Pharmaceuticals in the Built and Natural Water Environment of the United States
Abstract
:1. Introduction
2. Built and Natural Water Environment
2.1. General Overview

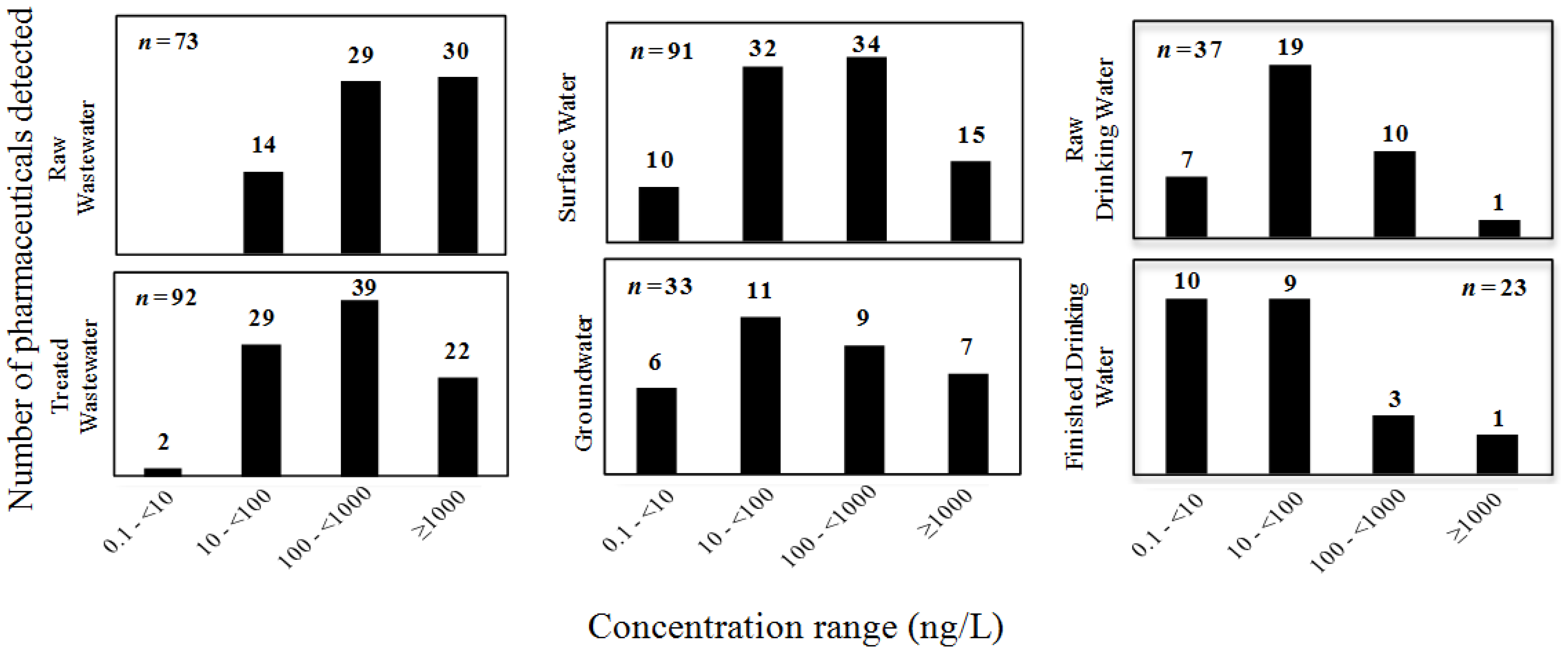
2.2. Occurrence and Distribution of Pharmaceuticals in the Built and Natural Water Environment
| Pharmaceuticals | PNEC (ng/L) | GW (ng/L) | RQ (GW) | FDW (ng/L) | RQ (FDW) |
|---|---|---|---|---|---|
| Acetaminophen | 1,000 [72] | 1,890 [68] | 1.890 | 28 [86] | 0.028 |
| Atenolol | 3.1 × 105 [73] | NA | – | 26 [37] | 0.01 |
| Caffeine | 1.0 × 107 [64] | 290 [68] | 0.01 | 180.8 [86] | 0.01 |
| Carbamazepine | 420 [74] | 420 [68,79] | 1.00 | 258 [87] | 0.614 |
| Ciprofloxacin | 5 [75] | 45 [8] | 9.0 | NA | – |
| Codeine | 2,900 a | 214 [68] | 0.07 | 30 [88] | 0.01 |
| Cotinine | 5,200 a | NA | – | 25 [87] | 0.01 |
| Dehydronifedipine | 15,000 a | 22 [80] | <0.01 | 4 [87] | 0.01 |
| Diazepam | 4,300 [76] | NA | – | 0.33 [14] | 0.01 |
| Diclofenac | 460 [77] | 46 [79] | 0.1 | NA | – |
| Dilantin (Phenytoin) | 1,800 a | 22 [79] | – | 32 [37] | 0.018 |
| Diltiazem | 920 a | 28 [80] | 0.03 | NA | – |
| 1,7-dimethylxanthine | 8,000 a | 57 [80] | 0.007 | NA | – |
| Erythromycin | 20 [77] | 2,380 [81] | 119 | 1.3 [89] | 0.065 |
| 17-α-ethinylestradiol | 1,800 a | 230 [8] | 0.128 | NA | – |
| Fluoxetine | 47 [77] | 56 [80] | 1.191 | 0.82 [14] | 0.017 |
| Gemfibrozil | 780 [77] | 6,860 [82] | – | 6.5 [90] | 0.008 |
| Genistein | 550 a | NA | – | 2.9 [37] | 0.005 |
| Ibuprofen | 1,000 [72] | 3,110 [80] | 3.11 | 1350 [91] | 1.350 |
| Iopromide | 460,000 a | NA | – | 31 [89] | 0.01 |
| Lincomycin | 13,000 a | 1,900 [83] | 0.025 | 4.4 [86] | 0.01 |
| Meprobamate | 110,000 a | 8.6 [79] | – | 43 [37] | 0.01 |
| Naproxen | 640 [77] | 0.7 [79] | – | 8 [89] | 0.013 |
| Oestriol | 14,000 a | 6.4 [79] | <0.01 | NA | – |
| Oestrone | 4,800 a | 1 [79] | <0.01 | NA | – |
| Oxybenzone | 3,500 a | 7.5 [79] | <0.01 | NA | – |
| Oxytetracycline | 200 [76] | 139 [84] | 0.695 | NA | – |
