Biological Complexities in Radiation Carcinogenesis and Cancer Radiotherapy: Impact of New Biological Paradigms
Abstract
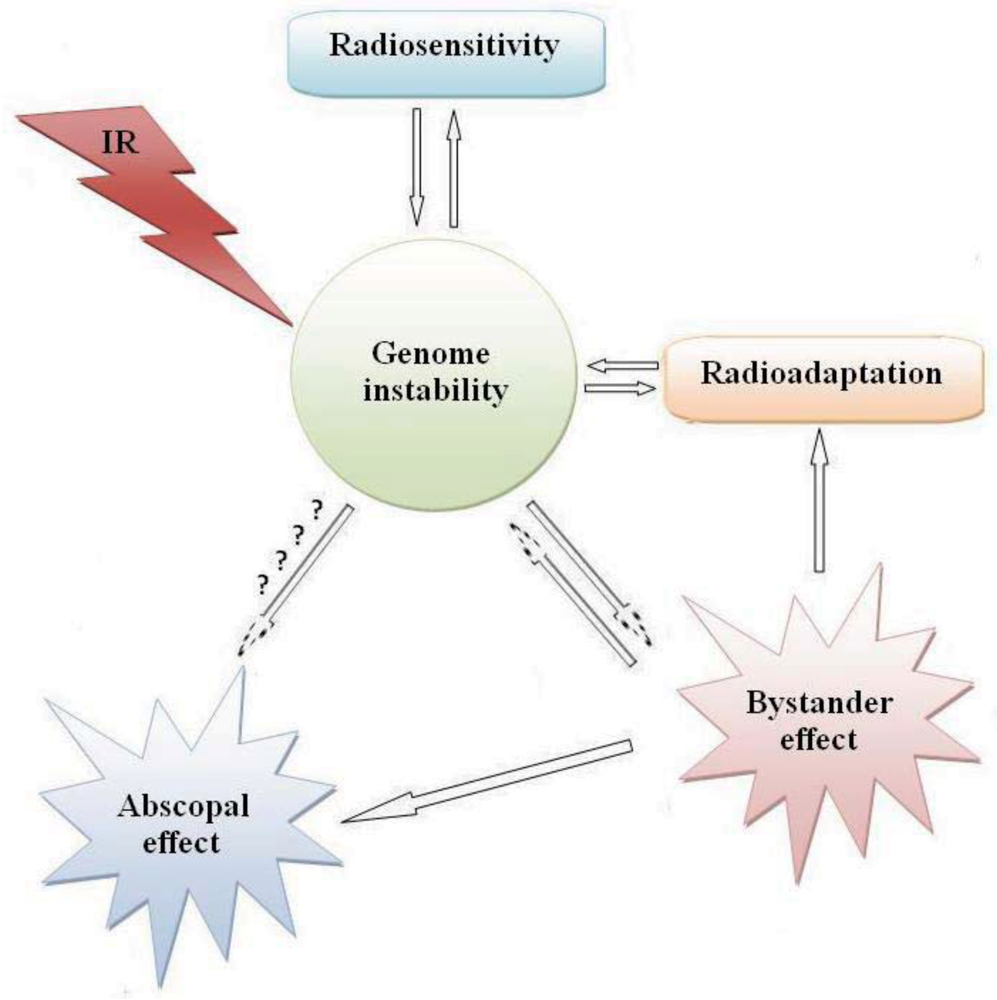
:1. Introduction
2. Radiation Carcinogenesis
3. Impact of Radiosensitivity on Carcinogenesis and Cancer Therapy
4. Radioadaptation and Carcinogenesis
5. Bystander Effect and Carcinogenesis
6. Distant Bystander (Abscopal) Effect
7. Conclusions
References
- Upton, A.C. Historical perspective on radiation carcinogenesis. In Radiation Carcinogenesis; Upton, A.C., Alber, R.E., Burns, F.J., Shore, R.E., Eds.; Elsevier: New York, NY, USA, 1986; pp. 1–10. [Google Scholar]
- Jacob, P.; Bogdanova, T.I.; Buglova, E.; Chepurniy, M.; Demidchik, Y.; Gavrilin, Y.; Kenigsberg, J.; Meckbach, R.; Schotola, C.; Shinkarev, S.; et al. Thyroid cancer risk in areas of Ukraine and Belarus affected by the Chernobyl accident. Radiat. Res. 2006, 165, 1–8. [Google Scholar] [CrossRef]
- National Research Council, Health Effects of Exposure to Low Levels of Ionizing Radiation, BEIR V; National Academy Press: Washington, DC, USA, 1990.
- Shadley, J.D.; Wolff, S. Very low doses of X-rays can cause human lymphocytes to become less susceptible to ionizing radiation. Mutagenesis 1987, 2, 95–96. [Google Scholar]
- Wang, Z.; Saigusa, S.; Sasaki, M.S. Adaptive response to chromosome damage incultured human lymphocytes primed with low doses of X-rays. Mutat. Res. 1991, 246, 179–186. [Google Scholar]
- Mortazavi, S.M.J.; Mozdarani, H. The search for a possible optimum adapting dose under the optimum irradiation time scheme in cultured human lymphocytes. Int. J. Low Radiat. 2006, 3, 74–82. [Google Scholar]
- Morgan, W.F.; Sowa, M.B. Non-targeted bystander effects induced by ionizing radiation. Mutat. Res. 2007, 616, 159–164. [Google Scholar]
- Bowater, R.; Doherty, A.J. Making ends meet: Repairing breaks in bacterial DNA by non-homologous end-joining. PloS Genet. 2006, 2, e8. [Google Scholar]
- Iliakis, G.; Wu, W.; Wang, M.; Terzoudi, G.I.; Pantelias, G.E. Backup pathways of nonhomologous end joining may have a dominant role in the formation of chromosome aberrations. In Chromosmeal Alterations: Methods, Results and Importance in Human Health; Obe, G., Vijayalaxmi, Eds.; Springer-Verlag: Berlin, Germany, 2007; pp. 67–85. [Google Scholar]
- Obe, G.; Pfeiffer, P.; Savage, J.R.K.; Johannes, C.; Goedecke, W.; Jeppesen, P.; Natarajan, A.T.; Martinez-Lopez, W.; Folle, G.A.; Derts, M.E. Chromosomal aberrations: Formation, identification and distribution. Mutat. Res. 2002, 504, 17–36. [Google Scholar]
- Goedeccke, W. Double starnd break repair mechanisms in mammalian cells. In Chromoosmeal Alterations: Methods, Results and Importance in Human Health; Obe, G., Vijayalaxmi, Eds.; Springer-Verlag: Berlin, Germany, 2007; pp. 55–66. [Google Scholar]
- Heyer, W.D.; Li, X.; Rolfsmeier, M.; Zhang, X.P. Rad54: The Swiss Army Knife of homologous recombinations? Nucleic Acids Res. 2006, 34, 4115–4125. [Google Scholar]
- Ruis, B.L.; Fattah, K.R.; Hendrickson, E.A. The catalytic subunit of DNA-dependent protein kinase regulates proliferation, telomere length, and genomic stability in human somatic cells. Mol. Cell Biol. 2008, 28, 6182–6195. [Google Scholar]
- Fukasawa, K.; Wiener, F.; Vande Woude, G.F.; Mai, S. Genomic instability and apoptosis are frequent in p53 deficient young mice. Oncogene 1997, 15, 1295–1302. [Google Scholar]
- Brusa, G.; Benvenuti, M.; Mazzacurati, L.; Mancini, M.; Pattacini, L.; Martinelli, G.; Barbieri, E.; Greenberger, J.S.; Baccarani, M.; Santucci, M.A. p53 loss of function enhances genomic instability and accelerates clonal evolution of murine myeloid progenitors expressing the p(210)BCR-ABL tyrosine kinase. Haematologia 2003, 88, 622–630. [Google Scholar]
- Dimitrova, N.; de Lange, T. Cell cycle-dependent role of MRN at dysfunctional telomeres: ATM signaling-dependent induction of nonhomologous end joining (NHEJ) in G1 and resection-mediated inhibition of NHEJ in G2. Mol. Cell Biol. 2009, 29, 5552–5563. [Google Scholar]
- Greenwood, J.; Cooper, J.P. Trapping Rap1 at the telomere to prevent chromosome end fusions. EMBO J. 2009, 28, 3277–3278. [Google Scholar]
- Bailey, S.M.; Meyne, J.; Chen, D.J.; Kurimasa, A.; Li, G.C.; Lehnert, B.E.; Goodwin, E.H. DNA double-strand break repair proteins are required to cap the ends of mammalian chromosomes. Proc. Natl. Acad. Sci. USA 1999, 96, 14899–14904. [Google Scholar]
- Hsiao, S.J.; Smith, S. Sister telomeres rendered dysfunctional by persistent cohesion are fused by NHEJ. J. Cell Biol. 2009, 184, 515–526. [Google Scholar]
- Bender, M.A.; Griggs, H.G.; Bedford, J.S. Mechanisms of chromosomal aberration production: III. Chemical and ionizing radiation. Mutat. Res. 1974, 23, 197–212. [Google Scholar]
