Inkjet Printing of Carbon Nanotubes
Abstract
:1. Introduction
2. Carbon Nanotube Network

3. Carbon Nanotube Ink
3.1. Carbon Nanotube Dispersion
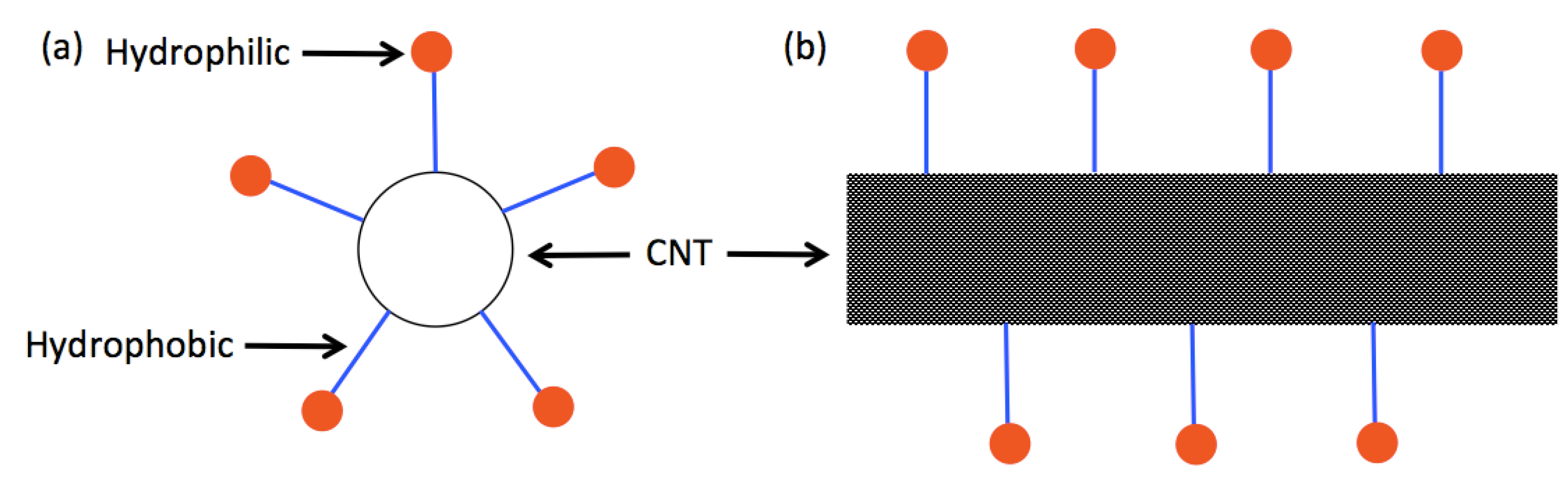
3.1.1. Functionalized Carbon Nanotube Dispersion
3.1.2. Organic Solvent-Based Carbon Nanotube Dispersion
3.1.3. Water-Based Carbon Nanotube Dispersion
3.2. Carbon Nanotube Ink Surface Tension

3.3. Carbon Nanotube Ink Preparation
4. Carbon Nanotube Inkjet Printing
4.1. Inkjet Printers
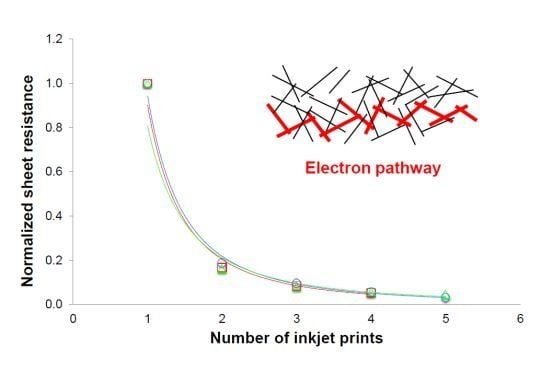
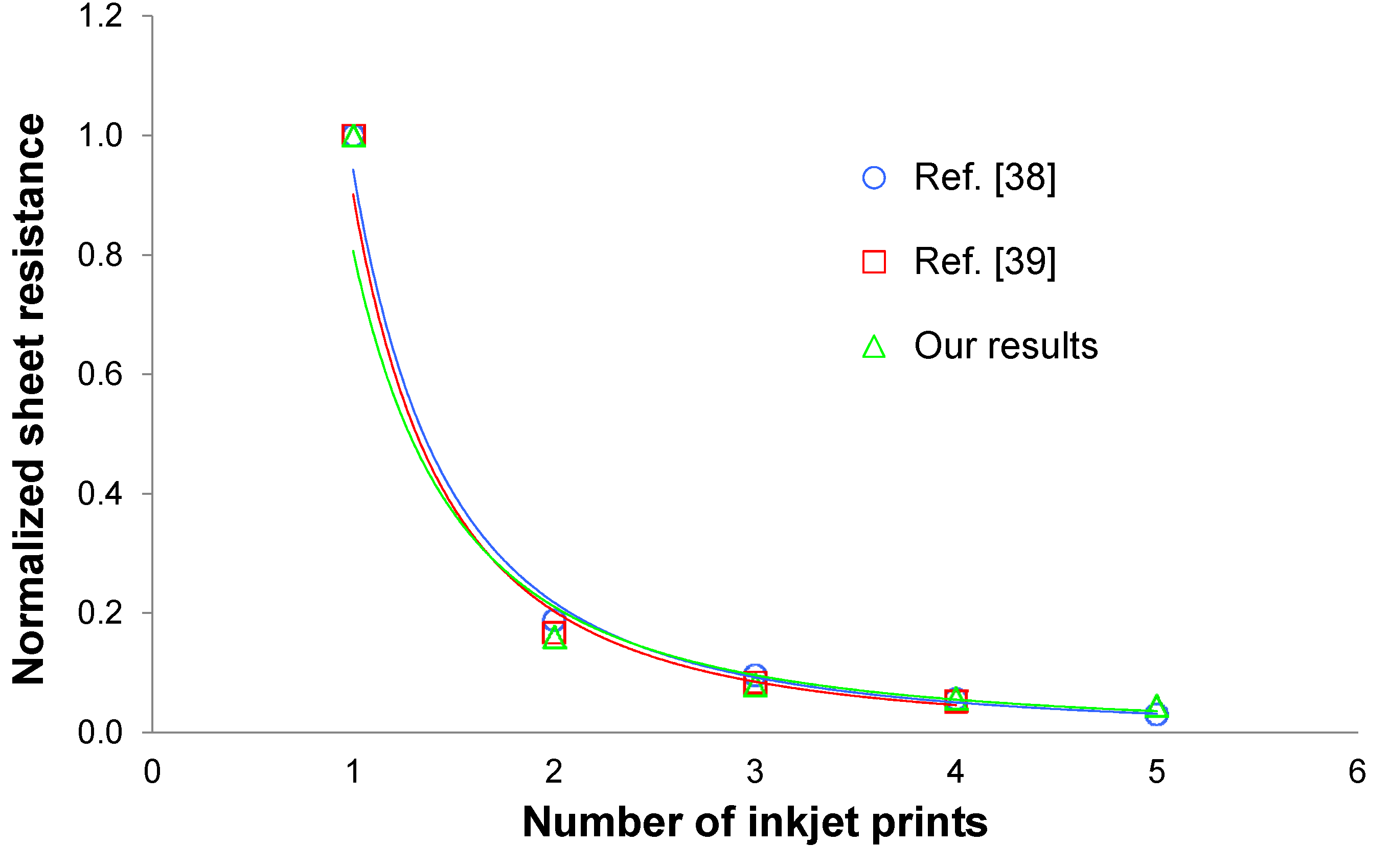
4.2. Sheet Resistance
4.3. Transparency
4.4. The Coffee Stain Effect
| Reference Number | Cited Papers | Printer | Solvent | Dispersant and Concentration | CNT Type and Concentration | Preparation | Best Sheet Resistance | Notable Feature |
|---|---|---|---|---|---|---|---|---|
| [43] | [39] | Canon | Water | Functionalized | MWCNT-COOH 0.26 mg/mL | Sonication Stirring Centrifuge | 40 kΩ/□ 90 prints | One of the first reported |
| [42] | [43,55] | Dimatix | Water | Functionalized | SWCNT-COOH 0.1 mg/mL | Sonication Centrifuge | Not reported | FET-like behavior |
| [41] | [42,43,49] | Dimatix | Water | Functionalized | SWCNT-COOH (carboxylic acid) SWCNT-CONH2 (amide) SWCNT-PEG (polyethylene glycol) SWCNT-PABS (polyaminobenzene sulfonic acid) 0.13 mg/mL | Sonication Centrifuge | Estimated 2 kΩ/□ (for COOH and PABS) 14 prints (Assumed) | Fully inkjet printed FET |
| [46] | - | MicroJet | DMF | n/a | SWCNT 20 µg/mL | Centrifuge | Not reported | Gas sensing |
| [47] | - | MicroJet | DMF | n/a | SWCNT 0.01 mg/mL | Sonication | Not reported | Field emission display |
