Gene Expression Profiling during Pregnancy in Rat Brain Tissue
Abstract
:1. Introduction
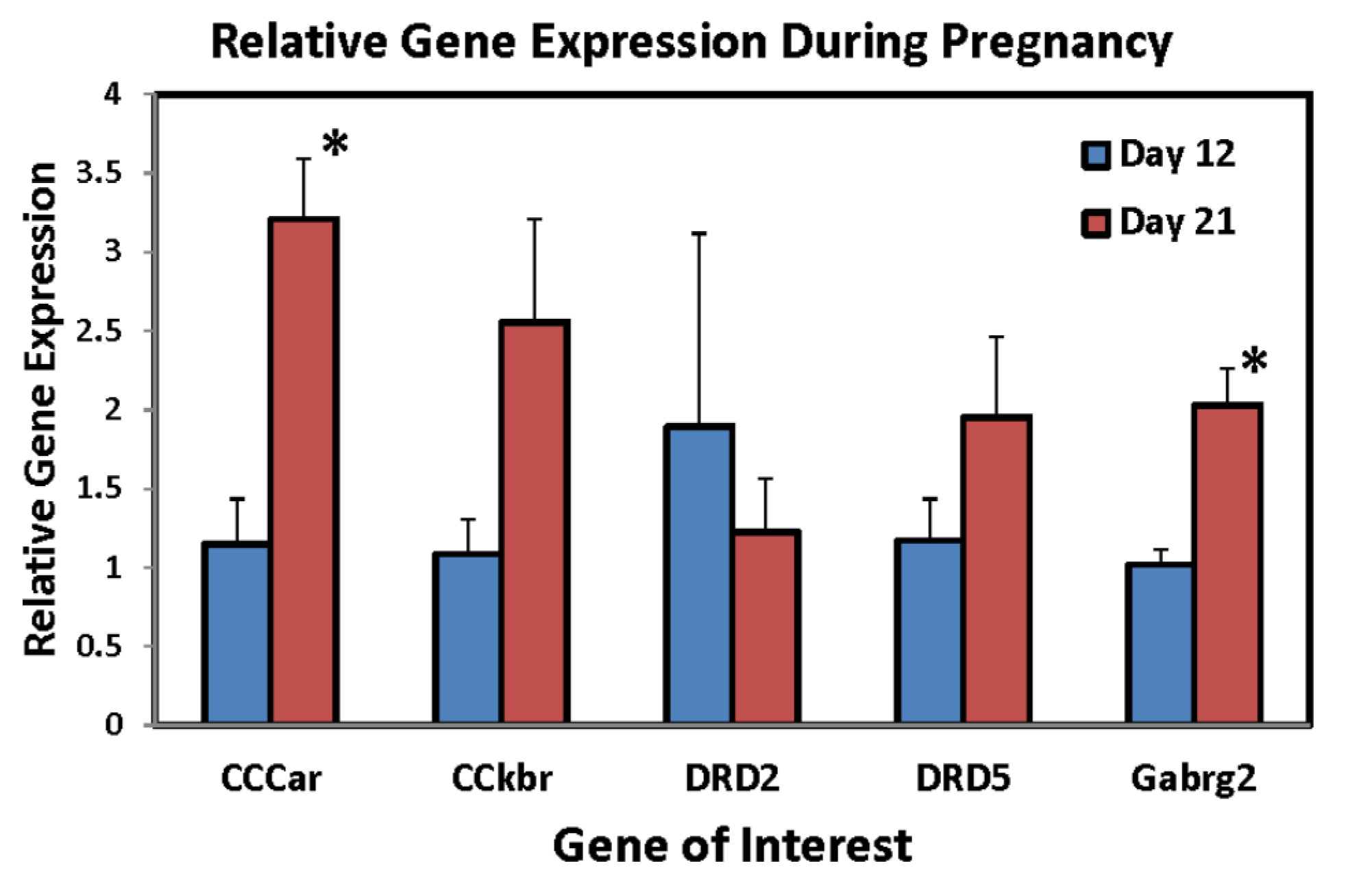
2. Results and Discussion
| A | Fold Difference |
| Acetylcholinesterase (Hache) | 2.92 |
| Cholinergic receptor, muscarinic 1 (Chrm1) | 10.99 |
| Cholinergic receptor, muscarinic 3 (Chrm3) | 22.56 |
| Cholinergic receptor, muscarinic 4 (M4) | 2.84 |
| Cholinergic receptor, nicotinic, alpha polypeptide 2 (ACHR) | 3.59 |
| Cholinergic receptor, nicotinic, beta polypeptide 2 (Chrnb2) | 2.08 |
| Cholecystokinin B receptor (CHOLREC) | 2.1 |
| Dopamine receptor D5 (Drd5) | 3.57 |
| G protein-coupled receptor 83 (Gir) | 5.53 |
| Gastrin releasing peptide receptor (Grpr) | 4.29 |
| Glutamic acid decarboxylase 1 (Gad67) | 2.83 |
| Neuropeptide FF receptor 1 (Gpr147/NPFF1) | 8.95 |
| Neuropeptide Y receptor Y1 (NPY-1) | 5.42 |
| Prokineticin receptor 2 (Gpr73l1) | 5.83 |
| Prolactin releasing hormone receptor (Gpr10/Uhr-1) | 8.22 |
| Gamma-aminobutyric-acid receptor alpha-2 subunit precursor (GABA(A) receptor) (LOC289606) | 2.1 |
| Somatostatin receptor 1 (Gpcrrna) | 3.65 |
| Somatostatin receptor 3 (Smstr28) | 3.29 |
| Somatostatin receptor 4 (Smstr4) | 3.19 |
| Tachykinin receptor 1 (Tac1r) | 3.54 |
| B | Fold Difference |
| Cholecystokinin A receptor (Cck-ar) | 2.64 |
| Dopamine receptor 2 (dopamine D2) | 2.67 |
| Dopamine receptor D1A (D1a/Drd-1) | 2.1 |
| Gamma-aminobutyric acid A receptor, gamma 2 (Gabrg2) | 2.24 |
| Neuropeptide FF receptor 2 (Gpr74/Npff2) | 3.09 |
3. Experimental Section
3.1. Animals
3.2. Tissue Collection
3.3. Sample Preparation
3.4. Quantitative Real-Time PCR (qPCR)
3.5. Reverse Transcription PCR (RT-PCR)
| Gene | Accession # | Direction | Sequence |
|---|---|---|---|
| Colecystokinin A receptor (CCKar) | NM_012688 | Forward | 5'- ATGCAGCAGTCCTGGCAAACATTC-3' |
| Reverse | 5'-TTTGGCAGATTTCTTCTGGCTGGC-3' | ||
| Colecystokinin B receptor (CCKbr) | NM_013165 | Forward | 5'-AGCGATACAGCGCCATCTG-3' |
| Reverse | 5'-CGTGGGAGCGTGTTTGC-3' | ||
| Dopamine receptor 2 (Drd2) | NM_012547 | Forward | 5'-TGACAGTCCTGCCAAACCAGAGAA-3' |
| Reverse | 5'-TGGGCATGGTCTGGATCTCAAAGA-3' | ||
| Dopamine receptor 5 (Drd5) | NM_012768 | Forward | 5'-TGTGTATCATCAGCGTGGACCGTT-3' |
| Reverse | 5'-ATTGAGTTGGACCGGGATGAAGGA-3' | ||
| Gamma-aminobutyric acid A receptor (Gabrg2) | NM_183327 | Forward | 5'-GCAACCGGAAACCAAGCAAGGATA-3' |
| Reverse | 5'-GGTGGGTGGCATTGTTCATTTGGA-3' |