- Natarajan, A.T.; Obe, G.; van Zeeland, A.A.; Palitti, F.; Meijers, M.; Verdegaal-Immerzell, E.A.M. Molecular mechanisms involved in the production of chromosomal aberrations. II. Utilization of Neurospora endonuclease for the study of aberration production by X-rays in G1 and G2 stages of the cell cycle. Mutat. Res. 1980, 69, 293–305. [Google Scholar] [CrossRef]
- Mozdarani, H.; Bryant, P.E. The effects of 9-b-D arabinofuranosylanine on the formation of X-ray induced chromatid aberrations in X-irradiated G2 human cells. Mutagenesis 1987, 2, 371–374. [Google Scholar]
- Mozdarani, H.; Bryant, P.E. Induction and rejoining of chromatid breaks in X-irradiated A-T and normal human G2 fibroblasts. Int. J. Radiat. Biol. 1989, 56, 645–659. [Google Scholar]
- Kemp, L.M.; Jeggo, P.A. Radiation induced chromosome damage in X-ray sensitive mutants (xrs) of the Chinese hamster ovary cell line. Mutat. Res. DNA Repair Rep. 1986, 166, 255–263. [Google Scholar]
- Bryant, P.E.; Birch, D.A.; Jeggo, P.A. High chromosomal sensitivity of Chinese hamster xrs5 cells to restriction endonuclease induced DNA dsb. Int. J. Radiat. Biol. 1987, 52, 537–554. [Google Scholar]
- Bryant, P.E.; Finnegan, C.E.; Mozdarani, H. G2 chromatid breaks in murine scid cells. Mutagenesis 1998, 13, 481–485. [Google Scholar]
- Natarajan, A.T.; Obe, G. Molecular mechanisms involved in the production of chromosomal aberrations. III. Restriction endonucleases. Chromosoma 1984, 90, 120–127. [Google Scholar] [CrossRef]
- Bryant, P.E. Use of restriction endonucleases to study relationship between DNA double strand breaks, chromosomal aberrations and other end points in mammalian cells. Int. J. Radiat. Biol. 1988, 54, 869–890. [Google Scholar]
- Heim, S.; Mitelman, F. Cancer Cytogenetics, 3rd ed; Wiley-Blackwell: Hoboken, NJ, USA, 2009. [Google Scholar]
- Fusco, A.; Santoro, M. 20 years of RET/PTC in thyroid cancer: Clinico-pathological correlations. Arq. Bras. Endocrinol. Metabol. 2007, 51, 731–735. [Google Scholar]
- Viglietto, G.; Chiappetta, G.; Martinez-Tello, F.J.; Fukunaga, F.H.; Tallini, G.; Rigopoulou, D.; Visconti, R.; Mastro, A.; Santoro, M.; Fusco, A. RET/PTC oncogene activation is an early event in thyroid carcinogenesis. Oncogene 1995, 11, 1207–1210. [Google Scholar]
- Nikiforova, M.N.; Stringer, J.R.; Blough, R.; Medvedovic, M.; Fagin, J.A.; Nikiforov, Y.E. Proximity of chromoso-mal loci that participate in radiation-induced rearrangements in human cells. Science 2000, 290, 138–141. [Google Scholar]
- Nikiforov, Y.; Rowland, J.M.; Bove, K.E.; Monfore Munoz, H.; Fagin, J.A. Distinct patterns of ret rearrangements in morphological variants of radiation-induced and sporadic thyroid papillary carcinomas in children. Cancer Res. 1997, 57, 1690–1694. [Google Scholar]
- Santoro, M.; Melillo, R.M.; Fusco, A. RET/PTC activation in papillary thyroid carcinoma. Eur. J. Endocrinol. 2006, 155, 645–653. [Google Scholar]
- Elisei, R.; Romel, C.; Vorontsova, T.; Cosci, B.; Veremeychick, V.; Kuchinskaya, E.; Basolo, F.; Demidchik, E.P.; Miccoli, P.; Pinchera, A.; et al. RET/PTC rearrangments in tyroid nodules: Studies in irradiated and not irradiated, malignant and benign thyroid lesions in children and adults. J. Clin. Endocrinol. Metab. 2001, 86, 3211–3216. [Google Scholar]
- Hagmar, L.; Stromberg, U.; Bonassi, S.; Hansteen, I.L.; Knudsen, L.E.; Lindholm, C.; Norppa, H. Impact of types of lymphocyte chromosomal aberrations on human cancer risk: Results from Nordic and Italian cohorts. Cancer Res. 2004, 64, 2258–2263. [Google Scholar]
- Bonassi, S.; Abbondandolo, A.; Camurri, L.; Dal Prá, L.; de Ferrari, M.; Degrassi, F.; Forni, A.; Lamberti, L.; Lando, C.; Padovani, P.; et al. Are chromosome aberrations in circulating lymphocytes predictive of future cancer onset in humans? Preliminary results of an Italian cohort study. Cancer Genet. Cytogenet. 1995, 79, 133–135. [Google Scholar]
- Rabbitts, T.H. Chromosomal translocations in human cancer. Nature 1994, 372, 143–149. [Google Scholar]
- Auletta, A.; Ashby, J. Workshop on the relationship between short-term information and carcinogenicity. Environ. Mol. Mutagen. 1988, 11, 135–145. [Google Scholar]
- Shelby, M. The genetic toxicity of human carcinogens and its implications. Mutat. Res. 1988, 204, 3–15. [Google Scholar]
- Au, W.W.; Lane, R.G.; Legator, M.S.; Whorton, E.B.; Wilkinson, G.S.; Gaberhort, G.J. Biomarker monitoring of a population residing near uranium mining activities. Environ. Health Perspect. 1995, 103, 466–470. [Google Scholar]
- Mondello, C.; Smirnova, A.; Giulotto, E. Gene amplification, radiation sensitivity and DNA double-strand breaks. Mutat. Res. 2010, 704, 29–37. [Google Scholar]
- ICRP, Annual Report of the International Commission on Radiological Protection; Elsevier Science: New York, NY, USA, 1999.
