Unique Responses are Observed in Transient Receptor Potential Ankyrin 1 and Vanilloid 1 (TRPA1 and TRPV1) Co-Expressing Cells
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
2.2. TRPV1, TRPA1, TRPA1V1 and pcDNA3 Control Cells
2.3. Measurement of Intracellular Calcium for Activation of TRA1V1, TRPA1 and TRPV1
3. Results and Discussion
3.1. Screening TRPA1V1, TRPA1 and TRPV1Receptor Containing Cells with 600 Compounds
3.1.1. Cell Culture Conditions

3.1.2. Identification of TRPA1, TRPV1 and TRPA1V1 Agonists
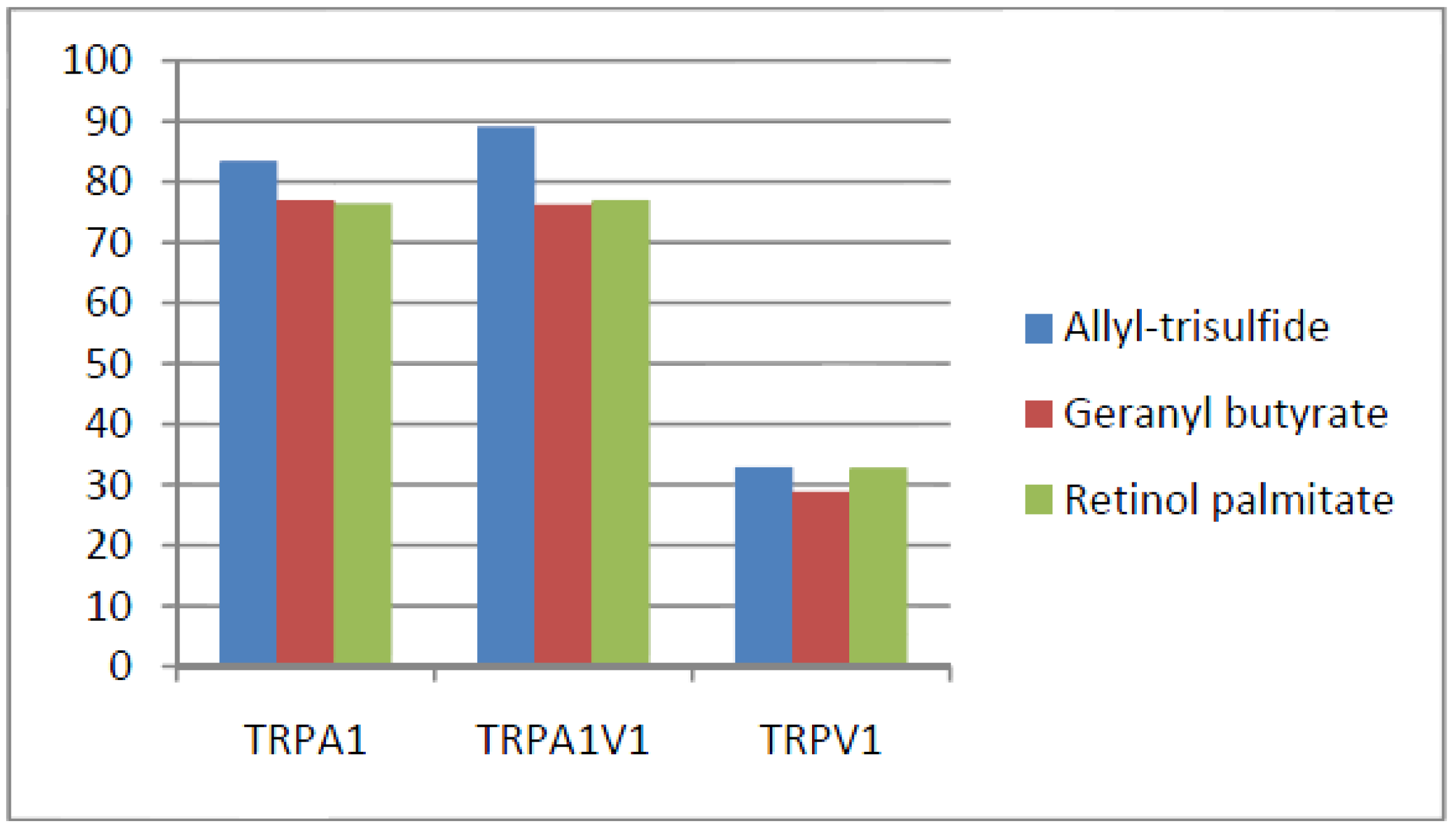
- Compounds that activated all three channels with TRPA1 = TRPA1V1 > TRPV1: We identified 18 compounds that activated TRPA1 and TRPA1V1 equally but had less agonist activity at TRPV1 (Figure 3). This result indicates that the TRPA1 constituent of the TRPA1V1 was the key driver of binding of this class of compounds.
- Compounds that activated all three channels with TRPA1 < TRPA1V1 = TRPV1: We identified 4 compounds that activated TRPV1 and TRPA1V1 equally but had less agonist activity at TRPA1 (Figure 4). This result indicates that the TRPV1 constituent of the TRA1V1 was the key driver of the binding of this class of compounds.
