1. Introduction
Skeletal muscle plays a central role in physiological, metabolic and nutritional processes in the human body as well facilitating locomotion. It undergoes continual adaptation during adult life, through its ability to activate robust anabolic processes involving increases in protein synthesis rates as well as utilizing stem cell populations to promote tissue growth and repair.
Recent work has shown that satellite cells, the resident stem cell population in skeletal muscle, are responsible for most if not all tissue repair following acute injury [
1,
2]. In resting undamaged muscle, satellite cells exist in a quiescent state on the sarcolemma of muscle fibers but under the endomysial extra-cellular matrix (ECM). Satellite cells are relatively metabolically inactive; evident by the sparse cytoplasmic content and associated organelles in particular mitochondria and are highly reliant on glycolysis for ATP production. Quiescent satellite cells are identifiable through their expression of Pax-7, a key gene that imbues stem cell properties [
3]. Following tissue damage, satellite cells undergo major transcriptional changes (termed activation) in particular the induction of the myogenic determination factor MyoD [
4]. Activated satellite cells remodel the ECM, eventually degrading it and assume a supra-basal position.
The ECM on which activated satellite cells perform their tissue repair properties differ greatly from traditionally used
in-vitro conditions. This is important as the ECM influences stem cell properties in terms of self-renewal versus myogenic commitment decisions but also on the mechanisms facilitating cell migration. Satellite cells cultured on plastic migrate through a mesenchymal form of movement reliant on a polarized cell forming a lamellipodia at the leading edge [
5]. The same cells on their native substrate move by a different mechanism, reliant on the formation and retraction of plasma membrane protrusions called blebs which enable migration of a variety of cell types during development and disease [
6,
7]. Bleb expansion is driven by hydrostatic pressure generated in the cytoplasm while the contractile actomyosin cortex underpins retraction [
8]. Blebbing-based migration allows satellite cells to move at high speeds and permits rapid changes in direction, two activities supporting tissue repair.
There is huge commercial interest in developing artificial matrixes that can be seeded with satellite cells to form contracting myofibers with a view of deploying them to replace damaged tissue following severe trauma e.g., blast wounds. Materials as diverse as polyethylene glycol (PEG) to electro-spun graphene have been made into scaffolds for skeletal muscles [
9,
10]. However, many studies examining the suitability of artificial scaffolds for muscle tissue repair only superficially scrutinize the impact of novel substrates on satellite cell behavior. We suggest that it is very important the artificial matrixes support the normal activities of satellite cells which ultimately control their robust tissue repair properties. To that end, it is vital that we determine the influences of the environment on stem cell behavior.
In this study we have investigated the influence of the ECM and glycolytic activity on satellite cell emergence and migration. We took advantage of the myostatin null mouse that contains much thinner ECM surrounding muscle fibers compared to that of a wild-type mouse. Secondly we exploited the availability of 3-Bromo pyruvate (3-BP) a small compound inhibitor that perturbs glycolysis [
11]. Both studies reveal unexpected findings. Firstly, our results show that decrease in ECM thickness does not translate into higher rates of satellite cells emergence and highlights the impact of endomysium composition on satellite cell behavior. Secondly we demonstrate that glycolysis impacts hugely on the migratory process; its inhibition leads to precocious emergence followed by an attenuation in migration. We suggest that both these parameters need to be given due consideration during the development of artificial muscle matrixes.
2. Methods
2.1. Ethical Approval
Animals were maintained and experiments performed under license from the UK Home Office in accordance with the Animals (Scientific Procedures) Act 1986 and complied with the policy and regulations of the journal.
2.2. Single Myofiber Isolation and Culture
Single muscle fibers were isolated from the extensor digitiorum longus (EDL) muscle of 12-week old male C57Bl6 and myostatin null mice on identical background [
4]. In brief an EDL was dissected with both tendons intact and individual myofibers were generated by digestion with 0.2% type I collagenase in Dulbecco modified eagle medium (DMEM) at 37 °C under 5% CO
2. Single fibers were plated in floating culture wells containing single fiber culture medium (SFCM, DMEM supplemented with 10% horse serum, 0.5% chick embryo extract and 1% penicillin/streptomycin) using tapered glass pipettes. When required 3-Bromo pyruvate (50, 100 and 500 µM) (Sigma-Aldrich, Gillingham, UK) was introduced to the SFCM either from the point of myofiber culture initiation or 24 h post myofiber isolation.