- Djuzenova, C.S.; Muhl, B.; Fehn, M.; Oppitz, U.; Muller, B.; Flentje, M. Radiosensitivity in breast cancer assessed by the Comet and micronucleus assays. Br. J. Cancer 2006, 94, 1194–1203. [Google Scholar]
- Shahidi, M.; Mozdarani, H.; Bryant, P.E. Radiation sensitivity of leukocytes from healthy individuals and breast cancer patients as measured by the alkaline and neutral comet assay. Cancer Lett. 2007, 257, 263–273. [Google Scholar]
- Shahidi, M.; Mozdarani, H.; Mueller, W.U. Radiosensitivity and repair kinetics of gamma-irradiated leukocytes from sporadic prostate cancer patients and healthy individuals assessed by alkaline comet assay. Iran. Biomed. J. 2010, 14, 67–75. [Google Scholar]
- Lengauer, C.; Kinzler, K.W.; Vogelstein, B. Genetic instabilities in human cancers. Nature 1998, 396, 643–649. [Google Scholar]
- Thompson, L.H.; Schild, D. Recombinational DNA repair and human disease. Mutat. Res. 2002, 509, 49–78. [Google Scholar]
- Rothfuss, A.; Schütz, P.; Bochum, S.; Volm, T.; Eberhardt, E.; Kreienberg, R.; Vogel, W.; Speit, G. Induced micronucleus frequencies in peripheral lymphocytes as a screening test for carriers of a BRCA1 mutation in breast cancer families. Cancer Res. 2000, 60, 390–394. [Google Scholar]
- Carney, J.P. Chromosomal breakage syndromes. Curr. Opin. Immunol. 1999, 11, 443–447. [Google Scholar]
- Pichierri, P.; Franchitto, A.; Palitti, F. Predisposition to cancer and radiosensitivity. Genet. Mol. Biol. 2000, 23, 1101–1105. [Google Scholar]
- Mohseni, A.; Mozdarani, H.; Mozdarani, S. DNA damage and repair of leukocytes from Fanconi anemia patients, carriers and healthy individuals as measured by the alkaline comet assay. Mutagenesis 2009, 24, 67–73. [Google Scholar]
- Taylor, A.M.R. Chromosome instability syndromes. Best Pract. Res. Clin. Haematol. 2001, 14, 3631–3644. [Google Scholar]
- Duker, N.J. Chromosome breakage syndromes and cancer. Am. J. Med. Genet. 2002, 115, 125–129. [Google Scholar]
- Higurashi, M.; Conen, P.E. In vitro chromosomal radiosensitivity in chromosomal breakage syndromes. Cancer 1973, 32, 380–383. [Google Scholar]
- Lavin, M.F.; Shiloh, Y. The genetic defect in ataxia-telangiectasia. Ann. Rev. Immunol. 1997, 15, 177–202. [Google Scholar]
- Carney, J.P.; Maser, R.S.; Olivares, H.; Davis, E.M.; Le Beau, M.; Yates, J.R., III; Hays, L.; Morgan, W.F.; Petrini, J.H.J. The hMre11/hRad50 protein complex and Nijmegen breakage syndrome: Linkage of double-strand break repair to the cellular DNA damage response. Cell 1998, 93, 477–486. [Google Scholar] [CrossRef]
- Varon, R.; Vissinga, C.; Platzer, M.; Cerosaletti, K.M.; Chrzanowska, K.H.; Saar, K.; Beckmann, G.; Seemanová, E.; Cooper, P.R.; Nowak, N.J.; et al. Nibrin, a novel DNA double-strand break repair protein, is mutated in Nijmegen breakage syndrome. Cell 1998, 93, 467–476. [Google Scholar]
- Stewart, G.S.; Maser, R.S.; Stankovic, T.; Bressan, D.A.; Kaplan, M.I.; Jaspers, N.G.J.; Raams, A.; Byrd1, P.J.; Petrini, J.H.J.; Taylor, A.M.R. The DNA double-strand break repair gene hmre11 is mutated in individuals with an ataxia-telangiectasia-like disorder. Cell 1999, 99, 577–587. [Google Scholar]
- Ellis, N.A.; Groden, J.; Ye, T.-Z.; Straughen, J.; Lennon, D.J.; Ciocci, S.; Proytcheva, M.; German, J. The Bloom’s syndrome gene product is homologous to RecQ helicases. Cell 1995, 83, 655–666. [Google Scholar] [CrossRef]
- Gray, M.D.; Shen, J.C.; Kamath-Loeb, A.S.; Blank, A.; Sopher, B.L.; Martin, G.M.; Oshima, J.; Loeb, L.A. The Werner syndrome protein is a DNA helicase. Nat. Genet. 1997, 17, 100–103. [Google Scholar]
- Howlett, N.G.; Taniguchi, T.; Olson, S.; Cox, B.; Waisfisz, Q.; de Die-Smulders, C.; Persky, N.; Grompe, M.; Joenje, H.; Pals, G.; et al. Biallelic inactivation of BRCA2 in Fanconi anemia. Science 2002, 297, 606–609. [Google Scholar]
- Patel, K.J.; Yu, V.P.; Lee, H.; Corcoran, A.; Thistlethwaite, F.C.; Evans, M.J.; Colledge, W.H.; Friedman, L.S.; Ponder, B.A.; Venkitaraman, A.R. Involvement of Brca2 in DNA repair. Mol. Cell. 1998, 1, 347–357. [Google Scholar]
- Stewart, G.; Elledge, S.J. The two faces of BRCA2, a FANCtastic discovery. Mol. Cell. 2002, 10, 2–4. [Google Scholar]
- Meybodi, M.A.; Mozdarani, H.; Vosogh, P. Cytogenetic sensitivity of G0 lymphocytes of Fanconi anemia patients and obligate carriers to mytomycin C and ionizing radiation. Cytogenet. Genome Res. 2007, 119, 191–195. [Google Scholar]
- Meybodi, M.A.; Mozdarani, H. DNA Damage in leukocytes from fanconi anemia (FA) patients and heterozygotes induced by mitomycin C and ionizing radiation as assessed by the comet and comet-FISH assay. Iran. Biomed. J. 2009, 13, 1–8. [Google Scholar]
- Sanford, K.K.; Parshad, R.; Price, F.M.; Gantt, R.; Jones, G.M.; Tarone, R.E. Deficient G2 phase repair of radiation-induced chromatin damage in the SENCAR mouse. Carcinogenesis 1989, 10, 1911–1916. [Google Scholar]
- Sanford, K.K.; Parshad, R.; Gantt, R.R.; Tarone, R.E. A deficiency in chromatin repair, genetic instability, and predisposition to cancer. Crit. Rev. Oncog. 1989, 1, 323–341. [Google Scholar]
- Parshad, R.; Price, F.M.; Bohr, V.A.; Cowans, K.H.; Zujewski, J.A.; Sanford, A.A. Deficient DNA repair capacity, a predisposing factor in breast cancer. Br. J. Cancer 1996, 74, 1–5. [Google Scholar]
- Baeyens, A.; Thierens, H.; Claes, K.; Poppe, B.; Messiaen, L.; de Ridder, L.; Vral, A. Chromosomal radiosensitivity in breast cancer patients with a known or putative genetic predisposition. Br. J. Cancer 2002, 87, 1379–1385. [Google Scholar]
