Water Diffusion through a Titanium Dioxide/Poly(Carbonate Urethane) Nanocomposite for Protecting Cultural Heritage: Interactions and Viscoelastic Behavior
Abstract
:1. Introduction
2. Results
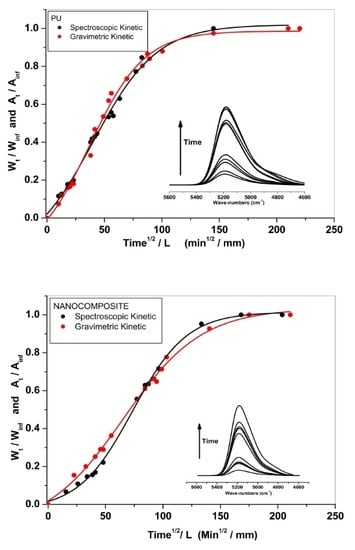
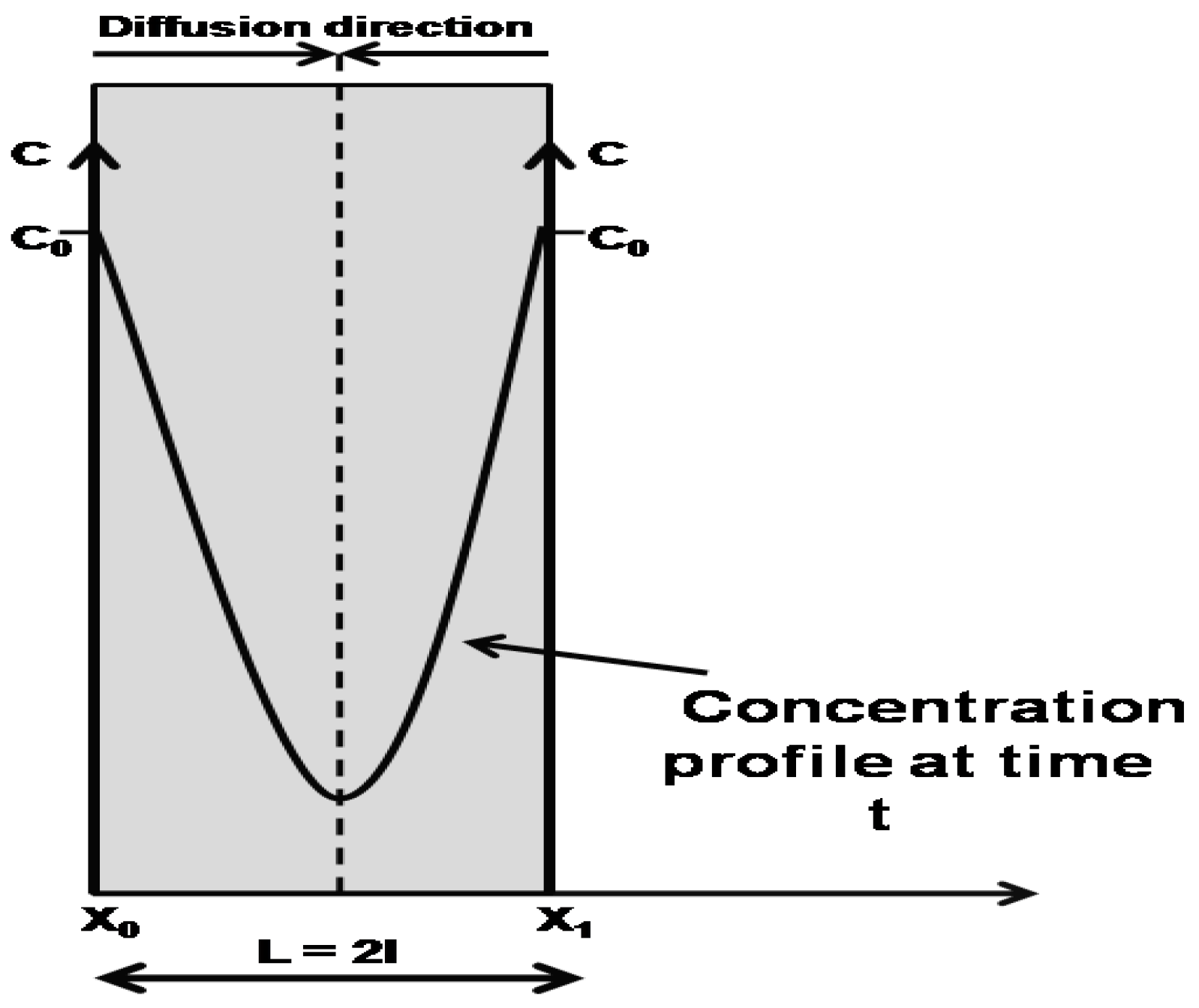
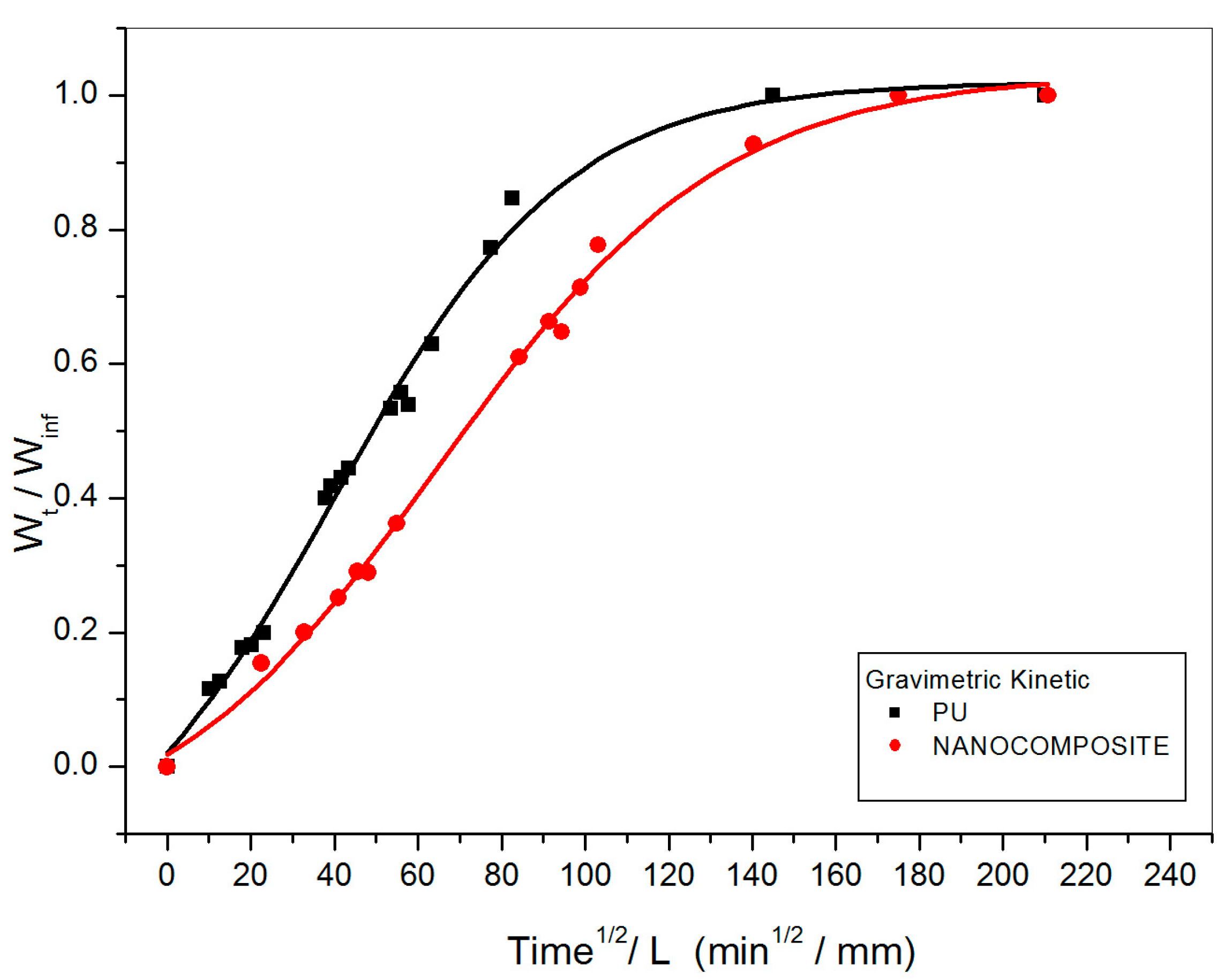
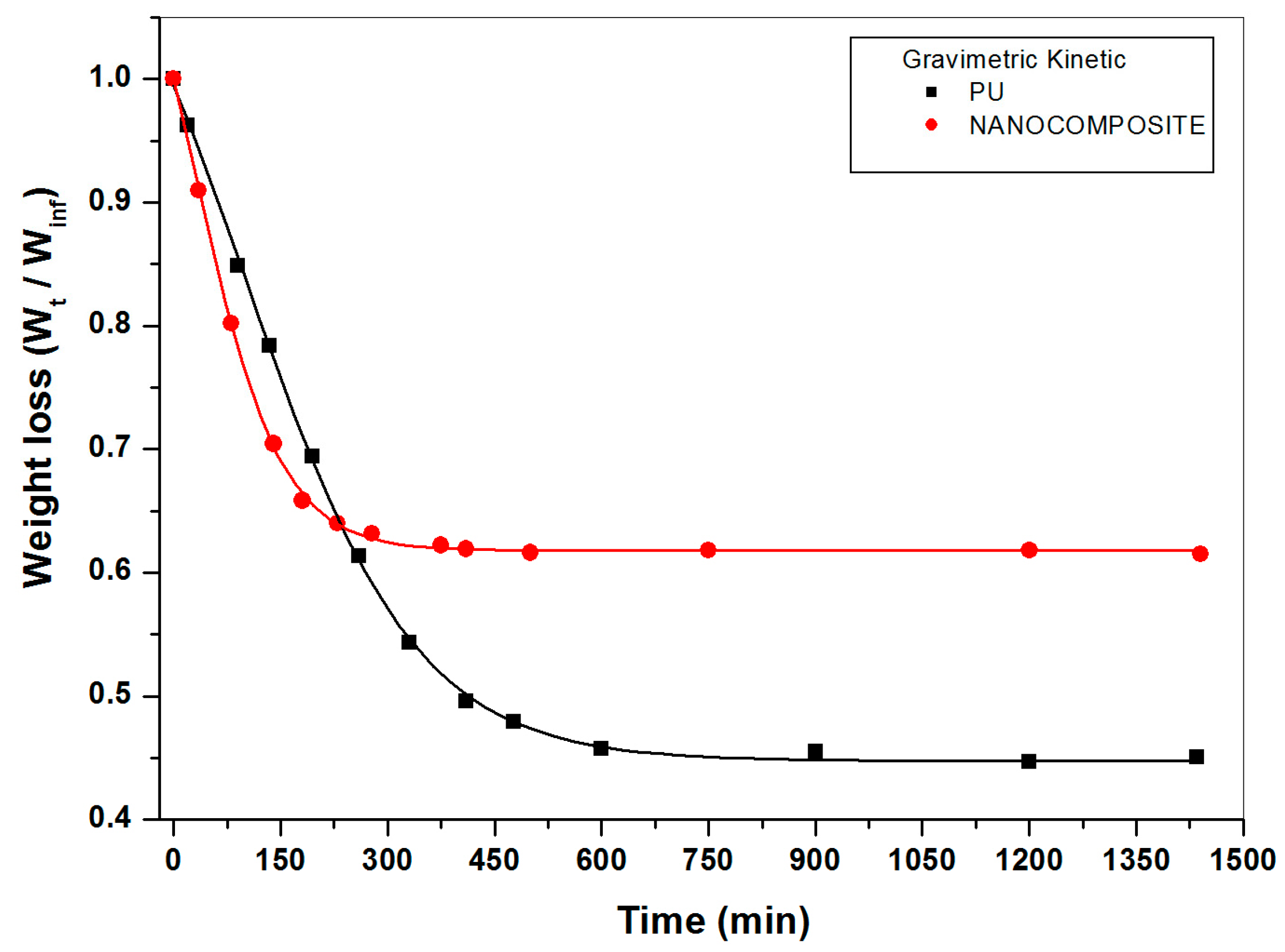
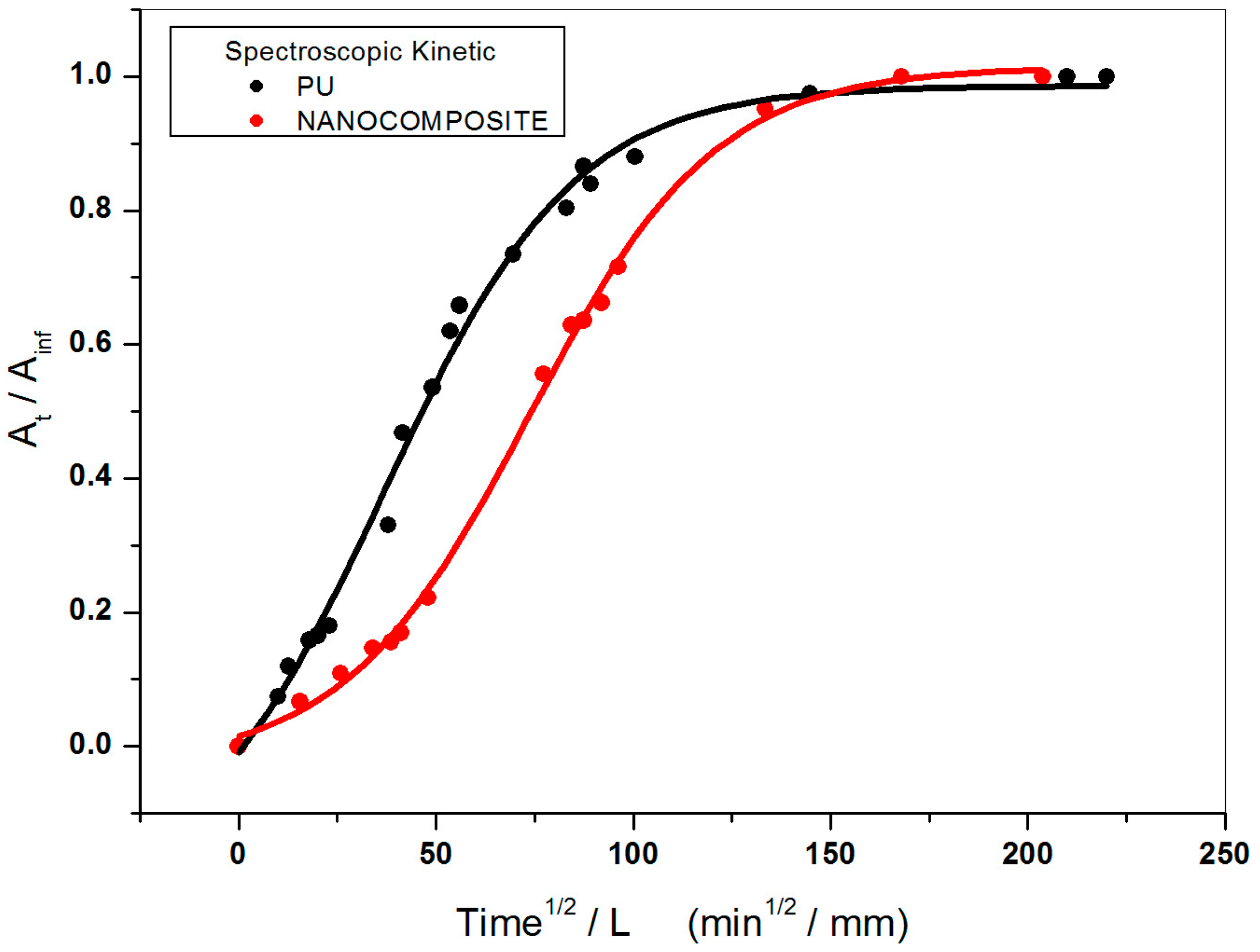
2.1. Gravimetric Measurements
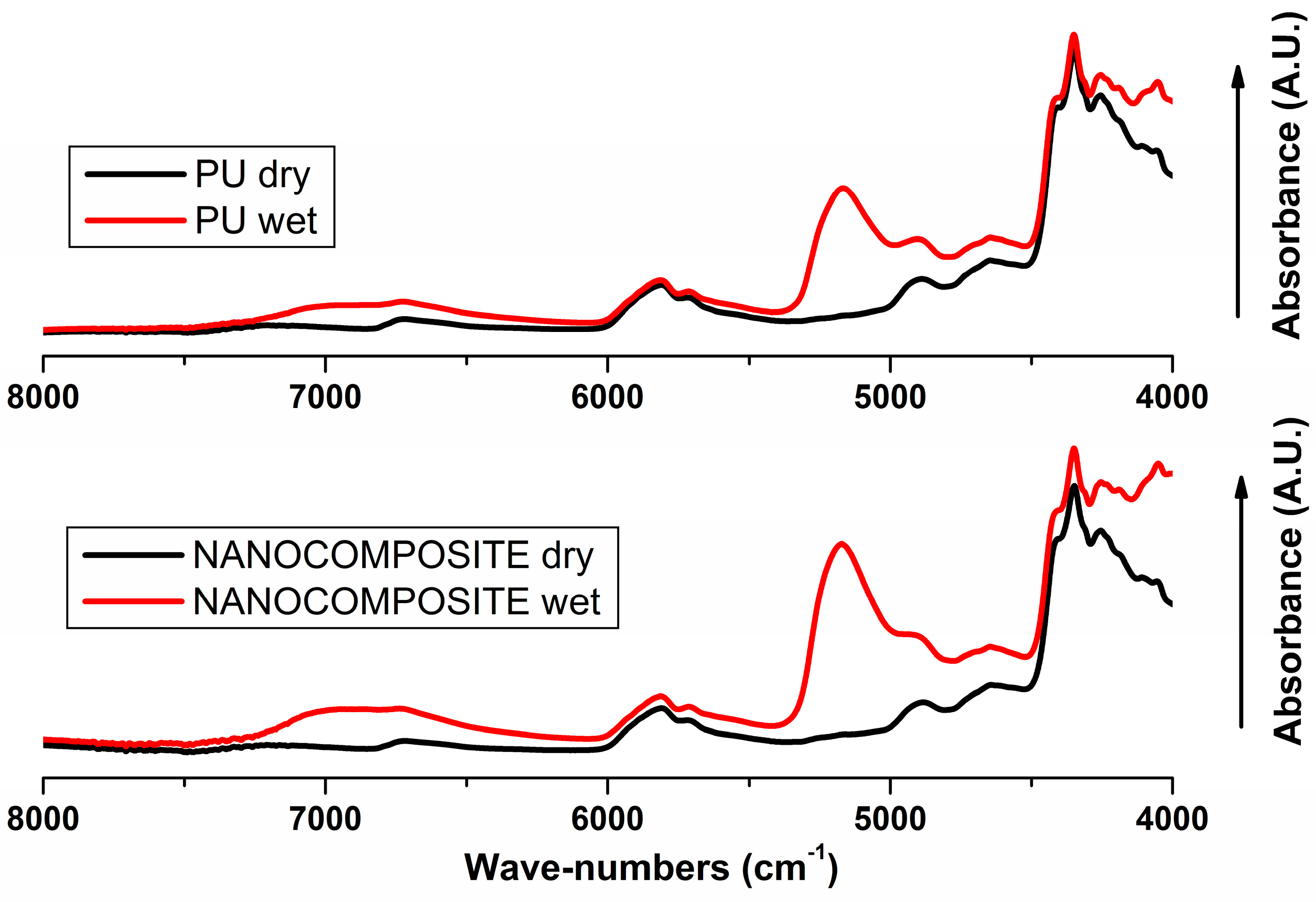
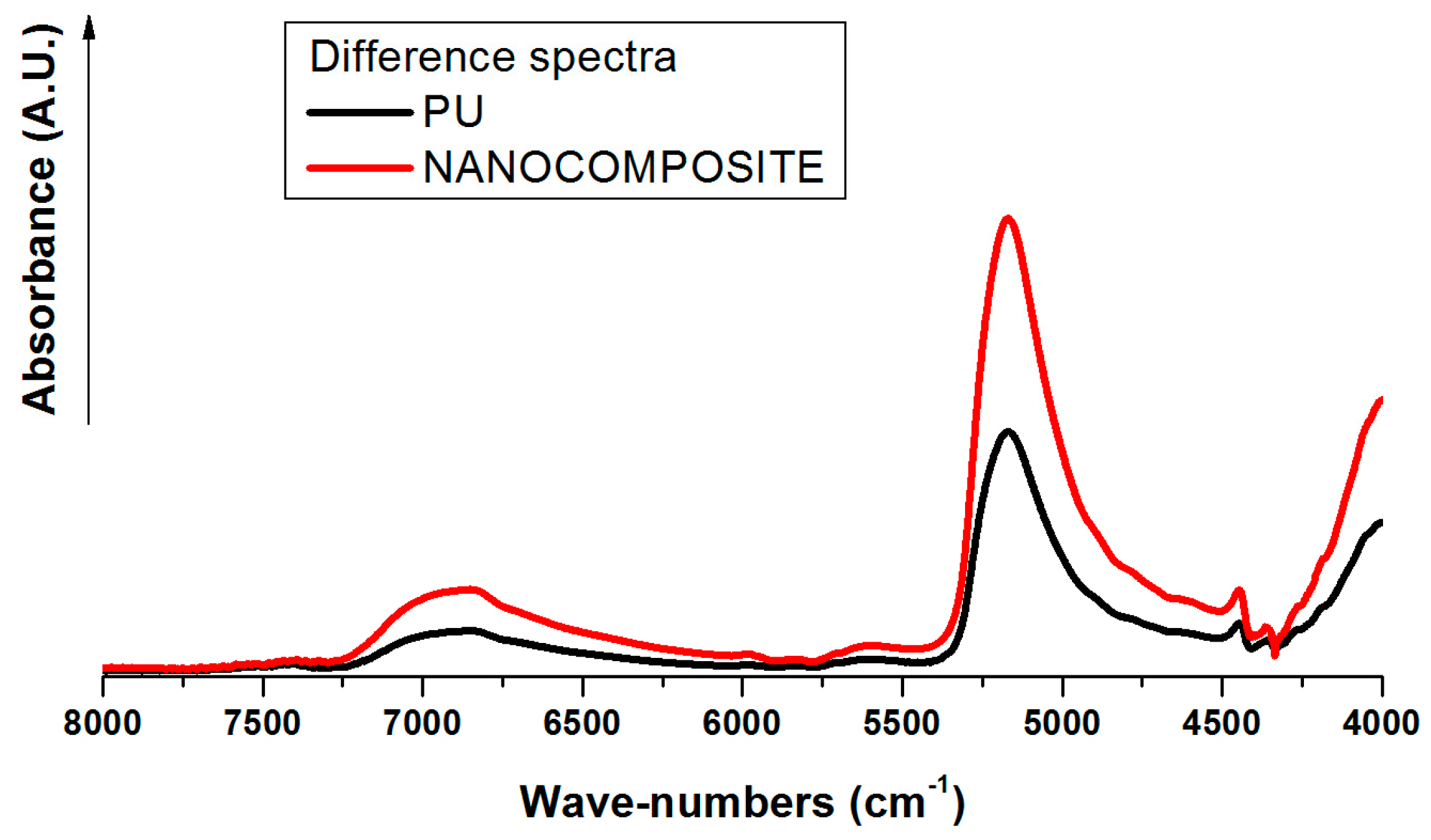
2.2. FT-NIR Measurements
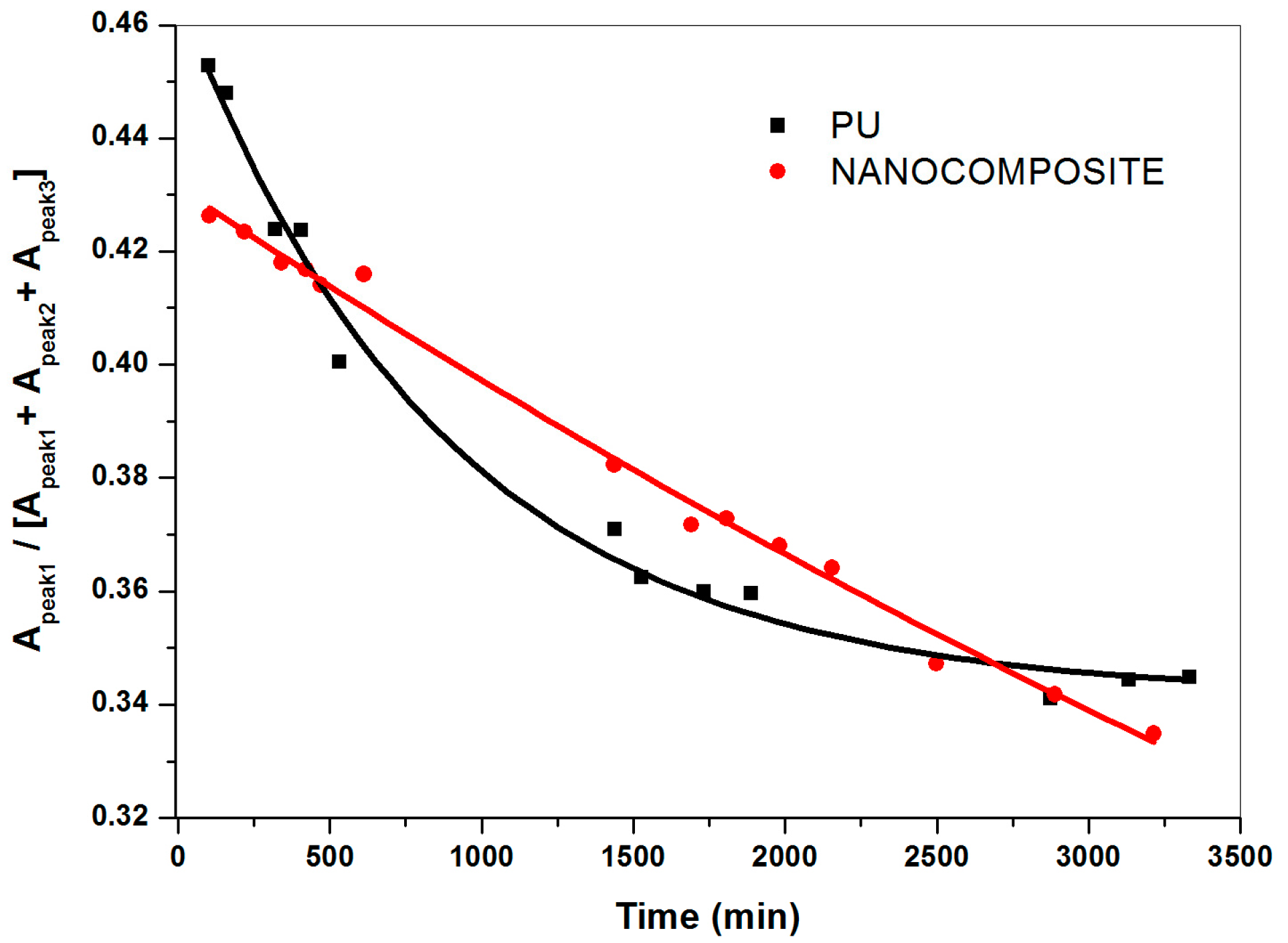
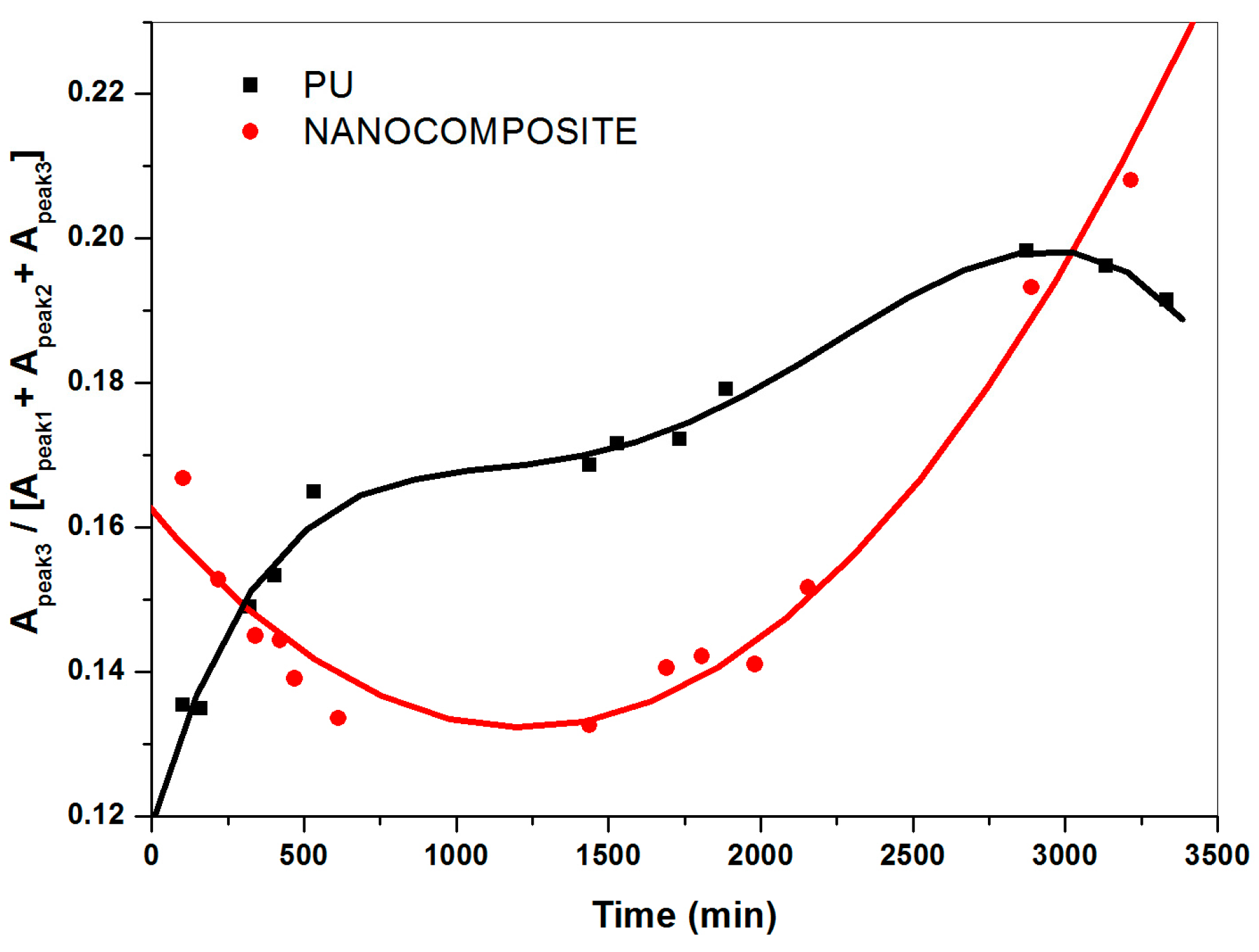
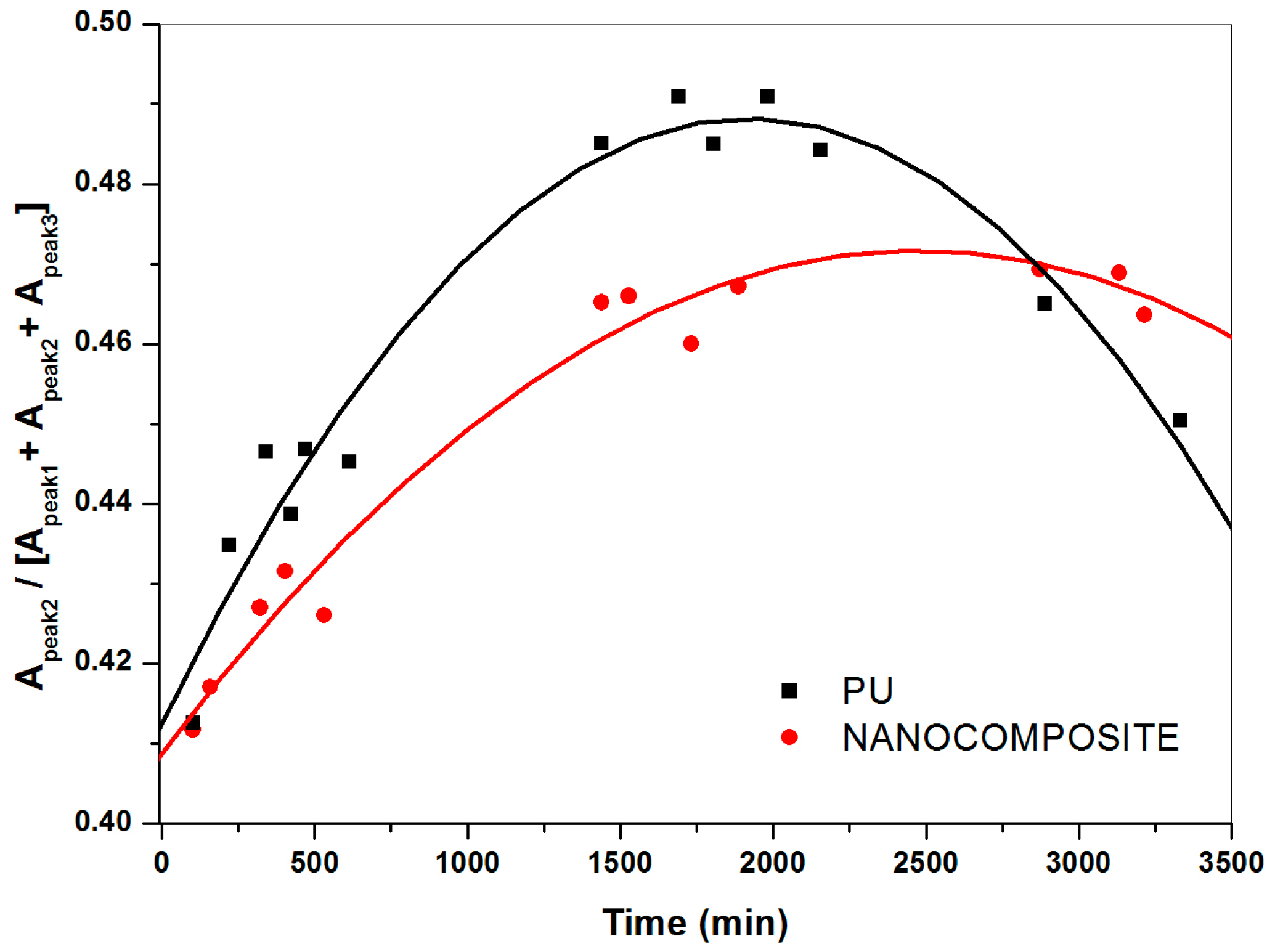
2.3. FT-NIR Curve-Fitting Analysis in the 5400–4600 cm−1 Wave-Number Range
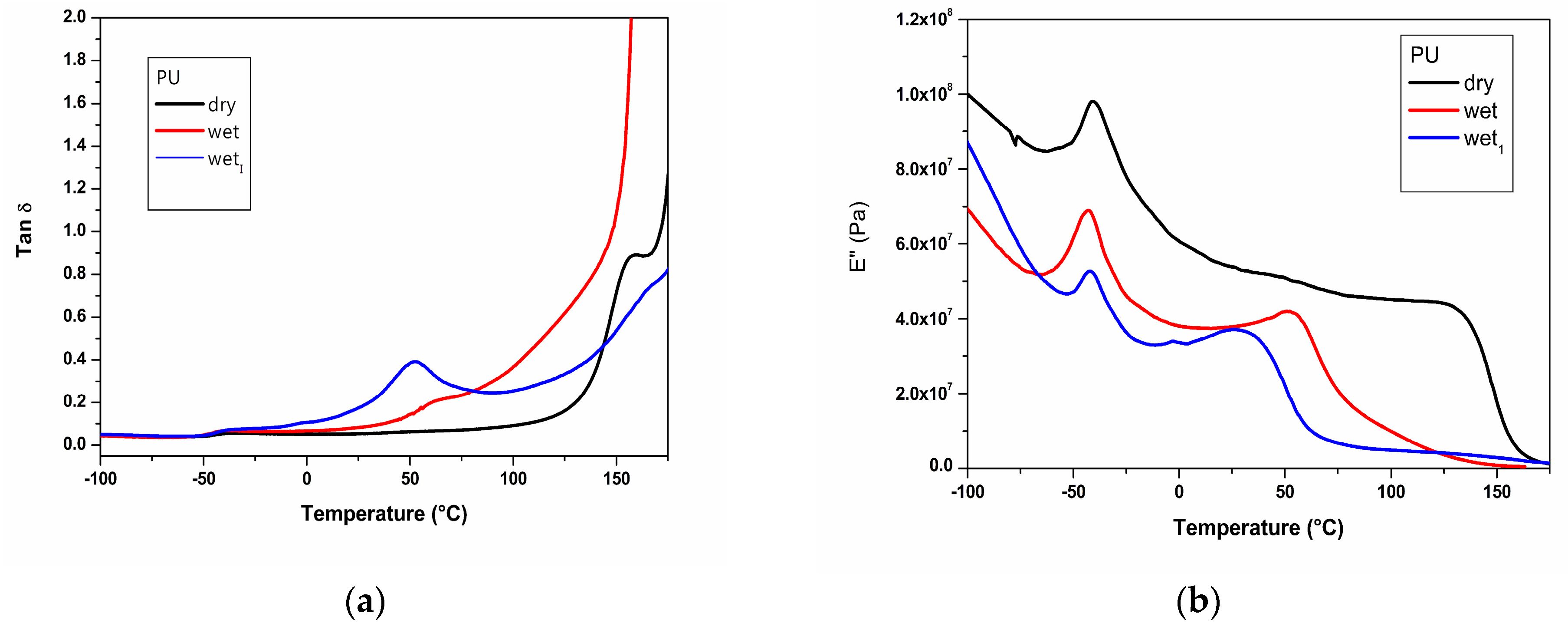
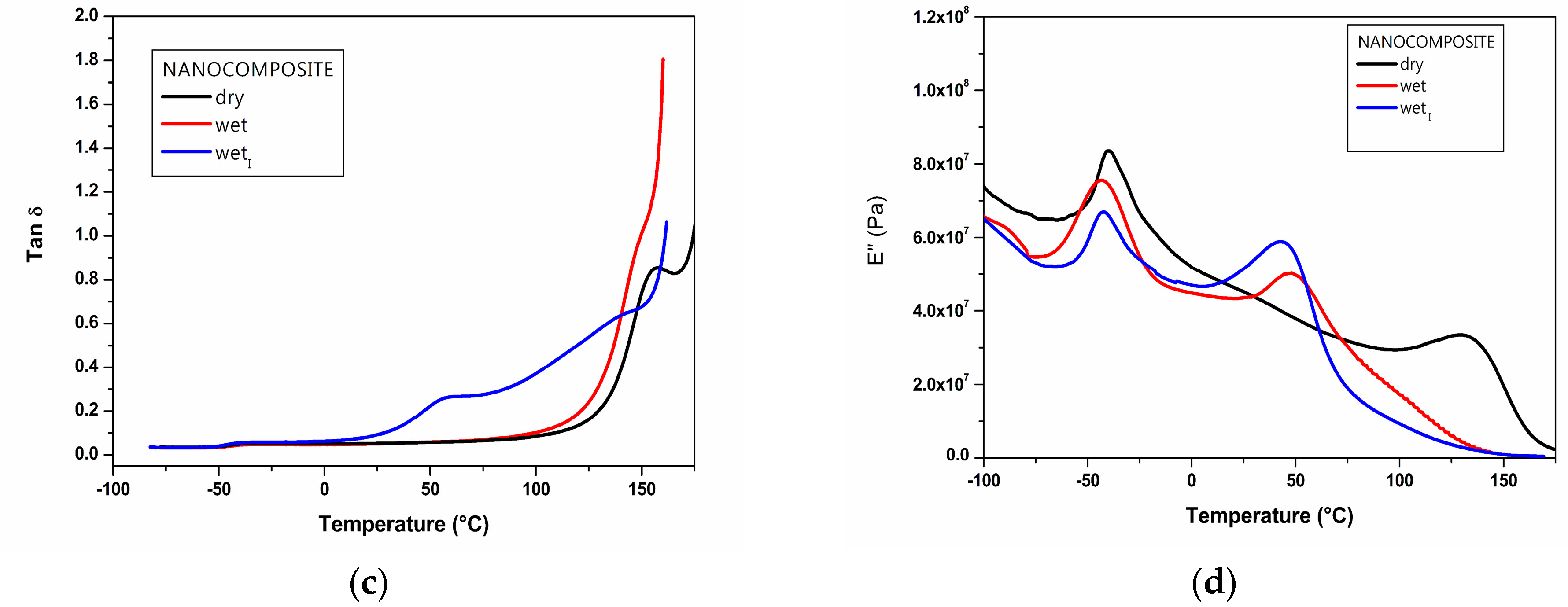
2.4. DMTA Analysis
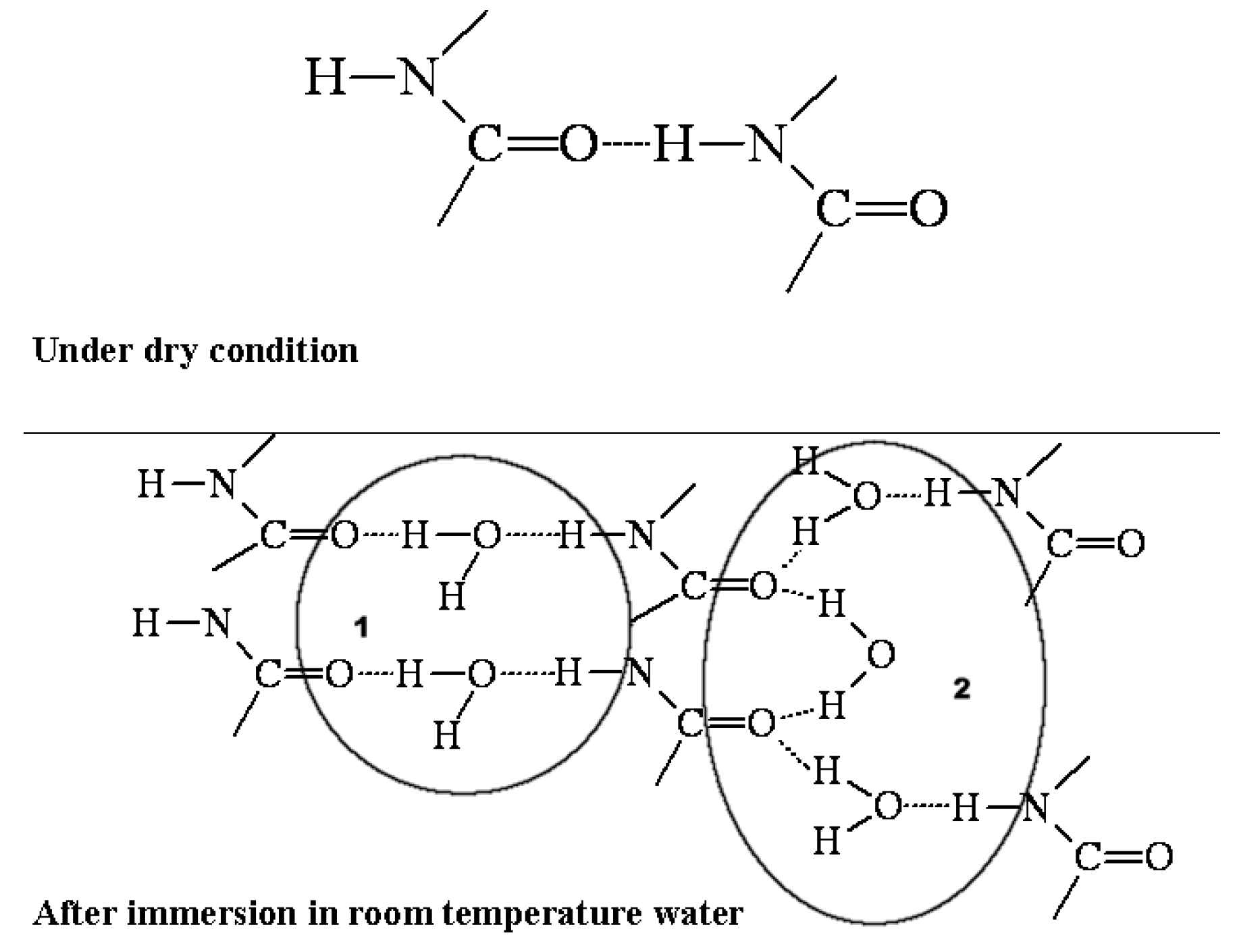
3. Discussion
4. Materials and Methods
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Brimblecombe, P. Urban Pollution and Changes to Materials and Building Surfaces; Imperial College Press: London, UK, 2016. [Google Scholar]
- Dornieden, T.H.; Gorbushina, A.A.; Krumbein, W.E. Biodecay of cultural heritage as a space/time-related ecological situation—An evaluation of a series of studies. Int. Biodeterior. Biodegrad. 2000, 46, 261–270. [Google Scholar] [CrossRef]
- Sabbioni, C.; Brimblecombe, P.; Cassar, M. The Atlas of Climate Change Impact on European Culture Heritage; Anthem Press: London, UK, 2010. [Google Scholar]
- Lazzarini, L.; Tabasso, M.L. Il Restauro Della Pietra; CEDAM: Obernai, France, 1986. [Google Scholar]