2.3. Time-Lapse Microscopy
Following 24 h of culture, myofiber cultures were monitored using a phase contrast microscope housed in an environmental chamber maintained at 37 °C and supplemented with 5% CO
2. Time-lapse video was taken at a rate of one frame/15 min over 24 h using 10× objectives [
3,
4]. High-power time-lapse microscopy was carried out using 100X objectives enabling the visualization of cell-surface blebbing. Fibers were cultured for 48 h and subsequently filmed at a rate of one frame/2 s for these studies.
2.4. Immunocytochemistry
Immunocytochemistry of satellite cells on single myofibers was carried out as described previously [
4]. Briefly, at pre-determined time points myofibers were fixed in 4% PFA/PBS for 15 min. Thereafter, fibers were washed three times in PBS. Myofibers were permeabilized in a solution of 20 mM HEPES, 300 mM sucrose, 50 mM NaCl, 3 mM MgCl
2, and 0.5% Triton X-100 (pH 7) at 4 °C for 15 min and incubated in blocking wash buffer (5% foetal calf serum in PBS containing 0.01% Triton X-100) for 30 min prior to antibody incubation. Antibodies were diluted and pre-blocked in wash buffer for 30 min prior to addition to the myofibers. Primary antibodies used: Polyclonal rabbit anti-Laminin (Sigma L-9393 1:200), monoclonal mouse anti-Pax7 (Developmental Studies Hybridoma Bank [DSHB] 1:1), polyclonal rabbit-anti-MyoD (Santa Cruz biotechnology 1:200) and monoclonal anti-α smooth muscle actin (Sigma A 2547 1:300). All primary antibodies incubations were for 12 h at 4 °C. Primary antibodies were visualized using the following secondary antibodies: Alexa Fluor goat anti-mouse 594 (Molecular probes A11032) and Alexa Fluor goat anti-rabbit 488 (Molecular probes A11034). Secondary antibodies were used at 1:200 and incubated at room temperature for 45 min. Myofibers were mounted in fluorescent mounting medium (DAKO) containing 7.5 mg/mL 4′,6-diamidino-2-phenylindole (DAPI) for nuclear visualization.
2.5. Fluorescence Microscopy
Mounted myofibers were analyzed using a Zeiss Axioscope fluorescent microscope, and images were captured using the Axiocam digital camera system and Axiovision image analysis software (version 4.7).
2.6. Electron Microscopy
Myofibers were fixed in 4% PFA for 15 min after 24, 48, or 72 h in standard culture conditions. Myofibers were dehydrated through 30%, 50%, 70%, 80%, 90%, and 100% ethanol solution series (15 min for each step) and transferred to a critical point drier (Balzers CPD 030, using liquid carbon dioxide). Myofibers were transferred to scanning electron microscopy (SEM) chucks using micro-forceps and gold-coated using an Edwards S150B sputter-coater. Coated myofibers were then analyzed with a FEI 600F SEM.
2.7. Image and Movie Analysis
Image analysis was carried out using ImageJ (version 1.49 m). Satellite cells were tracked manually using the MTrackJ plugin. Bleb dynamics on high magnification movies and bleb number on SEM images were manually quantified using ImageJ. Satellite cell emergence profile and morphologies were manually assessed by examining live images captured by an Axiovision digital camera system. Data was processed for statistical significance using independent sample t-tests and one-way ANOVA with Bonferroni’s post-hoc analysis where appropriate, at the 95% confidence interval. All data are presented as mean ± SEM.
4. Discussion
Satellite cells, the resident stem cells of skeletal muscle occupy a unique environment which has a major impact on these cells to carry out tissue repair [
2]. It is very important for tissue engineering purposes to understand the influence of the environment on the activity of satellite cells if they are going to be exploited for regenerative uses.
In this study, we examined the behavior of satellite cells on their native substrate in two settings; one in which there was a decrease in the ECM content and the other where there was a change in glycolytic metabolism.
