In Vitro Antibiofilm Efficacies of Different Antibiotic Combinations with Zinc Sulfate against Pseudomonas aeruginosa Recovered from Hospitalized Patients with Urinary Tract Infection
Abstract
:1. Introduction
2. Results and Discussion
2.1. Scanning Electron Microscopy of the Biofilm

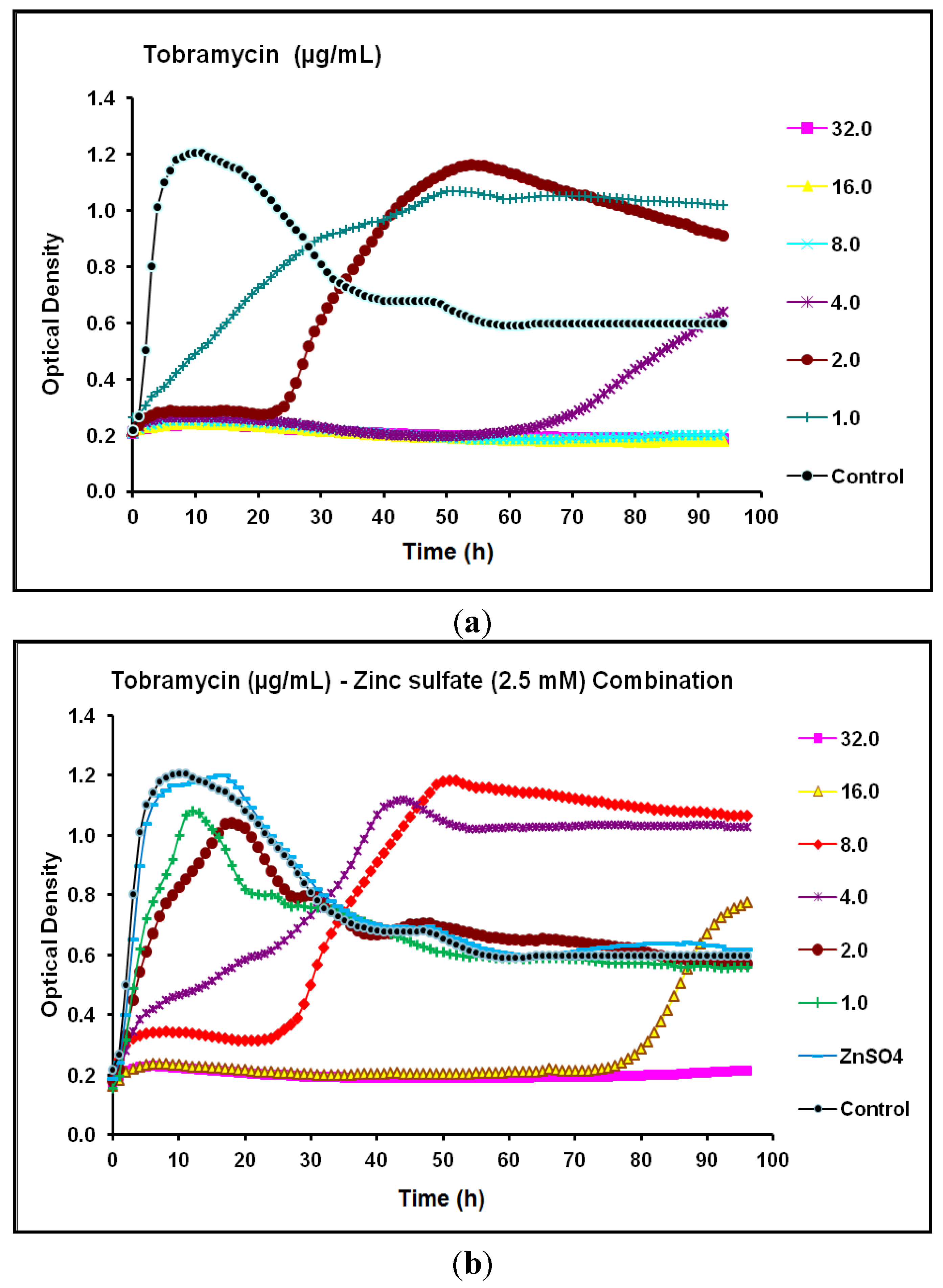
2.2. Real-Time Monitoring of P. aeruginosa CCIN34519 Biofilm
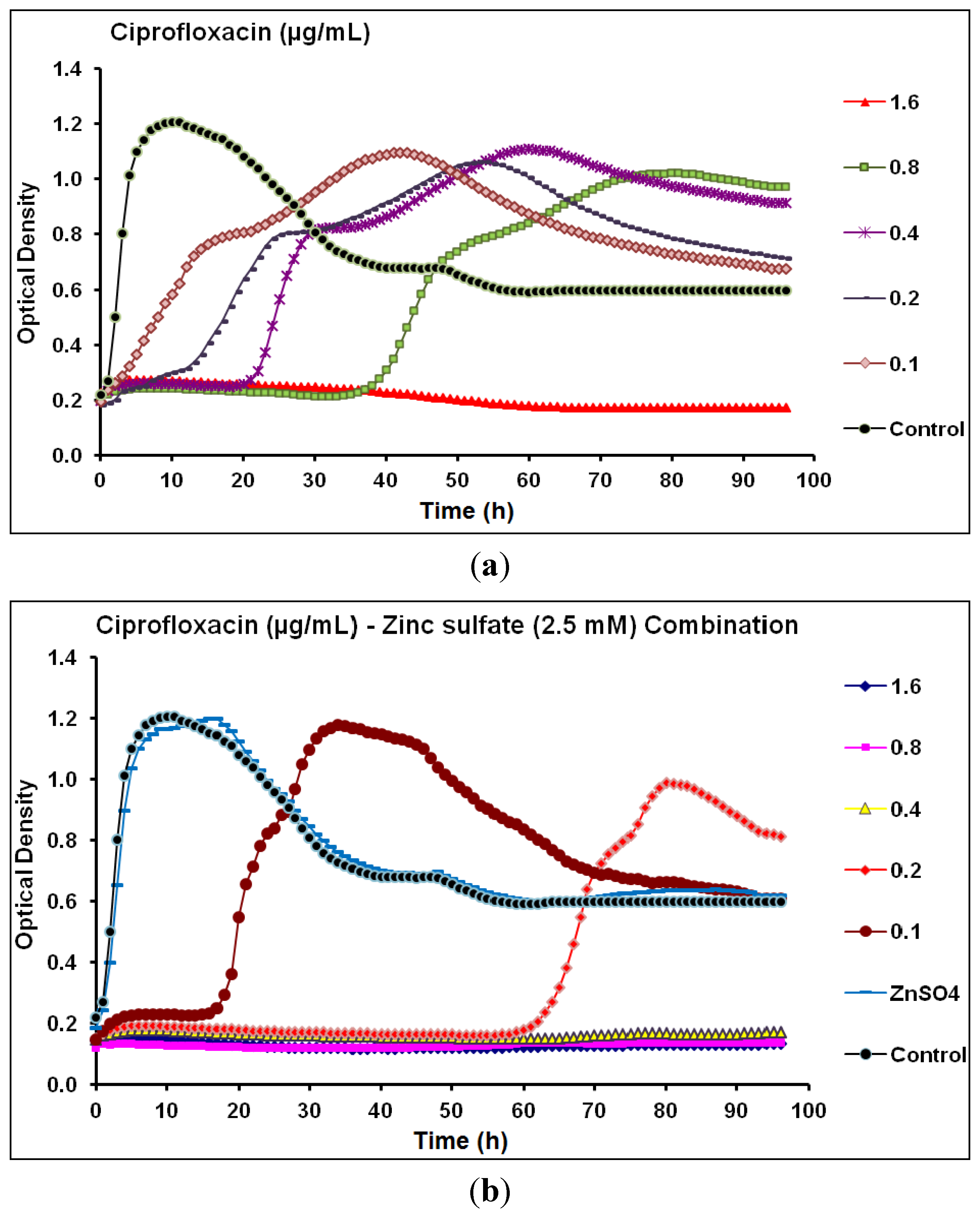


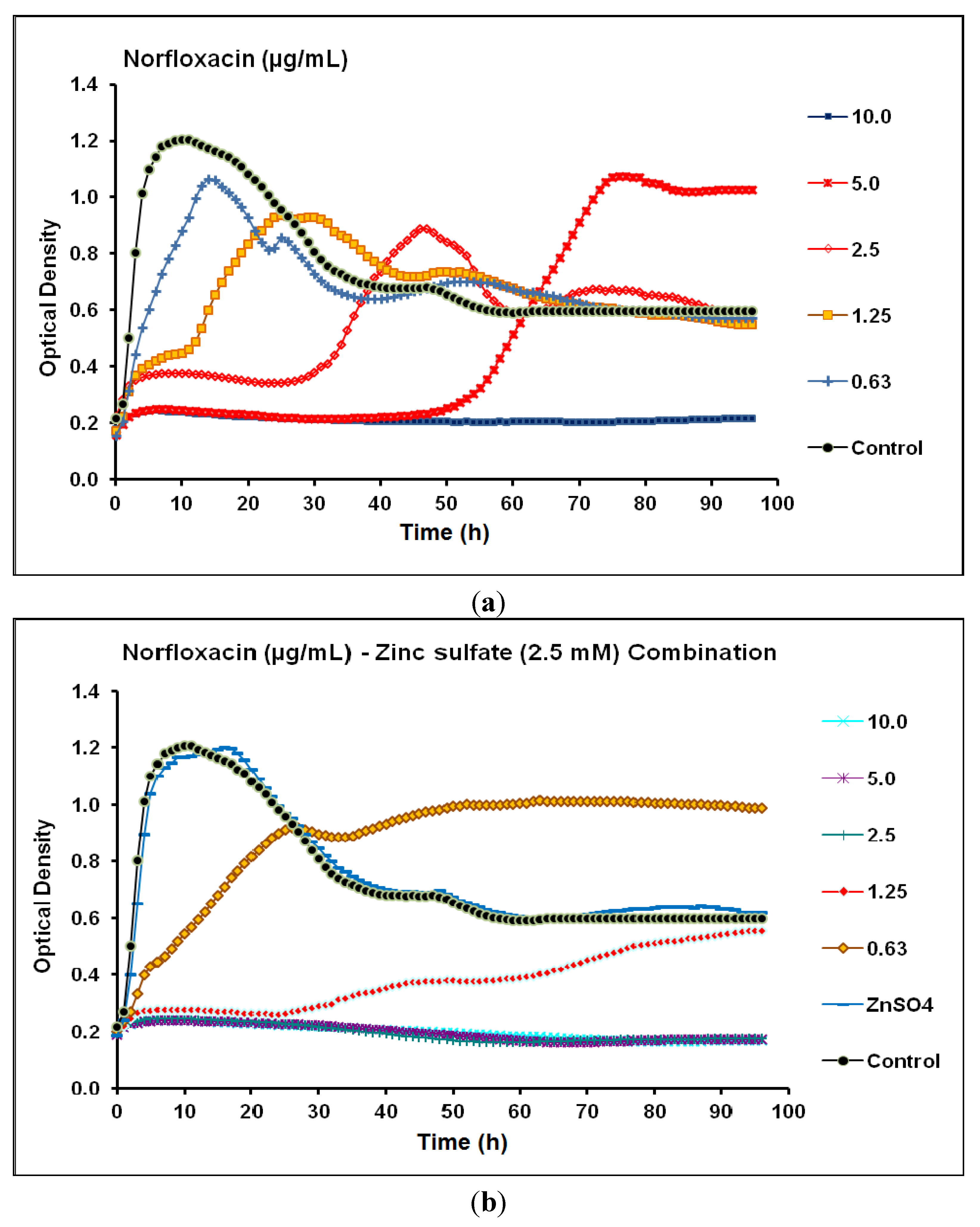

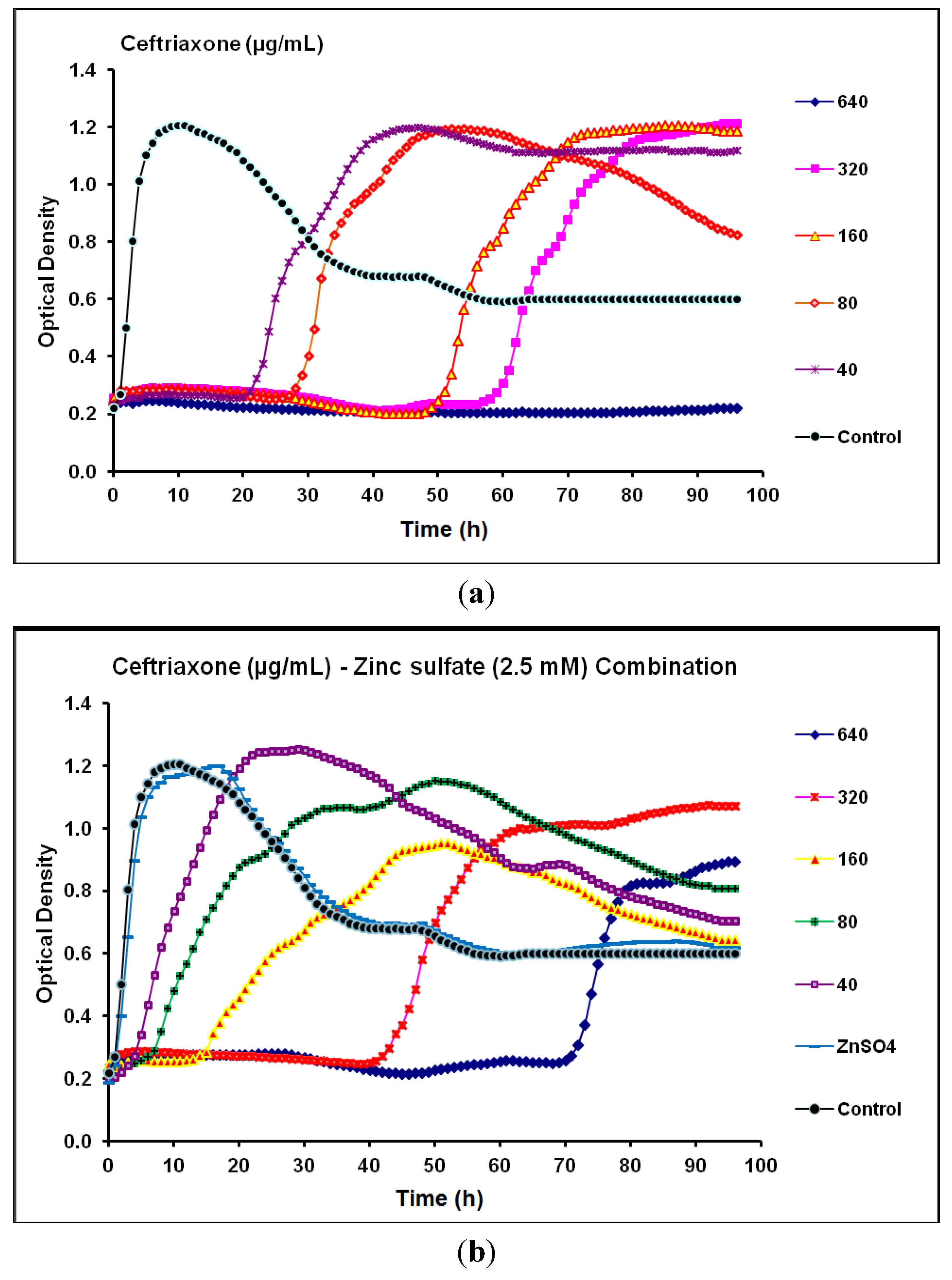
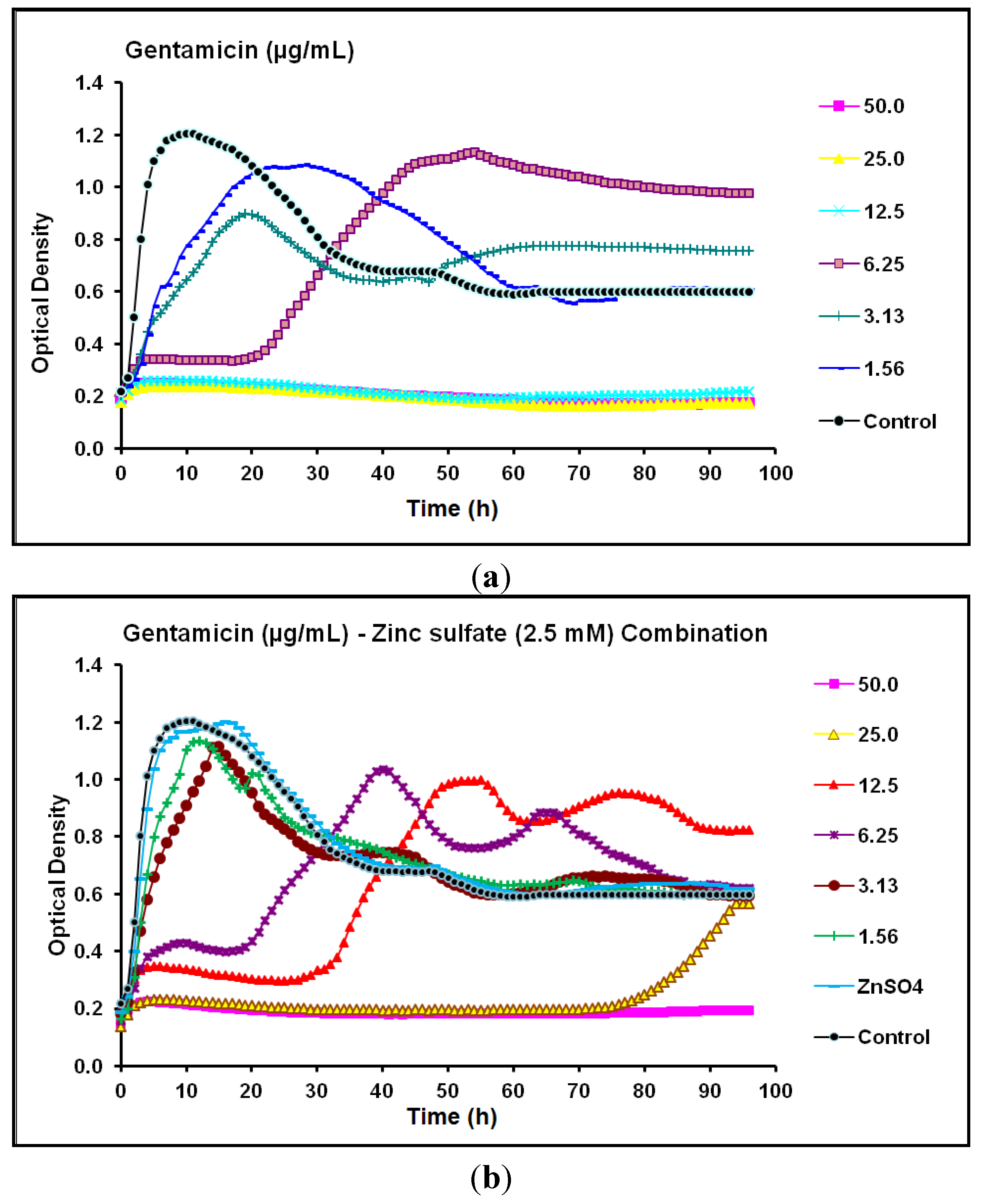
| Antibiotic | Class | Biofilm Without Zinc Sulfate | Biofilm With Zinc Sulfate * | Anti-biofilm Efficacy of Combination | ||
|---|---|---|---|---|---|---|
| MIC (24 h) | MPC (96 h) | MIC (24 h) | MPC (96 h) | |||
| Ciprofloxacin | Fluoroquinolone | 0.8 | 1.6 | 0.20 | 0.40 | Synergistic |
| Levofloxacin | Fluoroquinolone | 4.0 | 8.0 | 0.25 | 0.50 | Synergistic |
| Moxifloxacin | Fluoroquinolone | 16 | 32 | 2.0 | 8.0 | Synergistic |
| Norfloxacin | Fluoroquinolone | 5.0 | 10 | 1.25 | 2.5 | Synergistic |
| Ertapenem | Carbapenem | 20 | 20 | 10 | 10 | Synergistic |
| Ceftriaxone | Cephalosporin | 80 | 640 | 320 | > 640 | Antagonistic |
| Gentamicin | Aminoglycoside | 12.5 | 12.5 | 25 | 50 | Antagonistic |
| Tobramycin | Aminoglycoside | 4.0 | 8.0 | 16 | 32 | Antagonistic |
2.3. Discussion
3. Experimental
3.1. Bacterial Strain and Antimicrobial Agents
3.2. Scanning Electron Microscopy (SEM)
3.3. Real-Time Monitoring of P. aeruginosa CCIN34519 Biofilm
4. Conclusions
Abbreviations
| CLSI | Clinical and Laboratory Standards Institute |
| MIC | Minimum Inhibitory Concentration |
| MPC | Mutant Prevention Concentration |
| NAUTICA | North American Urinary Tract Infection Collaborative Alliance |
| OTC | Over The Counter |
| P. aeruginosa | Pseudomonas aeruginosa |
| SEM | Scanning Electron Microscope |
| UTI | Urinary Tract Infection |
Acknowledgment
Author Contributions
Conflicts of Interest
References
- Parsek, M.R.; Singh, P.K. Bacterial biofilms: An emerging link to disease pathogenesis. Annu. Rev. Microbiol. 2003, 57, 677–701. [Google Scholar] [CrossRef]
