Sex Steroid-Mediated Control of Oviductal Function in Cattle
Abstract
:1. Introduction
2. Ovarian Sex Steroids and Their Receptors
2.1. Progesterone Receptors and Mechanism of Action
2.2. Estradiol Receptors and Mechanism of Action
3. Sex Steroids Dictate Oviductal Function
3.1. Sex Steroids Control Oviductal Gene Expression
3.2. Sex Steroids Control Oviductal Protein Expression
3.3. Sex Steroids Modulate Morphological Characteristics and Activity of the Oviduct
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Brower, L.; Anderson, E. Cytological Events Associated with the Secretory Process in the Rabbit Oviduct. Biol. Reprod. 1969, 1, 130–148. [Google Scholar] [CrossRef] [PubMed]
- Boilard, M.; Reyes-Moreno, C.; Lachance, C.; Massicotte, L.; Bailey, J.L.; Sirard, M.A.; Leclerc, P. Localization of the chaperone proteins GRP78 and HSP60 on the luminal surface of bovine oviduct epithelial cells and their association with spermatozoa. Biol. Reprod. 2004, 71, 1879–1889. [Google Scholar] [CrossRef] [PubMed]
- Lachance, C.; Bailey, J.L.; Leclerc, P. Expression of Hsp60 and Grp78 in the human endometrium and oviduct, and their effect on sperm functions. Hum. Reprod. 2007, 22, 2606–2614. [Google Scholar] [CrossRef] [PubMed]
- Pollard, J.W.; Plante, C.; King, W.A.; Hansen, P.J.; Betteridge, K.J.; Suarez, S.S. Fertilizing capacity of bovine sperm may be maintained by binding of oviductal epithelial cells. Biol. Reprod. 1991, 44, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Caballero, J.N.; Gervasi, M.G.; Veiga, M.F.; Dalvit, G.C.; Perez-Martínez, S.; Cetica, P.D.; Vazquez-Levin, M.H. Epithelial cadherin is present in bovine oviduct epithelial cells and gametes, and is involved in fertilization-related events. Theriogenology 2014, 81, 1189–1206. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Winuthayanon, W. Oviduct: Roles in fertilization and early embryo development. J. Endocrinol. 2017, 232, R1–R26. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.M.; Wang, H.S.; Lee, C.L.; Lee, J.D.; Huang, H.Y.; Chang, F.H.; Lee, J.F.; Soong, Y.K. Insulin-like growth factor-binding proteins produced by Vero cells, human oviductal cells and human endometrial cells, and the role of insulin-like growth factor-binding protein-3 in mouse embryo co-culture systems. Hum. Reprod. 1996, 11, 1281–1286. [Google Scholar] [CrossRef] [PubMed]
- Saint-Dizier, M.; Sandra, O.; Ployart, S.; Chebrout, M.; Constant, F. Expression of nuclear progesterone receptor and progesterone receptor membrane components 1 and 2 in the oviduct of cyclic and pregnant cows during the post-ovulation period. Reprod. Biol. Endocrinol. 2012, 10, 76. [Google Scholar] [CrossRef] [PubMed]
- Menezo, Y.; Guerin, P.; Elder, K. The oviduct: A neglected organ due for re-assessment in IVF. Reprod. Biomed. Online 2015, 30, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Betteridge, K.J.; Eaglesome, M.D.; Flood, P.F. Embryo transport through the mare’s oviduct depends upon cleavage and is independent of the ipsilateral corpus luteum. J. Reprod. Fertil. Suppl. 1979, 27, 387–394. [Google Scholar]
- Wetscher, F.; Havlicek, V.; Huber, T.; Muller, M.; Brem, G.; Besenfelder, U. Effect of morphological properties of transferred embryonic stages on tubal migration Implications for in vivo culture in the bovine oviduct. Theriogenology 2005, 64, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Almiñana, C.; Caballero, I.; Heath, P.R.; Maleki-Dizaji, S.; Parrilla, I.; Cuello, C.; Gil, M.A.; Vazquez, J.L.; Vazquez, J.M.; Roca, J.; et al. The battle of the sexes starts in the oviduct: Modulation of oviductal transcriptome by X and Y-bearing spermatozoa. BMC Genom. 2014, 15, 293. [Google Scholar] [CrossRef] [PubMed]
- Sponchiado, M.; Gomes, N.S.; Fontes, P.K.; Martins, T.; Del Collado, M.; Pastore, A.A.; Pugliesi, G.; Nogueira, M.F.G.; Binelli, M. Pre-hatching embryo-dependent and -independent programming of endometrial function in cattle. PLoS ONE 2017, 12, e0175954. [Google Scholar] [CrossRef] [PubMed]
- Gandolfi, F.; Moor, R.M. Stimulation of early embryonic development in the sheep by co-culture with oviduct epithelial cells. J. Reprod. Fertil. 1987, 81, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Sakkas, D.; Trounson, A.O. Co-culture of mouse embryos with oviduct and uterine cells prepared from mice at different days of pseudopregnancy. J. Reprod. Fertil. 1990, 90, 109–118. [Google Scholar] [CrossRef] [PubMed]
- White, K.L.; Hehnke, K.; Rickords, L.F.; Southern, L.L.; Thompson, D.L., Jr.; Wood, T.C. Early embryonic development in vitro by coculture with oviductal epithelial cells in pigs. Biol. Reprod. 1989, 41, 425–430. [Google Scholar] [CrossRef] [PubMed]
- Eyestone, W.H.; First, N.L. Co-culture of early cattle embryos to the blastocyst stage with oviducal tissue or in conditioned medium. J. Reprod. Fertil. 1989, 85, 715–720. [Google Scholar] [CrossRef] [PubMed]
- Yeung, W.S.; Ho, P.C.; Lau, E.Y.; Chan, S.T. Improved development of human embryos in vitro by a human oviductal cell co-culture system. Hum. Reprod. 1992, 7, 1144–1149. [Google Scholar] [CrossRef] [PubMed]
- Rizos, D.; Lonergan, P.; Boland, M.P.; Arroyo-Garcia, R.; Pintado, B.; de la Fuente, J.; Gutiérrez-Adán, A. Analysis of differential messenger RNA expression between bovine blastocysts produced in different culture systems: Implications for blastocyst quality. Biol. Reprod. 2002, 66, 589–595. [Google Scholar] [CrossRef] [PubMed]
- Rizos, D.; Ward, F.; Duffy, P.; Boland, M.P.; Lonergan, P. Consequences of bovine oocyte maturation, fertilization or early embryo development in vitro versus in vivo: Implications for blastocyst yield and blastocyst quality. Mol. Reprod. Dev. 2002, 61, 234–248. [Google Scholar] [CrossRef] [PubMed]
- Rizos, D.; Fair, T.; Papadopoulos, S.; Boland, M.P.; Lonergan, P. Developmental, qualitative, and ultrastructural differences between ovine and bovine embryos produced in vivo or in vitro. Mol. Reprod. Dev. 2002, 62, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Lonergan, P.; Rizos, D.; Kanka, J.; Nemcova, L.; Mbaye, A.M.; Kingston, M.; Wade, M.; Duffy, P.; Boland, M.P. Temporal sensitivity of bovine embryos to culture environment after fertilization and the implications for blastocyst quality. Reproduction 2003, 126, 337–346. [Google Scholar] [CrossRef] [PubMed]
