Quality Control System for Beer Developed with Monoclonal Antibodies Specific to Barley Lipid Transfer Protein
Abstract
:1. Introduction
2. Results and Discussion
2.1. Barley Grain LTP

2.2. Allergenicity of bLTP
| Patient No. | Barley | Wheat | Apple | Sensitization |
|---|---|---|---|---|
| IgE kU/L | ||||
| 1 | 1.2 | - | 6.4 | wheat products, nuts, beans |
| 2 | 9.8 | 33.1 | 25.6 | egg white |
| 3 | 15.8 | 14.3 | 14.4 | wheat products, nuts, beans |
| 4 | - | - | 11.7 | apple, pear, carrot, pollen |
| 5 | 25.6 | 46.8 | 19.2 | peach, apple, pear, fish, meat |
| 6 | 9.5 | 17.8 | 0.9 | nuts, beans |
| 7 | 5.6 | 5.7 | 1.2 | grass pollen |
| 8 | 5.5 | 5.4 | 4.7 | fish |
| 9 | 6.4 | 2.9 | - | plum, dates |
| 10 | - | - | 11.5 | milk products, egg, nuts, beans |
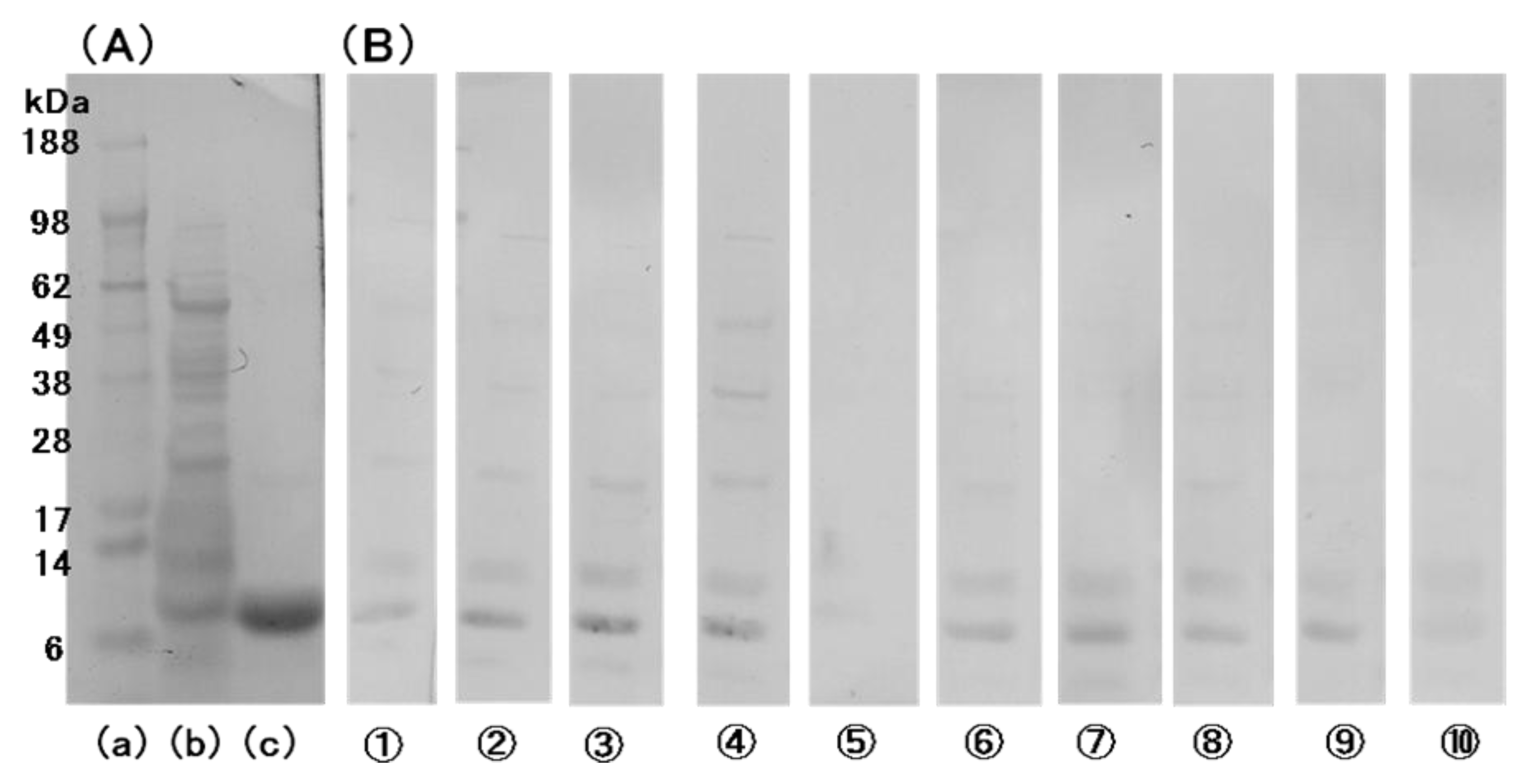
2.3. Preparation and Characterization of Monoclonal Antibodies to bLTP
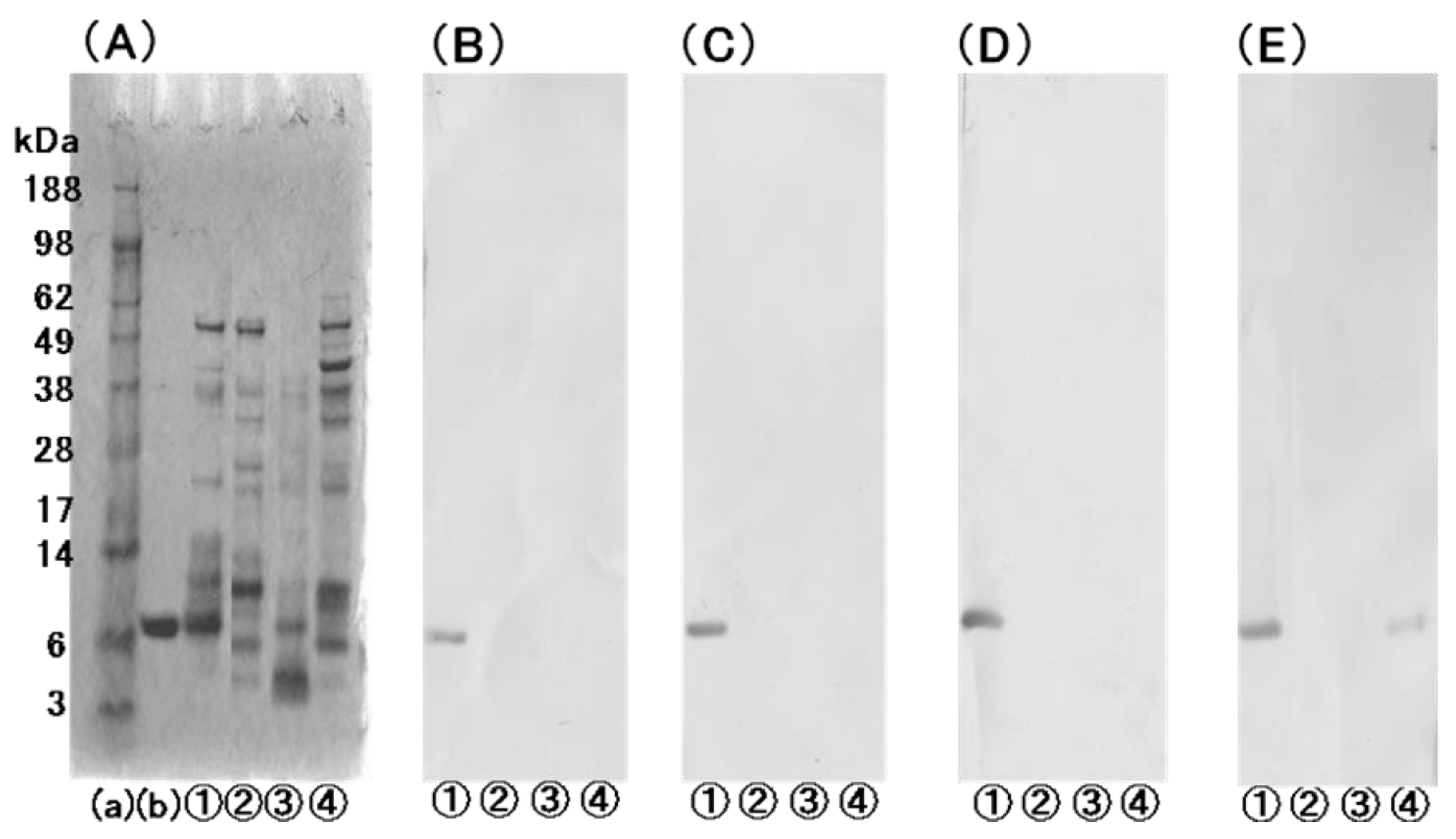
2.4. Sandwich ELISA for the Determination of bLTP
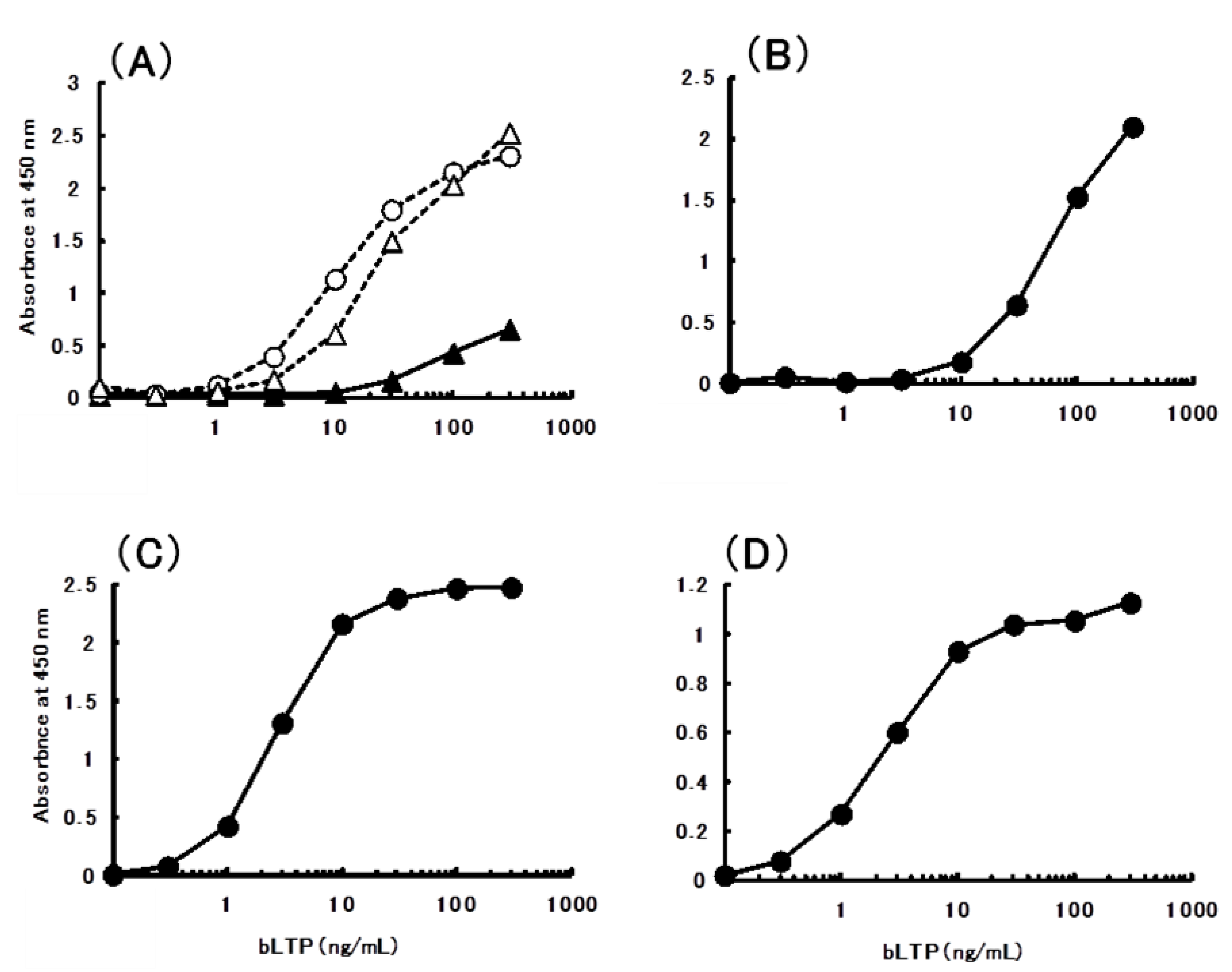
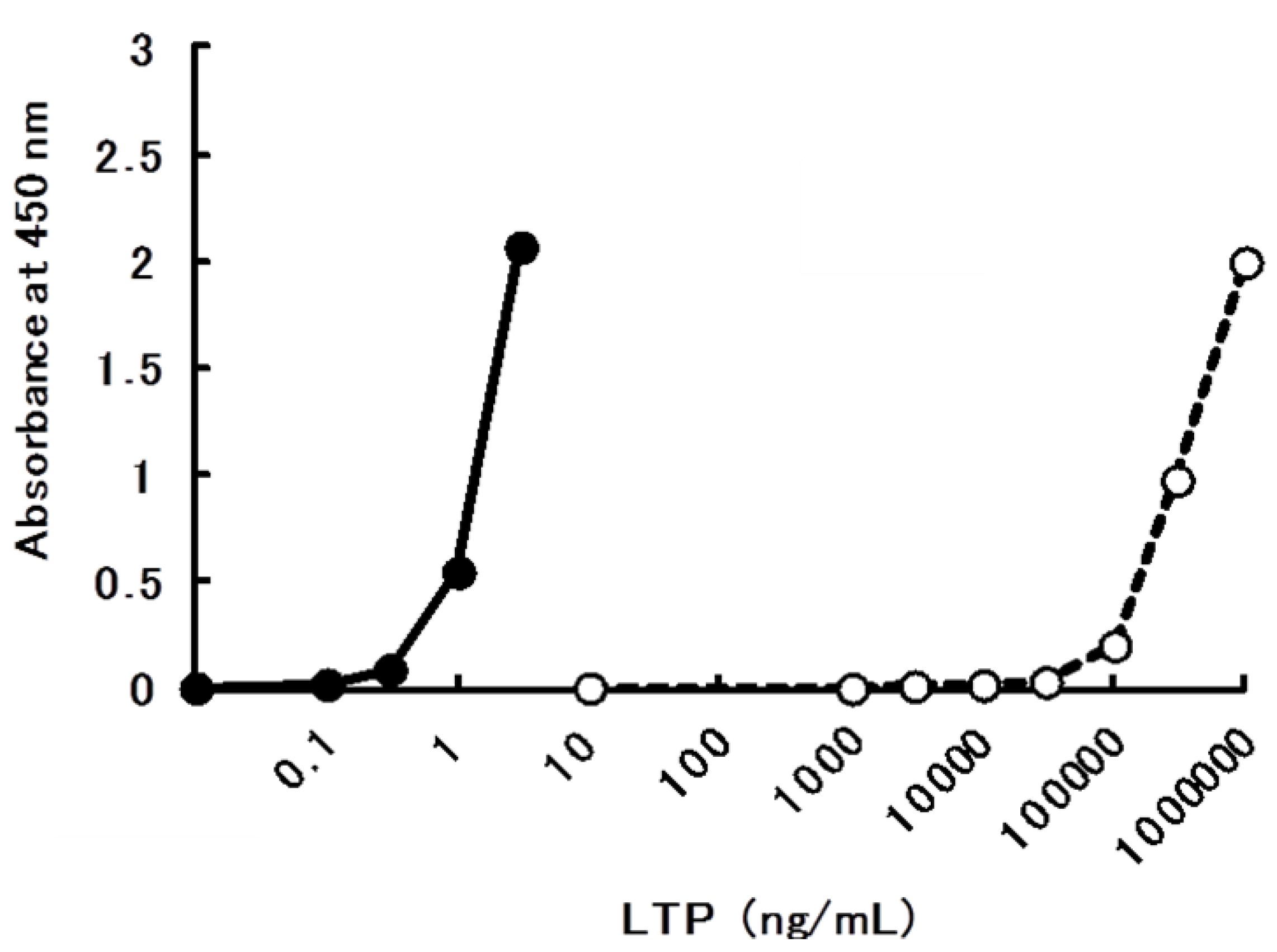
| Food | µg/g (mL) |
|---|---|
| Two-row barley flower | 393.7 |
| Two-row barley flower | 301.3 |
| Barley leaf | - |
| Hattaiko * | 0.1 |
| Roasted barley grain ** | - |
| Barley miso *** | 18.3 |
| Malt vinegar | 21.0 |
| beer | 2.6 |
