A Naturally Occurring Antioxidant Complex from Unripe Grapes: The Case of Sangiovese (v. Vitis vinifera)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Sangiovese Grapes
2.3. Wines
2.4. Preparation of Juice and Powder
2.5. General Analyses
2.6. Total Polyphenols (TP) Determination
2.7. Total Anthocyanins Determination
2.8. 2,2-Diphenyl-1-Picryhydrazil (DPPH) Antioxidant Test
2.9. Liquid Chromatography-High-Resolution Mass Spectrometry (LC-HRMS) Analysis
2.10. HPLC-Determination of Anthocyanins
2.11. Polyphenols Oxidative Medium (POM) Test
2.12. Statistics Analysis
3. Results and Discussion
3.1. Solid-Liquid Extraction and Juice Composition
3.2. Antioxidat Activity and Effect on White Wine
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dani, C.; Bonatto, D.; Salvador, M.; Pereira, M.D.; Henriques, J.A.P.; Eleutherio, E. Antioxidant protection of resveratrol and catechin in Saccharomyces cerevisiae. J. Agric. Food Chem. 2008, 56, 4268–4272. [Google Scholar] [CrossRef] [PubMed]
- Heim, K.E.; Tagliaferro, A.R.; Bobilya, D.J. Flavonoid antioxidants: Chemistry, metabolism and structure-activity relationships. J. Nutr. Biochem. 2002, 13, 572–584. [Google Scholar] [CrossRef]
- Williams, R.J.; Spencer, J.P.E.; Rice-Evans, C. Flavonoids: Antioxidants or signalling molecules? Free Radic. Biol. Med. 2004, 36, 838–849. [Google Scholar] [CrossRef] [PubMed]
- Tsao, R. Chemistry and biochemistry of dietary polyphenols. Nutrients 2010, 2, 1231–1246. [Google Scholar] [CrossRef] [PubMed]
- Xia, E.; He, X.; Li, H.; Wu, S.; Li, S.; Deng, G. Biological Activities of Polyphenols from Grapes. Int. J. Mol. Sci. 2010, 11, 622–646. [Google Scholar] [CrossRef] [PubMed]
