Spatial Distribution of Fecal Indicator Bacteria in Groundwater beneath Two Large On-Site Wastewater Treatment Systems
Abstract
:1. Introduction
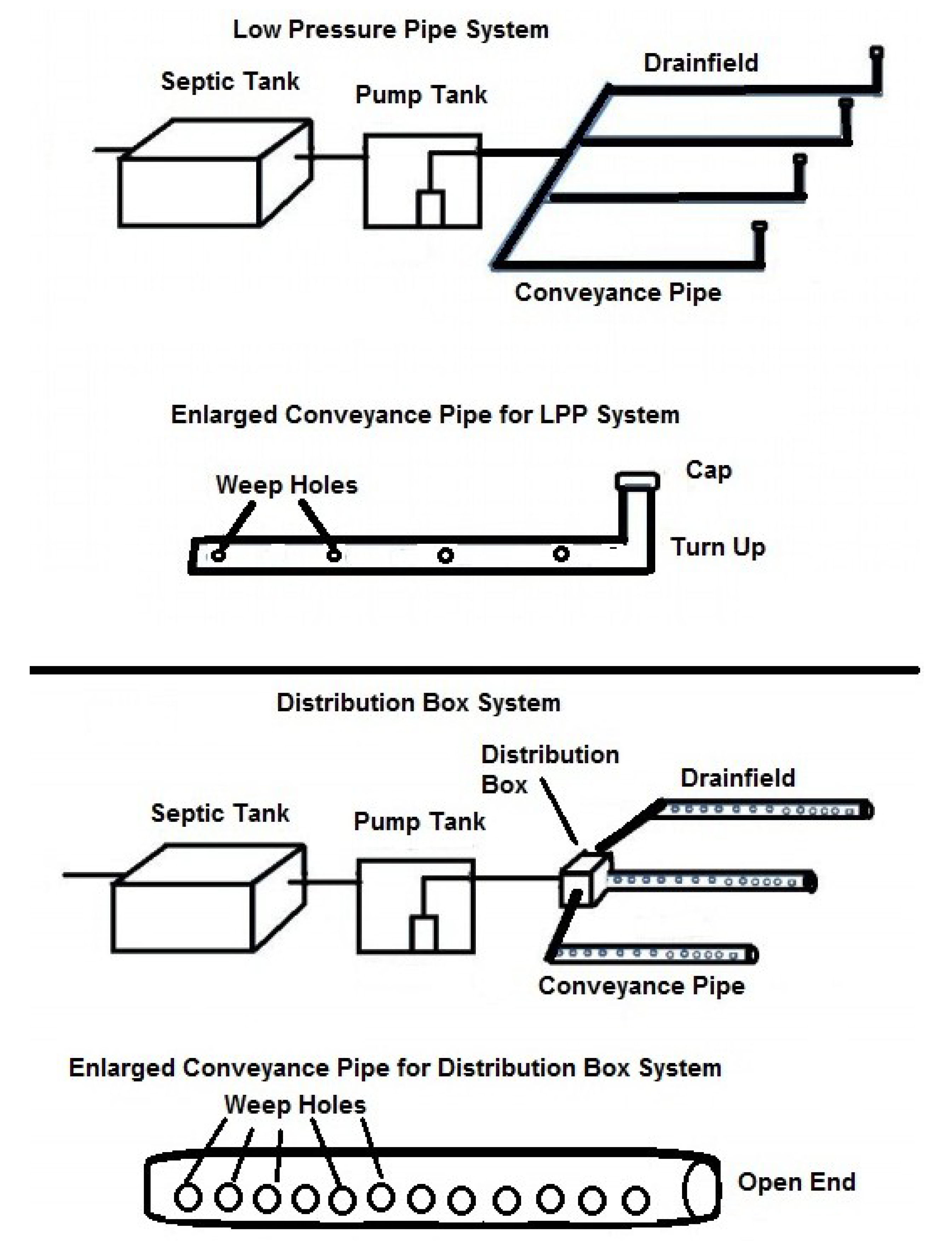
2. Materials and Methods
2.1. Site Selection
| Site | Install Date | Septic Tank Capacity (L) | Max Design Flow (L/d) | Grease Trap (L) | Pump Tank (L) | Distribution Device | Dispersal Area (m2) | Vertical Separation (m) | USDA Soil Series |
|---|---|---|---|---|---|---|---|---|---|
| JWS | 1987 | 37,800 | 37,800 | 3780 | 18,144 | D-box (2) | 892 | >4 m | Autryville |
| WCH | 1997 | 73,827 | 73,827 | 11,340 | 11,340 | LPP (2) | 1115 | >1 m | Tarboro |
2.2. Groundwater Monitoring Network
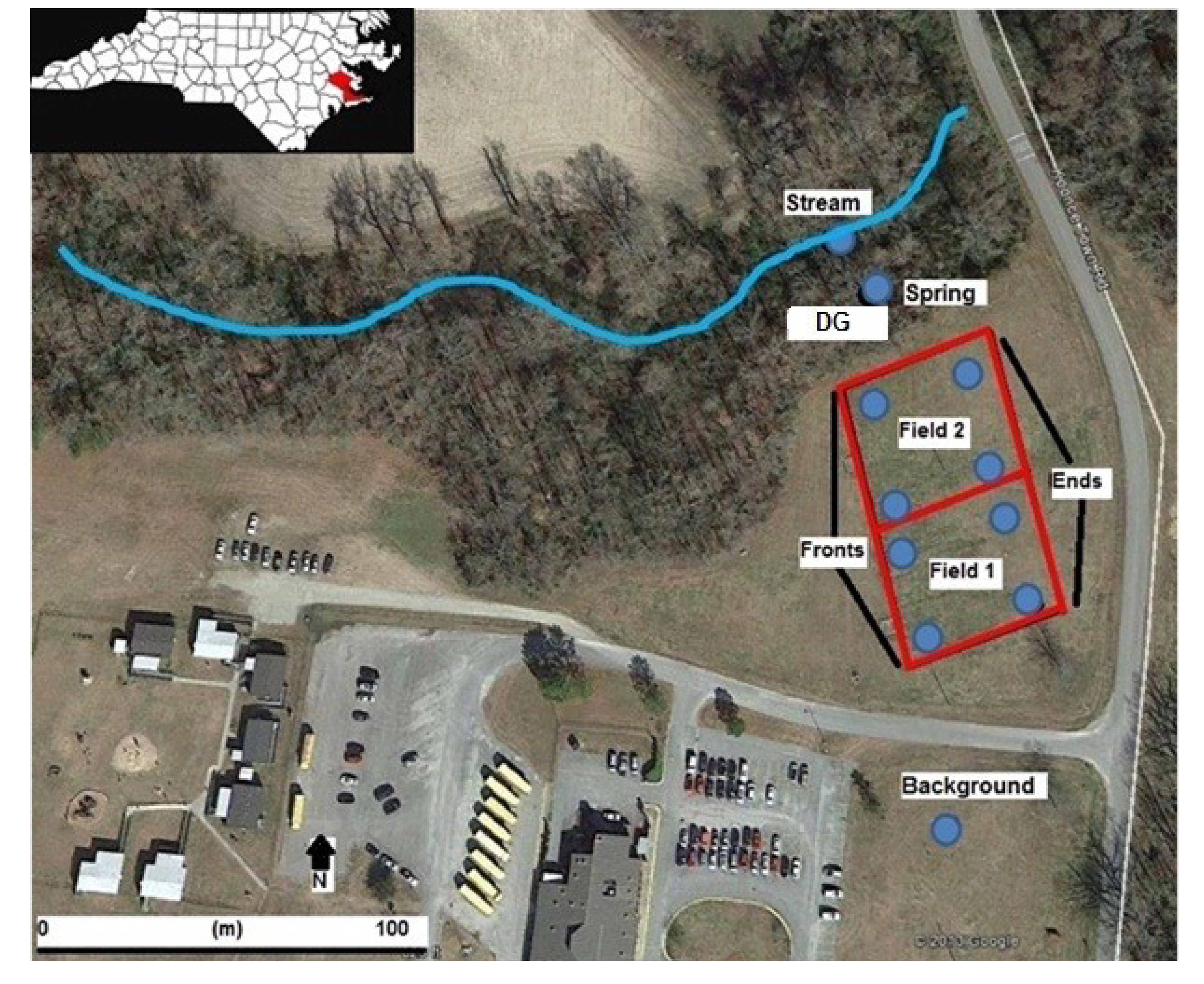

2.3. Groundwater Characterization and Analysis
2.4. Spatial Distribution of Fecal Indicators
2.5. Treatment Efficiency
3. Results
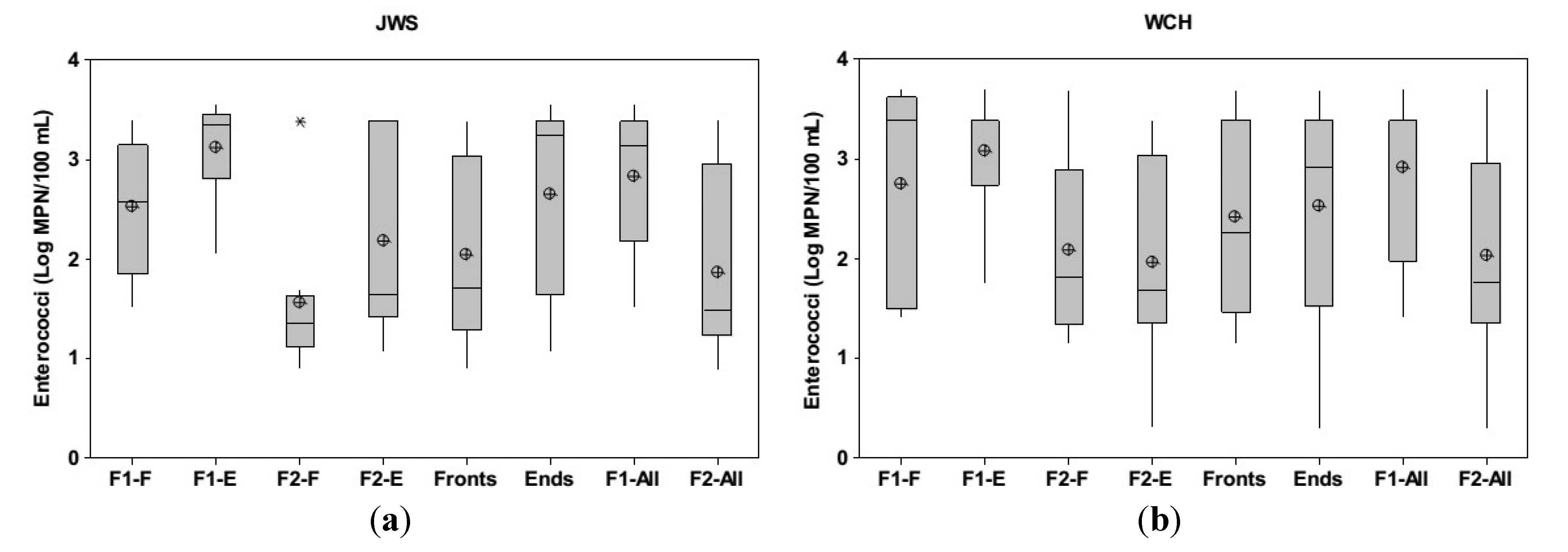
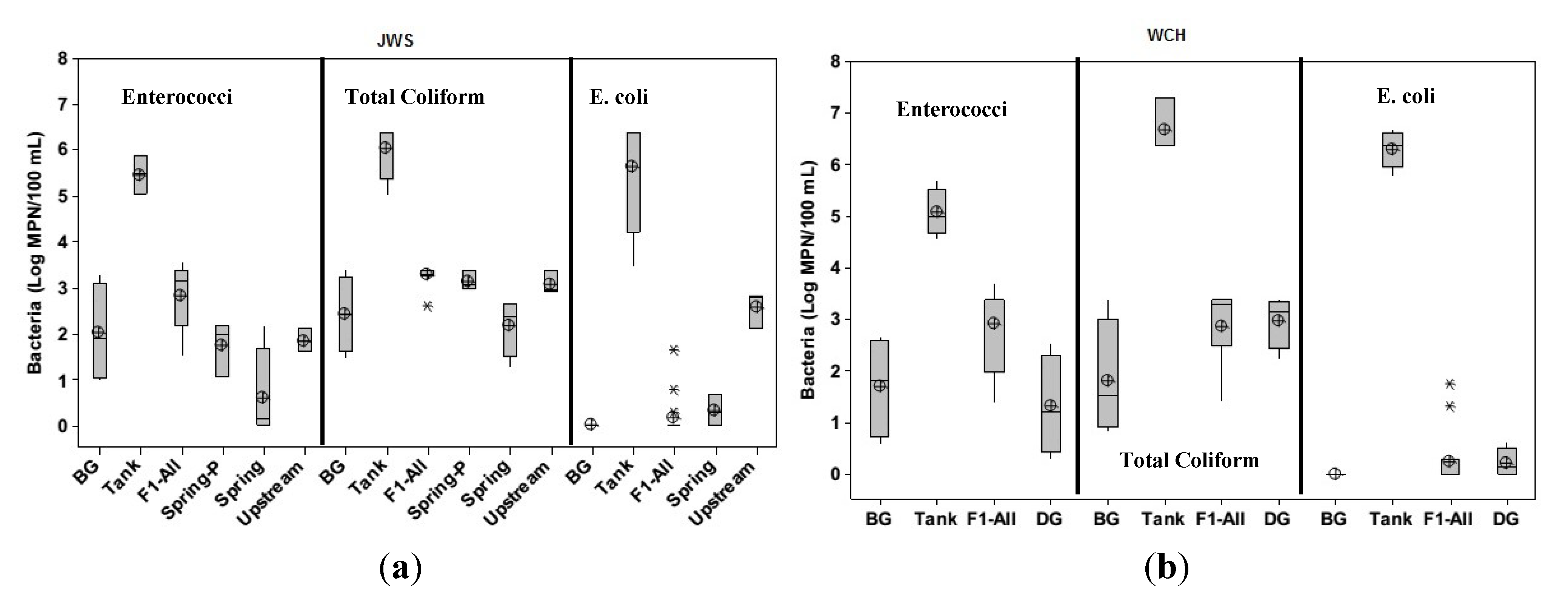
3.1. Groundwater Enterococci Distribution
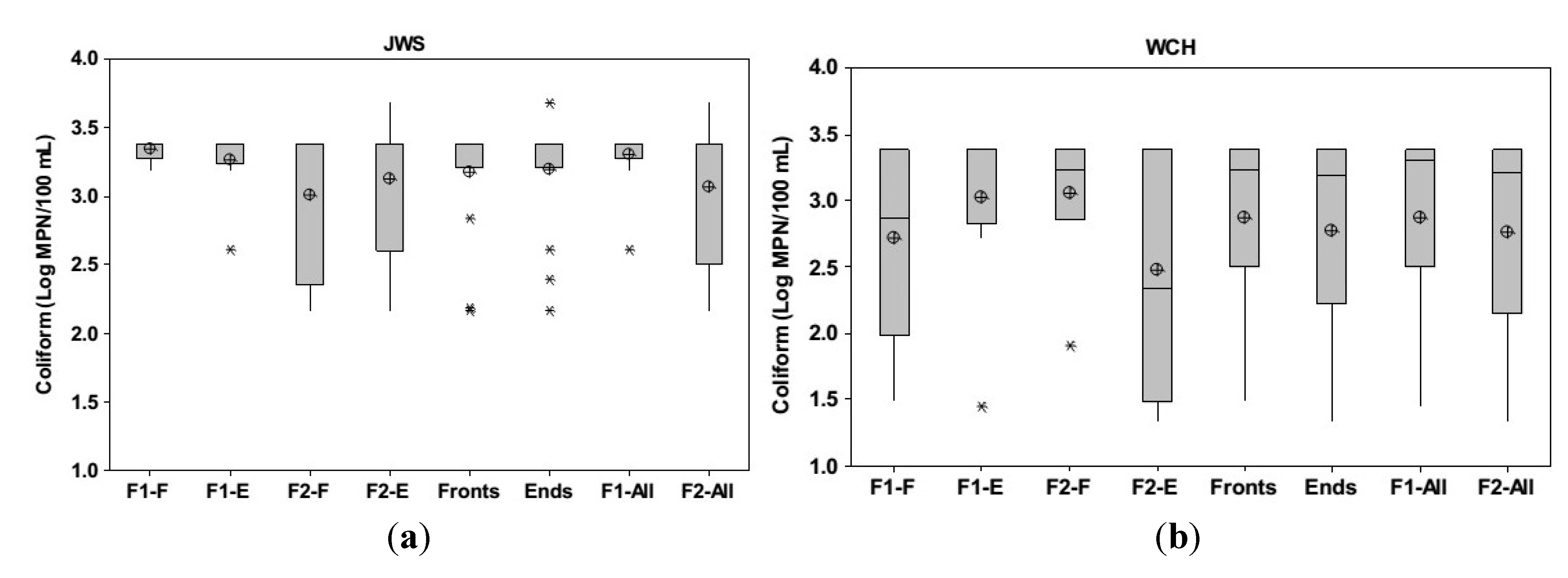
3.2. Groundwater Total Coliform Distribution
3.3. Groundwater E. coli Distribution
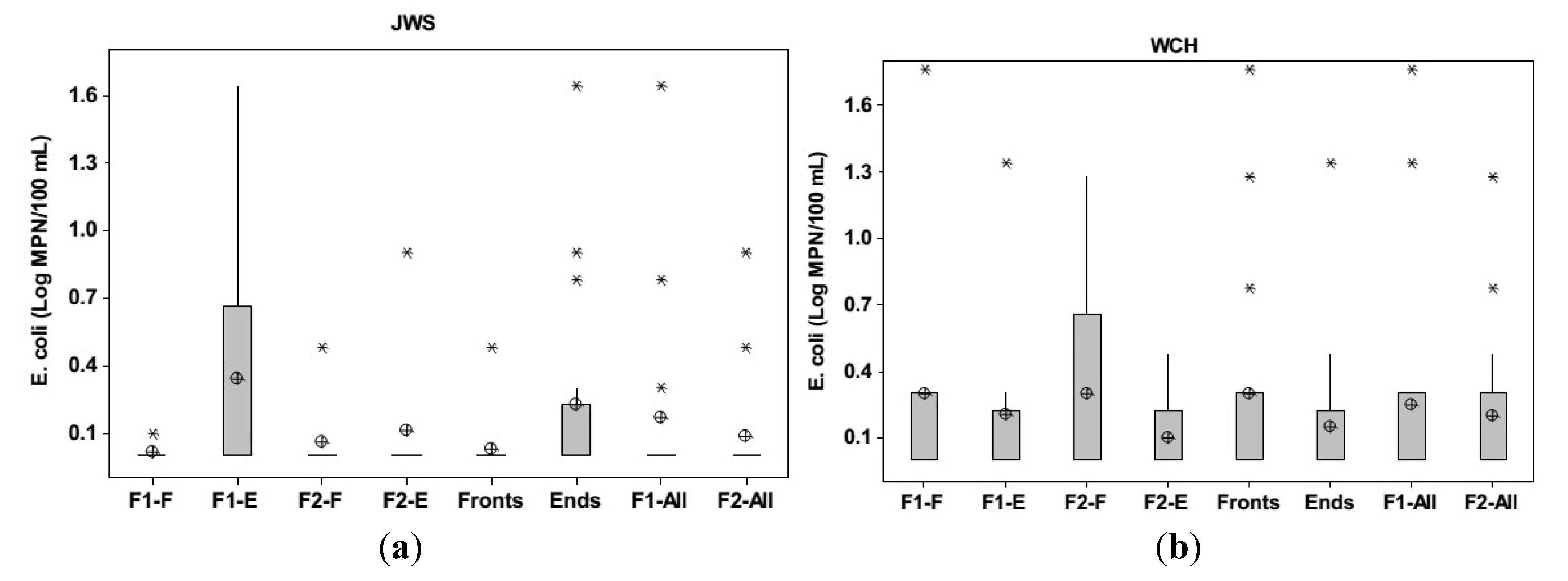
3.4. Microbial Treatment
3.5. Physical and Chemical Parameters
