The Construction of an Engineered Bacterial Strain and Its Application in Accumulating Mercury from Wastewater
Abstract
:1. Introduction
2. Materials and Methods
2.1. Strains and Chemicals
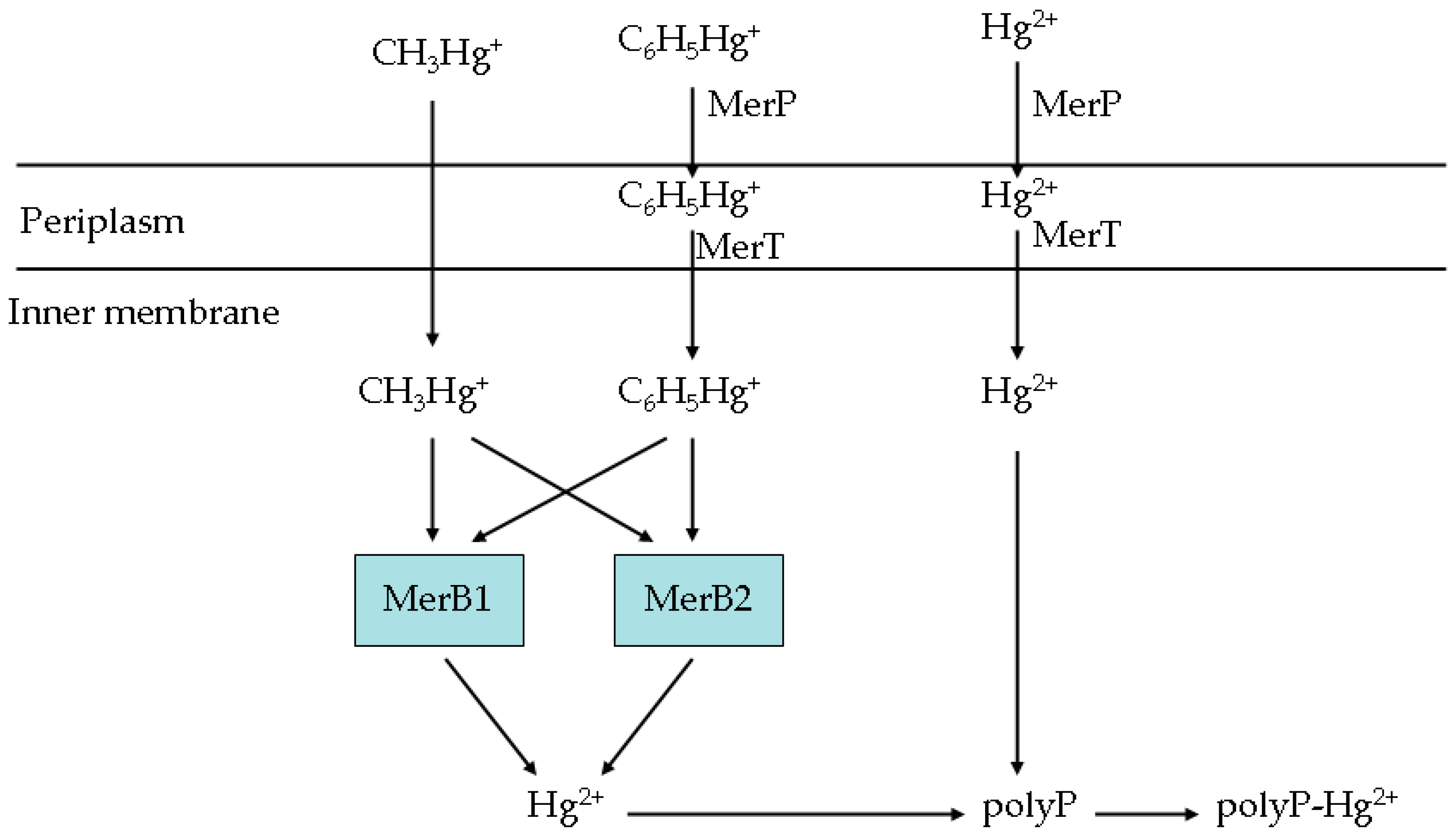
2.2. Constructing the Engineered Bacterial Strains
2.3. Real-Time Reverse-Transcription PCR
2.4. Measuring Bacterial Hg Tolerance
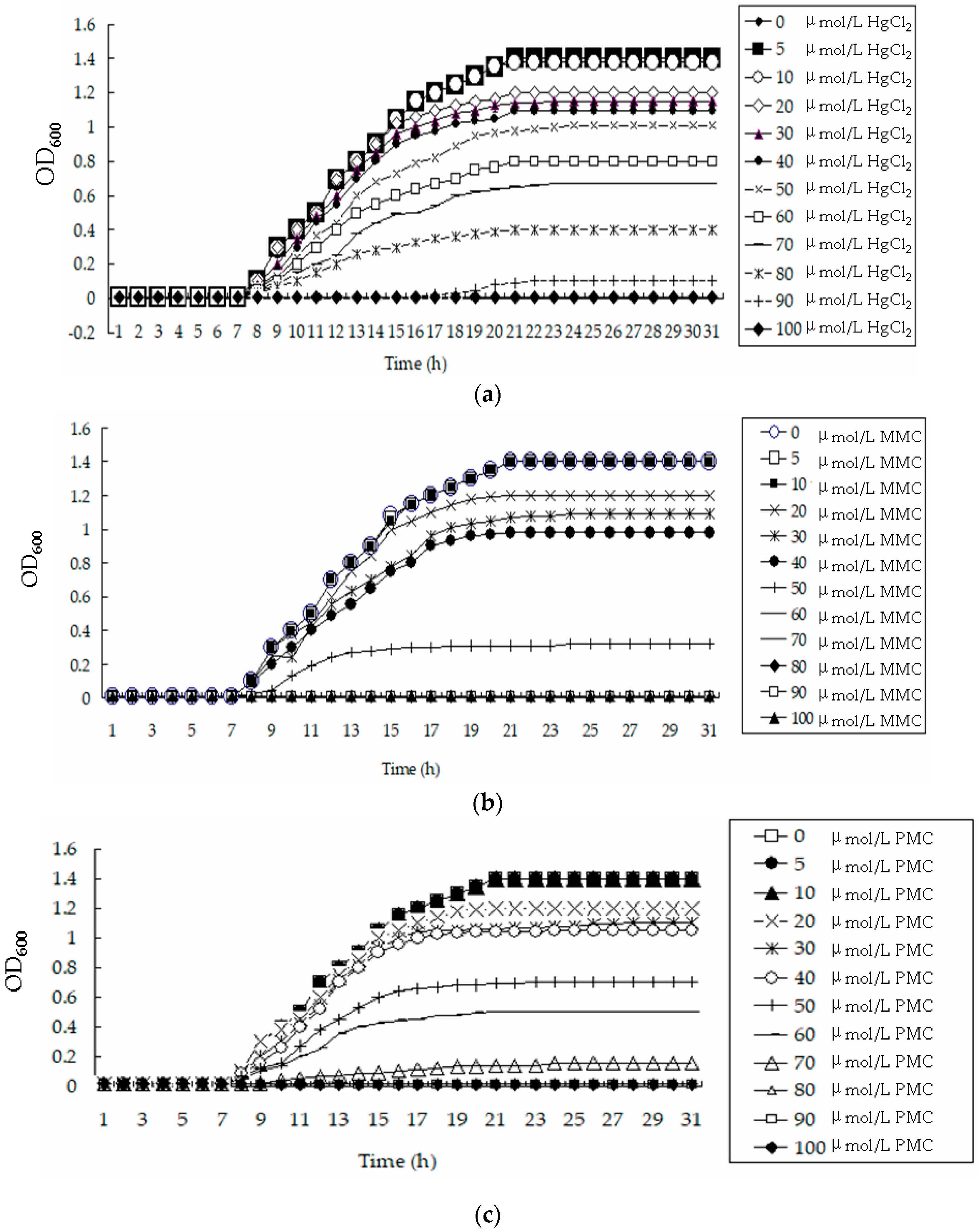
2.5. Measuring Minimum Inhibitory Concentration (MIC) Values
