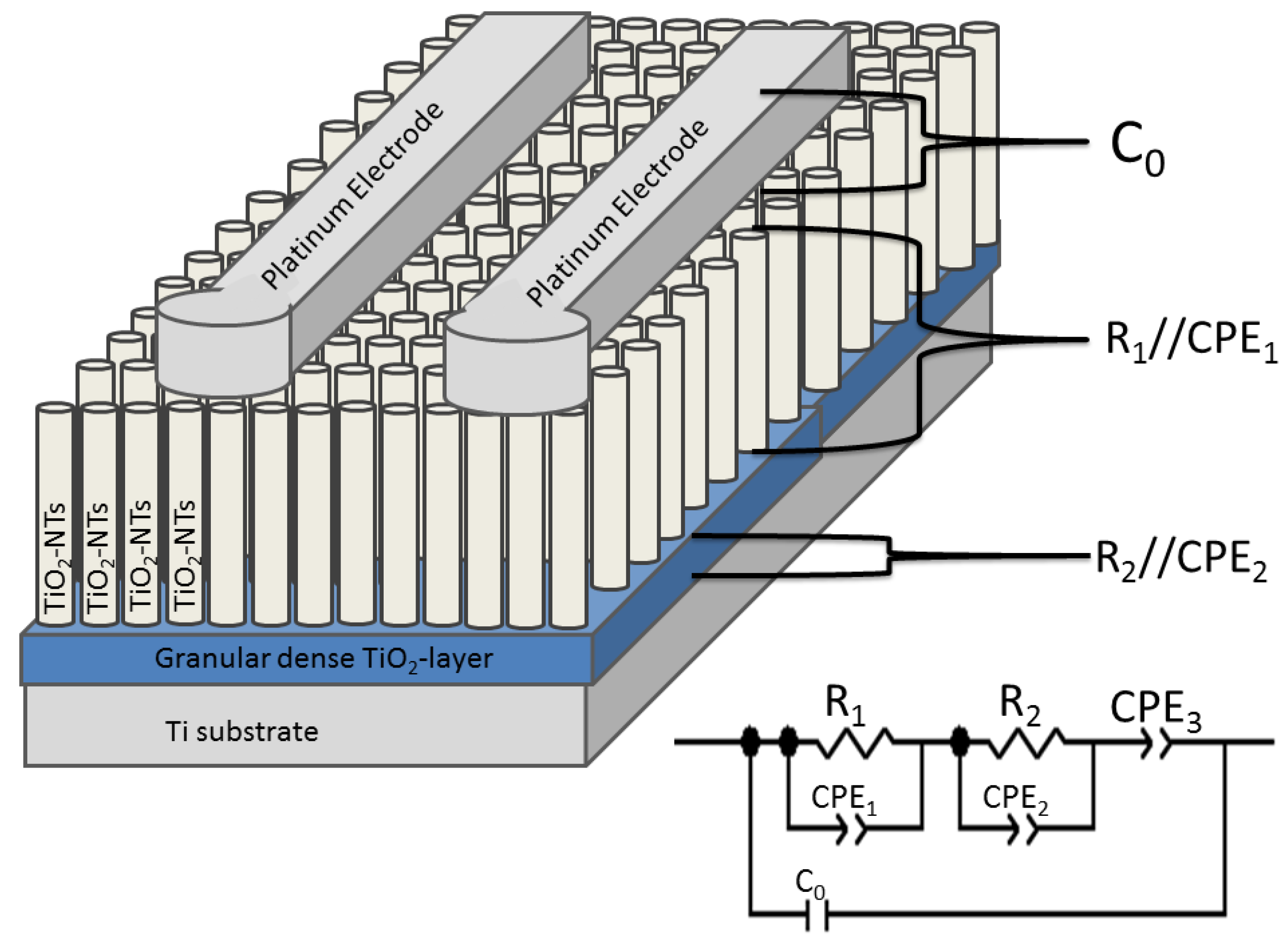
The equivalent circuit model employed in this study is given in
Figure 11. According to this model, a serial connected
R1||CPE1,
R2||CPE2 and
CPE3 was created, which was then parallel connected with a
C0 representing the capacity of platinum electrodes. In the equivalent circuit model, the first compound,
i.e.,
R1||CPE1 belongs to the TiO
2 NTs,
R1 representing the resistance of the TiO
2 NTs. The constant phase element (
CPE) is represented with the depressed semicircle in the corresponding Nyquist impedance plot as given in the literature [
11] and is a calculated parameter when the circuit element is behaving in between capacitor and resistor. The constant phase element (
CPE) is comprised by two components;
CPE-T and
CPE-P.