- Drenkard, E. Antimicrobial resistance of Pseudomonas aeruginosa biofilms. Microbes Infect. 2003, 5, 1213–1219. [Google Scholar] [CrossRef]
- Bjarnsholt, T.; Jensen, P.; Fiandaca, M.J.; Pedersen, J.; Hansen, C.R.; Andersen, C.B.; Pressler, T.; Givskov, M.; Høiby, N. Pseudomonas aeruginosa biofilms in the respiratory tract of cystic fibrosis patients. Pediatr. Pulmonol. 2009, 44, 547–558. [Google Scholar] [CrossRef]
- Wagner, V.E.; Iglewski, B.H. Pseudomonas aeruginosa biofilms in CF Infection. Clin. Rev. Allergy Immunol. 2008, 35, 124–134. [Google Scholar] [CrossRef]
- Chang, S.L.; Shortliffe, L.D. Pediatric urinary tract infections. Pediatr. Clin. North. Am. 2006, 53, 379–400. [Google Scholar]
- Mittal, R.; Aggarwal, S.; Sharma, S.; Chhibber, S.; Harjai, K. Urinary tract infections caused by Pseudomonas aeruginosa. J. Infect. Public Health 2009, 2, 101–111. [Google Scholar] [CrossRef]
- Foxman, B. Epidemiology of urinary tract infections: Incidence, morbidity, and economic costs. Dis. Mon. 2003, 49, 53–70. [Google Scholar] [CrossRef]
- Litwin, M.S.; Saigal, C.S.; Yano, E.M.; Avila, C.; Geschwind, S.A.; Hanley, J.M. Urologic Diseases in America Project: Analytical methods and principal findings. J. Urol. 2005, 173, 933–937. [Google Scholar] [CrossRef]
- Ghafoor, A.; Hay, I.; Rehm, B. Role of exopolysaccharides in Pseudomonas aeruginosa biofilm formation and architecture. Appl. Environ. Microbiol. 2011, 77, 5238–5246. [Google Scholar] [CrossRef]
- Ryder, C.; Byrd, M.; Wozniak, D.J. Role of polysaccharides in Pseudomonas aeruginosa biofilm development. Curr. Opin. Microbiol. 2007, 10, 644–648. [Google Scholar] [CrossRef]
- Colvin, K.; Gordon, V.; Murakami, K. The Pel polysaccharide can serve a structural and protective role in the biofilm matrix of Pseudomonas aeruginosa. PLoS Pathog. 2011, 7, e1001264. [Google Scholar] [CrossRef]
- Williamson, K.S.; Richards, L.A.; Perez-Osorio, A.C.; Pitts, B.; McInnerney, K.; Stewart, P.S.; Franklin, M.J. Heterogeneity in Pseudomonas aeruginosa biofilms includes expression of ribosome hibernation factors in the antibiotic-tolerant subpopulation and hypoxia-induced stress response in the metabolically active population. J. Bacteriol. 2012, 194, 2062–2073. [Google Scholar] [CrossRef]
