Application of Sub-Micrometer Vibrations to Mitigate Bacterial Adhesion
Abstract
:1. Introduction
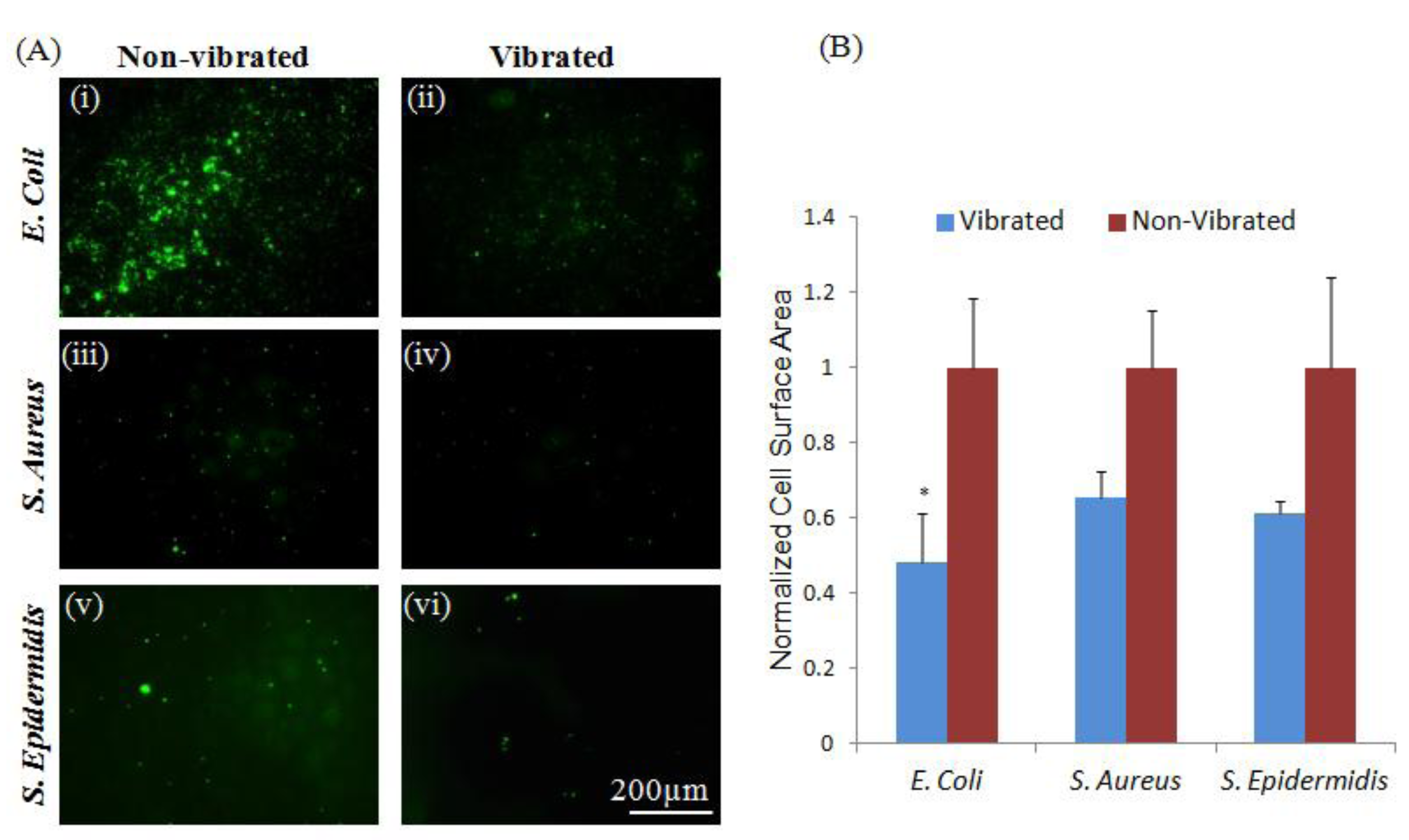
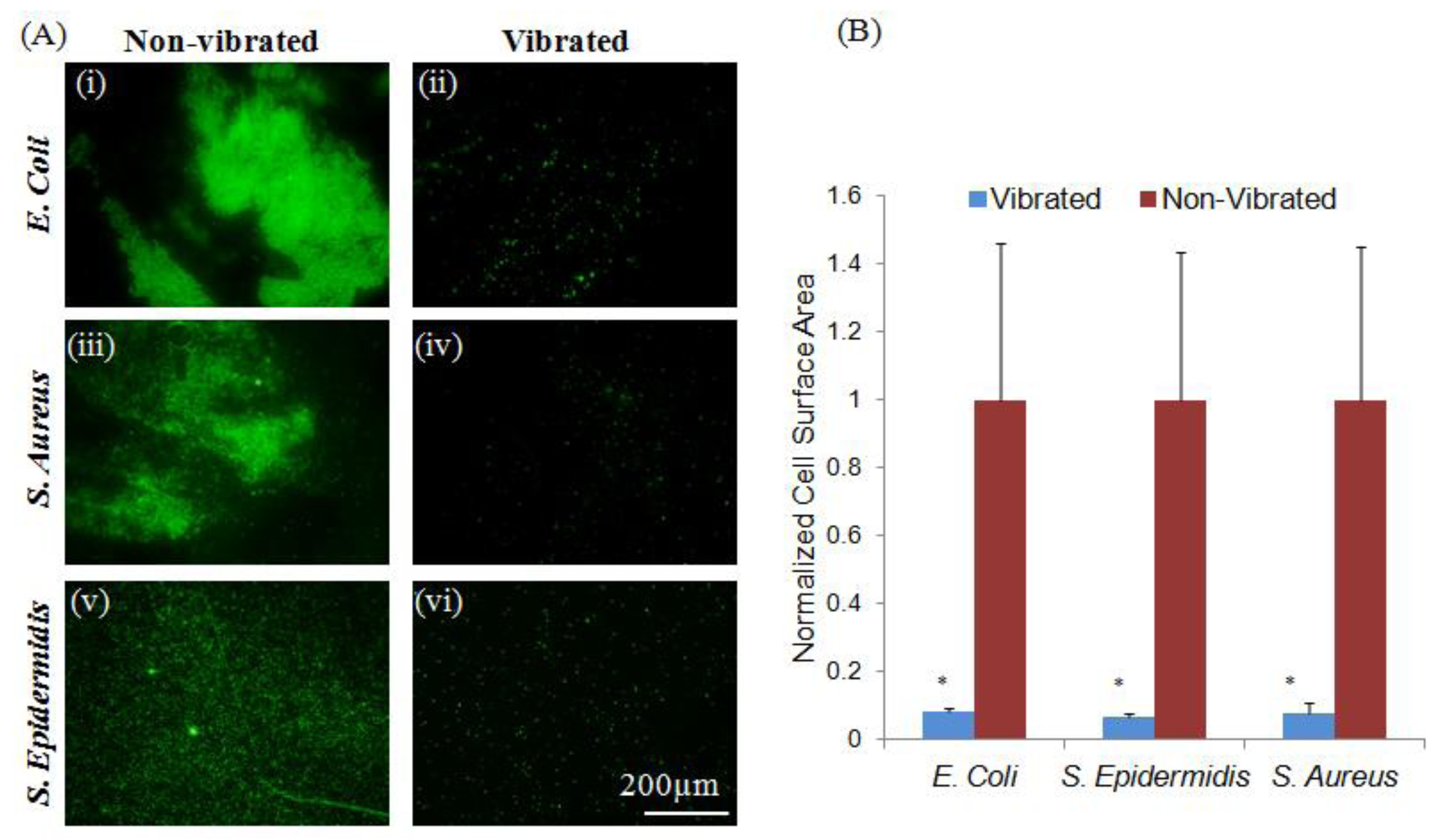
2. Results and Discussion

| Surface | Contact Angle (°θ) | Surface Roughness (RMS Average) | Surface Energy (mJ/m2) | Chemical Structure |
|---|---|---|---|---|
| Parylene-C | 84.2 | 9.01 | 19.6 [37] |  |
| Plasma Etched Parylene-C | 21.3 | 26.3 | 46.5 [38] | – |
| PLLA | 71.7 | 0.211 | 43.2 [39] |  |

3. Experimental Section
3.1. Preparation of Magnetoelastic (ME) Samples
3.1.1. Parylene-C Coating
3.1.2. Plasma Etching
3.1.3. Explanted Sensors
3.1.4. Poly-L-Lactide (PLLA)
3.1.5. Surface Characterization
3.2. Bacterial Culture
3.3. Colony Staining and Fluorescent Imaging
3.4. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Christensen, G.; Simpson, W.; Bisno, A.; Beachey, E. Adherence of slime-producing strains of Staphylococcus epidermidis to smooth surfaces. Infect. Immunol. 1982, 37, 318–326. [Google Scholar]
- Cunliffe, D.; Smart, C.; Alexander, C.; Vulfson, E. Bacterial adhesion at synthetic surfaces. Appl. Environ. Microbiol. 1999, 65, 4995–5002. [Google Scholar]
- Richards, M.; Edwards, J.; Culver, D.; Gaynes, R. Nosocomial infections in pediatric intensive care units in the United States. Pediatrics 1999, 103, e39. [Google Scholar] [CrossRef]
- Peters, G.; Locci, R.; Pulverer, G. Adherence and growth of coagulase-negative staphylococci on surfaces of intravenous catheters. J. Infect. Dis. 1982, 146, 479–482. [Google Scholar] [CrossRef]
- Hawkey, P.; Jones, A. The changing epidemiology of resistance. J. Antimicrob. Chemother. 2009, 64. [Google Scholar] [CrossRef]
- Balaban, N.; Giacometti, A.; Cirioni, O.; Gov, Y.; Ghiselli, R.; Mocchegiani, F.; Viticchi, C.; Del Prete, M.; Saba, V.; Scalise, G.; et al. Use of the quorum-sensing inhibitor RNAIII-inhibiting peptide to prevent biofilm formation in vivo by drug-resistant staphylococcus epidermidis. J. Infect. Dis. 2003, 187, 625–630. [Google Scholar] [CrossRef]
- Ionita, D.; Grecu, M.; Ungureanu, C.; Demetrescu, I. Antimicrobial activity of the surface coatings on TiAlZr implant biomaterial. J. Biosci. Bioeng. 2011, 112, 630–634. [Google Scholar] [CrossRef]
