Evolution of Microbial “Streamer” Growths in an Acidic, Metal-Contaminated Stream Draining an Abandoned Underground Copper Mine
Abstract
:1. Introduction
2. Results
2.1. Physico-Chemistry of Dyffryn Adda AMD
| Analyte | Mean value (standard error) |
|---|---|
| pH | 2.53 (0.04) |
| Redox potential Eh (mV) | 669 (2.6) |
| Conductivity (µS cm−1) | 2506 (66) |
| Temperature (°C) | 11.3 (0.1) |
| Oxygen (%) | 12.7 (0.4) |
| Sulfate-S | 800 (13) |
| Fe2+ | 378 (14) |
| Fetotal | 563 (11) |
| Zn | 67 (1.6) |
| Cu | 49 (2.2) |
| Mn | 15.8(0.7) |
| Al | 2.15(0.16) |
| DOC* | 4.50(0.29) |


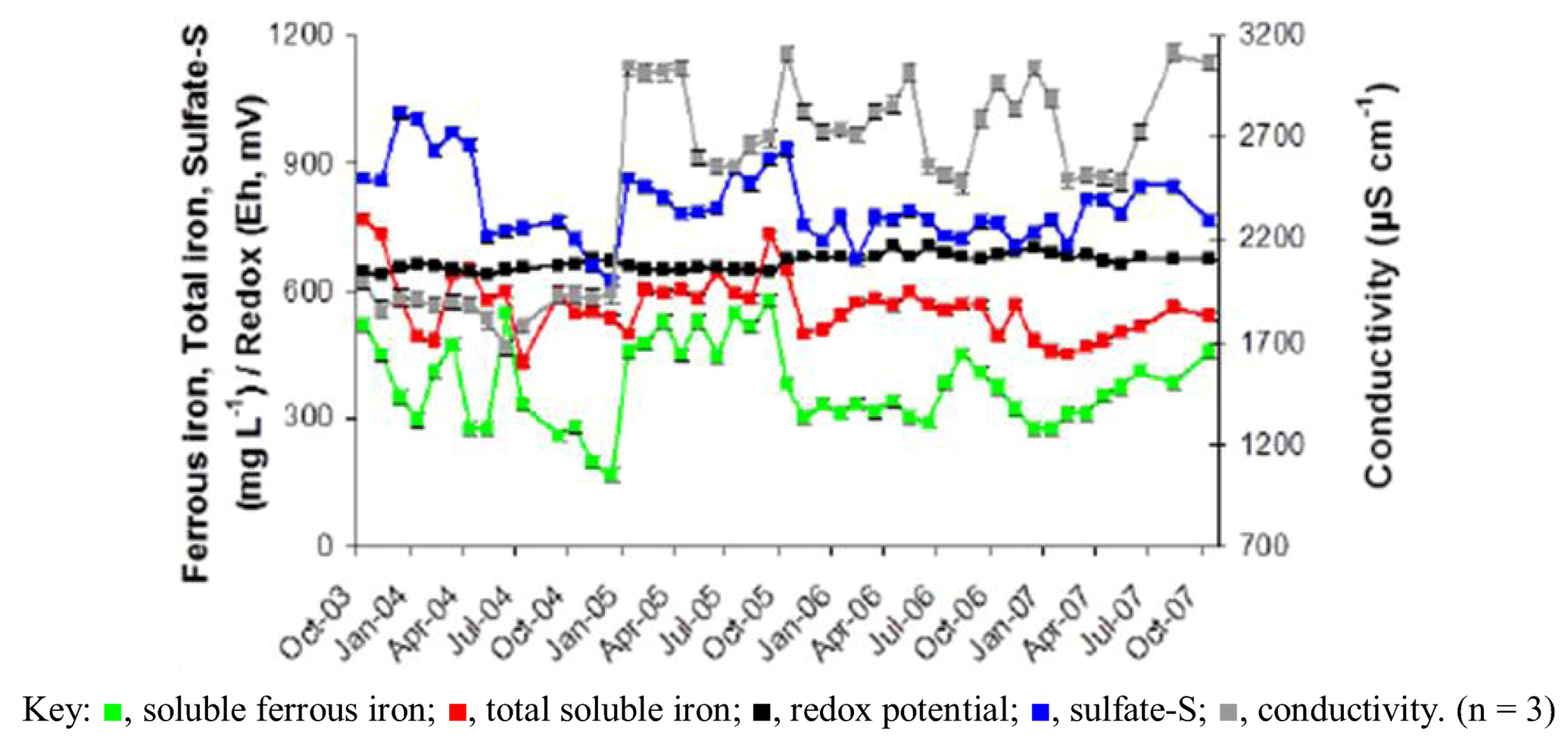
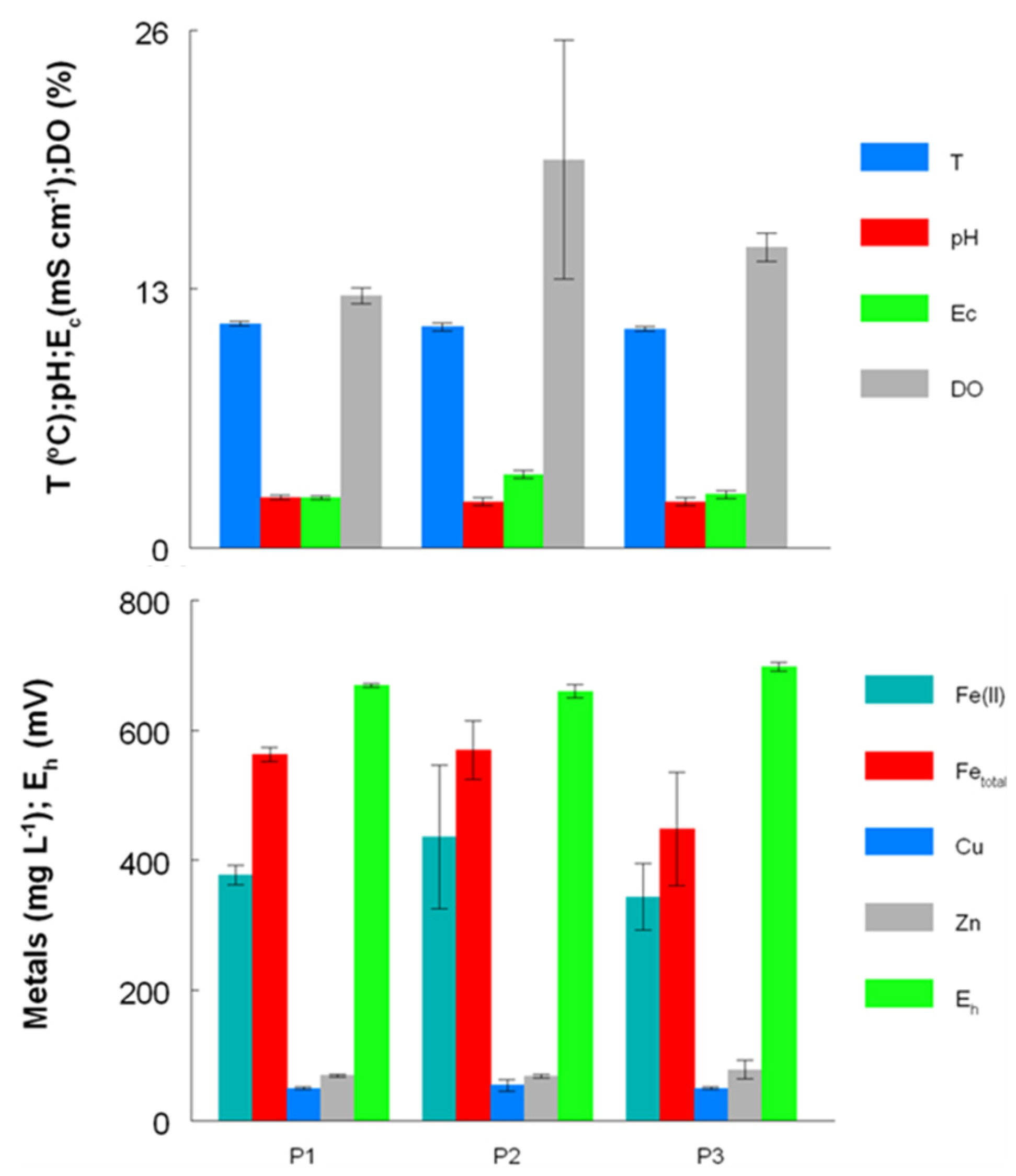
2.2. Growth of Acid Streamers within the Dyffryn Adda
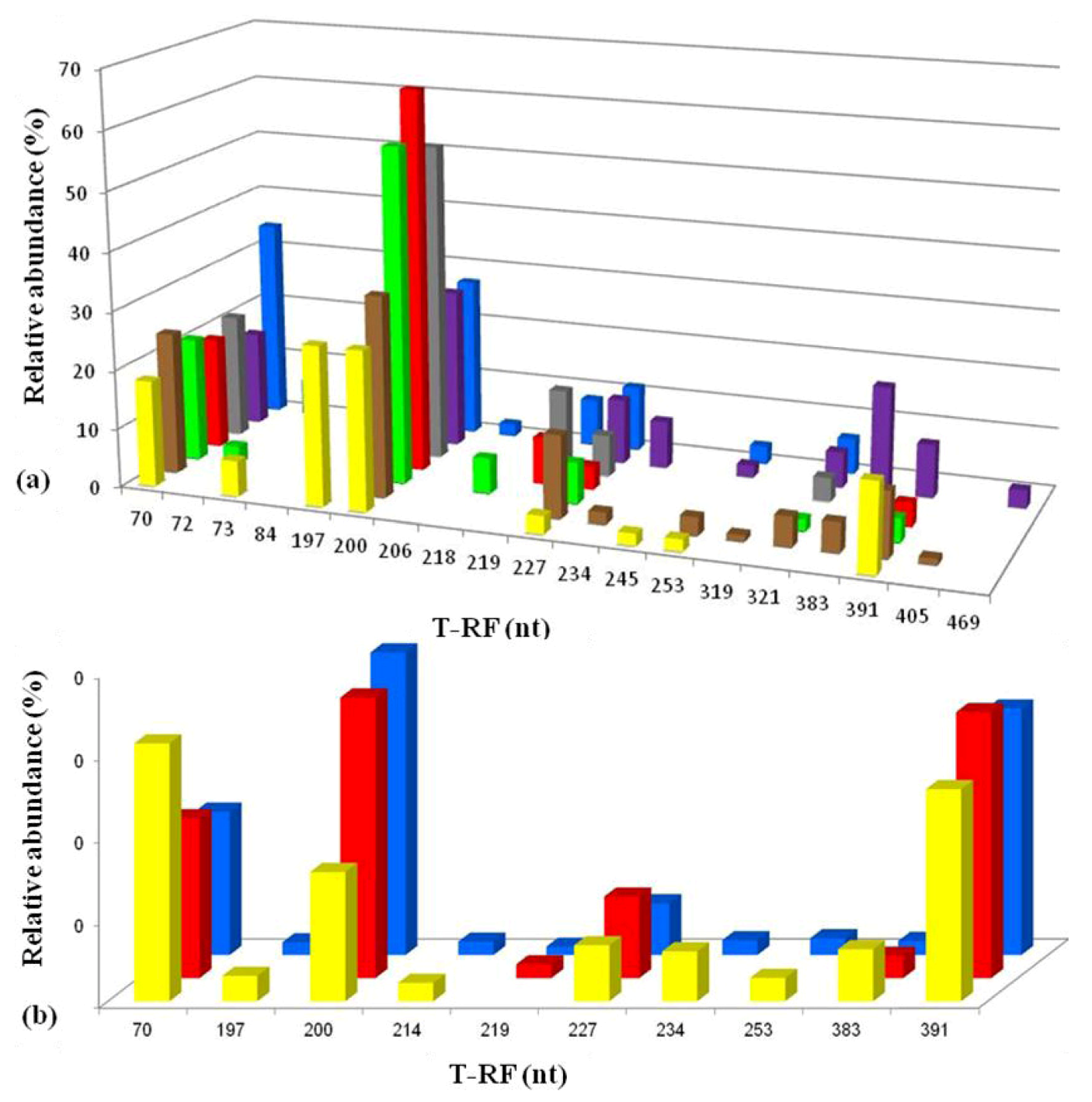
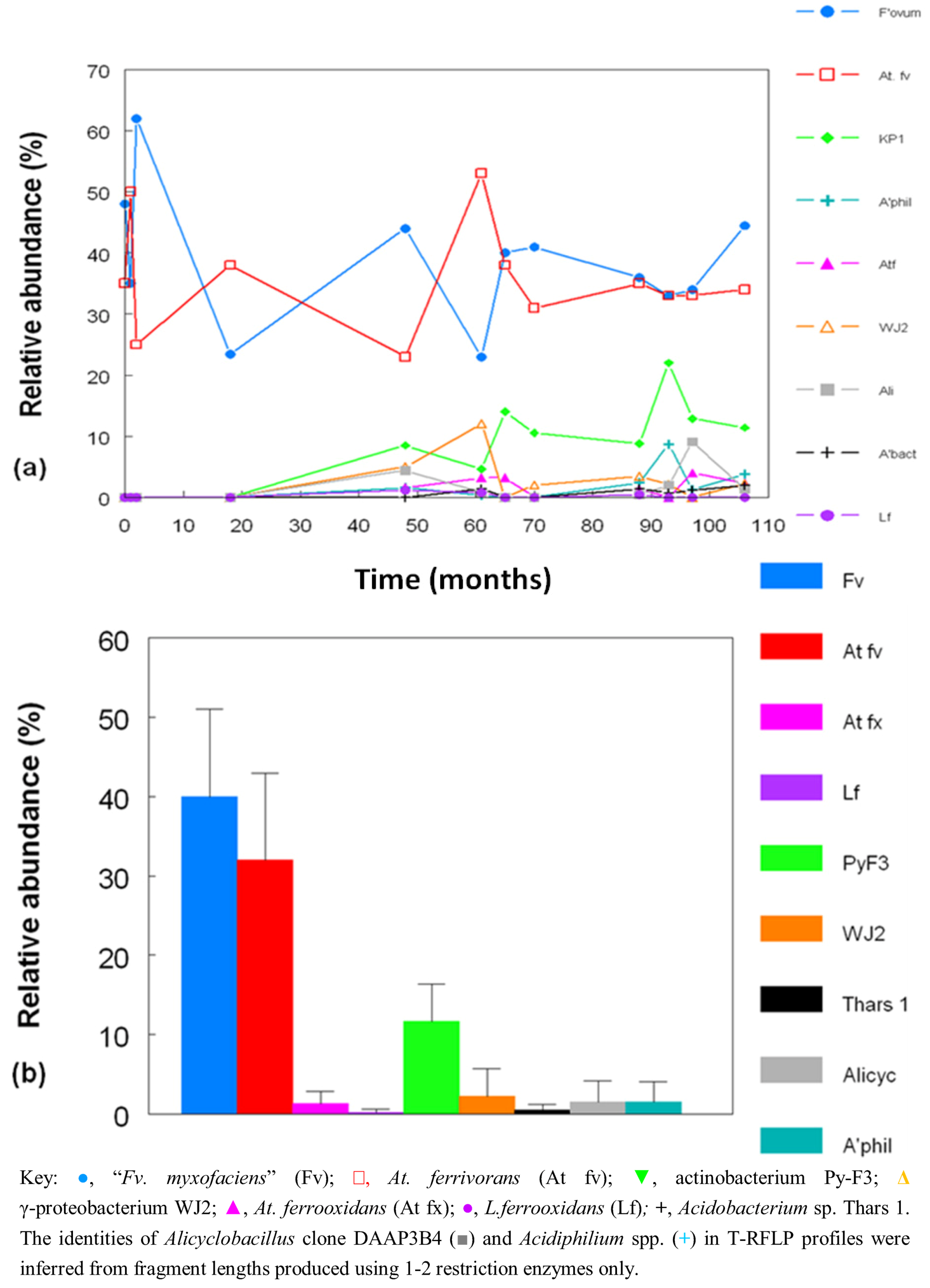
2.3. Molecular Analysis of Acid Streamer Growths: Bacteria


2.4. Bacterial Isolates and Cloned Genes

2.5. Molecular Analysis of Acid Streamer Growths: Archaea
| Isolate/clone designation (GenBank Accession No.) | Closest relative (GenBank Accession No.) | Identity (%) (16S rRNA gene) | Reference |
|---|---|---|---|
| Bacterial isolates | |||
| P3G (“Ferrovum myxofaciens”T) (HM044161) | “Ferrovum myxofaciens” PSTR (EF133508) | 100 | [10] |
| Py-H1 (KC208493) | Acidocella sp. strain M21(AY765998) | 100 | [10] |
| Acidocella aluminiidurans (AB362219) | 97.4 | [11] | |
| Py-H3 (KC208494) | Acidiphilium sp. CCP3(AY766000) | 99.9 | [10] |
| Acidiphilium sp. NO-17 (AF376026) | 99.5 | [12] | |
| Acidiphilium rubrumT (NR_025854) | 95.8 | [13] | |