CPE-T is a pseudo capacitance which is called Q and
CPE-P is related to the semi-circle in the Nyquist plot (depressed semicircle), normally used for the notation `n`. By using CPE-P and CPE-T and resistance, one can calculate the true capacitance for the electrodes. If
CPE-P equals approximately to 1, then the
CPE turns theoretically to a capacitor,
C [
12].
The second parallel contacted elements (i.e., R2 and CPE2) belong to the dense bulk TiO2 layer present between the TiO2 NTs and Ti metal. Hereby, R2 represents the resistance of bulk TiO2 layer.
Lastly,
CPE3 defines gas depended interface in order to figure out the effect of diffusion and can be regarded as the Warburg Element [
12].
CPE3-T value is related to the amount of the carrier electrons on the system with a different mechanism such as diffusion [
13,
14,
15]. Lack of diffusion in the system can cause a decrease in the
CPE3-T values. It can be seen in
Table 1 that
CPE3-T is getting smaller with the increase of nano-tubular layer thickness and even constant above a certain thickness, indicating the reduction of diffusion above this NT thickness.
Thus, these results can be used to define the relation between sensor signal and layer thickness. On the contrary to general opinion, the present results reveal that sensor signal of TiO2 NT layers does not deteriorate on increase of layer thickness above a critical length (dcrit) despite the fact that slower gas diffusion is anticipated as the thickness of diffusion path increases. An increase in the thickness of TiO2 NTs from 12 to 24 μm resulted in a reduction of gas diffusion in the TiO2 NTs, but not in the sensitivity. At very low thickness state (e.g., 6 μm), the resistance values of the TiO2 NTs and the bulk TiO2 layer are close to each other. The electrons might have been transferred from one electrode to another electrode through both TiO2 NTs and the bulk TiO2 layer. However, after an increase in the nanotube thickness to a critical point (dcrit), the electron flow to and within the bulk TiO2 layers starts to be inhibited. Since less gas diffuses through the bottom layer of the nanotubes, less electron flow occurs in the bulk TiO2 layer. Therefore, it can be stated that the TiO2 NTs show higher resistance changes (which means higher sensitivity) as the layer thickness increases up to a critical thickness (dcrit).
Figure 10.
Nyquist plots of the TiO2 NTs at different layer thickness (a) by exposing only to argon (b) towards 25 ppm NO2 in argon carrier gas at 300 °C.
Figure 10.
Nyquist plots of the TiO2 NTs at different layer thickness (a) by exposing only to argon (b) towards 25 ppm NO2 in argon carrier gas at 300 °C.
Figure 11.
Equivalent circuit model of the sensor with TiO2 NTs indicating the corresponding sensor parts C0: capacitive, R1 and R2: resistive.
Figure 11.
Equivalent circuit model of the sensor with TiO2 NTs indicating the corresponding sensor parts C0: capacitive, R1 and R2: resistive.
Table 1.
Change in resistance (R1, R2) and CPE-values of undoped TiO2 NTs layer and the bulk TiO2 depending on NTs thickness towards different amounts of NO2 at each selected measurement. Colored columns indicate the constant values, and thus reduced diffusion.
Table 1.
Change in resistance (R1, R2) and CPE-values of undoped TiO2 NTs layer and the bulk TiO2 depending on NTs thickness towards different amounts of NO2 at each selected measurement. Colored columns indicate the constant values, and thus reduced diffusion.