| [3] | [38,46] | Dimatix | DMF | n/a | SWCNT 0.4 mg/mL | Sonication | Estimated 150 Ω/□ 25 prints | RFID and gas detection |
| [38] | [43] | MicroJet | DMF | n/a | SWCNT 0.02 mg/mL | Centrifuge | Estimated 333 Ω/□ 8 prints | Uniform CNT network |
| [45] | [48,58] | MicroJet | DMF | n/a | SWCNT 0.001 µg/mL or 0.04 μg/mL (Assumed) | Sonication Filtering | Not reported | Doping of CNT Films |
| [48] | [42,43,49] | MicroJet | DMF | n/a | SWCNT 0.001 µg/mL or 0.04 μg/mL | Sonication Centrifuge Filtering | Not reported | Fully inkjet printed FET |
| [49] | [43] | Microdrop Autodrop | NMP | n/a | SWCNT 0.003 mg/mL | Sonication Centrifuge Filtering | Not reported | Use of CNT as active layer in TFT |
| [39] | - | Not reported | Water | Special dispersant S27000 | MWCNT 3 mg/mL | Centrifuge Sonication | 11.6 k Ω/□ 4 prints | One of the first reported |
| [52] | - | HP | Water | Gellan gum or xanthan gum <1 mg/mL | SWCNT or MWCNT Concentration not reported | Sonication | Not reported | Water vapor detection |
| [53] | [38,43,59] | Epson | Water | SDS 10 mg/mL | SWCNT 0.2 mg/mL | Sonication Centrifuge | 78 Ω/□ 200 prints | Supercapacitors |
| [50] | [39,41,43,51,57,60] | Epson | Water | Combination of 3 different dispersants 150 mg/mL | MWCNT 0.15 mg/mL | Mixing Ball-milling Centrifuge | 760 Ω/□ 12 prints | Low sheet resistance |
| [51] | [38,39,43,47,49,52,59] | Microfab | Water | Solsperce® 46000 5 mg/mL Byk 348 1 mg/mL | MWCNT 10 mg/mL | Sonication | Not reported | Electroluminescent device |
5. Conclusions and Future Outlook
Conflict of Interest
References