| Py-F1 (KC208495) | Acidithiobacillus ferrivoransT (AF376020) | 100 | [14] |
| Py-F2 (KC208496) | Ferrimicrobium acidiphilumT (NR_041798) | 99.5 | [15] |
| Py-F3 (“Acidithrix ferrooxidans”) (KC208497) | Heterotrophic iron-oxidizing bacterium KP1(AY765991) | 100 | [10] |
| Ferrimicrobium acidiphilumT (NR_041798) | 92.4 | [15] | |
| Bacterial clones | |||
| DAAP3B4 (KC208499) | Alicyclobacillus K23_bac (EF464642) | 99.3 | [16] |
| Alicyclobacillus ferrooxydansT (NR_044413) | 90.0 | [17] | |
| PMC25 (KC208498) | Actinobacterium U2V-bac_a5 (JN982098) | 95.9 | [18] |
| Ferrimicrobium acidiphilumT (NR_041798) | 94.8 | [15] | |
| Isolate Py-F3 | 91.3 | (this study) | |
| Archaeal clones | |||
| DAAP3A2 (KC208501) | Clone from sediment in an acidic pit lake (FJ228391) | 99.1 | (GenBank entry) |
| Methanomassiliicoccu luminyensisT (HQ896499) | 83.5 | [19] | |
| DAAP3A1 (KC208500) | Clone from sediment in an acidic pit lake (FJ228392) | 99.7 | (GenBank entry) |
| Methanomassiliicoccus luminyensisT (HQ896499) | 82.9 | [19] | |
| Clone DAAP3A3 | 77.1 | (this study) | |
| Clone DAAPA6 | 90.4 | (this study) | |
| DAAP3A3 (KC208502) | clone from coal-impacted forest wetland (AF523941) | 98.5 | [20] |
| Thermogymnomonas acidicolaT (NR_041513) | 90.6 | [21] | |
| Clone DAAP3A6 | 76.8 | (this study) | |
| DAAP3A6 (KC2084503) | Clone from AMD stream (HE653789) | 99.2 | [22] |
| Methanomassiliicoccus luminyensisT (HQ896499) | 80.6 | [19] | |
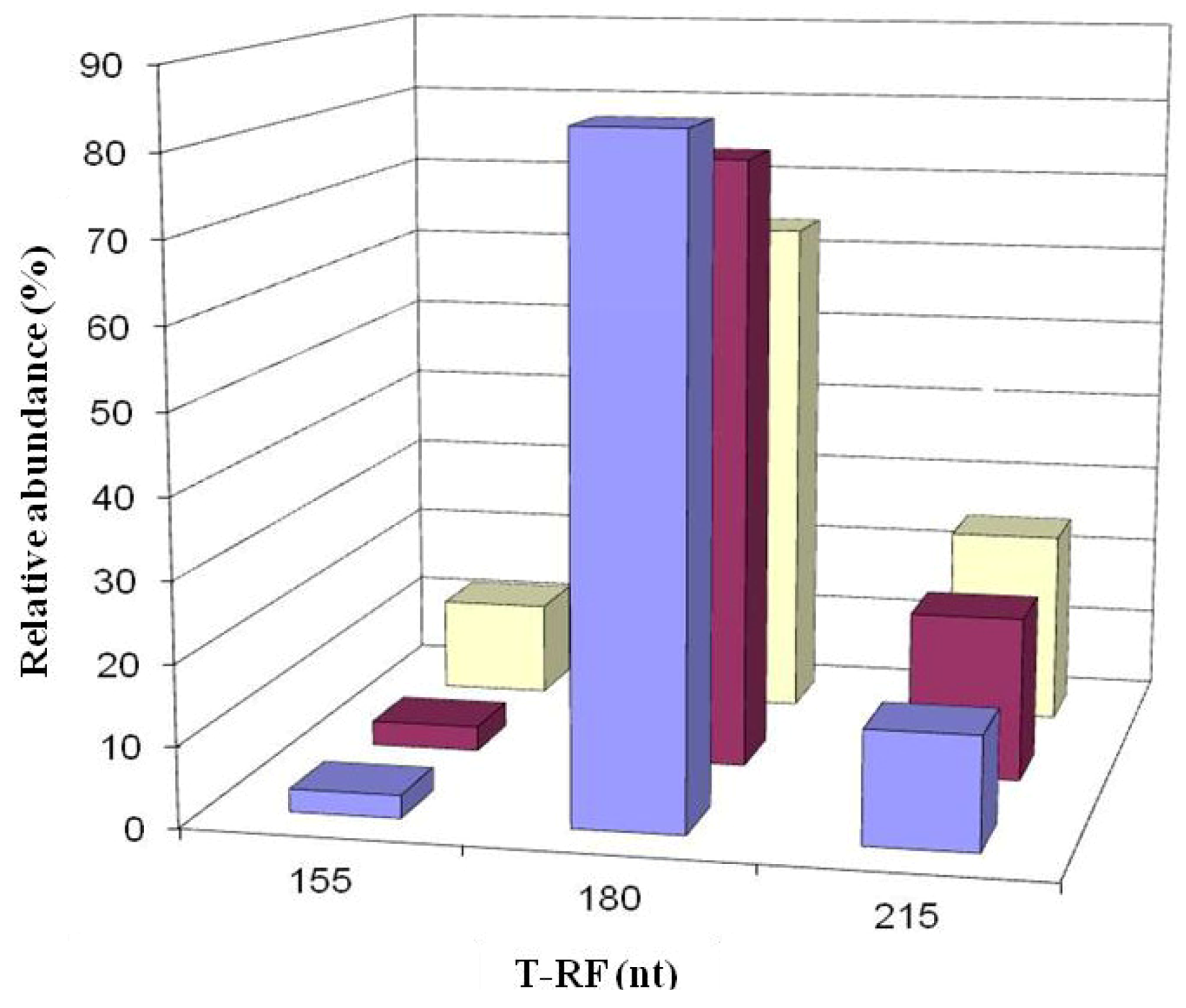