- Scott, D. Chromosomal radiosensitivity and low penetrance predisposition to cancer. Cytogenet. Genome Res. 2004, 104, 365–370. [Google Scholar]
- Scott, D.; Barber, J.B.; Spreadborough, A.R.; Burrill, W.; Roberts, S.A. Increased chromosomal radiosensitivity in breast cancer patients: A comparison of two assays. Int. J. Radiat. Biol. 1999, 75, 1–10. [Google Scholar]
- Sánchez, P.; Peñarroja, R.; Gallegos, F.; Bravo, J.L.; Rojas, E.; Benítez-Bribiesca, L. DNA damage in peripheral lymphocytes of untreated breast cancer patients. Arch. Med. Res. 2004, 35, 480–483. [Google Scholar]
- Mozdarani, H.; Mansouri, Z.; Haeri, S.A. Cytogenetic radiosensitivity of G0-lymphocytes of breast and esophageal cancer patients using the micronucleous assay. J. Radiat. Res. 2005, 46, 111–116. [Google Scholar]
- Helzlsouer, K.J.; Harris, E.L.; Parshad, R.; Perry, H.R.; Price, F.M.; Sanford, K.K. DNA repair proficiency: Potential susceptiblity factor for breast cancer. J. Natl. Cancer Inst. 1996, 88, 754–755. [Google Scholar]
- Patel, R.K.; Trivedi, A.H.; Arora, D.C.; Bhatavdekar, J.M.; Patel, D.D. DNA repair proficiency in breast cancer patients and their first-degree relatives. Int. J. Cancer 1997, 73, 20–24. [Google Scholar]
- Cybulski, C.; Huzarski, T.; Górski, B.; Masojć, B.; Mierzejewski, M.; Debniak, T.; Gliniewicz, B.; Matyjasik, J.; Złowocka, E.; Kurzawski, G.; et al. A novel founder CHEK2 mutation is associated with increased prostate cancer risk. Cancer Res. 2004, 64, 2677–2679. [Google Scholar]
- Angèle, S.; Jones, C.; Reis Filho, J.S.; Fulford, L.G.; Treilleux, I.; Lakhani, S.R.; Hall, J. Expression of ATM, p53, and the MRE11-Rad50-NBS1 complex in myoepithelial cells from benign and malignant proliferations of the breast. J. Clin. Pathol. 2004, 57, 1179–1184. [Google Scholar]
- Edwards, S.M.; Kote-Jarai, Z.; Meitz, J.; Hamoudi, R.; Hope, Q.; Osin, P.; Jackson, R.; Southgate, C.; Singh, R.; Falconer, A.; Dearnaley, D.P.; et al. Two percent of men with early-onset prostate cancer harbor germline mutations in the BRCA2 gene. Am. J. Hum. Genet. 2003, 72, 1–12. [Google Scholar] [CrossRef]
- Parshad, R.; Sanford, K.K.; Jones, G.M. Chromatid damage after G2 phase x-irradiation of cells from cancer-prone individuals implicates deficiency in DNA repair. Proc. Natl. Acad. Sci. USA 1983, 80, 5612–5616. [Google Scholar]
- Sanford, K.K.; Parshad, R.; Stanbridge, E.J.; Frost, J.K.; Jones, G.M.; Wilkinson, J.E.; Tarone, R.E. Chromosomal radiosensitivity during the G2 cell cycle period and cytopathology of human normal × tumor cell hybrids. Cancer Res. 1986, 46, 2045–2049. [Google Scholar]
- Mozdarani, H.; Liu, N.; Jones, N.J.; Bryant, P.E. The XRCC2 human repair gene influences recombinational rearrangements leading to chromatid breaks. Int. J. Radiat. Biol. 2001, 77, 859–865. [Google Scholar]
- Terzoudi, G.I.; Hatzi, V.I.; Donta-Bakoyianni, C.; Pantelias, G.E. Chromatin dynamics during cell cycle mediate conversion of DNA damage into chromatid breaks and affect formation of chromosomal aberrations: Biological and clinical significance. Mutat. Res. 2011, 711, 174–186. [Google Scholar]
- Belyaev, I.Y. Radiation-induced DNA repair foci: Spatio-temporal aspects of formation, application for assessment of radiosensitivity and biological dosimetry. Mutat. Res. 2010, 704, 132–141. [Google Scholar]
- Alsbeih, G.; Al-Harbi, N.; Al-Buhairi, M.; Al-Hadyan, K.; Al-Hamed, M. Association between TP53 codon 72 single-nucleotide polymorphism and radiation sensitivity of human fibroblasts. Radiat. Res. 2007, 167, 535–540. [Google Scholar]
- Andreassen, C.N.; Alsner, J.; Overgaard, J. Does variability in normal tissue reactions after radiotherapy have a genetic basis—Where and how to look for it? Radiother. Oncol. 2002, 64, 131–140. [Google Scholar] [CrossRef]
- Andreassen, C.N.; Alsner, J. Genetic variants and normal tissue toxicity after radiotherapy: A systematic review. Radiother. Oncol. 2009, 92, 299–309. [Google Scholar]
- Rosenstein, B.S. Identification of SNPs associated with susceptibility for development of adverse reactions to radiotherapy. Pharmacogenomics 2011, 12, 267–275. [Google Scholar]
- Azria, D.; Ozsahin, M.; Kramar, A.; Peters, S.; Atencio, D.P.; Crompton, N.E.A.; Mornex, F.; Pèlegrin, A.; Dubois, J.B.; Mirimanoff, R.O.; et al. Single nucleotide polymorphisms, apoptosis and the development of severe late adverse effects after radiotherapy. Clin. Cancer Res. 2008, 14, 6284–6288. [Google Scholar]
- Michikawa, Y.; Suga, T.; Ishikawa, A.; Hayashi, H.; Oka, A.; Inoko, H.; Iwakawa, M.; Imai, T. Genome wide screen identifies microsatellite markers associated with acute adverse effects following radiotherapy in cancer patients. BMC Med. Genet. 2010, 11, 123. [Google Scholar]
- Luckey, T.D. Radiation Hormesis; CRC Press: Boca Raton, FL, USA, 1991. [Google Scholar]
- Jolly, D.; Meyer, J. A brief review of radiationhormesis. Australas. Phys. Eng. Sci. Med. 2009, 32, 180–187. [Google Scholar]
- Muckerheide, J. Low-Level Radiation Health Effects: A Compilation of Data and Programs; RSH Inc.: Needham, MA, USA, 2001. [Google Scholar]
- McClintock, B. The significance of responses of the genome to challenge. Science 1984, 226, 792–801. [Google Scholar]