| Thickness-Gas | R1
[Ω] | CPE1 | R2
[Ω] | CPE2 | CPE3 |
|---|
T
[sP/Ω] | P | T
[sP/Ω] | P | T
[s] | P |
|---|
| 6 μm—Argon | 3.5E5 | 1E-7 | 0.9 | 1.4E5 | 3.8E-6 | 0.24 | 0.001 | 0.1 |
| 12 μm—Argon | 5.2E5 | 1E-7 | 0.7 | 2.3E3 | 9E-6 | 0.5 | 1.2E-4 | 0.2 |
| 18 μm—Argon | 5.5E5 | 1E-7 | 0.9 | 2.4E3 | 3.8E-6 | 0.24 | 1.3E-4 | 0.1 |
| 24 μm—Argon | 5.6E5 | 1E-7 | 0.7 | 2.3E3 | 9E-6 | 0.5 | 1.3E-4 | 0.2 |
| 6 μm—25 ppm NO2 in Argon | 4.2E5 | 1.2E-8 | 0.9 | 3.3E5 | 6.6E-6 | 0.27 | 0.004 | 0.1 |
| 12 μm—25 ppm NO2 in Argon | 7.2E5 | 1E-7 | 0.8 | 4.5E3 | 4.9E-7 | 0.6 | 2.1E-4 | 0.2 |
| 18 μm—25 ppm NO2 in Argon | 7.4E5 | 1.2E-8 | 0.9 | 3.5E3 | 6.6E-6 | 0.27 | 2.6E-4 | 0.1 |
| 24 μm—25 ppm NO2 in Argon | 7.2E5 | 1E-7 | 0.8 | 2.5E3 | 4.9E-7 | 0.6 | 2.6E-4 | 0.2 |
3.3.2. Effect of Doping on Gas Sensing Mechanism
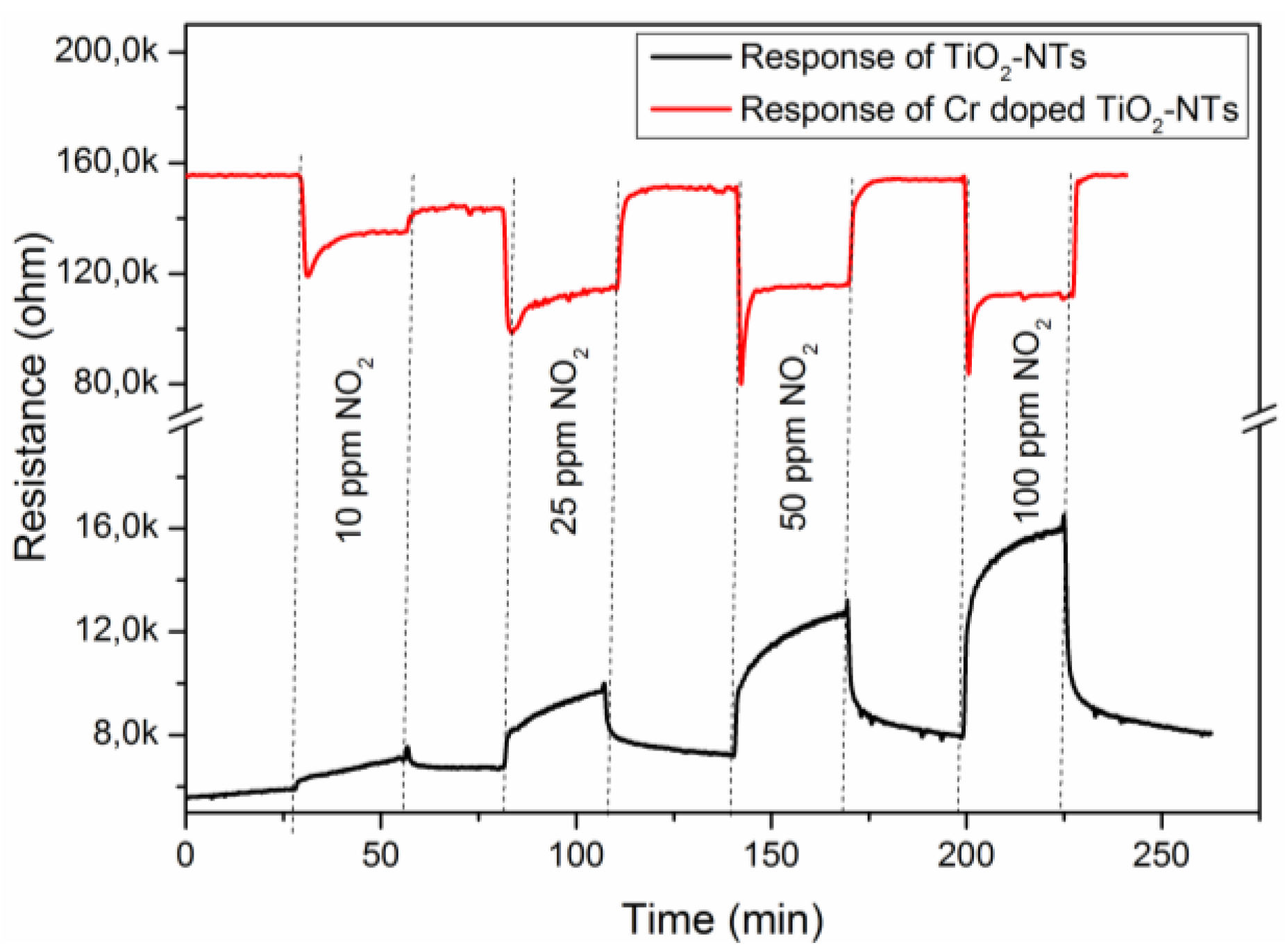
The sensor response curves obtained with a 12 µm thick TiO
2 NT layer in
Figure 12 exhibit that the undoped TiO