- Zhou, Y.; Gaur, A.; Hur, S.-H.; Kocabas, C.; Meitl, M.A.; Shim, M.; Rogers, J.A. P-channel, n-channel thin film transistors and p−n diodes based on single wall carbon nanotube networks. Nano Lett. 2004, 4, 2031–2035. [Google Scholar] [CrossRef]
- Artukovic, E.; Kaempgen, M.; Hecht, D.S.; Roth, S.; Grüner, G. Transparent and flexible carbon nanotube transistors. Nano Lett. 2005, 5, 757–760. [Google Scholar] [CrossRef]
- Yang, L.; Zhang, R.; Staiculescu, D.; Wong, C.P.; Tentzeris, M.M. A novel conformal RFID-enabled module utilizing inkjet-printed antennas and carbon nanotubes for gas-detection applications. IEEE Antennas Wirel. Propag. Lett. 2009, 8, 653–656. [Google Scholar] [CrossRef]
- Li, J.; Lu, Y.; Ye, Q.; Cinke, M.; Han, J.; Meyyappan, M. Carbon nanotube sensors for gas and organic vapor detection. Nano Lett. 2003, 3, 929–933. [Google Scholar] [CrossRef]
- Chopra, S.; McGuire, K.; Gothard, N.; Rao, A.M.; Pham, A. Selective gas detection using a carbon nanotube sensor. Appl. Phys. Lett. 2003, 83, 2280–2282. [Google Scholar] [CrossRef]
- Zhang, T.; Mubeen, S.; Myung, N.V.; Deshusses, M.A. Recent progress in carbon nanotube-based gas sensors. Nanotechnology 2008, 19, 332001. [Google Scholar] [CrossRef]
- Fu, D.; Okimoto, H.; Lee, C.W.; Takenobu, T.; Iwasa, Y.; Kataura, H.; Li, L.-J. Ultrasensitive detection of DNA molecules with high on/off single-walled carbon nanotube network. Adv. Mater. 2010, 22, 4867–4871. [Google Scholar] [CrossRef]
- Scardaci, V.; Coull, R.; Coleman, J.N.; Byrne, L.; Scott, G. Carbon Nanotube Network Based Sensors. In Proceedings of 2012 12th IEEE Conference on Nanotechnology (IEEE-NANO), Birmingham, UK, 20–23 August 2012; pp. 1–3.