- Nordmann, P.; Naas, T.; Fortineau, N.; Poirel, L. Superbugs in the coming new decade: Multidrug resistance and prospects for treatment of Staphylococcus aureus, Enterococcus spp. and Pseudomonas aeruginosa in 2010. Curr. Opin. Microbiol. 2007, 1010, 436–440. [Google Scholar]
- Algun, U.; Arisoy, A.; Gunduz, T.; Ozbakkaloglu, B. The resistance of Pseudomonas aeruginosa strains to fluoroquinolone group of antibiotics. Indian J. Med. Microbiol. 2004, 22, 112–114. [Google Scholar]
- Henrichfreise, B.; Wiegand, I.; Pfister, W.; Wiedemann, B. Resistance mechanisms of multiresistant Pseudomonas aeruginosa strains from Germany and correlation with hypermutation. Antimicrob. Agents Chemother. 2007, 51, 4062–4070. [Google Scholar] [CrossRef]
- Huang, S.S.; Lee, S.C.; Lee, N.; See, L.C.; Tsai, M.H.; Shieh, W.B. Comparison of in vitro activities of levofloxacin, ciprofloxacin, ceftazidime, cefepime, imipenem, and piperacillintazobactam against aerobic bacterial pathogens from patients with nosocomial infections. J. Microbiol. Immunol. Infect. 2007, 40, 134–140. [Google Scholar]
- Rogues, A.M.; Dumartin, C.; Amadeo, B.; Venier, A.G.; Marty, N.; Parneix, P. Relationship between rates of antimicrobial consumption and the incidence of antimicrobial resistance in Staphylococcus aureus and Pseudomonas aeruginosa isolates from 47 French hospitals. Infect. Control Hosp. Epidemiol. 2007, 28, 1389–1395. [Google Scholar] [CrossRef]
- Obritsch, M.D.; Fish, D.N.; MacLaren, R.; Jung, R. Nosocomial infections due to multidrug-resistant Pseudomonas aeruginosa: Epidemiology and treatment options. Pharmacotherapy 2005, 25, 1353–1364. [Google Scholar] [CrossRef]
- Rice, L.B. Unmet medical needs in antibacterial therapy. Biochem. Pharmacol. 2006, 71, 991–995. [Google Scholar] [CrossRef]
- Wolter, D.J.; Lister, P.D. Mechanisms of β-lactam resistance among Pseudomonas aeruginosa. Curr. Pharm. Des. 2013, 19, 209–222. [Google Scholar]