2.5. Quantitative Analysis of bLTP in Beer
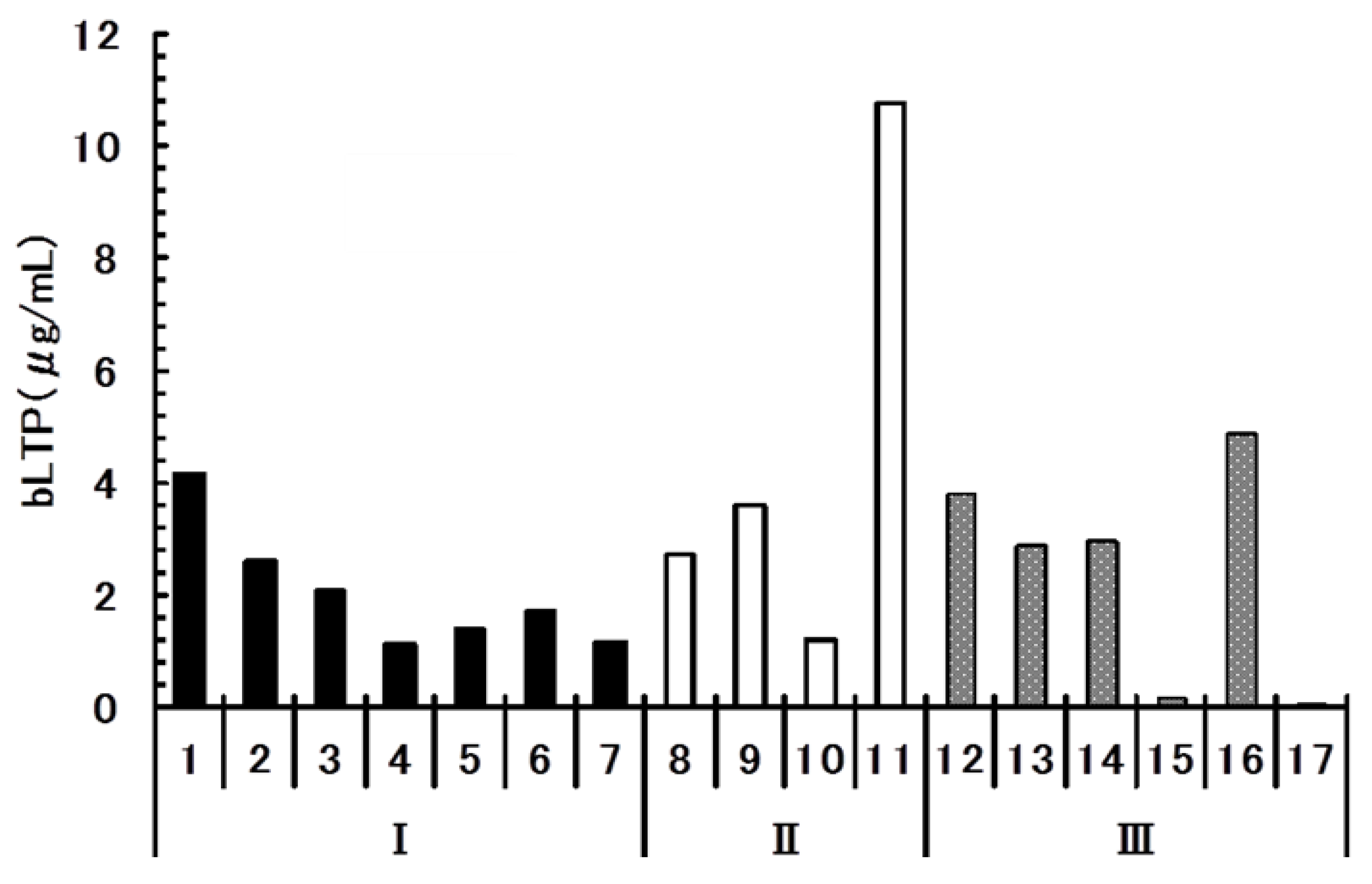
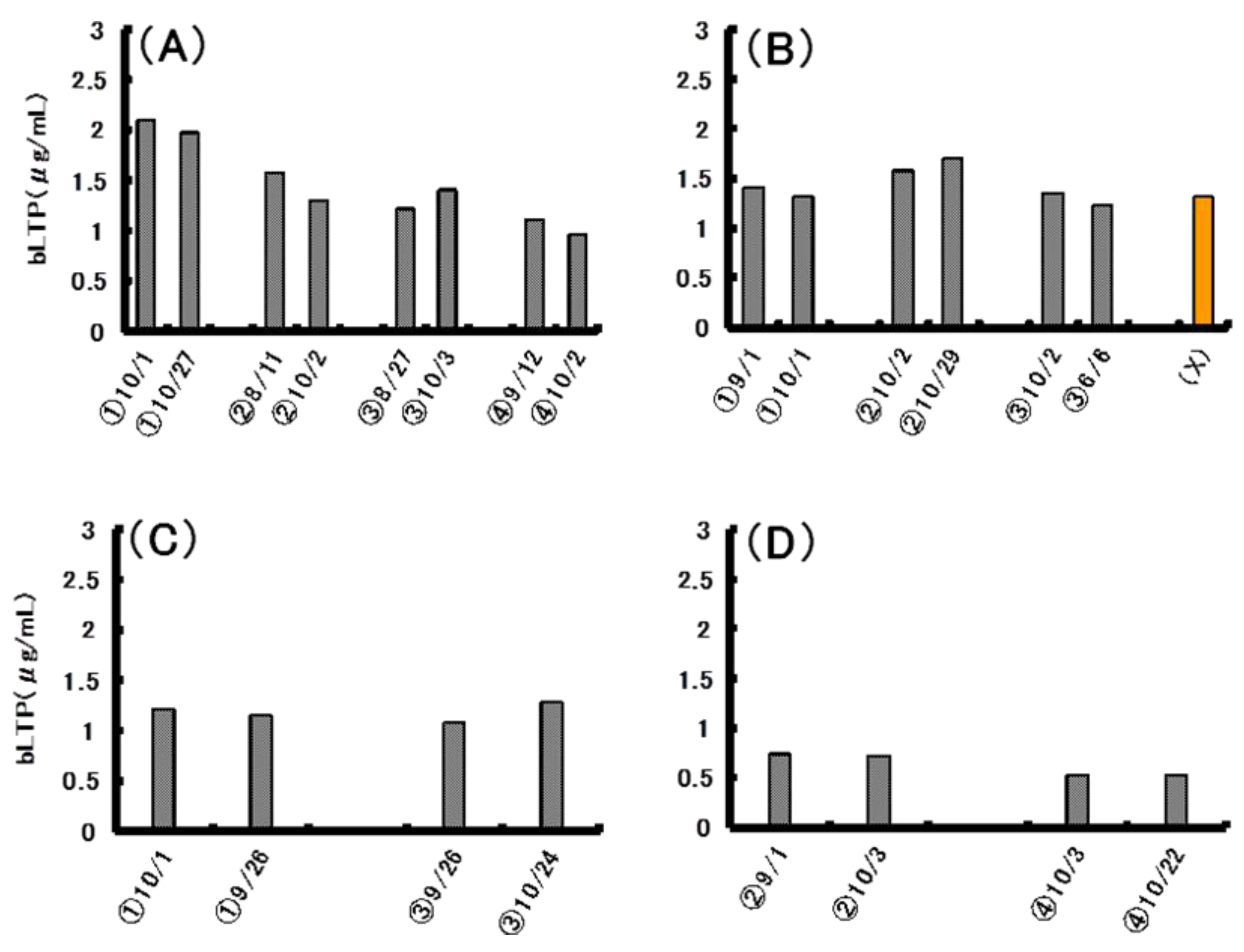
3. Experimental Section
3.1. Reagents and Apparatus
3.2. Isolation of Barley Grain LTP
3.3. Electrophoresis and Western Blotting
3.4. N-Terminal Amino Acid Sequencing
3.5. Production of Monoclonal Antibody
3.6. Preparation of Extract for LTP Determination
3.7. Enzyme-Linked Immunosorbent Assay (ELISA)
4. Conclusions
Conflict of Interest
Acknowledgements
References and Notes
- Kader, J.C. Lipid-transfer proteins in plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1996, 47, 627–654. [Google Scholar] [CrossRef]
- Douliez, J.P.; Michon, T.; Elmorjani, K.; Marion, D. Structure, biological and technological functions of lipid transfer protein and indolines, the major lipid binding protein from cereal kernels. J. Cereal Sci. 2000, 32, 1–20. [Google Scholar] [CrossRef]
- Garcia-Olmedo, F.; Molina, A.; Segura, A.; Moreno, M. The defensive role of nonspecific lipid-transfer proteins in plants. Trends Microbiol. 1995, 3, 72–74. [Google Scholar] [CrossRef]
- Kader, J.C. Lipid-transfer proteins: A puzzling family of plant proteins. Trends Plant Sci. Biol. 1997, 2, 66–70. [Google Scholar] [CrossRef]
