Calcium Orthophosphates as Bioceramics: State of the Art
Abstract
:1. Introduction
2. General Knowledge on Biomaterials and Bioceramics
| Ca/P molar ratio | Compound | Formula | Solubility at 25 °C, −log(Ks) | Solubility at 25 °C, g/L | pH stability range in aqueous solutions at 25 °C |
|---|---|---|---|---|---|
| 0.5 | Monocalcium phosphate monohydrate (MCPM) | Ca(H2PO4)2·H2O | 1.14 | ~18 | 0.0–2.0 |
| 0.5 | Monocalcium phosphate anhydrous (MCPA) | Ca(H2PO4)2 | 1.14 | ~17 | [c] |
| 1.0 | Dicalcium phosphate dihydrate (DCPD), mineral brushite | CaHPO4·2H2O | 6.59 | ~0.088 | 2.0–6.0 |
| 1.0 | Dicalcium phosphate anhydrous (DCPA), mineral monetite | CaHPO4 | 6.90 | ~0.048 | [c] |
| 1.33 | Octacalcium phosphate (OCP) | Ca8(HPO4)2(PO4)4·5H2O | 96.6 | ~0.0081 | 5.5–7.0 |
| 1.5 | α-Tricalcium phosphate (α-TCP) | α-Ca3(PO4)2 | 25.5 | ~0.0025 | [a] |
| 1.5 | β-Tricalcium phosphate (β-TCP) | β-Ca3(PO4)2 | 28.9 | ~0.0005 | [a] |
| 1.0–2.2 | Amorphous calcium phosphate (ACP) | CaxHy(PO4)z·nH2O, n = 3–4.5; 15–20% H2O | [b] | [b] | ~5–12 [d] |
| 1.5–1.67 | Calcium-deficient hydroxyapatite (CDHA)[e] | Ca10-x(HPO4)x(PO4)6-x(OH)2-x[f] (0 < x < 1) | ~85.1 | ~0.0094 | 6.5–9.5 |
| 1.67 | Hydroxyapatite (HA, HAp or OHAp) | Ca10(PO4)6(OH)2 | 116.8 | ~0.0003 | 9.5–12 |
| 1.67 | Fluorapatite (FA or FAp) | Ca10(PO4)6F2 | 120.0 | ~0.0002 | 7–12 |
| 1.67 | Oxyapatite (OA or OAp) | Ca10(PO4)6O | ~69 | ~0.087 | [a] |
| 2.0 | Tetracalcium phosphate (TTCP or TetCP), mineral hilgenstockite | Ca4(PO4)2O | 38–44 | ~0.0007 | [a] |
3. General Knowledge on Calcium Orthophosphates
4. Bioceramics of Calcium Orthophosphates
4.1. History
4.2. Chemical Composition and Preparation
4.3. Forming and Shaping
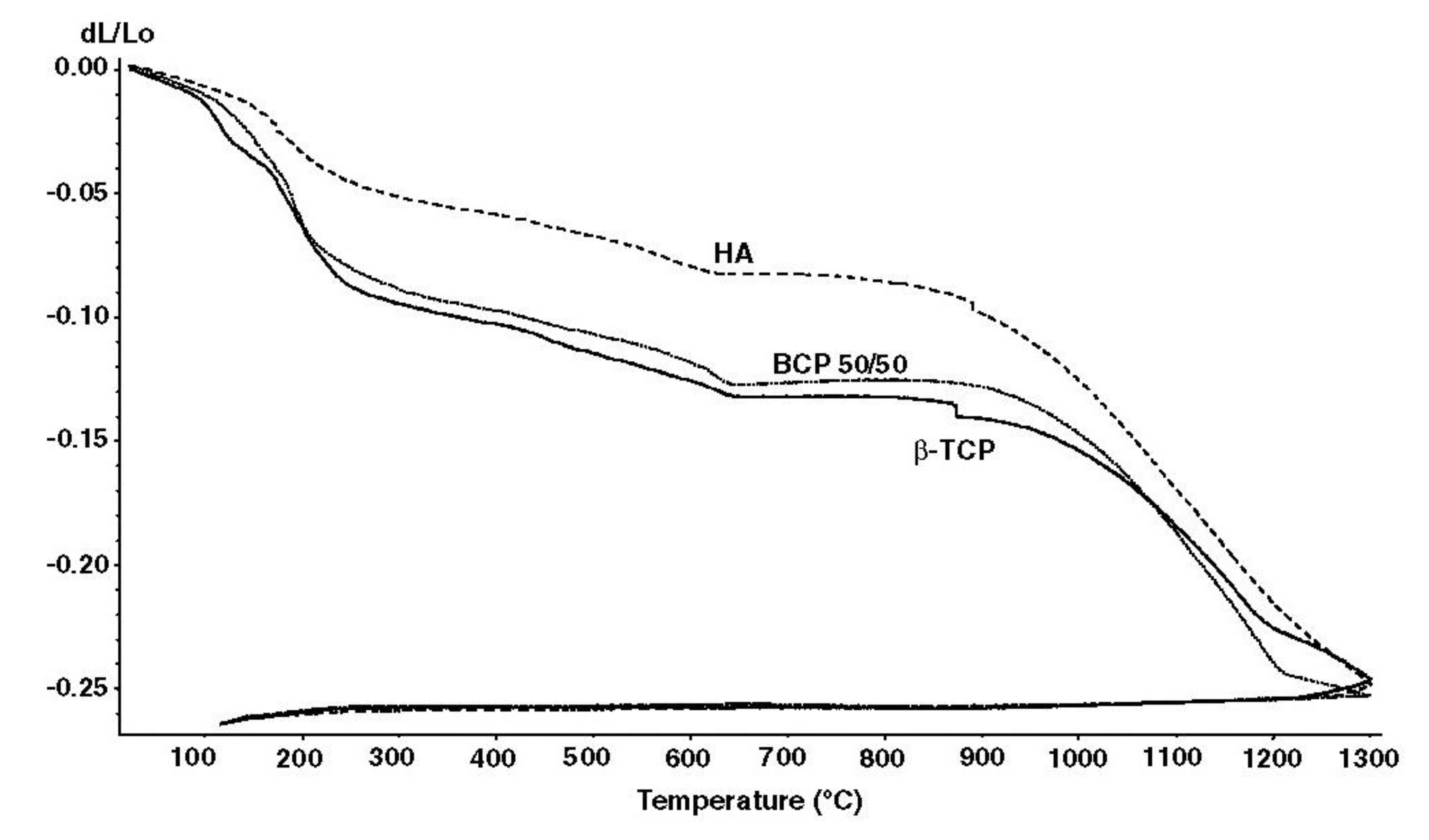
4.4. Sintering and Firing
5. The Major Properties
5.1. Mechanical Properties
5.2. Electrical Properties
5.3. Possible Transparency
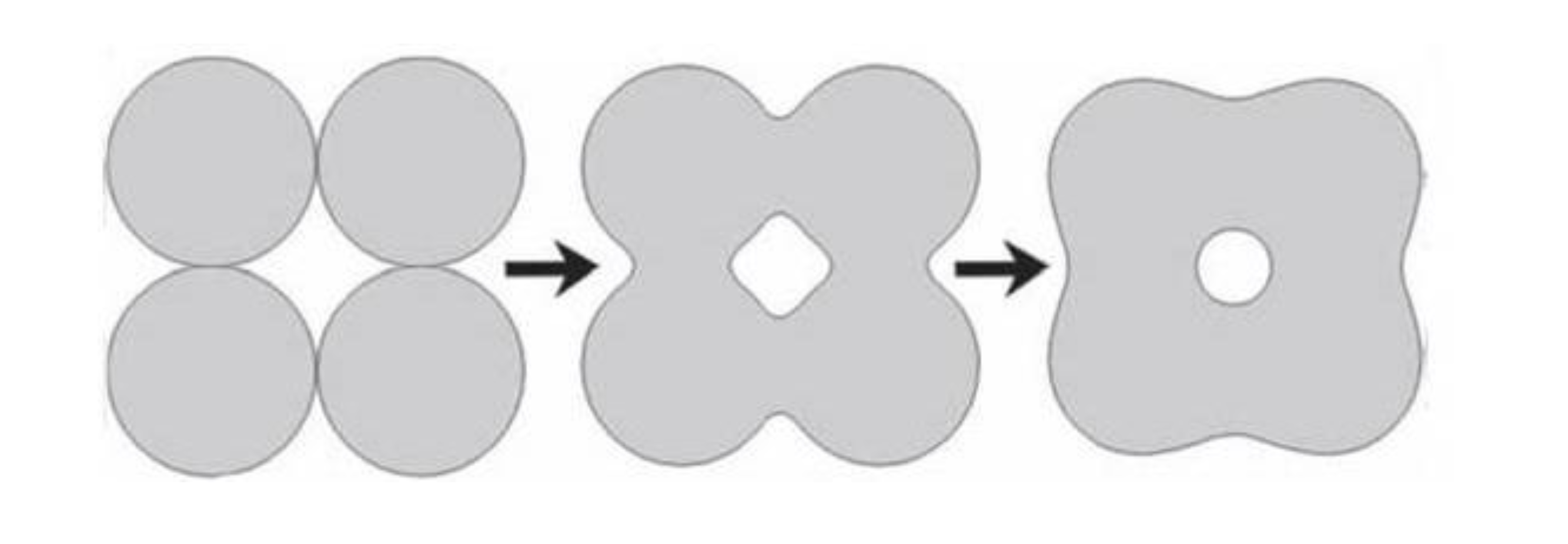
5.4. Porosity


| Year | Who and where | Process | Calcium orthophosphate | Sintering | Compressive strength | Pore sizes | Porosity |
|---|---|---|---|---|---|---|---|
| 2006 | Deville et al. Berkeley, CA | HA + ammonium methacrylate in PTFE mold, freeze dried and sintered. | HA | Yes: 1300 °C | 16 MPa, 65 MPa, 145 MPa | open unidirectional 50–150 μm | >60%, 56%, 47% |
| 2006 | Saiz et al. Berkeley, CA | Polymer foams coated, compressed after infiltration, then calcined. | HA powder | Yes: 700–1300 °C | – | 100–200 μm | – |
| 2006 | Murugan et al. Singapore + USA | Bovine bone cleaned, calcined. | Bovine bone | Yes: 500 °C | – | retention of nanopores | – |
| 2006 | Xu et al. Gaithersburg, MD | Directly injectable calcium orthophosphate cement, self hardens, mannitol as porogen. | Nanocrystalline HA | No | 2.2–4.2 MPa (flexural) | 0–50% macroporous | 65–82% |
| 2004 | Landi et al. Italy + Indonesia | Sponge impregnation, isotactic pressing, sintering of HA in simulated body fluid. | Calcium hydroxide + orthophosphoric acid | Yes: 1250 °C for 1 hr | 23 ± 3.8 MPa | closed 6%, open 60% | 66% |
| 2003 | Charriere et al. EPFL, Switzerland | Thermoplastic negative porosity by ink jet printing, slip casting process for HA | DCPD + Calcite | No: 90 °C for 1 day | 12.5 ± 4.6 MPa | – | 44% |
| 2003 | Almirall et al. Barcelona, Spain | α-TCP foamed with hydrogen peroxide at different conc., liq. ratios, poured in PTFE molds. | α-TCP + (10% and 20% peroxide) | No: 60 °C for 2 hr | 1.41 ± 0.27 MPa 2.69 ± 0.91 MPa | 35.7% macro 29.7% micro 26.8% macro 33.8% micro | 65.5% 60.7% |
| 2003 | Ramay et al. Seattle, WA | Slurries of HA prepared: gel-casting + polymer sponge technique. Sintered. | HA powder | Yes: 600 °C for 1 hr, 1350 °C for 2 hr | 0.5–5 MPa | 200–400 μm | 70–77% |
| 2003 | Miao et al. Singapore | TTCP to calcium orthophosphate cement. Slurry cast on polymer foam, sintered. | TTCP | Yes: 1200 °C for 2 hr | – | 1 mm macro, 5 μm micro | ~70% |
| 2003 | Uemura et al. China + Japan | Slurry of HA with polyoxyethylenelaurylether (crosslinked) and sintered. | HA powders | Yes: 1200 °C for 3 hr | 2.25 MPa (0 wk) 4.92 MPa (12 wks) 11.2 MPa (24 wks) | 500 micron 200 μm interconnects | ~77% |
| 2003 | Ma et al. Singapore + USA | Electrophoretic deposition of HA, sintering. | HA powders | Yes: 1200 °C for 2 hr | 860 MPa | 0.5 μm, 130 μm | ~20% |
| 2002 | Barralet et al. Birmingham, London | Calcium orthophosphate cement + sodium orthophosphate ice: evaporated. | Calcium carbonate + DCDP | 1st step: 1400 °C for 1 day | 0.6 ± 0.27 MPa | 2 μm | 62 ± 9% |

6. Biomedical Applications
| Calcium orthophosphate | Trade name and producer | ||
|---|---|---|---|
| CDHA | Cementek (Teknimed, France) | ||
| Osteogen (Impladent, NY, USA) | |||
| HA | Actifuse (ApaTech, UK) | ||
| Apaceram (Pentax, Japan) | |||
| ApaPore (ApaTech, UK) | |||
| Bioroc (Depuy-Bioland, France) | |||
| Bonefil (Pentax, Japan) | |||
| Bonetite (Pentax, Japan) | |||
| Boneceram (Sumitomo Osaka Cement, Japan) | |||
| BoneSource (Stryker Orthopaedics, NJ, USA) | |||
| Calcitite (Zimmer, IN, USA) | |||
| Cerapatite (Ceraver, France) | |||
| Neobone (Toshiba Ceramics, Japan) | |||
| Ostegraf (Ceramed, CO, USA) | |||
| Ostim (Heraeus Kulzer, Germany) | |||
| Synatite (SBM, France) | |||
| HA/collagen | Bioimplant (Connectbiopharm, Russia) | ||
| Bonject (Koken, Japan) | |||
| CollapAn (Intermedapatite, Russia) | |||
| HAPCOL (Polystom, Russia) | |||
| LitAr (LitAr, Russia) | |||
| HA/sodium alginate | Bialgin (Biomed, Russia) | ||
| HA/Poly-L-Lactic Acid | SuperFIXSORB30 (Takiron, Japan) | ||
| HA/polyethylene | HAPEX (Gyrus, TN, USA) | ||
| HA/CaSO4 | Hapset (LifeCore, MIN, USA) | ||
| coralline HA | Interpore (Interpore, CA, USA) | ||
| ProOsteon (Interpore, CA, USA) | |||
| algae-derived HA | Algipore (Dentsply Friadent, Germany) | ||
| bovine bone apatite (unsintered) | BioOss (Geitslich, Switzerland) | ||
| Laddec (Ost-Developpement, France) | |||
| Lubboc (Ost-Developpement, France) | |||
| Oxbone (Bioland biomateriaux, France) | |||
| Tutoplast (IOP, CA, USA) | |||
| bovine bone apatite (sintered) | BonAP | ||
| Cerabone (aap Implantate, Germany) | |||
| Endobon (Merck, Germany) | |||
| Osteograf (Ceramed, CO, USA) | |||
| PepGen P-15 (Dentsply Friadent, Germany) | |||
| β-TCP | Bioresorb (Sybron Implant Solutions, Germany) | ||
| Biosorb (SBM S.A., France) | |||
| Calciresorb (Ceraver, France) | |||
| Cerasorb (Curasan, Germany) | |||
| Ceros (Thommen Medical, Switzerland) | |||
| ChronOS (Synthes, PA, USA) | |||
| Conduit (DePuy Spine, USA) | |||
| JAX (Smith and Nephew Orthopaedics, USA) | |||
| Osferion (Olympus Terumo Biomaterials, Japan) | |||
| OsSatura TCP (Integra Orthobiologics, CA, USA) | |||
| Vitoss (Orthovita, PA, USA) | |||
| BCP (HA + β-TCP) | 4Bone (MIS, Israel) | ||
| BCP (Medtronic, MN, USA) | |||
| Biosel (Depuy Bioland, France) | |||
| BoneSave (Stryker Orthopaedics, NJ, USA) | |||
| Calciresorb (Ceraver, France) | |||
| CellCeram (Scaffdex, Finland) | |||
| Ceraform (Teknimed, France) | |||
| Ceratite (NGK Spark Plug, Japan) | |||
| Eurocer (FH Orthopedics, France) | |||
| Graftys BCP (Graftys, France) | |||
| Hatric (Arthrex, Naples, FL, USA) | |||
| Indost (Polystom, Russia) | |||
| Kainos (Signus, Germany) | |||
| MBCP (Biomatlante, France) | |||
| OptiMX (Exactech, USA) | |||
| OsSatura BCP (Integra Orthobiologics, CA, USA) | |||
| Osteosynt (Einco, Brazil) | |||
| SBS (Expanscience, France) | |||
| TCH (Kasios, France) | |||
| Triosite (Zimmer, IN, USA) | |||
| Tribone (Stryker, Europe) | |||
| BCP (HA + α-TCP) | Skelite (Millennium Biologix, ON, Canada) | ||
| BCP/collagen | Allograft (Zimmer, IN, USA) | ||
| BCP/fibrin | TricOS (Baxter BioScience, France) | ||
| BCP/silicon | FlexHA (Xomed, FL, USA) | ||
| FA + BCP (HA + β-TCP) | FtAP (Polystom, Russia) | ||
| carbonateapatite | Healos (Orquest, CA, USA) |
6.1. Cements and Concretes
| Form | Defect form | Mechanical stability | Resorption/bone formation | Handling |
|---|---|---|---|---|
| Granules (0.1–5 mm in diameter) | Open | Negligible | Throughout the defect | Fair (granule migration during and after surgery) |
| Macroporous blocks | Open and defined shape | Fair provided there is press-fitting into the defect | Throughout the defect | Very good (problems might arise to fit the block within the defect) |
| Cement paste | Closed | Fair | Peripheral | Fair to good (the paste might set too fast or might be poorly injectable) |
| Putty | Open or closed | Negligible | Peripheral or throughout the defect depending on the composition | Very good for pastes that have to be mixed in the operating room to excellent for ready-mixed pastes (the paste might be poorly-injectable) |

6.2. Coatings
| Technique | Thickness | Advantages | Disadvantages |
|---|---|---|---|
| Thermal spraying | 30–200 μm | High deposition rates; low cost | Line of sight technique; high temperatures induce decomposition; rapid cooling produces amorphous coatings |
| Sputter coating | 0.5–3 μm | Uniform coating thickness on flat substrates; dense coating | Line of sight technique; expensive; time consuming; produces amorphous coatings |
| Pulsed laser deposition | 0.05–5 μm | Coating by crystalline and amorphous phases; dense and porous coating | Line of sight technique |
| Dynamic mixing method | 0.05–1.3 μm | High adhesive strength | Line of sight technique; expensive; produces amorphous coatings |
| Dip coating | 0.05–0.5 mm | Inexpensive; coatings applied quickly; can coat complex substrates | Requires high sintering temperatures; thermal expansion mismatch |
| Sol-gel technique | <1 μm | Can coat complex shapes; low processing temperatures; relatively cheap as coatings are very thin | Some processes require controlled atmosphere processing; expensive raw materials |
| Electrophoreticdeposition | 0.1–2.0 mm | Uniform coating thickness; rapid deposition rates; can coat complex substrates | Difficult to produce crack-free coatings; requires high sintering temperatures |
| Biomimetic coating | <30 μm | Low processing temperatures; can form bonelike apatite; can coat complex shapes; can incorporate bone growth stimulating factors | Time consuming; requires replenishment and a pH constancy of simulated body fluid |
| Hot isostatic pressing | 0.2–2.0 μm | Produces dense coatings | Cannot coat complex substrates; high temperature required; thermal expansion mismatch; elastic property differences; expensive; removal/interaction of encapsulation material |
| Electrochemical deposition | 0.05–0.5 mm | Uniform coating thickness; rapid deposition rates; can coat complex substrates; moderate temperature, low cost | Thecoating/substrate bonding is not strong enough |

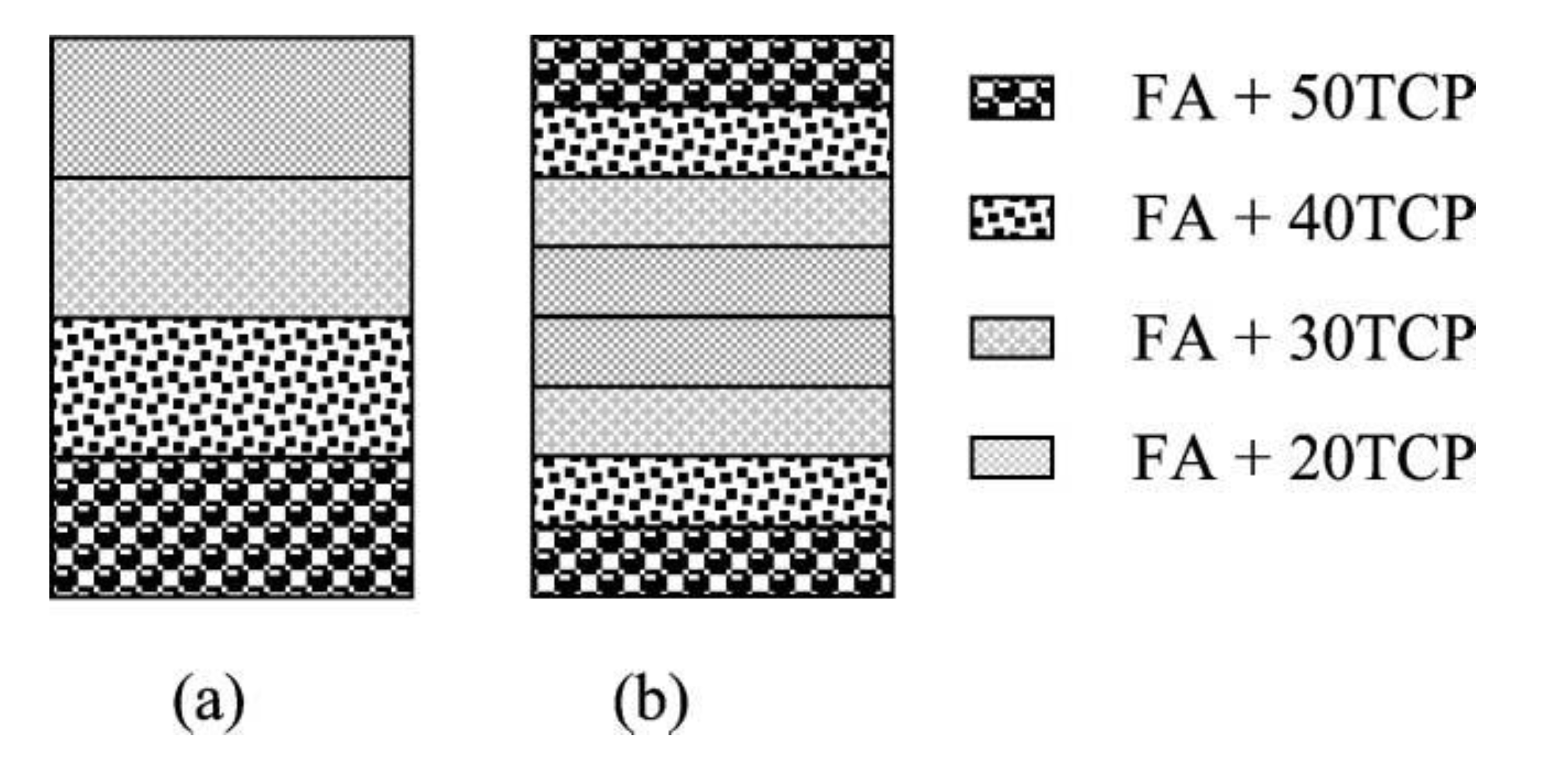
6.3. Functionally Graded Bioceramics
7. Biological Properties and In Vivo Behavior
7.1. Interaction with the Surrounding Tissues and the Host Responses
7.2. Osteoinduction
7.3. Biodegradation
7.4. Bioactivity


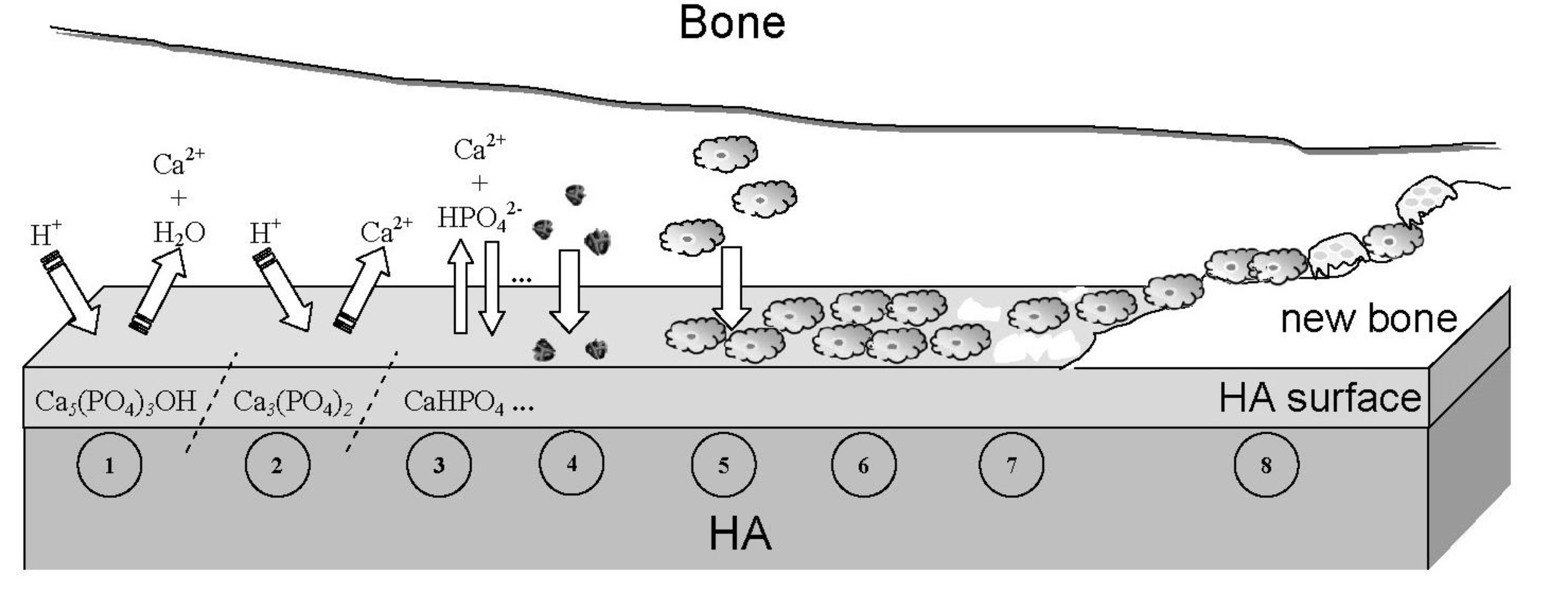
7.5. Cellular Response
8. Calcium Orthophosphate Bioceramics in Tissue Engineering
8.1. Tissue Engineering
8.2. Scaffolds and Their Properties
| Pore sizes of a 3D scaffold | Biochemical effect or function |
|---|---|
| <1 μm | Interaction with proteins |
| Responsible for bioactivity | |
| 1–20 μm | Type of cells attracted |
| Cellular development | |
| Orientation and directionality of cellular ingrowth | |
| 100–1000 μm | Cellular growth |
| Bone ingrowth | |
| Predominant function in the mechanical strength | |
| >1000 μm | Implant functionality |
| Implant shape | |
| Implant esthetics |
8.3. Scaffolds from Calcium Orthophosphate Bioceramics
8.4. A Clinical Experience
9. Conclusions and Outlook
- Improvement of the mechanical performance of existing types of bioceramics;
- Enhanced bioactivity in terms of gene activation;
- Improvement in the performance of biomedical coatings in terms of their mechanical stability and ability to deliver biological agents;
- Development of smart biomaterials capable of combining sensing with bioactivity;
- Development of improved biomimetic composites.