- Autar, R.; Khan, A.; Schad, M.; Hacker, J.; Liskamp, R.; Pieters, R. Adhesion inhibition of F1C-fimbriated Escherichia coli and Pseudomonas aeruginosa PAK and PAO by multivalent carbohydrate ligands. Chembiochem 2003, 4, 1317–1325. [Google Scholar] [CrossRef]
- Batisson, I.; Guimond, M.P.; Girard, F.; An, H.; Zhu, C.; Oswald, E.; Fairbrother, J.; Jaques, M.; Harel, J. Characterization of the novel factor PAA involved in the early steps of the adhesion mechanism of attaching and effacing Escherichia coli. Infect. Immun. 2003, 71, 4516–4525. [Google Scholar] [CrossRef]
- Yakovenko, O.; Sharma, S.; Forero, M.; Tchesnokova, V.; Aprikian, P.; Kidd, B.; Mach, A.; Vogel, V.; Sokurenko, E.; Thomas, W. FimH forms catch bonds that are enhanced by mechanical force due to allosteric regulation. J. Biol. Chem. 2008, 283, 11596–11605. [Google Scholar] [CrossRef]
- Azghani, A.; Idell, S.; Bains, M.; Hancock, R. Pseudomonas aeruginosa outer membrane protein f is an adhesin in bacterial binding to lung epithelial cells in culture. Microb. Pathog. 2002, 33, 109–114. [Google Scholar] [CrossRef]
- Merino, S.; Rubires, X.; Aguilar, A.; Tomás, J. The role of flagella and motility in the adherence and invasion to fish cell lines by Aeromonas hydrophila serogroup O:34 strains. FEMS Microb. Lett. 1997, 151, 213–217. [Google Scholar] [CrossRef]
- Doig, P.; Todd, T.; Sastry, P.; Lee, K.; Hodges, R.; Paranchych, W.; Irvin, R. Role of pili in adhesion of Pseudomonas aeruginosa to human respiratory epithelial cells. Infect. Immun. 1988, 56, 1641–1646. [Google Scholar]
- Miller, R.; Tomaras, A.; Barker, A.; Voelker, D.; Chan, E.; Vasil, A.; Vasil, M. Pseudomonas aeruginosa twitching motility-mediated chemotaxis towards phospholipids and fatty acids: specificity and metabolic requirements. J. Bacteriol. 2008, 190, 4038–4049. [Google Scholar] [CrossRef]
- Stewart, P.; Costerton, J. Antibiotic resistance of bacteria in biofilms. Lancet 2001, 358, 135–138. [Google Scholar] [CrossRef]
- Drago, L.; Mattina, R.; Nicola, L.; Rodighiero, V.; De Vecchi, E. Macrolide resistance and in vitro selection of resistance to antibiotics in Lactobacillus isolates. J. Microbiol. 2011, 49, 651–656. [Google Scholar] [CrossRef]
