Association of NF-κB and AP-1 with MMP-9 Overexpression in 2-Chloroethanol Exposed Rat Astrocytes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Astrocyte Enrichment and Culture
2.3. In Vitro Treatments
2.4. Immunofluorescence
2.5. Western Blotting
2.6. Quantitative Real-Time (RT)-PCR
2.7. ELISA
2.8. Statistical Analysis
3. Results
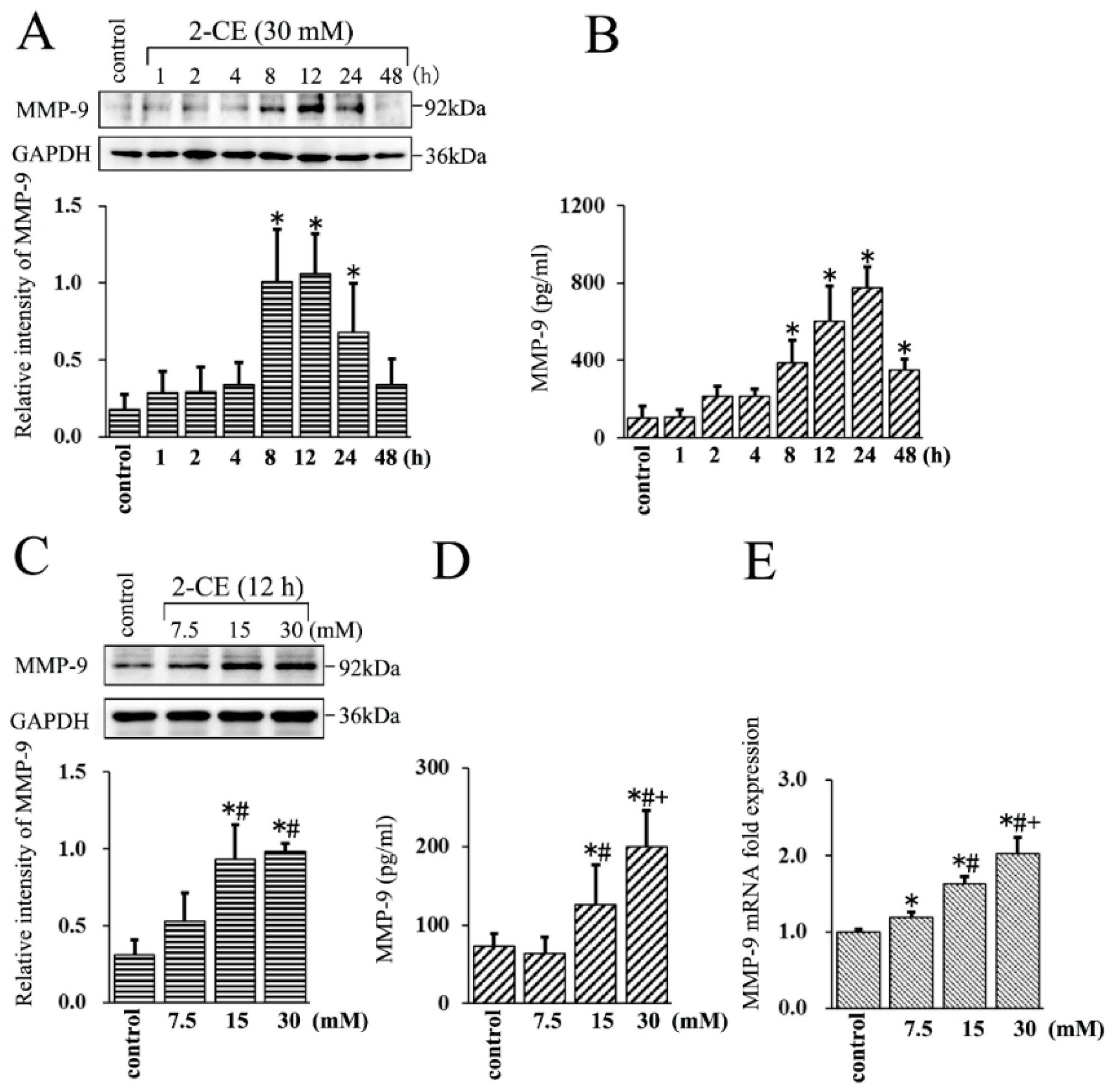
3.1. Upregulated MMP-9 Expression in 2-CE Treated Rat Astrocytes along with the Exposure Duration and Concentrations
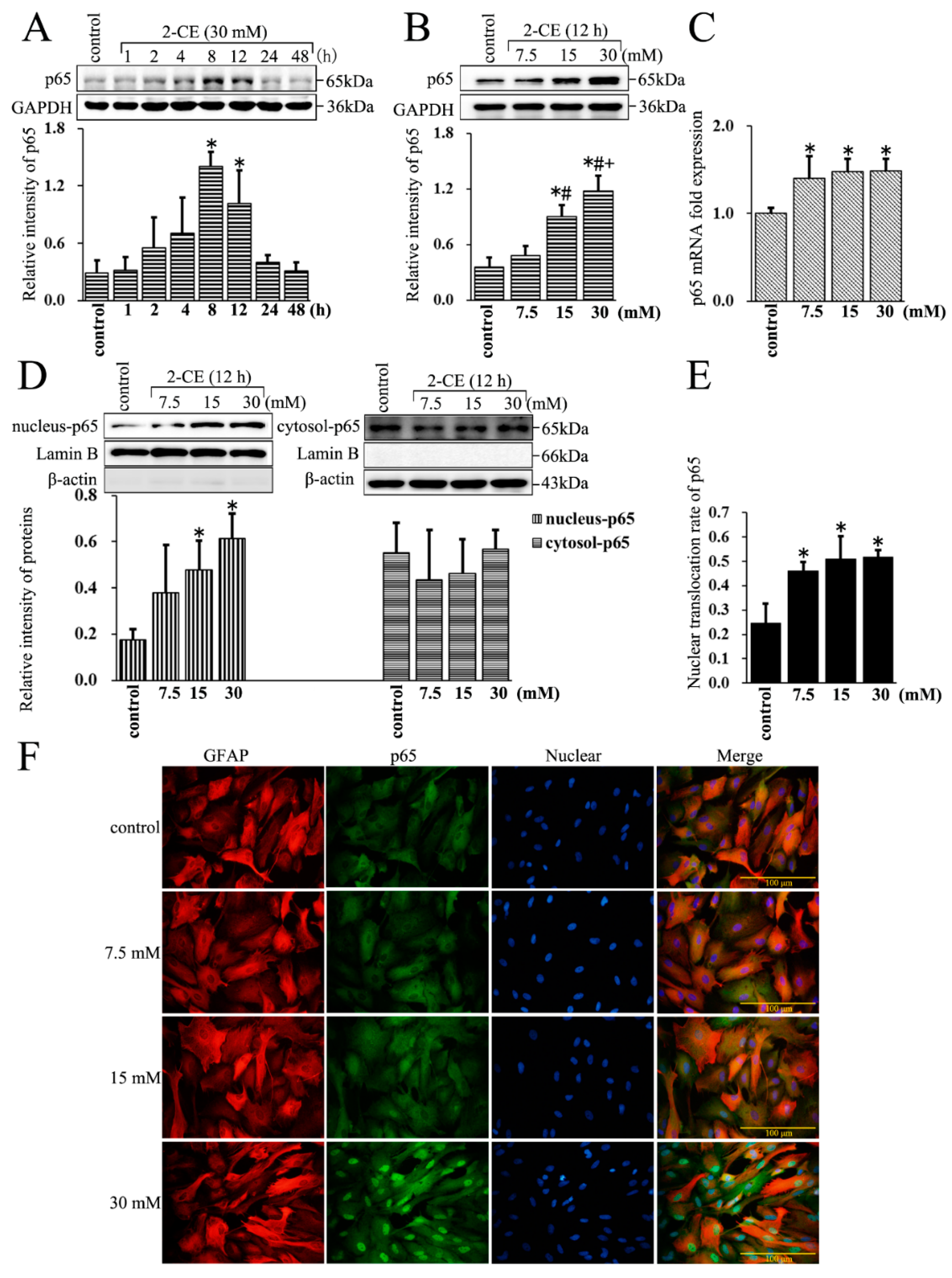
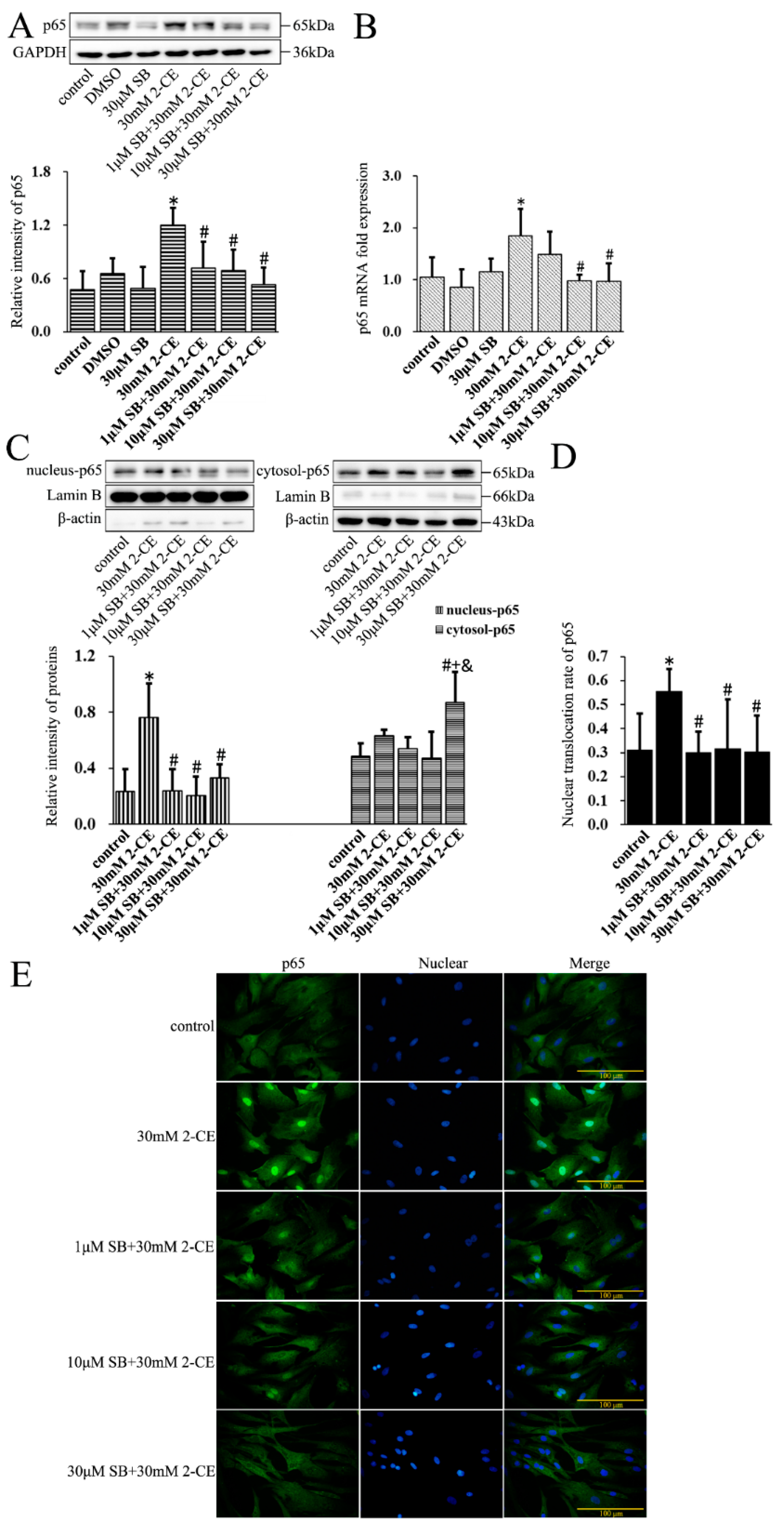
3.2. Increased Expression and Nuclear Translocation of p65 in 2-CE Treated Rat Astrocytes along with the Exposure Duration and Concentrations
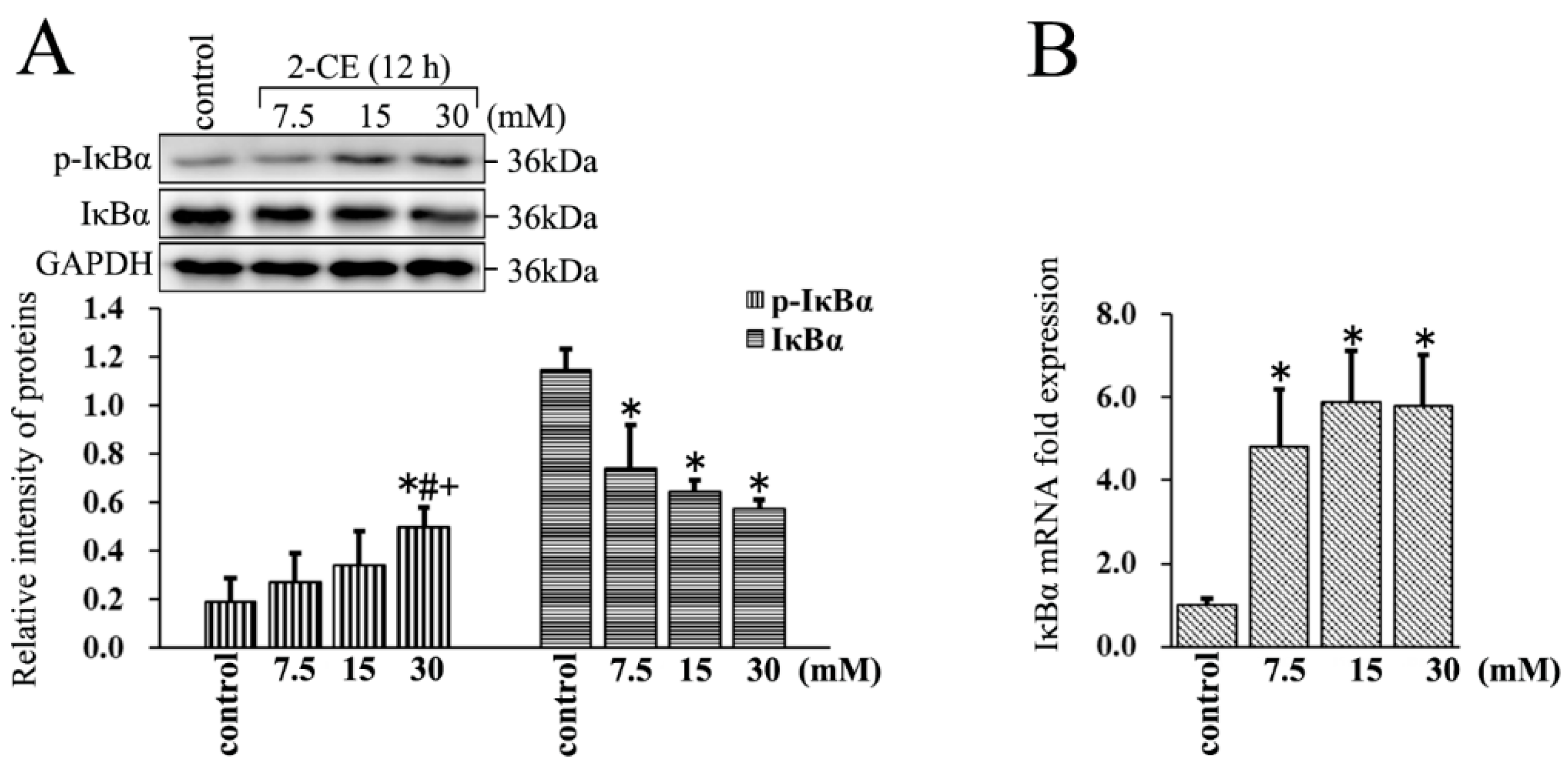
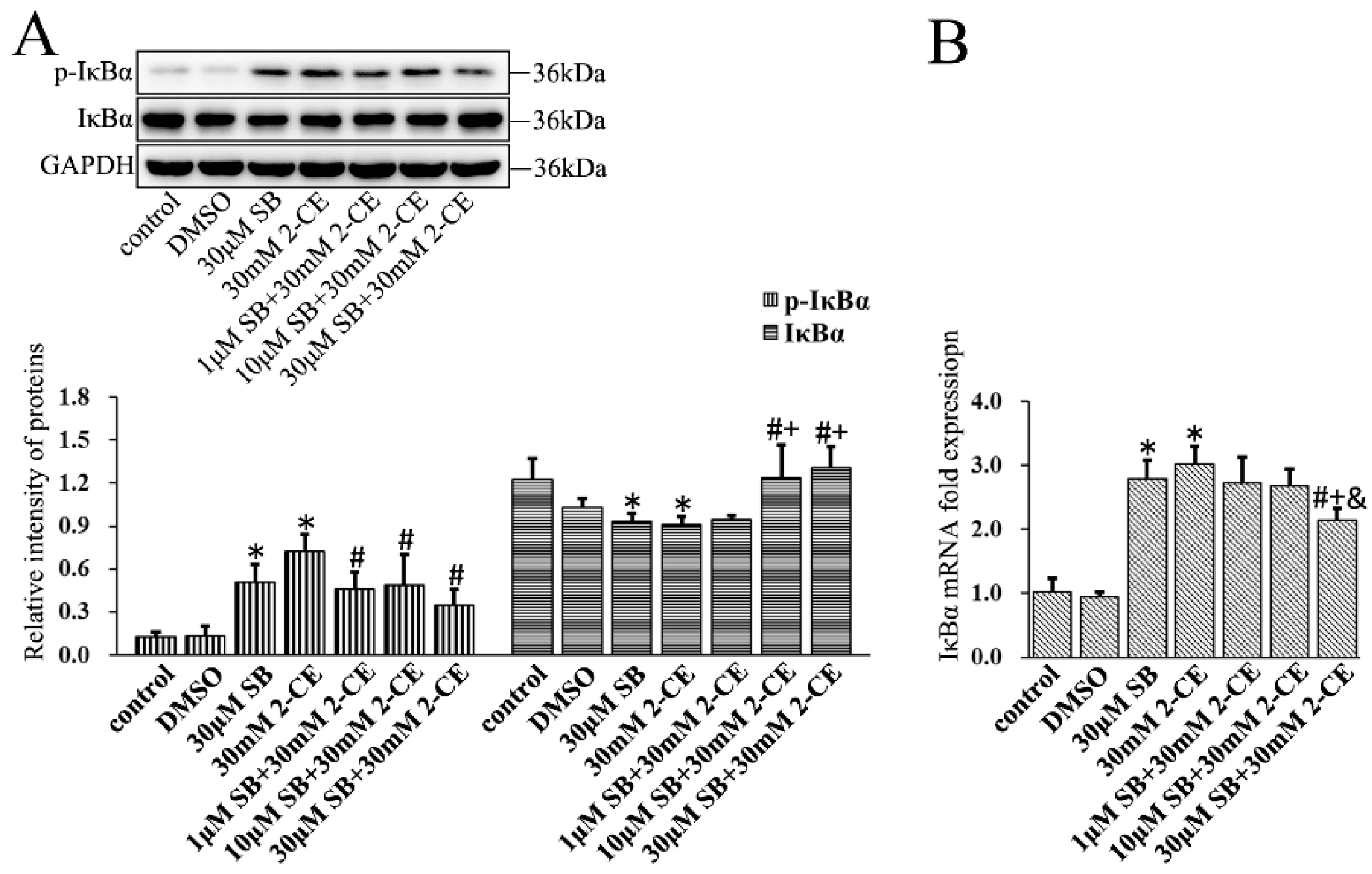
3.3. Increased Expression and Phosphorylation of IκBα in 2-CE Treated Rat Astrocytes along with the Exposure Concentrations
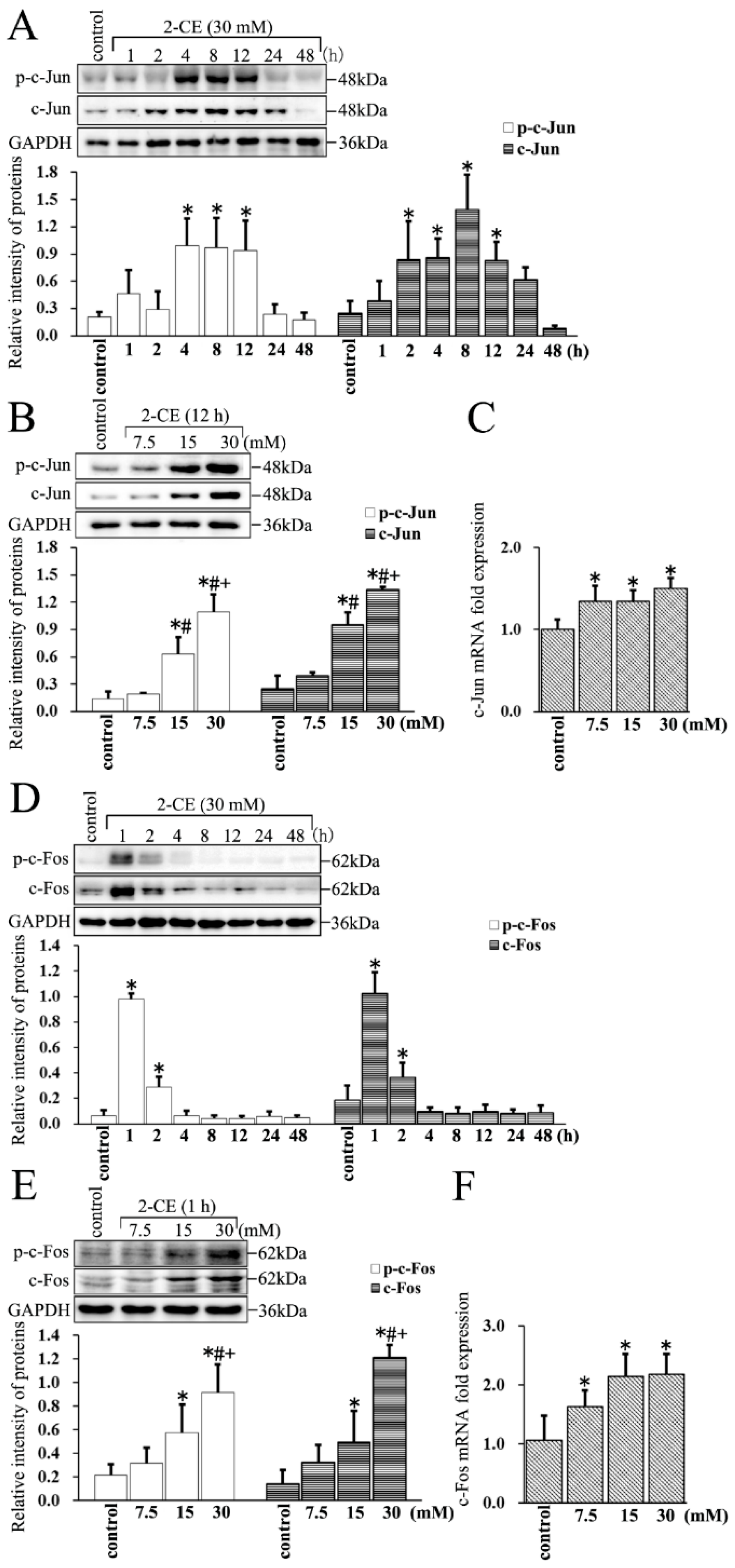
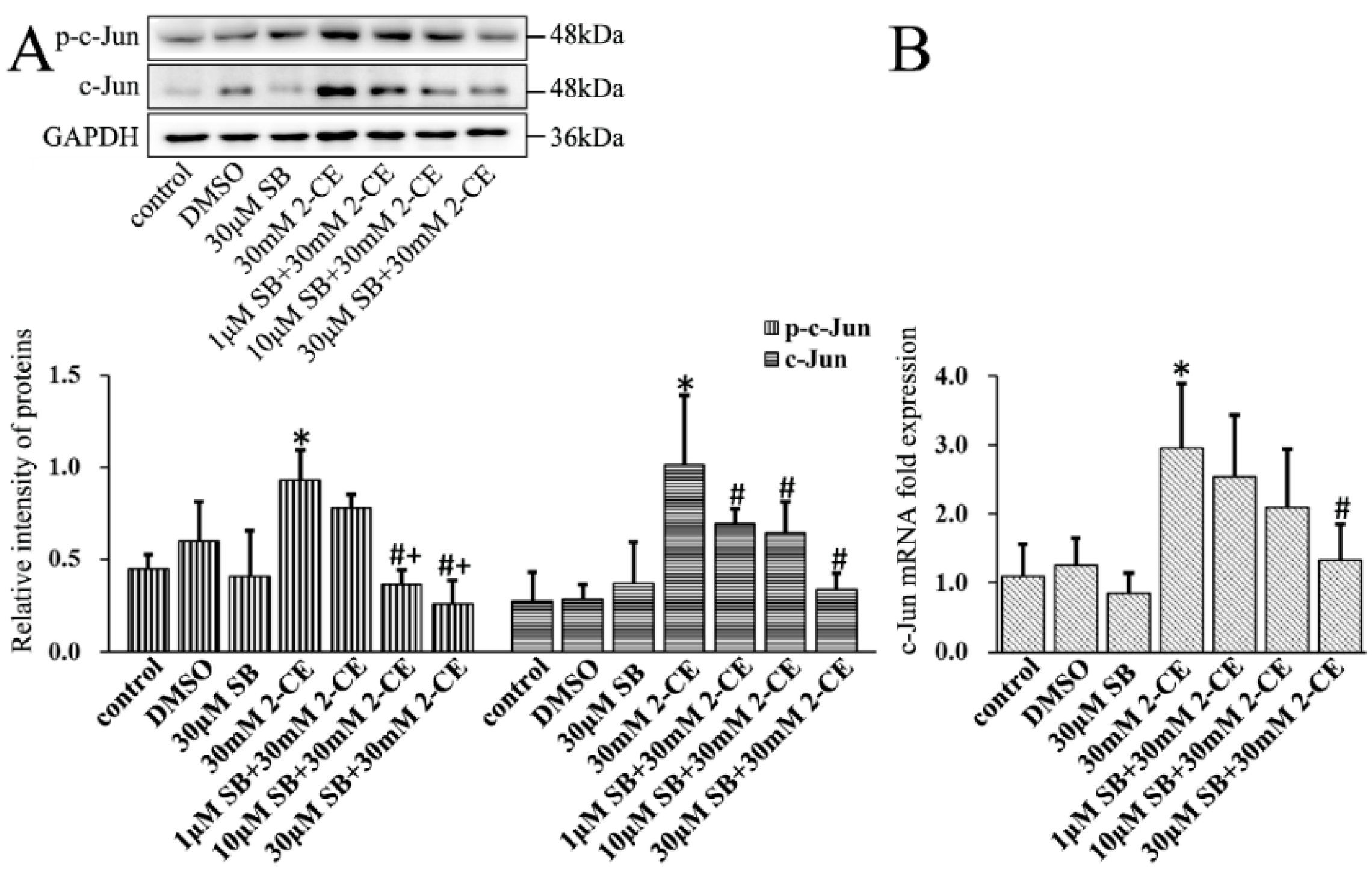
3.4. Upregulated Expression and Phosphorylation of c-Jun and c-Fos in 2-CE Treated Rat Astrocytes along with the Exposure Duration and Concentrations
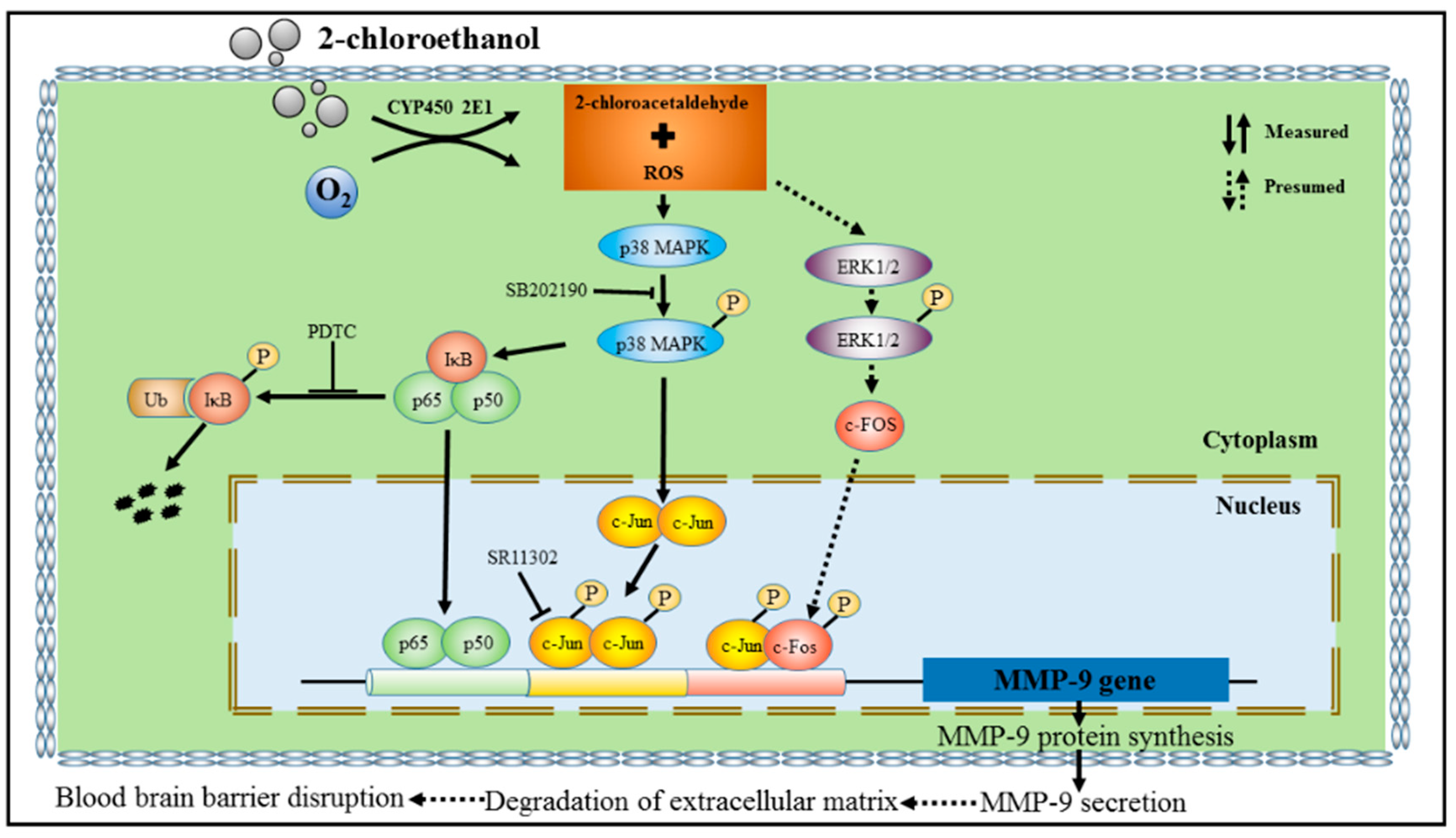
3.5. MMP-9 Overexpression Mediated by NF-κB and AP-1 in 2-CE-Treated Astrocytes
3.6. Roles of p38 MAPK Signaling Pathway in NF-κB and AP-1 Activation in 2-CE Treated Astrocytes