2.6. Hg Content Determination
2.7. Hg Content Determination Inside Bacterial Cells
2.8. Treating Wastewater Using the Engineered Bacteria
3. Results and Discussion
3.1. Construction of Engineered Bacterial Strain BL21-7
3.2. Transcription of the Artificial Operon P16S-g10-merT-merP-merB1-merB2-ppk-rpsT
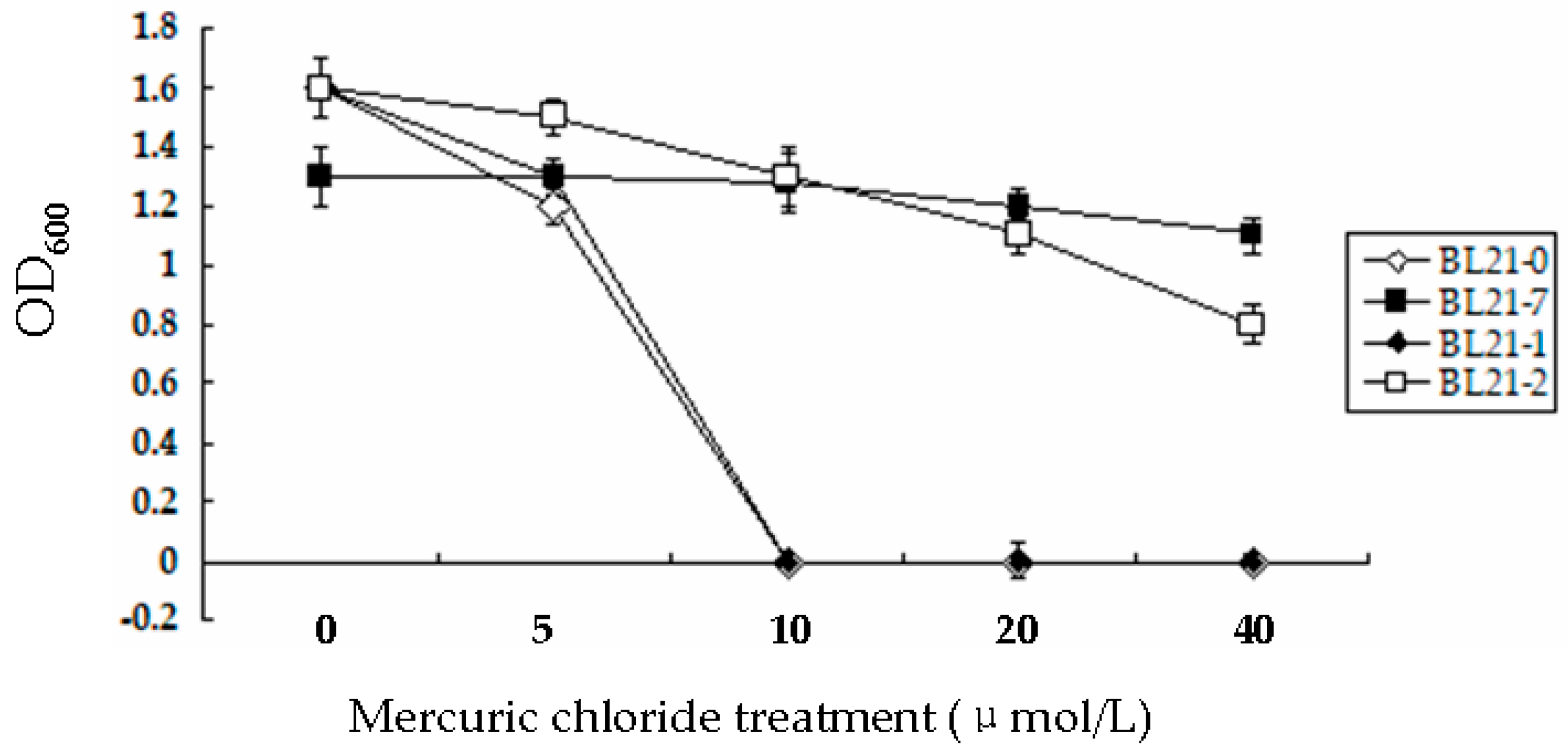
3.3. Bacterial Strain Tolerance to Hg
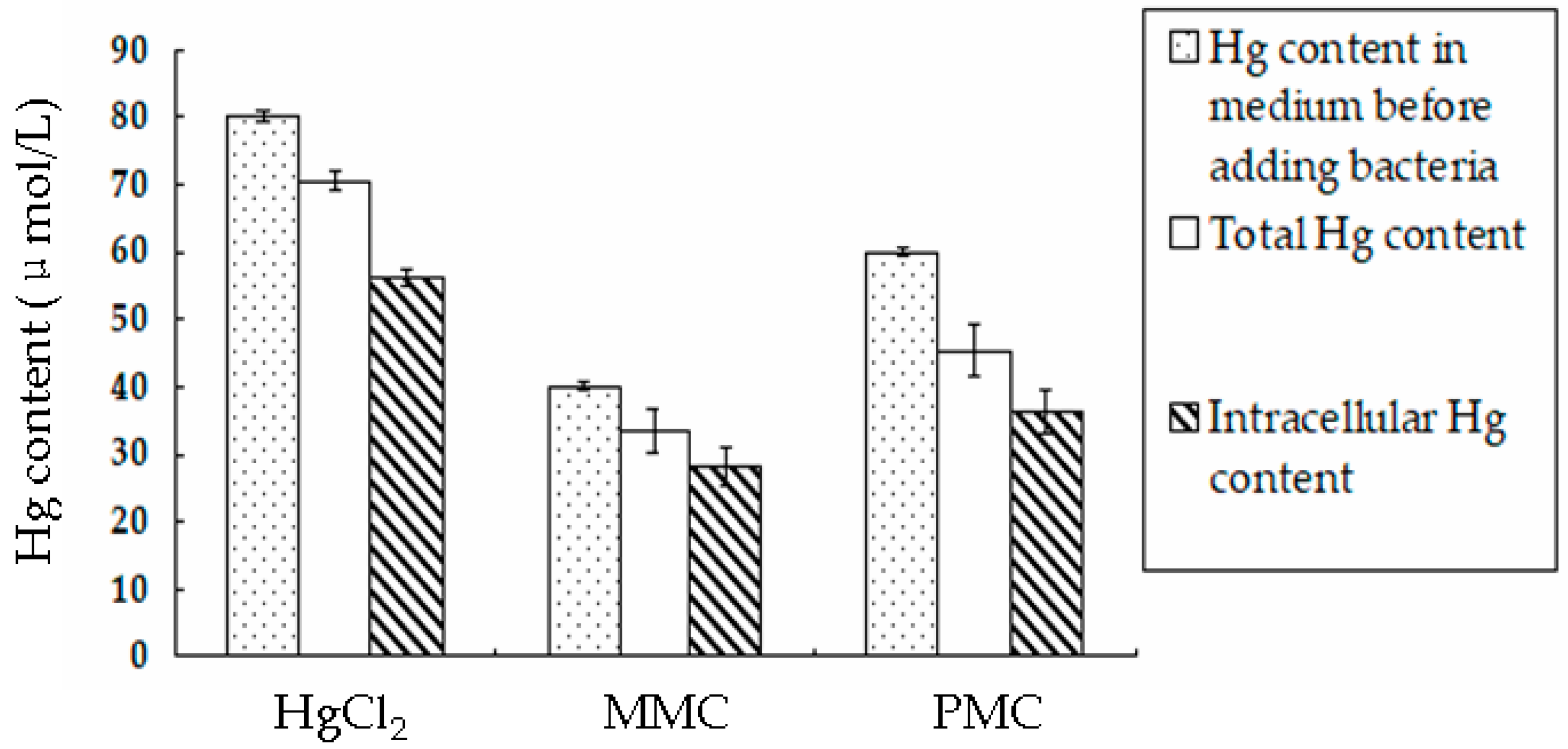