- Avouris, P.; Freitag, M.; Perebeinos, V. Carbon-nanotube photonics and optoelectronics. Nat. Photonics 2008, 2, 341–350. [Google Scholar] [CrossRef]
- Choi, W.B.; Chung, D.S.; Kang, J.H.; Kim, H.Y.; Jin, Y.W.; Han, I.T.; Lee, Y.H.; Jung, J.E.; Lee, N.S.; Park, G.S.; et al. Fully sealed, high-brightness carbon-nanotube field-emission display. Appl. Phys. Lett. 1999, 75, 3129–3131. [Google Scholar] [CrossRef]
- Abera, A.; Choi, J.-W. Quantitative lateral flow immunosensor using carbon nanotubes as label. Anal. Methods 2010, 2, 1819–1822. [Google Scholar] [CrossRef]
- Ng, M.H.A.; Hartadi, L.T.; Tan, H.; Poa, C.H.P. Efficient coating of transparent and conductive carbon nanotube thin films on plastic substrates. Nanotechnology 2008, 19, 205703. [Google Scholar] [CrossRef]
- Geng, H.-Z.; Kim, K.K.; So, K.P.; Lee, Y.S.; Chang, Y.; Lee, Y.H. Effect of acid treatment on carbon nanotube-based flexible transparent conducting films. J. Am. Chem. Soc. 2007, 129, 7758–7759. [Google Scholar] [CrossRef]
- Schrage, C.; Kaskel, S. Flexible and transparent SWCNT electrodes for alternating current electroluminescence devices. ACS Appl. Mater. Interfaces 2009, 1, 1640–1644. [Google Scholar] [CrossRef]
- Sarkar, A.; Daniels-Race, T. Electrophoretic deposition of carbon nanotubes on 3-amino-popyl-triethoxysilane (APTES) surface functionalized silicon substrates. Nanomaterials 2013, 3, 272–288. [Google Scholar] [CrossRef]
- Li, J.; Lei, W.; Zhang, X.; Zhou, X.; Wang, Q.; Zhang, Y.; Wang, B. Field emission characteristic of screen-printed carbon nanotube cathode. Appl. Surf. Sci. 2003, 220, 96–104. [Google Scholar] [CrossRef]
- Jones, C.S.; Lu, X.; Renn, M.; Stroder, M.; Shih, W.-S. Aerosol-jet-printed, high-speed, flexible thin-film transistor made using single-walled carbon nanotube solution. Microelectron. Eng. 2010, 87, 434–437. [Google Scholar] [CrossRef]
- Vaillancourt, J.; Zhang, H.; Vasinajindakaw, P.; Xia, H.; Lu, X.; Han, X.; Janzen, D.C.; Shih, W.-S.; Jones, C.S.; Stroder, M.; et al. All ink-jet-printed carbon nanotube thin-film transistor on a polyimide substrate with an ultrahigh operating frequency of over 5 GHz. Appl. Phys. Lett. 2008, 93, 243301. [Google Scholar] [CrossRef]
- Ha, M.; Xia, Y.; Green, A.A.; Zhang, W.; Renn, M.J.; Kim, C.H.; Hersam, M.C.; Frisbie, C.D. Printed, sub-3V digital circuits on plastic from aqueous carbon nanotube inks. ACS Nano 2010, 4, 4388–4395. [Google Scholar] [CrossRef]
- Zhou, Y.; Hu, L.; Grüner, G. A method of printing carbon nanotube thin films. Appl. Phys. Lett. 2006, 88, 123109. [Google Scholar] [CrossRef]
- Liu, C.-X.; Choi, J.-W. Patterning conductive PDMS nanocomposite in an elastomer using microcontact printing. J. Micromech. Microeng. 2009, 19, 085019. [Google Scholar] [CrossRef]
- Chow, E.; Herrmann, J.; Barton, C.S.; Raguse, B.; Wieczorek, L. Inkjet-printed gold nanoparticle chemiresistors: Influence of film morphology and ionic strength on the detection of organics dissolved in aqueous solution. Anal. Chim. Acta 2009, 632, 135–142. [Google Scholar] [CrossRef]
- Zhao, N.; Chiesa, M.; Sirringhaus, H.; Li, Y.; Wu, Y.; Ong, B. Self-aligned inkjet printing of highly conducting gold electrodes with submicron resolution. J. Appl. Phys. 2007, 101, 064513. [Google Scholar]