2 NT yields n-type sensor response, while the Cr doped TiO
2 NT behaves as p-type semi-conductor under NO
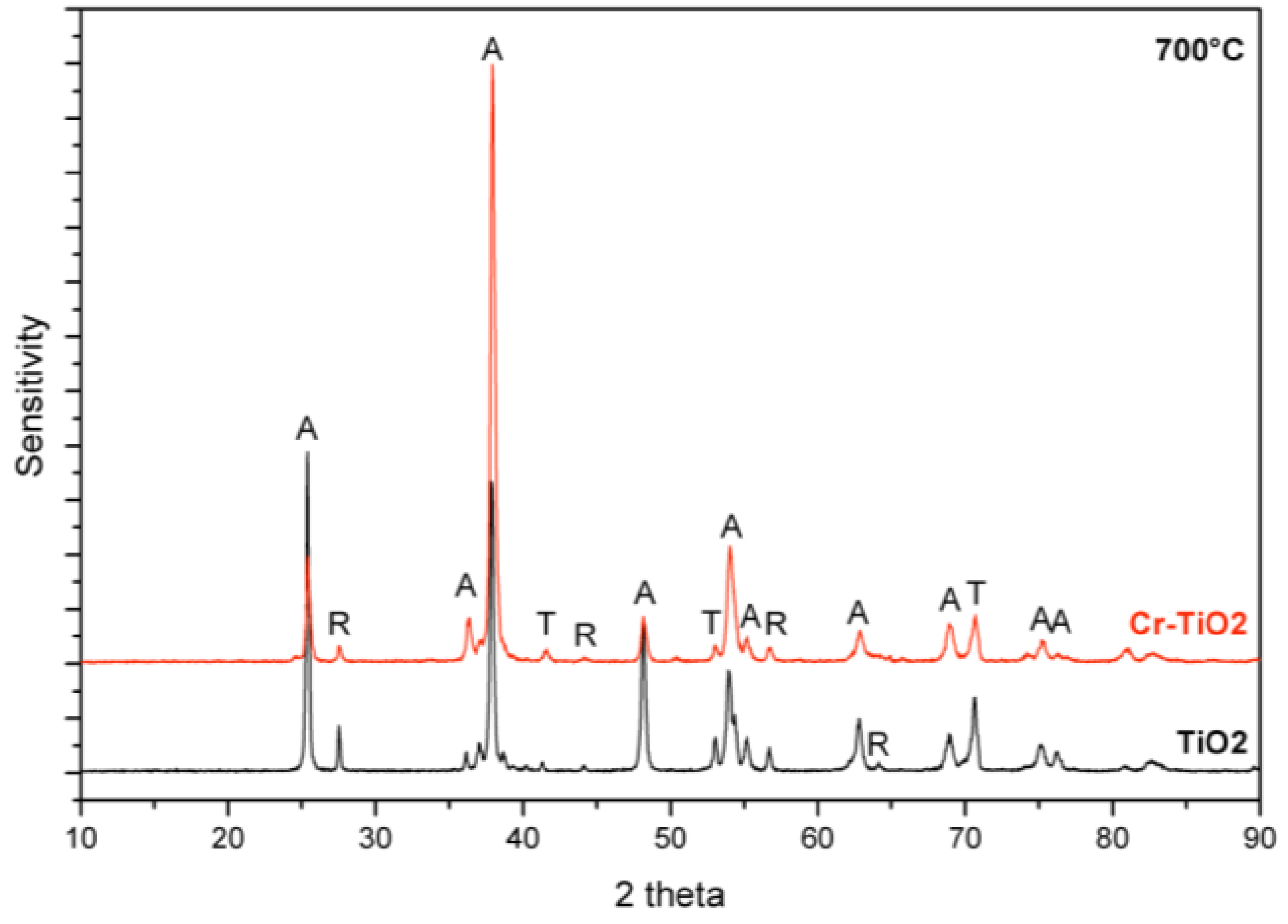
2-exposure. Considering the XRD investigation, this alteration in the sensor response type (
i.e., in the semiconductor type) can be firstly attributed to the presence of rutile-phase. However, according to Savage
et al., p-type semiconductor behavior can only be observed by the presence of almost pure rutile phase. After their observations, n-type semiconductor behavior is likely to be observed with a sensor layer (e.g., anatase and rutile phase mixture), which contains less than 75% rutile, being exposed to an oxidizing gas [
16]. Moreover, our TEM investigations showed that Cr doped TiO
2 NTs which were annealed at 700 °C contain solely anatase phase. Thus, the improved sensor response of the Cr doped TiO
2 NTs with p-type behavior can be attributed to the Cr
3+ doping which probably induces oxygen vacancies and thus facilitates the transport of charge carriers.