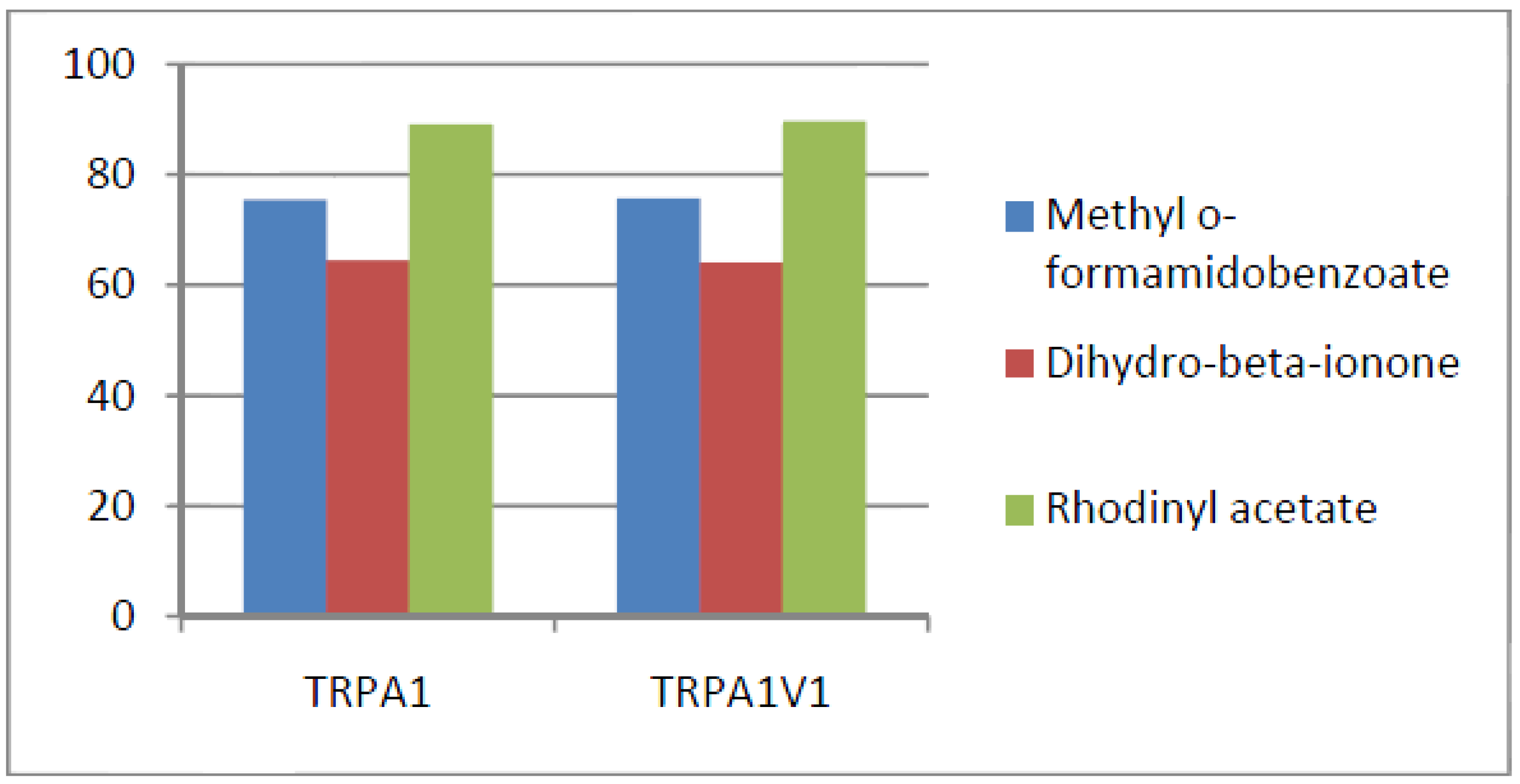
- Compounds that activate TRPA1 = TRPA1V1 but not TRPV1: We identified 66 compounds that activated TRPA1 and TRPA1V1 to similar levels but did not activate TRPV1 (Figure 5). This result indicates that the TRPA1 component of the TRPA1V1 was the key driver of binding of this class of compounds.