- UNESCO. Synthetic materials used in the conservation of cultural material. In The Conservation of the Cultural Property. Museum and Monuments 11; UNESCO: Paris, France, 1968; pp. 303–331. [Google Scholar]
- D’Orazio, L.; Gentile, G.; Mancarella, C.; Martuscelli, E.; Massa, V. Water-dispersed polymers for the conservation and restoration of cultural heritage: A molecular, thermal, structural and mechanical characterisation. Polym. Test. 2001, 20, 227–240. [Google Scholar] [CrossRef]
- Cocca, M.; D’Arienzo, L.; D’Orazio, L.; Gentile, G.; Martuscelli, E. Polyacrylates for conservation: Chemico-physical properties and durability of different commercial products. Polym. Test. 2004, 23, 333–342. [Google Scholar] [CrossRef]
- Alessandrini, G.; Aglietto, M.; Castelvetro, V.; Ciardelli, F.; Peruzzi, R.; Toniolo, L. Comparative evaluation of fluorinated and unfluorinated acrylic copolymers as water-repellent coating materials for stone. J. Appl. Polym. Sci. 2000, 76, 962–977. [Google Scholar] [CrossRef]
- ICR-CNR. Normal Protocol 20/85. Conservation Works: Planning, Execution and Preventive Evaluation; ICR-CNR: Rome, Italy, 1985. [Google Scholar]
- Paul, D.R.; Robeson, L.M. Polymer nanotechnology: Nanocomposites. Polymer 2008, 49, 3187–3204. [Google Scholar] [CrossRef]
- D’Orazio, L.; Grippo, A. A water dispersed Titanium dioxide/poly (carbonate urethane) nanocomposite for protecting cultural heritage: Preparation and properties. Prog. Org. Coat. 2015, 79, 1–7. [Google Scholar] [CrossRef]