3.6. Data Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Brunton, P.J.; Russell, J.A. The expectant brain: Adapting for motherhood. Nat. Rev. Neurosci. 2008, 9, 11–25. [Google Scholar] [CrossRef]
- Kinsley, C.H.; Amory-Meyer, E. Why the maternal brain? J. Neuroendocrinol. 2011, 23, 974–983. [Google Scholar] [CrossRef]
- Brunton, P.J.; Russell, J.A. Allopregnanolone and suppressed hypothalamo-pituitary-adrenal axis stress responses in late pregnancy in the rat. Stress 2011, 14, 6–12. [Google Scholar]
- Zimberknopf, E.; Xavier, G.F.; Kinsley, C.H.; Felicio, L.F. Prior parity positively regulates learning and memory in young and middle-aged rats. Comp. Med. 2011, 61, 366–377. [Google Scholar]
- Russell, J.A.; Douglas, A.J.; Ingram, C.D. Brain preparations for maternity—Adaptive changes in behavioral and neuroendocrine systems during pregnancy and lactation. An overview. Prog. Brain Res. 2001, 133, 1–38. [Google Scholar]
- Ladyman, S.R.; Augustine, R.A.; Grattan, D.R. Hormone interactions regulating energy balance during pregnancy. J. Neuroendocrinol. 2010, 22, 805–817. [Google Scholar]
- Ladyman, S.R.; Fieldwick, D.M.; Grattan, D.R. Suppression of leptin-induced hypothalamic JAK/STAT signalling and feeding response during pregnancy in the mouse. Reproduction 2012, 144, 83–90. [Google Scholar] [CrossRef]
- Elmquist, J.K.; Bjorbaek, C.; Ahima, R.S.; Flier, J.S.; Saper, C.B. Distributions of leptin receptor mRNA isoforms in the rat brain. J. Comp. Neurol. 1998, 395, 535–547. [Google Scholar] [CrossRef]
- Flanagan-Cato, L.M. Sex Differences in the neural circuit that mediates female sexual receptivity. Front. Neuroendocrinol. 2011, 32, 124–136. [Google Scholar] [CrossRef]
- Bridges, R.S.; Mann, P.E.; Coppeta, J.S. Hypothalamic involvement in the regulation of maternal behaviour in the rat: Inhibitory roles for the ventromedial hypothalamus and the dorsal/anterior hypothalamic areas. J. Neuroendocrinol. 1999, 11, 259–266. [Google Scholar] [CrossRef]
- Mann, P.E.; Babb, J.A. Disinhibition of maternal behavior following neurotoxic lesions of the hypothalamus in primigravid rats. Brain Res. 2004, 1025, 51–58. [Google Scholar]
- Slotnick, B.M.; Carpenter, M.L.; Fusco, R. Initiation of maternal behavior in pregnant nulliparous rats. Horm. Behav. 1973, 4, 53–59. [Google Scholar] [CrossRef]
- Mayer, A.D.; Rosenblatt, J.S. Prepartum changes in maternal responsiveness and nest defense in rattus norvegicus. J. Comp. Psychol. 1984, 98, 177–188. [Google Scholar] [CrossRef]