- Anderson, G.; Kenney, T.; MacLeod, D. Eradication of Pseudomonas aeruginosa biofilms on cultured airway cells by a fosfomycin/tobramycin antibiotic combination. Pathog. Dis. 2013, 67, 39–45. [Google Scholar] [CrossRef]
- Tamma, P.; Cosgrove, S.; Maragakis, L. Combination therapy for treatment of infections with gram-negative bacteria. Clin. Microbiol. Rev. 2012, 25, 450–470. [Google Scholar] [CrossRef]
- Yu, Q.; Griffin, E.; Marquis, S. In vitro evaluation of tobramycin and aztreonam versus Pseudomonas aeruginosa biofilms on cystic fibrosis-derived human airway epithelial cells. J. Antimicrob. Chemother. 2012, 67, 2673–2681. [Google Scholar] [CrossRef]
- Andreini, C.; Banci, L.; Bertini, I.; Rosato, A. Zinc through the three domains of life. J. Proteome Res. 2006, 5, 3173–3178. [Google Scholar] [CrossRef]
- Ellison, M.L.; Farrow, J.M., III.; Parrish, W.; Danell, A.S.; Pesci, E.C. The transcriptional Regulator Np20 is the zinc uptake regulator in Pseudomonas aeruginosa. PLoS One 2013, 8, e75389. [Google Scholar]
- Sobke, A.; Klinger, M.; Hermann, B.; Sachse, S.; Nietzsche, S.; Makarewicz, O.; Keller, P.M.; Pfister, W.; Straube, E. The urinary antibiotic 5-nitro-8-hydroxyquinoline (Nitroxoline) reduces the formation and induces the dispersal of Pseudomonas aeruginosa biofilms by chelation of iron and zinc. Antimicrob. Agents Chemother. 2012, 56, 6021–6025. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Susceptibility Testing. Twenty-First Informational Supplement. CLSI document M100-S21 (ISBN 1-56238-742-1); CLSI: Wayne, Pennsylvania, USA, 2011. [Google Scholar]
- Alkawareek, M.; Algwari, Q.; Laverty, G. Eradication of Pseudomonas aeruginosa biofilms by atmospheric pressure non-thermal plasma. PLoS One 2012, 7, e44289. [Google Scholar] [CrossRef] [Green Version]
- Costerton, W.; Veeh, R.; Shirtliff, M.; Pasmore, M.; Post, C.; Ehrlich, G. The application of biofilm science to the study and control of chronic bacterial infections. J. Clin. Invest. 2003, 112, 1466–1477. [Google Scholar] [CrossRef]