| Pentoxifylline | 4,600 a | 34 [79] | <0.01 | NA | – |
| Primidone | 4,300 a | 2.8 [85] | – | 1.3 [92] | 0.01 |
| Sulfadimethoxine | 248,000 a | 130 [83] | <0.01 | NA | – |
| Sulfamerazine | 116,000 a | 54 [81] | <0.01 | NA | – |
| Sulfamethazine | 1.2 × 106 a | 3,600 [83] | 0.001 | NA | – |
| Sulfamethoxazole | 27 [78] | 1,110 [80] | 41.11 | 20 [89] | 0.741 |
| Sulfathiazole | 5,000 a | 305 [81] | 0.061 | 10 [88] | 0.002 |
| Tetracycline | 90 [76] | 500 [7] | 5.6 | NA | – |
| Triclosan | 1,550 [72] | 53 [8] | 0.034 | 734 [91] | 0.474 |
| Trimethoprim | 1,000 [64] | 18 [68] | 0.018 | 1.7 [86] | 0.002 |
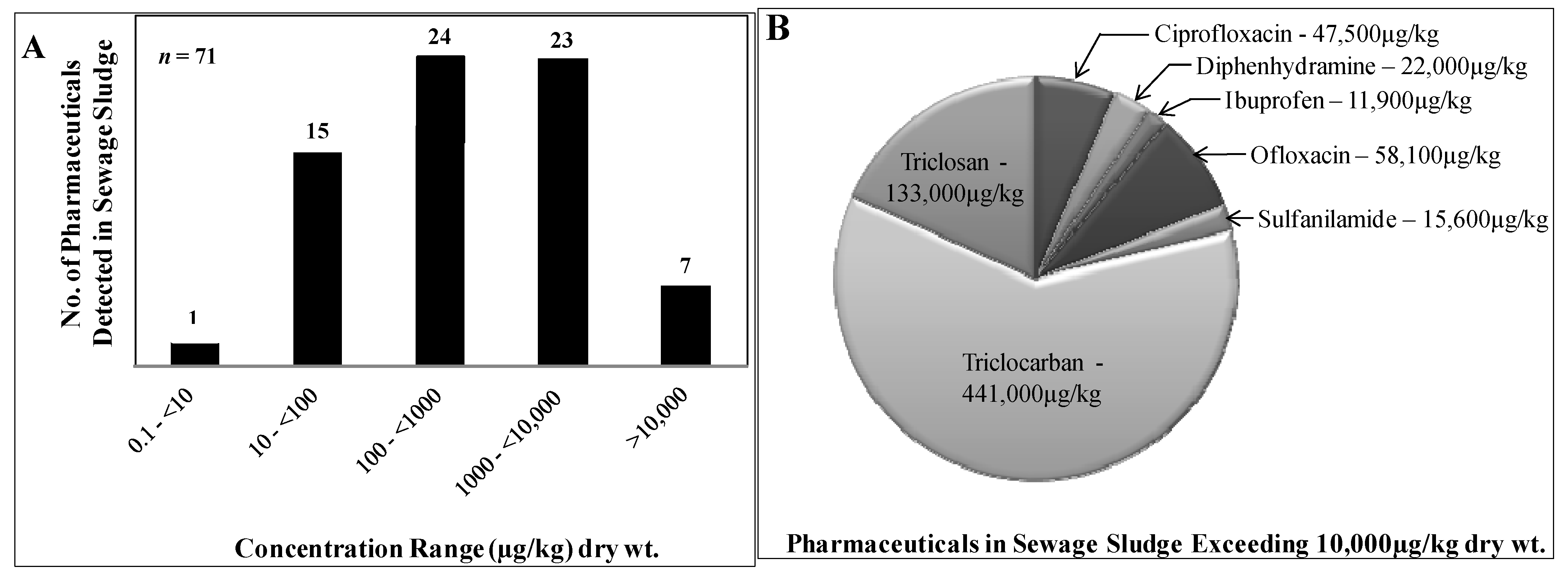
2.3. Pharmaceuticals in Municipal Sludge
3. Sustainable Management of Pharmaceuticals
4. Summary and Conclusions
- Long-term effects of low-level pharmaceutical contamination on human health;
- Short- and long-term effects of pharmaceutical contamination on non-target organisms;
- Comprehensive evaluations of treatment works (DWTPs and WWTPs) to identify infrastructure that improves the removal efficiencies of pharmaceuticals, and minimizes associated costs;
- Enhanced monitoring of pharmaceuticals in the built water environment to facilitate accurate evaluation of removal efficiencies of treatment works and to identify all possible avenues by which pharmaceuticals enter the natural water environment (including sewer overflows, leachates from solid waste, and biosolids runoff);
- More targeted monitoring of pharmaceuticals that are toxic to indicator organisms, are produced in high-volume, and possess persistent physico-chemical properties (i.e., long environmental half-life);
- Design of pharmaceuticals that are susceptible to transformation (or degradation) by treatment works and/or natural processes (e.g., photolysis);
- Identification and elimination of high-strength wastewaters from the pharmaceutical industry;
- Partnerships between academic institutions, pharmaceutical industries, and government to promote public education for proper use and disposal of pharmaceuticals.