- Hincha, D.K. Cryoprotectin: A plant lipid- transfer protein homologue that stabilized membranes during freezing. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2002, 357, 909–919. [Google Scholar] [CrossRef]
- Gaudet, D.; Laroche, A.; Frick, M.; Huel, R.; Puchalski, B. Cold induced expression of plant defensin and lipid transfer protein transcripts in winter wheat. Physiol. Plant 2003, 117, 195–205. [Google Scholar] [CrossRef]
- Bubier, J.; Schlappi, M. Cold induction of EARLI1, a putative Arabidopsis lipid transfer protein, is light and calcium dependent. Plant Cell Environ. 2004, 27, 929–936. [Google Scholar] [CrossRef]
- Gorjanović, S.; Sužnjević, D.; Beljanski, M.; Ostojić, S.; Gorjanović, R.; Vrvić, M.; Hranisavljević, J. Effects of lipid-transfer protein from malting barley grain on brewers yeast fermentation. J. Inst. Brew. 2004, 110, 297–302. [Google Scholar] [CrossRef]
- Sohal, A.K.; Pallas, J.A.; Jenkins, G.I. The promoter of a Brassica napus lipid transfer protein gene is active in a range of tissues and simulated by light and viral infection in transgenic Arabidopsis. Plant Mol. Biol. 1999, 41, 75–87. [Google Scholar] [CrossRef]
- Park, C.J.; Shin, R.; Park, J.M.; Lee, G.J.; You, J.S.; Paek, K.H. Induction of pepper cDNA encoding a lipid- transfer protein during the resistance response to tobacco mosaic virus. Plant Mol. Biol. 2002, 48, 243–254. [Google Scholar] [CrossRef]