2.6. Comparison of Acid Streamer and Planktonic Phase Prokaryotic Communities in the Dyffryn Adda
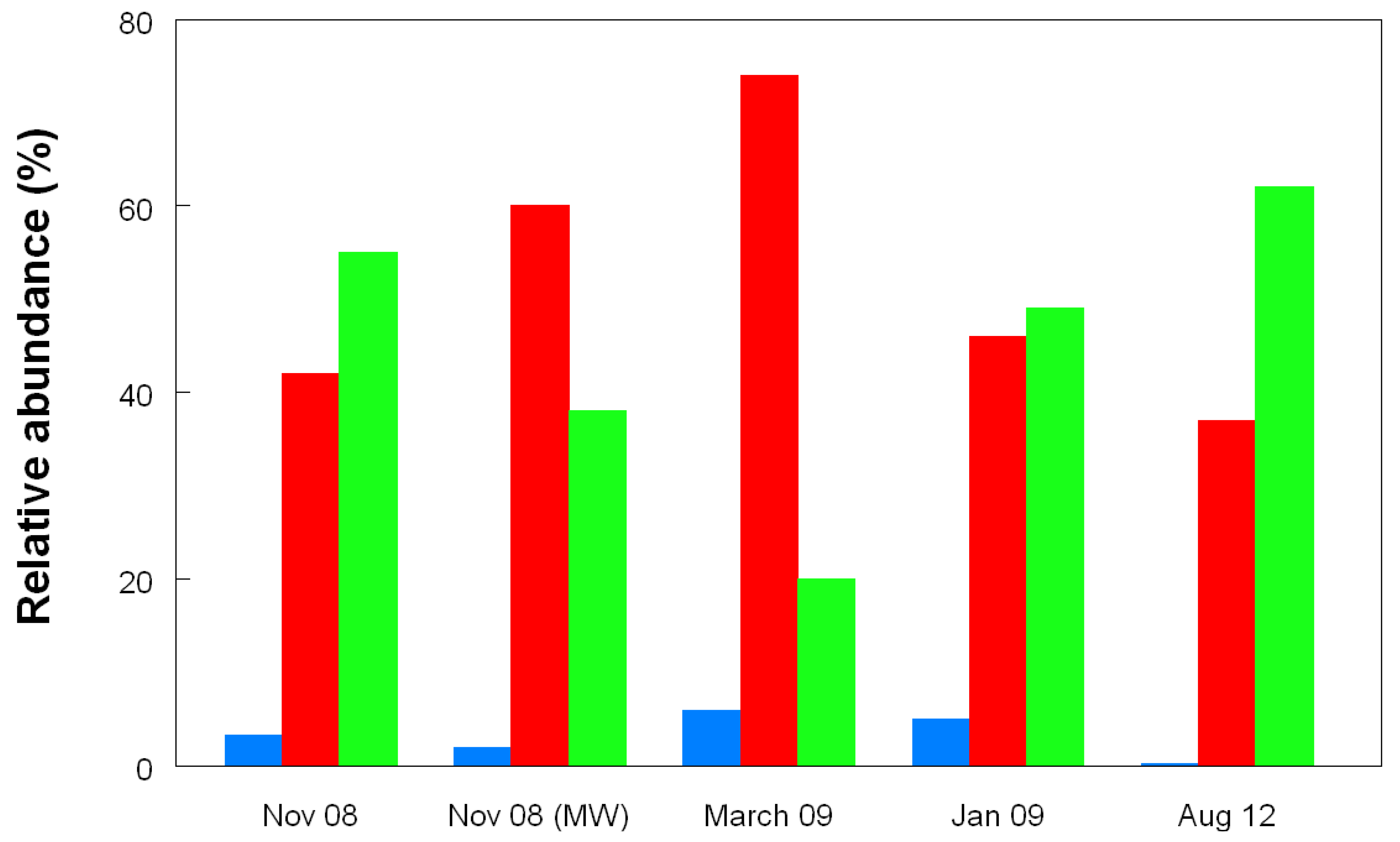
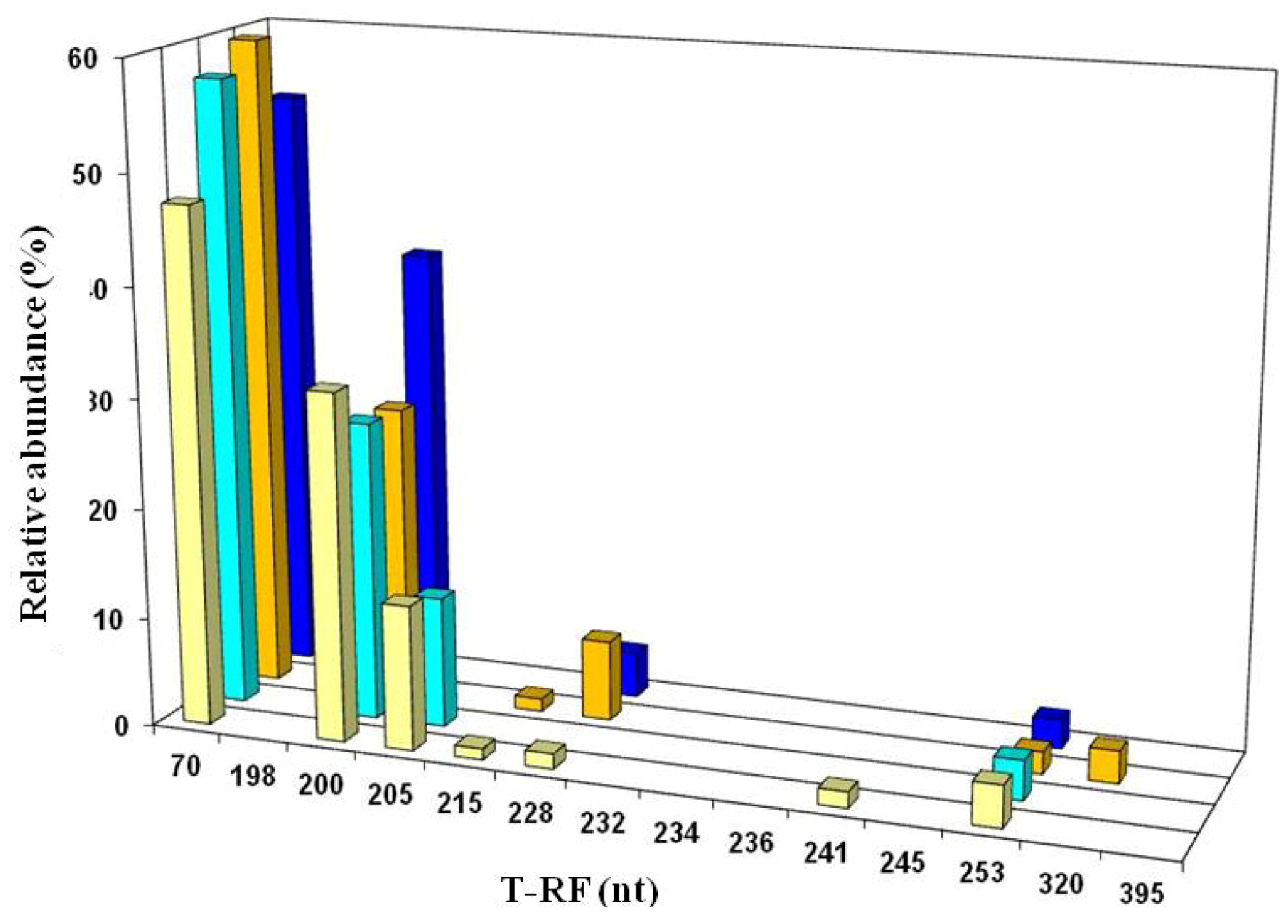
3. Discussion
4. Experimental Section
4.1. Site Description and Sampling Regime
4.2. Physico-Chemical Analyses
4.3. Molecular Analysis of Acid Streamer Growths
4.4. Molecular Analysis of Planktonic-Phase Bacteria
4.5. Clone Library Construction
4.6. Cultivation Analysis of Acid Streamer Bacteria
5. Conclusions
Acknowledgements
References
- Johnson, D.B. Extremophiles: acidic environments. In Encyclopaedia of Microbiology, 2nd Edition; Schaechter, M., Ed.; Elsevier: Oxford, UK, 2009; pp. 107–126. [Google Scholar]
- Johnson, D.B. Geomicrobiology of extremely acidic subsurface environments. FEMS Microbiol. Ecol. 2012, 81, 2–12. [Google Scholar] [CrossRef]
- Edwards, K.J.; Gihring, T.M.; Banfield, J.F. Seasonal variations in microbial populations and environmental conditions in an extreme acid mine drainage environment. Appl. Environ. Microbiol. 1999, 65, 3627–3632. [Google Scholar]
- Zeigler, S.; Ackermann, S.; Majzian, J.; Gescher, J. Matrix composition and community structure analysis of a novel bacterial leaching community. Environ. Microbiol. 2009, 11, 2329–2338. [Google Scholar] [CrossRef]
- Jones, D.S.; Albrecht, H.L.; Dawson, K.S.; Schaperdoth, I.; Freeman, K.H.; Pi, Y.; Pearson, A.; Macalady, J.L. Community genomic analysis of an extremely acidophilic sulfur-oxidizing biofilm. ISME J. 2012, 6, 158–170. [Google Scholar] [CrossRef]