Acknowledgments
Conflicts of Interest
References
- Ruhoy, I.S.; Daughton, C.G. Beyond the medicine cabinet: An analysis of where and why medications accumulate. Environ. Int. 2008, 34, 1157–1169. [Google Scholar] [CrossRef]
- Daughton, C.G.; Ternes, T.A. Pharmaceuticals and personal care products in the environment: Agents of subtle change? Environ. Health Perspect. 1999, 107, 907–938. [Google Scholar] [CrossRef]
- Dong, Z.; Senn, D.B.; Moran, R.E.; Shine, J.P. Prioritizing environmental risk of prescription pharmaceuticals. Regul. Toxicol. Pharmacol. 2013, 65, 60–67. [Google Scholar] [CrossRef]
- Kummerer, K. The presence of pharmaceuticals in the environment due to human use—Present knowledge and future challenges. J. Environ. Manag. 2009, 90, 2354–2366. [Google Scholar] [CrossRef]
- Kummerer, K. Pharmaceuticals in the Environment. In Annual Review of Environment and Resources; Gadgil, A., Liverman, D.M., Eds.; Annual Reviews: Palo Alto, CA, USA, 2010; pp. 57–75. [Google Scholar]
- Boxall, A.B.A.; Rudd, M.A.; Brooks, B.W.; Caldwell, D.J.; Choi, K.; Hickmann, S.; Innes, E.; Ostapyk, K.; Staveley, J.P.; Verslycke, T.; et al. Pharmaceuticals and Personal Care Products in the Environment: What Are the Big Questions? Environ. Health Perspect. 2012, 120, 1221–1229. [Google Scholar] [CrossRef]
- Monteiro, S.C.; Boxall, A.B.A. Occurrence and fate of human pharmceuticals in the environment. In Reviews of Environmental Contamination and Toxicology; Whitacre, D.M., Ed.; Springer: Summerfield, NC, USA, 2010; pp. 53–154. [Google Scholar]
- Karnjanapiboonwong, A.; Suski, J.G.; Shah, A.A.; Cai, Q.S.; Morse, A.N.; Anderson, T.A. Occurrence of PPCPs at a Wastewater Treatment Plant and in Soil and Groundwater at a Land Application Site. Water Air Soil Pollut. 2011, 216, 257–273. [Google Scholar] [CrossRef]
- Loganathan, B.; Phillips, M.; Mowery, H.; Jones-Lepp, T.L. Contamination profiles and mass loadings of macrolide antibiotics and illicit drugs from a small urban wastewater treatment plant. Chemosphere 2009, 75, 70–77. [Google Scholar] [CrossRef]
- Wilson, B.; Chen, R.F.; Cantwell, M.; Gontz, A.; Zhu, J.; Olsen, C.R. The partitioning of triclosan between aqueous and particulate bound phases in the Hudson River Estuary. Mar. Pollut. Bull. 2009, 59, 207–212. [Google Scholar] [CrossRef]
- Katz, B.G.; Griffin, D.W.; Davis, J.H. Groundwater quality impacts from the land application of treated municipal wastewater in a large karstic spring basin: Chemical and microbiological indicators. Sci. Total Environ. 2009, 407, 2872–2886. [Google Scholar] [CrossRef]
- Yu, C.P.; Chu, K.H. Occurrence of pharmaceuticals and personal care products along the West Prong Little Pigeon River in east Tennessee, USA. Chemosphere 2009, 75, 1281–1286. [Google Scholar] [CrossRef]
- Guo, Y.C.; Krasner, S.W. Occurrence of primidone, carbamazepine, caffeine, and precursors for n-nitrosodimethylamine in drinking water sources impacted by wastewater. J. Am. Water Resour. Assoc. 2009, 45, 58–67. [Google Scholar] [CrossRef]
- Benotti, M.J.; Trenholm, R.A.; Vanderford, B.J.; Holady, J.C.; Stanford, B.D.; Snyder, S.A. Pharmaceuticals and endocrine disrupting compounds in US drinking water. Environ. Sci. Technol. 2009, 43, 597–603. [Google Scholar] [CrossRef]
- Daughton, C.G.; Ruhoy, I.S. The afterlife of drugs and the role of PharmEcovigilance. Drug Saf. 2008, 31, 1069–1082. [Google Scholar]
- Daughton, C.G. Pharmaceutical ingredients in drinking water: Overview of occurrence and significance of human health considerations. In Contaminants of Emerging Concern in the Environment: Ecological and Human Health Considerations; Rolf, U.H., Ed.; American Chemical Society: Washington, DC, USA, 2010; pp. 9–68. [Google Scholar]
- Snyder, A.S. Occurrence of pharmaceuticals in U.S. drinking water. In Contaminants of Emerging Concern in the Environment: Ecological and Human Health Considerations; Rolf, U.H., Ed.; American Chemical Society: Washington, DC, USA, 2010; pp. 69–80. [Google Scholar]
- Aydin, E.; Talinli, I. Analysis, occurrence and fate of commonly used pharmaceuticals and hormones in the Buyukcekmece Watershed, Turkey. Chemosphere 2013, 90, 2004–2012. [Google Scholar] [CrossRef]