- Kim, D.; Moon, J. Highly conductive ink jet printed films of nanosilver particles for printable electronics. Electrochem. Solid State Lett. 2005, 8, J30–J33. [Google Scholar] [CrossRef]
- Lee, S.-H.; Shin, K.-Y.; Hwang, J.Y.; Kang, K.T.; Kang, H.S. Silver inkjet printing with control of surface energy and substrate temperature. J. Micromech. Microeng. 2008, 18, 075014. [Google Scholar] [CrossRef]
- Ham, H.T.; Choi, Y.S.; Chung, I.J. An explanation of dispersion states of single-walled carbon nanotubes in solvents and aqueous surfactant solutions using solubility parameters. J. Colloid Interface Sci. 2005, 286, 216–223. [Google Scholar] [CrossRef]
- O’Connell, M.J.; Bachilo, S.M.; Huffman, C.B.; Moore, V.C.; Strano, M.S.; Haroz, E.H.; Rialon, K.L.; Boul, P.J.; Noon, W.H.; Kittrell, C.; et al. Band gap fluorescence from individual single-walled carbon nanotubes. Science 2002, 297, 593–596. [Google Scholar] [CrossRef]
- Liu, J.; Rinzler, A.G.; Dai, H.; Hafner, J.H.; Bradley, R.K.; Boul, P.J.; Lu, A.; Iverson, T.; Shelimov, K.; Huffman, C.B.; et al. Fullerene pipes. Science 1998, 280, 1253–1256. [Google Scholar] [CrossRef]
- Islam, M.F.; Rojas, E.; Bergey, D.M.; Johnson, A.T.; Yodh, A.G. High weight fraction surfactant solubilization of single-wall carbon nanotubes in water. Nano Lett. 2003, 3, 269–273. [Google Scholar] [CrossRef]
- Moore, V.C.; Strano, M.S.; Haroz, E.H.; Hauge, R.H.; Smalley, R.E.; Schmidt, J.; Talmon, Y. Individually suspended single-walled carbon nanotubes in various surfactants. Nano Lett. 2003, 3, 1379–1382. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, T.; Sreekumar, T.V.; Kumar, S.; Moore, V.C.; Hauge, R.H.; Smalley, R.E. Poly(vinyl alcohol)/SWNT composite film. Nano Lett. 2003, 3, 1285–1288. [Google Scholar] [CrossRef]
- O’Connell, M.J.; Boul, P.; Ericson, L.M.; Huffman, C.; Wang, Y.; Haroz, E.; Kuper, C.; Tour, J.; Ausman, K.D.; Smalley, R.E. Reversible water-solubilization of single-walled carbon nanotubes by polymer wrapping. Chem. Phys. Lett. 2001, 342, 265–271. [Google Scholar] [CrossRef]
- Zheng, M.; Jagota, A.; Semke, E.D.; Diner, B.A.; Mclean, R.S.; Lustig, S.R.; Richardson, R.E.; Tassi, N.G. DNA-assisted dispersion and separation of carbon nanotubes. Nat. Mater. 2003, 2, 338–342. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Nguyen, S.U.; Phuong, D.T.; Nguyen, D.C.; Mai, A.T. Dispersion of denatured carbon nanotubes by using a dimethylformamide solution. Adv. Nat. Sci. Nanosci. Nanotechnol. 2011, 2, 035015. [Google Scholar] [CrossRef]
- Ausman, K.D.; Piner, R.; Lourie, O.; Ruoff, R.S.; Korobov, M. Organic solvent dispersions of single-walled carbon nanotubes: Toward solutions of pristine nanotubes. J. Phys. Chem. B 2000, 104, 8911–8915. [Google Scholar] [CrossRef]
- Hasan, T.; Scardaci, V.; Tan, P.; Rozhin, A.G.; Milne, W.I.; Ferrari, A.C. Stabilization and “debundling” of single-wall carbon nanotube dispersions in n-methyl-2-pyrrolidone (NMP) by polyvinylpyrrolidone (PVP). J. Phys. Chem. C 2007, 111, 12594–12602. [Google Scholar]