- Bevins, R.E. A Mineralogy of Wales; Geological Series No. 16; National Museum of Wales Press: Cardiff, UK, 1994. [Google Scholar]
- Walton, K.C.; Johnson, D.B. Microbiological and chemical characteristics of an acidic stream draining a disused copper mine. Environ. Pollut. 1992, 76, 169–175. [Google Scholar] [CrossRef]
- Coupland, K.; Johnson, D.B. Geochemistry and microbiology of an impounded subterranean acidic water body at Mynydd Parys, Anglesey, Wales. Geobiol. 2004, 2, 77–86. [Google Scholar] [CrossRef]
- Valente, T.M.; Gomez, C.L. The role of two acidophilic algae as ecological indicators of acid mine drainage sites. J. Iberian Geol. 2007, 33, 283–294. [Google Scholar]
- Hallberg, K.B.; Coupland, K.; Kimura, S.; Johnson, D.B. Macroscopic “acid streamer” growths in acidic, metal-rich mine waters in north Wales consist of novel and remarkably simple bacterial communities. Appl. Environ. Microbiol. 2006, 72, 2022–2030. [Google Scholar] [CrossRef]
- Kimoto, K.; Aizawa, T.; Urai, M.; Bao Ve, N.; Suzuki, K.; Nakajima, M.; Sunairi, M. Acidocella. aluminiidurans sp. nov., an aluminium-tolerant bacterium isolated from Panicum. repens grown in a highly acidic swamp in actual acid sulfate soil area of Vietnam. Int. J. Syst. Evol. Microbiol. 2010, 60, 764–768. [Google Scholar] [CrossRef]
- Johnson, D.B.; Rolfe, S.; Hallberg, K.B.; Iversen, E. Isolation and phylogenetic characterisation of acidophilic microorganisms indigenous to acidic drainage waters at an abandoned Norwegian copper mine. Environ. Microbiol. 2001, 3, 630–637. [Google Scholar] [CrossRef]