- D’Orazio, L.; Grippo, A. A water dispersed Titanium dioxide/poly (carbonate urethane) nanocomposite for protecting cultural heritage: Eco-sustainable treatments on Neapolitan Yellow Tuff. Prog. Org. Coat. 2016, 99, 412–419. [Google Scholar] [CrossRef]
- National Researh Council (CNR)—Istituto Centrale del Restauro (ICR) Recommendations. AA. VV.: NORMAL 11/85 Water Absorption by Capillarity; National Researh Council (CNR)—Istituto Centrale del Restauro (ICR) Recommendations: Rome, Italy, 1985. [Google Scholar]
- Pimentel, G.C.; McClellan, A.L. The Hydrogen Bond; Freeman: San Francisco, CA, USA, 1960. [Google Scholar]
- Crank, J.; Park, G.S. Diffusion in Polymers; Academic Press: London, UK; New York, NY, USA, 1968. [Google Scholar]
- Comyn, J. Polymer Permeability; Elsevier Applied Science Publishers: London, UK, 1985. [Google Scholar]
- Crank, J. The Mathematics of Diffusion, 2nd ed.; Oxford University Press: Oxford, UK, 1975. [Google Scholar]
- Liu, W.; Hoe, S.V.; Pugh, M. Water uptake of epoxy clay nanocomposites: Model development. Compos. Sci. Technol. 2008, 68, 156–163. [Google Scholar] [CrossRef]
- Shen, C.H.; Springer, G.S. Moisture Absorption and Desorption of Composite Materials. J. Comp. Mater. 1976, 10, 2. [Google Scholar] [CrossRef]
- Vergnaud, J.M. Liquid Transport Processes in Polymeric Materials: Modeling and Industrial Applications; Prentice Hall: London, UK, 1991. [Google Scholar]
- Jacobs, P.M.; Jones, F.R. Diffusion of moisture into two-phase polymers. J. Mater. Sci. 1989, 24, 2331–2336. [Google Scholar] [CrossRef]
- Maggana, C.; Pissis, P. Water sorption and diffusion studies in an epoxy resin system. J. Polym. Sci. Part B 1999, 37, 1165–1182. [Google Scholar] [CrossRef]