| Site | Sampling Location | Specific Conductivity (µS/cm) | pH | Temp (°C) | Depth to Water (m) |
|---|---|---|---|---|---|
| JWS | Field 1 | 753 (246) | 6.37 (0.57) | 18.3 (1.1) | 4.56 (0.91) |
| Field 2 | 547 (237) | 6.89 (0.75) | 18.9 (1.1) | 6.21 (1.02) | |
| Front | 547 (204) | 6.45 (0.80) | 18.4 (0.9) | 4.92 (1.25) | |
| Ends | 754 (268) | 6.78 (0.58) | 18.8 (1.2) | 5.85 (1.15) | |
| Background | 98 (51) | 5.12 (0.65) | 18.1 (0.5) | 4.32 (0.31) | |
| Tank | 1057 (387) | 7.31 (0.26) | 17.8 (2.9) | - | |
| Down-gradient | 620 (69) | 7.23 (0.27) | 17.5 (0.8) | 0.34 (0.02) | |
| Spring | 445 (29) | 6.91 (0.37) | 18.2 (0.6) | - | |
| Upstream | 144 (12) | 7.22 (0.25) | 13.7 (1.1) | - | |
| WCH | Field 1 | 312 (401) | 5.97 (0.65) | 16.4 (2.1) | 1.76 (0.16) |
| Field 2 | 550 (302) | 6.53 (0.34) | 16.7 (1.8) | 1.62 (0.11) | |
| Front | 521 (327) | 6.32 (0.55) | 16.6 (2.0) | 1.69 (0.16) | |
| Ends | 340 (397) | 6.17 (0.63) | 16.4 (1.9) | 1.69 (0.16) | |
| Background | 49 (12) | 6.56 (0.99) | 15.5 (2.2) | 1.53 (0.31) | |
| Tank | 1196 (432) | 6.91 (0.28) | 17.9 (3.5) | - | |
| Down-gradient | 710 (212) | 6.53 (0.22) | 17.2 (1.5) | 1.44 (0.13) |
4. Discussion
4.1. Microbial Distribution
4.2. Treatment