- Dumitriu, D.; Peinado, R.A.; Peinado, J.; de Lerma, N.L. Grape pomace extract improves the in vitro and in vivo antioxidant properties of wines from sun light dried Pedro Ximnez grapes. J. Funct. Foods 2015, 17, 380–387. [Google Scholar] [CrossRef]
- Hogan, S.; Zhang, L.; Li, J.; Zoecklein, B.; Zhou, K. Antioxidant properties and bioactive components of Norton (Vitis aestivalis) and Cabernet Franc (Vitis vinifera) wine grapes. LWT Food Sci. Technol. 2009, 42, 1269–1274. [Google Scholar] [CrossRef]
- Muñoz, A.; Ramos, F. Componentes fenólicos de la dieta y sus propiedades biomedicinales. Horiz. Med. 2007, 7, 23–31. [Google Scholar]
- Hollman, P.C.H.; Bijsman, M.N.C.P.; Van Gameren, Y.; Cnossen, E.P.J.; De Vries, J.H.M.; Katan, M.B. The sugar moiety is a major determinant of the absorption of dietary flavonoid glycosides in man. Free Radic. Res. 1999, 31, 569–573. [Google Scholar] [CrossRef] [PubMed]
- Makris, D.P.; Boskou, G.; Andrikopoulos, N.K. Polyphenolic content and in vitro antioxidant characteristics of wine industry and other Agri-food solid waste extracts. J. Food Compos. Anal. 2007, 20, 125–132. [Google Scholar] [CrossRef]
- Öncül, N.; Karabiyikli, Ş. Factors affecting the quality attributes of unripe grape functional food products. J. Food Biochem. 2015, 39, 689–695. [Google Scholar] [CrossRef]
- Kontoudakis, N.; Esteruelas, M.; Fort, F.; Canals, J.M.; Zamora, F. Use of unripe grapes harvested during cluster thinning as a method for reducing alcohol content and pH of wine. Aust. J. Grape Wine Res. 2011, 17, 230–238. [Google Scholar] [CrossRef]
- Tinello, F.; Lante, A. Evaluation of antibrowning and antioxidant activities in unripe grapes recovered during bunch thinning. Aust. J. Grape Wine Res. 2017, 23, 33–41. [Google Scholar] [CrossRef]