- Nagatomo, H.; Akizawa, H.; Sada, A.; Kishi, Y.; Yamanaka, K.; Takuma, T.; Sasaki, K.; Yamauchi, N.; Yanagawa, Y.; Nagano, M.; et al. Comparing spatial expression dynamics of bovine blastocyst under three different procedures: In-vivo, in-vitro derived, and somatic cell nuclear transfer embryos. Jpn. J. Vet. Res. 2015, 63, 159–171. [Google Scholar] [PubMed]
- Ponsuksili, S.; Tesfaye, D.; Schellander, K.; Hoelker, M.; Hadlich, F.; Schwerin, M.; Wimmers, K. Differential expression of miRNAs and their target mRNAs in endometria prior to maternal recognition of pregnancy associates with endometrial receptivity for in vivo- and in vitro-produced bovine embryos. Biol. Reprod. 2014, 91, 135. [Google Scholar] [CrossRef] [PubMed]
- Salilew-Wondim, D.; Fournier, E.; Hoelker, M.; Saeed-Zidane, M.; Tholen, E.; Looft, C.; Neuhoff, C.; Besenfelder, U.; Havlicek, V.; Rings, F.; et al. Genome-Wide DNA Methylation Patterns of Bovine Blastocysts Developed In Vivo from Embryos Completed Different Stages of Development In Vitro. PLoS ONE 2015, 10, e0140467. [Google Scholar] [CrossRef] [PubMed]
- Hunter, R.H. Components of oviduct physiology in eutherian mammals. Biol. Rev. Camb. Philos. Soc. 2012, 87, 244–255. [Google Scholar] [CrossRef] [PubMed]
- Crisman, R.O.; McDonald, L.E.; Thompson, F.N. Effects of progesterone or estradiol on uterine tubal transport of ova in the cow. Theriogenology 1980, 13, 141–154. [Google Scholar] [CrossRef]
- Besenfelder, U.; Havlicek, V.; Brem, G. Role of the oviduct in early embryo development. Reprod. Domest. Anim. 2012, 47 (Suppl. 4), 156–163. [Google Scholar] [CrossRef] [PubMed]
- Avilés, M.; Coy, P.; Rizos, D. The oviduct: A key organ for the success of early reproductive events. Anim. Front. 2015, 5, 25–31. [Google Scholar] [CrossRef]
- Coy, P.; Garcia-Vázquez, F.A.; Visconti, P.E.; Avilés, M. Roles of the oviduct in mammalian fertilization. Reproduction 2012, 144, 649–660. [Google Scholar] [CrossRef] [PubMed]
- Ellington, J. The bovine oviduct and its role in reproduction: A review of the literature. Cornell Vet. 1991, 81, 313–328. [Google Scholar] [PubMed]
- Ikawa, M.; Inoue, N.; Benham, A.M.; Okabe, M. Fertilization: A sperm’s journey to and interaction with the oocyte. J. Clin. Investig. 2010, 120, 984. [Google Scholar] [CrossRef] [PubMed]
- Kolle, S.; Reese, S.; Kummer, W. New aspects of gamete transport, fertilization, and embryonic development in the oviduct gained by means of live cell imaging. Theriogenology 2010, 73, 786–795. [Google Scholar] [CrossRef] [PubMed]
- Talevi, R.; Gualtieri, R. Molecules involved in sperm-oviduct adhesion and release. Theriogenology 2010, 73, 796–801. [Google Scholar] [CrossRef] [PubMed]
- Aviles, M.; Gutierrez-Adan, A.; Coy, P. Oviductal secretions: Will they be key factors for the future ARTs? Mol. Hum. Reprod. 2010, 16, 896–906. [Google Scholar] [CrossRef] [PubMed]
- Buhi, W.C. Characterization and biological roles of oviduct-specific, oestrogen-dependent glycoprotein. Reproduction 2002, 123, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Buhi, W.C.; Alvarez, I.M.; Kouba, A.J. Secreted proteins of the oviduct. Cells Tissues Organs 2000, 166, 165–179. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, J.; Reyley, M. The uterine tubal fluid: Secretion, composition and biological effects. Anim. Reprod. 2005, 2, 91–105. [Google Scholar]
- Mondejar, I.; Acuna, O.S.; Izquierdo-Rico, M.J.; Coy, P.; Aviles, M. The Oviduct: Functional Genomic and Proteomic Approach. Reprod. Domest. Anim. 2012, 47, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Leese, H.J. The formation and function of oviduct fluid. J. Reprod. Fertil. 1988, 82, 843–856. [Google Scholar] [CrossRef] [PubMed]
- Pugliesi, G.; Oliveria, M.L.; Scolari, S.C.; Lopes, E.; Pinaffi, F.V.; Miagawa, B.T.; Paiva, Y.N.; Maio, J.R.; Nogueira, G.P.; Binelli, M. Corpus Luteum Development and Function after Supplementation of Long-Acting Progesterone During the Early Luteal Phase in Beef Cattle. Reprod. Domest. Anim. 2014, 49, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Vasconcelos, J.L.M.; Sartori, R.; Oliveira, H.N.; Guenther, J.G.; Wiltbank, M.C. Reduction in size of the ovulatory follicle reduces subsequent luteal size and pregnancy rate. Theriogenology 2001, 56, 307–314. [Google Scholar] [CrossRef]
- Sa Filho, O.G.; Meneghetti, M.; Peres, R.F.G.; Lamb, G.C.; Vasconcelos, J.L.M. Fixed-time artificial insemination with estradiol and progesterone for Bos indicus cows II: Strategies and factors affecting fertility. Theriogenology 2009, 72, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Sa Filho, M.F.; Crespilho, A.M.; Santos, J.E.P.; Perry, G.A.; Baruselli, P.S. Ovarian follicle diameter at timed insemination and estrous response influence likelihood of ovulation and pregnancy after estrous synchronization with progesterone or progestin-based protocols in suckled Bos indicus cows. Anim. Reprod. Sci. 2010, 120, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Abe, H. The mammalian oviductal epithelium: Regional variations in cytological and functional aspects of the oviductal secretory cells. Histol. Histopathol. 1996, 11, 743–768. [Google Scholar] [PubMed]
- Hunter, R.H. Have the Fallopian tubes a vital role in promoting fertility? Acta Obstet. Gynecol. Scand. 1998, 77, 475–486. [Google Scholar] [CrossRef] [PubMed]
- Hunter, R.H.; Cook, B.; Poyser, N.L. Regulation of oviduct function in pigs by local transfer of ovarian steroids and prostaglandins: A mechanism to influence sperm transport. Eur. J. Obstet. Gynecol. Reprod. Biol. 1983, 14, 225–232. [Google Scholar] [CrossRef]
- Wijayagunawardane, M.P.B.; Miyamoto, A.; Cerbito, W.A.; Acosta, T.J.; Takagi, M.; Sato, K. Local distributions of oviductal estradiol, progesterone, prostaglandins, oxytocin and endothelin-1 in the cyclic cow. Theriogenology 1998, 49, 607–618. [Google Scholar] [CrossRef]
- Almiñana, C.; Corbin, E.; Tsikis, G.; Alcantara-Neto, A.S.; Labas, V.; Reynaud, K.; Galio, L.; Uzbekov, R.; Garanina, A.S.; Druart, X.; et al. Oviduct extracellular vesicles protein content and their role during oviduct-embryo cross-talk. Reproduction 2017, 154, 153–168. [Google Scholar] [CrossRef] [PubMed]
- Maillo, V.; Sanchez-Calabuig, M.J.; Lopera-Vasquez, R.; Hamdi, M.; Gutierrez-Adan, A.; Lonergan, P.; Rizos, D. Oviductal response to gametes and early embryos in mammals. Reproduction 2016, 152, R127–R141. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Einspanier, R.; Schoen, J. In Vitro Mimicking of Estrous Cycle Stages in Porcine Oviduct Epithelium Cells: Estradiol and Progesterone Regulate Differentiation, Gene Expression, and Cellular Function. Biol. Reprod. 2013, 89, 54. [Google Scholar] [CrossRef] [PubMed]