Acknowledgements
References and Notes
- Bohner, M. Resorbable biomaterials as bone graft substitutes. Mater. Today 2010, 13, 24–30. [Google Scholar] [CrossRef]
- Doremus, R.H. Bioceramics. J. Mater. Sci. 1992, 27, 285–297. [Google Scholar] [CrossRef]
- Vallet-Regí, M. Ceramics for medical applications. J. Chem. Soc. Dalton Trans. 2001, 97–108. [Google Scholar] [CrossRef]
- Best, S.M.; Porter, A.E.; Thian, E.S.; Huang, J. Bioceramics: Past, present and for the future. J. Eur. Ceram. Soc. 2008, 28, 1319–1327. [Google Scholar] [CrossRef]
- Vallet-Regí, M. Evolution of bioceramics within the field of biomaterials. C. R. Chimie 2010, 13, 174–185. [Google Scholar] [CrossRef]
- Rahaman, M.N.; Yao, A.; Bal, B.S.; Garino, J.P.; Ries, M.D. Ceramics for prosthetic hip and knee joint replacement. J. Am. Ceram. Soc. 2007, 90, 1965–1988. [Google Scholar] [CrossRef]
- Langstaff, S.D.; Sayer, M.; Smith, T.J.N.; Pugh, S.M.; Hesp, S.A.M.; Thompson, W.T. Resorbable bioceramics based on stabilized calcium phosphates. Part I: Rational design, sample preparation and material characterization. Biomaterials 1999, 20, 1727–1741. [Google Scholar] [CrossRef]
- Langstaff, S.D.; Sayer, M.; Smith, T.J.N.; Pugh, S.M. Resorbable bioceramics based on stabilized calcium phosphates. Part II: Evaluation of biological response. Biomaterials 2001, 22, 135–150. [Google Scholar] [CrossRef]
- Sayer, M.; Stratilatov, A.D.; Reid, J.W.; Calderin, L.; Stott, M.J.; Yin, X.; MacKenzie, M.; Smith, T.J.N.; Hendry, J.A.; Langstaff, S.D. Structure and composition of silicon-stabilized tricalcium phosphate. Biomaterials 2003, 24, 369–382. [Google Scholar] [CrossRef]
- Reid, J.W.; Pietak, A.M.; Sayer, M.; Dunfield, D.; Smith, T.J.N. Phase formation and evolution in the silicon substituted tricalcium phosphate/apatite system. Biomaterials 2005, 26, 2887–2897. [Google Scholar] [CrossRef]
- Sanchez-Sálcedo, S.; Arcos, D.; Vallet-Regí, M. Upgrading calcium phosphate scaffolds for tissue engineering applications. Key Eng. Mater. 2008, 377, 19–42. [Google Scholar] [CrossRef]
- Ito, A.; LeGeros, R.Z. Magnesium- and zinc-substituted beta-tricalcium phosphates as potential bone substitute biomaterials. Key Eng. Mater. 2008, 377, 85–98. [Google Scholar] [CrossRef]
- Ergun, C.; Webster, T.J.; Bizios, R.; Doremus, R.H. Hydroxylapatite with substituted magnesium, zinc, cadmium, and yttrium. I. Structure and microstructure. J. Biomed. Mater. Res. 2002, 59, 305–311. [Google Scholar] [CrossRef]
- Webster, T.J.; Ergun, C.; Doremus, R.H.; Bizios, R. Hydroxylapatite with substituted magnesium, zinc, cadmium, and yttrium. II. Mechanisms of osteoblast adhesion. J. Biomed. Mater. Res. 2002, 59, 312–317. [Google Scholar] [CrossRef]
- Kim, S.R.; Lee, J.H.; Kim, Y.T.; Riu, D.H.; Jung, S.J.; Lee, Y.J.; Chung, S.C.; Kim, Y.H. Synthesis of Si, Mg substituted hydroxyapatites and their sintering behaviors. Biomaterials 2003, 24, 1389–1398. [Google Scholar] [CrossRef]
- Patel, N.; Best, S.M.; Bonfield, W. Characterisation of hydroxyapatite and substituted-hydroxyapatites for bone grafting. J. Australas. Ceram. Soc. 2005, 41, 1–22. [Google Scholar]
- Vallet-Regí, M.; Arcos, D. Silicon substituted hydroxyapatites. A method to upgrade calcium phosphate based implants. J. Mater. Chem. 2005, 15, 1509–1516. [Google Scholar] [CrossRef]
- Landi, E.; Celotti, G.; Logroscino, G.; Tampieri, A. Carbonated hydroxyapatite as bone substitute. J. Eur. Ceram. Soc. 2003, 23, 2931–2937. [Google Scholar] [CrossRef]
- Gbureck, U.; Thull, R.; Barralet, J.E. Alkali ion substituted calcium phosphate cement formation from mechanically activated reactants. J. Mater. Sci. Mater. Med. 2005, 16, 423–427. [Google Scholar] [CrossRef]
- Kannan, S.; Ventura, J.M.; Ferreira, J.M.F. In situ formation and characterization of flourine-substituted biphasic calcium phosphate ceramics of varied F-HAP / β-TCP ratios. Chem. Mater. 2005, 17, 3065–3068. [Google Scholar] [CrossRef]
- Tas, A.C.; Bhaduri, S.B.; Jalota, S. reparation of Zn-doped β-tricalcium phosphate (β-Ca3(PO4)2) bioceramics. Mater. Sci. Eng. C 2007, 27, 394–401. [Google Scholar] [CrossRef]
- Gbureck, U.; Knappe, O.; Grover, L.M.; Barralet, J.E. Antimicrobial potency of alkali ion substituted calcium phosphate cements. Biomaterials 2005, 26, 6880–6886. [Google Scholar] [CrossRef]
- Tardei, C.; Grigore, F.; Pasuk, I.; Stoleriu, S. The study of Mg2+/Ca2+ substitution of β-tricalcium phosphate. J. Optoelectron. Adv. Mater. 2006, 8, 568–571. [Google Scholar]
- Pietak, A.M.; Reid, J.W.; Stott, M.J.; Sayer, M. Silicon substitution in the calcium phosphate bioceramics. Biomaterials 2007, 28, 4023–4032. [Google Scholar] [CrossRef]
- Yao, Z.P.; Liu, W.G.; Ni, G.X. Biology characteristics and clinical application of strontium substituted hydroxyapatite bone cement. J. Clin. Rehabil. Tissue Eng. Res. 2008, 12, 7151–7154. [Google Scholar]
- Landi, E.; Tampieri, A.; Mattioli-Belmonte, M.; Celotti, G.; Sandri, M.; Gigante, A.; Fava, P.; Biagini, G. Biomimetic Mg- and Mg,CO3-substituted hydroxyapatites: Synthesis characterization and in vitro behaviour. J. Eur. Ceram. Soc. 2006, 26, 2593–2601. [Google Scholar] [CrossRef]
- Landi, E.; Tampieri, A.; Celotti, G.; Sprio, S.; Sandri, M.; Logroscino, G. Sr-substituted hydroxyapatites for osteoporotic bone replacement. Acta Biomater. 2007, 3, 961–969. [Google Scholar] [CrossRef]
- Kannan, S.; Ventura, J.M.G.; Ferreira, J.M.F. Synthesis and thermal stability of potassium substituted hydroxyapatites and hydroxyapatite/β-tricalcium phosphate mixtures. Ceram. Int. 2007, 33, 1489–1494. [Google Scholar] [CrossRef]
- Kannan, S.; Rebelo, A.; Lemos, A.F.; Barba, A.; Ferreira, J.M.F. Synthesis and mechanical behaviour of chlorapatite and chlorapatite/β-TCP composites. J. Eur. Ceram. Soc. 2007, 27, 2287–2294. [Google Scholar] [CrossRef]
- Kannan, S.; Goetz-Neunhoeffer, F.; Neubauer, J.; Ferreira, J.M.F. Ionic substitutions in biphasic hydroxyapatite and β-tricalcium phosphate mixtures: Structural analysis by Rietveld refinement. J. Am. Ceram. Soc. 2008, 91, 1–12. [Google Scholar]
- Lafon, J.P.; Champion, E.; Bernache-Assollant, D. Processing of AB-type carbonated hydroxyapatite Ca10−x(PO4)6−x(CO3)x(OH)2−x−2y(CO3)y ceramics with controlled composition. J. Eur. Ceram. Soc. 2008, 28, 139–147. [Google Scholar] [CrossRef]
- Ren, F.; Xin, R.; Ge, X.; Leng, Y. An experimental and computational study on Zn-substituted hydroxyapatite. Adv. Mater. Res. 2008, 47-50, 1379–1382. [Google Scholar] [CrossRef]
- Meejoo, S.; Pon-On, W.; Charnchai, S.; Amornsakchai, T. Substitution of iron in preparation of enhanced thermal property and bioactivity of hydroxyapatite. Adv. Mater. Res. 2008, 55-57, 689–692. [Google Scholar] [CrossRef]
- Kannan, S.; Goetz-Neunhoeffer, F.; Neubauer, J.; Ferreira, J.M.F. Synthesis and structure refinement of zinc-doped β-tricalcium phosphate powders. J. Am. Ceram. Soc. 2009, 92, 1592–1595. [Google Scholar]
- Natasha, A.N.; Sopyan, I.; Zuraida, A. Fourier transform infrared study on sol-gel derived manganese-doped hydroxyapatite. Adv. Mater. Res. 2008, 47-50, 1185–1188. [Google Scholar] [CrossRef]
- Matsumoto, N.; Yoshida, K.; Hashimoto, K.; Toda, Y. Thermal stability of β-tricalcium phosphate doped with monovalent metal ions. Mater. Res. Bull. 2009, 44, 1889–1894. [Google Scholar] [CrossRef]
- Boanini, E.; Gazzano, M.; Bigi, A. Ionic substitutions in calcium phosphates synthesized at low temperature. Acta Biomater. 2010, 6, 1882–1894. [Google Scholar] [CrossRef]
- Williams, D.F. The Williams Dictionary of Biomaterials; Liverpool University Press: Liverpool, UK, 1999; p. 368. [Google Scholar]
- Williams, D.F. On the nature of biomaterials. Biomaterials 2009, 30, 5897–5909. [Google Scholar] [CrossRef]
- Jandt, K.D. Evolutions, revolutions and trends in biomaterials science—A perspective. Adv. Eng. Mater. 2007, 9, 1035–1050. [Google Scholar] [CrossRef]
- Biomimetic Materials Chemistry; Mann, S. (Ed.) Wiley-VCH: Weinheim, Germany, 1996; p. 400.
- Vallet-Regí, M. Bioceramics: Where do we come from and which are the future expectations. Key Eng. Mater. 2008, 377, 1–18. [Google Scholar] [CrossRef]
- Meyers, M.A.; Chen, P.Y.; Lin, A.Y.M.; Seki, Y. Biological materials: Structure and mechanical properties. Prog. Mater. Sci. 2008, 53, 1–206. [Google Scholar] [CrossRef]
- According to Wikipedia, the free encyclopedia: “The word ceramic comes from the Greek word κεραμικός (keramikos) meaning pottery, which is said to derive from the Indo-European word ker, meaning heat. A ceramic is an inorganic, non-metallic solid prepared by the action of heat and subsequent cooling. Ceramic materials may have a crystalline or partly crystalline structure, or may be amorphous (e.g., a glass). Because most common ceramics are crystalline, the definition of ceramic is often restricted to inorganic crystalline materials, as opposed to the non-crystalline glasses. Ceramic may be used as an adjective describing a material, product or process; or as a singular noun, or, more commonly, as a plural noun, ceramics.”. Available online: http://en.wikipedia.org/wiki/Ceramics (accessed on 11 November 2010).
- Biocompatibility is the ability of a material to perform with an appropriate host response in a specific application [46-48]
- Hench, L.L. Bioceramics: From concept to clinic. J. Am. Ceram. Soc. 1991, 74, 1487–1510. [Google Scholar] [CrossRef]
- Cao, W.; Hench, L.L. Bioactive materials. Ceram. Int. 1996, 22, 493–507. [Google Scholar] [CrossRef]
- Hench, L.L. Bioceramics. J. Am. Ceram. Soc. 1998, 81, 1705–1728. [Google Scholar] [CrossRef]
- Pinchuk, N.D.; Ivanchenko, L.A. Making calcium phosphate biomaterials. Powder Metall. Metal Ceram. 2003, 42, 357–371. [Google Scholar] [CrossRef]
- Heimann, R.B. Materials science of crystalline bioceramics: A review of basic properties and applications. CMU J. 2002, 1, 23–46. [Google Scholar]
- Dorozhkin, S.V. Bioceramics based on calcium orthophosphates. Glass Ceram. 2007, 64, 442–447. [Google Scholar] [CrossRef]
- Salinas, A.J.; Vallet-Regí, M. Evolution of ceramics with medical applications. Z. Anorg. Allg. Chem. 2007, 633, 1762–1773. [Google Scholar] [CrossRef]
- Tomoda, K.; Ariizumi, H.; Nakaji, T.; Makino, K. Hydroxyapatite particles as drug carriers for proteins. Colloids Surf. B Biointerfaces 2010, 76, 226–235. [Google Scholar] [CrossRef]
- Ducheyne, P.; Qiu, Q. Bioactive ceramics: The effect of surface reactivity on bone formation and bone cell function. Biomaterials 1999, 20, 2287–2303. [Google Scholar] [CrossRef]
- Lowenstam, H.A.; Weiner, S. On Biomineralization; Oxford University Press: Oxford, UK, 1989; p. 324. [Google Scholar]
- Weiner, S.; Wagner, H.D. Material bone: Structure-mechanical function relations. Ann. Rev. Mater. Sci. 1998, 28, 271–298. [Google Scholar] [CrossRef]
- Dorozhkin, S.V. Calcium orthophosphates. J. Mater. Sci. 2007, 42, 1061–1095. [Google Scholar] [CrossRef]
- Dorozhkin, S.V. Calcium orthophosphates in nature, biology and medicine. Materials 2009, 2, 399–498. [Google Scholar] [CrossRef]
- Bioactivity is defined as the property of the material to develop a direct, adherent and strong bonding with bone [46-48]
- Ong, J.L.; Chan, D.C.N. Hydroxyapatite and their use as coatings in dental implants: A review. Crit. Rev. Biomed. Eng. 1999, 28, 667–707. [Google Scholar]
- Davies, J.E. In vitro modeling of the bone/implant interface. Anat. Rec. 1996, 245, 426–445. [Google Scholar] [CrossRef]
- Anselme, K. Osteoblast adhesion on biomaterials. Biomaterials 2000, 21, 667–681. [Google Scholar] [CrossRef]
- Suchanek, W.L.; Yoshimura, M. Processing and properties of hydroxyapatite-based biomaterials for use as hard tissue replacement implants. J. Mater. Res. 1998, 13, 94–117. [Google Scholar] [CrossRef]
- Gauthier, O.; Bouler, J.M.; Weiss, P.; Bosco, J.; Daculsi, G.; Aguado, E. Kinetic study of bone ingrowth and ceramic resorption associated with the implantation of different injectable calcium-phosphate bone substitutes. J. Biomed. Mater. Res. 1999, 47, 28–35. [Google Scholar] [CrossRef]
- Hing, K.A.; Best, S.M.; Bonfield, W. Characterization of porous hydroxyapatite. J. Mater. Sci. Mater. Med. 1999, 10, 135–145. [Google Scholar] [CrossRef]
- Carotenuto, G.; Spagnuolo, G.; Ambrosio, L.; Nicolais, L. Macroporous hydroxyapatite as alloplastic material for dental applications. J. Mater. Sci. Mater. Med. 1999, 10, 671–676. [Google Scholar] [CrossRef]
- Bioceramics of Calcium Phosphate; De Groot, K. (Ed.) CRC Press: Boca Raton, FL, USA, 1983; p. 146.
- Narasaraju, T.S.B.; Phebe, D.E. Some physico-chemical aspects of hydroxylapatite. J. Mater. Sci. 1996, 31, 1–21. [Google Scholar] [CrossRef]
- LeGeros, R.Z. Calcium Phosphates in Oral Biology and Medicine; Karger: Basel, Switzerland, 1991; p. 201. [Google Scholar]
- Elliott, J.C. Structure and chemistry of the apatites and other calcium orthophosphates. In Studies in Inorganic Chemistry; Elsevier: Amsterdam, The Netherlands, 1994; Volume 18, p. 389. [Google Scholar]
- Hydroxyapatite and Related Materials; Brown, P.W.; Constantz, B. (Eds.) CRC Press: Boca Raton, FL, USA, 1994; p. 343.
- Calcium Phosphates in Biological and Industrial Systems; Amjad, Z. (Ed.) Kluwer Academic Publishers: Boston, MA, USA, 1997; p. 529.
- Phosphates: Geochemical, Geobiological and Materials Importance; Hughes, J.M.; Kohn, M.; Rakovan, J. (Eds.) Mineralogical Society of America: Washington, DC, USA, 2002; p. 742.
- Octacalcium phosphate. In Monographs in Oral Science; Chow, L.C.; Eanes, E.D. (Eds.) S. Karger AG: Basel, Switzerland, 2001; Vol. 18, p. 168.
- Les Matériaux en Phosphate de Calcium. Aspects fondamentaux/Calcium Phosphate Materials. Fundamentals; Brès, E.; Hardouin, P. (Eds.) Sauramps Medical: Montpellier, France, 1998; p. 176.
