Water-Soluble Electrospun Nanofibers as a Method for On-Chip Reagent Storage
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
2.2. Electrospun Nanofiber Preparation
2.3. Scanning Electronic Microscope (SEM)
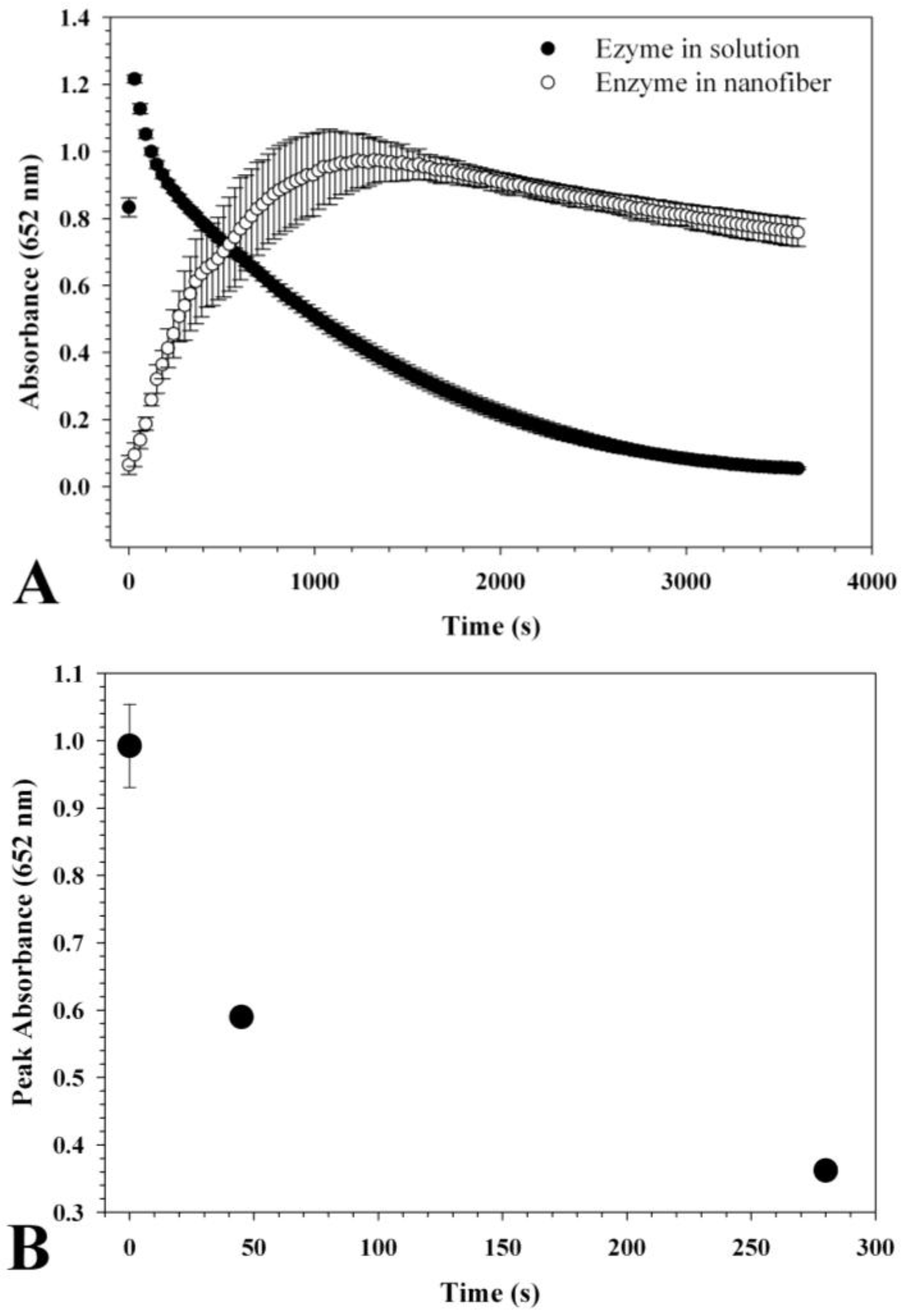
2.4. HRP Activity Measurement
2.5. On-Chip Microfluidic Devices with Nanofibers
3. Results and Discussion
3.1. Morphology of Nanofibers

3.2. HRP Enzyme Activity
3.3. On-Chip Microfluidic System

4. Conclusions
Acknowledgments
References
- Haeberle, S.; Zengerle, R. Microfluidic platforms for lab-on-a-chip applications RID B-2451-2012. Lab on a Chip 2007, 7, 1094–1110. [Google Scholar] [CrossRef]
- Nugen, S.R.; Asiello, P.J.; Connelly, J.T.; Baeumner, A.J. PMMA biosensor for nucleic acids with integrated mixer and electrochemical detection. Biosens. Bioelectron. 2009, 24, 2428–2433. [Google Scholar] [CrossRef]
- Zhou, P.; Young, L.; Chen, Z. Weak solvent based chip lamination and characterization of on-chip valve and pump. Biomed. Microdevices 2010, 12, 821–832. [Google Scholar] [CrossRef]
- Reneker, D.; Smith, D. Naonfiber Structures for Supporting Biological Materials. WO/2006/133118; PCT/US2006/021785. 2006. Available online: http://patentscope.wipo.int/search/en/WO2006133118 (accessed on 15 August 2012).