- Stephens, S.M.; Moley, K.H. Follicular origins of modern reproductive endocrinology. Am. J. Physiol. Endocrinol. Metab. 2009, 297, E1235–E1236. [Google Scholar] [CrossRef] [PubMed]
- Allen, E.; Doisy, E.A. An ovarian hormone: Preliminary report on its localization, extraction and partial purification, and action in test animals. JAMA 1923, 81, 819–821. [Google Scholar] [CrossRef]
- Simpson, E.; Santen, R.J. Celebrating 75 years of oestradiol. J. Mol. Endocrinol. 2015, 55, T1-20. [Google Scholar] [CrossRef] [PubMed]
- Genazzani, A.R.; Stomati, M.; Morittu, A.; Bernardi, F.; Monteleone, P.; Casarosa, E.; Gallo, R.; Salvestroni, C.; Luisi, M. Progesterone, progestagens and the central nervous system. Hum. Reprod. 2000, 15 (Suppl. 1), 14–27. [Google Scholar] [CrossRef] [PubMed]
- Grumbach, M.M.; Auchus, R.J. Estrogen: Consequences and implications of human mutations in synthesis and action. J. Clin. Endocrinol. Metab. 1999, 84, 4677–4694. [Google Scholar] [CrossRef] [PubMed]
- Grazzini, E.; Guillon, G.; Mouillac, B.; Zingg, H.H. Inhibition of oxytocin receptor function by direct binding of progesterone. Nature 1998, 392, 509–512. [Google Scholar] [CrossRef] [PubMed]
- Hirata, S.; Shoda, T.; Kato, J.; Hoshi, K. Novel isoforms of the mRNA for human female sex steroid hormone receptors. J. Steroid Biochem. Mol. Biol. 2002, 83, 25–30. [Google Scholar] [CrossRef]
- Wei, L.L.; Norris, B.M.; Baker, C.J. An N-terminally truncated third progesterone receptor protein, PR(C), forms heterodimers with PR(B) but interferes in PR(B)-DNA binding. J. Steroid Biochem. Mol. Biol. 1997, 62, 287–297. [Google Scholar] [CrossRef]
- Abdel-Hafiz, H.A.; Horwitz, K.B. Post-translational modifications of the progesterone receptors. J. Steroid Biochem. Mol. Biol. 2014, 140, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Hagan, C.R.; Lange, C.A. Molecular determinants of context-dependent progesterone receptor action in breast cancer. BMC Med. 2014, 12, 32. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Rice, C.D.; Pang, Y.; Pace, M.; Thomas, P. Cloning, expression, and characterization of a membrane progestin receptor and evidence it is an intermediary in meiotic maturation of fish oocytes. Proc. Natl. Acad. Sci. USA 2003, 100, 2231–2236. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.L.; Kupchak, B.R.; Garitaonandia, I.; Hoang, L.K.; Maina, A.S.; Regalla, L.M.; Lyons, T.J. Heterologous expression of human mPRalpha, mPRbeta and mPRgamma in yeast confirms their ability to function as membrane progesterone receptors. Steroids 2008, 73, 1160–1173. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.T.; Hu, T.; Arterburn, M.; Boyle, B.; Bright, J.M.; Emtage, P.C.; Funk, W.D. PAQR proteins: A novel membrane receptor family defined by an ancient 7-transmembrane pass motif. J. Mol. Evol. 2005, 61, 372–380. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Bond, J.; Thomas, P. Identification, classification, and partial characterization of genes in humans and other vertebrates homologous to a fish membrane progestin receptor. Proc. Natl. Acad. Sci. USA 2003, 100, 2237–2242. [Google Scholar] [CrossRef] [PubMed]
- Aparicio, I.M.; Garcia-Herreros, M.; O’Shea, L.C.; Hensey, C.; Lonergan, P.; Fair, T. Expression, regulation, and function of progesterone receptors in bovine cumulus oocyte complexes during in vitro maturation. Biol. Reprod. 2011, 84, 910–921. [Google Scholar] [CrossRef] [PubMed]
- Dressing, G.E.; Goldberg, J.E.; Charles, N.J.; Schwertfeger, K.L.; Lange, C.A. Membrane progesterone receptor expression in mammalian tissues: A review of regulation and physiological implications. Steroids 2011, 76, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Qiu, H.B.; Lu, S.S.; Ji, K.L.; Song, X.M.; Lu, Y.Q.; Zhang, M.; Lu, K.H. Membrane progestin receptor beta (mPR-beta): A protein related to cumulus expansion that is involved in in vitro maturation of pig cumulus-oocyte complexes. Steroids 2008, 73, 1416–1423. [Google Scholar] [CrossRef] [PubMed]
- Pang, Y.; Dong, J.; Thomas, P. Characterization, neurosteroid binding and brain distribution of human membrane progesterone receptors delta and (mPRdelta and mPR) and mPRdelta involvement in neurosteroid inhibition of apoptosis. Endocrinology 2013, 154, 283–295. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, M.S.; Pierron, V.; Michalovich, D.; Astle, S.; Thornton, S.; Peltoketo, H.; Lam, E.W.; Gellersen, B.; Huhtaniemi, I.; Allen, J.; et al. Regulated expression of putative membrane progestin receptor homologues in human endometrium and gestational tissues. J. Endocrinol. 2005, 187, 89–101. [Google Scholar] [CrossRef] [PubMed]
- Karteris, E.; Zervou, S.; Pang, Y.; Dong, J.; Hillhouse, E.W.; Randeva, H.S.; Thomas, P. Progesterone signaling in human myometrium through two novel membrane G protein-coupled receptors: Potential role in functional progesterone withdrawal at term. Mol. Endocrinol. 2006, 20, 1519–1534. [Google Scholar] [CrossRef] [PubMed]
- Falkenstein, E.; Meyer, C.; Eisen, C.; Scriba, P.C.; Wehling, M. Full-length cDNA sequence of a progesterone membrane-binding protein from porcine vascular smooth muscle cells. Biochem. Biophys. Res. Commun. 1996, 229, 86–89. [Google Scholar] [CrossRef] [PubMed]
- Filardo, E.J.; Thomas, P. Minireview: G protein-coupled estrogen receptor-1, GPER-1: Its mechanism of action and role in female reproductive cancer, renal and vascular physiology. Endocrinology 2012, 153, 2953–2962. [Google Scholar] [CrossRef] [PubMed]
- Barton, M.; Filardo, E.J.; Lolait, S.J.; Thomas, P.; Maggiolini, M.; Prossnitz, E.R. Twenty years of the G protein-coupled estrogen receptor GPER: Historical and personal perspectives. J. Steroid Biochem. Mol. Biol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Cheskis, B.J.; Greger, J.G.; Nagpal, S.; Freedman, L.P. Signaling by estrogens. J. Cell. Physiol. 2007, 213, 610–617. [Google Scholar] [CrossRef] [PubMed]
- Green, S.; Walter, P.; Kumar, V.; Krust, A.; Bornert, J.M.; Argos, P.; Chambon, P. Human oestrogen receptor cDNA: Sequence, expression and homology to v-erb-A. Nature 1986, 320, 134–139. [Google Scholar] [CrossRef] [PubMed]