- Fux, C.A.; Costerton, J.W.; Stewart, P.S.; Stoodley, P. Survival strategies of infectious biofilms. Trends Microbiol. 2005, 13, 34–40. [Google Scholar] [CrossRef]
- Mah, T.; Pitts, B.; Pellock, B.; Walker, G.C.; Stewart, P.S.; O’Toole, G.A. A genetic basis for Pseudomonas aeruginosa biofilm antibiotic resistance. Nature 2003, 426, 306–310. [Google Scholar] [CrossRef]
- Hall-Stoodley, L.; Costerton, J.W.; Stoodley, P. Bacterial biofilms: From the natural environment to infectious diseases. Nat. Rev. Microbiol. 2004, 2, 95–108. [Google Scholar] [CrossRef]
- Palmer, R.J.; Stoodley, P. Biofilms: Broadened horizons and new emphases. J. Bacteriol. 2007, 189, 7948–7960. [Google Scholar] [CrossRef]
- Lyczak, J.B.; Cannon, C.; Pier, G.B. Establishment of Pseudomonas aeruginosa infection: Lessons from a versatile opportunist. Microbes Infect. 2000, 2, 1051–1060. [Google Scholar] [CrossRef]
- Lu, Q.; Yu, J.; Yang, X. Ambroxol interferes with Pseudomonas aeruginosa quorum sensing. Int. J. Antimicrob. Agents 2010, 36, 211–215. [Google Scholar] [CrossRef]
- Moriarty, T.F.; Elborn, J.S.; Tunney, M.M. Effect of pH on the antimicrobial susceptibility of planktonic and biofilm-grown clinical Pseudomonas aeruginosa isolates. Br. J. Biomed. Sci. 2007, 64, 101–104. [Google Scholar]
- Walters, M.C.; Roe, F.; Bugnicourt, A.; Franklin, M.J.; Stewart, P.S. Contributions of antibiotic penetration, oxygen limitation, and low metabolic activity to tolerance of Pseudomonas aeruginosa biofilms to ciprofloxacin and tobramycin. Antimicrob. Agents Chemother. 2003, 47, 317–323. [Google Scholar] [CrossRef]
- Balaban, N.Q.; Merrin, J.; Chait, R.; Kowalik, L.; Leibler, S. Bacterial persistence as a phenotypic switch. Science 2004, 305, 1622–1625. [Google Scholar] [CrossRef]
- Gefen, O.; Balaban, N.Q. The importance of being persistent: Heterogeneity of bacterial populations under antibiotic stress. FEMS Microbiol. Rev. 2009, 33, 704–717. [Google Scholar] [CrossRef]
- Lewis, K. Persister cells, dormancy and infectious disease. Nat. Rev. Microbiol. 2007, 5, 48–56. [Google Scholar] [CrossRef]