- Huebsch, N.; Mooney, D.J. Inspiration and application in the evolution of biomaterials. Nature 2009, 462, 426–432. [Google Scholar] [CrossRef]
- Ring, M.E. Dentistry: An Illustrated History; Harry, N., Ed.; Abrams: New York, NY, USA, 1992; p. 320. [Google Scholar]
- Bobbio, A. The first endosseous alloplastic implant in the history of man. Bull. Hist. Dent. 1970, 20, 1–6. [Google Scholar]
- Weiner, S.; Dove, P.M. An overview of biomineralization processes and the problem of the vital effect. In Biomineralization, Series: Reviews in Mineralogy and Geochemistry; Dove, P.M., De Yoreo, J.J., Weiner, S., Eds.; Mineralogical Society of America: Washington, DC, USA, 2003; Volume 54, pp. 1–29. [Google Scholar]
- Weiner, S.; Traub, W.; Wagner, H.D. Lamellar bone: Structure-function relations. J. Struct. Biol. 1999, 126, 241–255. [Google Scholar] [CrossRef]
- Albee, F.H. Studies in bone growth: Triple calcium phosphate as a stimulus to osteogenesis. Ann. Surg. 1920, 71, 32–39. [Google Scholar] [CrossRef]
- Ray, R.D.; Ward, A.A., Jr. A preliminary report on studies of basic calcium phosphate in bone replacement. Surg. Form. 1951, 3, 429–434. [Google Scholar]
- Nery, E.B.; Lynch, K.L.; Hirthe, W.M.; Mueller, K.H. Bioceramic implants in surgically produced infrabony defects. J. Periodontol. 1975, 46, 328–347. [Google Scholar] [CrossRef]
- Denissen, H.W.; De Groot, K. Immediate dental root implants from synthetic dense calcium hydroxylapatite. J. Prosthet. Dent. 1979, 42, 551–556. [Google Scholar] [CrossRef]
- Blakeslee, K.C.; Condrate, R.A., Sr. Vibrational spectra of hydrothermally prepared hydroxyapatites. J. Am. Ceram. Soc. 1971, 54, 559–563. [Google Scholar] [CrossRef]
- Garrington, G.E.; Lightbody, P.M. Bioceramics and dentistry. J. Biomed. Mater. Res. 1972, 6, 333–343. [Google Scholar] [CrossRef]
- Cini, L.; Sandrolini, S.; Paltrinieri, M.; Pizzoferrato, A.; Trentani, C. Materiali bioceramici in funzione sostitutiva. Nota preventiva. (Bioceramic materials for replacement purposes. Preliminary note). Chir. Organi Mov. 1972, 60, 423–430. [Google Scholar]
- Rivault, M.A. Evolution, conception et technologie des travaux de prothèse fixe, réalisés en céramo-métallique. (Evolution, conception and technology of fixed prosthesis made of ceramic and metal). Rev. Fr. Odontostomatol. 1966, 13, 1367–1402. [Google Scholar]
- Dumont, A.; Appel, M.; Favard, E. Prothèses plurales en céramique sur métal. Soudage et artifices de jonction. (Multiple prostheses made of ceramics on metal. Soldering and artifacts of the junction). Ann. Odontostomatol. (Lyon) 1968, 25, 231–240. [Google Scholar]
- Hulbert, S.F.; Young, F.A.; Mathews, R.S.; Klawitter, J.J.; Talbert, C.D.; Stelling, F.H. Potential of ceramic materials as permanently implantable skeletal prostheses. J. Biomed. Mater. Res. 1970, 4, 433–456. [Google Scholar] [CrossRef]
- Hench, L.L.; Splinter, R.J.; Allen, W.C.; Greenlee, T.K. Bonding mechanisms at the interface of ceramic prosthetic materials. J. Biomed. Mater. Res. 1971, 2, 117–141. [Google Scholar]
- Hulbert, S.F.; Hench, L.L.; Forbers, D.; Bowman, L.S. History of bioceramics. Ceram. Int. 1982, 8, 131–140. [Google Scholar] [CrossRef]
- Hulbert, S.F.; Hench, L.L.; Forbers, D.; Bowman, L.S. History of bioceramics. In Ceramics in Surgery; Vincenzini, P., Ed.; Elsevier: Amsterdam, The Netherlands, 1983; pp. 3–29. [Google Scholar]
- Jarcho, M.; Bolen, C.H.; Thomas, M.B.; Bobick, J.; Kay, J.F.; Doremus, R.H. Hydroxylapatite synthesis and characterization in dense polycrystalline form. J. Mater. Sci. 1976, 11, 2027–2035. [Google Scholar] [CrossRef]
- Jarcho, M.; O’Connor, J.R.; Paris, D.A. Ceramic hydroxylapatite as a plaque growth and drug screening substrate. J. Dental Res. 1977, 56, 151–156. [Google Scholar] [CrossRef]
- Jarcho, M.; Salsbury, R.L.; Thomas, M.B.; Doremus, R.H. Synthesis and fabrication of β-tricalcium phosphate ceramics for potential prosthetic applications. J. Mater. Sci. 1979, 14, 142–150. [Google Scholar] [CrossRef]
- Jarcho, M. Calcium phosphate ceramics as hard tissue prosthetics. Clin. Orthop. Rel. Res. 1981, 157, 259–278. [Google Scholar]
- Rejda, B.V.; Peelen, J.G.J.; De Groot, K. Tricalcium phosphate as a bone substitute. J. Bioeng. 1977, 1, 93–97. [Google Scholar]
- De Groot, K. Bioceramics consisting of calcium phosphate salts. Biomaterials 1980, 1, 47–50. [Google Scholar] [CrossRef]
- Aoki, H.; Kato, K.M.; Ogiso, M.; Tabata, T. Studies on the application of apatite to dental materials. J. Dent. Eng. 1977, 18, 86–89. [Google Scholar]
- Kato, K.; Aoki, H.; Tabata, T.; Ogiso, M. Biocompatibility of apatite ceramics in mandibles. Biomater. Med. Dev. Artif. Organs 1979, 7, 291–297. [Google Scholar]
- Akao, M.; Aoki, H.; Kato, K. Mechanical properties of sintered hydroxyapatite for prosthetic applications. J. Mater. Sci. 1981, 16, 809–812. [Google Scholar] [CrossRef]
- Akao, M.; Aoki, H.; Kato, K.; Sato, A. Dense polycrystalline β-tricalcium phosphate for prosthetic applications. J. Mater. Sci. 1982, 17, 343–346. [Google Scholar] [CrossRef]
- Roy, D.M.; Linnehan, S.K. Hydroxyapatite formed from coral skeletal carbonate by hydrothermal exchange. Nature 1974, 247, 220–222. [Google Scholar] [CrossRef]
- Holmes, R.E. Bone regeneration within a coralline hydroxyapatite implant. Plast. Reconstr. Surg. 1979, 63, 626–633. [Google Scholar] [CrossRef]
- Elsinger, E.C.; Leal, L. Coralline hydroxyapatite bone graft substitutes. J. Foot Ankle Surg. 1996, 35, 396–399. [Google Scholar] [CrossRef]
- Shipman, P.; Foster, G.; Schoeninger, M. Burnt bones and teeth: An experimental study of color, morphology, crystal structure and shrinkage. J. Archaeol. Sci. 1984, 11, 307–325. [Google Scholar] [CrossRef]
- LeGeros, R.Z.; LeGeros, J.P. Calcium phosphate bioceramics: Past, present, future. Key Eng. Mater. 2003, 240-242, 3–10. [Google Scholar] [CrossRef]
- Rivera, E.M.; Araiza, M.; Brostow, W.; Castaño, V.M.; Díaz-Estrada, J.R.; Hernández, R.; Rodríguez, J.R. Synthesis of hydroxyapatite from eggshells. Mater. Lett. 1999, 41, 128–134. [Google Scholar] [CrossRef]
- Lee, S.J.; Oh, S.H. Fabrication of calcium phosphate bioceramics by using eggshell and phosphoric acid. Mater. Lett. 2003, 57, 4570–4574. [Google Scholar] [CrossRef]
- Balazsi, C.; Weber, F.; Kover, Z.; Horvath, E.; Nemeth, C. Preparation of calcium-phosphate bioceramics from natural resources. J. Eur. Ceram. Soc. 2007, 27, 1601–1606. [Google Scholar] [CrossRef]
- Murugan, R.; Ramakrishna, S. Crystallographic study of hydroxyapatite bioceramics derived from various sources. Cryst. Growth Des. 2005, 5, 111–112. [Google Scholar] [CrossRef]
- Ruksudjarit, A.; Pengpat, K.; Rujijanagul, G.; Tunkasiri, T. Synthesis and characterization of nanocrystalline hydroxyapatite from natural bovine bone. Curr. Appl. Phys. 2008, 8, 270–272. [Google Scholar] [CrossRef]
- Ooi, C.Y.; Hamdi, M.; Ramesh, S. Properties of hydroxyapatite produced by annealing of bovine bone. Ceram. Int. 2007, 33, 1171–1177. [Google Scholar] [CrossRef]
- Lee, S.J.; Lee, Y.C.; Yoon, Y.S. Characteristics of calcium phosphate powders synthesized from cuttlefish bone and phosphoric acid. J. Ceram. Proc. Res. 2007, 8, 427–430. [Google Scholar]
- Oktar, F.N. Microstructure and mechanical properties of sintered enamel hydroxyapatite. Ceram. Int. 2007, 33, 1309–1314. [Google Scholar] [CrossRef]
- Vallet-Regí, M.; González-Calbet, J.M. Calcium phosphates as substitution of bone tissues. Progr. Solid State Chem. 2004, 32, 1–31. [Google Scholar] [CrossRef]
- Lecomte, A.; Gautier, H.; Bouler, J.M.; Gouyette, A.; Pegon, Y.; Daculsi, G.; Merle, C. Biphasic calcium phosphate: A comparative study of interconnected porosity in two ceramics. J. Biomed. Mater. Res. B Appl. Biomater. 2008, 84B, 1–6. [Google Scholar] [CrossRef]
- Tancret, F.; Bouler, J.M.; Chamousset, J.; Minois, L.M. Modelling the mechanical properties of microporous and macroporous biphasic calcium phosphate bioceramics. J. Eur. Ceram. Soc. 2006, 26, 3647–3656. [Google Scholar] [CrossRef]
- Bouler, J.M.; Trecant, M.; Delecrin, J.; Royer, J.; Passuti, N.; Daculsi, G. Macroporous biphasic calcium phosphate ceramics: Influence of five synthesis parameters on compressive strength. J. Biomed. Mater. Res. 1996, 32, 603–609. [Google Scholar] [CrossRef]
- Kivrak, N.; Tas, A.C. Synthesis of calcium hydroxyapatite—tricalcium phosphate (HA-TCP) composite bioceramic powders and their sintering behavior. J. Am. Ceram. Soc. 1998, 81, 2245–2252. [Google Scholar] [CrossRef]
- Gauthier, O.; Bouler, J.M.; Aguado, E.; Pilet, P.; Daculsi, G. Macroporous biphasic calcium phosphate ceramics: Influence of macropore diameter and macroporosity percentage on bone ingrowth. Biomaterials 1998, 19, 133–139. [Google Scholar] [CrossRef]
- Wang, J.; Chen, W.; Li, Y.; Fan, S.; Weng, J.; Zhang, X. Biological evaluation of biphasic calcium phosphate ceramic vertebral laminae. Biomaterials 1998, 19, 1387–1392. [Google Scholar] [CrossRef]
- Daculsi, G. Biphasic calcium phosphate concept applied to artificial bone, implant coating and injectable bone substitute. Biomaterials 1998, 19, 1473–1478. [Google Scholar] [CrossRef]
- Daculsi, G.; Weiss, P.; Bouler, J.M.; Gauthier, O.; Millot, F.; Aguado, E. Biphasic calcium phosphate/hydrosoluble polymer composites: A new concept for bone and dental substitution biomaterials. Bone 1999, 25 Suppl. 2, 59S–61S. [Google Scholar] [CrossRef]
- LeGeros, R.Z.; Lin, S.; Rohanizadeh, R.; Mijares, D.; LeGeros, J.P. Biphasic calcium phosphate bioceramics: Preparation, properties and applications. J. Mater. Sci. Mater. Med. 2003, 14, 201–209. [Google Scholar] [CrossRef]
- Daculsi, G.; Laboux, O.; Malard, O.; Weiss, P. Current state of the art of biphasic calcium phosphate bioceramics. J. Mater. Sci. Mater. Med. 2003, 14, 195–200. [Google Scholar]
- Alam, I.; Asahina, I.; Ohmamiuda, K.; Enomoto, S. Comparative study of biphasic calcium phosphate ceramics impregnated with rhBMP-2 as bone substitutes. J. Biomed. Mater. Res. 2001, 54, 129–138. [Google Scholar] [CrossRef]
- Daculsi, G. Biphasic calcium phosphate granules concept for injectable and mouldable bone substitute. Adv. Sci. Technol. 2006, 49, 9–13. [Google Scholar] [CrossRef]
- Tanimoto, Y.; Shibata, Y.; Murakami, A.; Miyazaki, T.; Nishiyama, N. Effect of varying HAP/TCP ratios in tape-cast biphasic calcium phosphate ceramics on responcce in vitro. J. Hard Tissue Biology 2009, 18, 71–76. [Google Scholar] [CrossRef]
- O’Neill, W.C. The fallacy of the calcium—phosphorus product. Kidney Int. 2007, 72, 792–796. [Google Scholar] [CrossRef]
- da Silva, R.V.; Bertran, C.A.; Kawachi, E.Y.; Camilli, J.A. Repair of cranial bone defects with calcium phosphate ceramic implant or autogenous bone graft. J. Craniofac. Surg. 2007, 18, 281–286. [Google Scholar] [CrossRef]
- Metsger, D.S.; Driskell, T.D.; Paulsrud, J.R. Tricalcium phosphate ceramic—a resorbable bone implant: Review and current status. J. Am. Dent. Assoc. 1982, 105, 1035–1038. [Google Scholar]
- Reid, J.W.; Tuck, L.; Sayer, M.; Fargo, K.; Hendry, J.A. Synthesis and characterization of single-phase silicon substituted α-tricalcium phosphate. Biomaterials 2006, 27, 2916–2925. [Google Scholar] [CrossRef]
- Yin, X.; Stott, M.J.; Rubio, A. α- and β-tricalcium phosphate: A density functional study. Phys. Rev. B 2003, 68, 205205–205211. [Google Scholar] [CrossRef]
- Yin, X.; Stott, M.J. Theoretical insights into bone grafting Si-stabilized α-tricalcium phosphate. J. Chem. Phys. 2005, 122, 024709–024717. [Google Scholar] [CrossRef]
- Ruan, J.M.; Zou, J.P.; Zhou, J.N.; Hu, J.Z. Porous hydroxyapatite—tricalcium phosphate bioceramics. Powder Metall. 2006, 49, 66–69. [Google Scholar] [CrossRef]
- Yin, X.; Stott, M.J. Surface and adsorption properties of α-tricalcium phosphate. J. Chem. Phys. 2006, 124, 124701–124709. [Google Scholar] [CrossRef]
- Kamitakahara, M.; Ohtsuki, C.; Miyazaki, T. Review paper: Behavior of ceramic biomaterials derived from tricalcium phosphate in physiological condition. J. Biomater. Appl. 2008, 23, 197–212. [Google Scholar] [CrossRef]
- Oishi, M.; Ohtsuki, C.; Kitamura, M.; Kamitakahara, M.; Ogata, S.; Miyazaki, T.; Tanihara, M. Fabrication and chemical durability of porous bodies consisting of biphasic tricalcium phosphates. Phosphorus Res. Bull. 2004, 17, 95–100. [Google Scholar]
- Kamitakahara, M.; Ohtsuki, C.; Oishi, M.; Ogata, S.; Miyazaki, T.; Tanihara, M. Preparation of porous biphasic tricalcium phosphate and its in vivo behavior. Key Eng. Mater. 2005, 284-286, 281–284. [Google Scholar] [CrossRef]
- Wang, R.; Weng, W.; Deng, X.; Cheng, K.; Liu, X.; Du, P.; Shen, G.; Han, G. Dissolution behavior of submicron biphasic tricalcium phosphate powders. Key Eng. Mater. 2006, 309-311, 223–226. [Google Scholar] [CrossRef]
- Li, Y.; Weng, W.; Tam, K.C. Novel highly biodegradable biphasic tricalcium phosphates composed of α-tricalcium phosphate and β-tricalcium phosphate. Acta Biomater. 2007, 3, 251–254. [Google Scholar] [CrossRef]
- Li, Y.; Li, D.; Weng, W. In vitro dissolution behavior of biphasic tricalcium phosphate composite powders composed of α-tricalcium phosphate and β-tricalcium phosphate. Key Eng. Mater. 2008, 368-372, 1206–1208. [Google Scholar] [CrossRef]
- Vani, R.; Girija, E.K.; Elayaraja, K.; Prakash Parthiban, S.; Kesavamoorthy, R.; Narayana Kalkura, S. Hydrothermal synthesis of porous triphasic hydroxyapatite/(α and β) tricalcium phosphate. J. Mater. Sci. Mater. Med. 2009, 20 Suppl. 1, 43–48. [Google Scholar]
- Daculsi, G.; Bouler, J.M.; LeGeros, R.Z. Adaptive crystal formation in normal and pathological calcifications in synthetic calcium phosphate and related biomaterials. Int. Rev. Cytol. 1997, 172, 129–191. [Google Scholar] [CrossRef]
- Astala, R.; Calderin, L.; Yin, X.; Stott, M.J. Ab initio simulation of Si-doped hydroxyapatite. Chem. Mater. 2006, 18, 413–422. [Google Scholar] [CrossRef]
- Zhu, X.D.; Zhang, H.J.; Fan, H.S.; Li, W.; Zhang, X.D. Effect of phase composition and microstructure of calcium phosphate ceramic particles on protein adsorption. Acta Biomater. 2010, 6, 1536–1541. [Google Scholar] [CrossRef]
- Bohner, M. Calcium orthophosphates in medicine: From ceramics to calcium phosphate cements. Injury 2000, 31 Suppl. 4, D37–D47. [Google Scholar] [CrossRef]
- Norton, J.; Malik, K.R.; Darr, J.A.; Rehman, I. Recent developments in processing and surface modification of hydroxyapatite. Adv. Appl. Ceram. 2006, 105, 113–139. [Google Scholar] [CrossRef]
- Yang, S.; Leong, K.F.; Du, Z.; Chua, C.K. The design of scaffolds for use in tissue engineering. Part II. Rapid prototyping techniques. Tissue Eng. 2002, 8, 1–11. [Google Scholar] [CrossRef]
- Yeong, W.Y.; Chua, C.K.; Leong, K.F.; Chandrasekaran, M. Rapid prototyping in tissue engineering: Challenges and potential. Trends Biotechnol. 2004, 22, 643–652. [Google Scholar] [CrossRef]
- Hieu, L.C.; Zlatov, N.; Sloten, J.V.; Bohez, E.; Khanh, L.; Binh, P.H.; Oris, P.; Toshev, Y. Medical rapid prototyping applications and methods. Assembly Autom. 2005, 25, 284–292. [Google Scholar] [CrossRef]
- Eufinger, H.; Wehniöller, M.; Machtens, E.; Heuser, L.; Harders, A.; Kruse, D. Reconstruction of craniofacial bone defects with individual alloplastic implants based on CAD/CAM-manipulated CT-data. J. Craniomaxillofac. Surg. 1995, 23, 175–181. [Google Scholar] [CrossRef]
- Klein, M.; Glatzer, C. Individual CAD/CAM fabricated glass-bioceramic implants in reconstructive surgery of the bony orbital floor. Plast. Reconstr. Surg. 2006, 117, 565–570. [Google Scholar] [CrossRef]
- Yin, L.; Song, X.F.; Song, Y.L.; Huang, T.; Li, J. An overview of in vitro abrasive finishing & CAD/CAM of bioceramics in restorative dentistry. Int. J. Mach. Tools Manuf. 2006, 46, 1013–1026. [Google Scholar] [CrossRef]
- Yardimci, M.A.; Guceri, S.I.; Danforth, S.C. Process modeling for fused deposition of ceramics. Ceram. Eng. Sci. Proc. 1996, 17, 78–82. [Google Scholar] [CrossRef]
- Bellini, A.; Shor, L.; Guceri, S.I. New developments in fused deposition modeling of ceramics. Rapid Prototyping J. 2005, 11, 214–220. [Google Scholar] [CrossRef]
- Tan, K.H.; Chua, C.K.; Leong, K.F.; Cheah, C.M.; Cheang, P.; Abu Bakar, M.S.; Cha, S.W. Scaffold development using selective laser sintering of polyetheretherketone-hydroxyapatite biocomposite blends. Biomaterials 2003, 24, 3115–3123. [Google Scholar] [CrossRef]
- Wiria, F.E.; Leong, K.F.; Chua, C.K.; Liu, Y. Poly-ε-caprolactone/hydroxyapatite for tissue engineering scaffold fabrication via selective laser sintering. Acta Biomater. 2007, 3, 1–12. [Google Scholar]
- Zhou, W.Y.; Lee, S.H.; Wang, M.; Cheung, W.L.; Ip, W.Y. Selective laser sintering of porous tissue engineering scaffolds from poly(L-lactide)/carbonated hydroxyapatite nanocomposite microspheres. J. Mater. Sci. Mater. Med. 2008, 19, 2535–2540. [Google Scholar] [CrossRef]
- Sachs, E.; Cima, M.; Williams, P.; Brancazio, D.; Cornie, J. Three dimensional printing. Rapid tooling and prototypes directly from a CAD model. J. Eng. Ind. 1992, 114, 481–488. [Google Scholar] [CrossRef]
- Gbureck, U.; Hölzel, T.; Doillon, C.J.; Müller, F.A.; Barralet, J.E. Direct printing of bioceramic implants with spatially localized angiogenic factors. Adv. Mater. 2007, 19, 795–800. [Google Scholar] [CrossRef]
- Gbureck, U.; Hölzel, T.; Klammert, U.; Würzler, K.; Müller, F.A.; Barralet, J.E. Resorbable dicalcium phosphate bone substitutes prepared by 3D powder printing. Adv. Funct. Mater. 2007, 17, 3940–3945. [Google Scholar] [CrossRef]
- Vorndran, E.; Klarner, M.; Klammert, U.; Grover, L.M.; Patel, S.; Barralet, J.E.; Gbureck, U. 3D powder printing of β-tricalcium phosphate ceramics using different strategies. Adv. Eng. Mater. 2008, 10, B67–B71. [Google Scholar] [CrossRef]
- Seitz, H.; Rieder, W.; Irsen, S.; Leukers, B.; Tille, C. Three-dimensional printing of porous ceramic scaffolds for bone tissue engineering. J. Biomed. Mater. Res. B 2005, 74B, 782–788. [Google Scholar] [CrossRef]
- Leukers, B.; Gülkan, H.; Irsen, S.H.; Milz, S.; Tille, C.; Schieker, M.; Seitz, H. Hydroxyapatite scaffolds for bone tissue engineering made by 3D printing. J. Mater. Sci. Mater. Med. 2005, 16, 1121–1124. [Google Scholar] [CrossRef]
- Habibovic, P.; Gbureck, U.; Doillon, C.J.; Bassett, D.C.; Van Blitterswijk, C.A.; Barralet, J.E. Osteoconduction and osteoinduction of low-temperature 3D printed bioceramic implants. Biomaterials 2008, 29, 944–953. [Google Scholar]
- Fierz, F.C.; Beckmann, F.; Huser, M.; Irsen, S.H.; Leukers, B.; Witte, F.; Degistirici, O.; Andronache, A.; Thie, M.; Müller, B. The morphology of anisotropic 3D-printed hydroxyapatite scaffolds. Biomaterials 2008, 29, 3799–3806. [Google Scholar] [CrossRef]
- Seitz, H.; Deisinger, U.; Leukers, B.; Detsch, R.; Ziegler, G. Different calcium phosphate granules for 3-D printing of bone tissue engineering scaffolds. Adv. Eng. Mater. 2009, 11, B41–B46. [Google Scholar] [CrossRef]
- Chu, T.M.G.; Halloran, J.W.; Hollister, S.J.; Feinberg, S.E. Hydroxyapatite implants with designed internal architecture. J. Mater. Sci. Mater. Med. 2001, 12, 471–478. [Google Scholar] [CrossRef]
- Li, X.; Li, D.; Lu, B.; Tang, Y.; Wang, L.; Wang, Z. Design and fabrication of CAP scaffolds by indirect solid free form fabrication. Rapid Prototyping J. 2005, 11, 312–318. [Google Scholar] [CrossRef]
- Woesz, A.; Rumpler, M.; Stampfl, J.; Varga, F.; Fratzl-Zelman, N.; Roschger, P.; Klaushofer, K.; Fratzl, P. Towards bone replacement materials from calcium phosphates via rapid prototyping and ceramic gel casting. Mater. Sci. Eng. C 2005, 25, 181–186. [Google Scholar] [CrossRef]
- Li, X.; Li, D.; Lu, B.; Wang, C. Fabrication of bioceramic scaffolds with pre-designed internal architecture by gel casting and indirect stereolithography techniques. J. Porous Mater. 2008, 15, 667–671. [Google Scholar] [CrossRef]
- Leong, K.F.; Cheah, C.M.; Chua, C.K. Solid freeform fabrication of three-dimensional scaffolds for engineering replacement tissues and organs. Biomaterials 2003, 24, 2363–2378. [Google Scholar] [CrossRef]
- Bone Grafts and Bone Substitutes: Basic Science and Clinical Applications; Nather, A. (Ed.) World Scientific: Singapore, 2005; p. 592.
- Bio-Materials and Prototyping Applications in Medicine; Bártolo, P.; Bidanda, B. (Eds.) Springer: New York, NY, USA, 2008; p. 216.
- Narayan, R.J.; Jin, C.; Doraiswamy, A.; Mihailescu, I.N.; Jelinek, M.; Ovsianikov, A.; Chichkov, B.; Chrisey, D.B. Laser processing of advanced bioceramics. Adv. Eng. Mater. 2005, 7, 1083–1098. [Google Scholar] [CrossRef]
- Bioceramics and Their Clinical Applications; Kokubo, T. (Ed.) Woodhead Publishing: Sawston, UK, 2008; p. 784.
- Biomedical Materials; Narayan, R. (Ed.) Springer: New York, NY, USA, 2009; p. 566.
- Raksujarit, A.; Pengpat, K.; Rujijanagul, G.; Tunkasiri, T. Processing and properties of nanoporous hydroxyapatite ceramics. Mater. Des. 2010, 31, 1658–1660. [Google Scholar] [CrossRef]
- Park, J. Bioceramics: Properties, Characterizations, and Applications; Springer: Berlin, Germany, 2008; p. 364. [Google Scholar]
- Rodríguez-Lorenzo, L.M.; Vallet-Regí, M.; Ferreira, J.M.F. Fabrication of hydroxyapatite bodies by uniaxial pressing from a precipitated powder. Biomaterials 2001, 22, 583–588. [Google Scholar] [CrossRef]
- Nazarpak, M.H.; Solati-Hashjin, M.; Moztarzadeh, F. Preparation of hydroxyapatite ceramics for biomedical applications. J. Ceram. Proc. Res. 2009, 10, 54–57. [Google Scholar]
- Itoh, H.; Wakisaka, Y.; Ohnuma, Y.; Kuboki, Y. A new porous hydroxyapatite ceramic prepared by cold isostatic pressing and sintering synthesized flaky powder. Dental Mater. 1994, 13, 25–35. [Google Scholar] [CrossRef]
- Gautier, H.; Merle, C.; Auget, J.L.; Daculsi, G. Isostatic compression, a new process for incorporating vancomycin into biphasic calcium phosphate: Comparison with a classical method. Biomaterials 2000, 21, 243–249. [Google Scholar] [CrossRef]
- Tadic, D.; Epple, M. Mechanically stable implants of synthetic bone mineral by cold isostatic pressing. Biomaterials 2003, 24, 4565–4571. [Google Scholar] [CrossRef]
- Pecqueux, F.; Tancret, F.; Payraudeau, N.; Bouler, J.M. Influence of microporosity and macroporosity on the mechanical properties of biphasic calcium phosphate bioceramics: Modelling and experiment. J. Eur. Ceram. Soc. 2010, 30, 819–829. [Google Scholar] [CrossRef]
- Uematsu, K.; Takagi, M.; Honda, T.; Uchida, N.; Saito, K. Transparent hydroxyapatite prepared by hot isostatic pressing of filter cake. J. Am. Ceram. Soc. 1989, 72, 1476–1478. [Google Scholar] [CrossRef]
- Onoki, T.; Hashida, T. New method for hydroxyapatite coating of titanium by the hydrothermal hot isostatic pressing technique. Surf. Coat. Technol. 2006, 200, 6801–6807. [Google Scholar] [CrossRef]
- Takikawa, K.; Akao, M. Fabrication of transparent hydroxyapatite and application to bone marrow derived cell/hydroxyapatite interaction observation in-vivo. J. Mater. Sci. Mater. Med. 1996, 7, 439–445. [Google Scholar] [CrossRef]