- Greene, G.L.; Gilna, P.; Waterfield, M.; Baker, A.; Hort, Y.; Shine, J. Sequence and expression of human estrogen receptor complementary DNA. Science 1986, 231, 1150–1154. [Google Scholar] [CrossRef] [PubMed]
- Mosselman, S.; Polman, J.; Dijkema, R. ER beta: Identification and characterization of a novel human estrogen receptor. FEBS Lett. 1996, 392, 49–53. [Google Scholar] [CrossRef]
- Pelletier, G.; Labrie, C.; Labrie, F. Localization of oestrogen receptor alpha, oestrogen receptor beta and androgen receptors in the rat reproductive organs. J. Endocrinol. 2000, 165, 359–370. [Google Scholar] [CrossRef] [PubMed]
- Kuiper, G.G.; Carlsson, B.; Grandien, K.; Enmark, E.; Haggblad, J.; Nilsson, S.; Gustafsson, J.A. Comparison of the ligand binding specificity and transcript tissue distribution of estrogen receptors alpha and beta. Endocrinology 1997, 138, 863–870. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Eriksson, H.; Sahlin, L. Estrogen receptors alpha and beta in the female reproductive tract of the rat during the estrous cycle. Biol. Reprod. 2000, 63, 1331–1340. [Google Scholar] [CrossRef] [PubMed]
- Saji, S.; Jensen, E.V.; Nilsson, S.; Rylander, T.; Warner, M.; Gustafsson, J.A. Estrogen receptors alpha and beta in the rodent mammary gland. Proc. Natl. Acad. Sci. USA 2000, 97, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Kowalski, A.A.; Graddy, L.G.; Vale-Cruz, D.S.; Choi, I.; Katzenellenbogen, B.S.; Simmen, F.A.; Simmen, R.C. Molecular cloning of porcine estrogen receptor-beta complementary DNAs and developmental expression in periimplantation embryos. Biol. Reprod. 2002, 66, 760–769. [Google Scholar] [CrossRef] [PubMed]
- Szego, C.M.; Davis, J.S. Adenosine 3′,5′-monophosphate in rat uterus: Acute elevation by estrogen. Proc. Natl. Acad. Sci. USA 1967, 58, 1711–1718. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Yuhanna, I.S.; Galcheva-Gargova, Z.; Karas, R.H.; Mendelsohn, M.E.; Shaul, P.W. Estrogen receptor alpha mediates the nongenomic activation of endothelial nitric oxide synthase by estrogen. J. Clin. Investig. 1999, 103, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Dewing, P.; Boulware, M.I.; Sinchak, K.; Christensen, A.; Mermelstein, P.G.; Micevych, P. Membrane estrogen receptor-alpha interactions with metabotropic glutamate receptor 1a modulate female sexual receptivity in rats. J. Neurosci. 2007, 27, 9294–9300. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Haynes, M.P.; Bender, J.R. Plasma membrane localization and function of the estrogen receptor alpha variant (ER46) in human endothelial cells. Proc. Natl. Acad. Sci. USA 2003, 100, 4807–4812. [Google Scholar] [CrossRef] [PubMed]
- Arnal, J.F.; Lenfant, F.; Metivier, R.; Flouriot, G.; Henrion, D.; Adlanmerini, M.; Fontaine, C.; Gourdy, P.; Chambon, P.; Katzenellenbogen, B.; et al. Membrane and Nuclear Estrogen Receptor Alpha Actions: From Tissue Specificity to Medical Implications. Physiol. Rev. 2017, 97, 1045–1087. [Google Scholar] [CrossRef] [PubMed]
- Pietras, R.J.; Szego, C.M. Specific binding sites for oestrogen at the outer surfaces of isolated endometrial cells. Nature 1977, 265, 69–72. [Google Scholar] [CrossRef] [PubMed]
- Owman, C.; Blay, P.; Nilsson, C.; Lolait, S.J. Cloning of human cDNA encoding a novel heptahelix receptor expressed in Burkitt’s lymphoma and widely distributed in brain and peripheral tissues. Biochem. Biophys. Res. Commun. 1996, 228, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Carmeci, C.; Thompson, D.A.; Ring, H.Z.; Francke, U.; Weigel, R.J. Identification of a gene (GPR30) with homology to the G-protein-coupled receptor superfamily associated with estrogen receptor expression in breast cancer. Genomics 1997, 45, 607–617. [Google Scholar] [CrossRef] [PubMed]
- Filardo, E.J.; Quinn, J.A.; Bland, K.I.; Frackelton, A.R., Jr. Estrogen-induced activation of Erk-1 and Erk-2 requires the G protein-coupled receptor homolog, GPR30, and occurs via trans-activation of the epidermal growth factor receptor through release of HB-EGF. Mol. Endocrinol. 2000, 14, 1649–1660. [Google Scholar] [CrossRef] [PubMed]
- Revankar, C.M.; Cimino, D.F.; Sklar, L.A.; Arterburn, J.B.; Prossnitz, E.R. A transmembrane intracellular estrogen receptor mediates rapid cell signaling. Science 2005, 307, 1625–1630. [Google Scholar] [CrossRef] [PubMed]
- Morita, M.; Miyamoto, H.; Sugimoto, M.; Sugimoto, N.; Manabe, N. Alterations in cell proliferation and morphology of ampullar epithelium of the mouse oviduct during the estrous cycle. J. Reprod. Dev. 1997, 43, 235–241. [Google Scholar] [CrossRef]
- Eriksen, T.; Terkelsen, O.; Hyttel, P.; Greve, T. Ultrastructural features of secretory-cells in the bovine oviduct epithelium. Anat. Embryol. 1994, 190, 583–590. [Google Scholar] [CrossRef] [PubMed]
- Ayen, E.; Shahrooz, R.; Kazemie, S. Histological and histomorphometrical changes of different regions of oviduct during follicular and lutealphases of estrus cycle in adult Azarbaijan buffalo. IJVR 2012, 13, 42–48. [Google Scholar] [CrossRef]
- Bishop, D.W. Active secretion in the rabbit oviduct. Am. J. Physiol. 1956, 187, 347–352. [Google Scholar] [CrossRef] [PubMed]
- Wijayagunawardane, M.P.B.; Kodithuwakku, S.P.; De Silva, N.T.; Miyamoto, A. Angiotensin II Secretion by the Bovine Oviduct is Stimulated by Luteinizing Hormone and Ovarian Steroids. J. Reprod. Dev. 2009, 55, 570–575. [Google Scholar] [CrossRef] [PubMed]
- Wijayagunawardane, M.P.B.; Kodithuwakku, S.P.; Yamamoto, D.; Miyamoto, A. Vascular endothelial growth factor system in the cow oviduct: A possible involvement in the regulation of oviductal motility and embryo transport. Mol. Reprod. Dev. 2005, 72, 511–520. [Google Scholar] [CrossRef] [PubMed]
- Sostaric, E.; Dieleman, S.J.; Van De Lest, C.H.A.; Colenbrander, B.; Vos, P.; Garcia-Gil, N.; Gadella, B.M. Sperm binding properties and secretory activity of the bovine oviduct immediately before and after ovulation. Mol. Reprod. Dev. 2008, 75, 60–74. [Google Scholar] [CrossRef] [PubMed]
- Hamner, C.E.; Fox, S.B. Effect of oestrogen and progesterone on physical properties of rabbit oviduct fluid. J. Reprod. Fertil. 1968, 16, 121–122. [Google Scholar] [CrossRef] [PubMed]
- McDonald, M.F.; Bellve, A.R. Influence of oestrogen and progesterone on flow of fluid from the Fallopian tube in the ovariectomized ewe. J. Reprod. Fertil. 1969, 20, 51–61. [Google Scholar] [CrossRef] [PubMed]