- Brodin, T.; Fick, J.; Jonsson, M.; Klaminder, J. Dilute concentrations of a psychiatric drug alter behavior of fish from natural populations. Science 2013, 339, 814–815. [Google Scholar] [CrossRef]
- Fair, P.A.; Lee, H.B.; Adams, J.; Darling, C.; Pacepavicius, G.; Alaee, M.; Bossart, G.D.; Henry, N.; Muir, D. Occurrence of triclosan in plasma of wild Atlantic bottlenose dolphins (Tursiops truncatus) and in their environment. Environ. Pollut. 2009, 157, 2248–2254. [Google Scholar] [CrossRef]
- Owen, S.F.; Huggett, D.B.; Hutchinson, T.H.; Hetheridge, M.J.; Kinter, L.B.; Ericson, J.F.; Sumpter, J.P. Uptake of propranolol, a cardiovascular pharmaceutical, from water into fish plasma and its effects on growth and organ biometry. Aquati. Toxicol. 2009, 93, 217–224. [Google Scholar] [CrossRef]
- Christenson, T. Fish on morphine: Protecting Wisconsin’s natural resources through a comprehensive plan for proper disposal of pharmaceuticals. Wiscon. Law Rev. 2008, 141–179. [Google Scholar]
- Kwon, J.W.; Armbrust, K.L. Laboratory persistence and fate of fluoxetine in aquatic environments. Environ. Toxicol. Chem. 2006, 25, 2561–2568. [Google Scholar] [CrossRef]
- Mearns, A.J.; Reish, D.J.; Oshida, P.S.; Buchman, M.; Ginn, T.; Donnelly, R. Effects of pollution on marine organisms. Water Environ. Res. 2009, 81, 2070–2125. [Google Scholar] [CrossRef]
- Brozinski, J.-M.; Lahti, M.; Meierjohann, A.; Oikari, A.; Kronberg, L. The anti-inflammatory drugs diclofenac, naproxen and ibuprofen are found in the bile of wild fish caught downstream of a wastewater treatment plant. Environ. Sci. Technol. 2013, 47, 342–348. [Google Scholar] [CrossRef]
- Cecchini, M.; LoPresti, V. Drug residues store in the body following cessation of use: Impacts on neuroendocrine balance and behavior—Use of the Hubbard sauna regimen to remove toxins and restore health. Med. Hypotheses 2007, 68, 868–879. [Google Scholar] [CrossRef]
- Rudel, R.A.; Attfield, K.R.; Schifano, J.N.; Brody, J.G. Chemicals causing mammary gland tumors in animals signal new directions for epidemiology, chemicals testing, and risk assessment for breast cancer prevention. Cancer 2007, 109, 2635–2666. [Google Scholar] [CrossRef]
- Cunningham, V.L.; Binks, S.P.; Olson, M.J. Human health risk assessment from the presence of human pharmaceuticals in the aquatic environment. Regul. Toxicol. Pharmacolog. 2009, 53, 39–45. [Google Scholar] [CrossRef]
- Fent, K.; Weston, A.A.; Caminada, D. Ecotoxicology of human pharmaceuticals. Aquat. Toxicol. 2006, 76, 122–159. [Google Scholar] [CrossRef]
- Richardson, M.L.; Bowron, J.M. The fate of pharmaceutical chemicals in the aquatic environment. J. Pharm. Pharmacolog. 1985, 37, 1–12. [Google Scholar] [CrossRef]
- Daughton, C.G.; Brooks, B.W. Active pharmaceutical ingredients and aquatic organisms. In Environmental Contaminants in Biota: Interpreting Tissue Concentrations, 2nd ed.; Meador, W.B.J., Ed.; Taylor and Francis: Boca Raton, FL, USA, 2011; pp. 281–340. [Google Scholar]
- Le-Minh, N.; Khan, S.J.; Drewes, J.E.; Stuetz, R.M. Fate of antibiotics during municipal water recycling treatment processes. Water Res. 2010, 44, 4295–4323. [Google Scholar] [CrossRef]
- Ternes, T.A.; Joss, A.; Siegrist, H. Scrutinizing pharmaceuticals and personal care products in wastewater treatment. Environ. Sci. Technol. 2004, 38, 392A–399A. [Google Scholar] [CrossRef]
- Petrovic, M.; de Alda, M.J.L.; Diaz-Cruz, S.; Postigo, C.; Radjenovic, J.; Gros, M.; Barcelo, D. Fate and removal of pharmaceuticals and illicit drugs in conventional and membrane bioreactor wastewater treatment plants and by riverbank filtration. Philos. Trans. R. Soc. A 2009, 367, 3979–4003. [Google Scholar] [CrossRef]
- Jones, O.A.H.; Voulvoulis, N.; Lester, J.N. Human pharmaceuticals in wastewater treatment processes. Crit. Rev. Environ. Sci. Technol. 2005, 35, 401–427. [Google Scholar] [CrossRef]
- Onesios, K.M.; Yu, J.T.; Bouwer, E.J. Biodegradation and removal of pharmaceuticals and personal care products in treatment systems: A review. Biodegradation 2009, 20, 441–466. [Google Scholar] [CrossRef]
- Snyder, S.A. Occurrence, treatment, and toxicological relevance of EDCs and pharmaceuticals in water. Ozone Sci. Eng. 2008, 30, 65–69. [Google Scholar] [CrossRef]