3.4. Accumulation of Hg Derivatives by BL21-7 Cultured in LB Medium
3.5. Effects of Other Metals on the Accumulation of Hg by BL21-7
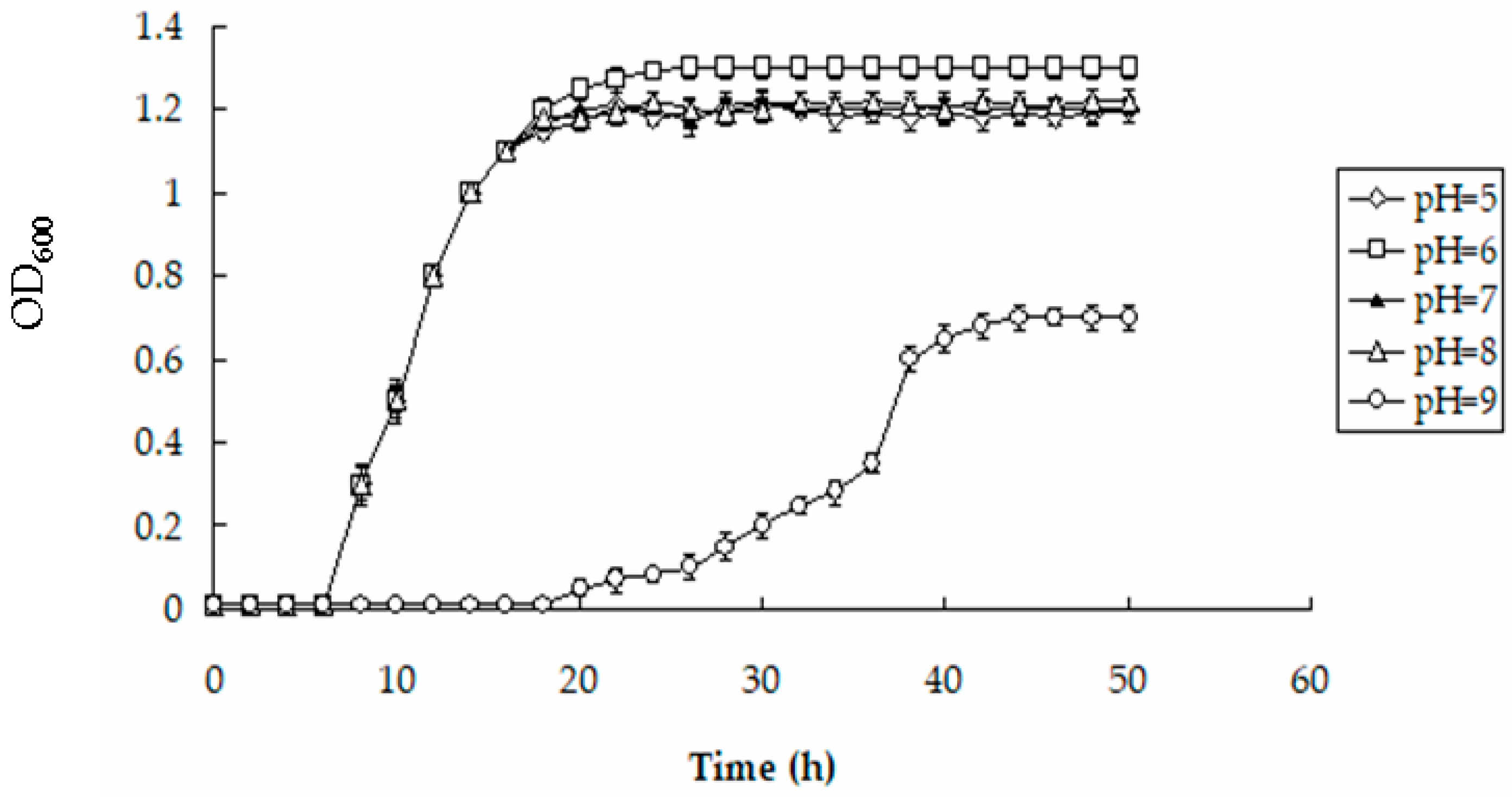
3.6. Effects of pH and Temperature on the Accumulation of Hg by BL21-7
3.7. Hg in Wastewater Can Be Removed by BL21-7
3.8. The Roles of the Genes in the Artificial Operon
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Jan, A.T.; Murtaza, I.; Ali, A.; Mohd, Q.; Haq, R. Mercury pollution: An emerging problem and potential bacterial remediation strategies. World J. Microbiol. Biotechnol. 2009, 25, 1529–1537. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency (US EPA). EPA’s Roadmap for Mercury. July 2006. Available online: http://www.epa.gov/mercury/roadmap.htm (accessed on 26 October 2007).
- U.S. Environmental Protection Agency (US EPA) and Environment Canada. Update: Binational Toxics Strategy—Mercury Sources and Regulations. 1999. Available online: http://www.epa.gov/bns/mercury/stephg.html (accessed on 26 October 2007).
- U.S. Environmental Protection Agency (US EPA). Great Lakes Pollution Prevention and Toxics Strategy. Background Information on Mercury Sources and Regulations. 2005. Available online: http://www.epa.gov/grtlakes/bnsdocs/mercsrce/merc srce.html (accessed on 26 October 2007).
