Effects of Medium and Temperature on Cellular Responses in the Superficial Zone of Hypo-Osmotically Challenged Articular Cartilage
Abstract
:1. Introduction
| Component | DMEM 290 mOsm | PBS 290 mOsm |
|---|---|---|
| Amino Acids | × | – |
| Vitamins | × | – |
| D-glucose (Dextrose) | × | – |
| Sodium Chloride (NaCl) | 6,400 mg/L | 8,000 mg/L |
| Potassium Chloride (KCl) | 400 mg/L | 200 mg/L |
| Sodium Phosphate monobasic (NaH2PO4) | 141 mg/L | – |
| Sodium Phosphate dibasic (Na2HPO4) | – | 1,440 mg/L |
| Potassium Phosphate monobasic (KH2PO4) | – | 240 mg/L |
| Calcium Chloride (CaCl2) anhyd. | 264 mg/L | – |
| Ferric Nitrate (Fe(NO3)3''9H2O) | 0.1 mg/L | – |
| Magnesium Sulfate (MgSO4) anhyd. | 200 mg/L | – |
| Sodium Bicarbonate (NaHCO3) | 3,700 mg/L | – |
2. Experimental Section
2.1. Sample Preparation
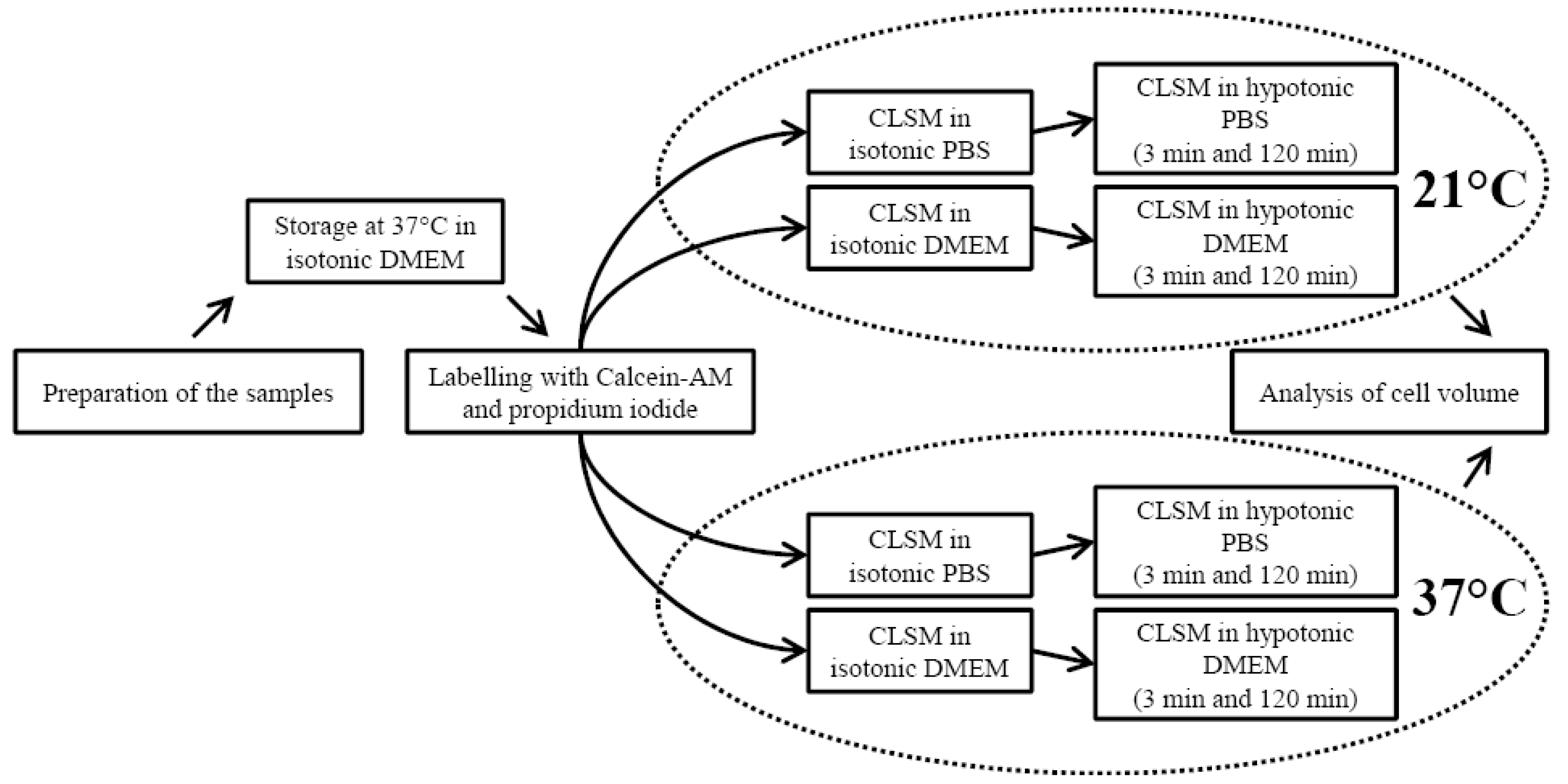
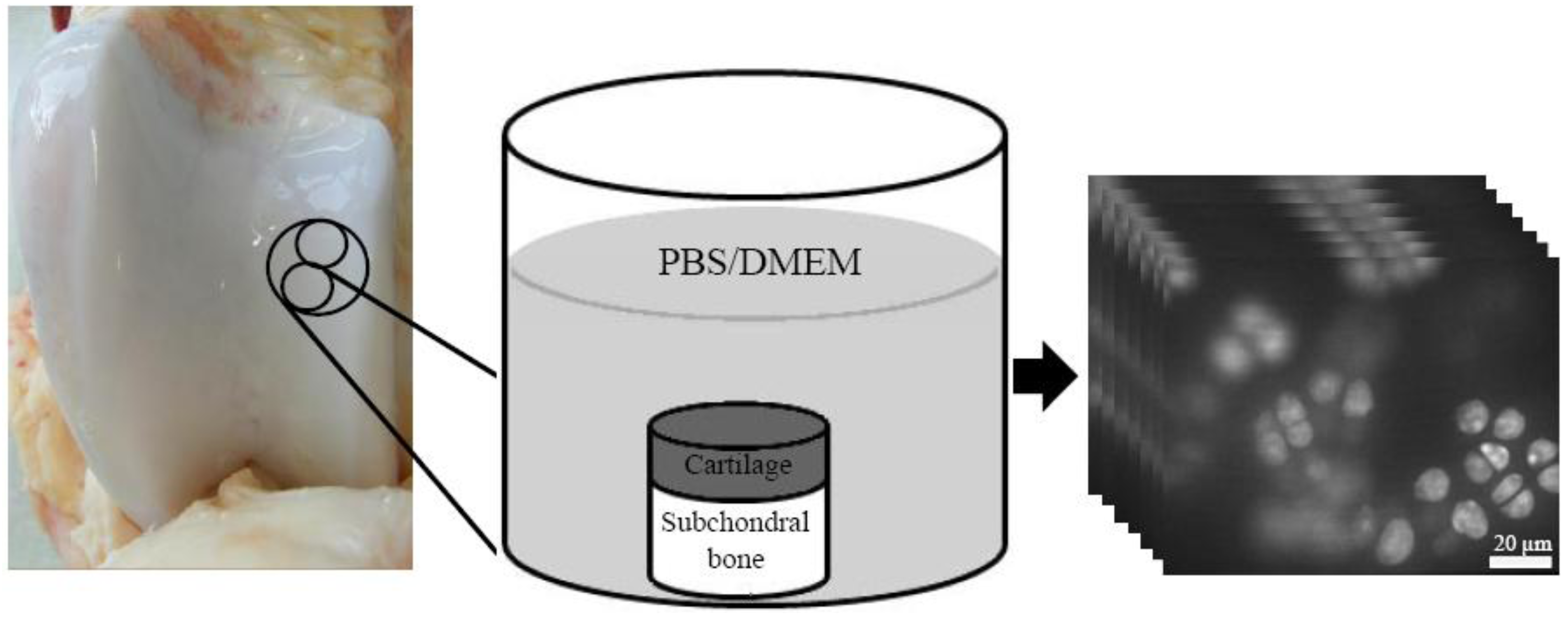

2.2. Confocal Laser Scanning Microscopy
2.3. Data Analysis
2.4. Statistical Analysis
3. Results

4. Discussion
5. Conclusions
Acknowledgments
References
- Bush, P.G.; Hall, A.C. Passive osmotic properties of in situ human articular chondrocytes within non-degenerate and degenerate cartilage. J. Cell Physiol. 2005, 204, 309–319. [Google Scholar] [CrossRef]
- Grushko, G.; Schneiderman, R.; Maroudas, A. Some biochemical and biophysical parameters for the study of the pathogenesis of osteoarthritis: A comparison between the processes of ageing and degeneration in human hip cartilage. Connect. Tissue Res. 1989, 19, 149–176. [Google Scholar] [CrossRef]
- Watson, P.J.; Carpenter, T.A.; Hall, L.D.; Tyler, J.A. Cartilage swelling and loss in a spontaneous model of osteoarthritis visualized by magnetic resonance imaging. Osteoarthritis Cartilage 1996, 4, 197–207. [Google Scholar] [CrossRef]
- Bush, P.G.; Hall, A.C. The volume and morphology of chondrocytes within non-degenerate and degenerate human articular cartilage. Osteoarthritis Cartilage 2003, 11, 242–251. [Google Scholar] [CrossRef]