- Ikehata, K.; Naghashkar, N.J.; Ei-Din, M.G. Degradation of aqueous pharmaceuticals by ozonation and advanced oxidation processes: A review. Ozone Sci. Eng. 2006, 28, 353–414. [Google Scholar] [CrossRef]
- Yargeau, V.; Leclair, C. Impact of operating conditions on decomposition of antibiotics during ozonation: A review. Ozone Sci. Eng. 2008, 30, 175–188. [Google Scholar] [CrossRef]
- Werner, J.J.; McNeill, K.; Arnold, W.A. Photolysis of chlortetracycline on a clay surface. J. Agric. Food Chem. 2009, 57, 6932–6937. [Google Scholar] [CrossRef]
- Santoke, H.; Song, W.H.; Cooper, W.J.; Greaves, J.; Miller, G.E. Free-radical-induced oxidative and reductive degradation of fluoroquinolone pharmaceuticals: kinetic studies and degradation mechanism. J. Phys. Chem. A 2009, 113, 7846–7851. [Google Scholar] [CrossRef]
- Hu, L.; Martin, H.M.; Arcs-Bulted, O.; Sugihara, M.N.; Keatlng, K.A.; Strathmann, T.J. Oxidation of carbamazepine by Mn(VII) and Fe(VI): Reaction kinetics and mechanism. Environ. Sci. Technol. 2009, 43, 509–515. [Google Scholar]
- Leech, D.M.; Snyder, M.T.; Wetzel, R.G. Natural organic matter and sunlight accelerate the degradation of 17 beta-estradiol in water. Sci. Total Environ. 2009, 407, 2087–2092. [Google Scholar] [CrossRef]
- Benotti, M.J.; Brownawell, B.J. Microbial degradation of pharmaceuticals in estuarine and coastal seawater. Environ. Pollut. 2009, 157, 994–1002. [Google Scholar] [CrossRef]
- Yu, T.H.; Lin, A.Y.C.; Lateef, S.K.; Lin, C.F.; Yang, P.Y. Removal of antibiotics and non-steroidal anti-inflammatory drugs by extended sludge age biological process. Chemosphere 2009, 77, 175–181. [Google Scholar] [CrossRef]
- Wu, C.X.; Spongberg, A.L.; Witter, J.D. Sorption and biodegradation of selected antibiotics in biosolids. J. Environ. Sci. Health Tox. Hazard. Subst. Environ. Eng. 2009, 44, 454–461. [Google Scholar] [CrossRef]
- Wu, C.X.; Spongberg, A.L.; Witter, J.D. Adsorption and degradation of triclosan and triclocarban in solis and biosolids-amended soils. J. Agric. Food Chem. 2009, 57, 4900–4905. [Google Scholar] [CrossRef]
- Chang, P.H.; Li, Z.H.; Yu, T.L.; Munkhbayer, S.; Kuo, T.H.; Hung, Y.C.; Jean, J.S.; Lin, K.H. Sorptive removal of tetracycline from water by palygorskite. J. Hazard. Mater. 2009, 165, 148–155. [Google Scholar] [CrossRef]
- Wilcox, J.D.; Bahr, J.M.; Hedman, C.J.; Hemming, J.D.C.; Barman, M.A.E.; Bradbury, K.R. Removal of organic wastewater contaminants in septic systems using advanced treatment technologies. J. Environ. Qual. 2009, 38, 149–156. [Google Scholar] [CrossRef]
- Phillips, P.J.; Smith, S.G.; Kolpin, D.W.; Zaugg, S.D.; Buxton, H.T.; Furlong, E.T.; Esposito, K.; Stinson, B. Pharmaceutical formulation facilities as sources of opioids and other pharmaceuticals to wastewater treatment plant effluents. Environ. Sci. Technol. 2010, 44, 4910–4916. [Google Scholar] [CrossRef]
- Nagarnaik, P.M.; Batt, A.L.; Boulanger, B. Healthcare facility effluents as point sources of select pharmaceuticals to municipal wastewater. Water Environ. Res. 2012, 84, 339–345. [Google Scholar]
- Daughton, C.G.; Ruhoy, I.S. Environmental footprint of pharmaceuticals: The significance of factors beyond direct excretion to sewers. Environ. Toxicol. Chem. 2009, 28, 2495–2521. [Google Scholar] [CrossRef]
- Ruhoy, I.S.; Daughton, C.G. Types and quantities of leftover drugs entering the environment via disposal to sewage—Revealed by coroner records. Sci. Total Environ. 2007, 388, 137–148. [Google Scholar] [CrossRef]
- Glassmeyer, S.T.; Hinchey, E.K.; Boehme, S.E.; Daughton, C.G.; Ruhoy, I.S.; Conerly, O.; Daniels, R.L.; Lauer, L.; McCarthy, M.; Nettesheim, T.G.; et al. Disposal practices for unwanted residential medications in the United States. Environ. Int. 2009, 35, 566–572. [Google Scholar] [CrossRef]
- Seehusen, D.A.; Edwards, J. Patient practices and beliefs concerning disposal of medications. J. Am. Board Fam. Med. 2006, 19, 542–547. [Google Scholar] [CrossRef]
- Kotchen, M.; Kallaos, J.; Wheeler, K.; Wong, C.; Zahller, M. Pharmaceuticals in wastewater: Behavior, preferences, and willingness to pay for a disposal program. J. Environ. Manag. 2009, 90, 1476–1482. [Google Scholar] [CrossRef]
- USEPA. Summary of the Clean Water Act. Available online: http://www2.epa.gov/laws-regulations/summary-clean-water-act (accessed on 1 August 2013).