- Mulcahy, L.R.; Burns, J.L.; Lory, S.; Lewis, K. Emergence of Pseudomonas aeruginosa strains producing high levels of persister cells in patients with cystic fibrosis. J. Bacteriol. 2010, 192, 6191–6199. [Google Scholar] [CrossRef]
- Kurosaka, Y.; Ishida, Y.; Yamamura, E.; Takase, H.; Otani, T.; Kumon, H. A non-surgical rat model of foreign body-associated urinary tract infection with Pseudomonas aeruginosa. Microbiol. Immunol. 2001, 45, 9–15. [Google Scholar] [CrossRef]
- Heydorn, A.; Ersbøll, B.; Kato, J. Statistical analysis of Pseudomonas aeruginosa biofilm development: Impact of mutations in genes involved in twitching motility, cell-to-cell signaling, and stationary-phase sigma factor expression. Appl. Environ. Microbiol. 2002, 68, 2008–2017. [Google Scholar] [CrossRef]
- Olmos, A.; Castillo, M.; Maiz, L. In vitro prevention of Pseudomonas aeruginosa early biofilm formation with antibiotics used in cystic fibrosis patients. Int. J. Antimicrob. Agent 2012, 40, 173–176. [Google Scholar] [CrossRef]
- Zhang, L.; Fritsch, M.; Hammond, L. Identification of genes involved in Pseudomonas aeruginosa biofilm-specific resistance to antibiotics. PLoS One 2013, 8, e61625. [Google Scholar] [CrossRef]
- Zhanel, G.G.; Hisanaga, T.L.; Laing, N.M.; DeCorby, M.R.; Nichol, K.A.; Palatnik, L.P.; Johnson, J.; Noreddin, A.; Harding, G.K.; Nicolle, L.E.; et al. Antibiotic resistance in outpatient urinary isolates: Final results from the North American Urinary Tract Infection Collaborative Alliance (NAUTICA). Int. J. Antimicrob. Agents 2005, 26, 380–388. [Google Scholar] [CrossRef]
- Pasquet, J.; Chevalier, Y.; Couval, E.; Bouvier, D.; Noizet, G.; Morlière, C.; Bolzinger, M.A. Antimicrobial activity of zinc oxide particles on five micro-organisms of the Challenge Tests related to their physicochemical properties. Int. J. Pharm. 2013, 460, 92–100. [Google Scholar]
- Ma, H.; Darmawan, E.T.; Zhang, M.; Zhang, L.; Bryers, J.D. Development of a poly(ether urethane) system for the controlled release of two novel anti-biofilm agents based on gallium or zinc and its efficacy to prevent bacterial biofilm formation. J. Control. Release 2013, 172, 1035–1044. [Google Scholar] [CrossRef]