- Segtan, V.H.; Šašic, Š.; Isaksson, T.; Ozaki, Y. Studies on the structure of water using two-dimensional Near-Infrared correlation spectroscopy and principal component analysis. Anal. Chem. 2001, 73, 3153–3161. [Google Scholar] [CrossRef]
- Musto, P.; Mensitieri, G.; Lavorgna, M.; Scarinzi, G.; Scherillo, G. Combining gravimetric and vibrational spectroscopy measurements to quantify first and second-shell hydration layers of polyimides with different molecular architectures. J. Phys. Chem. B 2012, 116, 1209–1220. [Google Scholar] [CrossRef] [PubMed]
- Cotugno, S.; Larobina, D.; Mensitieri, G.; Musto, P.; Ragosta, G. A novel spectroscopic approach to investigate transport processes in polymers: The case water-epoxy system. Polymer 2001, 42, 6431–6438. [Google Scholar] [CrossRef]
- Musto, P.; Abbate, M.; Lavorgna, M.; Ragosta, G.; Scarinzi, G. Microstructural features, diffusion and molecular relaxations in polyimide/silica hybrids. Polymer 2006, 47, 6172–6186. [Google Scholar] [CrossRef]
- Dıez-Pascual, A.M.; Diez-Vicente, A.L. Poly (3-hydroxybutyrate)/ZnO bionanocomposites with improved mechanical, barrier and antibacterial properties. Int. J. Mol. Sci. 2014, 15, 10950–10973. [Google Scholar] [CrossRef] [PubMed]
- Tien, Y.I.; Wie, K.H. Hydrogen bonding and mechanical properties of segmented montmorillonite/polyurethane nanocomposite with different hard segment ratios. Polymer 2001, 42, 3213–3221. [Google Scholar] [CrossRef]