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| 1,2-DCE | 1,2-dichloroethane |
| 2-CE | 2-chloroethanol |
| NF-κB | nuclear factor kappa B |
| IκBα | inhibitor of κBα |
| AP-1 | activator protein-1 |
| MMP-9 | matrix metalloproteinase-9 |
| MAPK | mitogen-activated protein kinase |
| PDTC | pyrrolidine dithiocarbamate |
| SR | SR11302 |
| SB | SB202190 |
References
- Liu, J.R.; Fang, S.; Ding, M.P.; Chen, Z.C.; Zhou, J.J.; Sun, F. Toxic encephalopathy caused by occupational exposure to 1,2-Dichloroethane. J. Neurol. Sci. 2010, 292, 111–113. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Niu, Q.; Li, L.Y.; Yang, L.; Guo, X.L.; Huang, J.X. Establishment of a poisoned animal model of toxic encephalopathy induced by 1,2-dichloroethane. Int. J. Immunopathol. Pharmacol. 2011, 24, 79S–83S. [Google Scholar] [PubMed]
- Chen, S.; Zhang, Z.; Lin, H.; Chen, Z.; Wang, Z.; Wang, W. 1,2-Dichloroethane-induced toxic encephalopathy: a case series with morphological investigations. J. Neurol. Sci. 2015, 351, 36–40. [Google Scholar] [CrossRef] [PubMed]
- Guengerich, F.P.; Crawford, W.M., Jr.; Domoradzki, J.Y.; Macdonald, T.L.; Watanabe, P.G. In vitro activation of 1,2-dichloroethane bymicrosomal and cytosolic enzymes. Toxicol. Appl. Pharmacol. 1980, 55, 303–317. [Google Scholar] [CrossRef]