- Kishimoto, N.; Kosako, Y.; Wakao, N.; Tano, T.; Hiraishi, A. Transfer of Acidiphilium facilis and Acidiphilium aminolytica to the genus Acidocella gen. nov., and emendation of the genus Acidiphilium. Syst. Appl. Microbiol. 1995, 18, 85–91. [Google Scholar] [CrossRef]
- Hallberg, K.B.; González-Toril, E.; Johnson, D.B. Acidithiobacillus ferrivorans sp. nov.; facultatively anaerobic, psychrotolerant, iron- and sulfur-oxidizing acidophiles isolated from metal mine-impacted environments. Extremophiles 2010, 14, 9–19. [Google Scholar] [CrossRef]
- Johnson, D.B.; Bacelar-Nicolau, P.; Okibe, N.; Thomas, A.; Hallberg, K.B. Characteristics of Ferrimicrobium acidiphilum gen. nov., sp. nov., and Ferrithrix thermotolerans gen. nov., sp. nov.: heterotrophic iron-oxidizing, extremely acidophilic Actinobacteria. Int. J. Syst. Evol. Microbiol. 2009, 59, 1082–1089. [Google Scholar] [CrossRef]
- Winch, S.; Mills, H.J.; Kostka, J.E.; Fortin, D.; Lean, D.R. Identification of sulfate-reducing bacteria in methylmercury-contaminated mine tailings by analysis of SSU rRNA genes. FEMS Microbiol. Ecol. 2009, 68, 94–107. [Google Scholar]
- Jiang, C.Y.; Liu, Y.; Liu, Y.Y.; You, X.Y.; Guo, X.; Liu, S.J. Alicyclobacillus ferrooxydans sp. nov., a ferrous-oxidizing bacterium from solfataric soil. Int. J. Syst. Evol. Microbiol. 2008, 58, 2898–2903. [Google Scholar] [CrossRef]
- Urbieta, M.S.; Gonzalez Toril, E.; Aguilera, A.; Giaveno, M.A.; Donati, E. First prokaryotic biodiversity assessment using molecular techniques of an acidic river in Neuquen, Argentina. Microb. Ecol. 2012, 64, 91–104. [Google Scholar]