- Brandt, O.; Mildner, M.; Egger, A.; Groessl, M.; Rix, U.; Posch, M.; Keppler, B.; Strupp, C.; Mueller, B.; Stingl, G. Nanoscalic silver possesses broad-spectrum antimicrobial activities and exhibits fewer toxicological side effects than silver sulfadiazine. Nanomedicine 2011, 8, 478–488. [Google Scholar]
- Wynne, J.; Fulmer, P.; McCluskey, D.; Mackey, N.; Buchanan, J. Synthesis and development of a multifunctional self-decontaminating polyurethane coating. ACS Appl. Mater. Interfaces 2001, 3, 2005–2011. [Google Scholar]
- Fortin, J.; Lu, T. A model for the chemical vapor deposition of poly (para-xylylene) (Parylene) thin films. Chem. Mater. 2002, 14, 1945–1949. [Google Scholar] [CrossRef]
- Wang, I.; Anderson, J.; Marchant, R. Staphylococcus epidermidis adhesion to hydrophobic biomedical polymer is mediated by platelets. J. Infect. Dis. 1993, 167, 329–336. [Google Scholar] [CrossRef]
- Yu, Q.; Deffeyes, J.; Yasuda, H. Engineering the surface and interface of Parylene C coatings by low-temperature plasmas. Prog. Org. Coat. 2001, 41, 247–253. [Google Scholar] [CrossRef]
- Chang, T.; Yadav, V.; De Leo, S.; Mohedas, A.; Rajalingam, B.; Chen, C.L.; Selvarasah, S.; Dokmeci, M.; Khademhosseini, A. Cell and protein compatibility of Parylene-C surface. Langmuir 2007, 23, 11718–11725. [Google Scholar] [CrossRef]
- Dror, N.; Mandel, M.; Hazan, Z.; Lavie, G. Advances in microbial biofilm prevention on indwelling medical devices with emphasis on usage of acoustic energy. Sensors 2009, 9, 2538–2554. [Google Scholar] [CrossRef]
- Pitt, W.; Ross, S. Ultrasound increases the rate of bacterial cell growth. Biotechnol. Prog. 2003, 19, 1038–1044. [Google Scholar] [CrossRef]
- Hazan, Z.; Zumeris, J.; Jacob, H.; Raskin, H.; Kratysh, G.; Vishnia, M.; Dror, N.; Barliya, T.; Mandel, M.; Lavie, G. Effective prevention of microbial biofilm formation on medical devices by low-energy surface acoustic waves. Antimicrob. Agents Chemother. 2006, 50, 4144–4152. [Google Scholar] [CrossRef]
- Qian, Z.; Stoodley, P.; Pitt, W. Effect of low-intensity ultrasound upon biofilm structure from confocal scanning laser microscopy observation. Biomaterials 1996, 17, 1975–1980. [Google Scholar] [CrossRef]