- Shojaee-Aliabadi, S.; Hosseini, S.M.; Tiwari, B.; Hashemi, M.; Fadavi, G.; Khaksar, R. Polyphenols content and antioxidant activity of Ghure (unripe grape) marc extract: Influence of extraction time, temperature and solvent type. Int. J. Food Sci. Technol. 2013, 48, 412–418. [Google Scholar] [CrossRef]
- Mattivi, F.; Guzzon, R.; Vrhovsek, U.; Stefanini, M.; Velasco, R. Metabolite profiling of grapes: Flavonols and anthocyanins. J. Agric. Food Chem. 2006, 54, 7692–7702. [Google Scholar] [CrossRef] [PubMed]
- Coombe, B.G. Adoption of a system for identifying grapevine growth stages. Aust. J. Grape Wine Res. 1995, 1, 104–110. [Google Scholar] [CrossRef]
- Gori, C.; Menichetti, S.; Fia, G. Multi-Functional Oenological Machine and Use in the Oenological Production Chain. European Patent 2,957,627, 22 June 2014. [Google Scholar]
- International Organisation of the Vine and Wine (OIV). International Organisation of the Vine and Wine Website. 2014. Available online: http://www.oiv.int/oiv/info/frmethodesanalyses (accessed on 15 June 2015).
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Di Stefano, R.; Cravero, M.C.; Gentilini, N. Metodi per lo studio dei polifenoli dei vini. Enotecnico 1989, 25, 83–89. [Google Scholar]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Muller-Spath, H. Der POM-test. Deutsche Weinbau 1992, 23, 1099–1100. [Google Scholar]
- Camuzzo, P.; Battistutta, F.; Vendrane, M.; Páez, M.S.; Luisi, G.; Zironi, R. Antioxidant properties of different products and additives in white wine. Food Chem. 2015, 168, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Gambacorta, G.; Antonacci, D.; Pati, S.; La Gatta, M.; Faccia, M.; Coletta, A.; La Notte, E. Influence of winemaking technologies on phenolic composition of Italian red wines. Eur. Food Res. Technol. 2011, 233, 1057–1066. [Google Scholar] [CrossRef]