- Igwe, O.J.; Que Hee, S.S.; Wagner, W.D. Inhalation pharmacokinetics of 1,2-dichloroethane after different dietary pretreatments of male Sprague-Dawley rats. Arch. Toxicol. 1986, 59, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Raucy, J.L.; Kraner, J.C.; Lasker, J.M. Bioactivation of halogenated hydrocarbons by cytochrome P4502E1. Crit. Rev. Toxicol. 1993, 23, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Liao, Y.; Sun, Q.; Tang, H.; Wang, G.; Zhao, F.; Jin, Y. Upregulation of Matrix Metalloproteinase-9 in Primary Cultured Rat Astrocytes Induced by 2-Chloroethanol Via MAPK Signal Pathways. Front. Cell. Neurosci. 2017, 11, 218. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Yuan, Y.; Zhang, J.; Gao, L.; Tan, X.; Yang, G.; Lv, X.; Jin, Y. Roles of aquaporins and matrix metalloproteinases in mouse brain edema formation induced by subacute exposure to 1,2-dichloroethane. Neurotoxicol. Teratol. 2014, 44, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Haorah, J.; Ramirez, S.H.; Schall, K.; Smith, D.; Pandya, R.; Persidsky, Y. Oxidative stress activates protein tyrosinkinase and matrix metalloproteinases leading to blood-brain barrier dysfunction. J. Neurochem. 2007, 101, 566–576. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, G.A. Matrix metalloproteinases in neuroinflammation. Glia 2002, 39, 279–291. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosell, A.; Ortega-Aznar, A.; Alvarez-Sabín, J.; Fernández-Cadenas, I.; Ribó, M.; Molina, C.A.; Lo, E.H.; Montaner, J. Increased brain expression of matrix metalloproteinase-9 after ischemic and hemorrhagic human stroke. Stroke 2006, 37, 1399–1406. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.; Liao, Y.; Wang, T.; Tang, H.; Wang, G.; Zhao, F.; Jin, Y. 2-Chloroethanol induced upregulation of matrix metalloproteinase-2 in primary cultured rat astrocytes via MAPK signal pathways. Front. Neurosci. 2017, 10, 593. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.H.; Hsieh, H.L.; Wu, C.Y.; Sun, C.C.; Yang, C.M. Oxidized low-density lipoprotein induces matrix metalloproteinase-9 expression via a p42/p44 and JNK-dependent AP-1 pathway in brain astrocytes. Glia 2009, 57, 24–38. [Google Scholar] [CrossRef] [PubMed]
- Speidl, W.S.; Kastl, S.P.; Hutter, R.; Katsaros, K.M.; Kaun, C.; Bauriedel, G.; Maurer, G.; Huber, K.; Badimon, J.J.; Wojta, J. The complement component C5a is present in human coronary lesions in vivo and induces the expression of MMP-1 and MMP-9 in human macrophages in vitro. FASEB J. 2011, 25, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.P.; Yun, H.J.; Choi, J.H.; Han, E.H.; Kim, H.G.; Song, G.Y.; Kwon, K.I.; Jeong, T.C.; Jeong, H.G. Suppression of EGF-induced tumor cell migration and matrix metalloproteinase-9 expression by capsaicin via the inhibition of EGFR-mediated FAK/Akt, PKC/Raf/ERK, p38 MAPK, and AP-1. Mol. Nutr. Food Res. 2011, 55, 594–605. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Kwak, C.H.; Ha, S.H.; Kwon, K.M.; Abekura, F.; Cho, S.H.; Chang, Y.C.; Lee, Y.C.; Ha, K.T.; Chung, T.W.; et al. Ganglioside GM3 suppresses lipopolysaccharide-induced inflammatory responses in rAW 264.7 macrophage cells through NF-κB, AP-1, and MAPKs signaling. J. Cell. Biochem. 2018, 119, 1173–1182. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, C.C.; Papaconstantinou, J. Akt/PKB and p38 MAPK signaling, translational initiation and longevity in Snell dwarf mouse livers. Mech. Ageing Dev. 2004, 125, 785–798. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.Y.; Hsieh, H.L.; Jou, M.J.; Yang, C.M. Involvement of p42/p44 MAPK, p38 MAPK, JNK and nuclear factor-kappa B in interleukin-1beta-induced matrix metalloproteinase-9 expression in rat brain astrocytes. J. Neurochem. 2004, 90, 1477–1488. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Herbein, G.; Varin, A.; Fulop, T. NF-kB, AP-1, Zinc-deficiency and aging. Biogerontology 2006, 7, 409–419. [Google Scholar] [CrossRef] [PubMed]
- Gosselin, K.; Abbadie, C. Involvement of Rel/NF-kappa B transcription factors in senescence. Exp. Gerontol. 2003, 38, 1271–1283. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Kumar, A. Differential signaling mechanism for HIV-1 Nef-mediated production of IL-6 and IL-8 in human astrocytes. Sci. Rep. 2015, 5, 9867. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shih, R.H.; Wang, C.Y.; Yang, C.M. NF-kappaB Signaling Pathways in Neurological Inflammation: A Mini Review. Front. Mol. Neurosci. 2015, 8, 77. [Google Scholar] [CrossRef] [PubMed]
- Carter, A.B.; Knudtson, K.L.; Monick, M.M.; Hunninghake, G.W. The p38 mitogen-activated protein kinase is required for NF-kappa B-dependent gene expression. The role of TATA-binding protein (TBP). J. Biol. Chem. 1999, 274, 30858–30863. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.W.; Feng, W.; Peh, M.T.; Peh, K.; Dymock, B.W.; Moore, P.K. A novel slow-releasing hydrogen sulfide donor, FW1256, exerts anti-inflammatory effects in mouse macrophages and in vivo. Pharmacol. Res. 2016, 113, 533–546. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.; Huang, Y.; Ning, B.; Gong, W.; Li, J.; Wang, H.; Chen, C.Y.; Huang, C. TNF-alpha induction by nickel compounds is specific through ERKs/AP-1-dependent pathway in human bronchial epithelial cells. Curr. Cancer Drug Targets 2009, 9, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Karin, M.; Liu, Z.; Zandi, E. AP-1 function and regulation. Curr. Opin. Cell Biol. 1997, 9, 240–246. [Google Scholar] [CrossRef]