- McDaniel, J.W.; Scalzi, H.; Black, D. Influence of ovarian hormones on histology and histochemistry of the bovine oviduct. J. Dairy Sci. 1968, 51, 754–761. [Google Scholar] [CrossRef]
- Ruckebusch, Y.; Bayard, F. Motility of the oviduct and uterus of the cow during the oestrous cycle. J. Reprod. Fertil. 1975, 43, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Verhage, H.G.; Abel, J.H.; Tietz, W.J.; Barrau, M.D. Development and maintenance of oviductal epithelium during estrous-cycle in bitch. Biol. Reprod. 1973, 9, 460–474. [Google Scholar] [CrossRef] [PubMed]
- Hunter, R.; Wilmut, I. The rate of functional sperm transport into the oviducts of mated cows. Anim. Reprod. Sci. 1983, 5, 167–173. [Google Scholar] [CrossRef]
- Ulbrich, S.E.; Kettler, A.; Einspanier, R. Expression and localization of estrogen receptor alpha, estrogen receptor beta and progesterone receptor in the bovine oviduct in vivo and in vitro. J. Steroid Biochem. Mol. Biol. 2003, 84, 279–289. [Google Scholar] [CrossRef]
- Valle, G.R.; Cassali, G.D.; Nogueira, J.C.; Castro, A.C.; Reis, A.M.; Cardoso, F.M.; Figueiredo, C.B.; Nascimento, E.F. Nuclear estrogen and progesterone receptors in the oviduct of heifers under natural and superovulated estrous cycles. Anim. Reprod. Sci. 2007, 101, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Bauersachs, S.; Blum, H.; Mallok, S.; Wenigerkind, H.; Rief, S.; Prelle, K.; Wolf, E. Regulation of ipsilateral and contralateral bovine oviduct epithelial cell function in the postovulation period: A transcriptomics approach. Biol. Reprod. 2003, 68, 1170–1177. [Google Scholar] [CrossRef] [PubMed]
- Bauersachs, S.; Rehfeld, S.; Ulbrich, S.E.; Mallok, S.; Prelle, K.; Wenigerkind, H.; Einspanier, R.; Blum, H.; Wolf, E. Monitoring gene expression changes in bovine oviduct epithelial cells during the oestrous cycle. J. Mol. Endocrinol. 2004, 32, 449–466. [Google Scholar] [CrossRef] [PubMed]
- Cerny, K.L.; Garrett, E.; Walton, A.J.; Anderson, L.H.; Bridges, P.J. A transcriptomal analysis of bovine oviductal epithelial cells collected during the follicular phase versus the luteal phase of the estrous cycle. Reprod. Biol. Endocrinol. 2015, 13, 84. [Google Scholar] [CrossRef] [PubMed]
- Gonella-Diaza, A.M.; Andrade, S.C.; Sponchiado, M.; Pugliesi, G.; Mesquita, F.S.; Van Hoeck, V.; de Francisco Strefezzi, R.; Gasparin, G.R.; Coutinho, L.L.; Binelli, M. Size of the Ovulatory Follicle Dictates Spatial Differences in the Oviductal Transcriptome in Cattle. PLoS ONE 2015, 10, e0145321. [Google Scholar] [CrossRef] [PubMed]
- Pugliesi, G.; Santos, F.B.; Lopes, E.; Nogueira, É.; Maio, J.R.; Binelli, M. Improved fertility in suckled beef cows ovulating large follicles or supplemented with long-acting progesterone after timed-AI. Theriogenology 2016, 85, 1239–1248. [Google Scholar] [CrossRef] [PubMed]
- Boice, M.L.; Geisert, R.D.; Blair, R.M.; Verhage, H.G. Identification and characterization of bovine oviductal glycoproteins synthesized at estrus. Biol. Reprod. 1990, 43, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Binelli, M.; Hampton, J.; Buhi, W.C.; Thatcher, W.W. Persistent dominant follicle alters pattern of oviductal secretory proteins from cows at estrus. Biol. Reprod. 1999, 61, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Einspanier, R.; Gabler, C.; Kettler, A.; Kloas, W. Characterization and localization of beta(2)-Adrenergic receptors in the bovine oviduct: Indication for progesterone-mediated expression. Endocrinology 1999, 140, 2679–2684. [Google Scholar] [CrossRef] [PubMed]
- Wijayagunawardane, M.P.B.; Gabler, C.; Killian, G.; Miyamoto, A. Tumor necrosis factor alpha in the bovine oviduct during the estrous cycle: Messenger RNA expression and effect on secretion of prostaglandins, endothelin-1, and angiotensin II. Biol. Reprod. 2003, 69, 1341–1346. [Google Scholar] [CrossRef] [PubMed]
- Lapointe, J.R.; Kimmins, S.; MacLaren, L.A.; Bilodeau, J.F. Estrogen selectively up-regulates the phospholipid hydroperoxide glutathione peroxidase in the oviducts. Endocrinology 2005, 146, 2583–2592. [Google Scholar] [CrossRef] [PubMed]
- Soleilhavoup, C.; Riou, C.; Tsikis, G.; Labas, V.; Harichaux, G.; Kohnke, P.; Reynaud, K.; de Graaf, S.P.; Gerard, N.; Druart, X. Proteomes of the Female Genital Tract During the Oestrous Cycle. Mol. Cell. Proteom. 2016, 15, 93–108. [Google Scholar] [CrossRef] [PubMed]
- Lauschova, I. Secretory cells and morphological manifestation of secretion in the mouse oviduct. Scripta Medica (BRNO) 2003, 76, 203–214. [Google Scholar]
- Buhi, W.C.; Ashworth, C.J.; Bazer, F.W.; Alvarez, I.M. In vitro synthesis of oviductal secretory proteins by estrogen-treated ovariectomized gilts. J. Exp. Zool. 1992, 262, 426–435. [Google Scholar] [CrossRef] [PubMed]
- Buhi, W.C.; Bazer, F.W.; Alvarez, I.M.; Mirando, M.A. In vitro synthesis of oviductal proteins associated with estrus and 17 beta-estradiol-treated ovariectomized ewes. Endocrinology 1991, 128, 3086–3095. [Google Scholar] [CrossRef] [PubMed]
- Lippes, J.; Krasner, J.; Alfonso, L.A.; Dacalos, E.D.; Lucero, R. Human oviductal fluid proteins. Fertil. Steril. 1981, 36, 623–629. [Google Scholar] [CrossRef]
- Wegner, C.C.; Killian, G.J. In vitro and in vivo association of an oviduct estrus-associated protein with bovine zona pellucida. Mol. Reprod. Dev. 1991, 29, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Araki, Y.; Nohara, M.; Yoshida-Komiya, H.; Kuramochi, T.; Ito, M.; Hoshi, H.; Shinkai, Y.; Sendai, Y. Effect of a null mutation of the oviduct-specific glycoprotein gene on mouse fertilization. Biochem. J. 2003, 374, 551–557. [Google Scholar] [CrossRef] [PubMed]
- Winuthayanon, W.; Bernhardt, M.L.; Padilla-Banks, E.; Myers, P.H.; Edin, M.L.; Lih, F.B.; Hewitt, S.C.; Korach, K.S.; Williams, C.J. Oviductal estrogen receptor alpha signaling prevents protease-mediated embryo death. Elife 2015, 4, e10453. [Google Scholar] [CrossRef] [PubMed]
- Kenngott, R.A.; Sinowatz, F. Prenatal development of the bovine oviduct. Anat Histol. Embryol. 2007, 36, 272–283. [Google Scholar] [CrossRef] [PubMed]
- Konishi, I.; Fujii, S.; Parmley, T.H.; Mori, T. Development of ciliated cells in the human fetal oviduct: An ultrastructural study. Anat. Rec. 1987, 219, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Agduhr, E. Studies on the structure and development of the bursa ovarica and the tuba uterina in the mouse. Acta Zool. 1927, 8, 1–133. [Google Scholar] [CrossRef]