- Bush, P.G.; Hall, A.C. The osmotic sensitivity of isolated and in situ bovine articular chondrocytes. J. Orthop. Res. 2001, 19, 768–778. [Google Scholar] [CrossRef]
- Bush, P.G.; Hall, A.C. Regulatory volume decrease (RVD) by isolated and in situ bovine articular chondrocytes. J. Cell Physiol. 2001, 187, 304–314. [Google Scholar] [CrossRef]
- Urban, J.P.G.; Hall, A.C.; Gehl, K.A. Regulation of matrix synthesis rates by the ionic and osmotic environment of articular chondrocytes. J. Cell. Physiol. 1993, 154, 262–270. [Google Scholar] [CrossRef]
- Korhonen, R.K.; Han, S.-K.; Herzog, W. Osmotic loading of in situ chondrocytes in their native environment. Mol. Cell. Biomech. 2010, 7, 125–134. [Google Scholar]
- Bush, P.G.; Hodkinson, P.D.; Hamilton, G.L.; Hall, A.C. Viability and volume of in situ bovine articular chondrocytes—Changes following a single impact and effects of medium osmolarity. Osteoarthritis Cartilage 2005, 13, 54–65. [Google Scholar] [CrossRef]
- Korhonen, R.K.; Han, S.-K.; Herzog, W. Osmotic loading of articular cartilage modulates cell deformations along primary collagen fibril directions. J. Biomech. 2010, 43, 783–787. [Google Scholar] [CrossRef]
- Guilak, F.; Erickson, G.R.; Ting-Beall, H.P. The effects of osmotic stress on the viscoelastic and physical properties of articular chondrocytes. Biophys. J. 2002, 82, 720–727. [Google Scholar] [CrossRef]
- Turunen, S.M.; Lammi, M.J.; Saarakkala, S.; Koistinen, A.; Korhonen, R.K. Hypotonic challenge modulates cell volumes differently in the superficial zone of intact articular cartilage and cartilage explants. Biomech. Model. Mechanobiol. 2012, 11, 665–675. [Google Scholar] [CrossRef]
- Kerrigan, M.J.P.; Hook, C.S.V.; Qusous, A.; Hall, A.C. Regulatory volume increase (RVI) by in situ and isolated bovine articular chondrocytes. J. Cell Physiol. 2006, 209, 481–492. [Google Scholar] [CrossRef]
- Glacken, M.W.R.; Fleischaker, J.; Sinskey, A.J. Reduction of waste product excretion via nutrient control: Possible strategies for maximizing product and cell yields on serum in cultures of mammalian cells. Biotechnol. Bioeng. 1986, 28, 1376–1389. [Google Scholar] [CrossRef]