- Zanoni, B.; Siliani, S.; Canuti, V.; Rosi, I.; Bertuccioli, M. A kinetic study on extraction and transformation phenomena of phenolic compounds during red wine fermentation. Int. J. Food Sci. Technol. 2010, 45, 2080–2088. [Google Scholar] [CrossRef]
- Singleton, V.L.; Salgues, M.; Zaya, J.; Trousdale, E. Caftaric acid disappearance and conversion to products of enzymic oxidation in grape must and wine. Am. J. Enol. Vitic. 1985, 36, 50–56. [Google Scholar]
- Kritzinger, E.C.; Bauer, F.F.; Du Toit, W.J. Role of glutathione in winemaking: A review. J. Agric. Food Chem. 2013, 61, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Zurbano, P.; Ferreira, V.; Peña, C.; Escudero, A.; Serrano, F.; Cacho, J. Prediction of oxidative browning in white wines as a function of their chemical composition. J. Agric. Food Chem. 1995, 43, 2813–2817. [Google Scholar] [CrossRef]
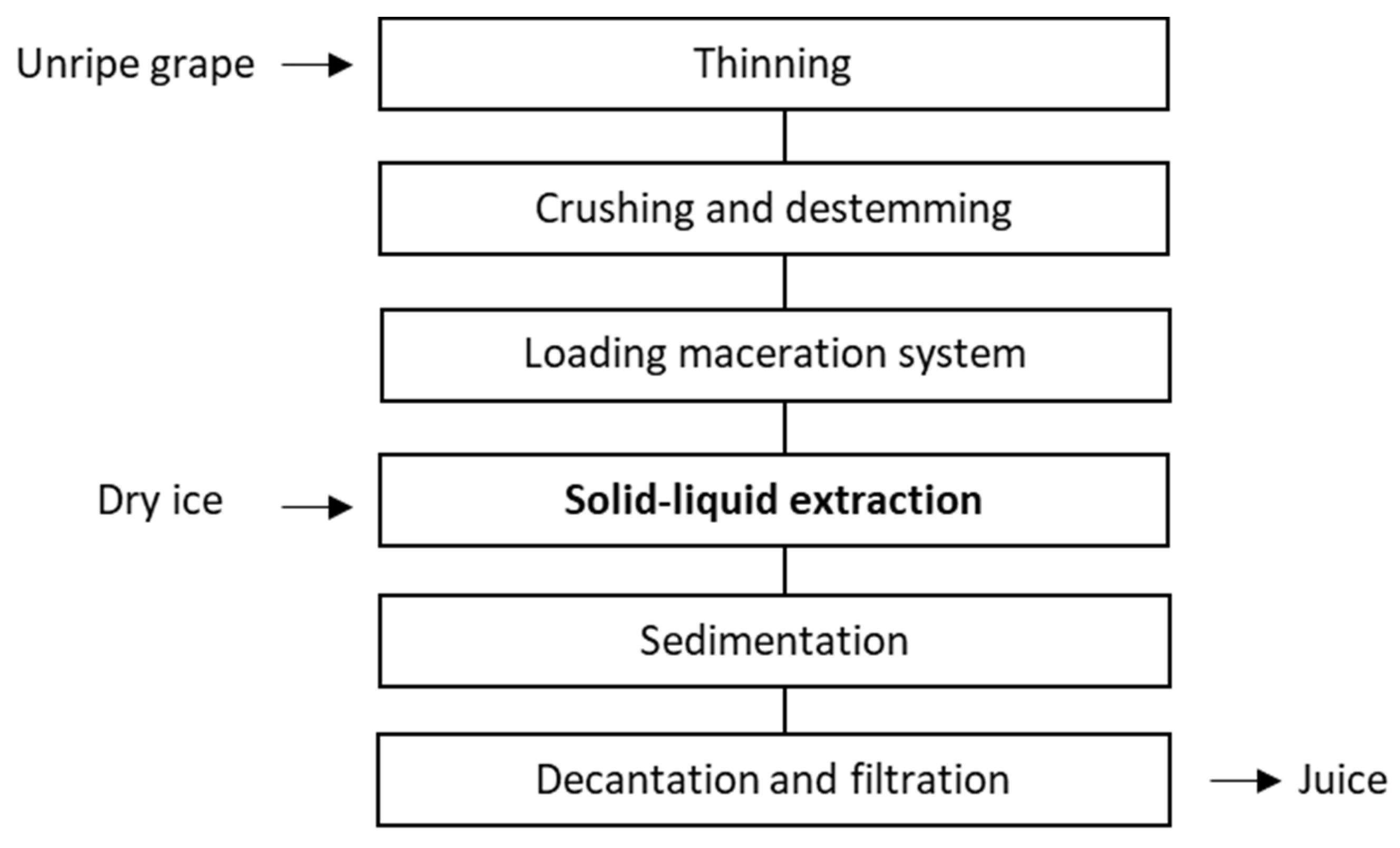

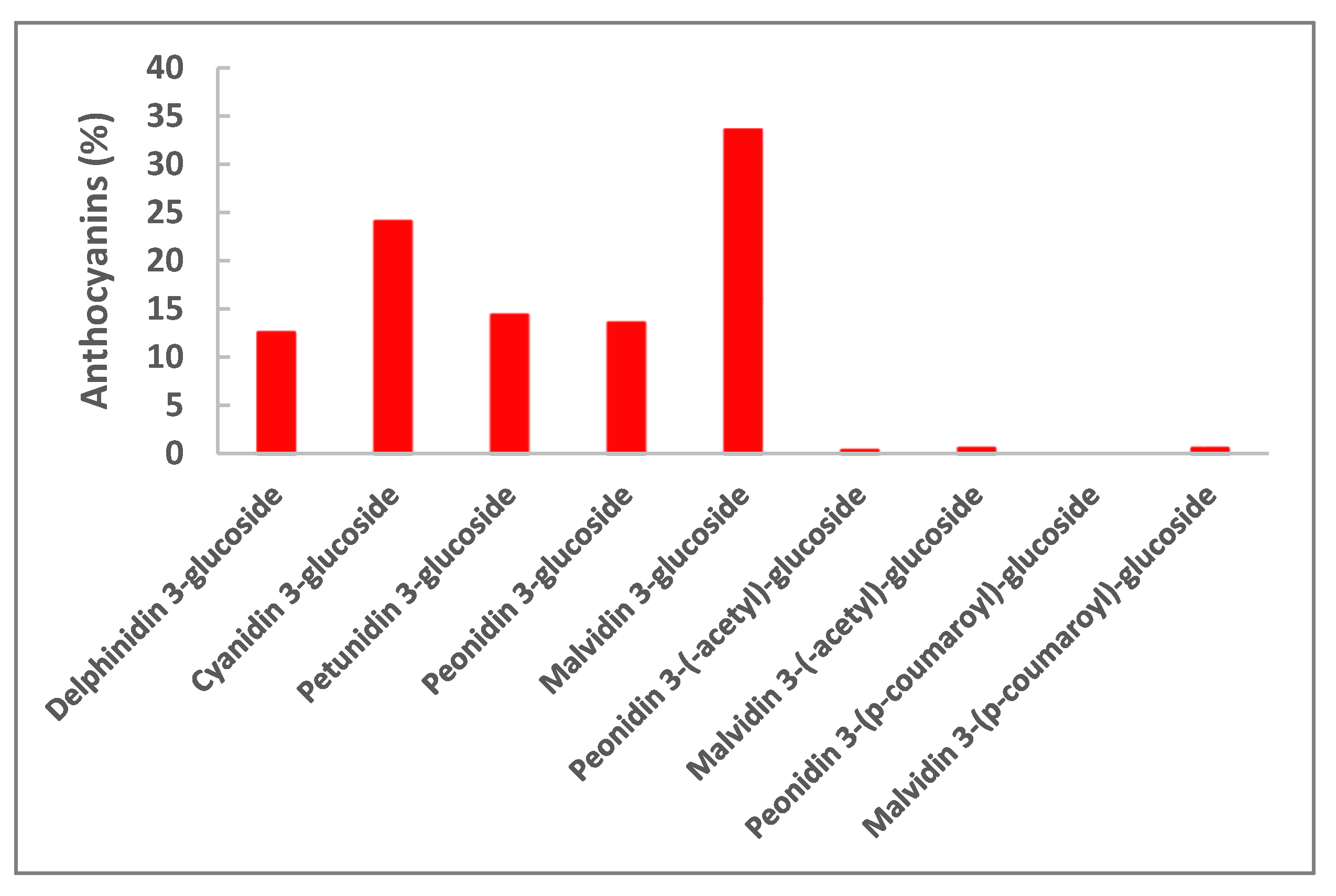
| Phenolic Compounds | Concentration (mg/L) of Juice * | Concentration (μg/g of Powder) * | |
|---|---|---|---|
| Time | S-D + AG | ||
| 2 h | 72 h | ||
| Phenolic Acids | |||
| Caffeic acid | 0.06 ± 0.01 a | 0.43 ± 0.16 b | 0.8 ± 0.0 |
| Coumaric acid | 0.03 ± 0.00 | nd | 0.5 ± 0.1 |
| Ferulic acid | 1.7 ± 0.4 b | 0.14 ± 0.00 a | 29.4 ± 4.7 |
| Caftaric acid | 50.0 ± 6.6 b | 27.5 ± 1.7 a | 191 ± 5 |
| Coutaric acid | 15.9 ± 1.8 b | 9.4 ± 0.1 a | 27.6 ± 4.4 |
| Fertaric acid | 16.8 ± 3.1 a | 30.1 ± 3.3 b | 291 ± 73 |
| Gallic acid | 0.05 ± 0.00 a | 9.4 ± 2.1 b | 9.5 ± 0.3 |
| Flavonols | |||
| Quercetin | 0.07 ± 0.00 a | 0.29 ± 0.01 b | 1.3 ± 0.0 |
| Quercetin 3-O-glucoside | 0.13 ± 0.02 a | 19.8 ± 0.8 b | 11.8 ± 0.8 |
| Quercetin 3-O-glucuronide | 0.38 ± 0.07 a | 27.0 ± 1.2 b | 56.6 ± 3.1 |
| Rutin | 0.02 ± 0.00 a | 0.40 ± 0.03 b | 0.4 ± 0.0 |
| Isorhamnetin | 0.04 ± 0.00 | nd | 0.7 ± 0.0 |
| Kaempferol | 0.02 ± 0.00 | nd | 0.5 ± 0.0 |