- Zasloff, M. Antimicrobial peptides of multicellular organisms. Nature 2002, 415, 389–395. [Google Scholar] [CrossRef]
- Van Loon, L.; Van Strien, E. The families of pathogenesis-related proteins, their activities, and comparative analysis ofPR-1 type proteins. Physiol. Mol. Plant Pathol. 1999, 55, 85–97. [Google Scholar] [CrossRef]
- Sanchez-Monge, R.; Lombardero, M.; Garcia-Selles, F. J.; Barber, D.; Salcedo, G. Lipid-transfer proteins are relevant allergen in fruit allergy. J. Allergy Clin. Immunol. 1999, 103, 514–519. [Google Scholar] [CrossRef]
- Palacin, A.; Cumplido, J.; Figueroa, J.; Ahrazem, O.; sanchez-Monge, R.; Carrillo, T.; Salcedo, G.; Blanco, C. Cabbage lipid transfer protein Bra o 3 is major allergen responsible for cross-reactivity between plant foods and pollens. J. Allergy Clin. Immunol. 2006, 117, 1423–1429. [Google Scholar] [CrossRef]
- Curioni, A.; Santucci, B.; Cristaudo, A.; Canistraci, C.; Pietravalle, M.; Simonato, B.; Giannattasio, M. Urticaria from beer: An immediate hypersensitivity reaction due to a 10 kDa protein derived from barley. Clin. Exp. Allergy 1999, 29, 407–413. [Google Scholar] [CrossRef]