- Rosenblatt, J.S. Nonhormonal basis of maternal behavior in the rat. Science 1967, 156, 1512–1514. [Google Scholar]
- Bridges, R.S.; Zarrow, M.X.; Goldman, B.D.; Denenberg, V.H. A developmental study of maternal responsiveness in the rat. Physiol. Behav. 1974, 12, 149–151. [Google Scholar] [CrossRef]
- Brunelli, S.A.; Hofer, M.A. Parental Behavior in Juvenile Rats: Environmental and Biological Determinants. In Mammalian Parenting; Krasnegor, N.A., Bridges, R.S., Eds.; Oxford University Press: New York, NY, USA, 1990; pp. 372–299. [Google Scholar]
- Stern, J.M. Pubertal decline in maternal responsiveness in long-evans rats: Maturational influences. Physiol. Behav. 1987, 41, 93–98. [Google Scholar] [CrossRef]
- Fleming, A.S.; Rosenblatt, J.S. Olfactory regulation of maternal behavior in rats. II. Effects of peripherally induced anosmia and lesions of lateral olfactory tract in pup-induced virgins. J. Comp. Physiol. Psychol. 1974, 86, 233–246. [Google Scholar] [CrossRef]
- Fleming, A.S.; Vaccarino, F.; Tambosso, L.; Chee, P. Vomeronasal and olfactory system modulation of maternal behavior in the rat. Science 1979, 203, 372–374. [Google Scholar]
- Fleming, A.S.; Vaccarino, F.; Luebke, C. Amygdaloid inhibition of maternal behavior in the nulliparous female rat. Physiol. Behav. 1980, 25, 731–743. [Google Scholar] [CrossRef]
- Numan, M.; Numan, M.J.; English, J.B. Excitotoxic amino acid injections into the medial amygdala facilitate maternal behavior in virgin female rats. Horm. Behav. 1993, 27, 56–81. [Google Scholar] [CrossRef]
- Numan, M.; Numan, M. A Lesion and neuroanatomical tract-tracing analysis of the role of the bed nucleus of the stria terminalis in retrieval behavior and other aspects of maternal responsiveness in rats. Dev. Psychobiol. 1996, 29, 23–51. [Google Scholar] [CrossRef]
- Kinsley, C.H.; Meyer, E.; Rafferty, K.A. Sex steroid hormone determination of the maternal brain: effects beyond reproduction. Mini Rev. Med. Chem. 2012, 12, 1063–1070. [Google Scholar] [CrossRef]
- Numan, M.; Woodside, B. Maternity: Neural mechanisms, motivational processes, and physiological adaptations. Behav. Neurosci. 2010, 124, 715–741. [Google Scholar] [CrossRef]
- Dwyer, J.B.; Broide, R.S.; Leslie, F.M. Nicotine and Brain Development. Birth Defects Res. C Embryo Today 2008, 84, 30–44. [Google Scholar] [CrossRef]
- Mao, C.; Lv, J.; Li, H.; Chen, Y.; Wu, J.; Xu, Z. Development of fetal nicotine and muscarinic receptors in utero. Braz. J. Med. Biol. Res. 2007, 40, 735–741. [Google Scholar] [CrossRef]