- Heidler, J.; Halden, R.U. Meta-analysis of mass balances examining chemical fate during wastewater treatment. Environ. Sci. Technol. 2008, 42, 6324–6332. [Google Scholar] [CrossRef]
- McClellan, K.; Halden, R.U. Pharmaceuticals and personal care products in archived US biosolids from the 2001 EPA national sewage sludge survey. Water Res. 2010, 44, 658–668. [Google Scholar] [CrossRef]
- Fick, J.; Soderstrom, H.; Lindberg, R.H.; Phan, C.; Tysklind, M.; Larsson, D.G.J. Contamination of surface, ground, and drinking water from pharmaceutical production. Environ. Toxicol. Chem. 2009, 28, 2522–2527. [Google Scholar] [CrossRef]
- Larsson, D.G.J.; de Pedro, C.; Paxeus, N. Effluent from drug manufactures contains extremely high levels of pharmaceuticals. J. Hazard. Mater. 2007, 148, 751–755. [Google Scholar] [CrossRef]
- Edwards, M.; Topp, E.; Metcalfe, C.D.; Li, H.; Gottschall, N.; Bolton, P.; Curnoe, W.; Payne, M.; Beck, A.; Kleywegt, S.; et al. Pharmaceutical and personal care products in tile drainage following surface spreading and injection of dewatered municipal biosolids to an agricultural field. Sci. Total Environ. 2009, 407, 4220–4230. [Google Scholar] [CrossRef]
- Lapen, D.R.; Topp, E.; Metcalfe, C.D.; Li, H.; Edwards, M.; Gottschall, N.; Bolton, P.; Curnoe, W.; Payne, M.; Beck, A. Pharmaceutical and personal care products in tile drainage following land application of municipal biosolids. Sci. Total Environ. 2008, 399, 50–65. [Google Scholar] [CrossRef]
- Lin, A.Y.C.; Yu, T.H.; Lin, C.F. Pharmaceutical contamination in residential, industrial, and agricultural waste streams: Risk to aqueous environments in Taiwan. Chemosphere 2008, 74, 131–141. [Google Scholar] [CrossRef]
- Phillips, P.; Chalmers, A. Wastewater effluent, combined sewer overflows, and other sources of organic compounds to Lake Champlain. J. Am. Water Resour. Assoc. 2009, 45, 45–57. [Google Scholar] [CrossRef]
- Shala, L.; Foster, G.D. Surface water concentrations and loading budgets of pharmaceuticals and other domestic-use chemicals in an urban watershed (Washington, DC, USA). Arch. Environ. Contam. Toxicol. 2010, 58, 551–561. [Google Scholar] [CrossRef]
- Halden, R.U.; Paull, D.H. Co-occurrence of triclocarban and triclosan in US water resources. Environ. Sci. Technol. 2005, 39, 1420–1426. [Google Scholar] [CrossRef]
- Fram, M.S.; Belitz, K. Occurrence and concentrations of pharmaceutical compounds in groundwater used for public drinking-water supply in California. Sci. Total Environ. 2011, 409, 3409–3417. [Google Scholar] [CrossRef]
- Deo, R.P.; Halden, R.U. Effect of sample filtration on the quality of monitoring data reported for organic compounds during wastewater treatment. J. Environ. Monit. 2010, 12, 478–483. [Google Scholar] [CrossRef]
- Sunkara, M.; Wells, M.J.M. Phase II pharmaceutical metabolites acetaminophen glucuronide and acetaminophen sulfate in wastewater. Environ. Chem. 2009, 7, 111–122. [Google Scholar] [CrossRef]
- Jones, O.A.; Lester, J.N.; Voulvoulis, N. Pharmaceuticals: A threat to drinking water? Trends Biotechnol. 2005, 23, 163–167. [Google Scholar] [CrossRef]
- Yu, Y.; Wu, L.; Chang, A.C. Seasonal variation of endocrine disrupting compounds, pharmaceuticals and personal care products in wastewater treatment plants. Sci. Total Environ. 2013, 442, 310–316. [Google Scholar] [CrossRef]
- Wilde, M.L.; Kummerer, K.; Martins, A.F. Multivariate optimization of analytical methodology and a first attempt to an environmental risk assessment of beta-blockers in hospital wastewater. J. Braz. Chem. Soc. 2012, 23, 1732–1740. [Google Scholar] [CrossRef]
- Ferrari, B.; Paxeus, N.; Lo Giudice, R.; Pollio, A.; Garric, J. Ecotoxicological impact of pharmaceuticals found in treated wastewaters: Study of carbamazepine, clofibric acid, and diclofenac. Ecotoxicol. Environ. Saf. 2003, 55, 359–370. [Google Scholar] [CrossRef]
- Vazquez-Roig, P.; Andreu, V.; Onghena, M.; Blasco, C.; Pico, Y. Assessment of the occurrence and distribution of pharmaceuticals in a Mediterranean wetland (L’Albufera, Valencia, Spain) by LC-MS/MS. Anal. Bioanal. Chem. 2011, 400, 1287–1301. [Google Scholar] [CrossRef]
- Carlsson, C.; Johansson, A.K.; Alvan, G.; Bergman, K.; Kuhler, T. Are pharmaceuticals potent environmental pollutants? Part II: Environmental risk assessments of selected pharmaceutical excipients. Sci. Total Environ. 2006, 364, 88–95. [Google Scholar] [CrossRef]
- Agerstrand, M.; Ruden, C. Evaluation of the accuracy and consistency of the Swedish environmental classification and information system for pharmaceuticals. Sci. Total Environ. 2010, 408, 2327–2339. [Google Scholar] [CrossRef]
- Zheng, Q.; Zhang, R.J.; Wang, Y.H.; Pan, X.H.; Tang, J.H.; Zhang, G. Occurrence and distribution of antibiotics in the Beibu Gulf, China: Impacts of river discharge and aquaculture activities. Mar. Environ. Res. 2012, 78, 26–33. [Google Scholar] [CrossRef]
- Miller, K.J.; Meek, J. Helena Valley Ground Water: Pharmaceuticals, Personal Care Products, Endocrine Disruptors (PPCPs) and Microbial Indicators of Faecal Contamination; Montana Department of Environmental Quality: Helena, MT, USA, 2006. [Google Scholar]