- Cachada, A.M.; Rodrigues, S.; Mieiro, C.; Ferreira da Silva, E.; Pereira, E.; Duarte, A.C. Controlling factors and environmental implications of mercury contamination in urban and agricultural soils under a long-term influence of a chlor-alkali plant in the North–West Portugal. Environ. Geol. 2009, 57, 91–98. [Google Scholar] [CrossRef]
- Ruiz, O.N.; Alvarez, D.; Gonzalez-Ruiz, G.; Torres, C. Characterization of mercury bioremediation by transgenic bacteria expressing metallothionein and polyphosphate kinase. BMC Biotechnol. 2011, 11, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Kiyono, M.; Omura, T.; Fujimori, H.; Pan-Hou, H. Lack of involvement of merT and merP in methylmercury transport in mercury resistant Pseudomonas K-62. FEMS Microbiol. Lett. 1995, 128, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Rojas, L.A.; Yanez, C.; Gonzalez, M.; Lobos, S.; Smalla, K.; Seeger, M. Characterization of the metabolically modified heavy metal-resistant Cupriavidus metallidurans strain MSR33 generated for mercury bioremediation. PLoS ONE 2011, 6, e17555. [Google Scholar] [CrossRef] [PubMed]
- Kiyono, M.; Omura, H.; Omura, T.; Murata, S.; Pan-Hou, H. Removal of inorganic and organic mercurials by immobilized bacteria having mer-ppk fusion plasmids. Appl. Microbiol. Biotechnol. 2003, 62, 274–278. [Google Scholar] [CrossRef] [PubMed]
- Francois, F.; Lombard, C.; Guigner, J.; Soreau, P.; Brian-Jaisson, F.; Martino, G.; Vandervennet, M.; Garcia, D.; Molinier, A.; Pignol, D.; et al. Isolation and characterization of environmental bacteria capable of extracellular biosorption of mercury. Appl. Environ. Microbiol. 2011, 78, 1097–1106. [Google Scholar] [CrossRef] [PubMed]
- Olins, P.O.; Rangwala, S.H. A novel sequence element derived from bacteriophage T7 mRNA acts as an enhancer of translation of the lacZ gene in Escherichia coli. J. Biol. Chem. 1989, 264, 16973–16976. [Google Scholar] [PubMed]
- Dennis, P.P.; Ehrenberg, M.; Fange, D.; Bremer, H. Varying rate of RNA chain elongation during rrn transcription in Escherichia coli. J. Bacteriol. 2009, 191, 3740–3746. [Google Scholar] [CrossRef] [PubMed]
- Newell, C.A.; Birch-Machin, I.; Hibberd, J.M.; Gray, J.C. Expression of green fluorescent protein from bacterial and plastid promoters in tobacco chloroplasts. Transgenic Res. 2003, 12, 631–634. [Google Scholar] [CrossRef] [PubMed]
- Kuroda, H.; Maliga, P. Complementarity of the 16S rRNA penultimate stem with sequences downstream of the AUG destabilizes the plastid mRNAs. Nucleic Acids Res. 2001, 29, 970–975. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shiina, T.; Allison, L.; Maliga, P. RbcL transcript levels in tobacco plastids are independent of light: Reduced dark transcription rate is compensated by increased mRNA stability. Plant Cell 1998, 10, 1713–1722. [Google Scholar] [CrossRef] [PubMed]
- Silver, S.; Phung, L.T. Bacterial heavy metal resistance: New surprises. Annu. Rev. Microbiol. 1996, 50, 753–789. [Google Scholar] [CrossRef] [PubMed]
- Kiyono, M.; Pan-Hou, H. Genetic engineering of bacteria for environmental remediation of mercury. J. Health Sci. 2006, 52, 199–204. [Google Scholar] [CrossRef]
- Pan-Hou, H.; Kiyono, M.; Kawase, T.; Omura, T.; Endo, G. Evaluation of ppk-specified polyphosphate as a mercury remedial tool. Biol. Pharm. Bull. 2001, 24, 1423–1426. [Google Scholar] [CrossRef] [PubMed]