- Fujishima, A.; Rao, T.N.; Tryk, D.A. Titanium dioxide photocatalysis. J. Photochem. Photobiol. C 2000, 1, 1–21. [Google Scholar] [CrossRef]
- Fahmi, A.; Minot, C. A theoretical investigation of water adsorption on Titanium dioxide surfaces. Surf. Sci. 1994, 304, 343–359. [Google Scholar] [CrossRef]
- Boehm, H.P.; Herrmann, Z. Über die chemie der oberfläche des Titandioxids. I. Bestimmung des aktiven wasserstoffs, thermische entwässerung und rehydroxy lierung. Anorg. Allg. Chem. 1967, 352, 156–167. (In German) [Google Scholar] [CrossRef]
- Lo, J.W.; Chung, Y.W.; Somorjai, G.A. Electron spectroscopy studies of the chemisorption of O2, H2 and H2O on the TiO2 (100) surfaces with varied stoichiometry: Evidence for the photogeneration of Ti+3 and for its importance in chemisorption. Surf. Sci. 1978, 71, 199–219. [Google Scholar]
- Suda, Y.; Morimoto, T. Molecularly adsorbed water on the bare surfaces of TiO2. Langmuir 1987, 3, 786–788. [Google Scholar] [CrossRef]
- Sun, C.H.; Liu, L.M.; Selloni, A.; Lu, G.Q.; Smith, S.C. Titania-water interactions: A review of theoretical studies. J. Mater.Chem. 2010, 20, 9559–9612. [Google Scholar] [CrossRef]
- Raju, M.; Kim, S.Y.; van Duin, A.C.T.; Fichthorn, K.A. ReaxFF Reactive Force Field study of the dissociation of water on Titania surfaces. J. Phys. Chem. C 2013, 117, 10558–10572. [Google Scholar] [CrossRef]
- Zhang, H.; Banfield, J.F. Structural characteristics and mechanical and thermodynamic properties of nanocrystalline TiO2. Chem. Rev. 2014, 114, 9613–9644. [Google Scholar] [CrossRef] [PubMed]
- Henderson, M.A. The interaction of water with solid surfaces: Fundamental aspects revisited. Surf. Sci. Rep. 2002, 46, 1–308. [Google Scholar] [CrossRef]