- Barnes, K.K.; Kolpin, D.W.; Furlong, E.T.; Zaugg, S.D.; Meyer, M.T.; Barber, L.B. A national reconnaissance of pharmaceuticals and other organic wastewater contaminants in the United States—I) Groundwater. Sci. Total Environ. 2008, 402, 192–200. [Google Scholar] [CrossRef]
- Bartelt-Hunt, S.; Snow, D.D.; Damon-Powell, T.; Miesbach, D. Occurrence of steroid hormones and antibiotics in shallow groundwater impacted by livestock waste control facilities. J. Contam. Hydrol. 2011, 123, 94–103. [Google Scholar] [CrossRef]
- Fang, Y.; Karnjanapiboonwong, A.; Chase, D.A.; Wang, J.F.; Morse, A.N.; Anderson, T.A. Occurrence, fate, and persistence of gemfibrozil in water and soil. Environ. Toxicol. Chem. 2012, 31, 550–555. [Google Scholar] [CrossRef]
- Watanabe, N.; Bergamaschi, B.A.; Loftin, K.A.; Meyer, M.T.; Harter, T. Use and environmental occurrence of antibiotics in freestall dairy farms with manured forage fields. Environ. Sci. Technol. 2010, 44, 6591–6600. [Google Scholar] [CrossRef]
- Mackie, R.I.; Koike, S.; Krapac, I.; Chee-Sanford, J.; Maxwell, S.; Aminov, R.I. Tetracycline residues and tetracycline resistance genes in groundwater impacted by swine production facilities. Anim. Biotechnol. 2006, 17, 157–176. [Google Scholar] [CrossRef]
- Zhao, S.; Zhang, P.F.; Crusius, J.; Kroeger, K.D.; Bratton, J.F. Use of pharmaceuticals and pesticides to constrain nutrient sources in coastal groundwater of northwestern Long Island, New York, USA. J. Environ. Monit. 2011, 13, 1337–1343. [Google Scholar] [CrossRef]
- Wang, C.A.; Shi, H.L.; Adams, C.D.; Gamagedara, S.; Stayton, I.; Timmons, T.; Ma, Y.F. Investigation of pharmaceuticals in Missouri natural and drinking water using high performance liquid chromatography-tandem mass spectrometry. Water Res. 2011, 45, 1818–1828. [Google Scholar] [CrossRef]
- Stackelberg, P.E.; Furlong, E.T.; Meyer, M.T.; Zaugg, S.D.; Henderson, A.K.; Reissman, D.B. Persistence of pharmaceutical compounds and other organic wastewater contaminants in a conventional drinking-watertreatment plant. Sci. Total Environ. 2004, 329, 99–113. [Google Scholar] [CrossRef]
- Stackelberg, P.E.; Gibs, J.; Furlong, E.T.; Meyer, M.T.; Zaugg, S.D.; Lippincott, R.L. Efficiency of conventional drinking-water-treatment processes in removal of pharmaceuticals and other organic compounds. Sci. Total Environ. 2007, 377, 255–272. [Google Scholar] [CrossRef]
- Snyder, S.A.; Adham, S.; Redding, A.M.; Cannon, F.S.; DeCarolis, J.; Oppenheimer, J.; Wert, E.C.; Yoon, Y. Role of membranes and activated carbon in the removal of endocrine disruptors and pharmaceuticals. Desalination 2007, 202, 156–181. [Google Scholar] [CrossRef]
- Kim, S.D.; Cho, J.; Kim, I.S.; Vanderford, B.J.; Snyder, S.A. Occurrence and removal of pharmaceuticals and endocrine disruptors in South Korean surface, drinking, and waste waters. Water Res. 2007, 41, 1013–1021. [Google Scholar] [CrossRef]
- Loraine, G.A.; Pettigrove, M.E. Seasonal variations in concentrations of pharmaceuticals and personal care products in drinking water and reclaimed wastewater in Southern California. Environ. Sci. Technol. 2006, 40, 687–695. [Google Scholar] [CrossRef]
- Trenholm, R.A.; Vanderford, B.J.; Snyder, S.A. On-line solid phase extraction LC-MS/MS analysis of pharmaceutical indicators in water: A green alternative to conventional methods. Talanta 2009, 79, 1425–1432. [Google Scholar] [CrossRef]
- PBT Profiler software. Available online: http://www.pbtprofiler.net (accessed on 1 August 2013).
- Hernando, M.D.; Mezcua, M.; Fernandez-Alba, A.R.; Barcelo, D. Environmental risk assessment of pharmaceutical residues in wastewater effluents, surface waters and sediments. Talanta 2006, 69, 334–342. [Google Scholar] [CrossRef]
- Carlsson, C.; Johansson, A.K.; Alvan, G.; Bergman, K.; Kuhler, T. Are pharmaceuticals potent environmental pollutants? Part I: Environmental risk assessments of selected active pharmaceutical ingredients. Sci. Total Environ. 2006, 364, 67–87. [Google Scholar] [CrossRef]
- Hernando, M.D.; Gomez, M.J.; Aguera, A.; Fernandez-Alba, A.R. LC-MS analysis of basic pharmaceuticals (beta-blockers and anti-ulcer agents) in wastewater and surface water. Trac Trends Anal. Chem. 2007, 26, 581–594. [Google Scholar] [CrossRef]
- Clarke, B.O.; Smith, S.R. Review of ‘emerging’ organic contaminants in biosolids and assessment of international research priorities for the agricultural use of biosolids. Environ. Int. 2011, 37, 226–247. [Google Scholar] [CrossRef]
- Clarke, B.O.; Porter, N.A. Persistent organic pollutants in sewage sludge: Levels, sources, and trends. In Contaminants of Emerging Concern in the Environment: Ecological and Human Health Considerations; Rolf, U.H., Ed.; American Chemical Society: Washington, DC, USA, 2010; pp. 137–171. [Google Scholar]
- Harrison, E.Z.; Oakes, S.R.; Hysell, M.; Hay, A. Organic chemicals in sewage sludges. Sci. Total Environ. 2006, 367, 481–497. [Google Scholar] [CrossRef]
- Rogers, H.R. Sources, behaviour and fate of organic contaminants during sewage treatment and in sewage sludges. Sci. Total Environ. 1996, 185, 3–26. [Google Scholar] [CrossRef]
- New England Bicycle Racing Association, A National Biosolids Regulation, Quality, End Use & Disposal Survey; Final Report; New England Bicycle Racing Association: Tamworth, NH, UK, 2007.