- Grimes, C.; Mungle, C.; Zeng, K.; Jain, M.; Dreschel, W.; Paulose, M.; Ong, K.G. Wireless magnetoelastic resonance sensors: A critical review. Sensors 2002, 2, 294–313. [Google Scholar]
- Huang, S.; Yang, H.; Lakshmanan, R.; Johnson, M.; Wan, J.; Chen, I.H.; Wikle, H., III; Petrenko, V.; Barbaree, J.; Chin, B. Sequential detection of Salmonella typhimurium and Bacillus anthracis spores using magnetoelastic biosensors. Biosens. Bioelectron. 2009, 24, 1730–1736. [Google Scholar] [CrossRef]
- Xiao, X.; Guo, M.; Li, Q.; Cai, Q.; Yao, S.; Grimes, C. In-situ monitoring of breast cancer cell (MCF-7) growth and quantification of the cytotoxicity of anticancer drugs fluorouracil and cisplatin. Biosen. Bioelectron. 2008, 24, 247–252. [Google Scholar] [CrossRef]
- Vlaisavljevich, E.; Janka, J.; Ong, K.G.; Rajachar, R. Magnetoelastic materials as novel bioactive coatings for the control of cell adhesion. IEEE Trans. Biomed. Eng. 2011, 58, 698–704. [Google Scholar] [CrossRef]
- Pankhurst, Q.A.; Connolly, J.; Jones, S.K.; Dobson, J. Applications of magnetic nanoparticles in biomedicine. J. Phys. D 2003, 36. [Google Scholar] [CrossRef]
- Banobre-Lopez, M.; Pineiro-Redondo, Y.; De Santis, R.; Gloria, A.; Ambrosio, L.; Tampieri, A.; Dediu, V.; Rivas, J. Poly(caprolactone) based magnetic scaffolds for bone tissue engineering. J. Appl. Phys. 2011, 109. [Google Scholar] [CrossRef]
- De Santis, R.; Gloria, A.; Russo, T.; D’Amora, U.; Zeppetelli, S.; Dionigi, C.; Sytcheva, A.; Herrmannsdo, T.; Dediu, V.; Ambrosio, L. Magnetic poly(ε-caprolactone)/iron-doped hydroxyapatite nanocomposite substrates for advanced bone tissue engineering. J. R. Soc. Interface 2013, 10. [Google Scholar] [CrossRef]
- Meng, E.; Li, P.; Tai, Y. Plasma removal of Parylene C. J. Micromech. Microeng. 2008, 18, 5004–5017. [Google Scholar]
- Vitagliano, L.; Berisio, R.; De Simone, A. Role of hydration in collagen recognition by bacterial adhesions. Biophys. J. 2011, 100, 2253–2261. [Google Scholar] [CrossRef]
- Antão, E.; Weilar, L.; Ewers, C. Adhesive threads of extraintestinal pathogenic Escherichia coli. Gut Pathog. 2009, 1, 1–22. [Google Scholar] [CrossRef]