- Pastrorello, E.A.; Farioli, L.; Pravettoni, V.; Robino, A.M.; Scibilia, J.; Fortunato, D.; Conti, A.; Borgonovo, L.; Bengtsson, A.; Ortolani, C. Lipid transfer protein and vicilin are important walnut allergens in patients not allergic to pollen. J. Allergy Clin. Immunol. 2004, 114, 908–914. [Google Scholar] [CrossRef]
- Hiller, K.M.; Lubahan, B.C.; Klapper, D.G. Cloning and expression of ragweed allergen Amb a 6, Scand. J. Immunol. 1998, 48, 26–36. [Google Scholar]
- Charvolin, D.; Douliez, J.P.; Marion, D.; Cohen-Addad, C.; Pebay-Peyroula, E. The crystal structure of a wheat nonspecific lipid transfer protein (ns-LTP1) complexed with two molecules of phospholipids at 2.1 Å resolution. Eur. J. Biochem. 1999, 264, 562–568. [Google Scholar] [CrossRef]
- Lerche, M.H.; Poulsen, F.M. Solution structure of barley lipid transfer protein complexed with palmitate. Two different binding modes of palmitate in the homologous maize and different nonspecific lipid transfer protein. Protein Sci. 1998, 7, 2490–2498. [Google Scholar] [CrossRef]
- Salcedo, G.; Sanchez-Monge, R.; Diaz-Perales, A.; Garcia-Casado, G.; Barber, D. Plant non-specific lipid transfer proteins as food and pollen allergens. Clin. Exp. Allergy 2004, 34, 1336–1341. [Google Scholar] [CrossRef]
- Salcedo, G.; Diaz-Perales, A.; Sanchez-Monge, R. Fruits allergy: Plant defence protein as noval potential panallergen. Clin. Exp. Allergy 1999, 29, 1158–1160. [Google Scholar] [CrossRef]