- Serebrianyĭ, A.M. Radiation adaptive response as a stress reaction of a cell (in Russian). Radiats. Biol. Radioecol. 2011, 51, 399–404. [Google Scholar]
- Samson, L.; Cairns, J. A new pathway for DNA repair in Escherichia coli. Nature 1977, 267, 281–283. [Google Scholar]
- Laval, F.; Laval, J. Adaptive response in mammalian cells: Crossreactivity of different pretreatments on cytotoxicity as contrasted to mutagenicity. Proc. Natl. Acad. Sci. USA 1984, 81, 1062–1066. [Google Scholar]
- Olivieri, G.; Bodycote, J.; Wolff, S. Adaptive response of human lymphocytes to low concentrations of radioactive thymidine. Science 1984, 223, 594–597. [Google Scholar]
- Wolff , S. Failla Memorial Lecture. Is radiation all bad? The search for adaptation. Radiat. Res. 1992, 131, 117–123. [Google Scholar] [CrossRef]
- Wolff, S. The adaptive response in radiobiology: Evolving insights and implications. Environ. Health Perspect. 1998, 106, S277–S283. [Google Scholar]
- Rigaud, O.; Moustacchi, E. Radioadaptation for gene mutation and the possible molecular mechanisms of the adaptive response. Mutat. Res. 1996, 358, 127–134. [Google Scholar]
- Stoilov, L.M.; Mullenders, L.H.; Darroudi, F.; Natarajan, A.T. Adaptive response to DNA and chromosomal damage induced by X-rays in human blood lymphocytes. Mutagenesis 2007, 22, 117–122. [Google Scholar]
- Bryant, P.E. Origin of chromosome anerrations: Mechanisms. In Chromoosmeal Alterations: Methods, Results and Importance in Human Health; Obe, G., Vijayalaxmi, Eds.; Springer-Verlag: Berlin, Germany, 2007; pp. 177–199. [Google Scholar]
- Bryant, P.E.; Mozdarani, H. Mechanisms underlying the conversion of DNA double strand breaks into chromatid breaks. Int. J. Low Radiat. 2004, 1, 223–230. [Google Scholar]
- Tedeschi, B.; Caporossi, D.; Vernole, P.; Padovani, L.; Appolloni, M.; Anzidei, P.; Mauro, F. Do human lymphocytes exposed to the fallout of the Chernobyl accident exhibit an adaptive response? II. Challenge with bleomycin. Mutat. Res. 1995, 332, 39–44. [Google Scholar] [CrossRef]
- Mozdarani, H.; Saberi, A.H. Induction of cytogenetic adaptive response of bone marrow cells to radiation by therapeutic doses of bleomycin sulfate and actionmycin D as assayed by the micronucleus test. Cancer Lett. 1994, 78, 141–150. [Google Scholar]
- Barquinero, J.F.; Barrios, L.; Caballin, M.R.; Miro, R.; Ribas, M. Occupational exposure to radiation induces an adaptive response in human lymphocytes. Int. J. Radiat. Biol. 1995, 67, 187–191. [Google Scholar]
- Barquinero, J.F.; Barrios, L.; Caballin, M.R.; Miro, R.; Ribas, M.; Subias, A.; Egozcue, J. Decreased sensitivity to the cytogenetic effects of bleomycin in individuals occupationally exposed to ionizing radiation. Mutat. Res. 1996, 354, 81–86. [Google Scholar]
- Gourabi, H.; Mozdarani, H. A cytokinesis blocked micronucleus study of the radiadaptive response of lymphocytes of individuals ocupationally exposed to chronic doses ofradiation. Mutagenesis 1998, 13, 475–480. [Google Scholar]
- Mozdarani, H.; Gourabi, H. Chromosomal aberrations and micronuclei in lymphocytes of medical radiation workers exposed to ionizing radiation below dose limit. Yakhteh Med. J. 2001, 3, 153–158. [Google Scholar]
- Tedeschi, B. Do human lymphocytes exposed to fall out of Chernobyl accident exhibit an adaptive response. Mutat. Res. 1996, 354, 77–80. [Google Scholar]
- Mitchel, R.E.J.; Jackson, J.S.; McCann, R.A.; Boreham, D.R. Adaptive response modification of latency for radiation-induced myeloid leukemia in CBA/H mice. Radiat. Res. 1999, 152, 273–279. [Google Scholar]
- Mitchel, R.E.J.; Jackson, J.S.; Morrison, D.P.; Carlisle, S.M. Low doses of radiation increase the latency of spontaneous lymphomas and spinal osteosarcomas in cancer-prone, radiation-sensitive Trp53 heterozygous mice. Radiat. Res. 2003, 159, 320–327. [Google Scholar]
- Cohen, B.L. Test of the linear no-threshold theory of radiation carcinogenesis for inhaled radon decay products. Health Phys. 1995, 68, 157–174. [Google Scholar]
- Deetjen, P. Biological and therapeutical properties of radon. In Radon and Thoron in the Human Environment; Katase, A., Shimo, M., Eds.; WorldScientific: Singapore, 1998; pp. 515–522. [Google Scholar]
- Becker, K. Health effects of high radon environments in central Europe: Another test for the LNT hypothesis. Nonlinearity Biol. Toxicol. Med. 2005, 1, 3–35. [Google Scholar]
- Chen, W.L.; Luan, Y.C.; Shici, M.C.; Chen, S.T.; Kung, H.T.; Soong, K.L.; Yeh, Y.C.; Chou, S.H.; Mong, S.H.; Wu, J.T.; et al. Is chronic radiation an effective prophylaxis against cancer? J. Am. Phys. Surg. 2004, 9, 6–10. [Google Scholar]
- Sakamoto, K.; Myonin, M.; Hosor, Y.; Ogawa, Y.; Nemoto, K.; Takai, Y.; Kakuto, Y.; Yamada, S.; Watabe, N. Fundamental and clinical studies on cancer control with total or upper half body irradiation. J. Jpn. Soc. Ther. Radiol. Oncol. 1997, 9, 161–175. [Google Scholar]
- Aurengo, A.; Averbeck, D.; Bonnin, A.; LeGuen, B.; Masse, R.; Monier, R.; Tubiana, M.; Valleron, A.J.; de Vathaire, F. Dose-Effect Relationships and Estimation of the Carcinogenic Effectsof Low Doses of Ionizing Radiation; Academies of Sciences and Medicine: Paris, France, 2005. [Google Scholar]
- UNSCEAR (United Nations Scientific Committee on the Effects of Atomic Radiation), Health Effects Due to Radiation from the Chernobyl Accident; Report to the General Assembly with Scientific Annexes, Volume II, Annex D; United Nations: New York, NY, USA, 2011.