- Shaulian, E.; Karin, M. AP-1 as a regulator of cell life and death. Nat. Cell Biol. 2002, 4, E131. [Google Scholar] [CrossRef] [PubMed]
- Bogoyevitch, M.A.; Kobe, B. Uses for JNK: The many and varied substrates of the c-Jun N-terminal kinases. Microbiol. Mol. Biol. Rev. 2006, 70, 1061–1095. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miraglia, M.C.; Scian, R.; Samartino, C.G.; Barrionuevo, P.; Rodriguez, A.M.; Ibañez, A.E.; Coria, L.M.; Velásquez, L.N.; Baldi, P.C.; Cassataro, J.; et al. Brucella abortus induces TNF-α-dependent astroglial MMP-9 secretion through mitogen-activated protein kinases. J. Neuroinflamm. 2013, 10, 819. [Google Scholar] [CrossRef] [PubMed]
- Lappas, M.; Riley, C.; Lim, R.; Barker, G.; Rice, G.E.; Menon, R.; Permezel, M. MAPK and AP-1 proteins are increased in term pre-labour fetal membranes overlying the cervix: Regulation of enzymes involved in the degradation of fetal membranes. Placenta 2011, 32, 1016–1025. [Google Scholar] [CrossRef] [PubMed]
- Lian, S.; Xia, Y.; Khoi, P.N.; Ung, T.T.; Yoon, H.J.; Kim, N.H.; Kim, K.K.; Jung, Y.D. Cadmium induces matrix metalloproteinase-9 expression via ROS-dependent EGFR, NF-κB, and AP-1 pathways in human endothelial cells. Toxicology 2015, 338, 104–116. [Google Scholar] [CrossRef] [PubMed]

| Gene | Primer Sequence (5′-3′) | |
|---|---|---|
| MMP-9 | Forward | 5′-ATCCGCAGTCCAAGAAGATT-3′ |