- Viana, M.; Désiré, A.; Chevalier, E.; Champion, E.; Chotard, R.; Chulia, D. Interest of high shear wet granulation to produce drug loaded porous calcium phosphate pellets for bone filling. Key Eng. Mater. 2009, 396-398, 535–538. [Google Scholar] [CrossRef]
- Reikerås, O.; Johansson, C.B.; Sundfeldt, M. Bone ingrowths to press-fit and loose-fit implants: Comparisons between titanium and hydroxyapatite. J. Long. Term. Eff. Med. Implants 2006, 16, 157–164. [Google Scholar] [CrossRef]
- Rao, R.R.; Kannan, T.S. Dispersion and slip casting of hydroxyapatite. J. Am. Ceram. Soc. 2001, 84, 1710–1716. [Google Scholar]
- Sakka, Y.; Takahashi, K.; Matsuda, N.; Suzuki, T.S. Effect of milling treatment on texture development of hydroxyapatite ceramics by slip casting in high magnetic field. Mater. Trans. 2007, 48, 2861–2866. [Google Scholar] [CrossRef]
- Zhang, Y.; Yokogawa, Y.; Feng, X.; Tao, Y.; Li, Y. Preparation and properties of bimodal porous apatite ceramics through slip casting using different hydroxyapatite powders. Ceram. Int. 2010, 36, 107–113. [Google Scholar] [CrossRef]
- Sepulveda, P.; Ortega, F.S.; Innocentini, M.D.M.; Pandolfelli, V.C. Properties of highly porous hydroxyapatite obtained by the gel casting of foams. J. Am. Ceram. Soc. 2000, 83, 3021–3024. [Google Scholar] [CrossRef]
- Padilla, S.; Vallet-Regí, M.; Ginebra, M.P.; Gil, F.J. Processing and mechanical properties of hydroxyapatite pieces obtained by the gel-casting method. J. Eur. Ceram. Soc. 2005, 25, 375–383. [Google Scholar]
- Sánchez-Salcedo, S.; Werner, J.; Vallet-Regí, M. Hierarchical pore structure of calcium phosphate scaffolds by a combination of gel-casting and multiple tape-casting methods. Acta Biomater. 2008, 4, 913–922. [Google Scholar] [CrossRef]
- Chen, B.; Zhang, T.; Zhang, J.; Lin, Q.; Jiang, D. Microstructure and mechanical properties of hydroxyapatite obtained by gel-casting process. Ceram. Int. 2008, 34, 359–364. [Google Scholar] [CrossRef]
- Marcassoli, P.; Cabrini, M.; Tirillò, J.; Bartuli, C.; Palmero, P.; Montanaro, L. Mechanical characterization of hydroxiapatite micro/macro-porous ceramics obtained by means of innovative gel-casting process. Key Eng. Mater. 2010, 417-418, 565–568. [Google Scholar]
- Padilla, S.; Roman, J.; Vallet-Regí, M. Synthesis of porous hydroxyapatites by combination of gel casting and foams burn out methods. J. Mater. Sci. Mater. Med. 2002, 13, 1193–1197. [Google Scholar]
- Fomin, A.S.; Barinov, S.M.; Ievlev, V.M.; Smirnov, V.V.; Mikhailov, B.P.; Belonogov, E.K.; Drozdova, N.A. Nanocrystalline hydroxyapatite ceramics produced by low-temperature sintering after high-pressure treatment. Dokl. Chem. 2008, 418, 22–25. [Google Scholar]
- Kankawa, Y.; Kaneko, Y.; Saitou, K. Injection molding of highly-purified hydroxylapatite and TCP utilizing solid phase reaction method. J. Ceram. Soc. Jpn. 1991, 99, 438–442. [Google Scholar]
- Kwon, S.H.; Jun, Y.K.; Hong, S.H.; Lee, I.S.; Kim, H.E.; Won, Y.Y. Calcium phosphate bioceramics with various porosities and dissolution rates. J. Am. Ceram. Soc. 2002, 85, 3129–3131. [Google Scholar]
- Fooki, A.C.B.M.; Aparecida, A.H.; Fideles, T.B.; Costa, R.C.; Fook, M.V.L. Porous hydroxyapatite scaffolds by polymer sponge method. Key Eng. Mater. 2009, 396-398, 703–706. [Google Scholar] [CrossRef]
- Sopyan, I.; Kaur, J. Preparation and characterization of porous hydroxyapatite through polymeric sponge method. Ceram. Int. 2009, 35, 3161–3168. [Google Scholar] [CrossRef]
- Bellucci, D.; Cannillo, V.; Sola, A. Shell scaffolds: A new approach towards high strength bioceramic scaffolds for bone regeneration. Mater. Lett. 2010, 64, 203–206. [Google Scholar]
- Velayudhan, S.; Ramesh, P.; Sunny, M.C.; Varma, H.K. Extrusion of hydroxyapatite to clinically significant shapes. Mater. Lett. 2000, 46, 142–146. [Google Scholar]
- Yang, H.Y.; Thompson, I.; Yang, S.F.; Chi, X.P.; Evans, J.R.G.; Cook, R.J. Dissolution characteristics of extrusion freeformed hydroxyapatite—tricalcium phosphate scaffolds. J. Mater. Sci. Mater. Med. 2008, 19, 3345–3353. [Google Scholar]
- Yang, S.; Yang, H.; Chi, X.; Evans, J.R.G.; Thompson, I.; Cook, R.J.; Robinson, P. Rapid prototyping of ceramic lattices for hard tissue scaffolds. Mater. Des. 2008, 29, 1802–1809. [Google Scholar] [CrossRef]
- Muthutantri, A.I.; Huang, J.; Edirisinghe, M.J.; Bretcanu, O.; Boccaccini, A.R. Dipping and electrospraying for the preparation of hydroxyapatite foams for bone tissue engineering. Biomed. Mater. 2008, 3, 25009–25022. [Google Scholar] [CrossRef]
- Roncari, E.; Galassi, C.; Pinasco, P. Tape casting of porous hydroxyapatite ceramics. J. Mater. Sci. Lett. 2000, 19, 33–35. [Google Scholar] [CrossRef]
- Tian, T.; Jiang, D.; Zhang, J.; Lin, Q. Aqueous tape casting process for hydroxyapatite. J. Eur. Ceram. Soc. 2007, 27, 2671–2677. [Google Scholar] [CrossRef]
- Suzuki, S.; Itoh, K.; Ohgaki, M.; Ohtani, M.; Ozawa, M. Preparation of sintered filter for ion exchange by a doctor blade method with aqueous slurries of needlelike hydroxyapatite. Ceram. Int. 1999, 25, 287–291. [Google Scholar] [CrossRef]
- Inoue, K.; Sassa, K.; Yokogawa, Y.; Sakka, Y.; Okido, M.; Asai, S. Control of crystal orientation of hydroxyapatite by imposition of a high magnetic field. Mater. Trans. 2003, 44, 1133–1137. [Google Scholar]
- Iwai, K.; Akiyama, J.; Asai, S. Structure control of hydroxyapatite using a magnetic field. Mater. Sci. Forum 2007, 561-565, 1565–1568. [Google Scholar] [CrossRef]
- Iwai, K.; Akiyama, J.; Tanase, T.; Asai, S. Alignment of HAp crystal using a sample rotation in a static magnetic field. Mater. Sci. Forum 2007, 539-543, 716–719. [Google Scholar] [CrossRef]
- Sakka, Y.; Takahashi, K.; Suzuki, T.S.; Ito, S.; Matsuda, N. Texture development of hydroxyapatite ceramics by colloidal processing in a high magnetic field followed by sintering. Mater. Sci. Eng. A 2008, 475, 27–33. [Google Scholar]
- Kang, J.; Hadfield, M. Parameter optimization by Taguchi methods for finishing advanced ceramic balls using a novel eccentric lapping machine. Proc. Inst. Mech. Eng. B 2001, 215, 69–78. [Google Scholar] [CrossRef]
- Kurella, A.; Dahotre, N.B. Surface modification for bioimplants: The role of laser surface engineering. J. Biomater. Appl. 2005, 20, 5–50. [Google Scholar] [CrossRef]
- Oktar, F.N.; Genc, Y.; Goller, G.; Erkmen, E.Z.; Ozyegin, L.S.; Toykan, D.; Demirkiran, H.; Haybat, H. Sintering of synthetic hydroxyapatite compacts. Key Eng. Mater. 2004, 264-268, 2087–2090. [Google Scholar] [CrossRef]
- He, Z.; Ma, J.; Wang, C. Constitutive modeling of the densification and the grain growth of hydroxyapatite ceramics. Biomaterials 2005, 26, 1613–1621. [Google Scholar] [CrossRef]
- Fellah, B.H.; Layrolle, P. Sol-gel synthesis and characterization of macroporous calcium phosphate bioceramics containing microporosity. Acta Biomater. 2009, 5, 735–742. [Google Scholar] [CrossRef]
- Khalil, K.A.; Kim, S.W.; Dharmaraj, N.; Kim, K.W.; Kim, H.Y. Novel mechanism to improve toughness of the hydroxyapatite bioceramics using high-frequency induction heat sintering. J. Mater. Proc. Technol 2007, 187-188, 417–420. [Google Scholar]
- Landi, E.; Tampieri, A.; Celotti, G.; Sprio, S. Densification behaviour and mechanisms of synthetic hydroxyapatites. J. Eur. Ceram. Soc. 2000, 20, 2377–2387. [Google Scholar] [CrossRef]
- Ruys, A.J.; Wei, M.; Sorrell, C.C.; Dickson, M.R.; Brandwood, A.; Milthorpe, B.K. Sintering effect on the strength of hydroxyapatite. Biomaterials 1995, 16, 409–415. [Google Scholar] [CrossRef]
- Van Landuyt, P.; Li, F.; Keustermans, J.P.; Streydio, J.M.; Delannay, F.; Munting, E. The influence of high sintering temperatures on the mechanical properties of hydroxylapatite. J. Mater. Sci. Mater. Med. 1995, 6, 8–13. [Google Scholar] [CrossRef]
- Ben Ayed, F.; Bouaziz, J.; Bouzouita, K. Pressureless sintering of fluorapatite under oxygen atmosphere. J. Eur. Ceram. Soc. 2000, 20, 1069–1076. [Google Scholar] [CrossRef]
- Haberko, K.; Bućko, M.M.; Brzezińska-Miecznik, J.; Haberko, M.; Mozgawa, W.; Panz, T.; Pyda, A.; Zarebski, J. Natural hydroxyapatite—its behaviour during heat treatment. J. Eur. Ceram. Soc. 2006, 26, 537–542. [Google Scholar] [CrossRef]
- Haberko, K.; Bućko, M.M.; Mozgawa, W.; Pyda, A.; Brzezińska-Miecznik, J.; Carpentier, J. Behaviour of bone origin hydroxyapatite at elevated temperatures and in O2 and CO2 atmospheres. Ceram. Int. 2009, 35, 2537–2540. [Google Scholar] [CrossRef]
- Janus, A.M.; Faryna, M.; Haberko, K.; Rakowska, A.; Panz, T. Chemical and microstructural characterization of natural hydroxyapatite derived from pig bones. Mikrochim. Acta 2008, 161, 349–353. [Google Scholar] [CrossRef]
- Bahrololoom, M.E.; Javidi, M.; Javadpour, S.; Ma, J. Characterisation of natural hydroxyapatite extracted from bovine cortical bone ash. J. Ceram. Proc. Res. 2009, 10, 129–138. [Google Scholar]
- Dorozhkina, E.I.; Dorozhkin, S.V. Mechanism of the solid-state transformation of a calcium-deficient hydroxyapatite (CDHA) into biphasic calcium phosphate (BCP) at elevated temperatures. Chem. Mater. 2002, 14, 4267–4272. [Google Scholar] [CrossRef]
- Li, Y.; Kong, F.; Weng, W. Preparation and characterization of novel biphasic calcium phosphate powders (α-TCP/HA) derived from carbonated amorphous calcium phosphates. J. Biomed. Mater. Res. B Appl. Biomater. 2009, 89B, 508–517. [Google Scholar] [CrossRef]
- Mostafa, N.Y. Characterization, thermal stability and sintering of hydroxyapatite powders prepared by different routes. Mater. Chem. Phys. 2005, 94, 333–341. [Google Scholar] [CrossRef]
- Suchanek, W.; Yashima, M.; Kakihana, M.; Yoshimura, M. Hydroxyapatite ceramics with selected sintering additives. Biomaterials 1997, 18, 923–933. [Google Scholar] [CrossRef]
- Kalita, S.J.; Bose, S.; Bandyopadhyay, A.; Hosick, H.L. Oxide based sintering additives for HAp ceramics. Ceram. Trans. 2003, 147, 63–72. [Google Scholar]
- Kalita, S.J.; Bose, S.; Hosick, H.L.; Bandyopadhyay, A. CaO-P2O5-Na2O-based sintering additives for hydroxyapatite (HAp) ceramics. Biomaterials 2004, 25, 2331–2339. [Google Scholar] [CrossRef]
- Safronova, T.V.; Putlyaev, V.I.; Shekhirev, M.A.; Tretyakov, Y.D.; Kuznetsov, A.V.; Belyakov, A.V. Densification additives for hydroxyapatite ceramics. J. Eur. Ceram. Soc. 2009, 29, 1925–1932. [Google Scholar] [CrossRef]
- Chen, S.; Wang, W.; Kono, H.; Sassa, K.; Asai, S. Abnormal grain growth of hydroxyapatite ceramic sintered in a high magnetic field. J. Cryst. Growth 2010, 312, 323–326. [Google Scholar]
- Muralithran, G.; Ramesh, S. Effects of sintering temperature on the properties of hydroxyapatite. Ceram. Int. 2000, 26, 221–230. [Google Scholar] [CrossRef]
- Bernache-Assollant, D.; Ababou, A.; Champion, E.; Heughebaert, M. Sintering of calcium phosphate hydroxyapatite Ca10(PO4)6(OH)2 I. Calcination and particle growth. J. Eur. Ceram. Soc. 2003, 23, 229–241. [Google Scholar] [CrossRef]
- Ramesh, S.; Tan, C.Y.; Bhaduri, S.B.; Teng, W.D. opyan I. Densification behaviour of nanocrystalline hydroxyapatite bioceramics. J. Mater. Proc. Technol. 2008, 206, 221–230. [Google Scholar]
- Kobayashi, S.; Kawai, W.; Wakayama, S. The effect of pressure during sintering on the strength and the fracture toughness of hydroxyapatite ceramics. J. Mater. Sci. Mater. Med. 2006, 17, 1089–1093. [Google Scholar] [CrossRef]
- Kasuga, T.; Ota, Y.; Tsuji, K.; Abe, Y. Preparation of high-strength calcium phosphate ceramics with low modulus of elasticity containing β-Ca(PO3)2 fibers. J. Am. Ceram. Soc. 1996, 79, 1821–1824. [Google Scholar] [CrossRef]
- Suchanek, W.L.; Yoshimura, M. Preparation of fibrous, porous hydroxyapatite ceramics from hydroxyapatite whiskers. J. Am. Ceram. Soc. 1998, 81, 765–767. [Google Scholar] [CrossRef]
- Kim, Y.; Kim, S.R.; Song, H.; Yoon, H. Preparation of porous hydroxyapatite/TCP composite block using a hydrothermal hot pressing method. Mater. Sci. Forum 2005, 486-487, 117–120. [Google Scholar] [CrossRef]
- Li, J.G.; Hashida, T. Preparation of hydroxyapatite ceramics by hydrothermal hot-pressing method at 300 °C. J. Mater. Sci. 2007, 42, 5013–5019. [Google Scholar] [CrossRef]
- Li, J.G.; Hashida, T. In situ formation of hydroxyapatite-whisker ceramics by hydrothermal hot-pressing method. J. Am. Ceram. Soc. 2006, 89, 3544–3546. [Google Scholar] [CrossRef]
- Halouani, R.; Bernache-Assolant, D.; Champion, E.; Ababou, A. Microstructure and related mechanical properties of hot pressed hydroxyapatite ceramics. J. Mater. Sci. Mater. Med. 1994, 5, 563–568. [Google Scholar] [CrossRef]
- Nakahira, A.; Murakami, T.; Onoki, T.; Hashida, T.; Hosoi, K. Fabrication of porous hydroxyapatite using hydrothermal hot pressing and post-sintering. J. Am. Ceram. Soc. 2005, 88, 1334–1336. [Google Scholar] [CrossRef]
- Auger, M.A.; Savoini, B.; Muñoz, A.; Leguey, T.; Monge, M.A.; Pareja, R.; Victoria, J. Mechanical characteristics of porous hydroxyapatite/oxide composites produced by post-sintering hot isostatic pressing. Ceram. Int. 2009, 35, 2373–2380. [Google Scholar] [CrossRef]
- Fang, Y.; Agrawal, D.K.; Roy, D.M.; Roy, R. Microwave sintering of hydroxyapatite ceramics. J. Mater. Res. 1994, 9, 180–187. [Google Scholar] [CrossRef]
- Yang, Y.; Ong, J.L.; Tian, J. Rapid sintering of hydroxyapatite by microwave processing. J. Mater. Sci. Lett. 2002, 21, 67–69. [Google Scholar] [CrossRef]
- Nath, S.; Basu, B.; Sinha, A. A comparative study of conventional sintering with microwave sintering of hydroxyapatite synthesized by chemical route. Trends Biomater. Artif. Organs 2006, 19, 93–98. [Google Scholar]
- Ramesh, S.; Tan, C.Y.; Bhaduri, S.B.; Teng, W.D. Rapid densification of nanocrystalline hydroxyapatite for biomedical applications. Ceram. Int. 2007, 33, 1363–1367. [Google Scholar] [CrossRef]
- Silva, C.C.; Graça, M.P.F.; Sombra, A.S.B.; Valente, M.A. Structural and electrical study of calcium phosphate obtained by a microwave radiation assisted procedure. Phys. Rev. B—Condens. Matter. 2009, 404, 1503–1508. [Google Scholar] [CrossRef]
- Chanda, A.; Dasgupta, S.; Bose, S.; Bandyopadhyay, A. Microwave sintering of calcium phosphate ceramics. Mater. Sci. Eng. C 2009, 29, 1144–1149. [Google Scholar] [CrossRef]
- Veljović, D.; Zalite, I.; Palcevskis, E.; Smiciklas, I.; Petrović, R.; Janaćković, D. Microwave sintering of fine grained HAP and HAP/TCP bioceramics. Ceram. Int. 2010, 36, 595–603. [Google Scholar] [CrossRef]
- Kalita, S.J.; Verma, S. Nanocrystalline hydroxyapatite bioceramic using microwave radiation: Synthesis and characterization. Mater. Sci. Eng. C 2010, 30, 295–303. [Google Scholar] [CrossRef]
- Gu, Y.W.; Loh, N.H.; Khor, K.A.; Tor, S.B.; Cheang, P. Spark plasma sintering of hydroxyapatite powders. Biomaterials 2002, 23, 37–43. [Google Scholar] [CrossRef]
- Guo, X.; Xiao, P.; Liu, J.; Shen, Z. Fabrication of nanostructured hydroxyapatite via hydrothermal synthesis and spark plasma sintering. J. Am. Ceram. Soc. 2005, 88, 1026–1029. [Google Scholar] [CrossRef]
- Drouet, C.; Largeot, C.; Raimbeaux, G.; Estournès, C.; Dechambre, G.; Combes, C.; Rey, C. Bioceramics: Spark plasma sintering (SPS) of calcium phosphates. Adv. Sci. Technol. 2006, 49, 45–50. [Google Scholar] [CrossRef]
- Li, H.; Khor, K.A.; Chow, V.; Cheang, P. Nanostructural characteristics, mechanical properties and osteoblast response of spark plasma sintered hydroxyapatite. J. Biomed. Mater. Res. A 2007, 82A, 296–303. [Google Scholar]
- Nakamura, T.; Fukuhara, T.; Izui, H. Mechanical properties of hydroxyapatites sintered by spark plasma sintering. Ceram. Trans. 2006, 194, 265–272. [Google Scholar]
- Kawagoe, D.; Ioku, K.; Fujimori, H.; Goto, S. Transparent β-tricalcium phosphate ceramics prepared by spark plasma sintering. J. Ceram. Soc. Jpn. 2004, 112, 462–463. [Google Scholar] [CrossRef]
- Zhang, F.; Lin, K.; Chang, J.; Lu, J.; Ning, C. Spark plasma sintering of macroporous calcium phosphate scaffolds from nanocrystalline powders. J. Eur. Ceram. Soc. 2008, 28, 539–545. [Google Scholar] [CrossRef]
- Grossin, D.; Rollin-Martinet, S.; Estournès, C.; Rossignol, F.; Champion, E.; Combes, C.; Rey, C.; Geoffroy, C.; Drouet, C. Biomimetic apatite sintered at very low temperature by spark plasma sintering: Physico-chemistry and microstructure aspects. Acta Biomater. 2010, 6, 577–585. [Google Scholar] [CrossRef] [Green Version]
- Chesnaud, A.; Bogicevic, C.; Karolak, F.; Estournès, C.; Dezanneau, G. Preparation of transparent oxyapatite ceramics by combined use of freeze-drying and spark-plasma sintering. Chem. Comm. 2007, 1550–1552. [Google Scholar]
- Ishihara, S.; Matsumoto, T.; Onoki, T.; Sohmura, T.; Nakahira, A. New concept bioceramics composed of octacalcium phosphate (OCP) and dicarboxylic acid-intercalated OCP via hydrothermal hot-pressing. Mater. Sci. Eng. C 2009, 29, 1885–1888. [Google Scholar]
- Gross, K.A.; Berndt, C.C. Biomedical application of apatites. In Phosphates: Geochemical, Geobiological And Materials Importance; Series: Reviews in Mineralogy and Geochemistry; Hughes, J.M., Kohn, M., Rakovan, J., Eds.; Mineralogical Society of America: Washington, DC, USA, 2002; Volume 48, pp. 631–672. [Google Scholar]
- LeGeros, R.Z.; LeGeros, J.P. Dense hydroxyapatite. In An Introduction to Bioceramics; Hench, L.L., Wilson, J., Eds.; World Scientific: London, UK, 1993; pp. 139–180. [Google Scholar]
- Evans, J.R.G. Seventy ways to make ceramics. J. Eur. Ceram. Soc. 2008, 28, 1421–1432. [Google Scholar] [CrossRef]
- Benaqqa, C.; Chevalier, J.; Saädaoui, M.; Fantozzi, G. Slow crack growth behaviour of hydroxyapatite ceramics. Biomaterials 2005, 26, 6106–6112. [Google Scholar] [CrossRef]
- Linhart, W.; Briem, D.; Amling, M.; Rueger, J.M.; Windolf, J. Mechanical failure of porous hydroxyapatite ceramics 7.5 years after implantation in the proximal tibial. Unfallchirurg. 2004, 107, 154–157. [Google Scholar]
- In materials science, fracture toughness is a property, which describes the ability of a material containing a crack to resist fracture and is one of the most important properties of any material for virtually all design applications
- Ramesh, S.; Tan, C.Y.; Sopyan, I.; Hamdi, M.; Teng, W.D. Consolidation of nanocrystalline hydroxyapatite powder. Sci. Technol. Adv. Mater. 2007, 8, 124–130. [Google Scholar] [CrossRef]
- The nature of flaws in most ceramics is statistical. As such, the strength of ceramics is not one specific value, but a distribution of strengths. The Weibull modulus is a measure of the distribution of flaws, usually for a brittle material. The modulus is a dimensionless number corresponding to the ability in measured strength and reflects the distribution of flaws in the material
- Suzuki, S.; Sakamura, M.; Ichiyanagi, M.; Ozawa, M. Internal friction of hydroxyapatite and fluorapatite. Ceram. Int. 2004, 30, 625–627. [Google Scholar] [CrossRef]
- Strength can be defined as the maximum tensile stresses that a material can endure before fracture occurs. It might also be defined as force per unit area required to initiate and propagate a crack to the fracture point [179]
- In materials science, fatigue is the progressive and localized structural damage that occurs when a material is subjected to cyclic loading
- le Huec, J.C.; Schaeverbeke, T.; Clement, D.; Faber, J.; le Rebeller, A. Influence of porosity on the mechanical resistance of hydroxyapatite ceramics under compressive stress. Biomaterials 1995, 16, 113–118. [Google Scholar] [CrossRef]
- Hsu, Y.H.; Turner, I.G.; Miles, A.W. Mechanical properties of three different compositions of calcium phosphate bioceramic following immersion in Ringer’s solution and distilled water. J. Mater. Sci. Mater. Med. 2009, 20, 2367–2374. [Google Scholar] [CrossRef]
- The elastic modulus is the ratio of stress to strain below the proportional limit. It represents the magnitude of the interatomic stiffness of a material within the elastic range when tensile or compressive forces are applied. It is also an indication of the amount of reversible deformation that will ur in a structure when a load is applied [179]
- De Aza, P.N.; De Aza, A.H.; De Aza, S. Crystalline bioceramic materials. Bol. Soc. Esp. Ceram. V. 2005, 44, 135–145. [Google Scholar] [CrossRef]
- Ching, W.Y.; Rulis, P.; Misra, A. Ab initio elastic properties and tensile strength of crystalline hydroxyapatite. Acta Biomater. 2009, 5, 3067–3075. [Google Scholar] [CrossRef]
- Hardness of a material usually is considered as a measure of the resistance to permanent indentation. A standardized force or weight is applied to an indenter pressing into the surface of a material to create a symmetrical shaped indentation [179]
- Poisson’s ratio (ν), named after Siméon Poisson, is the ratio, when a sample object is stretched, of the contraction or transverse strain (perpendicular to the applied load), to the extension or axial strain (in the direction of the applied load)
- Burger, E.L.; Patel, V. Calcium phosphates as bone graft extenders. Orthopedics 2007, 30, 939–942. [Google Scholar]
- Rodriguez-Lorenzo, L.M.; Vallet-Regí, M.; Ferreira, J.M.F.; Ginebra, M.P.; Aparicio, C.; Planell, J. A hydroxyapatite ceramic bodies with tailored mechanical properties for different applications. J. Biomed. Mater. Res. 2002, 60, 159–166. [Google Scholar]
- Dorozhkin, S.V. Calcium orthophosphate-based biocomposites and hybrid biomaterials. J. Mater. Sci. 2009, 44, 2343–2387. [Google Scholar] [CrossRef]
- Bouslama, N.; Ben Ayed, F.; Bouaziz, J. Sintering and mechanical properties of tricalcium phosphate – fluorapatite composites. Ceram. Int. 2009, 35, 1909–1917. [Google Scholar] [CrossRef]
- Suchanek, W.; Yashima, M.; Kakihana, M.; Yoshimura, M. Processing and mechanical properties of hydroxyapatite reinforced with hydroxyapatite whiskers. Biomaterials 1996, 17, 1715–1723. [Google Scholar] [CrossRef]
- Suchanek, W.; Yashima, M.; Kakihana, M.; Yoshimura, M. Hydroxyapatite/hydroxyapatite-whisker composites without sintering additives: Mechanical properties and microstructural evolution. J. Am. Ceram. Soc. 1997, 80, 2805–2813. [Google Scholar]
- Simsek, D.; Ciftcioglu, R.; Guden, M.; Ciftcioglu, M.; Harsa, S. Mechanical properties of hydroxyapatite composites reinforced with hydroxyapatite whiskers. Key Eng. Mater. 2004, 264-268, 1985–1988. [Google Scholar] [CrossRef]
- Bose, S.; Banerjee, A.; Dasgupta, S.; Bandyopadhyay, A. Synthesis, processing, mechanical, and biological property characterization of hydroxyapatite whisker-reinforced hydroxyapatite composites. J. Am. Ceram. Soc. 2009, 92, 323–330. [Google Scholar] [CrossRef]
- Shiota, T.; Shibata, M.; Yasuda, K.; Matsuo, Y. Influence of β-tricalcium phosphate dispersion on mechanical properties of hydroxyapatite ceramics. J. Ceram. Soc. Jpn. 2009, 116, 1002–1005. [Google Scholar]
- Dorozhkin, S.V.; Ajaal, T. Toughening of porous bioceramic scaffolds by bioresorbable polymeric coatings. Proc. Inst. Mech. Eng. Part H: J. Eng. Med. 2009, 223, 459–470. [Google Scholar] [CrossRef]