- Lodish, H.; Baltimore, D.; Berk, A.; Zipursky, S.L.; Matsudaira, P.; Darnell, J. Molecular Cell Biology; Scientific American Books: New York, NY, USA, 1995. [Google Scholar]
- Rajpurohit, R.; Koch, C.J.; Tao, Z.; Teixeira, C.M.; Shapiro, I.M. Adaptation of chondrocytes to low oxygen tension: Relationship between hypoxia and cellular metabolism. J. Cell Physiol. 1996, 168, 424–432. [Google Scholar] [CrossRef]
- Lang, F. Mechanisms and significance of cell volume regulation. J. Am. Coll. Nutr. 2007, 26, S613–S623. [Google Scholar]
- McCarty, N.A.; O’Neil, R.G. Calcium signaling in cell volume regulation. Physiol. Rev. 1992, 72, 1037–1061. [Google Scholar]
- Hall, A.C. Volume-sensitive taurine transport in bovine articular chondrocytes. J. Physiol. 1995, 484, 755–766. [Google Scholar]
- Tan, S.C.W.; Pan, W.X.; Ma, G.; Cai, N.; Leong, K.W.; Liao, K. Viscoelastic behaviour of human mesenchymal stem cells. BMC Cell Biol. 2008, 9, 40. [Google Scholar] [CrossRef]
- Hall, A.C.; Bush, P.G. The role of a swelling-activated taurine transport pathway in the regulation of articular chondrocyte volume. Eur. J. Physiol. 2001, 442, 771–781. [Google Scholar] [CrossRef]
- Han, S.-K.; Seerattan, R.; Herzog, W. Mechanical loading of in situ chondrocytes in lapine retropatellar cartilage after anterior cruciate ligament transaction. J. R. Soc. Interface 2010, 7, 895–903. [Google Scholar] [CrossRef]
- Király, K.; Lapveteläinen, T.; Arokoski, J.; Törrönen, K.; Módis, L.; Kiviranta, I; Helminen, H.J. Application of selected cationic dyes for the semiquantitative estimation of glycosaminoglycans in histological sections of articular cartilage by microspectrophotometry. Histochem. J. 1996, 28, 577–590. [Google Scholar] [CrossRef]
- Kiviranta, I.; Jurvelin, J.; Tammi, M.; Säämänen, A.M.; Helminen, H.J. Microspectrophotometric quantitation of glycosaminoglycans in articular cartilage sections stained with Safranin O. Histochemistry 1985, 82, 249–255. [Google Scholar] [CrossRef]