- Lindroff-Larsen, K.; Winther, J.R. Surprisingly high stability of barley lipid transfer protein, LTP1, towards denaturant, heat and proteases. FEBS Lett. 2001, 488, 145–148. [Google Scholar] [CrossRef]
- Van Nierop, S.N.E.; Evans, D.E.; Axcel, B.C.; Cantrell, I.C.; Rautenbach, M. Impact of different wort boiling temperatures on the beer foam stabilizing properties of lipid transfer protein 1. J. Agric. Food Chem. 2004, 52, 3120–3129. [Google Scholar] [CrossRef]
- Garcia-Casado, G.; Crespo, J.F.; Rodriguez, J.; Salcedo, G. Isolation and charasterization of barley lipid transfer protein and protein Z as beer allergens. J. Allergy Clin. Immunol. 2001, 108, 647–649. [Google Scholar] [CrossRef]
- Quercia, O.; Zoccatelli, G.; Stefanini, G.F.; Mistrello, G.; Amato, S.; Bolla, M.; Emiliani, F.; Asero, R. Allergy to beer in LTP-sensitized patients: Beers are not all the same. Allergy 2012, 67, 1186–1189. [Google Scholar] [CrossRef]
- Akiyama, H.; Imai, T.; Ebisawa, M. Japan food allergen labeling regulation—History and Evaluation. In Advances in Food and Nutrition Research; Taylor, S., Ed.; Academic Press: Burlington, MA, USA, 2011; Volume 62, pp. 139–171. [Google Scholar]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar]