- Bacha, W.J.; Bacha, L.M. Color Atlas of Veterinary Histology, 3rd ed.; Wiley-blackwell: Hoboken, NJ, USA, 2012; 368p, ISBN 10 0470958510. [Google Scholar]
- Mokhtar, D.M. Microscopic and histochemical characterization of the bovine uterine tube during the follicular and luteal phases of estrous cycle. JAMU 2015, 3, 44–52. [Google Scholar] [CrossRef]
- Restall, B.J. Histological observations on the reproductive tract of the ewe. Aust. J. Biol. Sci. 1966, 19, 673–686. [Google Scholar] [CrossRef] [PubMed]
- Okada, A.; Ohta, Y.; Brody, S.L.; Watanabe, H.; Krust, A.; Chambon, P.; Iguchi, T. Role of foxj1 and estrogen receptor alpha in ciliated epithelial cell differentiation of the neonatal oviduct. J. Mol. Endocrinol. 2004, 32, 615–625. [Google Scholar] [CrossRef] [PubMed]
- Akison, L.K.; Boden, M.J.; Kennaway, D.J.; Russell, D.L.; Robker, R.L. Progesterone receptor-dependent regulation of genes in the oviducts of female mice. Physiol. Genom. 2014, 46, 583–592. [Google Scholar] [CrossRef] [PubMed]
- Murray, M.K. Epithelial lining of the sheep ampulla oviduct undergoes pregnancy-associated morphological-changes in secretory status and cell height. Biol. Reprod. 1995, 53, 653–663. [Google Scholar] [CrossRef] [PubMed]
- Shirley, B.; Reeder, R.L. Cyclic changes in the ampulla of the rat oviduct. J. Exp. Zool. 1996, 276, 164–173. [Google Scholar] [CrossRef]
- Kolle, S.; Dubielzig, S.; Reese, S.; Wehrend, A.; Konig, P.; Kummer, W. Ciliary Transport, Gamete Interaction, and Effects of the Early Embryo in the Oviduct: Ex Vivo Analyses Using a New Digital Videomicroscopic System in the Cow. Biol. Reprod. 2009, 81, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Ozen, A.; Ergun, E.; Kurum, A. Histomorphology of the oviduct epithelium in the Angora rabbit. Turk. J. Vet. Anim. Sci. 2010, 34, 219–226. [Google Scholar]
- Gonella-Diaza, A.M.; Mesquita, F.S.; da Silva, K.R.; de Carvalho Balieiro, J.C.; dos Santos, N.P.; Pugliesi, G.; de Francisco Strefezzi, R.; Binelli, M. Sex Steroids Modulate Morphological and Functional Features of the Bovine Oviduct. Cell Tissue Res. 2017, 370, 319–333. [Google Scholar] [CrossRef] [PubMed]
- Samuelson, D.A. Textbook of Veterinary Histology; Saunders-Elsevier: St. Louis, MO, USA, 2007; ISBN 10 0721681743. [Google Scholar]
- Donnez, J.; Casanas-Roux, F.; Caprasse, J.; Ferin, J.; Thomas, K. Cyclic changes in ciliation, cell height, and mitotic activity in human tubal epithelium during reproductive life. Fertil. Steril. 1985, 43, 554–559. [Google Scholar] [CrossRef]
- Bylander, A.; Nutu, M.; Wellander, R.; Goksor, M.; Billig, H.; Larsson, D.G. Rapid effects of progesterone on ciliary beat frequency in the mouse fallopian tube. Reprod. Biol. Endocrinol. 2010, 8, 48. [Google Scholar] [CrossRef] [PubMed]
- Bylander, A.; Lind, K.; Goksor, M.; Billig, H.; Larsson, D.G. The classical progesterone receptor mediates the rapid reduction of fallopian tube ciliary beat frequency by progesterone. Reprod. Biol. Endocrinol. 2013, 11, 33. [Google Scholar] [CrossRef] [PubMed]
- Teilmann, S.C.; Clement, C.A.; Thorup, J.; Byskov, A.G.; Christensen, S.T. Expression and localization of the progesterone receptor in mouse and human reproductive organs. J. Endocrinol. 2006, 191, 525–535. [Google Scholar] [CrossRef] [PubMed]
- Nutu, M.; Weijdegard, B.; Thomas, P.; Thurin-Kjellberg, A.; Billig, H.; Larsson, D.G. Distribution and hormonal regulation of membrane progesterone receptors beta and gamma in ciliated epithelial cells of mouse and human fallopian tubes. Reprod. Biol. Endocrinol. 2009, 7, 89. [Google Scholar] [CrossRef] [PubMed]
- Orihuela, P.A.; Croxatto, H.B. Acceleration of oviductal transport of oocytes induced by estradiol in cycling rats is mediated by nongenomic stimulation of protein phosphorylation in the oviduct. Biol. Reprod. 2001, 65, 1238–1245. [Google Scholar] [CrossRef] [PubMed]
- Orihuela, P.A.; Rios, M.; Croxatto, H.B. Disparate effects of estradiol on egg transport and oviductal protein synthesis in mated and cyclic rats. Biol. Reprod. 2001, 65, 1232–1237. [Google Scholar] [CrossRef] [PubMed]
- Orihuela, P.A.; Parada-Bustamante, A.; Cortes, P.P.; Gatica, C.; Croxatto, H.B. Estrogen receptor, cyclic adenosine monophosphate, and protein kinase A are involved in the nongenomic pathway by which estradiol accelerates oviductal oocyte transport in cyclic rats. Biol. Reprod. 2003, 68, 1225–1231. [Google Scholar] [CrossRef] [PubMed]
- Gandolfi, F.; Brevini, T.A.L.; Richardson, L.; Brown, C.R.; Moor, R.M. Characterization of proteins secreted by sheep oviduct epithelial-cells and their function in embryonic-development. Development 1989, 106, 303–312. [Google Scholar] [PubMed]
- Priedkalns, J.; Leiser, R. Female Reproductive System. In Dellmann’s Textbook of Veterinary Histology, 6th ed.; Blackwell Publishing: Hoboken, NJ, USA, 2006; ISBN 978-0-7817-4148-4. [Google Scholar]
- Hunter, R.H.F.; Wilmut, I. Sperm transport in the cow-peri-ovulatory redistribution of viable cells within the oviduct. Reprod. Nutr. Dev. 1984, 24, 597–608. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, A.; Syed, S.M.; Tanwar, P.S. In vivo genetic cell lineage tracing reveals that oviductal secretory cells self-renew and give rise to ciliated cells. Development 2017, 144, 3031–3041. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Kobayashi, Y.; Yamamoto, Y.; Kimura, K.; Okuda, K. Remodeling of bovine oviductal epithelium by mitosis of secretory cells. Cell Tissue Res. 2016, 366, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Demetrio, D.G.; Santos, R.M.; Demetrio, C.G.; Vasconcelos, J.L. Factors affecting conception rates following artificial insemination or embryo transfer in lactating Holstein cows. J. Dairy Sci. 2007, 90, 5073–5082. [Google Scholar] [CrossRef] [PubMed]
- Ashworth, C.J.; Sales, D.I.; Wilmut, I. Evidence of an association between the survival of embryos and the periovulatory plasma progesterone concentration in the ewe. J. Reprod. Fertil. 1989, 87, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Morris, D.; Diskin, M. Effect of progesterone on embryo survival. Animal 2008, 2, 1112–1119. [Google Scholar] [CrossRef] [PubMed]
- Mesquita, F.S.; Pugliesi, G.; Scolari, S.C.; Franca, M.R.; Ramos, R.S.; Oliveira, M.; Papa, P.C.; Bressan, F.F.; Meirelles, F.V.; Silva, L.A.; et al. Manipulation of the periovulatory sex steroidal milieu affects endometrial but not luteal gene expression in early diestrus Nelore cows. Theriogenology 2014, 81, 861–869. [Google Scholar] [CrossRef] [PubMed]