- Luckey, T.D. Low dose irradiation reduces cancer death rates. Radiat. Protect. Manag. 1997, 14, 58–64. [Google Scholar]
- Luckey, T.D. Radiation hormesis in cancer mortality. Int. J. Occup. Med. Tox. 1994, 3, 175–191. [Google Scholar]
- Vaiserman, A.M. Radiationhormesis: Historical perspective and implications for low-dose cancer risk assessment. Dose Response 2010, 8, 172–191. [Google Scholar]
- Luckey, T.D. Nuclear triage and the dirty bomb. Radiat. Protect. Manag. 2004, 20, 11–18. [Google Scholar]
- NEA Committee, Chernobyl: Ten Years on Radiological and Health Impact; Nuclear Energy Agency, OCED (Organization for Economic Co-Operation and Development): Paris, France, 1995.
- National Research Council, Health Effects of Exposure to Radon, BEIR VI; National Academy Press: Washington, DC, USA, 1999.
- National Research Council, Health Risks from Exposure to Low Levels of Ionizing Radiation, BEIR VII, Phase 2; National Academy Press: Washington, DC, USA, 2005.
- Jaworowski, Z. Radiationhormesis—A remedy for fear. Hum. Exp. Toxicol. 2010, 29, 263–270. [Google Scholar]
- Azzam, E.I.; Raaphorst, G.P.; Mitchel, R.E.J. Radiation-induced adaptive response for protection against micronucleus formation and neoplastic transformation in C3H 10T1/2 mouse embryo cells. Radiat. Res. 1994, 138, S28–S31. [Google Scholar]
- Broome, E.J.; Brown, D.L.; Mitchel, R.E. Dose responses for adaption to low doses of (60)Co gamma rays and (3)H beta particles in normal human fibroblasts. Radiat. Res. 2002, 158, 181–186. [Google Scholar]
- Hamada, N.; Maeda, M.; Otsuka, K.; Tomita, M. Signaling pathways underpinning the manifestations of ionizing radiation-induced bystander effects. Curr. Mol. Pharmacol. 2011, 4, 79–95. [Google Scholar]
- Feinendegen, L.E.; Bond, V.P.; Booz, J.; Mühlensiepen, H. Biochemical and cellular mechanisms of low-dose effects. Int. J. Radiat. Biol. Relat. Stud. Phys. Chem. Med. 1988, 53, 23–37. [Google Scholar]
- Wolff, S. Are radiation-induced effects hormetic? Science 1989, 245, 575–621. [Google Scholar]
- Sankaranarayanan, K.; von Duyn, A.; Loos, M.J.; Natarajan, A.T. Adaptive response of human lymphocytes to low-level radiation from radioisotopes or X-rays. Mutat. Res. 1989, 211, 7–12. [Google Scholar]
- Bosi, A.; Olivieri, G. Variability of the adaptive response to ionizing radiations in humans. Mutat. Res. 1989, 211, 13–17. [Google Scholar]
- Davalos, A.; Yannone, S.; Kawahara, M.; Campisi, J. Molecular Mediators of Radio-Adaptation in Human Cells. DOE Low Dose Radiation Research Investigators’ Workshop VI, Washington, DC, USA, 30 July–2 August 2006.
- UNSCEAR (United Nations Scientific Committee on the Effects of Atomic Radiation), Effects of Ionizing Radiation; Report to the General Assembly with Scientific Annexes, Volume I; United Nations, New York, NY, USA, 2006.
- Liu, S.Z. Biological effects of low level exposures to ionizing radiation: Theory and practice. Hum. Exp. Toxicol. 2010, 29, 275–281. [Google Scholar]
- Baverstock, K. Radiation-induced genomic instability: A paradigm-breaking phenomenon and its relevance to environmentally induced cancer. Mutat. Res. Fund. Mol. Mech. Mutagen. 2000, 454, 89–109. [Google Scholar]
- Mothersill, C.; Seymour, C. Radiation-induced bystander effects: Past history and future directions. Radiat. Res. 2001, 155, 757–765. [Google Scholar]
- Goldberg, Z.; Lehnert, B.E. Radiation-induced effects in unirradiated cells: A review and implications in cancer. Int. J. Oncol. 2002, 21, 337–349. [Google Scholar]
- Lorimore, S.A.; Coates, P.J.; Wright, E.G. Radiation-induced genomic instability and bystander effects: Inter-related nontargeted effects of exposure to ionizing radiation. Oncogene 2003, 22, 7058–7069. [Google Scholar]
- Mothersill, C.; Seymour, C. Radiation-induced bystander effects, carcinogenesis and models. Oncogene 2003, 22, 7028–7033. [Google Scholar]
- Wright, E.G. Manifestations and mechanisms of non-targeted effects of ionizing radiation. Mutat. Res. 2010, 687, 28–33. [Google Scholar]
- Mothersill, C.; Seymour, C. Medium from irradiated human epithelial cells but not human fibroblasts reduces the clonogenic survival of unirradiated cells. Int. J. Radiat. Biol. 1997, 71, 421–427. [Google Scholar]
- Lehnert, B.E.; Goodwin, E.H. A new mechanism for DNA alterations induced by alpha particles such as those emitted by radon and radon progeny. Environ. Health Perspect. 1997, 105, 1095–1101. [Google Scholar]
- Prise, K.M.; Belyakov, O.V.; Folkard, M.; Michael, B.D. Studies of bystander effects in human fibroblasts using a charged particle microbeam. Int. J. Radiat. Biol. 1998, 74, 793–798. [Google Scholar]
- Shao, C.; Stewart, V.; Folkard, M.; Michael, B.D.; Prise, K.M. Nitric oxide-mediated signaling in the bystandard response of individually targeted glioma cells. Cancer Res. 2003, 63, 8437–8442. [Google Scholar]
- Lorimore, S.A.; Kadhim, M.A.; Pocock, D.A.; Papworth, D.; Stevens, D.L.; Goodhead, D.T.; Wright, E.G. Chromosomal instability in the descendants of unirradiated surviving cells after alpha-particle irradiation. Proc. Natl. Acad. Sci. USA 1998, 95, 5730–5733. [Google Scholar]
- Azzam, E.I.; de Toledo, S.M.; Gooding, T.; Little, J.B. Intercellular communication is involved in the bystander regulation of gene expression in human cells exposed to very low fluences of alpha particles. Radiat. Res. 1998, 150, 497–504. [Google Scholar]