- Van Paridon, P. A.; Gadella, T.W.J., Jr.; Wirtz, K.W.A. The effect ofpolyphosphoinositides and phosphatidic acid on the phosphatidylinositol transfer protein from bovine brain: Kinetic study. Biochim. Biophys. Acta 1988, 943, 76–86. [Google Scholar] [CrossRef]
- Lusk, L.T.; Navarro, A.L.; Goldstein, H.; Wagner, R.J.; Ryder, D.S. Monoclonal antibodies for assaying lipid transfer proteins. U.S. Patent 6423546, 23 July 2002. [Google Scholar]
- Hirose, J.; Kitabatake, N.; Kimura, A.; Narita, H. Recognition of native and/or thermally induced denatured forms of the major food allergen, ovomucoid, by human IgE and mouse monoclonal IgG antibodies. Biosci. Biotechnol. Biochem. 2004, 68, 2490–2497. [Google Scholar] [CrossRef]
- Hirose, J.; Murakami-Yamaguchi, Y.; Ikeda, M.; Kitabatake, N.; Narita, H. Oligoclonal enzyme-linked immunosorbent assay capable of determining the major food allergen, ovomucoid, irrespective of the degree of heat denaturation. Cytotechnology 2005, 47, 145–149. [Google Scholar]
- Honjoh, T.; Muraoka, S.; Mamekoshi, S.; Sakai, M. Detection methods for allergic substances in foods by enzyme linked immunesorbent assay. Foods Food Ingredients J. Jpn. 2002, 206, 13–22. [Google Scholar]