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- United States Environmental Protection Agency (USEPA). On-site Wastewater Treatment Systems Manual; EPA/625/R-00/008; USEPA: Washington, DC, USA, 2002; pp. 1–11.
- Carroll, S.; Hargreaves, M.; Goonetilleke, A. Sourcing faecal pollution from onsite wastewater treatment systems in surface waters using antibiotic resistance analysis. J. Appl. Microbiol. 2005, 99, 471–482. [Google Scholar] [CrossRef] [Green Version]
- Harman, J.; Robertson, W.D.; Cherry, J.A.; Zanini, L. Impacts on a sand aquifer from an old septic system: Nitrate and phosphate. Groundwater 1996, 34, 1105–1114. [Google Scholar] [CrossRef]
- Pang, L.; Close, M.; Goltz, M.; Sinton, L.; Davies, H.; Hall, C.; Stanton, G. Estimation of septic tank setback distances based on transport of E. coli and F-RNA phages. Environ. Int. 2003, 29, 907–921. [Google Scholar]
- Patel, T.; O’Luanaigh, N.O.; Gill, L.W. A Comparison of gravity distribution devices used in On-site domestic wastewater treatment systems. Water Air Soil Pollut. 2008, 191, 55–69. [Google Scholar] [CrossRef]
- Lowe, K.S.; Rothe, N.K.; Tomaras, J.M.B.; DeJong, K.; Tucholke, M.B.; Drewes, J.; McCray, J.E.; Munakata-Marr, J. Influent constituent characteristics of the modern waste stream rom single sources. In Literature Review; Water Environment Research Foundation: Alexandria, VA, USA, 2007; pp. 3–19. [Google Scholar]
- Humphrey, C.P.; O’Driscoll, M.A.; Zarate, M.A. Evaluation of on-site wastewater system Escherichia coli contributions to shallow groundwater in coastal North Carolina. Water Sci. Technol. 2011, 63, 789–795. [Google Scholar] [CrossRef]
- Conn, K.E.; Habteselassie, M.Y.; Blackwood, A.D.; Noble, R.T. Microbial water quality before and after the repair of a failing onsite wastewater treatment system adjacent to coastal waters. J. Appl. Microbiol. 2011, 112, 214–224. [Google Scholar]
- Habteselassie, M.Y.; Kirs, M.; Conn, K.E.; Blackwood, A.D.; Kelly, G.; Noble, R.T. Tracking microbial transport through four onsite wastewater treatment systems to receiving waters in eastern North Carolina. J. Appl. Microbiol. 2011, 111, 835–847. [Google Scholar] [CrossRef]
- Harris, J.; Humphrey, C.; O’Driscoll, M. Transport of indicator microorganisms from an onsite wastewater system to adjacent stream. Univers. J. Environ. Res. Technol. 2013, 3, 423–426. [Google Scholar]