- Belloni, P.; Latini, P.; Palitti, F. Radiation-induced bystander effect in healthy G0 human lymphocytes: Biological and clinical significance. Mutat. Res. 2011, 713, 32–38. [Google Scholar] [CrossRef]
- Little, J.B. Cellular radiation effects and the bystander response. Mutat. Res. 2006, 597, 113–118. [Google Scholar]
- Kanasugi, Y.; Hamada, N.; Wada, S.; Funayama, T.; Sakashita, T.; Kakizaki, T.; Kobayashi, Y.; Takakura, K. Role of DNA-PKcs in the bystander effect after low- or high-LET irradiation. Int. J. Radiat. Biol. 2007, 83, 73–80. [Google Scholar]
- Konopacka, M.; Rzeszowska-Wolny, R.J. The bystander effect-induced formation of micronucleated cells is inhibited by antioxidants, but the parallel induction of apoptosis and loss of viability are not affected. Mutat. Res. 2006, 593, 32–38. [Google Scholar]
- Rzeszowska-Wolny, J.; Przybyszewski, W.M.; Widel, M. Ionizing radiation-induced bystander effects, potential targets for modulation of radiotherapy. Eur. J. Pharmacol. 2009, 625, 156–164. [Google Scholar]
- Kashino, G.; Suzuki, K.; Matsuda, N.; Kodama, S.; Ono, K.; Watanabe, M.; Prise, K.M. Radiation induced bystander signals are independent of DNA damage and DNA repair capacity of the irradiated cells. Mutat. Res. 2007, 619, 134–138. [Google Scholar] [CrossRef]
- Sokolov, M.V.; Smilenov, L.B.; Hall, E.J.; Panyutin, I.G.; Bonner, W.M.; Sedelnikova, O.A. Ionizing radiation induces DNA double-strand breaks in bystander primary human fibroblasts. Oncogene 2005, 24, 7257–7265. [Google Scholar]
- Harada, T.; Kashino, G.; Suzuki, K.; Matsuda, N.; Kodama, S.; Watanabe, M. Different involvement of radical species in irradiated and bystander cells. Int. J. Radiat. Biol. 2008, 84, 809–814. [Google Scholar]
- Sokolov, M.V.; Dickey, J.S.; Bonner, W.M.; Sedelnikova, O.A. Gamma-H2AX in bystander cells: Not just a radiation-triggered event, a cellular response to stress mediated by intercellular communication. Cell Cycle 2007, 6, 2210–2212. [Google Scholar]
- Sedelnikova, O.A.; Nakamura, A.; Kovalchuk, O.; Koturbash, I.; Mitchell, S.A.; Marino, S.A.; Brenner, D.J.; Bonner, W.M. DNA double-strand breaks form in bystander cells after microbeam irradiation of three-dimensional human tissue models. Cancer Res. 2007, 67, 4295–4302. [Google Scholar]
- Belyakov, O.; Malcolmson, A.; Folkard, M.; Prise, K.; Michael, B. Direct evidence for a bystander effect of ionizing radiation in primary human fibroblasts. Br. J. Cancer 2001, 84, 674–679. [Google Scholar]
- Ballarini, F.; Biaggi, M.; Ottolenghi, A.; Sapora, O. Cellular communication and bystander effects: A critical review for modelling low-dose radiation action. Mutat. Res. 2002, 501, 1–12. [Google Scholar]
- Prise, K.M.; O'Sullivan, J.M. Radiation-induced bystander signalling in cancer therapy. Nat. Rev. Cancer 2009, 9, 351–360. [Google Scholar]
- Lorimore, S.A.; Coates, P.J.; Scobie, G.E.; Milne, G.; Wright, E.G. Inflammatory-type responses after exposure to ionizing radiation in vivo: A mechanism for radiation-induced bystander effects? Oncogene 2001, 20, 7085–7095. [Google Scholar] [CrossRef]
- Marples, B.; Collis, S.J. Low-dose hyper-radiosensitivity: Past, present, and future. Int. J. Radiat. Oncol. Biol. Phys. 2008, 70, 1310–1318. [Google Scholar]
- Iyer, R.; Lehnert, B.E. Low dose, low-LET ionizing radiation-induced radioadaptation and associated early responses in unirradiated cells. Mutat.Res. 2002, 503, 1–9. [Google Scholar]
- Rothkamm, K.; Löbrich, M. Evidence for a lack of DNA double-strand break repair in human cells exposed to very low x-ray doses. Proc. Natl. Acad. Sci. USA 2003, 100, 5057–5062. [Google Scholar]
- Ojima, M.; Ban, N.; Kai, M. DNA double-strand breaks induced by very low X-ray doses are largely due to bystander effects. Radiat. Res. 2008, 170, 365–371. [Google Scholar] [CrossRef]
- Little, M.P. Do non-targeted effects increase or decrease low dose risk in relation to the linear-non-threshold (LNT) model? Mutat. Res. 2010, 687, 17–27. [Google Scholar] [CrossRef]
- Suit, H.; Goldberg, S.; Niemierko, A.; Ancukiewicz, M.; Hall, E.; Goitein, M.; Wong, W.; Paganetti, H. Secondary carcinogenesis in patients treated with radiation: A review of data on radiation-induced cancers in human, non-human primate, canine and rodent subjects. Radiat. Res. 2007, 167, 12–42. [Google Scholar]
- Tubiana, M. Can we reduce the incidence of second primary malignancies occurring after radiotherapy? A critical review. Radiother. Oncol. 2009, 91, 4–15. [Google Scholar]
- Wideł, M.; Przybyszewski, W.; Rzeszowska-Wolny, J. Radiation-induced bystander effect: The important part of ionizing radiation response (in Polish). Potential clinical implications. Postepy Hig. Med. Dosw. (Online) 2009, 63, 377–388. [Google Scholar]
- Sjostedt, S.; Bezak, E. Non-targeted effects of ionising radiation and radiotherapy. Australas. Phys. Eng. Sci. Med. 2010, 33, 219–231. [Google Scholar]
- Blyth, B.J.; Sykes, P.J. Radiation-induced bystander effects: What are they, and how relevant are they to human radiation exposures? Radiat. Res. 2011, 176, 139–157. [Google Scholar] [CrossRef]
- Mackonis, E.C.; Suchowerska, N.; Zhang, M.; Ebert, M.; McKenzie, D.R.; Jackson, M. Cellular response to modulated radiation fields. Phys. Med. Biol. 2007, 52, 5469–5482. [Google Scholar]