- Kornberg, A. Inorganic polyphosphate: Toward making a forgotten polymer unforgettable. J. Bacteriol. 1995, 177, 491–496. [Google Scholar] [CrossRef] [PubMed]
- Pan-Hou, H.; Kuyono, M.; Omura, H.; Omura, T.; Endo, G. Polyphosphate produced in recombinant Escherichia coli confers mercury resistance. FEMS Microbiol. Lett. 2002, 207, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Sone, Y.; Nakamura, R.; Pan-Hou, H.; Itoh, T.; Kiyono, M. Role of MerC, MerE, MerF, MerT, and/or MerP in resistance to mercurials and the transport of mercurials in Escherichia coli. Biol. Pharm. Bull. 2013, 36, 1835–1841. [Google Scholar] [CrossRef] [PubMed]
- Misra, T.K. Bacterial resistances to inorganic mercury salts and organomercurials. Plasmid 1992, 27, 1, 4–16. [Google Scholar] [CrossRef]
- Tezuka, T.; Tonomura, K. Purification and properties of an enzyme catalyzing the splitting of carbon-mercury linkages from mercury-resistant Pseudomonas K-62 strain. J. Biochem. 1976, 80, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Tezuka, T.; Tonomura, K. Purification and properties of a second enzyme catalyzing the splitting of carbon-mercury linkages from mercury-resistant Pseudomonas K-62. J. Bacteriol. 1978, 135, 138–143. [Google Scholar] [PubMed]
- Tonomura, K.; Maeda, K.; Futai, F.; Nakagami, T.; Yamada, M. Stimulative vaporization of phenylmercuric acetate by mercury-resistant bacteria. Nature 1968, 217, 644–646. [Google Scholar] [CrossRef] [PubMed]
- Studier, F.W.; Daegelen, P.; Lenski, R.E.; Maslov, S.; Kim, J.F. Understanding the differences between genome sequences of Escherichia coli B strains REL606 and BL21(DE3) and comparision of the E. coli B and K-12 genomes. J. Mol. Biol. 2009, 394, 653–680. [Google Scholar] [CrossRef] [PubMed]
- Barkay, T.; Wagner-Dobler, I. Microbial transformations of mercury: Potentials, challenges and achievements in controlling mercury toxicity in the environment. Adv. Appl. Microbiol. 2005, 57, 1–52. [Google Scholar] [PubMed]





| Primer | Sequence | Plasmid |
|---|---|---|
| PPPKF | 5′-ATAAGAATGCGGCCGCATTTACCACGTCCTGTGATT-3′; Not I restriction site is underlined. | p2 |
| PPPKR | 5′-CCGATCCGCTCGAGGGTTAATCGGGTTGCTCGAG-3′; Xho I restriction site is underlined. | p2 |
| P1F | 5′-CGACATATGGCTCCCCCGCCGTCGTTCAATGAGAATGGATAAGAGGCTCGTGGGATTGACGTGAGGGGGCAGGGATGGCTATATTTCTGGGAGCGAACTCCGGGCGAATACGAAGCGCTTGGATACAGTTGTAGGGAGGGATTTATCTTTTAACTTTAAGAAGGAGTGGCCAAGCGCTATTCGATCGACATATGGGAC-3′ Nde I and Bal I restriction sites are underlined. | p3 |
| P1R | 5′-GTCCCATATGTCGATCGAATAGCGCTTGGCCACTCCTTCTTAAAGTTAAAAGATAAATCCCTCCCTACAACTGTATCCAAGCGCTTCGTATTCGCCCGGAGTTCGCTCCCAGAAATATAGCCATCCCTGCCCCCTCACGTCAATCCCACGAGCCTCTTATCCATTCTCATTGAACGACGGCGGGGGAGCCATATGTCG-3′ Nde I and Bal I restriction sites are underlined. | p3 |
| P2F | 5′-CGGTGGCCAGTATGTCTGAACCACAAAAACG-3′; Bal I restriction site is underlined. | p4 |
| P2R | 5′-CGCGAATTCTTATCATTTATCGGCATAGGT-3′; EcoR I restriction site is underlined. | p4 |
| P3F | 5′-CCGGAATTCCGTGTTCTATAACGACAAAA-3′; EcoR I restriction site is underlined. | p5 |
| P3R | 5’-CCGGAATTCAAGAAAATATCGAAGAAAAAT-3′; EcoR I restriction site is underlined. | p5 |
| Bacterial Strains | Plasmids Harboring Artificial Operons |
|---|---|
| BL21-0 | pET28a |
| BL21-1 | pET28a (g10-ppk-rpsT) |
| BL21-2 | pET28a (P16S-g10-ppk-rpsT) |