- Cahoon, L.B.; Hales, J.C.; Carey, E.S.; Loucaides, S.; Rowland, K.R.; Nearhoof, J.E. Shellfishing closures in southwest Brunswick County, North Carolina: Septic tanks vs. stormwater runoff as fecal coliform sources. J. Coast. Res. 2006, 22, 319–327. [Google Scholar]
- Meeroff, D.; Bloetscher, F.; Bocca, T.; Morin, F. Evaluation of water quality impacts of on-site wastewater treatment and disposal systems on urban coastal waters. Water Air Soil Pollut. 2008, 192, 11–24. [Google Scholar] [CrossRef]
- Borchardt, M.A.; Po-Huang, C.; DeVries, E.O.; Belongia, E.A. Septic system density and infectious diarrhea in a defined population of children. Environ. Health Perspect. 2003, 111, 742–748. [Google Scholar] [CrossRef]
- Scandura, J.E.; Sobsey, M.D. Viral and bacterial contamination of groundwater from on-site sewage treatment systems. Water Sci. Technol. 1997, 35, 141–146. [Google Scholar] [CrossRef]
- Karathanasis, A.D.; Mueller, T.G.; Boone, B.; Thompson, Y.L. Effect of soil depth and texture on fecal bacteria removal from septic effluents. J. Water Health 2006, 4, 395–404. [Google Scholar]
- Ijzerman, M.M.; Hagedorn, C.; Reneau, R.B. Microbial tracers to evaluate an on-site shallow-placed low pressure distribution system. Water Res. 1993, 27, 343–347. [Google Scholar] [CrossRef]