| Myricetin | nd | 0.03 ± 0.00 b | nd |
| Flava-3-Ols | |||
| (−)-Epicatechin | 0.24 ± 0.02 a | 39.5 ± 1.6 b | 64 ± 12 |
| (+) Catechin | 6.3 ± 0.2 a | 38.4 ± 1.3 b | 327 ± 19 |
| Procyanidins | |||
| Procyanidin B1 | 0.82 ± 0.11 a | 23.9 ± 0.9 b | 19.0 ± 1.5 |
| Procyanidin B2 | 0.15 ± 0.00 a | 2.0 ± 0.1 b | 10.1 ± 1.0 |
| Stilbenes | |||
| Resveratrol | 0.01 ± 0.00 a | ± 0.00 b | 0.2 ± 0.0 |
| Sample | Total Polyphenols * (mg CATeq/L of Juice or g of Powder) | Antioxidant Capacity * (µmoL TEAC/L of Juice or g of Powder) |
|---|---|---|
| Juice | 1214.6 ± 37.8 | 5345.8 ± 119.3 |
| S-D + AG | 2.3 ± 0.01 | 24.4 ± 0.00 |
| Sample | PH | Total Acidity (g/L as Tartaric Acid Equivalents) | Free SO2 (mg/L) | Total SO2 (mg/L) |
|---|---|---|---|---|
| Viognier | 3.35 | 5.5 | 5.0 | 11.0 |
| Chardonnay | 3.06 | 5.6 | 15.5 | 42.3 |
| Bellone | 2.90 | 7.1 | 12.8 | 16.6 |
| Wine | Wine | Wine + Powder |
|---|---|---|
| % OXH2O2 | % OXH2O2 | |
| Viognier | 12 | −15 |
| Chardonnay | 34 | 5 |
| Bellone | 9 | 0 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fia, G.; Gori, C.; Bucalossi, G.; Borghini, F.; Zanoni, B. A Naturally Occurring Antioxidant Complex from Unripe Grapes: The Case of Sangiovese (v. Vitis vinifera). Antioxidants 2018, 7, 27. https://doi.org/10.3390/antiox7020027
Fia G, Gori C, Bucalossi G, Borghini F, Zanoni B. A Naturally Occurring Antioxidant Complex from Unripe Grapes: The Case of Sangiovese (v. Vitis vinifera). Antioxidants. 2018; 7(2):27. https://doi.org/10.3390/antiox7020027
Chicago/Turabian StyleFia, Giovanna, Claudio Gori, Ginevra Bucalossi, Francesca Borghini, and Bruno Zanoni. 2018. "A Naturally Occurring Antioxidant Complex from Unripe Grapes: The Case of Sangiovese (v. Vitis vinifera)" Antioxidants 7, no. 2: 27. https://doi.org/10.3390/antiox7020027