- Sanchez-Andrade, G.; Kendrick, K.M. The main olfactory system and social learning in mammals. Behav. Brain Res. 2009, 200, 323–335. [Google Scholar] [CrossRef]
- Kaufman, L.S.; McEwen, B.S.; Pfaff, D.W. Cholinergic mechanisms of lordotic behavior in rats. Physiol. Behav. 1988, 43, 507–514. [Google Scholar] [CrossRef]
- Kow, L.M.; Pfaff, D.W. Transmitter and peptide actions on hypothalamic neurons in vitro: Implications for lordosis. Brain Res. Bull. 1988, 20, 857–861. [Google Scholar] [CrossRef]
- Kow, L.M.; Tsai, Y.F.; Weiland, N.G.; McEwen, B.S.; Pfaff, D.W. In vitro electro-pharmacological and autoradiographic analyses of muscarinic receptor subtypes in rat hypothalamic ventromedial nucleus: implications for cholinergic regulation of lordosis. Brain Res. 1995, 694, 29–39. [Google Scholar] [CrossRef]
- Rainbow, T.C.; Snyder, L.; Berck, D.J.; McEwen, B.S. Correlation of muscarinic receptor induction in the ventromedial hypothalamic nucleus with the activation of feminine sexual behavior by estradiol. Neuroendocrinology 1984, 39, 476–480. [Google Scholar] [CrossRef]
- Voogt, J.L.; Lee, Y.; Yang, S.; Arbogast, L. Regulation of prolactin secretion during pregnancy and lactation. Prog. Brain Res. 2001, 133, 173–185. [Google Scholar] [CrossRef]
- Arbogast, L.A.; Voogt, J.L. Mechanisms of tyrosine hydroxylase regulation during pregnancy: Evidence for protein dephosphorylation during the prolactin surges. Endocrinology 1991, 129, 2575–2582. [Google Scholar] [CrossRef]
- Andrews, Z.B.; Kokay, I.C.; Grattan, D.R. Dissociation of Prolactin secretion from tuberoinfundibular dopamine activity in late pregnant rats. Endocrinology 2001, 142, 2719–2724. [Google Scholar]
- Bakowska, J.C.; Morell, J.I. Quantitative autoradiographic analysis of D1 and D2 dopamine receptors in rat brain in early and late pregnancy. Brain Res. 1995, 703, 191–200. [Google Scholar] [CrossRef]
- Stern, J.M.; Keer, S.E. Maternal motivation of lactating rats is disrupting by low dosages of haloperidol. Behav. Brain Res. 1999, 99, 231–239. [Google Scholar] [CrossRef]
- Silva, M.R.P.; Bernardi, M.M.; Felicio, L.F. Effects of dopamine receptor antagonists on ongoing maternal behavior in rats. Pharmacol. Biochem. Behav. 2001, 68, 461–468. [Google Scholar] [CrossRef]
- Miller, S.M.; Lonstein, J.S. Dopamine D1 and D2 receptor antagonism in the preoptic area produces different effects on maternal behavior in lactating rats. Behav. Neurosci. 2005, 119, 1072–1083. [Google Scholar] [CrossRef]