- Dorozhkin, S.V.; Ajaal, T. Strengthening of dense bioceramic samples using bioresorbable polymers—A statistical approach. J. Biomim. Biomater. Tissue Eng. 2009, 4, 27–39. [Google Scholar] [CrossRef]
- He, L.H.; Standard, O.C.; Huang, T.T.; Latella, B.A.; Swain, M.V. Mechanical behaviour of porous hydroxyapatite. Acta Biomater. 2008, 4, 577–586. [Google Scholar] [CrossRef]
- Nagai, M.; Nishino, T. Surface conduction of porous hydroxyapatite ceramics at elevated temperatures. Solid State Ionics 1988, 28-30, 1456–1461. [Google Scholar] [CrossRef]
- Yamashita, K.; Owada, H.; Umegaki, T.; Kanazawa, T.; Futagamu, T. Ionic conduction in apatite solid solutions. Solid State Ionics 1988, 28-30, 660–663. [Google Scholar] [CrossRef]
- Mahabole, M.P.; Aiyer, R.C.; Ramakrishna, C.V.; Sreedhar, B.; Khairnar, R.S. Synthesis, characterization and gas sensing property of hydroxyapatite ceramic. Bull. Mater. Sci. 2005, 28, 535–545. [Google Scholar]
- Fanovich, M.A.; Castro, M.S.; Lopez, J.M.P. Analysis of the microstructural evolution in hydroxyapatite ceramics by electrical characterisation. Ceram. Int. 1999, 25, 517–522. [Google Scholar] [CrossRef]
- Valdes, J.J.P.; Rodriguez, A.V.; Carrio, J.G. Dielectric properties and structure of hydroxyapatite ceramics sintered by different conditions. J. Mater. Res. 1995, 10, 2174–2177. [Google Scholar] [CrossRef]
- Bensaoud, A.; Bouhaouss, A.; Ferhat, M. Electrical properties in compressed poorly crystalline apatite. J. Solid State Electrochem. 2001, 5, 362–365. [Google Scholar] [CrossRef]
- Tanaka, Y.; Nakamura, M.; Nagai, A.; Toyama, T.; Yamashita, K. Ionic conduction mechanism in Ca-deficient hydroxyapatite whiskers. Mater. Sci. Eng. B 2009, 161, 115–119. [Google Scholar] [CrossRef]
- Tanaka, Y.; Takata, S.; Shimoe, K.; Nakamura, M.; Nagai, A.; Toyama, T.; Yamashita, K. Conduction properties of non-stoichiometric hydroxyapatite whiskers for biomedical use. J. Ceram. Soc. Jpn. 2008, 116, 815–821. [Google Scholar] [CrossRef]
- Nakamura, S.; Takeda, H.; Yamashita, K. Proton transport polarization and depolarization of hydroxyapatite ceramics. J. Appl. Phys. 2001, 89, 5386–5392. [Google Scholar] [CrossRef]
- Gittings, J.P.; Bowen, C.R.; Turner, I.G.; Baxter, F.R.; Chaudhuri, J.B. Polarisation behaviour of calcium phosphate based ceramics. Mater. Sci. Forum 2008, 587-588, 91–95. [Google Scholar] [CrossRef]
- Itoh, S.; Nakamura, S.; Kobayashi, T.; Shinomiya, K.; Yamashita, K.; Itoh, S. Effect of electrical polarization of hydroxyapatite ceramics on new bone formation. Calcif. Tissue Int. 2006, 78, 133–142. [Google Scholar] [CrossRef]
- Iwasaki, T.; Tanaka, Y.; Nakamura, M.; Nagai, A.; Hashimoto, K.; Toda, Y.; Katayama, K.; Yamashita, K. Rate of bonelike apatite formation accelerated on polarized porous hydroxyapatite. J. Am. Ceram. Soc. 2008, 91, 3943–3949. [Google Scholar] [CrossRef]
- Itoh, S.; Nakamura, S.; Kobayashi, T.; Shinomiya, K.; Yamashita, K. Enhanced bone ingrowth into hydroxyapatite with interconnected pores by electrical polarization. Biomaterials 2006, 27, 5572–5579. [Google Scholar] [CrossRef]
- Kobayashi, T.; Itoh, S.; Nakamura, S.; Nakamura, M.; Shinomiya, K.; Yamashita, K. Enhanced bone bonding of hydroxyapatite-coated titanium implants by electrical polarization. J. Biomed. Mater. Res. A 2007, 82A, 145–151. [Google Scholar] [CrossRef]
- Bodhak, S.; Bose, S.; Bandyopadhyay, A. Role of surface charge and wettability on early stage mineralization and bone cell-materials interactions of polarized hydroxyapatite. Acta Biomater. 2009, 5, 2178–2188. [Google Scholar] [CrossRef]
- Yamashita, K.; Oikawa, N.; Umegaki, T. Acceleration and deceleration of bone-like crystal growth on ceramic hydroxyapatite by electric poling. Chem. Mater. 1996, 8, 2697–2700. [Google Scholar] [CrossRef]
- Teng, N.C.; Nakamura, S.; Takagi, Y.; Yamashita, Y.; Ohgaki, M.; Yamashita, K. A new approach to enhancement of bone formation by electrically polarized hydroxyapatite. J. Dental Res. 2001, 80, 1925–1929. [Google Scholar] [CrossRef]
- Kobayashi, T.; Nakamura, S.; Yamashita, K. Enhanced osteobonding by negative surface charges of electrically polarized hydroxyapatite. J. Biomed. Mater. Res. 2001, 57, 477–484. [Google Scholar] [CrossRef]
- Park, Y.J.; Hwang, K.S.; Song, J.E.; Ong, J.L.; Rawls, H.R. Growth of calcium phosphate on poling treated ferroelectric BaTiO3 ceramics. Biomaterials 2002, 23, 3859–3864. [Google Scholar]
- Hwang, K.S.; Song, J.E.; Jo, J.W.; Yang, H.S.; Park, Y.J.; Ong, J.L.; Rawls, H.R. Effect of poling conditions on growth of calcium phosphate crystal in ferroelectric BaTiO3 ceramics. J. Mater. Sci. Mater. Med. 2002, 13, 133–138. [Google Scholar] [CrossRef]
- Yamashita, K. Enhanced bioactivity of electrically poled hydroxyapatita ceramics and coatings. Mater. Sci. Forum 2003, 426-432, 3237–3242. [Google Scholar]
- Nakamura, S.; Kobayashi, T.; Yamashita, K. Highly orientated calcification in newly formed bones on negatively charged hydroxyapatite electrets. Key Eng. Mater. 2005, 284-286, 897–900. [Google Scholar] [CrossRef]
- Kato, R.; Nakamura, S.; Katayama, K.; Yamashita, K. Electrical polarization of plasma-spray-hydroxyapatite coatings for improvement of osteoconduction of implants. J. Biomed. Mater. Res. A 2005, 74A, 652–658. [Google Scholar] [CrossRef]
- Nakamura, S.; Kobayashi, T.; Nakamura, M.; Itoh, S.; Yamashita, K. Electrostatic surface charge acceleration of bone ingrowth of porous hydroxyapatite/β-tricalcium phosphate ceramics. J. Biomed. Mater. Res. A 2010, 92A, 267–275. [Google Scholar] [CrossRef]
- Itoh, S.; Nakamura, S.; Nakamura, M.; Shinomiya, K.; Yamashita, K. Enhanced bone regeneration by electrical polarization of hydroxyapatite. Artif. Organs 2006, 30, 863–869. [Google Scholar]
- Nakamura, M.; Nagai, A.; Ohashi, N.; Tanaka, Y.; Sekilima, Y.; Nakamura, S. Regulation of osteoblast-like cell behaviors on hydroxyapatite by electrical polarization. Key Eng. Mater. 2008, 361-363, 1055–1058. [Google Scholar] [CrossRef]
- Nakamura, M.; Nagai, A.; Tanaka, Y.; Sekilima, Y.; Yamashita, K. Polarized hydroxyapatite promotes spread and motility of osteoblastic cells. J. Biomed. Mater. Res. A 2010, 92A, 783–790. [Google Scholar]
- Nakamura, S.; Kobayashi, T.; Yamashita, K. Extended bioactivity in the proximity of hydroxyapatite ceramic surfaces induced by polarization charges. J. Biomed. Mater. Res. 2002, 61, 593–599. [Google Scholar] [CrossRef]
- Wang, W.; Itoh, S.; Tanaka, Y.; Nagai, A.; Yamashita, K. Comparison of enhancement of bone ingrowth into hydroxyapatite ceramics with highly and poorly interconnected pores by electrical polarization. Acta Biomater. 2009, 5, 3132–3140. [Google Scholar] [CrossRef]
- Nakamura, M.; Niwa, K.; Nakamura, S.; Sekijima, Y.; Yamashita, K. Interaction of a blood coagulation factor on electrically polarized hydroxyapatite surfaces. J. Biomed. Mater. Res. B Appl. Biomater. 2007, 82B, 29–36. [Google Scholar]
- Nagai, M.; Shibuya, Y.; Nishino, T.; Saeki, T.; Owada, H.; Yamashita, K.; Umegaki, T. Electrical conductivity of calcium phosphate ceramics with various Ca/P ratios. J. Mater. Sci. 1991, 26, 2949–2953. [Google Scholar] [CrossRef]
- Laghzizil, A.; Elherch, N.; Bouhaouss, A.; Lorente, G.; Coradin, T.; Livage, J. Electrical behavior of hydroxyapatites M10(PO4)6(OH)2 (M = Ca, Pb, Ba). Mater. Res. Bull. 2001, 36, 953–962. [Google Scholar]
- Louati, B.; Guidara, K.; Gargouri, M. Dielectric and ac ionic conductivity investigations in the monetite. J. Alloys Compd. 2009, 472, 347–351. [Google Scholar] [CrossRef]
- Gittings, J.P.; Bowen, C.R.; Dent, A.C.; Turner, I.G.; Baxter, F.R.; Chaudhuri, J.B. Electrical characterization of hydroxyapatite-based bioceramics. Acta Biomater. 2009, 5, 743–754. [Google Scholar] [CrossRef]
- Tofail, S.A.M.; Baldisserri, C.; Haverty, D.; McMonagle, J.B.; Erhart, J. Pyroelectric surface charge in hydroxyapatite ceramics. J. Appl. Phys. 2009, 106, 106104. [Google Scholar] [CrossRef]
- Fang, Y.; Agrawal, D.K.; Roy, D.M.; Roy, R. Fabrication of transparent hydroxyapatite ceramics by ambient-pressure sintering. Mater. Lett. 1995, 23, 147–151. [Google Scholar] [CrossRef]
- Kotobuki, N.; Kawagoe, D.; Fujimori, H.; Goto, S.; Loku, K.; Ohgushi, H. In vitro osteogenic activity of rat bone marrow derived mesenchymal stem cells cultured on transparent hydroxyapatite ceramics. Key Eng. Mater. 2004, 254-256, 1055–1058. [Google Scholar] [CrossRef]
- Kotobuki, N.; Ioku, K.; Kawagoe, D.; Nomura, D.; Fujimori, H.; Goto, S.; Ohgushi, H. In vitro osteogenic activity of rat mesenchymal cells cultured on transparent β-tricalcium phosphate ceramics. Key Eng. Mater. 2005, 284-286, 663–666. [Google Scholar] [CrossRef]
- Varma, H.; Vijayan, S.P.; Babu, S.S. Transparent hydroxyapatite ceramics through gel-casting and low-temperature sintering. J. Am. Ceram. Soc. 2002, 85, 493–495. [Google Scholar] [CrossRef]
- Watanabe, Y.; Ikoma, T.; Monkawa, A.; Suetsugu, Y.; Yamada, H.; Tanaka, J.; Moriyoshi, Y. Fabrication of transparent hydroxyapatite sintered body with high crystal orientation by pulse electric current sintering. J. Am. Ceram. Soc. 2005, 88, 243–245. [Google Scholar]
- Kotobuki, N.; Ioku, K.; Kawagoe, D.; Fujimori, H.; Goto, S.; Ohgushi, H. Observation of osteogenic differentiation cascade of living mesenchymal stem cells on transparent hydroxyapatite ceramics. Biomaterials 2005, 26, 779–785. [Google Scholar]
- John, A.; Varma, H.K.; Vijayan, S.; Bernhardt, A.; Lode, A.; Vogel, A.; Burmeister, B.; Hanke, T.; Domaschke, H.; Gelinsky, M. In vitro investigations of bone remodeling on a transparent hydroxyapatite ceramic. Biomed. Mater. 2009, 4, 015007–015015. [Google Scholar]
- Kotobuki, N.; Kawagoe, D.; Nomura, D.; Katou, Y.; Muraki, K.; Fujimori, H.; Goto, S.; Ioku, K.; Ohgushi, H. Observation and quantitative analysis of rat bone marrow stromal cells cultured in vitro on newly formed transparent β-tricalcium phosphate. J. Mater. Sci. Mater. Med. 2006, 17, 33–41. [Google Scholar] [CrossRef]
- Tancred, D.C.; McCormack, B.A.; Carr, A.J. A synthetic bone implant macroscopically identical to cancellous bone. Biomaterials 1998, 19, 2303–2311. [Google Scholar] [CrossRef]
- Miao, X.; Sun, D. Graded/gradient porous biomaterials. Materials 2010, 3, 26–47. [Google Scholar] [CrossRef]
- Bucholz, R.W.; Carlton, A.; Holmes, R. Interporous hydroxyapatite as a bone graft substitute in tibial plateau fractures. Clin. Orthop. 1989, 240, 53–62. [Google Scholar]
- Cavagna, R.; Daculsi, G.; Bouler, J.M. Macroporous calcium phosphate ceramic: A prospective study of 106 cases in lumbar spinal fusion. J. Long Term Eff. Med. Implants 1999, 9, 403–412. [Google Scholar]
- Lu, J.X.; Flautre, B.; Anselme, K.; Hardouin, P.; Gallur, A.; Descamps, M.; Thierry, B. Role of interconnections in porous bioceramics on bone recolonization in vitro and in vivo. J. Mater. Sci. Mater. Med. 1999, 10, 111–120. [Google Scholar] [CrossRef]
- Ayers, R.A.; Simske, S.J.; Nunes, C.R.; Wolford, L.M. Long-term bone ingrowth and residual microhardness of porous block hydroxyapatite implants in humans. J. Oral Maxillof. Surg. 1998, 56 Suppl. 5, 1297–1302. [Google Scholar] [CrossRef]
- Jones, A.C.; Arns, C.H.; Sheppard, A.P.; Hutmacher, D.W.; Milthorpe, B.K.; Knackstedt, M.A. Assessment of bone ingrowth into porous biomaterials using MICRO-CT. Biomaterials 2007, 28, 2491–2504. [Google Scholar]
- Karageorgiou, V.; Kaplan, D. Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials 2005, 26, 5474–5491. [Google Scholar] [CrossRef]
- Ohgushi, H.; Goldberg, V.M.; Caplan, A.I. Heterotopic osteogenesis in porous ceramics induced by marrow cells. J. Orthop. Res. 1989, 7, 568–578. [Google Scholar] [CrossRef]
- Cheung, H.S.; Haak, M.H. Growth of osteoblasts on porous calcium phosphate ceramic: An in vitro model for biocompatibility study. Biomaterials 1989, 10, 63–67. [Google Scholar] [CrossRef]
- Zyman, Z.; Ivanov, I.; Glushko, V.; Dedukh, N.; Malyshkina, S. Inorganic phase composition of remineralisation in porous CaP ceramics. Biomaterials 1998, 19, 1269–1273. [Google Scholar] [CrossRef]
- Yoshikawa, T.; Ohgushi, H.; Tamai, S. Immediate bone forming capability of prefabricated osteogenic hydroxyapatite. J. Biomed. Mater. Res. 1996, 32, 481–492. [Google Scholar] [CrossRef]
- Chang, B.S.; Lee, C.K.; Hong, K.S.; Youn, H.J.; Ryu, H.S.; Chung, S.S.; Park, K. Osteoconduction at porous hydroxyapatite with various pore configurations. Biomaterials 2000, 21, 1291–1298. [Google Scholar]
- Flautre, B.; Descamps, M.; Delecourt, C.; Blary, M.C.; Hardouin, P. Porous HA ceramic for bone replacement: Role of the pores and interconnections-experimental study in the rabbits. J. Mater. Sci. Mater. Med. 2001, 12, 679–682. [Google Scholar] [CrossRef]
- McAfee, P.C.; Cunningham, B.W.; Orbegoso, D.O.; Sefter, J.C.; Dmitriev, A.E.; Fedder, I.L. Analysis of porous ingrowth in intervertebral disc prostheses. Spine 2003, 28, 332–340. [Google Scholar]
- Tamai, N.; Myoui, A.; Tomita, T.; Nakase, T.; Tanaka, J.; Ochi, T.; Yoshikawa, H. Novel hydroxyapatite ceramics with an interconnective porous structure exhibit superior osteoconduction in vivo. J. Biomed. Mater. Res. 2002, 59, 110–117. [Google Scholar]
- Mastrogiacomo, M.; Scaglione, S.; Martinetti, R.; Dolcini, L.; Beltrame, F.; Cancedda, R.; Quarto, R. Role of scaffold internal structure on in vivo bone formation in macroporous calcium phosphate bioceramics. Biomaterials 2006, 27, 3230–3237. [Google Scholar]
- Okamoto, M.; Dohi, Y.; Ohgushi, H.; Shimaoka, H.; Ikeuchi, M.; Matsushima, A.; Yonemasu, K.; Hosoi, H. Influence of the porosity of hydroxyapatite ceramics on in vitro and in vivo bone formation by cultured rat bone marrow stromal cells. J. Mater. Sci. Mater. Med. 2006, 17, 327–336. [Google Scholar] [CrossRef]
- Zhang, L.; Hanagata, N.; Maeda, M.; Minowa, T.; Ikoma, T.; Fan, H.; Zhang, X. Porous hydroxyapatite and biphasic calcium phosphate ceramics promote ectopic osteoblast differentiation from mesenchymal stem cells. Sci. Technol. Adv. Mater. 2009, 10, 025003–025011. [Google Scholar] [CrossRef]
- Omae, H.; Mochizuki, Y.; Yokoya, S.; Adachi, N.; Ochi, M. Effects of interconnecting porous structure of hydroxyapatite ceramics on interface between grafted tendon and ceramics. J. Biomed. Mater. Res. A 2006, 79A, 329–337. [Google Scholar] [CrossRef]
- Yoshikawa, H.; Tamai, N.; Murase, T.; Myoui, A. Interconnected porous hydroxyapatite ceramics for bone tissue engineering. J. R. Soc. Interface 2009, 6, S341–S348. [Google Scholar] [CrossRef]
- Takagi, S.; Chow, L.C. Formation of macropores in calcium phosphate cement implants. J. Biomed. Mater. Res. 2001, 12, 135–139. [Google Scholar]
- Walsh, D.; Tanaka, J. Preparation of a bone-like apatite foam cement. J. Mater. Sci. Mater. Med. 2001, 12, 339–344. [Google Scholar] [CrossRef]
- Chevalier, E.; Chulia, D.; Pouget, C.; Viana, M. Fabrication of porous substrates: A review of processes using pore forming agents in the biomaterial field. J. Pharm. Sci. 2008, 97, 1135–1154. [Google Scholar] [CrossRef]
- Komlev, V.S.; Barinov, S.M. Porous hydroxyapatite ceramics of bi-modal pore size distribution. J. Mater. Sci. Mater. Med. 2002, 13, 295–299. [Google Scholar] [CrossRef]
- Sepulveda, P.; Binner, J.G.; Rogero, S.O.; Higa, O.Z.; Bressiani, J.C. Production of porous hydroxyapatite by the gel-casting of foams and cytotoxic evaluation. J. Biomed. Mater. Res. 2000, 50, 27–34. [Google Scholar] [CrossRef]
- Hsu, Y.H.; Turner, I.G.; Miles, A.W. Mechanical characterization of dense calcium phosphate bioceramics with interconnected porosity. J. Mater. Sci. Mater. Med. 2007, 18, 2319–2329. [Google Scholar] [CrossRef]
- Zhang, H.G.; Zhu, Q. Preparation of porous hydroxyapatite with interconnected pore architecture. J. Mater. Sci. Mater. Med. 2007, 18, 1825–1829. [Google Scholar] [CrossRef]
- le Ray, A.M.; Gautier, H.; Bouler, J.M.; Weiss, P.; Merle, C. A new technological procedure using sucrose as porogen compound to manufacture porous biphasic calcium phosphate ceramics of appropriate micro- and macrostructure. Ceram. Int. 2010, 36, 93–101. [Google Scholar] [CrossRef] [Green Version]
- Guda, T.; Appleford, M.; Oh, S.; Ong, J.L. A cellular perspective to bioceramic scaffolds for bone tissue engineering: The state of the art. Curr. Topics Med. Chem. 2008, 8, 290–299. [Google Scholar] [CrossRef]
- Ota, Y.; Kasuga, T.; Abe, Y. Preparation and compressive strength behaviour of porous ceramics with β-Ca3(PO3)2 fiber skeletons. J. Am. Ceram. Soc. 1997, 80, 225–231. [Google Scholar] [CrossRef]
- White, E.; Shors, E.C. Biomaterial aspects of Interpore-200 porous hydroxyapatite. Dent. Clin. North Am. 1986, 30, 49–67. [Google Scholar]
- Descamps, M.; Duhoo, T.; Monchau, F.; Lu, J.; Hardouin, P.; Hornez, J.C.; Leriche, A. Manufacture of macroporous β-tricalcium phosphate bioceramics. J. Eur. Ceram. Soc. 2008, 28, 149–157. [Google Scholar] [CrossRef]
- Engin, N.O.; Tas, A.C. Manufacture of macroporous calcium hydroxyapatite bioceramics. J. Eur. Ceram. Soc. 1999, 19, 2569–2572. [Google Scholar]
- Liu, D.M. Fabrication of hydroxyapatite ceramic with controlled porosity. J. Mater. Sci. Mater. Med. 1997, 8, 227–232. [Google Scholar] [CrossRef]
- Rivera-Munoz, E.; Diaz, J.R.; Rodriguez, J.R.; Brostow, W.; Castano, V.M. Hydroxyapatite spheres with controlled porosity for eye ball prosthesis: Processing and characterization. J. Mater. Sci. Mater. Med. 2001, 12, 305–311. [Google Scholar] [CrossRef]
- Hosoi, K.; Hashida, T.; Takahashi, H.; Yamasaki, N.; Korenaga, T. New processing technique for hydroxyapatite ceramics by the hydrothermal hot-pressing method. J. Am. Ceram. Soc. 1996, 79, 2771–2774. [Google Scholar]
- Walsh, D.; Hopwood, J.D.; Mann, S. Crystal tectonics: Construction of reticulated calcium phosphate frameworks in bicontinuous reverse microemulsions. Science 1994, 264, 1576–1578. [Google Scholar]
- Walsh, D.; Mann, S. Chemical synthesis of microskeletal calcium phosphate in bicontinuous microemulsions. Chem. Mater. 1996, 8, 1944–1953. [Google Scholar] [CrossRef]
- Charriere, E.; Lemaitre, J.; Zysset, P. Hydroxyapatite cement scaffolds with controlled macroporosity: Fabrication protocol and mechanical properties. Biomaterials 2003, 24, 809–817. [Google Scholar] [CrossRef]
- Zhou, L.; Wang, D.; Huang, W.; Yao, A.; Kamitakahara, M.; Ioku, K. Preparation and characterization of periodic porous frame of hydroxyapatite. J. Ceram. Soc. Jpn. 2009, 117, 521–524. [Google Scholar] [CrossRef]
- Moroni, L.; De Wijn, J.R.; Van Blitterswijk, C.A. Integrating novel Technologies to fabricate smart scaffolds. J. Biomater. Sci. Polymer Ed. 2008, 19, 543–572. [Google Scholar] [CrossRef]
- Aizawa, M.; Howell, S.F.; Itatani, K.; Yokogawa, Y.; Nishizawa, K.; Toriyama, M.; Kameyama, T. Fabrication of porous ceramics with well-controlled open pores by sintering of fibrous hydroxyapatite particles. J. Ceram. Soc. Jpn. 2000, 108, 249–253. [Google Scholar] [CrossRef]
- Kawata, M.; Uchida, H.; Itatani, K.; Okada, I.; Koda, S.; Aizawa, M. Development of porous ceramics with well-controlled porosities and pore sizes from apatite fibers and their evaluations. J. Mater. Sci. Mater. Med. 2004, 15, 817–823. [Google Scholar] [CrossRef]
- Li, S.H.; De Wijn, J.R.; Layrolle, P.; De Groot, K. Novel method to manufacture porous hydroxyapatite by dual-phase mixing. J. Am. Ceram. Soc. 2003, 86, 65–72. [Google Scholar] [CrossRef]
- Koh, Y.H.; Kim, H.W.; Kim, H.E.; Halloran, J.W. Fabrication of macrochannelled-hydroxyapatite bioceramic by a coextrusion process. J. Am. Ceram. Soc. 2002, 85, 2578–2580. [Google Scholar] [CrossRef]
- Nakahira, A.; Tamai, M.; Sakamoto, K.; Yamaguchi, S. Sintering and microstructure of porous hydroxyapatite. J. Ceram. Soc. Jpn. 2000, 108, 99–104. [Google Scholar] [CrossRef]
- De Sousa, F.C.G.; Evans, J.R.G. Tubular hydroxyapatite scaffolds. Adv. Appl. Ceram. 2005, 104, 30–34. [Google Scholar] [CrossRef]
- Kitamura, M.; Ohtsuki, C.; Ogata, S.I.; Kamitakahara, M.; Tanihara, M.; Miyazaki, T. Mesoporous calcium phosphate via post-treatment of α-TCP. J. Am. Ceram. Soc. 2005, 88, 822–826. [Google Scholar] [CrossRef]
- Walsh, D.; Boanini, E.; Tanaka, J.; Mann, S. Synthesis of tri-calcium phosphate sponges by interfacial deposition and thermal transformation of self-supporting calcium phosphate films. J. Mater. Chem. 2005, 15, 1043–1048. [Google Scholar] [CrossRef]
- Gonzalez-McQuire, R.; Green, D.; Walsh, D.; Hall, S.; Chane-Ching, J.Y.; Oreffo, R.O.C.; Mann, S. Fabrication of hydroxyapatite sponges by dextran sulphate/amino acid templating. Biomaterials 2005, 26, 6652–6656. [Google Scholar] [CrossRef] [Green Version]
- Ioku, K.; Kawachi, G.; Nakahara, K.; Ishida, E.H.; Minagi, H.; Okuda, T.; Yonezawa, I.; Kurosawa, H.; Ikeda, T. Porous granules of β-tricalcium phosphate composed of rod-shaped particles. Key Eng. Mater. 2006, 309-311, 1059–1062. [Google Scholar]
- Kitamura, M.; Ohtsuki, C.; Ogata, S.; Kamitakahara, M.; Tanihara, M. Microstructure and bioresorbable properties of α-TCP ceramic porous body fabricated by direct casting method. Mater. Trans. 2004, 45, 983–988. [Google Scholar] [CrossRef]
- Ramay, H.R.; Zhang, M. Preparation of porous hydroxyapatite scaffolds by combination of the gel-casting and polymer sponge methods. Biomaterials 2003, 24, 3293–3302. [Google Scholar] [CrossRef]
- Potoczek, M.; Zima, A.; Paszkiewicz, Z.; Ślósarczyk, A. Manufacturing of highly porous calcium phosphate bioceramics via gel-casting using agarose. Ceram. Int. 2009, 35, 2249–2254. [Google Scholar] [CrossRef]
- Xu, S.; Li, D.; Lu, B.; Tang, Y.; Wang, C.; Wang, Z. Fabrication of a calcium phosphate scaffold with a three dimensional channel network and its application to perfusion culture of stem cells. Rapid Prototyping J. 2007, 13, 99–106. [Google Scholar] [CrossRef]
- Li, S.H.; De Wijn, J.R.; Layrolle, P.; De Groot, K. Synthesis of macroporous hydroxyapatite scaffolds for bone tissue engineering. J. Biomed. Mater. Res. 2002, 61, 109–120. [Google Scholar] [CrossRef]
- Almirall, A.; Larrecq, G.; Delgado, J.A.; Martínez, S.; Planell, J.A.; Ginebra, M.P. Fabrication of low temperature macroporous hydroxyapatite scaffolds by foaming and hydrolysis of an α-TCP paste. Biomaterials 2004, 25, 3671–3680. [Google Scholar] [CrossRef]
- Kamitakahara, M.; Ohtsuki, C.; Kawachi, G.; Wang, D.; Ioku, K. Preparation of hydroxyapatite porous ceramics with different porous structures using a hydrothermal treatment with different aqueous solutions. J. Ceram. Soc. Jpn. 2008, 116, 6–9. [Google Scholar] [CrossRef]
- Peña, J.; Román, J.; Cabañas, M.V.; Vallet-Regí, M. An alternative technique to shape scaffolds with hierarchical porosity at physiological temperature. Acta Biomater. 2010, 6, 1288–1296. [Google Scholar] [CrossRef]
- Marcassoli, P.; Cabrini, M.; Tirillò, J.; Bartuli, C.; Palmero, P.; Montanaro, L. Mechanical characterization of hydroxyapatite micro/macro-porous ceramics obtained by means of innovative gel-casting process. Key Eng. Mater. 2010, 417-418, 565–568. [Google Scholar]