- Andrady, A. Science and Technology of Polymer Nanofibers; Wiley-Interscience: Hoboken, NJ, USA, 2008. [Google Scholar]
- Wang, Z.; Wan, L.; Liu, Z.; Huang, X.; Xu, Z. Enzyme immobilization on electrospun polymer nanofibers: An overview RID A-7659-2010 RID A-5635-2008. J. Molecul. Catal. B Enzym. 2009, 56, 189–195. [Google Scholar] [CrossRef]
- Gorji, M.; Jeddi, A.A.A.; Gharehaghaji, A.A. Fabrication and characterization of polyurethane electrospun nanofiber membranes for protective clothing applications. J. Appl. Polym. Sci. 2012, 125, 4135–4141. [Google Scholar] [CrossRef]
- Kanafchian, M.; Valizadeh, M.; Haghi, A.K. Fabrication of nanostructured and multicompartmental fabrics based on electrospun nanofibers. Korean J. Chem. Eng. 2011, 28, 763–769. [Google Scholar] [CrossRef]
- Feng, C.; Khulbe, K.C.; Matsuura, I.; Gopal, R.; Kaur, S.; Rarnakrishna, S.; Khayet, A. Production of drinking water from saline water by air-gap membrane distillation using polyvinylidene fluoride nanofiber membrane. J. Membr. Sci. 2008, 311, 1–6. [Google Scholar] [CrossRef]
- Prince, J.A.; Singh, G.; Rana, D.; Matsuura, T.; Anbharasi, V.; Shanmugasundaram, T.S. Preparation and characterization of highly hydrophobic poly(vinylidene fluoride)—Clay nanocomposite nanofiber membranes (PVDF-clay NNMs) for desalination using direct contact membrane distillation. J. Membr. Sci. 2012, 397, 80–86. [Google Scholar] [CrossRef]
- Gouma, P. Electrospun Enzyme-Nanocompositebiosensing Material. U.S. Patent 2006/0134716 A1, June 2006. [Google Scholar]
- Stasiak, M.; Studer, A.; Greiner, A.; Wendorff, J.H. Polymer fibers as carriers for homogeneous catalysts. Chem. Eur. J. 2007, 13, 6150–6156. [Google Scholar] [CrossRef]
- Luu, Y.; Kim, K.; Hsiao, B.; Chu, B.; Hadjiargyrou, M. Development of a nanostructured DNA delivery scaffold via electrospinning of PLGA and PLA-PEG block copolymers. J. Control. Release 2003, 89, 341–353. [Google Scholar] [CrossRef]
- Huang, X.; Ge, D.; Xu, Z. Preparation and characterization of stable chitosan nanofibrous membrane for lipase immobilization RID A-2073-2010 RID B-1462-2009. Eur. Polym. J. 2007, 43, 3710–3718. [Google Scholar] [CrossRef]
- Yu, D.-G.; Nie, W.; Zhu, L.M.; Branford-White, C. Fast dissolution nanofiber membrane of ferulic acid prepared using electrospinning. In Proceedings of 2010 4th International Conference on Bioinformatics and Biomedical Engineering (iCBBE), Chengdu, China, 18-20 June 2010; pp. 1–4.
- Haaf, F.; Sanner, A.; Straub, F. Polymers of N-Vinylpyrrolidone—Synthesis, Characterization and Uses. Polym. J. 1985, 17, 143–152. [Google Scholar] [CrossRef]
- Salalha, W.; Kuhn, J.; Dror, Y.; Zussman, E. Encapsulation of bacteria and viruses in electrospunnanofibres. Nanotechnology 2006, 17, 4675–4681. [Google Scholar] [CrossRef]
- Volpe, G.; Compagnone, D.; Draisci, R.; Palleschi, G. 3,3',5,5'-tetramethylbenzidine as electrochemical substrate for horseradish peroxidase based enzyme immunoassays. A comparative study. Analyst 1998, 123, 1303–1307. [Google Scholar] [CrossRef]
- Nugen, S.R.; Asiello, P.J.; Baeumner, A.J. Design and fabrication of a microfluidic device for near-single cell mRNA isolation using a copper hot embossing master. Microsyst. Technol. 2008, 15, 477–483. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Dai, M.; Jin, S.; Nugen, S.R. Water-Soluble Electrospun Nanofibers as a Method for On-Chip Reagent Storage. Biosensors 2012, 2, 388-395. https://doi.org/10.3390/bios2040388
Dai M, Jin S, Nugen SR. Water-Soluble Electrospun Nanofibers as a Method for On-Chip Reagent Storage. Biosensors. 2012; 2(4):388-395. https://doi.org/10.3390/bios2040388
Chicago/Turabian StyleDai, Minhui, Shengquan Jin, and Sam R. Nugen. 2012. "Water-Soluble Electrospun Nanofibers as a Method for On-Chip Reagent Storage" Biosensors 2, no. 4: 388-395. https://doi.org/10.3390/bios2040388