Figure 12.
Responses of the undoped and Cr doped TiO2–NTs sensors having a thickness of 12 µm and annealed at 700 °C towards various NO2 concentrations in argon carrier gas at 500 °C.
Figure 12.
Responses of the undoped and Cr doped TiO2–NTs sensors having a thickness of 12 µm and annealed at 700 °C towards various NO2 concentrations in argon carrier gas at 500 °C.
Relying on their close ionic radius (Ti4+ in hexa-coordination is 0.745 Å; while 0.755 Å for Cr3+), it can be postulated that the incorporation of trivalent Cr3+ into the TiO2-lattice will occur without any distortion of the lattice, simply via displacement of Ti4+-sites. Upon substitution of Ti4+ by the Cr3+; the original oxygen atom of one Ti–O bond becomes electronegative resulting in a shift from its original site. Cr-substitution in Ti-sites and Oo denotes an oxygen atom at its normal lattice site, resulting in oxygen vacancies, VO. As a result of Cr doping, oxygen vacancies with a positive charge will be formed.
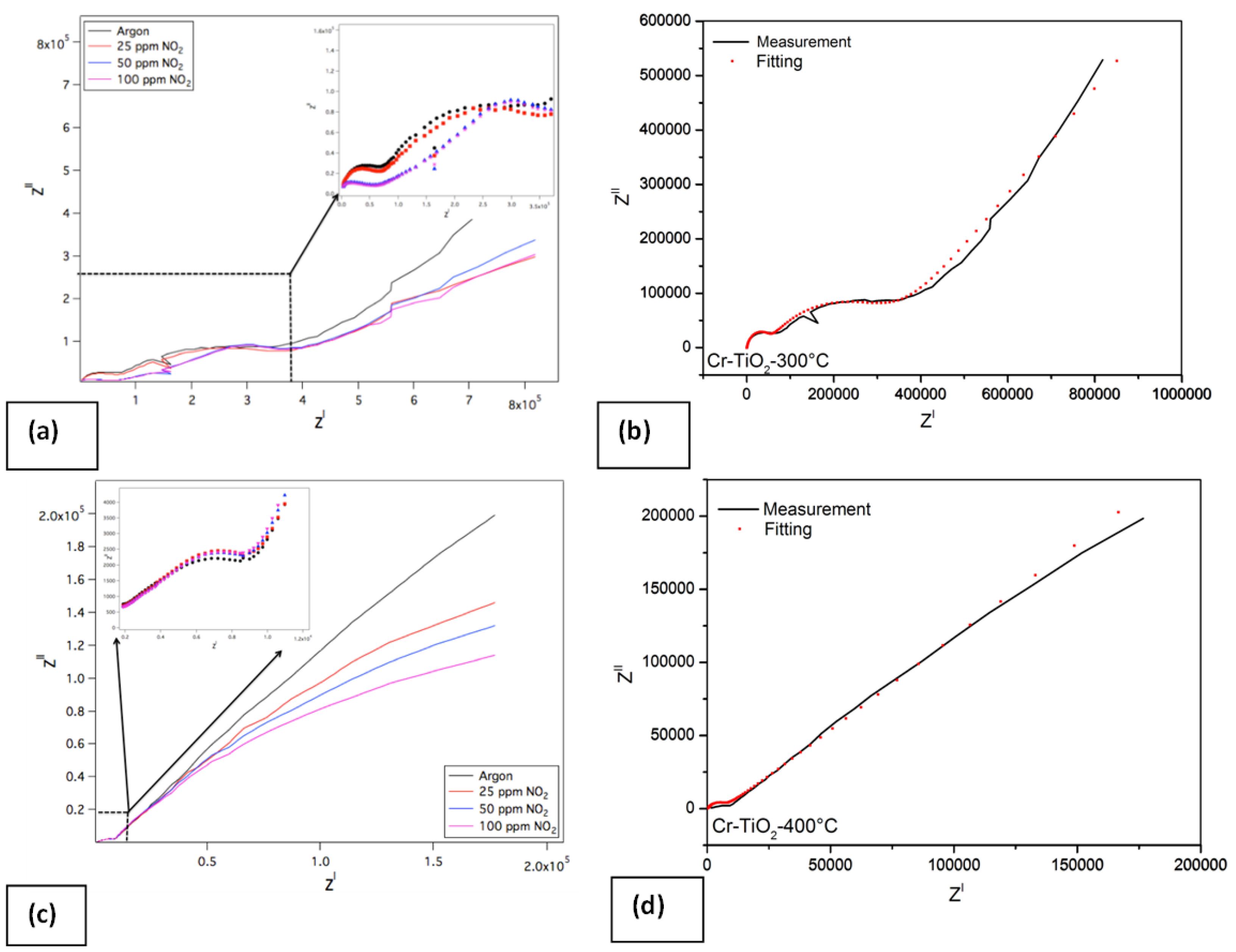
The Nyquist plot of the Cr doped TiO
2 NTs for different concentrations of NO
2 (25–50 and 100 ppm) at 300 °C and their fitting results are given in
Figure 13. The impedance spectrum of the Cr doped TiO
2 NTs differs from that of undoped TiO
2 NTs, displaying two semi-circle parts. The first semi-circle is related to the grain boundary conductivity and the second one is to the bulk conductivity. Furthermore, there is one linear part that is related with the response of electrode [
17]. The linear part of the impedance spectra shrinks with the NO
2-concentrations rising to 25, 50 and 100 ppm respectively in argon atmosphere (see
Figure 13a). The same equivalent circuit model which is given in
Figure 11 is used here too in order to obtain the fitting curve. The equivalent circuit model and measurements, which were carried out at 300 °C in argon, yield a perfect fit, as can be seen in