- O’Luanaigh, N.D.; Gill, L.W.; Misstear, B.D.R.; Johnston, P.M. The attenuation of microorganisms in on-site wastewater effluent discharged into highly permeable subsoils. J. Contam. Hydrol. 2012, 142–143, 126–139. [Google Scholar] [CrossRef]
- Motz, E.C.; Cey, E.; Ryan, M.C.; Chu, A. Vadose zone microbial transport below at-grade distribution of wastewater effluent. Water Air Soil Pollut. 2012, 223, 771–785. [Google Scholar] [CrossRef]
- Carlile, B.L.; Cogger, C.G.; Sobsey, M.D.; Scandura, J.; Steinbeck, S.J. Movement and Fate of Septic Tank Effluent in Soils of the North Carolina Coastal Plain; Report to the Coastal Plains Regional Commission: Raleigh, NC, USA, October 1981; p. 37. [Google Scholar]
- Ijzerman, M.M.; Hagedorn, C.; Reneau, R.B. Fecal indicator organisms below an on-site wastewater system with low pressure pipe distribution. Water Air Soil Pollut. 1992, 63, 201–210. [Google Scholar] [CrossRef]
- North Carolina Division of Environmental Health: On-site Wastewater Section. Laws and Rules for Sewage Treatment and Disposals Systems; Raleigh, NC, USA, 1999. Available online: http://ehs.ncpublichealth.com/oswp/resources.htm (accessed on 11 March 2014).