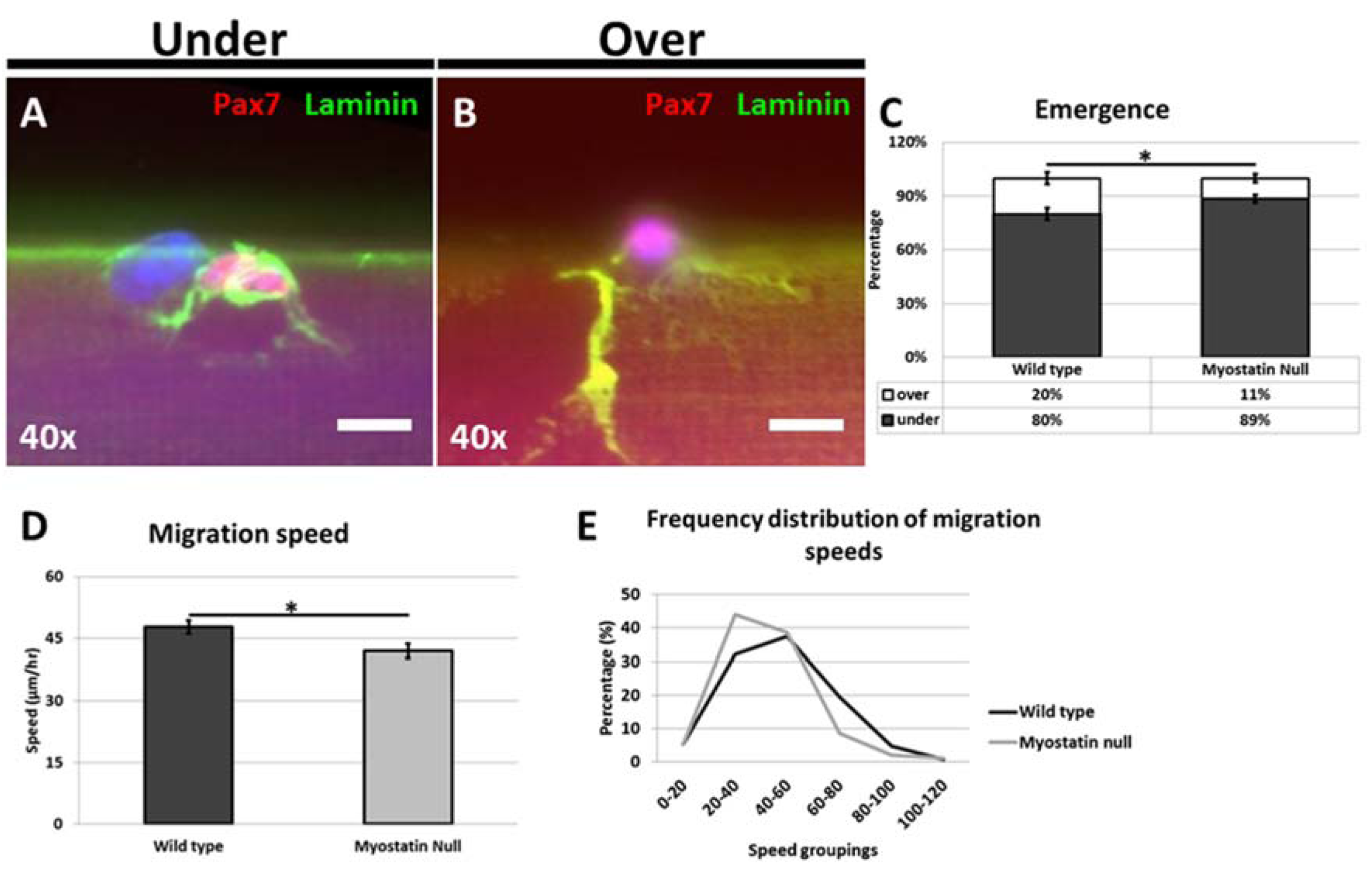
As a start point we suggest that any differences revealed in the Mstn
-/- experiments depend on properties of the muscle fiber and not the satellite cells. This assumption is based on the findings that muscle fibers express both the ligand (Mstn) and its receptors and therefore are influenced by deletion of the gene, evident by changes in size and composition. In contrast, the satellite cells from Mstn
-/- mice are equivalent to their WT counterparts as neither express the Mstn ligand or receptor [
13]. In the first series of experiments we showed that in the absence of Mstn, satellite cells emerged from their sub-basal lamina position far slower than WT cells. This result was surprising given that the endomysium of Mstn null muscle is thinner than in WT muscle [
14]. Possible explanation for these findings could lie in the relationship between the ECM and enzymes responsible for its degradation during satellite cell activation. A number of studies have shown that matrix metalloproteinases (MMPs) are required for activation of satellite cells [
15]. We suggest that the ECM of Mstn
-/- fibers could have lower levels of these MMPs that would attenuate the emergence program. Studies are underway to test this line of investigation.