- Rossolini, G.M.; Mantengoli, E. Treatment and control of severe infections caused by multiresistant Pseudomonas aeruginosa. Clin. Microbiol. Infect. 2005, 11, 17–32. [Google Scholar] [CrossRef]
- Adi, H.; Young, P.; Chan, H. Co-spray-dried mannitol-ciprofloxacin dry powder inhaler formulation for cystic fibrosis and chronic obstructive pulmonary disease. Eur. J. Pharm. Sci. 2010, 40, 239–247. [Google Scholar] [CrossRef]
- Karlowsky, J.A.; Adam, H.J.; Desjardins, M.; Lagacé-Wiens, P.R.; Hoban, D.J.; Zhanel, G.G. Canadian Antimicrobial Resistance Alliance. Changes in fluoroquinolone resistance over 5 years (CANWARD 2007-11) in bacterial pathogens isolated in Canadian hospitals. J. Antimicrob. Chemother. 2013, 68, 39–46. [Google Scholar] [CrossRef]
- Bruchmann, S.; Dötsch, A.; Nouri, B.; Chaberny, I.; Häussler, S. Quantitative contributions of target alteration and decreased drug accumulation to Pseudomonas aeruginosa fluoroquinolone resistance. Antimicrob. Agents Chemother. 2013, 57, 1361–1368. [Google Scholar] [CrossRef]
- Hansen, G.T.; Blondeau, J.M. Comparison of the minimum inhibitory, mutant prevention and minimum bactericidal concentrations of ciprofloxacin, levofloxacin and garenoxacin against enteric Gram-negative urinary tract infection pathogens. J. Chemother. 2005, 17, 484–492. [Google Scholar] [CrossRef]
- DeRyke, C.A.; Lee, S.Y.; Kuti, J.L.; Nicolau, D.P. Optimising dosing strategies of antibacterials utilizing pharmacodynamic principles: Impact on the development of resistance. Drugs 2006, 66, 1–14. [Google Scholar]
- Hansen, G.T.; Zhao, X.; Drlica, K.; Blondeau, J.M. Mutant prevention concentration for ciprofloxacin and levofloxacin with Pseudomonas aeruginosa. Int. J. Antimicrob. Agents 2006, 27, 120–124. [Google Scholar] [CrossRef]
- Ruiz, J.P.; Quero, Q.J. Pharmacodynamic and pharmacokinetic evaluation of respiratory fluoroquinolones. Guideline to selection of the most appropriate fluoroquinolone. Rev. Esp. Quimioter. 2012, 25, 245–251. [Google Scholar]