| Reverse | 5′-GCCAGAGAACTCGTTATCCA-3′ | |
| p38 | Forward | 5′-CCGAGCGATACCAGAACCT-3′ |
| Reverse | 5′-AACACATCCAACAGACCAATCA-3′ | |
| p65 | Forward | 5′-TTAGCCATCATCCACCTTC-3′ |
| Reverse | 5′-AGTCCTCCACCACATCTT-3′ | |
| IκBα | Forward | 5′-GAGGATTACGAGCAGATGG-3′ |
| Reverse | 5′-ATGGTCAGTGTCTTCTCTTC-3′ | |
| c-Jun | Forward | 5′-ACGACCTTCTACGACGAT-3′ |
| Reverse | 5′-CATTGCTGGACTGGATGAT-3′ | |
| c-Fos | Forward | 5′-TCCGAAGGGAAAGGAATAAG-3′ |
| Reverse | 5′-AGTCAAGTCCAGGGAGGTC-3′ | |
| GAPDH | Forward | 5′-GCAAGAGAGAGGCCCTCAG-3′ |
| Reverse | 5′-TGTGAGGGAGATGCTCAGTG-3′ | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, T.; Jin, X.; Liao, Y.; Sun, Q.; Luo, C.; Wang, G.; Zhao, F.; Jin, Y. Association of NF-κB and AP-1 with MMP-9 Overexpression in 2-Chloroethanol Exposed Rat Astrocytes. Cells 2018, 7, 96. https://doi.org/10.3390/cells7080096
Wang T, Jin X, Liao Y, Sun Q, Luo C, Wang G, Zhao F, Jin Y. Association of NF-κB and AP-1 with MMP-9 Overexpression in 2-Chloroethanol Exposed Rat Astrocytes. Cells. 2018; 7(8):96. https://doi.org/10.3390/cells7080096
Chicago/Turabian StyleWang, Tong, Xiaoxia Jin, Yingjun Liao, Qi Sun, Chaohong Luo, Gaoyang Wang, Fenghong Zhao, and Yaping Jin. 2018. "Association of NF-κB and AP-1 with MMP-9 Overexpression in 2-Chloroethanol Exposed Rat Astrocytes" Cells 7, no. 8: 96. https://doi.org/10.3390/cells7080096