- Murakami-Yamaguchi, Y.; Hirose, J.; Honjoh, T.; Narita, H. Evaluation of wheat content with monoclonal antibodies against lipid transfer protein. J. Jpn. Soc. Food Ssi. Technol. 2009, 56, 16–24. [Google Scholar]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Kohler, G.; Milstein, C. Continuous cultures of fused cells secreting antibody ofpredefined specificity. Nature 1975, 256, 495–497. [Google Scholar] [CrossRef]
- Nakane, P.K.; Kawaoi, A. Perioxidase-labelled antibody. New method of conjugation. J. Histochem. Cytochem. 1974, 22, 1084–1091. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Murakami-Yamaguchi, Y.; Hirose, J.; Kizu, K.; Okazaki, F.; Fujii, W.; Narita, H. Quality Control System for Beer Developed with Monoclonal Antibodies Specific to Barley Lipid Transfer Protein. Antibodies 2012, 1, 259-272. https://doi.org/10.3390/antib1030259
Murakami-Yamaguchi Y, Hirose J, Kizu K, Okazaki F, Fujii W, Narita H. Quality Control System for Beer Developed with Monoclonal Antibodies Specific to Barley Lipid Transfer Protein. Antibodies. 2012; 1(3):259-272. https://doi.org/10.3390/antib1030259
Chicago/Turabian StyleMurakami-Yamaguchi, Yukie, Junko Hirose, Kumiko Kizu, Fumiko Okazaki, Wataru Fujii, and Hiroshi Narita. 2012. "Quality Control System for Beer Developed with Monoclonal Antibodies Specific to Barley Lipid Transfer Protein" Antibodies 1, no. 3: 259-272. https://doi.org/10.3390/antib1030259