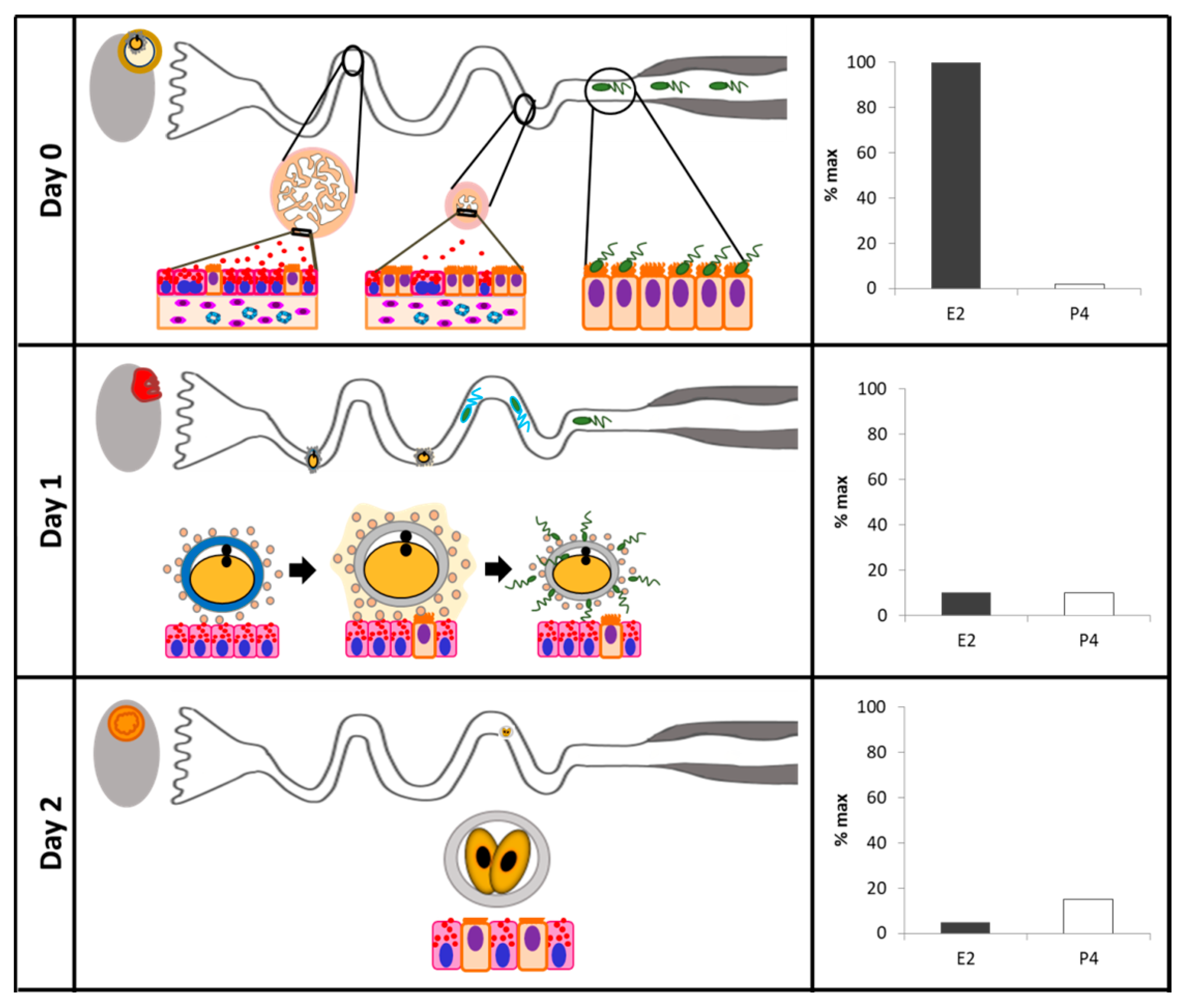
| Species | Studied Effect | Sample Type | Phase of the Cycle | Method Used | Sex Steroids-Mediated Effect | Source |
|---|---|---|---|---|---|---|
| Cattle | Characterization of Glycoproteins | Tissue sections of ampulla and isthmus | Days 0, 5, 10, 15, and 18 | Electrophoresis and fluorography, Western blot, light microscopy, and colloidal gold immunolabeling | The cattle oviductal epithelium synthesizes and secretes a class of oviduct-specific glycoprotein that is present in luminal fluid at the time when fertilization occurs. | Boice et al., 1990 [114] |
| Cattle | Oviductal secretory proteins in cows with persistent dominant follicle | Infundibulum, ampulla and isthmus explants | Cows with fresh or persistent dominant follicle (estrus) | In vitro culture of explants, two-dimensional electrophoresis, densitometry | Rate of incorporation of [3H]leucine into macromolecules was greater in the infundibulum, ampulla, and isthmus of cows with a fresh dominant follicle. | Binelli et al., 1999 [115] |
| Cattle | Expression of β2-adrenergic receptors | Oviductal epithelial cells collected after flushing the oviductal lumen. | Secretory, early to midluteal, proliferative, and preovulatory phases | cDNA cloning, in vitro autoradiography | P4 upregulated the β2-adrenergic receptor. | Einspanier et al., 1999 [116] |
| Cattle | Expression of E2 and P4 receptors in vivo and in vitro | Ampulla and isthmus epithelial cells | Early-luteal, mid-luteal, late-luteal, and follicular stages | PCR, Western blot, and immunohistochemistry | PR and ER mRNA transcripts were elevated in vivo during the follicular phase. The highest PR and ER protein expression was detected during the early-luteal phase. In vitro, E2-supplementation resulted in an upregulation of PR and ER. | Ulbrich et al., 2003 [107] |
| Cattle | Tumor necrosis factor expression during the estrous cycle | Whole oviductal tissue sections | Follicular, postovulatory and luteal phases | In vitro microdialysis in organ culture chamber and qPCR | Infusion of TNFa stimulated oviductal secretion of PG, ET-1, and Ang II during the follicular and postovulatory stages, but not during the luteal stage. High expression of both TNFaR types and ligands was detected during the follicular and postovulatory stages, whereas low expression was detected during the luteal stage. | Wijayagunawardane et al., 2003 [117] |
| Cattle | Comparison of ipsilateral vs. contralateral to corpus luteum (CL) oviducts | Oviductal epithelial cells | Day 3.5 | Subtraction cDNA libraries and cDNA array hybridization. | 35 cDNAs differentially expressed. The regulated genes or their products include a variety of functional classes such as cell-surface proteins, cell–cell interaction proteins, members of signal transduction pathways, immune-related proteins, and enzymes. | Bauersachs et al., 2003 [109] |
| Cattle | Comparison of oviductal samples of estrous vs. diestrus phases | Oviductal epithelial cells | Day 1 vs. Day 12 | cDNA libraries and cDNA array hybridization | 77 differently expressed cDNAs. Thirty-seven were expressed at a higher level at estrus. During estrus genes involved in the regulation of protein secretion and protein modification were upregulated, whereas during diestrus, particularly, transcripts of genes involved in transcription regulation were upregulated. | Bauersachs et al., 2004 [110] |
| Cattle | Expression of phospholipid hydroperoxide glutathione peroxidase (GPx-4) | Tissue fragments of isthmus, ishtmic-ampullary junction, and ampulla | Day 16 after E2 intrauterine infusion | In situ hybridization, qPCR, and activity assay for GPx-4. | GPx-4 expression was 2-fold higher in the oviducts of cows treated with uterine infusions of E2. | Lapointe et al., 2005 [118] |
| Postovulatory, mid-luteal, late luteal, and follicular stages | There is a cell-specific distribution for GPx mRNAs in the different oviduct segments. GPx-4 expression was highest during the follicular, postovulatory, and late luteal stages. | |||||
| Cattle | Expression of E2 and P4 receptors in natural and superovulated estrous cycles | Tissue sections of infundibulum, ampulla, ampullary/isthmic transition, and isthmus of both oviducts | For natural estrous cycle, samples were collected at 17 h, 4, and 11 days after estrus. In the superovulatory treatment, samples were collected 17 h and 4 days after estrus | Immunohistochemistry | There is a positive correlation between circulating concentrations of E2 and P4 and ER staining. There is a negative correlation of PR staining with P4 circulating concentrations. There was no effect of superovulation. | Ribeiro Valle et al., 2007 [108] |
| Cattle | Expression of P4 receptors (nuclear and membrane components) of cyclic and pregnant cows | Sections of the whole oviduct | 1.5, 4, and 5 days post-ovulation | RT-PCR, qPCR, Western blot, and immunohistochemistry | No obvious differences in localization patterns of PR, PGRMC1 and PGRMC2 were observed between ipsi- and contralateral oviducts or according to the stage post-ovulation. | Saint-Dizier et al., 2012 [8] |
| Cattle | Gene expression in oviductal samples of follicular vs. luteal phases | Epithelial cells from ampulla or isthmus | 36 h after PGF vs. Days 11 and 12 | Microarray | 972 and 597 transcripts in the ampulla and 946 and 817 transcripts in the isthmus were up- and downregulated in the Follicular phase compared to Luteal phase. Upregulated genes were involved in cholesterol biosynthesis and cell cycle pathways, while downregulated genes were found in numerous inflammatory response pathways. | Cerny et al., 2015 [111] |
| Cattle | Large vs. small pre-ovulatory follicle | Ampulla and isthmus ipsilateral to the ovary containing the dominant follicle | Day 4 | RNA sequencing (RNAseq), ERa and PGR immunohistochemistry | There was a greater abundance of PGR and ERa in the oviducts of cows having a Large preovulatory follicle. 325 and 274 transcripts were upregulated in the large follicle group, for ampulla and isthmus, respectively. | Gonella-Diaza et al., 2015 [112] |