- Cocca, M.; D’Orazio, L. Novel silver/polyurethane nanocomposite by in situ reduction: Effects of the silver nanoparticles on phase and viscoelastic behavior. J. Polym. Sci. Part B 2008, 46, 344–350. [Google Scholar] [CrossRef]
- Thompson, T.L.; Yates, J.T. Surface science studies on the photoactivation of TiO2: New photochemical processes. Chem. Rev. 2006, 106, 4428–4453. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Nakajima, A.; Wang, R.; Minabe, M.; Koizumi, S.; Fujishima, A.; Hashimoto, K. Photocatalytic activity and photoinduced hydrophilicity of titanium dioxide coated glass. Thin Solid Films 1999, 351, 260–263. [Google Scholar] [CrossRef]
- Xu, G.; Gryte, C.C.; Nowick, A.S.; Li, S.Z.; Pak, Y.S.; Greenbaum, S.G. Dielectric relaxation and deuteron NMR of water in polyimide films. J. Appl. Phys. 1989, 66, 5290–5296. [Google Scholar] [CrossRef]
- Lim, L.T.; Britt, U.; Tung, M.A. Sorption and transport of water vapor in nylon 6,6 film. J. Appl. Polym. Sci. 1999, 71, 197–206. [Google Scholar] [CrossRef]
- Puffr, R.; Sebenda, J. On the structure and properties of polyamides. The mechanism of water sorption in polyamides. J. Polym. Sci. C 1967, 16, 79–93. [Google Scholar] [CrossRef]
- Berens, A.R. Solubility of vinyl chloride in poly (vinyl chloride). Die Angew. Makromol. Chem. 1975, 47, 97–110. [Google Scholar] [CrossRef]
- Stapf, S.; Kimmich, R.; Seitter, R.O. Proton and deuteron field cycling NMR relaxometry of liquids in porous glasses: Evidence for Lévy-Walk Statistics. Phys. Rev. Lett. 1995, 75, 2855–2858. [Google Scholar] [CrossRef] [PubMed]