- Dridi, B.; Fardeau, M.L.; Ollivier, B.; Raoult, D.; Drancourt, M. Methanomassiliicoccus luminyensis gen. nov., sp. nov., a methanogenic archaeon isolated from human faeces. Int. J. Syst. Evol. Microbiol. 2012, 62, 1902–1907. [Google Scholar] [CrossRef]
- Brofft, J.E.; McArthur, J.V.; Shimkets, L.J. Recovery of novel bacterial diversity from a forested wetland impacted by reject coal. Environ. Microbiol. 2002, 4, 764–769. [Google Scholar] [CrossRef]
- Itoh, T.; Yoshikawa, N.; Takashina, T. Thermogymnomonas acidicola gen. nov., sp. nov., a novel thermoacidophilic, cell wall-less archaeon in the order Thermoplasmatales, isolated from a solfataric soil in Hakone, Japan. Int. J. Syst. Evol. Microbiol. 2007, 57, 2557–2561. [Google Scholar]
- Volant, A.; Desoeuvre, A.; Casiot, C.; Lauga, B.; Delpoux, S.; Morin, G.; Personne, J.C.; Hery, M.; Elbaz-Poulichet, F.; Bertin, P.N.; et al. Archaeal diversity: temporal variation in the arsenic-rich creek sediments of Carnoulès Mine, France. Extremophiles. 2012, 16, 645–657. [Google Scholar] [CrossRef]
- Heinzel, E.; Janneck, E.; Glombitza, F.; Schlömann, M.; Seifert, J. Population dynamics of iron-oxidizing communities in pilot plants for the treatment of acid mine waters. Environ. Sci. Technol. 2009, 43, 6138–6144. [Google Scholar] [CrossRef]
- Hedrich, S.; Schlömann, M.; Johnson, D.B. The iron-oxidizing Proteobacteria. Microbiol. 2011, 157, 1551–1564. [Google Scholar] [CrossRef]