- Kokkonen, H.T.; Mäkelä, J.; Kulmala, K.A.M.; Rieppo, L.; Jurvelin, J.S.; Tiitu, V.; Karjalainen, H.M.; Korhonen, R.K.; Kovanen, V.; Töyräs, J. Computed tomography detects changes in contrast agent diffusion after collagen cross-linking typical to natural aging of articular cartilage. Osteoarthritis Cartilage 2011, 19, 1190–1198. [Google Scholar]
- Oh, T.; Sung, J.H.; Tatosian, D.A.; Shuler, M.L.; Kim, D. Real-time fluorescence detection of multiple microscale cell culture analog devices in situ. Cytometry Part A 2007, 71A, 857–865. [Google Scholar] [CrossRef]
- Alyassin, A.M.; Lancaster, J.L.; Downs, J.H.; Fox, P.T. Evaluation of new algorithms for the interactive measurement of surface area and volume. Med. Eng. Phys. 1994, 21, 741–752. [Google Scholar]
- Brown, H.; Prescott, R. Applied Mixed Models in Medicine, 2nd ed; John Wiley & Sons, Inc.: New York, NY, USA, 2006. [Google Scholar]
- Korhonen, R.K.; Julkunen, P.; Jurvelin, J.S.; Saarakkala, S. Structural and compositonal changes in peri- and extracellular matrix of osteoarthritic cartilage modulate chondrocyte morphology. Cell. Mol. Bioeng. 2011, 4, 484–494. [Google Scholar] [CrossRef]
- Saarakkala, S.; Julkunen, P.; Kiviranta, P.; Mäkitalo, J.; Jurvelin, J.S.; Korhonen, R.K. Depth-wise progression of osteoarthritis in human articular cartilage: Investigation of composition, structure and biomechanics. Osteoarthritis Cartilage 2010, 18, 73–81. [Google Scholar]
- Hall, A.C. Volume regulation by single isolated bovine articular chondrocytes studied by fluorescence imaging. J. Physiol. 1999, 517P, 55–56. [Google Scholar]
- Uesono, Y. Environmental stresses and clinical drugs paralyze a cell. Commun. Integr. Biol. 2009, 2, 275–278. [Google Scholar] [CrossRef]
- Uesono, Y.; Ashe, M.P.; Toh-e, A. Simultaneous yet independent regulation of actin cytoskeletal organization and translation initiation by glucose in Saccharomyces cerevisiae. Mol. Biol. Cell 2004, 15, 1544–1556. [Google Scholar] [CrossRef]