Figure 13b. The effect of temperature on sensing mechanism was also figured out and it was given in
Figure 13c,d. At higher temperatures, the impedance spectra display no longer two semi-circles; instead, contains one semi-circle which is related to the grain boundary conductivity at higher frequency and one linear part at low frequencies corresponding to the diffusion of sensing gas. Nevertheless, the impedance spectra of the Cr doped TiO
2 NTs yield the same behavior at 400 °C and at 300 °C. At higher concentrations of NO
2, the linear part of the spectra shrinks.
Figure 13.
Impedance spectra of Cr doped TiO2 NTs under different concentrations of NO2 at 300 °C (a) measurement results (b) fitting result combined with measurement and at 400 °C (c) measurements (d) fitting result combined with measurement.
Figure 13.
Impedance spectra of Cr doped TiO2 NTs under different concentrations of NO2 at 300 °C (a) measurement results (b) fitting result combined with measurement and at 400 °C (c) measurements (d) fitting result combined with measurement.
The numerical values of the equivalent circuit’s elements for Cr doped TiO
2 NTs are given in
Table 2. It is known that if CPE-P values of constant phase elements equals to approximately 1, then the CPE turns theoretically to a capacitor [
13]. As it can be seen from
Table 2, CPE1-P values are 1, which means CPE elements for Cr doped TiO
2 NTs layers behave like capacitor at all temperature ranges. The use of this information can make an important inference for the determination of which
R||CPE group represents which TiO
2 layer. Above, it is described that the same element group (
i.e., parallel connected
R||CPE) is used to define and differentiate between two different layers,
i.e., TiO
2 NTs layer and dense bulk TiO
2 layer lying between nanotubular layer and titanium.
Table 2.
Fitting parameters of the Cr doped TiO2 NTs.
Table 2.
Fitting parameters of the Cr doped TiO2 NTs.