| BL21-T | pET28a (P16S-g10-merT-rpsT) |
| BL21-P | pET28a (P16S-g10-merP-rpsT) |
| BL21-B1 | pET28a (P16S-g10-merB1-rpsT) |
| BL21-B2 | pET28a (P16S-g10-merB2-rpsT) |
| BL21-TP | pET28a (P16S-g10-merT-merP-rpsT) |
| BL21-B1B2 | pET28a (P16S-g10-merB1-merB2-rpsT) |
| BL21-TPK | pET28a (P16S-g10-merT-merP-ppk-rpsT) |
| BL21-B1B2K | pET28a (P16S-g10-merB1-merB2-ppk-rpsT) |
| BL21-TPB1B2 | pET28a (P16S-g10-merT-merP-merB1-merB2-rpsT) |
| BL21-7 | pET28a (P16S-g10-merT-merP-merB1-merB2-ppk-rpsT) |
| Metals | HgCl2 Removed (µmol/L) | MMC Removed (µmol/L) | PMC Removed (µmol/L) |
|---|---|---|---|
| None | 8.80 ± 0.16 | 5.47 ± 0.28 | 7.80 ± 0.17 |
| Cd2+ | 8.66 ± 0.15 | 5.53 ± 0.23 | 7.76 ± 0.15 |
| Pb2+ | 8.85 ± 0.09 | 5.41 ± 0.25 | 7.73 ± 0.18 |
| Cu2+ | 8.95 ± 0.12 | 5.44 ± 0.26 | 7.83 ± 0.17 |
| Cd2+ + Pb2+ + Cu2+ | 8.98 ± 0.19 | 5.59 ± 0.27 | 7.85 ± 0.19 |
| Strains | HgCl2 (µmol/L) | MMC (µmol/L) | PMC (µmol/L) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 h | 5 h | 10 h | 15 h | 20 h | 0 h | 5 h | 10 h | 15 h | 20 h | 0 h | 5 h | 10 h | 15 h | 20 h | |
| BL21-T | 20.0 | 17.1 | 16.3 | 15.2 | 15.0 | 20.0 | 19.8 | 19.5 | 19.3 | 19.0 | 20.0 | 19.9 | 19.9 | 19.8 | 19.6 |
| BL21-B1 | 20.0 | 19.8 | 19.8 | 19.8 | 19.4 | 20.0 | 19.8 | 19.2 | 18.5 | 18.1 | 20.0 | 18.2 | 17.5 | 16.3 | 15.5 |
| BL21-B2 | 20.0 | 19.8 | 19.8 | 19.7 | 19.6 | 20.0 | 19.3 | 18.9 | 18.8 | 18.8 | 20.0 | 18.2 | 17.3 | 16.5 | 16.2 |
| BL21-B1B2 | 20.0 | 19.9 | 19.8 | 19.5 | 19.2 | 20.0 | 19.5 | 18.5 | 18.0 | 17.5 | 20.0 | 17.4 | 16.5 | 15.3 | 15.1 |
| BL21-TP | 20.0 | 14.8 | 13.2 | 11.5 | 11.1 | 20.0 | 19.9 | 19.7 | 19.5 | 19.5 | 20.0 | 19.9 | 19.9 | 19.8 | 19.5 |
| BL21-P | 20.0 | 19.6 | 18.5 | 18.3 | 18.0 | 20.0 | 19.9 | 19.8 | 19.8 | 19.5 | 20.0 | 19.8 | 19.7 | 19.5 | 19.5 |
| BL21-0 | 20.0 | 19.9 | 19.8 | 19.7 | 19.2 | 20.0 | 19.8 | 19.9 | 19.6 | 19.6 | 20.0 | 19.8 | 19.7 | 19.5 | 19.3 |
| BL21-1 | 20.0 | 19.8 | 19.7 | 19.9 | 19.5 | 20.0 | 19.9 | 19.5 | 19.4 | 19.4 | 20.0 | 19.9 | 19.8 | 19.5 | 19.2 |
| BL21-2 | 20.0 | 12.5 | 11.2 | 10.0 | 9.5 | 20.0 | 19.8 | 19.9 | 19.6 | 19.6 | 20.0 | 19.8 | 19.8 | 19.6 | 19.5 |
| BL21-B1B2K | 20.0 | 13.1 | 12.5 | 11.3 | 10.0 | 20.0 | 16.1 | 15.3 | 14.2 | 14.1 | 20.0 | 10.2 | 8.5 | 6.8 | 6.1 |
| BL21-TPK | 20.0 | 7.9 | 4.5 | 3.8 | 2.2 | 20.0 | 19.8 | 19.6 | 19.5 | 19.3 | 20.0 | 19.9 | 19.8 | 19.8 | 19.5 |
| BL21-7 | 20.0 | 6.8 | 3.7 | 2.5 | 1.1 | 20.0 | 6.7 | 3.2 | 1.4 | 1.1 | 20.0 | 5.2 | 2.3 | 1.5 | 1.0 |
| BL21-TPB1B2 | 20.0 | 14.6 | 13.2 | 12.3 | 12.2 | 20.0 | 19.5 | 19.2 | 18.3 | 17.2 | 20.0 | 17.5 | 17.1 | 15.2 | 15.1 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, S.; Wu, Z.; Sun, W.; Shu, H. The Construction of an Engineered Bacterial Strain and Its Application in Accumulating Mercury from Wastewater. Appl. Sci. 2018, 8, 1572. https://doi.org/10.3390/app8091572
Chang S, Wu Z, Sun W, Shu H. The Construction of an Engineered Bacterial Strain and Its Application in Accumulating Mercury from Wastewater. Applied Sciences. 2018; 8(9):1572. https://doi.org/10.3390/app8091572
Chicago/Turabian StyleChang, Shenghe, Zhengjing Wu, Wei Sun, and Haiyan Shu. 2018. "The Construction of an Engineered Bacterial Strain and Its Application in Accumulating Mercury from Wastewater" Applied Sciences 8, no. 9: 1572. https://doi.org/10.3390/app8091572