- Obradovic, B.; Carrier, R.L.; Vunjak-Novakovic, G.; Freed, L.E. Gas exchange is essential for bioreactor cultivation of tissue engineered cartilage. Biotechnol. Bioeng. 1999, 63, 197–205. [Google Scholar] [CrossRef]
- Hoffmann, E.K.; Dunham, P.B. Membrane mechanisms and intracellular signaling in cell volume regulation. Int. Rev. Cytol. 1995, 161, 173–260. [Google Scholar] [CrossRef]
- Mow, V.C.; Proctor, C.S.; Kelly, M.A. Biomechanics of articular cartilage. In Basic Biomechanics of the Locomotor System; Nordin, M., Frankel, V.H., Eds.; Lea and Febiger: Philadelphia, PA, USA, 1989; pp. 31–58. [Google Scholar]
- Poole, A.R.; Pidoux, I.; Reiner, A.; Rosenberg, L. An immunoelectron microscope study of the organization of proteoglycan monomer, link protein, and collagen in the matrix of articular cartilage. J. Cell Biol. 1982, 93, 921–937. [Google Scholar] [CrossRef]
- Poole, A.R.; Pidoux, I.; Reiner, A.; Tang, L.H.; Choi, H.; Rosenberg, L. Localization of proteoglycan monomer and link protein in the matrix of bovine articular cartilage: An immunohistochemical study. J. Histochem. Cytochem. 1980, 28, 621–635. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Huttu, M.; Turunen, S.; Sokolinski, V.; Tiitu, V.; Lammi, M.; Korhonen, R.K. Effects of Medium and Temperature on Cellular Responses in the Superficial Zone of Hypo-Osmotically Challenged Articular Cartilage. J. Funct. Biomater. 2012, 3, 544-555. https://doi.org/10.3390/jfb3030544
Huttu M, Turunen S, Sokolinski V, Tiitu V, Lammi M, Korhonen RK. Effects of Medium and Temperature on Cellular Responses in the Superficial Zone of Hypo-Osmotically Challenged Articular Cartilage. Journal of Functional Biomaterials. 2012; 3(3):544-555. https://doi.org/10.3390/jfb3030544
Chicago/Turabian StyleHuttu, Mari, Siru Turunen, Viktoria Sokolinski, Virpi Tiitu, Mikko Lammi, and Rami K. Korhonen. 2012. "Effects of Medium and Temperature on Cellular Responses in the Superficial Zone of Hypo-Osmotically Challenged Articular Cartilage" Journal of Functional Biomaterials 3, no. 3: 544-555. https://doi.org/10.3390/jfb3030544