- Numan, M.; Numan, M.J.; Pliakou, N.; Stolzenberg, D.S.; Mullins, O.J.; Murphy, J.M.; Smith, C.D. The effects of D1 or D2 dopamine receptor antagonism in the medial preoptic area, ventral pallidum, or nucleus accumbens on the maternal retrieval response and other aspects of maternal behavior in rats. Behav. Neurosci. 2005, 119, 1588–1604. [Google Scholar] [CrossRef]
- Silva, M.R.; Bernardi, M.M.; Cruz-Casallas, P.E.; Felicio, L.F. Pimozide injections into the nucleus accumbens disrupt maternal behaviour in lactating rats. Pharmacol. Toxicol. 2003, 93, 42–47. [Google Scholar] [CrossRef]
- Hansen, S.; Harthon, C.; Wallin, E.; Lofberg, L.; Svensson, K. The effects of 6-OHDA-induced dopamine depletions in the ventral or dorsal striatum on maternal and sexual behavior in the female rats. Pharmacol. Biochem. Behav. 1991, 39, 71–77. [Google Scholar] [CrossRef]
- Stolzenberg, D.S.; McKenna, J.B.; Keough, S.; Hancock, R.; Numan, M.J.; Numan, M. Dopamine D1 receptor stimulation of the nucleus accumbens or the medial preoptic area promotes the onset of maternal behavior in pregnancy-terminated rats. Behav. Neurosci. 2007, 121, 907–919. [Google Scholar] [CrossRef]
- Stolzenberg, D.S.; Zhang, K.Y.; Luskin, K.; Ranker, L.; Bress, J.; Numan, M. Dopamine D1 receptor activation of adenylyl cyclase, not phospholipase c, in the nucleus accumbens promotes maternal behavior onset in rats. Horm. Behav. 2010, 57, 96–104. [Google Scholar] [CrossRef]
- Follesa, P.; Floris, S.; Tuligi, G.; Mostallino, M.C.; Concas, A.; Biggio, G. Molecular and functional adaptation of the GABAA receptor complex during pregnancy and after delivery in the rat brain. Eur. J. Neurosci. 1998, 10, 2905–2912. [Google Scholar] [CrossRef]
- Fenelon, V.S.; Herbison, A.E. Plasticity in GABAA receptor subunit mRNA expression by hypothalamic magnocellular neurons in the adult rat. J. Neurosci. 1996, 16, 4872–4880. [Google Scholar]
- Carretero, M.I.; Segovia, S.; Gomez, F.; del Cerro, M.C.R. Bicuculline Infusion into the accessory olfactory bulb facilitates the induction of maternal behavior in rats. Scand. J. Psychol. 2003, 44, 273–277. [Google Scholar] [CrossRef]
- Concas, A.; Mostallino, M.C.; Porcu, P.; Follesa, P.; Barbaccia, M.L.; Trabucchi, M.; Purdy, R.H.; Grisenti, P.; Biggio, G. Role of brain allopregnanolone in the plasticity of gamma-aminobutyric acid type A receptor in rat brain during pregnancy and after delivery. Proc. Natl. Acad. Sci. USA 1998, 95, 13284–13289. [Google Scholar] [CrossRef]