- Iyer, R.; Lehnert, B.E. Effects of ionizing radiation in targeted and nontargeted cells. Arch. Biochem. Biophys. 2000, 376, 14–25. [Google Scholar]
- Hei, T.K.; Zhou, H.; Chai, Y.; Ponnaiya, B.; Ivanov, V.N. Radiation induced non-targeted response: Mechanism and potential clinical implications. Curr. Mol. Pharmacol. 2011, 4, 96–105. [Google Scholar]
- Mole, R.H. Whole body irradiation-radiobiology or medicine? Br. J. Radiol. 1953, 26, 234–241. [Google Scholar] [CrossRef]
- Nobler, M.P. The abscopal effect in malignant lymphoma and its relationship to lymphocyte circulation. Radiology 1969, 93, 410–412. [Google Scholar]
- Antoniades, J.; Brady, L.W.; Lightfoot, D.A. Lymphangiographic demonstration of the abscopal effect in patients with malignant lymphomas. Int. J. Radiat. Oncol. Biol. Phys. 1977, 2, 141–147. [Google Scholar]
- Rees, G.J. Abscopal regression in lymphoma: A mechanism in common with total body irradiation? Clin. Radiol. 1981, 32, 475–480. [Google Scholar] [CrossRef]
- Ehlers, G.; Fridman, M. Abscopal effect of radiation in papillary adenocarcinoma. Br. J. Radiol. 1973, 46, 220–222. [Google Scholar]
- Kingsley, D.P. An interesting case of possible abscopal effect in malignant melanoma. Br. J. Radiol. 1975, 48, 863–866. [Google Scholar]
- Rees, G.J.; Ross, C.M. Abscopal regression following radiotherapy for adenocarcinoma. Br. J. Radiol. 1983, 56, 63–66. [Google Scholar]
- Sham, R.L. The abscopal effect and chronic lymphocytic leukemia. Am. J. Med. 1995, 98, 307–308. [Google Scholar]
- Ohba, K.; Omagari, K.; Nakamura, T.; Ikuno, N.; Saeki, S.; Matsuo, I.; Kinoshita, H.; Masuda, J.; Hazama, H.; Sakamoto, I.; et al. Abscopal regression of hepatocellular carcinoma after radiotherapy for bone metastasis. Gut 1998, 43, 575–577. [Google Scholar] [CrossRef]
- Takaya, M.; Niibe, Y.; Tsunoda, S.; Jobo, T.; Imai, M.; Kotani, S.; Unno, N.; Hayakawa, K. Abscopal effect of radiation on toruliform para-aortic lymph node metastases of advanced uterine cervical carcinoma—A case report. Anticancer Res. 2007, 27, 499–503. [Google Scholar]
- Okuma, K.; Yamashita, H.; Niibe, Y.; Hayakawa, K.; Nakagawa, K. Abscopal effect of radiation on lung metastases of hepatocellular carcinoma: A case report. J. Med. Case Reports 2011, 5, 111. [Google Scholar]
- Perego, D.; Faravelli, A. Unexpected consequence of splenectomy in composite lymphoma. The abscopal effect. Haematologica 2000, 85, 211. [Google Scholar]
- Vartak, S.; George, K.C.; Singh, B.B. Antitumor effects of local hyperthermia on a mouse fibrosarcoma. Anticancer Res. 1993, 13, 727–729. [Google Scholar]
- Formenti, S.C.; Demaria, S. Systemic effects of local radiotherapy. Lancet Oncol. 2009, 10, 718–726. [Google Scholar]
- Dewan, M.Z.; Galloway, A.E.; Kawashima, N.; Dewyngaert, J.K.; Babb, J.S.; Formenti, S.C.; Demaria, S. Fractionated but not single-dose radiotherapy induces an immune-mediated abscopal effect when combined with anti-CTLA-4 antibody. Clin. Cancer Res. 2009, 15, 5379–5388. [Google Scholar]
- Demaria, S.; Ng, B.; Devitt, M.L.; Babb, J.S.; Kawashima, N.; Liebes, L.; Formenti, S.C. Ionizing radiation inhibition of distant untreated tumors (abscopal effect) is immune mediated. Int. J. Radiat. Oncol. Biol. Phys. 2004, 58, 862–870. [Google Scholar]
- Camphausen, K.; Moses, M.A.; Ménard, C.; Sproull, M.; Beecken, W.D.; Folkman, J.; O'Reilly, M.S. Radiation abscopal antitumor effect is mediated through p53. Cancer Res. 2003, 63, 1990–1993. [Google Scholar]
- Kinashi, Y.; Tanaka, H.; Masunaga, S.; Suzuki, M.; Kashino, G.; Yong, L.; Takahashi, S.; Ono, K. Ascorbic acid 2-glucocide reduces micronucleus induction in distant splenic T lymphocytes following head irradiation. Mutat. Res. 2010, 695, 69–74. [Google Scholar]
- Ilnytskyy, Y.; Kovalchuk, O. Non-targeted radiation effects-an epigenetic connection. Mutat. Res. 2011, 714, 113–125. [Google Scholar]
- Kaminski, J.M.; Shinohara, E.; Summers, J.B.; Niermann, K.J.; Morimoto, A.; Brousal, J. The controversial abscopal effect. Cancer Treat. Rev. 2005, 31, 159–172. [Google Scholar]
- Joiner, M.C.; Marples, B.; Lambin, P.; Short, S.C.; Turesson, I. Low-dose hypersensitivity: Current status and possible mechanisms. Int. J. Radiat. Oncol. Biol. Phys. 2001, 49, 379–389. [Google Scholar]
- Mortazavi, S.M.J.; Ikushima, T.; Mozdarani, H. Variablity of chromosomal radioadaptive response in human lymphocytes. Iran. J. Radiat. Res. 2003, 1, 55–61. [Google Scholar]
- Kadhim, M.A.; Moore, S.R.; Goodwin, E.H. Interrelationships amongst radiation-induced genomic instability, bystander effects, and the adaptive response. Mutat. Res. 2004, 568, 21–32. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mozdarani, H. Biological Complexities in Radiation Carcinogenesis and Cancer Radiotherapy: Impact of New Biological Paradigms. Genes 2012, 3, 90-114. https://doi.org/10.3390/genes3010090
Mozdarani H. Biological Complexities in Radiation Carcinogenesis and Cancer Radiotherapy: Impact of New Biological Paradigms. Genes. 2012; 3(1):90-114. https://doi.org/10.3390/genes3010090
Chicago/Turabian StyleMozdarani, Hossein. 2012. "Biological Complexities in Radiation Carcinogenesis and Cancer Radiotherapy: Impact of New Biological Paradigms" Genes 3, no. 1: 90-114. https://doi.org/10.3390/genes3010090