- Sammon, C.; Mura, C.; Yarwood, J.; Everall, N.; Swart, R.; Hodge, D. FTIR-ATR studies on the structure and dynamics of water molecules in polymeric matrixes. A comparison of PET and PVC. J. Phys. Chem. B 1998, 102, 3402–3411. [Google Scholar] [CrossRef]
- Rittigstein, P.; Priestley, R.D.; Broadbelt, L.J.; Torkelson, J.M. Model polymer nanocomposites provide an understanding of confinement effects in real nanocomposites. Nat. Mater. 2007, 6, 278–282. [Google Scholar] [CrossRef] [PubMed]
- Lan, T.; Torkelson, J.M. Methacrylate-based polymer films useful in lithographic applications exhibit different glass transition temperature-confinement effects at high and low molecular weight. Polymer 2014, 55, 1249–1258. [Google Scholar] [CrossRef]
- Tortora, M.; Gorrasi, G.; Vittoria, V.; Galli, G.; Ritrovati, S.; Chiellini, E. Structural characterization and transport properties of organically modified montmorillonite/polyurethane nanocomposites. Polymer 2002, 43, 6147–6157. [Google Scholar] [CrossRef]
- Mohan, T.P.; Kanny, K. Water barrier properties of nanoclay filled sisal fibre reinforced epoxy composites. Composites Part A 2011, 42, 385–393. [Google Scholar] [CrossRef]

| Sample | D (mm2/min) | Thickness (mm) | Absorbed Water (wt. %) |
|---|---|---|---|
| PU (equilibrium) | Gravimetric: 1.93 × 10−5 Spectroscopic: 2.09 × 10−5 | 0.652 | 15 |
| Nanocomposite (equilibrium) | Gravimetric: 1.04 × 10−5 Spectroscopic: 9.22 × 10−6 | 1.037 | 9.95 |
| PU (saturation) | Gravimetric: 2.00 × 10−4 | 1.023 | 85 |