| Temperature—Gas content | R1
[Ω] | CPE1 | R2
[Ω] | CPE2 | CPE3 |
|---|
T
[sP/Ω] | P | T
[sP/Ω] | P | T
[s] | P |
|---|
| 300 °C—pure Argon | 43,000 | 5.9E-9 | 1 | 285,000 | 9E-7 | 0,58 | 1.7E-5 | 0.5 |
| 300 °C—25 ppm NO2 in Argon | 40,000 | 1.6E-8 | 1 | 336,000 | 1.9E-6 | 0.56 | 2.5E-5 | 0.65 |
| 300 °C—50 ppm NO2 in Argon | 23,000 | 4E-9 | 1 | 600,000 | 2E-6 | 0.55 | 3.5E5 | 0.65 |
| 300 °C—100 ppm NO2 in Argon | 22,000 | 8E-9 | 1 | 620,000 | 2E-6 | 0.7 | 3.8E-5 | 0.85 |
| 400 °C—pure Argon | 5,000 | 3E-6 | 1 | 6,000 | 0.002 | 0.3 | 7.4E-5 | 0.58 |
| 400 °C—25 ppm NO2 in Argon | 4,100 | 3E-6 | 1 | 6,000 | 0.002 | 0.3 | 6E-5 | 0.59 |
| 400 °C—50 ppm NO2 in Argon | 3,300 | 3E-6 | 1 | 6,000 | 0.002 | 0.3 | 5.9E-5 | 0.69 |
| 400 °C—100 ppm NO2 in Argon | 2,900 | 3E-6 | 1 | 6,000 | 0.002 | 0.3 | 5.7E-5 | 0.71 |
| 500 °C—pure Argon | 800 | 5E-7 | 1 | 450 | 0.0002 | 0.5 | 1.9E-4 | 0.5 |
| 500 °C—25 ppm NO2 in Argon | 700 | 5E-7 | 1 | 450 | 0.0002 | 0.5 | 1.7E-4 | 0.51 |
| 500 °C—50 ppm NO2 in Argon | 450 | 5E-7 | 1 | 450 | 0.0002 | 0.5 | 1.6E-4 | 0.51 |
| 500 °C—100 ppm NO2 in Argon | 400 | 5E-7 | 1 | 450 | 0.0002 | 0.5 | 1.6E-4 | 0.54 |
The resistance values of the first compound
R1, which represents the TiO
2 NTs, decreases as the amount of the test gas NO
2 concentration increases. On the other hand, the resistance values of the second compound
R2, which represents bulk TiO
2 layer, increases with increasing NO
2 amount. As explained previously, a decrease in resistance towards oxidizing gases is a typical behavior for p-type semi-conductive metal-oxides; while an increase in the resistance for n- type semi-conductive metal-oxide is expected. TEM investigations and EDX-mapping results showed that Cr is situated only inside the TiO
2 NTs, while no presence of Cr was detected by TEM in the bulk TiO
2 layer which lies beneath the nanotubular TiO
2 (see
Figure 6). As a result, while Cr doped TiO
2 NTs exhibits p-type sensor behavior, the bulk TiO
2 layer contributes to the behavior of n-type semiconductor. After all, the different responses at
R1 and
R2 values towards NO
2 support this idea correlating the relation between the different circuit elements with the different morphological parts of TiO
2.
Impedance spectra show a reduction at the linear part of the graph (i.e., low frequency part) with the increase of test-gas concentrations. At equivalent circuit model, the linear part of the spectra is found to depend on the value of CPE3, which is responsible for the gas related interface. It is known that the CPE-T value varies mostly with the amount of the transported electrons at the system. Increasing the amount of NO2 in the system causes more reactions on the walls of the TiO2 NTs. This means that the more reactions occur on the wall surface, the more electrons are transported within the system. Thus, the observed increase in the value of CPE3-T is plausible and this causes a lowering at the linear part of the impedance spectra as the amount of the test gas increases.