- Xu, J.; Chen, W.P.; Wu, L.S.; Green, R.; Chang, A.C. Leachability of some emerging contaminants in reclaimed municipal wastewater-irrigated turf grass fields. Environ. Toxicol. Chem. 2009, 28, 1842–1850. [Google Scholar] [CrossRef]
- Xia, K.; Hundal, L.S.; Kumar, K.; Armbrust, K.; Cox, A.E.; Granato, T.C. Triclocarban, triclosan, polybrominated diphenyl ethers, and 4-nonylphenol in biosolids and in soil receiving 33-year biosolids application. Environ. Toxicol. Chem. 2010, 29, 597–605. [Google Scholar] [CrossRef]
- Smith, S.R. Organic contaminants in sewage sludge (biosolids) and their significance for agricultural recycling. Philos. Trans. R. Soc. A 2009, 367, 4005–4041. [Google Scholar] [CrossRef]
- Wu, C.X.; Spongberg, A.L.; Witter, J.D.; Fang, M.; Ames, A.; Czajkowski, K.P. Detection of Pharmaceuticals and Personal Care Products in Agricultural Soils Receiving Biosolids Application. Clean Soil Air Water 2010, 38, 230–237. [Google Scholar] [CrossRef]
- Dolliver, H.; Kumar, K.; Gupta, S. Sulfamethazine uptake by plants from manure-amended soil. J. Environ. Qual. 2007, 36, 1224–1230. [Google Scholar] [CrossRef]
- Kumar, K.; Gupta, S.C.; Baidoo, S.K.; Chander, Y.; Rosen, C.J. Antibiotic uptake by plants from soil fertilized with animal manure. J. Environ. Qual. 2005, 34, 2082–2085. [Google Scholar] [CrossRef]
- Holling, C.S.; Bailey, J.L.; Heuvel, B.V.; Kinney, C.A. Uptake of human pharmaceuticals and personal care products by cabbage (Brassica campestris) from fortified and biosolids-amended soils. J. Environ. Monit. 2012, 14, 3029–3036. [Google Scholar] [CrossRef]
- Klavarioti, M.; Mantzavinos, D.; Kassinos, D. Removal of residual pharmaceuticals from aqueous systems by advanced oxidation processes. Environ. Int. 2009, 35, 402–417. [Google Scholar] [CrossRef]
- Sharma, V.K. Oxidative transformations of environmental pharmaceuticals by Cl-2, ClO2, O-3, and Fe(VI): Kinetics assessment. Chemosphere 2008, 73, 1379–1386. [Google Scholar] [CrossRef]
- Ikehata, K.; Gamal El-Din, M.; Snyder, S.A. Ozonation and advanced oxidation treatment of emerging organic pollutants in water and wastewater. Ozone Sci. Eng. J. Int. Ozone Assoc. 2008, 30, 21–26. [Google Scholar] [CrossRef]
- Esplugas, S.; Bila, D.M.; Krause, L.G.T.; Dezotti, M. Ozonation and advanced oxidation technologies to remove endocrine disrupting chemicals (EDCs) and pharmaceuticals and personal care products (PPCPs) in water effluents. J. Hazard. Mater. 2007, 149, 631–642. [Google Scholar] [CrossRef]
- Ellis, W.C.; Tran, C.T.; Roy, R.; Rusten, M.; Fischer, A.; Ryabov, A.D.; Blumberg, B.; Collins, T.J. Designing green oxidation catalysts for purifying environmental waters. J. Am. Chem. Soc. 2010, 132, 9774–9781. [Google Scholar]
- Daughton, C.G. Cradle-to-cradle stewardship of drugs for minimizing their environmental disposition while promoting human health. II. Drug disposal, waste reduction, and future directions. Environ. Health Perspect. 2003, 111, 775–785. [Google Scholar] [CrossRef]
- Kummerer, J.; Hempel, M. Green and Sustainable Pharmacy, 1st ed.; Springer-Verlag: Berlin Heidelberg, Germany, 2010. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Deo, R.P.; Halden, R.U. Pharmaceuticals in the Built and Natural Water Environment of the United States. Water 2013, 5, 1346-1365. https://doi.org/10.3390/w5031346
Deo RP, Halden RU. Pharmaceuticals in the Built and Natural Water Environment of the United States. Water. 2013; 5(3):1346-1365. https://doi.org/10.3390/w5031346
Chicago/Turabian StyleDeo, Randhir P., and Rolf U. Halden. 2013. "Pharmaceuticals in the Built and Natural Water Environment of the United States" Water 5, no. 3: 1346-1365. https://doi.org/10.3390/w5031346