- Tan, C.; Craighead, H. Surface engineering and patterning using Parylene for biological applications. Materials 2010, 3, 1803–1832. [Google Scholar] [CrossRef]
- Subbiahdoss, G.; Kuijer, R.; Grijpma, D.; Van Der Mei, H.; Busscher, H. Microbial biofilm growth vs. tissue integration: The race for the surface, experimentally studied. Acta Biomater. 2009, 5, 1399–1404. [Google Scholar] [CrossRef]
- Yang, J.; Shi, G.; Bei, J.; Wang, S.; Cao, Y.; Shang, Q.; Yang, G.; Wang, W. Fabrication and surface modification of macroporous poly(L-lactic acid) and poly(L-lactic-co-glycolic acid) (70/30) cell scaffolds for human skin fibroblast cell culture. J. Biomed. Mater. Res. 2002, 62, 438–446. [Google Scholar] [CrossRef]
- Ham, Y.; Shutov, D.; Baek, K.; Do, L.; Kim, K.; Lee, C.; Kwon, K. Surface characteristics of Parylene-C films in an inductively coupled O2/CF4 gas plasma. Thin Solid Films 2010, 518, 6378–6381. [Google Scholar] [CrossRef]
- Modzelewski, C.; Savage, H.; Kabacoff, L.; Clark, A. Magnetomechanical coupling and permeability in transversely annealed metglas 2605 alloys. IEEE Trans. Magn. 1981, 17, 2837–2839. [Google Scholar] [CrossRef]
- Holmes, H.R.; Tan, E.L.; Ong, K.G.; Rajachar, R.M. Fabrication of biocompatible, vibrational magnetoelastic materials for controlling cellular adhesion. Biosensors 2012, 2, 57–62. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Paces, W.R.; Holmes, H.R.; Vlaisavljevich, E.; Snyder, K.L.; Tan, E.L.; Rajachar, R.M.; Ong, K.G. Application of Sub-Micrometer Vibrations to Mitigate Bacterial Adhesion. J. Funct. Biomater. 2014, 5, 15-26. https://doi.org/10.3390/jfb5010015
Paces WR, Holmes HR, Vlaisavljevich E, Snyder KL, Tan EL, Rajachar RM, Ong KG. Application of Sub-Micrometer Vibrations to Mitigate Bacterial Adhesion. Journal of Functional Biomaterials. 2014; 5(1):15-26. https://doi.org/10.3390/jfb5010015
Chicago/Turabian StylePaces, Will R., Hal R. Holmes, Eli Vlaisavljevich, Katherine L. Snyder, Ee Lim Tan, Rupak M. Rajachar, and Keat Ghee Ong. 2014. "Application of Sub-Micrometer Vibrations to Mitigate Bacterial Adhesion" Journal of Functional Biomaterials 5, no. 1: 15-26. https://doi.org/10.3390/jfb5010015