- Liu, C.-X.; Choi, J.-W. Improved dispersion of carbon nanotubes in polymers at high concentrations. Nanomaterials 2012, 2, 329–347. [Google Scholar] [CrossRef]
- Song, J.-W.; Kim, J.; Yoon, Y.-H.; Choi, B.-S.; Kim, J.-H.; Han, C.-S. Inkjet printing of single-walled carbon nanotubes and electrical characterization of the line pattern. Nanotechnology 2008, 19, 095702. [Google Scholar] [CrossRef]
- Fan, Z.; Wei, T.; Luo, G.; Wei, F. Fabrication and characterization of multi-walled carbon nanotubes-based ink. J. Mater. Sci. 2005, 40, 5075–5077. [Google Scholar] [CrossRef]
- Hecht, D.; Hu, L.; Grüner, G. Conductivity scaling with bundle length and diameter in single walled carbon nanotube networks. Appl. Phys. Lett. 2006, 89, 133112. [Google Scholar] [CrossRef]
- Gracia-Espino, E.; Sala, G.; Pino, F.; Halonen, N.; Luomahaara, J.; Mäklin, J.; Tóth, G.; Kordás, K.; Jantunen, H.; Terrones, M.; et al. Electrical transport and field-effect transistors using inkjet-printed SWCNT films having different functional side groups. ACS Nano 2010, 4, 3318–3324. [Google Scholar] [CrossRef]
- Mustonen, T.; Mäklin, J.; Kordás, K.; Halonen, N.; Tóth, G.; Saukko, S.; Vähäkangas, J.; Jantunen, H.; Kar, S.; Ajayan, P.M.; et al. Controlled ohmic and nonlinear electrical transport in inkjet-printed single-wall carbon nanotube films. Phys. Rev. B 2008, 77, 125430. [Google Scholar] [CrossRef]
- Kordás, K.; Mustonen, T.; Tóth, G.; Jantunen, H.; Lajunen, M.; Soldano, C.; Talapatra, S.; Kar, S.; Vajtai, R.; Ajayan, P.M. Inkjet printing of electrically conductive patterns of carbon nanotubes. Small 2006, 2, 1021–1025. [Google Scholar] [CrossRef]
- Hecht, D.S.; Hu, L.; Irvin, G. Emerging transparent electrodes based on thin films of carbon nanotubes, graphene, and metallic nanostructures. Adv. Mater. 2011, 23, 1482–1513. [Google Scholar] [CrossRef]
- Matsuzaki, S.; Nobusa, Y.; Shimizu, R.; Yanagi, K.; Kataura, H.; Takenobu, T. Continuous electron doping of single-walled carbon nanotube films using inkjet technique. Jpn. J. Appl. Phys. 2012, 51, 06FD18:1–06FD18:3. [Google Scholar]
- Yun, J.-H.; Chang-Soo, H.; Kim, J.; Song, J.-W.; Shin, D.-H.; Park, Y.-G. Fabrication of Carbon Nanotube Sensor Device by Inkjet Printing. In Proceedings of 3rd IEEE International Conference on Nano/Micro Engineered and Molecular Systems, Sanya, China, 6–9 January 2008; 2008; pp. 506–509. [Google Scholar]
- Song, J.-W.; Kim, Y.-S.; Yoon, Y.-H.; Lee, E.-S.; Han, C.-S.; Cho, Y.; Kim, D.; Kim, J.; Lee, N.; Ko, Y.-G.; et al. The production of transparent carbon nanotube field emitters using inkjet printing. Physica E 2009, 41, 1513–1516. [Google Scholar] [CrossRef]
- Okimoto, H.; Takenobu, T.; Yanagi, K.; Miyata, Y.; Shimotani, H.; Kataura, H.; Iwasa, Y. Tunable carbon nanotube thin-film transistors produced exclusively via inkjet printing. Adv. Mater. 2010, 22, 3981–3986. [Google Scholar] [CrossRef]
- Beecher, P.; Servati, P.; Rozhin, A.; Colli, A.; Scardaci, V.; Pisana, S.; Hasan, T.; Flewitt, A.J.; Robertson, J.; Hsieh, G.W.; et al. Ink-jet printing of carbon nanotube thin film transistors. J. Appl. Phys. 2007, 102, 043710. [Google Scholar] [CrossRef]