| Nanocomposite (saturation) | Gravimetric: 1.16 × 10−4 | 0.975 | 52 |
| PU | ||||||
|---|---|---|---|---|---|---|
| Peak | Center (cm−1) | Height (a.u.) | Left (cm−1) | Right (cm−1) | Fwhh a (cm−1) | Area (a.u.) |
| Peak 1 | 5212 | 0.346 | 5500 | 4900 | 123 | 53.6 |
| Peak 2 | 5113 | 0.336 | 5600 | 4600 | 171 | 72.0 |
| Peak 3 | 4931 | 0.099 | 5500 | 4500 | 239 | 29.8 |
| Nanocomposite | ||||||
| Peak 1 | 5211 | 0.643 | 5500 | 4900 | 122 | 98.6 |
| Peak 2 | 5214 | 0.630 | 5600 | 4600 | 170 | 134.5 |
| Peak 3 | 4931 | 0.198 | 5500 | 4500 | 246 | 61.3 |
| Sample | Tg (°C) | Tg (°C) |
|---|---|---|
| PUdry | 130 | −40 |
| PUwet | 49 | −43 |
| PUwetI | 30 | −42 |
| Nanocompositedry | 132 | −40 |
| Nanocompositewet | 53 | −43 |
| NanocompositewetI | 44 | −42 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abbate, M.; D’Orazio, L. Water Diffusion through a Titanium Dioxide/Poly(Carbonate Urethane) Nanocomposite for Protecting Cultural Heritage: Interactions and Viscoelastic Behavior. Nanomaterials 2017, 7, 271. https://doi.org/10.3390/nano7090271
Abbate M, D’Orazio L. Water Diffusion through a Titanium Dioxide/Poly(Carbonate Urethane) Nanocomposite for Protecting Cultural Heritage: Interactions and Viscoelastic Behavior. Nanomaterials. 2017; 7(9):271. https://doi.org/10.3390/nano7090271
Chicago/Turabian StyleAbbate, Mario, and Loredana D’Orazio. 2017. "Water Diffusion through a Titanium Dioxide/Poly(Carbonate Urethane) Nanocomposite for Protecting Cultural Heritage: Interactions and Viscoelastic Behavior" Nanomaterials 7, no. 9: 271. https://doi.org/10.3390/nano7090271