- U.S. Department of Agriculture, Natural Resources Conservation Service. Soil Survey of Craven County, North Carolina, USA. 1989. Available online: http://soils.usda.gov/survey/printed_surveys/ (accessed on 11 March 2014).
- Blott, S.; Pye, K. Gradistat: A grain size distribution and statistics package for the analysis of uncosolidated sediments. Earth Surf. Process. Landf. 2001, 26, 1237–1248. [Google Scholar] [CrossRef]
- Domenico, P.A.; Schwartz, W. Physical and Chemical Hydrogeology, 2nd ed.; John Wiley & Sons, Inc.: New York, NY, USA, 1998; p. 36. [Google Scholar]
- Del Rosario, K.L.; Humphrey, C.P.; Mitra, S.; O’Driscoll, M.A. Nitrogen and carbon dynamics beneath on-site wastewater treatment systems in pitt county, North Carolina. Water Sci. Technol. 2014, 69, 663–671. [Google Scholar] [CrossRef]
- Humphrey, C.P.; O’Driscoll, M.A. Biogeochemistry of groundwater beneath on-site wastewater systems in a coastal watershed. Univers. J. Environ. Res. Technol. 2011, 1, 320–328. [Google Scholar]
- United States Environmental Protection Agency (USEPA). Managing Urban Watershed Pathogen Contamination; EPA/600/R-03/111; USEPA: Cincinnati, OH, USA, 2003.