| Sheep | Oviductal proteome | Oviductal fluid | Day 0 vs. Day 10 | In-gel digestion coupled with mass spectrometric analysis (GeLC-MS/MS) | The proteins more abundant at estrus included several families such as the heat shock proteins, mucins, complement cascade proteins, and several redox enzymes. The proteins more abundant during the luteal phase were associated with the immune system and tissue remodeling. | Soleilhavoup et al., 2016 [119] |
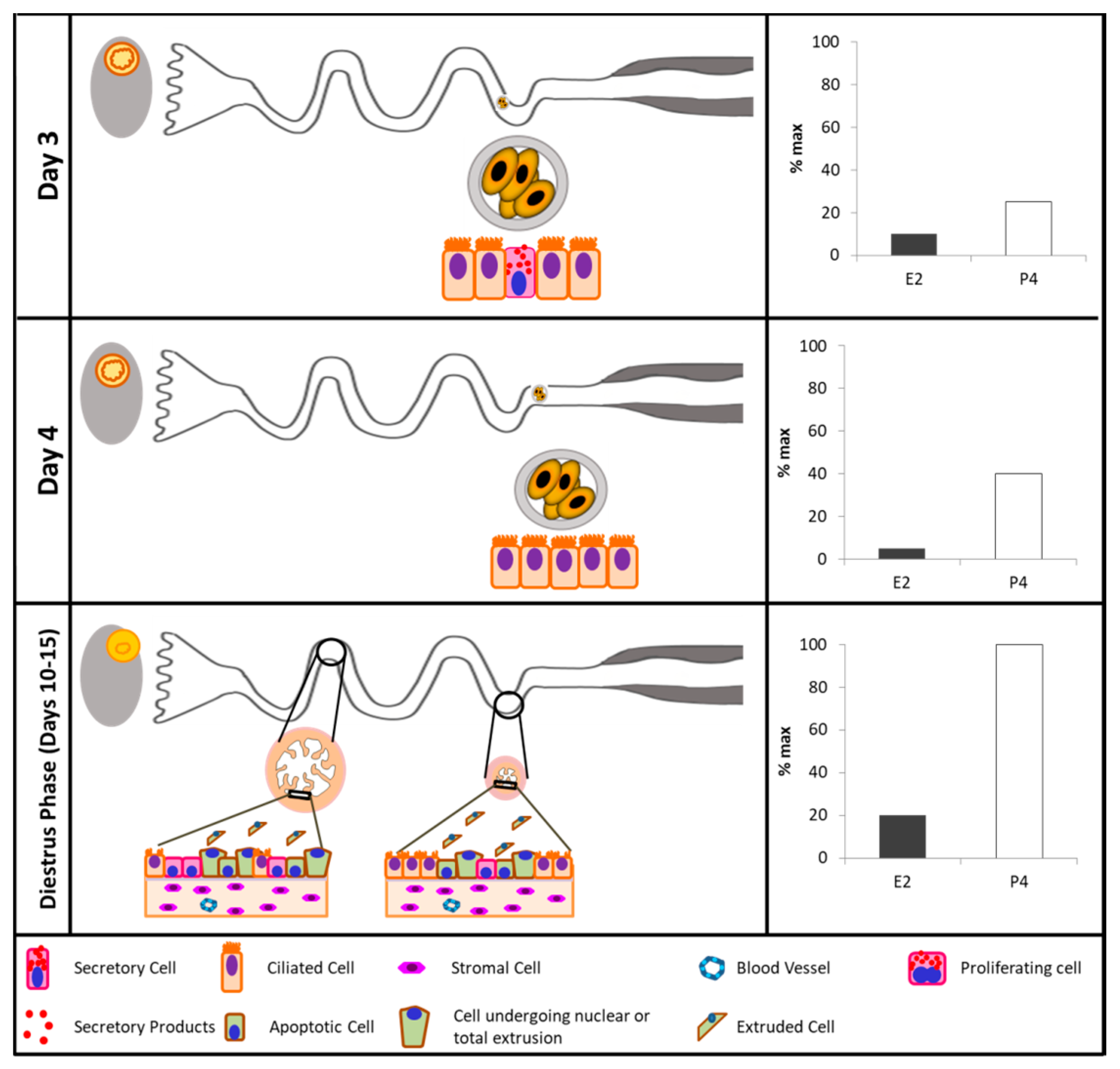
| Species | Studied Effect | Sample Type | Phase of the Cycle | Method Used | Sex Steroids-Mediated Effect | Source |
|---|---|---|---|---|---|---|
| Cattle | P4 effect of oviductal cytology | Tissue sections of infundibulum, ampulla, and isthmus | Estrus and early postestrus, early and middle luteal phase, late-luteal phase, and proestrus-follicular phase. | Frozen sections stained with calcium-cobalt method, methyl greenpyronin Y, Best’s carmine staining for glycogen, |, alcian blue, and toluidine blue | Cell heights and cytoplasmic content of ciliated and secretory cells in ampulla and isthmus increased in the presence of E2 and decreased in the presence of P4. In luteal phase or P4-treated animals, secretory cells had extruded nuclei and epithelial desquamation. | McDaniel et al., 1968 [103] |
| Ovariectomized animals treated with E2, P4, or E2 + P4 | ||||||
| Cattle | Oviductal motility | Recordings of electrical activity | Diestrous, proestrus, estrous, and metestrus | Electrical activity was directly recorded on a polygraph | Electrical activity start to increase at proestrus and was more intense (amplitude and frequency) at estrus and metestrus. | Ruckebusch & Bayard, 1975 [104] |
| Cattle | Effect of supplementation with P4 or E2 on oviductal transport | Oviductal flushing | 60 h post-estrous | The oviduct was cut into 8 equal segments; each one was flushed with 1 mL of saline. Flushings were evaluated to determine the presence of an oocyte or zygote | The transport rate was increased by P4 but not affected by estradiol benzoate. | Crisman et al., 1980 [27] |
| Sheep | Ampullary epithelium morphology during early pregnancy | Tissue section of the ampulla | Days 0, 1.5, 2, 3, 4, 6, and 16 of pregnancy | Toluidine blue staining and electron microscopy | The ampullary secretory cells undergo morphological alterations in protein-synthesizing organelles and apical specializations that vary with the stage of pregnancy.The secretory products contained in cytoplasmatic granules are released by the process of exocytosis until Days 3 and 4. Cell death appears to occur at Day 16 by shedding of epithelial cells into the oviductal lumen. | Murray, 1995 [135] |
| Rat | Cyclic changes in the ampulla | Tissue sections of the ampulla | Estrus, metestrus, diestrus-1, diestrus-2, and proestrus | Electron microscopy | The days in which oocytes can be found inside the ampullary lumen are the same days when the ampullary secretory cells exhibited morphological changes indicative of increased activity. | Shirley & Reeder, 1996 [136] |
| Mice | Secretory cells in adult or prepubertal E2-treated mice | Oviductal tissue fragments | Proestrus, oestrus, metestrus, diestrus | Transmission electron microscopy | The proportion of secretory cells and production of secretory products increased around and after ovulation in cycling animals. E2 administration accelerates the differentiation and maturation of the secretory cells. | Lauschová, 2003 [120] |
| Cattle | Ciliary transport, gamete interaction, and effects of the early embryo | Ex vivo analyses with digital video microscopic system | Days 2.5, 3.5, and 4.5 of pregnancy. | Video microscopy | There are secretory and ciliated cells in the ampullary epithelium. Entering the isthmus, secretory and ciliated cells are observed. In the end portion of the isthmus, only ciliated cells are present in the oviductal epithelium. | Kolle et al., 2009 [137] |
| Rabbit | Oviductal morphology | Tissue sections of ampulla and isthmus | Estrus and luteal stages | Mallory’s triple stain, PAS, Alcian blue, PAS/Ab reaction, Aldehyde fuchsin/Alcian blue (AF/Ab), and electron microscopy | The numbers of secretory cells and the amount of secretions increases in the estrus stage.The amount of secretions and the number of cilia were demonstrated to decrease evidently in the luteal stage. | Özen et al., 2010 [138] |
| Buffalo | Oviductal morphology | Tissue sections of infundibulum, ampulla, and isthmus. | Follicular vs. luteal phase | Haematoxylin and eosin, verhoffe, toluidine blue, and PAS staining methods. | The ampulla epithelium is highly folded, taller, and presents more cells with secretory activity at the follicular phase. There is no difference in the microscopic structure of the isthmus at follicular and luteal phases. The thickness of tunica muscularis at luteal phase is significantly greater than the follicular phase in all regions. | Ayen et al., 2012 [96] |
| Cattle | Oviductal cell and tissue morphology | Tissue sections of infundibulum and ampulla | Luteal and follicular phases | Harris haematoxylin and eosin, van Gieson resorcin fuchsin, Goldener’s trichome stain, alcian blue stain, PAS stain, Sudan black, Gomori lead nitrate, toluidine blue staining | The secretory cells were predominant in the luteal phase with numerous apical cytoplasmic protrusions. In the ampulla the number of mucosal folds and length of the primary folds were greater at the follicular phase | Mokhtar, 2015 [131] |
| Cattle | Oviductal cell and tissue morphology | Ipsilateral and contralateral isthmus and ampulla | Day 4 | Hematoxylin and eosine staining, PAS staining, and Ki67 immunedetection. | Animals that ovulated a large preovulatory follicle presented more primary mucosal folds, a greater mucosal-folding grade and luminal perimeter, and more secretory and proliferating cells. | Gonella-Diaza et al., 2017 [139] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Binelli, M.; Gonella-Diaza, A.M.; Mesquita, F.S.; Membrive, C.M.B. Sex Steroid-Mediated Control of Oviductal Function in Cattle. Biology 2018, 7, 15. https://doi.org/10.3390/biology7010015
Binelli M, Gonella-Diaza AM, Mesquita FS, Membrive CMB. Sex Steroid-Mediated Control of Oviductal Function in Cattle. Biology. 2018; 7(1):15. https://doi.org/10.3390/biology7010015
Chicago/Turabian StyleBinelli, Mario, Angela Maria Gonella-Diaza, Fernando Silveira Mesquita, and Claudia Maria Bertan Membrive. 2018. "Sex Steroid-Mediated Control of Oviductal Function in Cattle" Biology 7, no. 1: 15. https://doi.org/10.3390/biology7010015