- Nakamura, S.; Nakahira, A. Synthesis and evaluation of porous hydroxyapatite prepared by hydrothermal treatment and subsequent sintering method. J. Ceram. Soc. Jpn. 2008, 116, 42–45. [Google Scholar] [CrossRef]
- Song, H.Y.; Islam, S.; Lee, B.T. A novel method to fabricate unidirectional porous hydroxyapatite body using ethanol bubbles in a viscous slurry. J. Am. Ceram. Soc. 2008, 91, 3125–3127. [Google Scholar] [CrossRef]
- Impens, S.; Schelstraete, R.; Luyten, J.; Mullens, S.; Thijs, I.; Van Humbeeck, J.; Schrooten, J. Production and characterisation of porous calcium phosphate structures with controllable hydroxyapatite/β-tricalcium phosphate ratios. Adv. Appl. Ceram. 2009, 108, 494–500. [Google Scholar] [CrossRef]
- Von Doernberg, M.C.; Von Rechenberg, B.; Bohner, M.; Grünenfelder, S.; Van Lenthe, G.H.; Müller, R.; Gasser, B.; Mathys, R.; Baroud, G.; Auer, J. In vivo behavior of calcium phosphate scaffolds with four different pore sizes. Biomaterials 2006, 27, 5186–5198. [Google Scholar] [CrossRef]
- Mygind, T.; Stiehler, M.; Baatrup, A.; Li, H.; Zou, X.; Flyvbjerg, A.; Kassem, M.; Bunger, C. Mesenchymal stem cell ingrowth and differentiation on coralline hydroxyapatite scaffolds. Biomaterials 2007, 28, 1036–1047. [Google Scholar] [CrossRef]
- Tsuruga, E.; Takita, H.; Wakisaka, Y.; Kuboki, Y. Pore size of porous hydoxyapatite as the cell-substratum controls BMP-induced osteogenesis. J. Biochem. 1997, 121, 317–324. [Google Scholar] [CrossRef]
- Hing, K.; Annaz, B.; Saeed, S.; Revell, P.; Buckland, T. Microporosity enhances bioactivity of synthetic bone graft substitutes. J. Mater. Sci. Mater. Med. 2005, 16, 467–475. [Google Scholar] [CrossRef]
- Woodard, J.R.; Hilldore, A.J.; Lan, S.K.; Park, C.J.; Morgan, A.W.; Eurell, J.A.C.; Clark, S.G.; Wheeler, M.B.; Jamison, R.D.; Wagoner, J.A.J. The mechanical properties and osteoconductivity of hydroxyapatite bone scaffolds with multi-scale porosity. Biomaterials 2007, 28, 45–54. [Google Scholar] [CrossRef]
- Ruksudjarit, A.; Pengpat, K.; Rujijanagul, G.; Tunkasiri, T. The fabrication of nanoporous hydroxyapatite ceramics. Adv. Mater. Res. 2008, 47-50, 797–800. [Google Scholar] [CrossRef]
- LeGeros, R.Z. Calcium phosphate-based osteoinductive materials. Chem. Rev. 2008, 108, 4742–4753. [Google Scholar] [CrossRef]
- Prokopiev, O.; Sevostianov, I. Dependence of the mechanical properties of sintered hydroxyapatite on the sintering temperature. Mater. Sci. Eng. A 2006, 431, 218–227. [Google Scholar] [CrossRef]
- Daculsi, G.; Jegoux, F.; Layrolle, P. The micro macroporous biphasic calcium phosphate concept for bone reconstruction and tissue engineering. In Advanced Biomaterials: Fundamentals, Processing and Applications; Basu, B., Katti, D.S., Kumar, A., Eds.; Wiley-American Ceramic Society: oken, NJ, USA, 2009; p. 768. [Google Scholar]
- Tadic, D.; Beckmann, F.; Schwarz, K.; Epple, M. A novel method to produce hydroxylapatite objects with interconnecting porosity that avoids sintering. Biomaterials 2004, 25, 3335–3340. [Google Scholar] [CrossRef]
- Woyansky, J.S.; Scott, C.E.; Minnear, W.P. Processing of porous ceramics. Am. Ceram. Soc. Bull. 1992, 71, 1674–1682. [Google Scholar]
- Vaz, L.; Lopes, A.B.; Almeida, M. Porosity control of hydroxyapatite implants. J. Mater. Sci. Mater. Med. 1999, 10, 239–242. [Google Scholar] [CrossRef]
- Yao, X.; Tan, S.; Jiang, D. Improving the properties of porous hydroxyapatite ceramics by fabricating methods. J. Mater. Sci. 2005, 40, 4939–4942. [Google Scholar] [CrossRef]
- Zhang, J.; Fujiwara, M.; Xu, Q.; Zhu, Y.; Iwasa, M.; Jiang, D. Synthesis of mesoporous calcium phosphate using hybrid templates. Micropor. Mesopor. Mater. 2008, 111, 411–416. [Google Scholar] [CrossRef]
- Simske, S.J.; Ayers, R.A.; Bateman, T.A. Porous materials for bone engineering. Mater. Sci. Forum 1997, 250, 151–182. [Google Scholar] [CrossRef]
- Wang, H.; Zhai, L.; Li, Y.; Shi, T. Preparation of irregular mesoporous hydroxyapatite. Mater. Res. Bull. 2008, 43, 1607–1614. [Google Scholar] [CrossRef]
- Cyster, L.A.; Grant, D.M.; Howdle, S.M.; Rose, F.R.A.J.; Irvine, D.J.; Freeman, D.; Scotchford, C.A.; Shakesheff, K.M. The influence of dispersant concentration on the pore morphology of hydroxyapatite ceramics for bone tissue engineering. Biomaterials 2005, 26, 697–702. [Google Scholar] [CrossRef]
- Tian, J.; Tian, J. Preparation of porous hydroxyapatite. J. Mater. Sci. 2001, 36, 3061–3066. [Google Scholar] [CrossRef]
- Rodriguez-Lorenzo, L.M.; Vallet-Regí, M.; Ferreira, J.M.F. Fabrication of porous hydroxyapatite bodies by a new direct consolidation method: Starch consolidation. J. Biomed. Mater. Res. 2002, 60, 232–240. [Google Scholar] [CrossRef]
- Lee, E.J.; Koh, Y.H.; Yoon, B.H.; Kim, H.E.; Kim, H.W. Highly porous hydroxyapatite bioceramics with interconnected pore channels using camphene-based freeze casting. Mater. Lett. 2007, 61, 2270–2273. [Google Scholar] [CrossRef]
- Jones, J.R.; Hench, L.L. Regeneration of trabecular bone using porous ceramics. Curr. Opin. Solid State Mater. Sci. 2003, 7, 301–307. [Google Scholar] [CrossRef]
- Ota, T.; Eitsuka, T.; Yoshida, H.; Adachi, N. Porous apatite ceramics derived from woods. Adv. Mater. Res. 2006, 11-12, 247–250. [Google Scholar] [CrossRef]
- Tas, A.C. Preparation of porous apatite granules from calcium phosphate cement. J. Mater. Sci. Mater. Med. 2008, 19, 2231–2239. [Google Scholar] [CrossRef]
- Fu, Q.; Rahaman, M.N.; Dogan, F.; Bal, B.S. Freeze casting of porous hydroxyapatite scaffolds. I. Processing and general microstructure. J. Biomed. Mater. Res. B Appl. Biomater. 2008, 86B, 125–135. [Google Scholar] [CrossRef]
- Tang, Y.J.; Tang, Y.F.; Lv, C.T.; Zhou, Z.H. Preparation of uniform porous hydroxyapatite biomaterials by a new method. Appl. Surf. Sci. 2008, 254, 5359–5362. [Google Scholar] [CrossRef]
- Fan, J.; Lei, J.; Yu, C.; Tu, B.; Zhao, D. Hard-templating synthesis of a novel rod-like nanoporous calcium phosphate bioceramics and their capacity as antibiotic carriers. Mater. Chem. Phys. 2007, 103, 489–493. [Google Scholar] [CrossRef]
- Sopyan, I.; Mel, M.; Ramesh, S.; Khalid, K.A. Porous hydroxyapatite for artificial bone applications. Sci. Technol. Adv. Mater. 2007, 8, 116–123. [Google Scholar] [CrossRef]
- Rice, R.W. Porosity of ceramics. Marcel Dekker: New York, NY, USA, 1998; 560 pp. [Google Scholar]
- Fu, Y.C.; Ho, M.L.; Wu, S.C.; Hsieh, H.S.; Wang, C.K. Porous bioceramic bead prepared by calcium phosphate with sodium alginate gel and PE powder. Mater. Sci. Eng. C 2008, 28, 1149–1158. [Google Scholar] [CrossRef]
- Hsu, Y.H.; Turner, I.G.; Miles, A.W. Fabrication of porous bioceramics with porosity gradients similar to the bimodal structure of cortical and cancellous bone. J. Mater. Sci. Mater. Med. 2007, 18, 2251–2256. [Google Scholar] [CrossRef]
- Munch, E.; Franco, J.; Deville, S.; Hunger, P.; Saiz, E.; Tomsia, A.P. Porous ceramic scaffolds with complex architectures. JOM 2008, 60, 54–59. [Google Scholar]
- Macchetta, A.; Turner, I.G.; Bowen, C.R. Fabrication of HA/TCP scaffolds with a graded and porous structure using a camphene-based freeze-casting method. Acta Biomater. 2009, 5, 1319–1327. [Google Scholar] [CrossRef]
- Deville, S.; Saiz, E.; Tomsia, A.P. Freeze casting of hydroxyapatite scaffolds for bone tissue engineering. Biomaterials 2006, 27, 5480–5489. [Google Scholar] [CrossRef]
- Zuo, K.H.; Zeng, Y.P.; Jiang, D. Effect of polyvinyl alcohol additive on the pore structure and morphology of the freeze-cast hydroxyapatite ceramics. Mater. Sci. Eng. C 2010, 30, 283–287. [Google Scholar] [CrossRef]
- Yan, X.; Yu, C.; Zhou, X.; Tang, J.; Zhao, D. Highly ordered mesoporous bioactive glasses with superior in vitro bone-forming bioactivities. Angew. Chem. Int. Ed. Engl. 2004, 43, 5980–5984. [Google Scholar] [CrossRef]
- Izquierdo-Barba, I.; Ruiz-González, L.; Doadrio, J.C.; González-Calbet, J.M.; Vallet-Regí, M. Tissue regeneration: A new property of mesoporous materials. Solid State Sci. 2005, 7, 983–989. [Google Scholar] [CrossRef]
- Cosijns, A.; Vervaet, C.; Luyten, J.; Mullens, S.; Siepmann, F.; Van Hoorebeke, L.; Masschaele, B.; Cnudde, V.; Remon, J.P. Porous hydroxyapatite tablets as carriers for low-dosed drugs. Eur. J. Pharm. Biopharm. 2007, 67, 498–506. [Google Scholar] [CrossRef]
- Uchida, A.; Shinto, Y.; Araki, N.; Ono, K. Slow release of anticancer drugs from porous calcium hydroxyapatite ceramic. J. Orthop. Res. 1992, 10, 440–445. [Google Scholar] [CrossRef]
- Shinto, Y.; Uchida, A.; Korkusuz, F.; Araki, N.; Ono, K. Calcium hydroxyapatite ceramic used as a delivery system for antibiotics. J. Bone. Joint. Surg. Br. 1992, 74, 600–604. [Google Scholar]
- Martin, R.B.; Chapman, M.W.; Sharkey, N.A.; Zissimos, S.L.; Bay, B.; Shors, E.C. Bone ingrowth and mechanical properties of coralline hydroxyapatite 1 yr after implantation. Biomaterials 1993, 14, 341–348. [Google Scholar] [CrossRef]
- Kazakia, G.J.; Nauman, E.A.; Ebenstein, D.M.; Halloran, B.P.; Keaveny, T.M. Effects of in vitro bone formation on the mechanical properties of a trabeculated hydroxyapatite bone substitute. J. Biomed. Mater. Res. A 2006, 77A, 688–699. [Google Scholar] [CrossRef]
- Hing, K.A.; Best, S.M.; Tanner, K.E.; Bonfield, W.; Revell, P.A. Mediation of bone ingrowth in porous hydroxyapatite bone graft substitutes. J. Biomed. Mater. Res. A 2004, 68A, 187–200. [Google Scholar] [CrossRef]
- Vuola, J.; Taurio, R.; Goransson, H.; Asko-Seljavaara, S. Compressive strength of calcium carbonate and hydroxyapatite implants after bone marrow induced osteogenesis. Biomaterials 1998, 19, 223–227. [Google Scholar] [CrossRef]
- Yang, L.; Ning, X.; Chen, K.; Zhou, H. Preparation and properties of hydroxyapatite filters for microbial filtration. Ceram. Int. 2007, 33, 483–489. [Google Scholar] [CrossRef]
- Levitt, G.E.; Crayton, P.H.; Monroe, E.A.; Condrate, R.A. Forming methods for apatite prosthesis. J. Biomed. Mater. Res. 1969, 3, 683–685. [Google Scholar] [CrossRef]
- Tadic, D.; Epple, M. A thorough physicochemical characterization of 14 calcium phosphate-based bone substitution materials in comparison to natural bone. Biomaterials 2004, 25, 987–994. [Google Scholar] [CrossRef]
- Bodde, E.W.H.; Wolke, J.G.C.; Kowalski, R.S.Z.; Jansen, J.A. Bone regeneration of porous β-tricalcium phosphate (Conduit™ TCP) and of biphasic calcium phosphate ceramic (Biosel®) in trabecular defects in sheep. J. Biomed. Mater. Res. A 2007, 82A, 711–722. [Google Scholar] [CrossRef]
- le Guéhennec, L.; Layrolle, P.; Daculsi, G. A review of bioceramics and fibrin sealant. Eur. Cell Mater. 2004, 8, 1–11. [Google Scholar]
- LeGeros, R.Z. Properties of osteoconductive biomaterials: Calcium phosphates. Clin. Orthop. Rel. Res. 2002, 395, 81–98. [Google Scholar] [CrossRef]
- Jordan, D.R.; Gilberg, S.; Bawazeer, A. Coralline hydroxyapatite orbital implant (Bio-Eye): Experience with 158 patients. Ophthal. Plast. Reconstr. Surg. 2004, 20, 69–74. [Google Scholar] [CrossRef]
- Yoon, J.S.; Lew, H.; Kim, S.J.; Lee, S.Y. Exposure rate of hydroxyapatite orbital implants a 15-year experience of 802 cases. Ophthalmology 2008, 115, 566–572. [Google Scholar] [CrossRef]
- Schnettler, R.; Stahl, J.P.; Alt, V.; Pavlidis, T.; Dingeldein, E.; Wenisch, S. Calcium phosphate-based bone substitutes. Eur. J. Trauma 2004, 4, 219–229. [Google Scholar]
- Zyman, Z.Z.; Glushko, V.; Dedukh, N.; Malyshkina, S.; Ashukina, N. Porous calcium phosphate ceramic granules and their behaviour in differently loaded areas of skeleton. J. Mater. Sci. Mater. Med. 2008, 19, 2197–2205. [Google Scholar] [CrossRef]
- Constantz, B.R.; Ison, I.C.; Fulmer, M.T.; Poser, R.D.; Smith, S.T.; Vanwagoner, M.; Ross, J.; Goldstein, S.A.; Jupiter, J.B.; Rosenthal, D.I. Skeletal repair by in situ formation of the mineral phase of bone. Science 1995, 267, 1796–1799. [Google Scholar]
- Dorozhkin, S.V. Calcium orthophosphate cements for biomedical application. J. Mater. Sci. 2008, 43, 3028–3057. [Google Scholar] [CrossRef]
- Dorozhkin, S.V. Calcium orthophosphate cements and concretes. Materials 2009, 2, 221–291. [Google Scholar] [CrossRef]
- Chow, L.C. Next generation calcium phosphate-based biomaterials. Dent. Mater. J. 2009, 28, 1–10. [Google Scholar] [CrossRef]
- Del Real, R.P.; Wolke, J.G.C.; Vallet-Regí, M.; Jansen, J.A. A new method to produce macropores in calcium phosphate cements. Biomaterials 2002, 23, 3673–3680. [Google Scholar] [CrossRef]
- Victor, S.P.; Kumar, T.S.S. Processing and properties of injectable porous apatitic cements. J. Ceram. Soc. Jpn. 2008, 116, 105–107. [Google Scholar] [CrossRef]
- Hesaraki, S.; Nemati, R.; Nosoudi, N. Preparation and characterisation of porous calcium phosphate bone cement as antibiotic carrier. Adv. Appl. Ceram. 2009, 108, 231–240. [Google Scholar] [CrossRef]
- Weiss, D.D.; Sachs, M.A.; Woodard, C.R. Calcium phosphate bone cements: A comprehensive review. J. Long Term Eff. Med. Implants 2003, 13, 41–47. [Google Scholar] [CrossRef]
- Ambard, A.J.; Mueninghoff, L. Calcium phosphate cement: Review of mechanical and biological properties. J. Prosthodont. 2006, 15, 321–328. [Google Scholar] [CrossRef]
- Costantino, P.D.; Friedman, C.D.; Jones, K.; Chow, L.C.; Pelzer, H.J.; Sisson, G.A. Hydroxyapatite cement. I—Basic chemistry and histologic properties. Arch. Otolaryngol. Head Neck Surg. 1991, 117, 379–384. [Google Scholar] [CrossRef]
- Costantino, P.D.; Friedman, C.D.; Jones, K.; Chow, L.C.; Pelzer, H.J.; Sisson, G.A. Hydroxyapatite cement. II—Obliteration and reconstruction of the cat frontal sinus. Arch. Otolaryngol. Head Neck Surg. 1991, 117, 385–389. [Google Scholar] [CrossRef]
- De Groot, K.; Wolke, J.G.C.; Jansen, J.A. Calcium phosphate coatings for medical implants. Proc. Inst. Mech. Eng. Part H: J. Eng. Med. 1998, 212, 137–147. [Google Scholar] [CrossRef]
- Campbell, A.A. Bioceramics for implant coatings. Mater. Today 2003, 6, 26–30. [Google Scholar] [CrossRef]
- Epinette, J.A.M.D.; Geesink, R.G.T. Hydroxyapatite Coated Hip and Knee Arthroplasty; Elsevier: Amsterdam, The Netherlands, 1995; p. 394. [Google Scholar]
- Willmann, G. Coating of implants with hydroxyapatite—Material connections between bone and metal. Adv. Eng. Mater. 1999, 1, 95–105. [Google Scholar] [CrossRef]
- Schliephake, H.; Scharnweber, D.; Roesseler, S.; Dard, M.; Sewing, A.; Aref, A. Biomimetic calcium phosphate composite coating of dental implants. Int. J. Oral Max. Impl. 2006, 21, 738–746. [Google Scholar]
- Kokubo, T.; Kim, H.M.; Kawashita, M. Novel bioactive materials with different mechanical properties. Biomaterials 2003, 24, 2161–2175. [Google Scholar] [CrossRef]
- Habibovic, P.; Li, J.; Van der Valk, C. M.; Meijer, G.; Layrolle, P.; Van Blitterswijk, C.A.; De Groot, K. Biological performance of uncoated and octacalcium phosphate-coated Ti6Al4V. Biomaterials 2005, 26, 23–36. [Google Scholar] [CrossRef]
- Hahn, B.D.; Park, D.S.; Choi, J.J.; Ryu, J.; Yoon, W.H.; Kim, K.H.; Park, C.; Kim, H.E. Dense nanostructured hydroxyapatite coating on titanium by aerosol deposition. J. Am. Ceram. Soc. 2009, 92, 683–687. [Google Scholar] [CrossRef]
- Gineste, L.; Gineste, M.; Ranz, X.; Ellefterion, A.; Guilhem, A.; Rouquet, N.; Frayssinet, P. Degradation of hydroxylapatite, fluorapatite, and fluorhydroxyapatite coatings of dental implants in dogs. J. Biomed. Mater. Res. 1999, 48, 224–234. [Google Scholar] [CrossRef]
- Okada, M.; Furuzono, T. Hydroxyapatite nanocrystal coating on biodegradable microspheres. Mater. Sci. Eng. B 2010, 173, 199–203. [Google Scholar] [CrossRef]
- Habibovic, P.; Barrère, F.; Van Blitterswijk, C.A.; De Groot, K.; Layrolle, P. Biomimetic hydroxyapatite coating on metal implants. J. Am. Ceram. Soc. 2002, 85, 517–522. [Google Scholar]
- Sun, L.; Berndt, C.C.; Gross, K.A.; Kucuk, A. Review: Material fundamentals and clinical performance of plasma sprayed hydroxyapatite coatings. J. Biomed. Mater. Res. Appl. Biomater. 2001, 58, 570–592. [Google Scholar] [CrossRef]
- Yang, Y.; Kim, K.H.; Ong, J.L. A review on calcium phosphate coatings produced using a sputtering process—An alternative to plasma spraying. Biomaterials 2005, 26, 327–337. [Google Scholar] [CrossRef]
- Oliveira, A.L.; Mano, J.F.; Reis, R.L. Nature-inspired calcium phosphate coatings: Present status and novel advances in the science of mimicry. Curr. Opin. Solid State Mater. Sci. 2003, 7, 309–318. [Google Scholar] [CrossRef] [Green Version]
- Manso, M.; Jiménez, C.; Morant, C.; Herrero, P.; Martínez-Duart, J. Electrodeposition of hydroxyapatite coatings in basic conditions. Biomaterials 2000, 21, 1755–1761. [Google Scholar] [CrossRef]
- Kuo, M.C.; Yen, S.K. The process of electrochemical deposited hydroxyapatite coatings on biomedical titanium at room temperature. Mater. Sci. Eng. C 2002, 20, 153–160. [Google Scholar] [CrossRef]
- Eliaz, N.; Sridhar, T.M.; Mudali, U.K.; Raj, B. Electrochemical and electrophoretic deposition of hydroxyapatite for orthopaedic applications. Surf. Eng. 2005, 21, 238–242. [Google Scholar] [CrossRef]
- Ma, M.; Ye, W.; Wang, X.X. Effect of supersaturation on the morphology of hydroxyapatite crystals deposited by electrochemical deposition on titanium. Mater. Lett. 2008, 62, 3875–3877. [Google Scholar] [CrossRef]
- Ong, J.L.; Appleford, M.; Oh, S.; Yang, Y.; Chen, W.H.; Bumgardner, J.D.; Haggard, W.O. The characterization and development of bioactive hydroxyapatite coatings. JOM 2006, 58, 67–69. [Google Scholar]
- Qu, H.; Wei, M. The effect of temperature and initial pH on biomimetic apatite coating. J. Biomed. Mater. Res. B Appl. Biomater. 2008, 87B, 204–212. [Google Scholar] [CrossRef]
- Esfehanian, M.; Raissi-Dehkordi, B.; Moztarzadeh, F. Electrophoretic deposition of hydroxyapatite on metal implants. Inter. Ceram. 2005, 54, 92–98. [Google Scholar]
- Narayanan, R.; Seshadri, S.K.; Kwon, T.Y.; Kim, K.H. Calcium phosphate-based coatings on titanium and its alloys. J. Biomed. Mater. Res. B Appl. Biomater. 2008, 85B, 279–299. [Google Scholar] [CrossRef]
- Sugino, A.; Tsuru, K.; Hayakawa, S.; Kikuta, K.; Kawachi, G.; Osaka, A.; Ohtsuki, C. Induced deposition of bone-like hydroxyapatite on thermally oxidized titanium substrates using a spatial gap in a solution that mimics a body fluid. J. Ceram. Soc. Jpn. 2009, 117, 515–520. [Google Scholar] [CrossRef]
- Ye, G.; Troczynski, T. Hydroxyapatite coatings by pulsed ultrasonic spray pyrolysis. Ceram. Int. 2008, 34, 511–516. [Google Scholar] [CrossRef]
- Zheng, M.; Fan, D.; Li, X.K.; Zhang, J.B.; Liu, Q.B. Microstructure and in vitro bioactivity of laser-cladded bioceramic coating on titanium alloy in a simulated body fluid. J. Alloys Compd. 2010, 489, 211–214. [Google Scholar] [CrossRef]
- Geesink, R.G.T. Osteoconductive coating for total joint arthroplasty. Clin. Orthop. Rel. Res. 2002, 395, 53–65. [Google Scholar] [CrossRef]
- Mukherjee, D.P.; Dorairaj, N.R.; Mills, D.K.; Graham, D.; Krauser, J.T. Fatigue properties of hydroxyapatite-coated dental implants after exposure to a periodontal pathogen. J. Biomed. Mater. Res. 2000, 53, 467–474. [Google Scholar] [CrossRef]
- Gledhill, H.C.; Turner, I.G.; Doyle, C. In vitro fatigue behaviour of vacuum plasma and detonation gun sprayed hydroxyapatite coatings. Biomaterials 2001, 22, 1233–1240. [Google Scholar] [CrossRef]
- Cheng, K.; Ren, C.; Weng, W.; Du, P.; Shen, G.; Han, G.; Zhang, S. Bonding strength of fluoridated hydroxyapatite coatings: A comparative study on pull-out and scratch analysis. Thin Solid Films 2009, 517, 5361–5364. [Google Scholar] [CrossRef]
- De Groot, K.; Geesink, R.G.T.; Klein, C.P.A.T.; Serekian, P. Plasma sprayed coatings of hydroxylapatite. J. Biomed. Mater. Res. 1987, 21, 1375–1381. [Google Scholar] [CrossRef]
- Furlong, R.J.; Osborn, J.F. Fixation of hip prostheses by hydroxyapatite ceramic coating. J. Bone Joint Surg. B 1991, 73, 741–745. [Google Scholar]
- Saithna, A. The influence of hydroxyapatite coating of external fixator pins on pin loosening and pin track infection: A systematic review. Injury 2010, 41, 128–132. [Google Scholar] [CrossRef]
- Chang, J.K.; Chen, C.H.; Huang, K.Y.; Wang, G.J. Eight-year results of hydroxyapatite-coated hip arthroplasty. J. Arthroplasty 2006, 21, 541–546. [Google Scholar] [CrossRef]
- Matsumine, A.; Myoui, A.; Kusuzaki, K.; Araki, N.; Seto, M.; Yoshikawa, H.; Uchida, A. Calcium hydroxyapatite ceramic implants in bone tumor surgery. A long-term follow-up study. J. Bone Joint Surg. B 2004, 86, 719–725. [Google Scholar]
- Shetty, A.A.; Slack, R.; Tindall, A.; James, K.D.; Rand, C. 1 Results of a hydroxyapatite-coated (Furlong) total hip replacement. A 13- to 15-year follow-up. J. Bone Joint Surg. B 2005, 87, 1050–1054. [Google Scholar]
- Buchanan, J.M. 16 year review of hydroxyapatite ceramic coated hip implants—A clinical and histological evaluation. Key Eng. Mater. 2005, 284-286, 1049–1052. [Google Scholar] [CrossRef]
- Buchanan, J.M. 17 year review of hydroxyapatite ceramic coated hip implants—A clinical and histological evaluation. Key Eng. Mater. 2006, 309-311, 1341–1344. [Google Scholar] [CrossRef]
- Buchanan, J.M.; Goodfellow, S. Nineteen years review of hydroxyapatite ceramic coated hip implants: A clinical and histological evaluation. Key Eng. Mater. 2008, 361-363, 1315–1318. [Google Scholar] [CrossRef]
- Rajaratnam, S.S.; Jack, C.; Tavakkolizadeh, A.; George, M.D.; Fletcher, R.J.; Hankins, M.; Shepperd, J.A.N. Long-term results of a hydroxyapatite-coated femoral component in total hip replacement: A 15- to 21-year follow-up study. J. Bone Joint Surg. B 2008, 90, 27–30. [Google Scholar] [CrossRef]
- Binahmed, A.; Stoykewych, A.; Hussain, A.; Love, B.; Pruthi, V. Long-term follow-up of hydroxyapatite-coated dental implants—A clinical trial. Int. J. Oral Max. Impl. 2007, 22, 963–968. [Google Scholar]
- Iezzi, G.; Scarano, A.; Petrone, G.; Piattelli, A. Two human hydroxyapatite-coated dental implants retrieved after a 14-year loading period: A histologic and histomorphometric case report. J. Periodontol. 2007, 78, 940–947. [Google Scholar] [CrossRef]
- Bauer, T.W.; Geesink, R.G.T.; Zimmerman, R.; McMahon, J.T. Hydroxyapatite-coated femoral stems. Histological analysis of components retrieved at autopsy. J. Bone Joint Surg. A 1991, 73, 1439–1452. [Google Scholar]
- Buma, P.; Gardeniers, J.W. Tissue reactions around a hydroxyapatite-coated hip prostheses: Case report of a retrieveal specimen. J. Arthroplasty 1995, 10, 389–395. [Google Scholar] [CrossRef]
- Capello, W.D.; D’Antonio, J.A.; Feinberg, J.R.; Manley, M.T. Hydroxyapatite-coated total hip femoral components in patients less than fifty years old. Clinical and radiographic results after five to eight years of follow-up. J. Bone Joint Surg. A 1997, 79, 1023–1029. [Google Scholar]
- Paital, S.R.; Dahotre, N.B. Calcium phosphate coatings for bio-implant applications: Materials, performance factors, and methodologie. Mater. Sci. Eng. R 2009, 66, 1–70. [Google Scholar] [CrossRef]
- Thin Calcium Phosphate Coatings for Medical Implants; León, B.; Jansen, J.A. (Eds.) Springer: Berlin, Germany, 2009; p. 326.