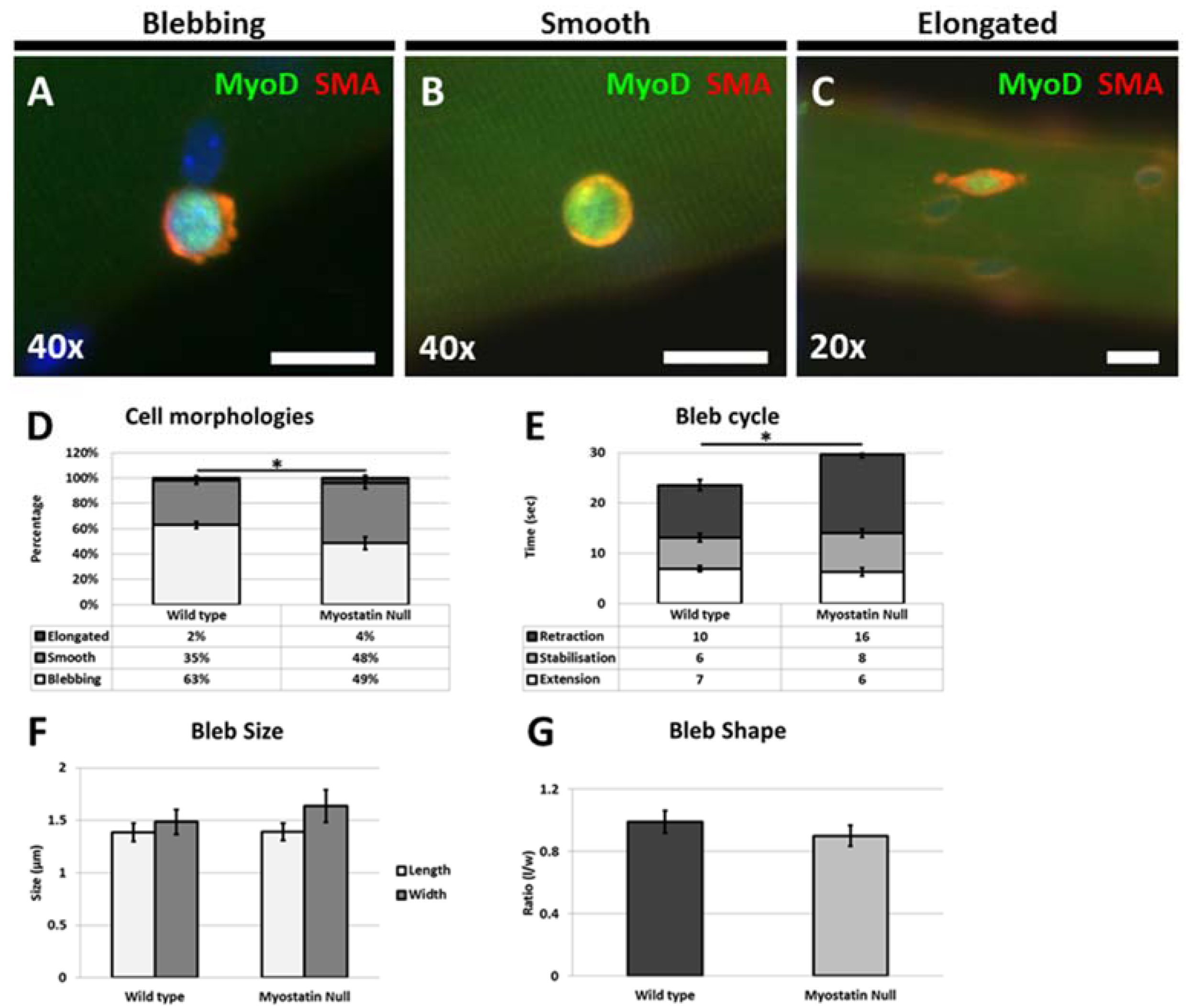
Our results also show for the first time that satellite cells from the hypertrophic mouse migrate at a significantly lower rate on their native substrate and that the migrating cells displayed altered morphologies. Interestingly we found that the even though the number of cells displaying a blebbing characteristic was significantly decreased they had not developed characteristics of migrating mesenchymal cells with the presence of a flattened leading edge composed of a lamellipodia [
5]. Previous work performed in cancer cells has shown the existence of antagonistic mechanisms that control cell shape and migration; the action of Rho mediated through ROCK promotes blebbing which acts to inhibit the action of WAVE that supports lamellipodia formation [
16]. In addition, WAVE2 inhibits the action of ROCK, leading to the scenario that attenuation of mechanisms supporting blebbing lead to a cell changing its morphology to develop a lamellipodia [
6,
16]. Here we show that this relationship is violated with the presence of rounded non-blebbing cells, which nevertheless fail to develop a lamellipodia. We suggest that ECM changes due to Mstn deletion results in modifications in the adhesion properties of the endomysium that permits some features of blebbing cells (rounding and the formation of an ordered cortical actin layer) but does not fully support dynamic membrane extension and retraction. However, the changes in the ECM in the absence of Mstn fail to result in a substrate that delivers the high level of stiffness that promotes the formation of a lamellipodia [
17].
Satellite cells in resting muscle exist in a quiescent state exemplified by the relative lack of cytoplasm and its associated organelles in relation to the size of the nucleus [