- United States Environmental Protection Agency. Ambient Water Quality Criteria for Bacteria; EPA440/5–84–002; USEPA: Washington, DC, USA, 1986.
- Stevik, T.K.; Aa, K.; Ausland, G.; Hanssen, J.F. Retention and removal of pathogenic bacteria in wastewater percolating through porous media: A review. Water Res. 2004, 38, 1355–1367. [Google Scholar] [CrossRef]
- Hoover, M.T.; Disy, T.A.; Pfieffer, M.A.; Dudley, N.; Mayer, R.B.; Buffington, B. North Carolina Subsurface Wastewater Operators Training School Manual; Soil Science Department, College of Agriculture and Life Sciences, North Carolina State University: Raleigh, NC, USA; North Carolina Department of Environment, Health and Natural Resources: Raleigh, NC, USA, 1996. [Google Scholar]
- Beal, C.D.; Gardner, E.A.; Kirchhof, G.; Menzies, N.W. Long-term flow rates and biomat zone hydrology in soil columns receiving septic tank effluent. Water Res. 2006, 40, 2327–2338. [Google Scholar] [CrossRef]
- Siegrist, R.; Boyle, W.C. Wastewater-induced soil clogging development. J. Environ. Eng. 1987, 113, 550–566. [Google Scholar] [CrossRef]
- Stevik, T.K.; Ausland, G.; Jenssen, P.D.; Siegrist, R. Removal of E. coli during intermittent filtration of wastewater effluent as affected by dosing rate and media type. Water Res. 1999, 33, 2088–2098. [Google Scholar] [CrossRef]
- Alhajjar, B.J.; Chesters, G.; Harkin, J.M. Indicators of Chemical Pollution from Septic Systems. Groundwater 1990, 28, 559–568. [Google Scholar] [CrossRef]
- Humphrey, C.P.; Deal, N.E.; O’Driscoll, M.A.; Lindbo, D.L. Characterization of on-site wastewater nitrogen plumes in shallow coastal aquifers, North Carolina. In Proceedings of the 2010 World Environmental & Water Resources Congress, Providence, RI, USA, 16–20 May 2010; pp. 949–958.
- Wright, M.E.; Solo-Gabriele, H.M.; Elmir, S.; Fleming, L.E. Microbial load from animal feces at a recreational beach. Mar. Pollut. Bull. 2009, 58, 1649–1656. [Google Scholar] [CrossRef]
- Liwimbi, L.; Graves, A.K.; Israel, D.W.; van Heugten, E.; Robinson, B.; Cahoon, C.W.; Lubbers, J.F. Microbial source tracking in a watershed dominated by swine. Water 2010, 2, 587–604. [Google Scholar] [CrossRef]
- Whitlock, J.E.; Jones, D.T.; Harwood, V.J. Identification of the sources of fecal coliforms in an urban watershed using antibiotic resistance analysis. Water Res. 2002, 36, 4237–4282. [Google Scholar]
- Sanders, E.C.; Yuan, Y.; Pitchford, A. Fecal coliform and E. coli concentrations in effluent-dominated streams of the Upper Santa Cruz Watershed. Water 2013, 5, 243–261. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Humphrey, C.; O'Driscoll, M.; Harris, J. Spatial Distribution of Fecal Indicator Bacteria in Groundwater beneath Two Large On-Site Wastewater Treatment Systems. Water 2014, 6, 602-619. https://doi.org/10.3390/w6030602
Humphrey C, O'Driscoll M, Harris J. Spatial Distribution of Fecal Indicator Bacteria in Groundwater beneath Two Large On-Site Wastewater Treatment Systems. Water. 2014; 6(3):602-619. https://doi.org/10.3390/w6030602
Chicago/Turabian StyleHumphrey, Charles, Michael O'Driscoll, and Jonathan Harris. 2014. "Spatial Distribution of Fecal Indicator Bacteria in Groundwater beneath Two Large On-Site Wastewater Treatment Systems" Water 6, no. 3: 602-619. https://doi.org/10.3390/w6030602