- To narrow the subject of this review, functionally graded bioceramics prepared from calcium orthophosphates with various dopants is not considered. The interested readers are advised to read the original papers [292,520,521]
- Manjubala, I.; Kumar, T.S.S. Effect of TiO2—Ag2O additives on the formation of calcium phosphate based functionally graded bioceramics. Biomaterials 2000, 21, 1995–2002. [Google Scholar] [CrossRef]
- Wei, T.; Ruys, A.; Milthorpe, B. Hydroxyapatite-zirconia functionally graded bioceramics prepared by hot isostatic pressing. Key Eng. Mater. 2003, 240-242, 591–594. [Google Scholar] [CrossRef]
- Kon, M.; Ishikawa, K.; Miyamoto, Y.; Asaoka, K. Development of calcium phosphate based functional gradient bioceramics. Biomaterials 1995, 16, 709–714. [Google Scholar] [CrossRef]
- Wong, L.H.; Tio, B.; Miao, X. Functionally graded tricalcium phosphate/fluoroapatite composites. Mater. Sci. Eng. C 2002, 20, 111–115. [Google Scholar] [CrossRef]
- Tampieri, A.; Celotti, G.; Sprio, S.; Delcogliano, A.; Franzese, S. Porosity-graded hydroxyapatite ceramics to replace natural bone. Biomaterials 2001, 22, 1365–1370. [Google Scholar] [CrossRef]
- Lu, W.W.; Zhao, F.; Luk, K.D.K.; Yin, Y.J.; Cheung, K.M.C.; Cheng, G.X.; Yao, K.D.; Leong, J.C.Y. Controllable porosity hydroxyapatite ceramics as spine cage: Fabrication and properties evaluation. J. Mater. Sci. Mater. Med. 2003, 14, 1039–1046. [Google Scholar] [CrossRef]
- Werner, J.; Linner-Krcmar, B.; Friess, W.; Greil, P. Mechanical properties and in vitro cell compatibility of hydroxyapatite ceramics with graded pore structure. Biomaterials 2002, 23, 4285–4294. [Google Scholar] [CrossRef]
- Kaito, T.; Mukai, Y.; Nishikawa, M.; Ando, W.; Yoshikawa, H.; Myoui, A. Dual hydroxyapatite composite with porous and solid parts: Experimental study using canine lumbar interbody fusion model. J. Biomed. Mater. Res. B 2006, 78B, 378–384. [Google Scholar] [CrossRef]
- Rodriguez-Lorenzo, L.M.; Ferreira, J.M.F. Development of porous ceramic bodies for applications in tissue engineering and drug delivery systems. Mater. Res. Bull. 2004, 39, 83–91. [Google Scholar] [CrossRef]
- Lu, W.W.; Zhao, F.; Luk, K.D.K.; Yin, Y.J.; Cheung, K.M.C.; Cheng, G.X.; Yao, K.D.; Leong, J.C.Y. Controllable porosity hydroxyapatite ceramics as spine cage: Fabrication and properties evaluation. J. Mater. Sci. Mater. Med. 2003, 14, 1039–1046. [Google Scholar] [CrossRef]
- Dubok, V.A. Bioceramics—Yesterday, today, tomorrow. Powder Metallurgy Metal Ceram. 2000, 39, 381–394. [Google Scholar] [CrossRef]
- Heness, G.; Ben-Nissan, B. Innovative bioceramics. Mater. Forum 2004, 27, 104–114. [Google Scholar]
- Ohtsuki, C.; Kamitakahara, M.; Miyazaki, T. Bioactive ceramic-based materials with designed reactivity for bone tissue regeneration. J. R. Soc. Interface 2009, 6, S349–S360. [Google Scholar] [CrossRef]
- The term “bioinert” should be used with care, since it is clear that any material introduced into the physiological environment will induce a response. However, for the purposes of biomedical implants, the term can be defined as a minimal level of response from the host tissue [4]
- Yuan, H.; Fernandes, H.; Habibovic, P.; De Boer, J.; Barradas, A.M.C.; De Ruiter, A.; Walsh, W.R.; Van Blitterswijk, C.A.; De Bruijn, J.D. Osteoinductive ceramics as a synthetic alternative to autologous bone grafting. Proc. Natl. Acad. Sci. USA 2010, 107, 13614–13619. [Google Scholar] [CrossRef]
- Greenspan, D.C. Bioactive ceramic implant materials. Curr. Opin. Solid State Mater. Sci. 1999, 4, 389–393. [Google Scholar] [CrossRef]
- Blokhuis, T.J.; Termaat, M.F.; Den Boer, F.C.; Patka, P.; Bakker, F.C.; Haarman, H.J.T.M. Properties of calcium phosphate ceramics in relation to their in vivo behavior. J. Trauma 2000, 48, 179–189. [Google Scholar] [CrossRef]
- Kim, H.M. Bioactive ceramics: Challenges and perspectives. J. Ceram. Soc. Jpn. 2001, 109, S49–S57. [Google Scholar] [CrossRef]
- Okuda, T.; Ioku, K.; Yonezawa, I.; Minagi, H.; Gonda, Y.; Kawachi, G.; Kamitakahara, M.; Shibata, Y.; Murayama, H.; Kurosawa, H.; Ikeda, T. The slow resorption with replacement by bone of a hydrothermally synthesized pure calcium-deficient hydroxyapatite. Biomaterials 2008, 29, 9–2728. [Google Scholar]
- Seeley, Z.; Bandyopadhyay, A.; Bose, S. Tricalcium phosphate based resorbable ceramics: Influence of NaF and CaO addition. Mater. Sci. Eng. C 2008, 28, 11–17. [Google Scholar] [CrossRef]
- Descamps, M.; Richart, O.; Hardouin, P.; Hornez, J.C.; Leriche , A. Synthesis of macroporous β-tricalcium phosphate with controlled porous architectural. Ceram. Int. 2008, 34, 1131–1137. [Google Scholar] [CrossRef]
- Cushnie, E.K.; Khan, Y.M.; Laurencin, C.T. Amorphous hydroxyapatite-sintered polymeric scaffolds for bone tissue regeneration: Physical characterization studies. J. Biomed. Mater. Res. A 2008, 84A, 54–62. [Google Scholar] [CrossRef]
- Hench, L.L. Challenges for bioceramics in the 21st century. Am. Ceram. Soc. Bull. 2005, 84, 18–21. [Google Scholar]
- Nagase, M.; Baker, D.G.; Schumacher, H.R. Prolonged inflammatory reactions induced by artificial ceramics in the rat pouch model. J. Rheumatol. 1988, 15, 1334–1338. [Google Scholar]
- Rooney, T.; Berman, S.; Indersano, A.T. Evaluation of porous block hydroxylapatite for augmentation of alveolar ridges. J. Oral Maxillof. Surg. 1988, 46, 15–18. [Google Scholar] [CrossRef]
- Prudhommeaux, F.; Schiltz, C.; Lioté, F.; Hina, A.; Champy, R.; Bucki, B.; Ortiz-Bravo, E.; Meunier, A.; Rey, C.; Bardin, T. Variation in the inflammatory properties of basic calcium phosphate crystals according to crystal type. Arthritis Rheum. 1996, 39, 1319–1326. [Google Scholar] [CrossRef]
- Uchida, A.; Araki, N.; Shinto, Y.; Yoshikawa, H.; Kurisaki, E.; Ono, K. The use of calcium hydroxyapatite ceramic in bone tumour surgery. J. Bone Joint Surg. B 1990, 72, 298–302. [Google Scholar]
- Osteogenesis is the process of laying down new bone material by osteoblasts
- Osteoinduction is the property of the material to induce bone formation de novo or ectopically (i.e., in non-bone forming sites). It happens due to a stimulation and activation of host mesenchymal stem cells from the surrounding tissues, which differentiate into bone-forming osteoblasts [549]
- Giannoudis, P.V.; Dinopoulos, H.; Tsiridis, E. Bone substitutes: An update. Injury 2005, 36 Suppl. 1, S20–S27. [Google Scholar] [CrossRef]
- Osteoid is a bioorganic portion of the matrix of bone tissue. Osteoblasts begin the process of forming bone tissue by secreting the osteoid as several specific proteins. When the osteoid becomes mineralized, it and the adjacent bone cells have developed into new bone tissues
- Yamasaki, H.; Sakai, H. Osteogenic response to porous hydroxyapatite ceramics under the skin of dogs. Biomaterials 1992, 13, 308–312. [Google Scholar] [CrossRef]
- Klein, C.; De Groot, K.; Chen, W.; Li, Y.; Zhang, X. Osseous substance formation induced in porous calcium phosphate ceramics in soft tissues. Biomaterials 1994, 15, 31–34. [Google Scholar] [CrossRef]
- Yuan, H.; Yang, Z.; Li, Y.; Zhang, Z.; De Bruijn, J.D.; De Groot, K. Osteoinduction by calcium phosphate biomaterials. J. Mater. Sci. Mater. Med. 1998, 9, 723–726. [Google Scholar] [CrossRef]
- Ripamonti, U. Osteoinduction in porous hydroxyapatite implanted in heterotopic sites of different animal models. Biomaterials 1996, 17, 31–35. [Google Scholar] [CrossRef]
- Ripamonti, U.; Crooks, J.; Kirkbride, A.N. Sintered porous hydroxyapatites with intrinsic osteoinductive activity: Geometric induction of bone formation. S. Afr. J. Sci. 1999, 95, 335–343. [Google Scholar]
- Yuan, H.P.; De Bruijn, J.D.; Li, Y.B.; Feng, J.Q.; Yang, Z.J.; De Groot, K.; Zhang, X.D. Bone formation induced by calcium phosphate ceramics in soft tissue of dogs: A comparative study between porous α-TCP and β-TCP. J. Mater. Sci. Mater. Med. 2001, 12, 7–13. [Google Scholar] [CrossRef]
- Matsushita, N.; Terai, H.; Okada, T.; Nozaki, K.; Inoue, H.; Miyamoto, S.; Takaoka, K. A new bone-inducing biodegradable porous β-tricalcium phosphate. J. Biomed. Mater. Res. A 2004, 70A, 450–458. [Google Scholar] [CrossRef]
- Le Nihouannen, D.; Daculsi, G.; Saffarzadeh, A.; Gauthier, O.; Delplace, S.; Pilet, P.; Layrolle, P. Ectopic bone formation by microporous calcium phosphate ceramic particles in sheep muscles. Bone 2005, 36, 1086–1093. [Google Scholar] [CrossRef]
- Yuan, H.; Kurashina, K.; De Bruijn, D.J.; Li, Y.; De Groot, K.; Zhang, X. A preliminary study of osteoinduction of two kinds of calcium phosphate bioceramics. Biomaterials 1999, 20, 1799–1806. [Google Scholar] [CrossRef]
- Gosain, A.K.; Song, L.; Riordan, P.; Amarante, M.T.; Nagy, P.G.; Wilson, C.R.; Toth, J.M.; Ricci, J.L. A 1-year study of osteoinduction in hydroxyapatite-derived biomaterials in an adult sheep model: Part I. Plast. Reconstr. Surg. 2002, 109, 619–630. [Google Scholar] [CrossRef]
- Cheng, L.; Ye, F.; Yang, R.; Lu, X.; Shi, Y.; Li, L.; Fan, H.; Bu, H. Osteoinduction of hydroxyapatite/β-tricalcium phosphate bioceramics in mice with a fractured fibula. Acta Biomater. 2010, 6, 1569–1574. [Google Scholar]
- Barrere, F.; Van der Valk, C.M.; Dalmeijer, R.A.; Meijer, G.; Van Blitterswijk, C.A.; De Groot, K.; Layrolle, P. Osteogenecity of octacalcium phosphate coatings applied on porous titanium. J. Biomed. Mater. Res. A 2003, 66A, 779–788. [Google Scholar] [CrossRef]
- Habibovic, P.; Van der Valk, C.M.; Van Blitterswijk, C.A.; De Groot, K.; Meijer, G. Influence of octacalcium phosphate coating on osteoinductive properties of biomaterials. J. Mater. Sci. Mater. Med. 2004, 15, 373–380. [Google Scholar]
- Habibovic, P.; Yuan, H.; Van der Valk, C.M.; Meijer, G.; Van Blitterswijk, C.A.; De Groot, K. 3D microenvironment as essential element for osteoinduction by biomaterials. Biomaterials 2005, 26, 3565–3575. [Google Scholar] [CrossRef]
- Habibovic, P.; Sees, T.M.; Van den Doel, M.A.; Van Blitterswijk, C.A.; De Groot, K. Osteoinduction by biomaterials—physicochemical and structural influences. J. Biomed. Mater. Res. A 2006, 77A, 747–762. [Google Scholar] [CrossRef]
- Reddi, A.H. Morphogenesis and tissue engineering of bone and cartilage: Inductive signals, stem cells and biomimetic biomaterials. Tissue Eng. 2000, 6, 351–359. [Google Scholar] [CrossRef]
- Ripamonti, U. The morphogenesis of bone in replicas of porous hydroxyapatite obtained by conversion of calcium carbonate exoskeletons of coral. J. Bone Joint Surg. A 1991, 73, 692–703. [Google Scholar]
- Kuboki, Y.; Takita, H.; Kobayashi, D. BMP-induced osteogenesis on the surface of hydroxyapatite with geometrically feasible and nonfeasible structures: Topology of osteogenesis. J. Biomed. Mater. Res. 1998, 39, 190–199. [Google Scholar] [CrossRef]
- Diaz-Flores, L.; Gutierrez, R.; Lopez-Alonso, A.; Gonzalez, R.; Varela, H. Pericytes as a supplementary source of osteoblasts in periosteal osteogenesis. Clin. Orthop. Relat. Res. 1992, 275, 280–286. [Google Scholar]
- Dorozhkin, S.V. Inorganic chemistry of the dissolution phenomenon, the dissolution mechanism of calcium apatites at the atomic (ionic) level. Comment Inorg. Chem. 1999, 20, 285–299. [Google Scholar] [CrossRef]
- Dorozhkin, S.V. A review on the dissolution models of calcium apatites. Prog. Cryst. Growth Charact. 2002, 44, 45–61. [Google Scholar] [CrossRef]
- Teitelbaum, S.L. Bone resorption by osteoclasts. Science 2000, 289, 1504–1508. [Google Scholar] [CrossRef]
- Okuda, T.; Ioku, K.; Yonezawa, I.; Minagi, H.; Kawachi, G.; Gonda, Y.; Murayama, H.; Shibata, Y.; Minami, S.; Kamihara, S.; Kurosawa, H.; Ikeda, T. The effect of the microstructure of β-tricalcium phosphate on the metabolism of subsequently formed bone tissue. Biomaterials 2007, 28, 2–2621. [Google Scholar]
- Orly, I.; Gregoire, M.; Menanteau, J.; Heughebaert, M.; Kerebel, B. Chemical changes in hydroxyapatite biomaterial under in vivo and in vitro biological conditions. Calcif. Tissue Int. 1989, 45, 20–26. [Google Scholar] [CrossRef]
- Bertazzo, S.; Zambuzzi, W.F.; Campos, D.D.P.; Ogeda, T.L.; Ferreira, C.V.; Bertran, C.A. Hydroxyapatite surface solubility and effect on cell adhesion. Colloids Surf. B Biointerfaces 2010, 78, 177–184. [Google Scholar] [CrossRef]
- Schwartz, Z.; Boyan, B.D. Underlying mechanisms at the bone-biomaterial interface. J. Cell. Biochem. 1994, 56, 340–347. [Google Scholar] [CrossRef]
- Puleo, D.A.; Nanci, A. Understanding and controlling the bone-implant interface. Biomaterials 1999, 20, 2311–2321. [Google Scholar] [CrossRef]
- Xin, R.; Leng, Y.; Chen, J.; Zhang, Q. A comparative study of calcium phosphate formation on bioceramics in vitro and in vivo. Biomaterials 2005, 26, 6477–6486. [Google Scholar] [CrossRef]
- Girija, E.K.; Parthiban, S.P.; Suganthi, R.V.; Elayaraja, K.; Joshy, M.I.A.; Vani, R.; Kularia, P.; Asokan, K.; Kanjilal, D.; Yokogawa, Y.; Narayana Kalkura, S. High energy irradiation—A tool for enhancing the bioactivity of hydroxyapatite. J. Ceram. Soc. Jpn. 2008, 116, 320–324. [Google Scholar] [CrossRef]
- Okada, M.; Furukawa, K.; Serizawa, T.; Yanagisawa, Y.; Tanaka, H.; Kawai, T.; Furuzono, T. Interfacial interactions between calcined hydroxyapatite nanocrystals and substrates. Langmuir 2009, 25, 6300–6306. [Google Scholar]
- Malik, M.A.; Puleo, D.A.; Bizios, R.; Doremus, R.H. Osteoblasts on hydroxyapatite, alumina and bone surfaces in vitro: Morphology during the first 2 h of attachment. Biomaterials 1992, 13, 123–128. [Google Scholar] [CrossRef]
- Norman, M.E.; Elgendy, H.M.; Shors, E.C.; El-Amin, S.F.; Laurencin, C.T. An in-vitro evaluation of coralline porous hydroxyapatite as a scaffold for osteoblast growth. Clin. Mater. 1994, 17, 85–91. [Google Scholar] [CrossRef]
- Shu, R.; McMullen, R.; Baumann, M.J.; McCabe, L.R. Hydroxyapatite accelerates differentiation and suppresses growth of MC3T3-E1 osteoblasts. J. Biomed. Mater. Res. A 2003, 67A, 1196–1204. [Google Scholar] [CrossRef]
- Gomi, K.; Lowenberg, B.; Shapiro, G.; Davies, J.E. Resorption of sintered synthetic hydroxyapatite by osteoclasts in vitro. Biomaterials 1992, 20, 91–96. [Google Scholar]
- Suzuki, T.; Ohashi, R.; Yokogawa, Y.; Nishizawa, K.; Nagata, F.; Kawamoto, Y.; Kameyama, T.; Toriyama, M. Initial anchoring and proliferation of fibroblast L-929 cells on unstable surface of calcium phosphate ceramics. J. Biosci. Bioeng. 1999, 87, 320–327. [Google Scholar] [CrossRef]
- Arinzeh, T.L.; Tran, T.; McAlary, J.; Daculsi, G. A comparative study of biphasic calcium phosphate ceramics for human mesenchymal stem-cell-induced bone formation. Biomaterials 2005, 26, 3631–3638. [Google Scholar] [CrossRef]
- Oh, S.; Oh, N.; Appleford, M.; Ong, J.L. Bioceramics for tissue engineering applications—A review. Am. J. Biochem. Biotechnol. 2006, 2, 49–56. [Google Scholar] [CrossRef]
- Appleford, M.; Oh, S.; Cole, J.A.; Carnes, D.L.; Lee, M.; Bumgardner, J.D.; Haggard, W.O.; Ong, J.L. Effects of trabecular calcium phosphate scaffolds on stress signaling in osteoblast precursor cells. Biomaterials 2007, 28, 2747–2753. [Google Scholar] [CrossRef]
- Ohgushi, H.; Caplan, A.I. Stem cell technology and bioceramics: From cell to gene engineering. J. Biomed. Mater. Res. 1999, 48, 913–927. [Google Scholar] [CrossRef]
- Okumura, M.; Ohgushi, H.; Tamai, S. Bonding osteogenesis in coralline hydroxyapatite combined with bone marrow cells. Biomaterials 1990, 12, 28–37. [Google Scholar]
- Unger, R.E.; Sartoris, A.; Peters, K.; Motta, A.; Migliaresi, C.; Kunkel, M.; Bulnheim, U.; Rychly, J.; Kirkpatrick, C.J. Tissue-like self-assembly in cocultures of endothelial cells and osteoblasts and the formation of microcapillary like structures on three-dimensional porous biomaterials. materials 2007, 28, 3965–3976. [Google Scholar]
- Saiz, E.; Gremillard, L.; Menendez, G.; Miranda, P.; Gryn, K.; Tomsia, A.P. Preparation of porous hydroxyapatite scaffolds. Mater. Sci. Eng. C 2007, 27, 546–550. [Google Scholar] [CrossRef]
- Tamai, N.; Myoui, A.; Hirao, M.; Kaito, T.; Ochi, T.; Tanaka, J.; Takaoka, K.; Yoshikawa, H. A new biotechnology for articular cartilage repair: Subchondral implantation of a composite of interconnected porous hydroxyapatite, synthetic polymer (PLAPEG), and bone morphogenetic protein-2 (rhBMP-2). Osteoarthritis Cartilage 2005, 13, 405–417. [Google Scholar] [CrossRef]
- Bignon, A.; Chouteau, J.; Chevalier, J.; Fantozzi, G.; Carret, J.P.; Chavassieux, P.; Boivin, G.; Melin, M.; Hartmann, D. Effect of micro- and macroporosity of bone substitutes on their mechanical properties and cellular response. J. Mater. Sci. Mater. Med. 2003, 14, 1089–1097. [Google Scholar] [CrossRef]
- Griffith, L.G.; Naughton, G. Tissue engineering—Current challenges and expanding opportunities. Science 2002, 295, 1009–1014. [Google Scholar] [CrossRef]
- Tissue Engineering; Van Blitterswijk, C.A.; Thomsen, P.; Hubbell, J.; Cancedda, R.; De Bruijn, J.D.; Lindahl, A.; Sohier, J.; Williams, D.F. (Eds.) Academic Press: Burlington, MA, USA, 2008; p. 760.
- Repair is usually a rapid occurrence that is required for the survival of the individual but is not necessary for its optimal function. It usually results in a dense connective tissue scar that fills the space; however, it may not be responsive to the highly loaded mechanical environment required of culoskeletal tissue. Regeneration is a slow process that is usually characterized by the recapitulation of aspects of embryonic development [598]
- Goldberg, V.M.; Caplan, A.I. Orthopedic Tissue Engineering Basic Science and Practice; Marcel Dekker: New York, NY, USA, 2004; p. 338. [Google Scholar]
- In 2003, the NSF published a report titled: “The emergence of tissue engineering as a research field”, which provides a thorough description of the history of this field. Available online: http://www.nsf.gov/pubs/2004/nsf0450/start.htm (accessed on October 2010).
- Ikada, Y. Challenges in tissue engineering. J. R. Soc. Interface 2006, 3, 589–601. [Google Scholar] [CrossRef]
- Cima, L.G.; Langer, R. Engineering human tissue. Chem. Eng. Prog. 1993, 89, 46–54. [Google Scholar]
- Langer, R.; Vacanti, J.P. Tissue engineering. Science 1993, 260, 920–926. [Google Scholar]
- El-Ghannam, A. Bone reconstruction: From bioceramics to tissue engineering. Expert Rev. Med. Dev. 2005, 2, 87–101. [Google Scholar] [CrossRef]
- In general, a scaffold is a temporary structure used to support people and material in the construction or repair of buildings and other large structures. It is usually a modular system of metal pipes (termed tubes in Britain), although it can be made out of other materials
- Yang, S.; Leong, K.F.; Du, Z.; Chua, C.K. The design of scaffolds for use in tissue engineering. Part I—Traditional factors. Tissue Eng. 2001, 7, 679–689. [Google Scholar] [CrossRef]
- Hutmacher, D.W. Scaffolds in tissue engineering bone and cartilage. Biomaterials 2000, 21, 2529–2543. [Google Scholar] [CrossRef]
- Ma, P.X. Scaffolds for tissue fabrication. Mater. Today 2004, 7, 30–40. [Google Scholar]
- Yasuhiko, T. Biomaterial Technology for tissue engineering applications. J. R. Soc. Interface 2009, 6, S311–S324. [Google Scholar] [CrossRef]
- Scaffolding in Tissue Engineering; Ma, P.X.; Elisseeff, J. (Eds.) CRC Press: Boca Raton, FL, USA, 2006; p. 638.