18]. The metabolic activity of the mitochondria in quiescent satellite cells is thought to be low due to its inactive state and to minimize the generation of reactive oxygen species (ROS) that would otherwise impact on the control of cell proliferation, survival, stem cell self-renewal as well as influence the molecular pathways that regulate differentiation.
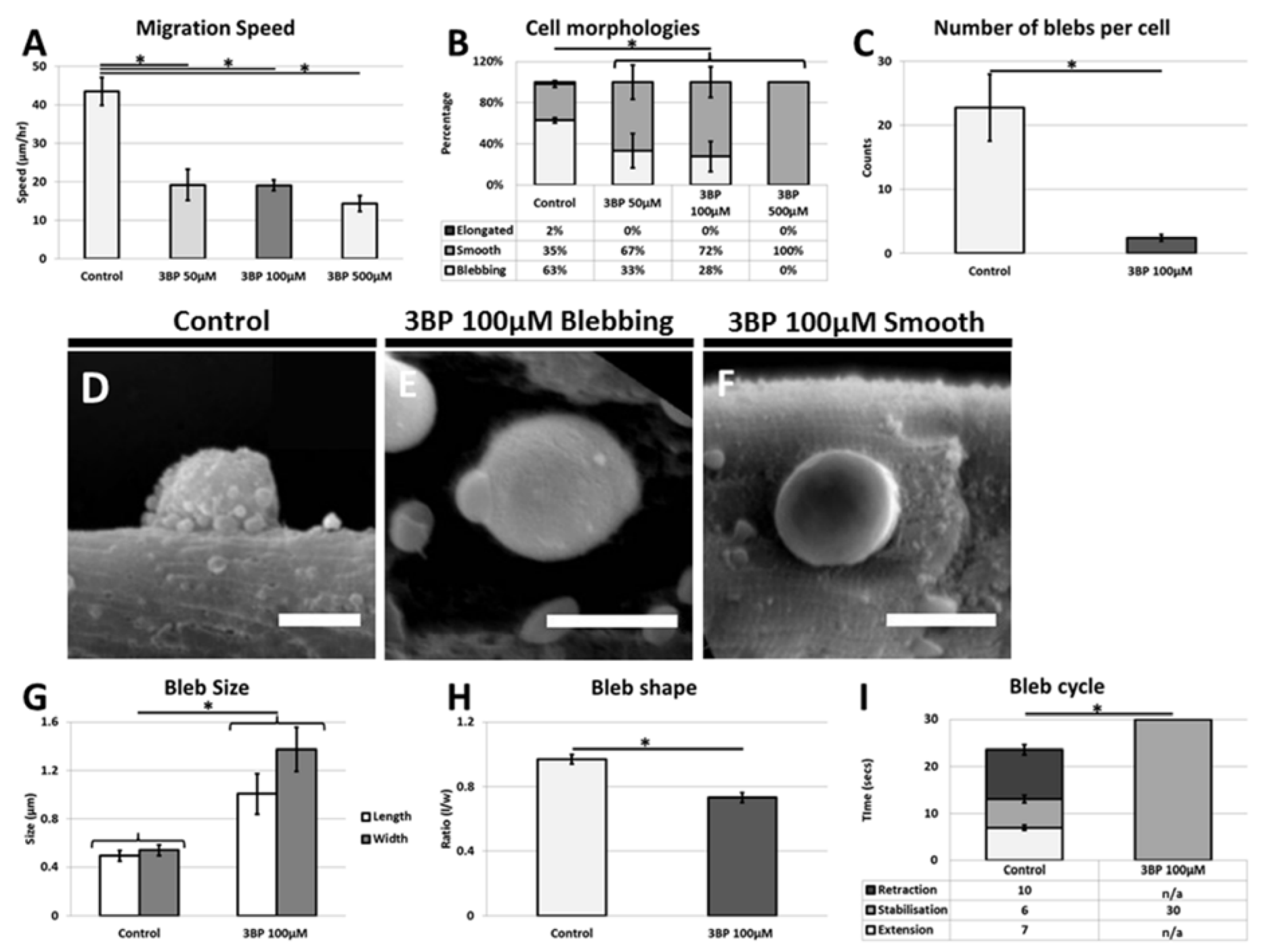
We showed that satellite cells are induced into activity following exposure of 3-BP a potent inhibitor of glycolysis [
11]. However once they have emerged from their sub-basal position, we found that their migratory processes were attenuated. We suggest that following the inhibition of glycolysis, satellite cells activate a stress response program as their major form of energy production has been blocked. These finding are congruent with previous studies on other stem cells populations including those of the hematopoietic system that too displays precocious activation after stress [
19].
However once they have emerged onto the muscle fiber, migration, one of the key activities undertaken by the stem cells is severely limited by the absence of glycolytic activity [
7,
20]. Our results show that this process must be highly energy dependent since the cells unable to fully undertake glycolysis decrease their migration rate by over 50%. Blebbing itself seems to be reliant on the glycolytic process since very few cells display anywhere near the normal number of plasma membrane protrusions. However, it is noteworthy that cells do not revert to using the lamellipodia means of translocation but still move. We suggest that glycolysis is used by the cells to promote cell migration through the highly energy demanding process of membrane extension and retraction. However, during decreased glycolysis, satellite cells still migrate but less rapidly. In such cases cell migration may be mediated by the rounded cells through a rolling action as seen during neutrophil extravasation process [