Temperature dependent change in resistance for semiconductors is given with the Equation (1):
The applied sensor behaves as a chemo-resistive system therefore the Equation (2) which applies generally for resistors is used for TCR (Temperature Coefficient of Resistance) calculations [
18].
where R
0 is the resistance at the temperature T
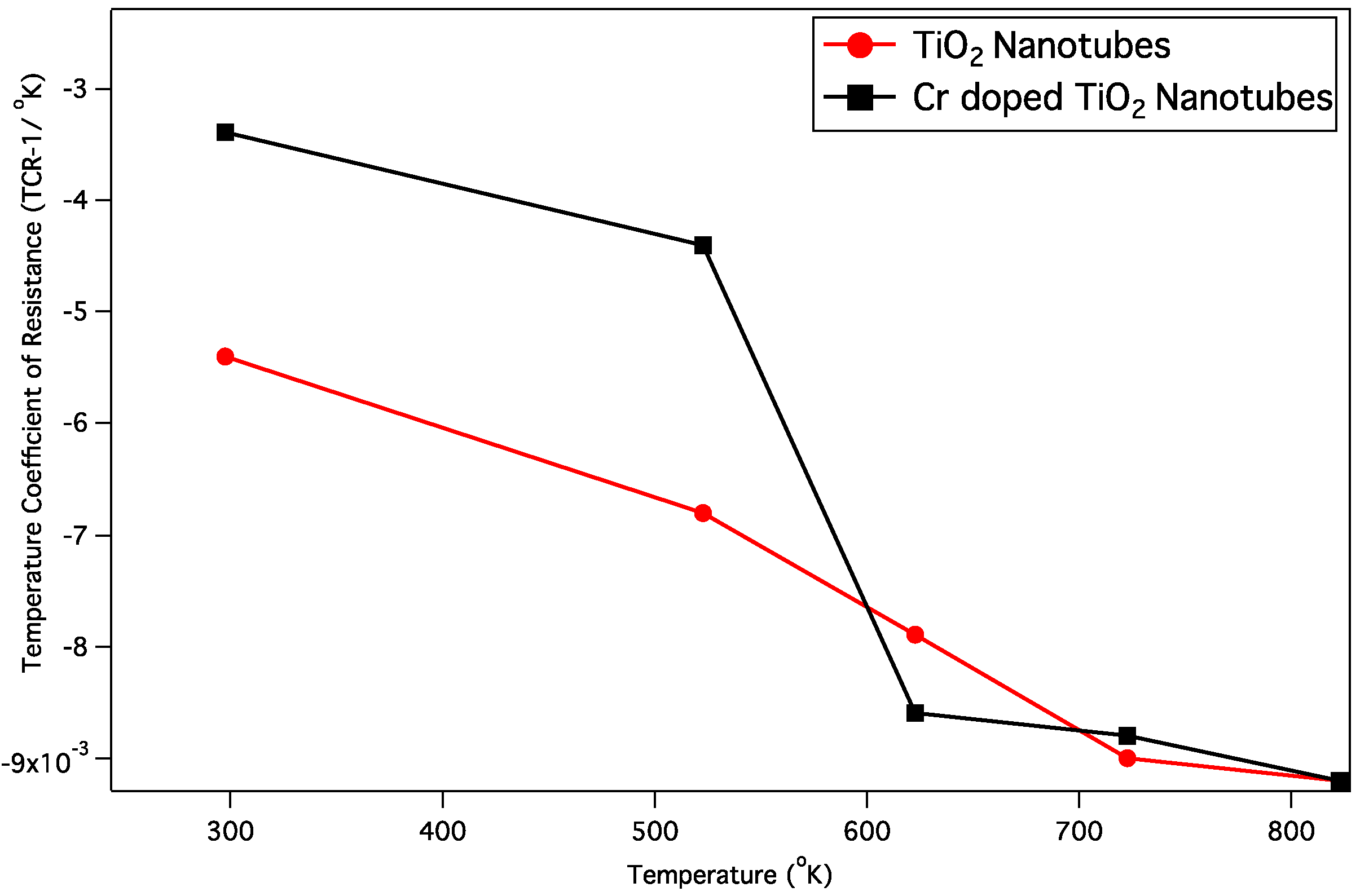
0 and α is the temperature coefficient of resistance (TCR) which is usually positive for metals at room temperature and negative for semiconductors and insulators. By applying this equation, easier experimental measurement is possible to demonstrate that the employed NTs exhibit negative TCR values. As
Figure 14 shows, both TiO
2 NTs samples (undoped and Cr doped) have negative TCR-values, which means that the temperature increase causes a decrease in the surface resistance of semi-conductive nanotubular layer.
Figure 14.
Temperature coefficient of resistance (TCR) values of the undoped and Cr doped TiO2 NTs depending on different temperatures.
Figure 14.
Temperature coefficient of resistance (TCR) values of the undoped and Cr doped TiO2 NTs depending on different temperatures.
The used equivalent circuit parameters showed that the increase of the temperature causes a decrease in both resistance values,
R1 and
R2, as expected. The resistance of the Cr doped TiO
2 NTs which is represented by
R1 decreases also depending on NO
2 concentration at higher concentrations. On the other hand, the resistance of the bulk TiO
2 layer towards test gas, NO
2 changes only at lower temperature (below 400 °C). Above 400 °C, the bulk TiO
2 layer shows no response to the NO
2 concentration changes. At the temperatures above 400 °C, the bulk conductivity of bulk TiO
2 becomes less effective as the grain boundary of the nano-tubes becomes more dominant. In addition, CPE
2-T value of the bulk TiO
2 layer remains stable even at higher NO
2 concentrations. The constant CPE
2-T rate is a sign of constant electron transfer in the bulk TiO
2 layer. It is already reported in the literature that the grain boundary conductivity increases with the increase of temperature [
19,
20,
21]. As indicated in the literature as well as the obtained impedance results show, the grain boundary conductivity of the Cr doped TiO
2 NTs became more dominant than the conductivity of thin bulk TiO
2 layer as temperature increases from 300 °C to 400 °C. In other words, increased grain boundary conductivity achieved by Cr doping provides an easy pathway for electron transfer eliminating the role of denser thin bulk TiO
2-layer beneath the TiO
2 NTs. Thus, it can be attributed that the doped transient oxides (e.g., TiO
2-NTs) may lead to higher sensitivity in gas sensors. However, no further influence of grain boundary or bulk conductivity can be anticipated for the sensors selectivity [
22]. It is more likely that selectivity of a sensor layer will relate to the band gap structure of the sensing material.