- Schieker, M.; Seitz, H.; Drosse, I.; Seitz, S.; Mutschler, W. Biomaterials as scaffold for bone tissue engineering. Eur. J. Trauma 2006, 32, 114–124. [Google Scholar] [CrossRef]
- Okumura, M.; Ohgushi, H.; Dohi, Y.; Katuda, T.; Tamai, S.; Koerten, H.K.; Tabata, S. Osteoblastic phenotype expression on the surface of hydroxyapatite ceramics. J. Biomed. Mater. Res. 1997, 37, 122–129. [Google Scholar] [CrossRef]
- Service, R.F. Tissue engineers build new bone. Science 2000, 289, 1498–1500. [Google Scholar] [CrossRef]
- Fini, M.; Giardino, R.; Borsari, V.; Torricelli, P.; Rimondini, L.; Giavaresi, G.; Aldini, N.N. In vitro behaviour of osteoblasts cultured on orthopaedic biomaterials with different surface roughness, uncoated and fluorohydroxyapatite-coated, relative to the in vivo osteointegration rate. Int. J. Artif. ans 2003, 26, 520–528. [Google Scholar]
- Sato, M.; Webster, T.J. Designing orthopedic implant surfaces: Harmonization of nanotopographical and chemical aspects. Nanomedicine 2006, 1, 351–354. [Google Scholar] [CrossRef]
- Annaz, B.; Hing, K.A.; Kayser, M.; Buckland, T.; Di Silvio, L. Porosity in phase-pure hydroxyapatite. J. Microscopy 2004, 216, 97–109. [Google Scholar] [CrossRef]
- Li, S.; De Wijn, J.R.; Li, J.; Layrolle, P.; De Groot, K. Macroporous biphasic calcium phosphate scaffold with high permeability/porosity ratio. Tissue Eng. 2003, 9, 535–548. [Google Scholar] [CrossRef]
- Ebaretonbofa, E.; Evans, J.R. High porosity hydroxyapatite foam scaffolds for bone substitute. J. Porous Mater. 2002, 9, 257–263. [Google Scholar] [CrossRef]
- Specchia, N.; Pagnotta, A.; Cappella, M.; Tampieri, A.; Greco, F. Effect of hydroxyapatite porosity on growth and differentiation of human osteoblast-like cells. J. Mater. Sci. 2002, 37, 577–584. [Google Scholar] [CrossRef]
- Malmström, J.; Adolfsson, E.; Arvidsson, A.; Thomsen, P. Bone response inside free-form fabricated macroporous hydroxyapatite scaffolds with and without an open microporosity. Clin. Implant Dentistry Rel. Res. 2007, 9, 79–88. [Google Scholar] [CrossRef]
- Teixeira, C.C.; Nemelivsky, Y.; Karkia, C.; LeGeros, R.Z. Biphasic calcium phosphate: A scaffold for growth plate chondrocyte maturation. Tissue Eng. 2006, 12, 2283–2289. [Google Scholar] [CrossRef]
- Teixeira, S.; Oliveira, S.; Ferraz, M.P.; Monteiro, F.J. Three dimensional macroporous calcium phosphate scaffolds for bone tissue engineering. Key Eng. Mater. 2008, 361-363, 947–950. [Google Scholar] [CrossRef]
- Stevens, M.M. Biomaterials for bone tissue engineering. Mater. Today 2008, 11, 18–25. [Google Scholar] [CrossRef]
- Artzi, Z.; Weinreb, M.; Givol, N.; Rohrer, M.D.; Nemcovsky, C.E.; Prasad, H.S.; Tal, H. Biomaterial resorbability and healing site morphology of inorganic bovine bone and beta tricalcium phosphate in the canine: A 24-month longitudinal histologic study and morphometric analysis. Int. J. Oral. Impl. 2004, 19, 357–368. [Google Scholar]
- Burg, K.J.L.; Porter, S.; Kellam, J.F. Biomaterial developments for bone tissue engineering. Biomaterials 2000, 21, 2347–2359. [Google Scholar] [CrossRef]
- Ajaal, T.T.; Smith, R.W. Employing the Taguchi method in optimizing the scaffold production process for artificial bone grafts. J. Mater. Proc. Technol. 2009, 209, 1521–1532. [Google Scholar] [CrossRef]
- Boyde, A.; Corsi, A.; Quarto, R.; Cancedda, R.; Bianco, P. Osteoconduction in large macroporous hydroxyapatite ceramic implants: Evidence for a complementary integration and disintegration mechanism. Bone 1999, 24, 579–589. [Google Scholar] [CrossRef]
- Hing, K.A. Bioceramic bone graft substitutes: Influence of porosity and chemistry. Int. J. Appl. Ceram. Technol. 2005, 2, 184–199. [Google Scholar] [CrossRef]
- Peppas, N.A.; Langer, R. New challenges in biomaterials. Science 1994, 263, 1715–1720. [Google Scholar]
- Hench, L.L. Biomaterials: A forecast for the future. Biomaterials 1998, 19, 1419–1423. [Google Scholar] [CrossRef]
- Barrère, F.; Mahmood, T.A.; De Groot, K.; Van Blitterswijk, C.A. Advanced biomaterials for skeletal tissue regeneration: Instructive and smart functions. Mater. Sci. Eng. R 2008, 59, 38–71. [Google Scholar] [CrossRef]
- Dellinger, J.G.; Eurell, J.A.C.; Jamison, R.D. Bone response to 3D periodic hydroxyapatite scaffolds with and without tailored microporosity to deliver bone morphogenetic protein 2. J. Biomed. Mater. Res. A 2006, 76A, 366–376. [Google Scholar] [CrossRef]
- Pal, K.; Pal, S. Development of porous hydroxyapatite scaffolds. Mater. Manuf. Proc. 2006, 21, 325–328. [Google Scholar] [CrossRef]
- Bae, C.J.; Kim, H.W.; Koh, Y.H.; Kim, H.E. Hydroxyapatite (HA) bone scaffolds with controlled macrochannel pores. J. Mater. Sci. Mater. Med. 2006, 17, 517–521. [Google Scholar] [CrossRef]
- Guo, H.; Wei, J.; Kong, H.; Liu, C.; Pan, K. Biocompatibility and osteogenesis of calcium phosphate cement scaffolds for bone tissue engineering. Adv. Mater. Res. 2008, 47-50, 1383–1386. [Google Scholar] [CrossRef]
- Guo, H.; Su, J.; Wei, J.; Kong, H.; Liu, C. Biocompatibility and osteogenicity of degradable Ca-deficient hydroxyapatite scaffolds from calcium phosphate cement for bone tissue engineering. Acta Biomater. 2009, 5, 268–278. [Google Scholar] [CrossRef]
- Liu, H.; Webster, T.J. Nanomedicine for implants: A review of studies and necessary experimental tools. Biomaterials 2007, 28, 354–369. [Google Scholar] [CrossRef]
- Wang, C.; Duan, Y.; Markovic, B.; Barbara, J.; Howlett, C.R.; Zhang, X.; Zreiqat, H. Proliferation and bone-related gene expression of osteoblasts grown on hydroxyapatite ceramics sintered at different temperature. Biomaterials 2004, 25, 2949–2956. [Google Scholar] [CrossRef]
- Matsumoto, T.; Okazaki, M.; Nakahira, A.; Sasaki, J.; Egusa, H.; Sohmura, T. Modification of apatite materials for bone tissue engineering and drug delivery carriers. Curr. Med. Chem. 2007, 14, 2726–2733. [Google Scholar] [CrossRef]
- Traykova, T.; Aparicio, C.; Ginebra, M.P.; Planell, J.A. Bioceramics as nanomaterials. Nanomedicine 2006, 1, 91–106. [Google Scholar] [CrossRef]
- Kalita, S.J.; Bhardwaj, A.; Bhatt, H.A. Nanocrystalline calcium phosphate ceramics in biomedical engineering. Mater. Sci. Eng. C 2007, 27, 441–449. [Google Scholar] [CrossRef]
- Dorozhkin, S.V. Nano-sized and nanocrystalline calcium orthophosphates in biomedical engineering. J. Biomimetics Biomaterials Tissue Eng. 2009, 3, 59–92. [Google Scholar] [CrossRef]
- Dorozhkin, S.V. Nanodimensional and nanocrystalline apatites and other calcium orthophosphates in biomedical engineering, biology and medicine. Materials 2009, 2, 1975–2045. [Google Scholar] [CrossRef]
- Dorozhkin, S.V. Nanosized and nanocrystalline calcium orthophosphates. Acta Biomater. 2010, 6, 715–734. [Google Scholar] [CrossRef]
- Aizawa, M.; Porter, A.E.; Best, S.M.; Bonfield, W. Ultrastructural observation of single-crystal apatite fibres. Biomaterials 2005, 26, 3427–3433. [Google Scholar] [CrossRef]
- Park, Y.M.; Ryu, S.C.; Yoon, S.Y.; Stevens, R.; Park, H.C. Preparation of whisker-shaped hydroxyapatite/β-tricalcium phosphate composite. Mater. Chem. Phys. 2008, 109, 440–447. [Google Scholar] [CrossRef]
- Morisue, H.; Matsumoto, M.; Chiba, K.; Matsumoto, H.; Toyama, Y.; Aizawa, M.; Kanzawa, N.; Fujimi, T.J.; Uchida, H.; Okada, I. A novel hydroxyapatite fiber mesh as a carrier for recombinant human bone morphogenetic protein-2 enhances bone union in rat posterolateral fusion model. Spine 2006, 31, 1194–1200. [Google Scholar]
- Matsuda, A.; Ikoma, T.; Kobayashi, H.; Tanaka, J. Preparation and mechanical property of core-shell type chitosan/calcium phosphate composite fiber. Mater. Sci. Eng. C 2004, 24, 723–728. [Google Scholar] [CrossRef]
- Tas, A.C. Molten salt synthesis of calcium hydroxyapatite whiskers. J. Am. Ceram. Soc. 2001, 84, 295–300. [Google Scholar] [CrossRef]
- Aizawa, M.; Ueno, H.; Itatani, K.; Okada, I. Syntheses of calcium-deficient apatite fibres by a homogeneous precipitation method and their characterizations. J. Eur. Ceram. Soc. 2006, 26, 501–507. [Google Scholar] [CrossRef]
- Wu, Y.; Hench, L.L.; Du, J.; Choy, K.L.; Guo, J. Preparation of hydroxyapatite fibers by electrospinning technique. J. Am. Ceram. Soc. 2004, 87, 1988–1991. [Google Scholar]
- Ramanan, S.R.; Venkatesh, R. A study of hydroxyapatite fibers prepared via sol-gel route. Mater. Lett. 2004, 58, 3320–3323. [Google Scholar] [CrossRef]
- Seo, D.S.; Lee, J.K. Synthesis of hydroxyapatite whiskers through dissolution-reprecipitation process using EDTA. J. Cryst. Growth 2008, 310, 2162–2167. [Google Scholar] [CrossRef]
- Tas, A.C. Formation of calcium phosphate whiskers in hydrogen peroxide (H2O2) solutions at 90 °C. J. Am. Ceram. Soc. 2007, 90, 2358–2362. [Google Scholar] [CrossRef]
- Neira, I.S.; Guitián, F.; Taniguchi, T.; Watanabe, T.; Yoshimura, M. Hydrothermal synthesis of hydroxyapatite whiskers with sharp faceted hexagonal morphology. J. Mater. Sci. 2008, 43, 2171–2178. [Google Scholar] [CrossRef]
- Yang, H.Y.; Yang, S.F.; Chi, X.P.; Evans, J.R.G.; Thompson, I.; Cook, R.J.; Robinson, P. Sintering behaviour of calcium phosphate filaments for use as hard tissue scaffolds. J. Eur. Ceram. Soc. 2008, 28, 159–167. [Google Scholar] [CrossRef]
- Zhang, H.; Zhu, Q. Synthesis of nanospherical and ultralong fibrous hydroxyapatite and reinforcement of biodegradable chitosan/hydroxyapatite composite. Modern Phys. Lett. B 2009, 23, 3967–3976. [Google Scholar] [CrossRef]
- Ribeiro, C.C.; Barrias, C.C.; Barbosa, M.A. Preparation and characterisation of calcium-phosphate porous microspheres with a uniform size for biomedical applications. J. Mater. Sci. Mater. Med. 2006, 17, 455–463. [Google Scholar] [CrossRef]
- Ribeiro, C.C.; Barrias, C.C.; Barbosa, M.A. Calcium phosphate-alginate microspheres as enzyme delivery matrices. Biomaterials 2004, 25, 4363–4373. [Google Scholar] [CrossRef]
- Zhou, W.Y.; Wang, M.; Cheung, W.L.; Guo, B.C.; Jia, D.M. Synthesis of carbonated hydroxyapatite nanospheres through nanoemulsion. J. Mater. Sci. Mater. Med. 2008, 19, 103–110. [Google Scholar]
- Kim, H.W.; Gu, H.J.; Lee, H.H. Microspheres of collagen-apatite nanocomposites with osteogenic potential for tissue engineering. Tissue Eng. 2007, 13, 965–973. [Google Scholar] [CrossRef]
- Kawai, T.; Sekikawa, H.; Unuma, H. Preparation of hollow hydroxyapatite microspheres utilizing poly(divinylbenzene) as a template. J. Ceram. Soc. Jpn. 2009, 117, 340–343. [Google Scholar] [CrossRef]
- Descamps, M.; Hornez, J.C.; Leriche, A. Manufacture of hydroxyapatite beads for medical applications. J. Eur. Ceram. Soc. 2009, 29, 369–375. [Google Scholar] [CrossRef]
- Cho, J.S.; Jung, D.S.; Han, J.M.; Kang, Y.C. Spherical shape hydroxyapatite powders prepared by flame spray pyrolysis. J. Ceram. Proc. Res. 2008, 9, 348–352. [Google Scholar]
- Kimura, I.; Honma, T.; Riman, R.E. Preparation of hydroxyapatite microspheres by interfacial reaction in a multiple emulsion. J. Ceram. Soc. Jpn. 2007, 115, 888–893. [Google Scholar] [CrossRef]
- Yao, A.; Ai, F.; Liu, X.; Wang, D.; Huang, W.; Xu, W. Preparation of hollow hydroxyapatite microspheres by the conversion of borate glass at near room temperature. Mater. Res. Bull. 2010, 45, 25–28. [Google Scholar] [CrossRef]
- Jiang, D.; Chen, M.; Li, D.; Zhu, J.; Lü, X.; Xie, J. One-pot synthesis of hierarchical fluorapatite hollow microparticles. Mater. Lett. 2009, 63, 2639–2642. [Google Scholar] [CrossRef]
- Lim, J.H.; Park, J.H.; Park, E.K.; Kim, H.J.; Park, I.K.; Shin, H.Y.; Shin, H.I. Fully interconnected globular porous biphasic calcium phosphate ceramic scaffold facilitates osteogenic repair. Key Eng. Mater. 2008, 361-363, 119–122. [Google Scholar] [CrossRef]
- Cho, J.S.; Ko, Y.N.; Koo, H.Y.; Kang, Y.C. Synthesis of nano-sized biphasic calcium phosphate ceramics with spherical shape by flame spray pyrolysis. J. Mater. Sci. Mater. Med. 2010, 21, 1143–1149. [Google Scholar] [CrossRef]
- Ye, F.; Guo, H.; Zhang, H.; He, X. Polymeric micelle-templated synthesis of hydroxyapatite hollow nanoparticles for a drug delivery system. Acta Biomater. 2010, 6, 2212–2218. [Google Scholar] [CrossRef]
- Simon, J.L.; Michna, S.; Lewis, J.A.; Rekow, E.D.; Thompson, V.P.; Smay, J.E.; Yampolsky, A.; Parsons, J.R.; Ricci, J.L. In vivo bone response to 3D periodic hydroxyapatite scaffolds assembled by direct ink writing. J. Biomed. Mater. Res. A 2007, 83A, 747–758. [Google Scholar] [CrossRef]
- Chu, T.M.; Orton, D.G.; Hollister, S.J.; Feinberg, S.E.; Halloran, J.W. Mechanical and in vivo performance of hydroxyapatite implants with controlled architectures. Biomaterials 2002, 23, 1283–1293. [Google Scholar] [CrossRef]
- Yoshikawa, H.; Myoui, A. Bone tissue engineering with porous hydroxyapatite ceramics. J.Artif. Organs 2005, 8, 131–136. [Google Scholar] [CrossRef]
- Detsch, R.; Uhl, F.; Deisinger, U.; Ziegler, G. In vitro-studies of cell growth on three differently fabricated hydroxyapatite ceramic scaffolds for bone tissue engineering. Key Eng. Mater. 2008, 361-363, 1181–1184. [Google Scholar] [CrossRef]
- Min, S.H.; Jin, H.H.; Park, H.Y.; Park, I.M.; Park, H.C.; Yoon, S.Y. Preparation of porous hydroxyapatite scaffolds for bone tissue engineering. Mater. Sci. Forum 2006, 510-511, 754–757. [Google Scholar] [CrossRef]
- Deville, S.; Saiz, E.; Nalla, R.K.; Tomsia, A.P. Strong biomimetic hydroxyapatite scaffolds. Adv. Sci. Technol. 2006, 49, 148–152. [Google Scholar] [CrossRef]
- Yang, W.; Zhou, D.; Yin, G.; Chen, H. Progress of biphasic calcium phosphate bioceramic as scaffold materials of bone tissue engineering. J. Chinese Ceram. Soc. 2004, 32, 1143–1149. [Google Scholar]
- Ramay, H.R.R.; Zhang, M. Biphasic calcium phosphate nanocomposite porous scaffolds for load-bearing bone tissue engineering. Biomaterials 2004, 25, 5171–5180. [Google Scholar] [CrossRef]
- Yuan, H.; Van Blitterswijk, C.A.; De Groot, K.; De Bruijn, J.D. Cross-species comparison of ectopic bone formation in biphasic calcium phosphate (BCP) and hydroxyapatite (HA) scaffolds. Tissue Eng. 2006, 12, 1607–1615. [Google Scholar] [CrossRef]
- Chen, G.; Li, W.; Zhao, B.; Sun, K. A novel biphasic bone scaffold: β-calcium phosphate and amorphous calcium polyphosphate. J. Am. Ceram. Soc. 2009, 92, 945–948. [Google Scholar] [CrossRef]
- Guo, D.; Xu, K.; Han, Y. The in situ synthesis of biphasic calcium phosphate scaffolds with controllable compositions, structures, and adjustable properties. J. Biomed. Mater. Res. A 2009, 88A, 43–52. [Google Scholar] [CrossRef]
- Furuichi, K.; Oaki, Y.; Ichimiya, H.; Komotori, J.; Imai, H. Preparation of hierarchically organized calcium phosphate-organic polymer composites by calcification of hydrogel. Sci. Technol. Adv. Mater. 2006, 7, 219–225. [Google Scholar] [CrossRef]
- Wei, J.; Jia, J.; Wu, F.; Wei, S.; Zhou, H.; Zhang, H.; Shin, J.W.; Liu, C. Hierarchically microporous/macroporous scaffold of magnesium-calcium phosphate for bone tissue regeneration. Biomaterials 2010, 31, 1260–1269. [Google Scholar] [CrossRef]
- Gbureck, U.; Grolms, O.; Barralet, J.E.; Grover, L.M.; Thull, R. Mechanical activation and cement formation of β-tricalcium phosphate. Biomaterials 2003, 24, 4123–4131. [Google Scholar] [CrossRef]
- Gbureck, U.; Barralet, J.E.; Hofmann, M.; Thull, R. Mechanical activation of tetracalcium phosphate. J. Am. Ceram. Soc. 2004, 87, 311–313. [Google Scholar] [CrossRef]
- Bohner, M.; Luginbühl, R.; Reber, C.; Doebelin, N.; Baroud, G.; Conforto, E. A physical approach to modify the hydraulic reactivity of α-tricalcium phosphate powder. Acta Biomater. 2009, 5, 3524–3535. [Google Scholar] [CrossRef]
- Hagio, T.; Tanase, T.; Akiyama, J.; Iwai, K.; Asai, S. Formation and biological affinity evaluation of crystallographically aligned hydroxyapatite. J. Ceram. Soc. Jpn. 2008, 116, 79–82. [Google Scholar] [CrossRef]
- Hench, L.L.; Polak, J.M. Third-generation biomedical materials. Science 2002, 295, 1014–1017. [Google Scholar] [CrossRef]
- Blawas, A.S.; Reichert, W.M. Protein patterning. Biomaterials 1998, 19, 595–609. [Google Scholar] [CrossRef]
- Sánchez-Salcedo, S.; Izquierdo-Barba, I.; Arcos, D.; Vallet-Regí, M. In vitro evaluation of potential calcium phosphate scaffolds for tissue engineering. Tissue Eng. 2006, 12, 279–290. [Google Scholar] [CrossRef]
- Meganck, J.A.; Baumann, M.J.; Case, E.D.; McCabe, L.R.; Allar, J.N. Biaxial flexure testing of calcium phosphate bioceramics for use in tissue engineering. J. Biomed. Mater. Res. A 2005, 72A, 115–126. [Google Scholar] [CrossRef]
- Case, E.D.; Smith, I.O.; Baumann, M.J. Microcracking and porosity in calcium phosphates and the implications for bone tissue engineering. Mater. Sci. Eng. A 2005, 390, 246–254. [Google Scholar] [CrossRef]
- Sibilla, P.; Sereni, A.; Aguiari, G.; Banzi, M.; Manzati, E.; Mischiati, C.; Trombelli, L.; Del Senno, L. Effects of a hydroxyapatite-based biomaterial on gene expression in osteoblast-like cells. J. Dent. Res. 2006, 85, 354–358. [Google Scholar] [CrossRef]
- Ono, I.; Ohura, T.; Murata, M.; Yamaguchi, H.; Ohnuma, Y.; Kuboki, Y. A study on bone induction in hydroxyapatite combined with bone morphogenetic protein. Plast. Reconstr. Surg. 1992, 90, 870–879. [Google Scholar] [CrossRef]
- Ono, I.; Yamashita, T.; Jin, H.Y.; Ito, Y.; Hamada, H.; Akasaka, Y.; Nakasu, M.; Ogawa, T.; Jimbow, K. Combination of porous hydroxyapatite and cationic liposomes as a vector for BMP-2 gene therapy. Biomaterials 2004, 25, 4709–4718. [Google Scholar] [CrossRef]
- Sawyer, A.A.; Hennessy, K.M.; Bellis, S.L. The effect of adsorbed serum proteins, RGD and proteoglycan-binding peptides on the adhesion of mesenchymal stem cells to hydroxyapatite. Biomaterials 2007, 28, 383–392. [Google Scholar] [CrossRef]
- Mastrogiacomo, M.; Muraglia, A.; Komlev, V.; Peyrin, F.; Rustichelli, F.; Crovace, A.; Cancedda, R. Tissue engineering of bone: Search for a better scaffold. Orthod. Craniofac. Res. 2005, 8, 277–284. [Google Scholar] [CrossRef]
- Schek, R.M.; Taboas, J.M.; Hollister, S.J.; Krebsbach, P.H. Tissue engineering osteochondral implants for temporomandibular joint repair. Orthod. Craniofac. Res. 2005, 8, 313–319. [Google Scholar] [CrossRef]
- Krylova, E.A.; Ivanov, A.A.; Krylov, S.E.; Plashchina, I.G.; Grigorjan, A.S.; Goldstein, D.V.; Pulin, A.A.; Fatkhudinov, T.H. Hydroxyapatite—Alginate structure as living cells supporting system. Minerva Biotecnol. 2006, 18, 17–22. [Google Scholar]
- Nishikawa, M.; Myoui, A.; Ohgushi, H.; Ikeuchi, M.; Tamai, N.; Yoshikawa, H. Bone tissue engineering using novel interconnected porous hydroxyapatite ceramics combined with marrow mesenchymal cells: Quantitative and three-dimensional image analysis. Cell Transplant. 2004, 3, 367–376. [Google Scholar]
- Quarto, R.; Mastrogiacomo, M.; Cancedda, R.; Kutepov, S.M.; Mukhachev, V; Lavroukov, A.; Kon, E.; Marcacci, M. Repair of large bone defects with the use of autologous bone marrow stromal cells. N. Engl. J. Med. 2001, 344, 385–386. [Google Scholar] [CrossRef]
- Vacanti, C.A.; Bonassar, L.J.; Vacanti, M.P.; Shufflebarger, J. Replacement of an avulsed phalanx with tissue-engineered bone. N. Engl. J. Med. 2001, 344, 1511–1514. [Google Scholar] [CrossRef]
- Morishita, T.; Honoki, K.; Ohgushi, H.; Kotobuki, N.; Matsushima, A.; Takakura, Y. Tissue engineering approach to the treatment of bone tumors: Three cases of cultured bone grafts derived from patients’ mesenchymal stem cells. Artif. Organs 2006, 30, 115–118. [Google Scholar] [CrossRef]
- Eniwumide, J.O.; Yuan, H.; Cartmell, S.H.; Meijer, G.J.; De Bruijn, J.D. Ectopic bone formation in bone marrow stem cell seeded calcium phosphate scaffolds as compared to autograft and (cell seeded) allograft. Eur. Cell Mater. 2007, 14, 30–39. [Google Scholar]
- Freed, L.E.; Langer, R.; Martin, I.; Pellis, N.R.; Vunjak-Novakovic, G. Tissue engineering of cartilage in space. Proc. Natl. Acad. Sci. USA 1997, 94, 13885–13890. [Google Scholar] [CrossRef]
- Holy, C.E.; Shoichet, M.S.; Davies, J.E. Engineering three-dimensional bone tissue in vitro using biodegradable scaffolds: Investigating initial cell-seeding density and culture period. J. Biomed. Mater. Res. 2000, 51, 376–382. [Google Scholar] [CrossRef]
- Shimizu, K.; Ito, A.; Honda, H. Enhanced cell-seeding into 3D porous scaffolds by use of magnetite nanoparticles. J. Biomed. Mater. Res. B Appl. Biomater. 2006, 77B, 265–272. [Google Scholar] [CrossRef]
- Hench, L.L.; Wilson, J. Surface-active biomaterials. Science 1984, 226, 630–636. [Google Scholar]
- Navarro, M.; Michiardi, A.; Castano, O.; Planell, J.A. Biomaterials in orthopaedics. J. R. Soc. Interface 2008, 5, 1137–1158. [Google Scholar] [CrossRef]
- Anderson, J.M. The future of biomedical materials. J. Mater. Sci. Mater. Med. 2006, 17, 1025–1028. [Google Scholar] [CrossRef]
- Chevalier, J.; Gremillard, L. Ceramics for medical applications: A picture for the next 20 years. J. Eur. Ceram. Soc. 2009, 29, 1245–1255. [Google Scholar] [CrossRef]
- Hartgerink, J.D.; Beniash, E.; Stupp, S.I. Self-assembly and mineralization of peptide-amphiphile nanofibers. Science 2001, 294, 1684–1688. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Dorozhkin, S.V. Calcium Orthophosphates as Bioceramics: State of the Art. J. Funct. Biomater. 2010, 1, 22-107. https://doi.org/10.3390/jfb1010022
Dorozhkin SV. Calcium Orthophosphates as Bioceramics: State of the Art. Journal of Functional Biomaterials. 2010; 1(1):22-107. https://doi.org/10.3390/jfb1010022
Chicago/Turabian StyleDorozhkin, Sergey V. 2010. "Calcium Orthophosphates as Bioceramics: State of the Art" Journal of Functional Biomaterials 1, no. 1: 22-107. https://doi.org/10.3390/jfb1010022