- Kwon, O.-S.; Kim, H.; Ko, H.; Lee, J.; Lee, B.; Jung, C.-H.; Choi, J.-H.; Shin, K. Fabrication and characterization of inkjet-printed carbon nanotube electrode patterns on paper. Carbon 2013, 58, 116–127. [Google Scholar] [CrossRef]
- Azoubel, S.; Shemesh, S.; Magdassi, S. Flexible electroluminescent device with inkjet-printed carbon nanotube electrodes. Nanotechnology 2012, 23, 344003. [Google Scholar] [CrossRef]
- In het Panhuis, M.; Heurtematte, A.; Small, W.R.; Paunov, V.N. Inkjet printed water sensitive transparent films from natural gum–carbon nanotube composites. Soft Matter 2007, 3, 840–843. [Google Scholar] [CrossRef]
- Chen, P.; Chen, H.; Qiu, J.; Zhou, C. Inkjet printing of single-walled carbon nanotube/RuO2 nanowire supercapacitors on cloth fabrics and flexible substrates. Nano Res. 2010, 3, 594–603. [Google Scholar] [CrossRef]
- Noh, J.; Jung, M.; Jung, K.; Lee, G.; Lim, S.; Kim, D.; Kim, S.; Tour, J.M.; Cho, G. Integrable single walled carbon nanotube (SWNT) network based thin film transistors using roll-to-roll gravure and inkjet. Org. Electron. 2011, 12, 2185–2191. [Google Scholar] [CrossRef]
- Mustonen, T.; Kordás, K.; Saukko, S.; Tóth, G.; Penttilä, J.S.; Helistö, P.; Seppä, H.; Jantunen, H. Inkjet printing of transparent and conductive patterns of single-walled carbon nanotubes and PEDOT-PSS composites. Phys. Status Solidi B 2007, 244, 4336–4340. [Google Scholar] [CrossRef]
- Deegan, R.D.; Bakajin, O.; Dupont, T.F.; Huber, G.; Nagel, S.R.; Witten, T.A. Capillary flow as the cause of ring stains from dried liquid drops. Nature 1997, 389, 827–829. [Google Scholar] [CrossRef]
- Denneulin, A.; Bras, J.; Carcone, F.; Neuman, C.; Blayo, A. Impact of ink formulation on carbon nanotube network organization within inkjet printed conductive films. Carbon 2011, 49, 2603–2614. [Google Scholar] [CrossRef]
- Nobusa, Y.; Yomogida, Y.; Matsuzaki, S.; Yanagi, K.; Kataura, H.; Takenobu, T. Inkjet printing of single-walled carbon nanotube thin-film transistors patterned by surface modification. Appl. Phys. Lett. 2011, 99, 183106. [Google Scholar] [CrossRef]
- Small, W.R.; in het Panhuis, M. Inkjet printing of transparent, electrically conducting single-walled carbon-nanotube composites. Small 2007, 3, 1500–1503. [Google Scholar] [CrossRef]
- Denneulin, A.; Bras, J.; Blayo, A.; Khelifi, B.; Roussel-Dherbey, F.; Neuman, C. The influence of carbon nanotubes in inkjet printing of conductive polymer suspensions. Nanotechnology 2009, 20, 385701. [Google Scholar] [CrossRef]
- Takagi, Y.; Nobusa, Y.; Gocho, S.; Kudou, H.; Yanagi, K.; Kataura, H.; Takenobu, T. Inkjet printing of aligned single-walled carbon-nanotube thin films. Appl. Phys. Lett. 2013, 102, 143107. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tortorich, R.P.; Choi, J.-W. Inkjet Printing of Carbon Nanotubes. Nanomaterials 2013, 3, 453-468. https://doi.org/10.3390/nano3030453
Tortorich RP, Choi J-W. Inkjet Printing of Carbon Nanotubes. Nanomaterials. 2013; 3(3):453-468. https://doi.org/10.3390/nano3030453
Chicago/Turabian StyleTortorich, Ryan P., and Jin-Woo Choi. 2013. "Inkjet Printing of Carbon Nanotubes" Nanomaterials 3, no. 3: 453-468. https://doi.org/10.3390/nano3030453