21]. These experiments clearly show the influence of 3-BP on the satellite cells. The muscle fiber too undertakes glycolysis and therefore it too must be influenced by 3-BP. We believe that the majority of changes in satellite cell behavior are due to the direct consequences of the 3-BP on the stem cells as opposed to indirect influences through changes in properties associated with the muscle fiber. This conclusion is based on the fact that many of the responses were detected within minutes after the introduction of 3-BP, whereas changes in the ECM of muscle fibers would be expected to take longer to alter. Future work is underway to examine in detail the changes in the muscle fiber properties following the inhibition glycolysis.
5. Conclusions
Our study examined the influence of ECM composition and metabolism on stem cell emergence, a key component of skeletal muscle regeneration. Results from the study were intriguing and unexpected. Firstly we showed that a thinner connective tissue covering the satellite cells does not result in a greater rate of emergence. In fact, the reduced endomysium of Mstn-/- mice impedes emergence. These results suggest that the composition of the endomysium rather than its quantity is the dominant determining factor controlling satellite cell emergence.
Secondly, we showed satellite cells need to perform glycolysis to maintain their sub-lamina position and that inhibition of glycolysis induces a stress process that forces them to emerge from their niche. However, the cells are unable to migrate in their normal manner, which would detrimentally impact on their ability to carry out muscle repair.