- Kimura, S.; Bryan, C.G.; Hallberg, K.B.; Johnson, D.B. Biodiversity and geochemistry of an extremely acidic, low temperature subterranean environment sustained by chemolithotrophy. Environ. Microbiol. 2011, 13, 2092–2104. [Google Scholar]
- Rowe, O.F.; Sánchez-España, J.; Hallberg, K.B.; Johnson, D.B. Microbial communities and geochemical dynamics in an extremely acidic, metal-rich stream at an abandoned sulfide mine (Huelva, Spain) underpinned by two functional primary production systems. Environ. Microbiol. 2007, 9, 1761–1771. [Google Scholar] [CrossRef]
- ancucheo, I.; Johnson, D.B. Acidophilic algae isolated from mine-impacted environments and their roles in sustaining heterotrophic acidophiles. Front. Microbiol. 2012, 3, 325. [Google Scholar]
- Johnson, D.B.; Ghauri, M.A.; Said, M.F. Isolation and characterisation of an acidophilic heterotrophic bacterium capable of oxidizing ferrous iron. Appl. Environ. Microbiol. 1992, 58, 1423–1428. [Google Scholar]
- ancucheo, I.; Johnson, D.B. Selective removal of transition metals from acidic mine waters by novel consortia of acidophilic sulfidogenic bacteria. Microb. Biotechnol. 2012, 5, 34–44. [Google Scholar] [CrossRef]
- Kolmert, A.; Wikstrom, P.; Hallberg, K.B. A fast and simple turbidometric method for the determination of sulfate in sulfate-reducing bacterial cultures. J. Microbiol. Meth. 2000, 41, 179–184. [Google Scholar] [CrossRef]
- Lovley, D.R.; Phillips, E.J.P. Rapid assay for microbially reducible ferric iron in aquatic sediments. Appl. Environ. Microbiol. 1987, 53, 1536–1540. [Google Scholar]