- Mao, W.; Warren, M.S.; Lee, A.; Mistry, A.; Lomovskaya, O. MexXY-OprM efflux pump is required for antagonism of aminoglycosides by divalent cations in Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2001, 45, 2001–2007. [Google Scholar] [CrossRef]
- Murphy, T.A.; Catto, L.E.; Halford, S.E.; Hadfield, A.T.; Minor, W.; Walsh, T.R.; Spencer, J. Crystal structure of Pseudomonas aeruginosa SPM-1 provides insights into variable zinc affinity of metallo-beta-lactamases. J. Mol. Biol. 2006, 357, 890–903. [Google Scholar] [CrossRef]
- Wen, J.; Zhao, K.; Gu, T.; Raad, I. A green biocide enhancer for the treatment of sulfate-reducing bacteria (SRB) biofilms on carbon steel surfaces using glutaraldehyde. Int. Biodeterior. Biodegrad. 2009, 63, 1102–1106. [Google Scholar]
- Elkhatib, W.F.; Haynes, V.L.; Noreddin, A.M. Microbiological appraisal of levofloxacin activity against Pseudomonas aeruginosa biofilm in combination with different calcium channel blockers in vitro. J. Chemother. 2009, 21, 135–143. [Google Scholar] [CrossRef]
- Smith, H.J.; Nichol, K.A.; Hoban, D.J.; Zhanel, G.G. Stretching the mutant prevention concentration (MPC) beyond its limits. J. Antimicrob. Chemother. 2003, 51, 1323–1325. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Elkhatib, W.; Noreddin, A. In Vitro Antibiofilm Efficacies of Different Antibiotic Combinations with Zinc Sulfate against Pseudomonas aeruginosa Recovered from Hospitalized Patients with Urinary Tract Infection. Antibiotics 2014, 3, 64-84. https://doi.org/10.3390/antibiotics3010064
Elkhatib W, Noreddin A. In Vitro Antibiofilm Efficacies of Different Antibiotic Combinations with Zinc Sulfate against Pseudomonas aeruginosa Recovered from Hospitalized Patients with Urinary Tract Infection. Antibiotics. 2014; 3(1):64-84. https://doi.org/10.3390/antibiotics3010064
Chicago/Turabian StyleElkhatib, Walid, and Ayman Noreddin. 2014. "In Vitro Antibiofilm Efficacies of Different Antibiotic Combinations with Zinc Sulfate against Pseudomonas aeruginosa Recovered from Hospitalized Patients with Urinary Tract Infection" Antibiotics 3, no. 1: 64-84. https://doi.org/10.3390/antibiotics3010064