- Russell, J.A.; Douglas, A.J.; Brunton, P.J. Reduced hypothalamo-pituitary-adrenal axis stress responses in late pregnancy: Central opioid inhibition and noradrenergic mechanisms. Ann. N. Y. Acad. Sci. 2008, 1148, 428–438. [Google Scholar]
- Dockray, G.J. Cholecystokinin. Curr. Opin. Endocrinol. Diabetes Obes. 2012, 19, 8–12. [Google Scholar] [CrossRef]
- Noble, F.; Roques, B.P. Cholecystokinin Peptides in Brain Function. In Handbook of Neurochemistry and Molecular Neurobiology; Springer: New York, NY, USA, 2006; pp. 545–571. [Google Scholar]
- Ladyman, S.R.; Sapsford, T.J.; Grattan, D.R. Loss of acute satiety response to cholecystokinin in pregnant rats. J. Neuroendocrinol. 2011, 23, 1091–1098. [Google Scholar] [CrossRef]
- Wank, S.A. Cholecystokinin receptors. Am. J. Physiol. 1995, 269, G628–G646. [Google Scholar]
- Linden, A.; Uvnas-Moberg, K.; Eneroth, P.; Sodersten, P. Stimulation of maternal behaviour in rats with cholecystokinin octapeptide. J. Neuroendocrinol. 1989, 1, 389–392. [Google Scholar] [CrossRef]
- Mann, P.E.; Felicio, L.F.; Bridges, R.S. Investigation into the role of cholecystokinin (CCK) in the induction and maintenance of maternal behavior in rats. Horm. Behav. 1995, 29, 392–406. [Google Scholar] [CrossRef]
- Sabatier, N.; Leng, G. Responses to cholecystokinin in the ventromedial nucleus of the rat hypothalamus in vivo. Eur. J. Neurosci. 2010, 31, 1127–1135. [Google Scholar] [CrossRef]
- Popper, P.; Priest, C.A.; Micevych, P.E. Regulation of cholecystokinin receptors in the ventromedial nucleus of the hypothalamus: Sex steroid hormone effects. Brain Res. 1996, 712, 335–339. [Google Scholar] [CrossRef]
- Palkovits, M.; Brownstein, M.J. Maps and Guide to Microdissection of the Rat Brain; Elsevier: New York, NY, USA, 1988. [Google Scholar]
- Neurotransmitter Receptors and Regulators PCR Array. Available online: http://www.sabiosciences.com/rt_pcr_product/HTML/PARN-060A.html (accessed on 24 February 2014).
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mann, P.E. Gene Expression Profiling during Pregnancy in Rat Brain Tissue. Brain Sci. 2014, 4, 125-135. https://doi.org/10.3390/brainsci4010125
Mann PE. Gene Expression Profiling during Pregnancy in Rat Brain Tissue. Brain Sciences. 2014; 4(1):125-135. https://doi.org/10.3390/brainsci4010125
Chicago/Turabian StyleMann, Phyllis E. 2014. "Gene Expression Profiling during Pregnancy in Rat Brain Tissue" Brain Sciences 4, no. 1: 125-135. https://doi.org/10.3390/brainsci4010125