- Suzuki, M.T.; Giovannoni, S.J. Bias caused by template annealing in the amplification of mixtures of 16S rRNA genes by PCR. Appl. Environ. Microbiol. 1996, 62, 625–630. [Google Scholar]
- Sambrook, J.; Fritsch, E.F.; Maniatis, T. Molecular Cloning: A Laboratory Manual, 2nd Ed. ed; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 1989. [Google Scholar]
- Johnson, D.B.; Hallberg, K.B. Techniques for detecting and identifying acidophilic mineral-oxidizing microorganisms. In Biomining; Rawlings, D.E., Johnson, D.B., Eds.; Springer-Verlag: Heidelberg, Germany, 2007; pp. 237–262. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kay, C.M.; Rowe, O.F.; Rocchetti, L.; Coupland, K.; Hallberg, K.B.; Johnson, D.B. Evolution of Microbial “Streamer” Growths in an Acidic, Metal-Contaminated Stream Draining an Abandoned Underground Copper Mine. Life 2013, 3, 189-210. https://doi.org/10.3390/life3010189
Kay CM, Rowe OF, Rocchetti L, Coupland K, Hallberg KB, Johnson DB. Evolution of Microbial “Streamer” Growths in an Acidic, Metal-Contaminated Stream Draining an Abandoned Underground Copper Mine. Life. 2013; 3(1):189-210. https://doi.org/10.3390/life3010189
Chicago/Turabian StyleKay, Catherine M., Owen F. Rowe, Laura Rocchetti, Kris Coupland, Kevin B. Hallberg, and D. Barrie Johnson. 2013. "Evolution of Microbial “Streamer” Growths in an Acidic, Metal-Contaminated Stream Draining an Abandoned Underground Copper Mine" Life 3, no. 1: 189-210. https://doi.org/10.3390/life3010189