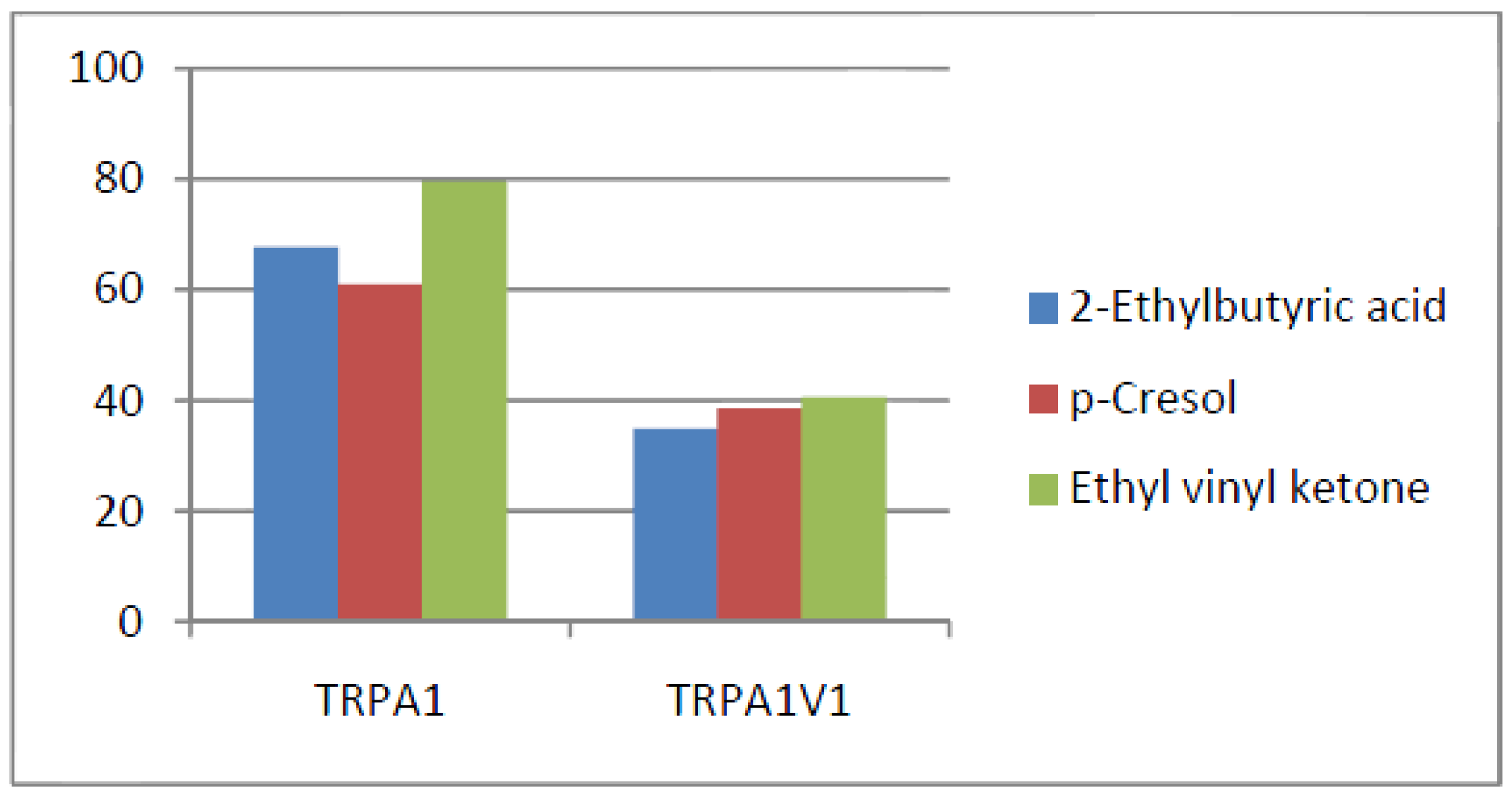
- Compounds that activate TRPV1 = TRPA1V1 but not TRPA1: We identified 6 compounds that activated TRPV1 and TRPA1V1 to similar levels but did not activate TRPA1 (Figure 6). This result indicates that the TRPV1 component of the TRPA1V1 was the key driver of binding of this class of compounds.
- Compounds that activate TRPA1 > TRPA1V1 but not TRPV1: We identified 13 compounds that activated TRPA1 to a greater extent than TRPA1V1 but did not activate TRPV1 (Figure 7). This result indicates that the TRPV1 component of the TRPA1V1 had a negative effect on binding of the compound to TRPA1V1.
- Compounds that activate TRPV1 > TRPA1V1 but not TRPA1: We identified 6 compounds that activated TRPV1 to a greater extent than TRPA1V1 but did not activate TRPA1 (Figure 8). This result indicates that the TRPA1 component of the TRPA1V1 had a negative effect on the binding of the compound to the TRPA1V1.
- Compounds that activate TRPA1 < TRPA1V1 but not TRPV1: We identified 17 compounds that activated the TRPA1V1 to a greater extent than TRPA1 but with no activity at the TRPV1 receptor (Figure 9). This finding indicates that the TRPV1 association with TRPA1 had a positive effect on compound activation of TRPA1V1.
- Compounds that activate TRPV1 < TRPA1V1 but not TRPA1: We identified 3 compounds that activate the TRPA1V1 to a greater extent than TRPV1 but with no activity at the TRPA1 receptor (Figure 10). This finding indicates that the TRPA1 association with TRPV1 had a positive effect on compound activation of the TRPA1V1.
- Compounds that activate all three TRP receptors with complex effects on TRPA1, TRPA1V1 and TRPV1: We identified 2 compounds that activated TRPA1 > TRPA1V1 > TRPV1, 2 compounds that activated TRPA1 < TRPA1V1 < TRPV1, 2 compounds that activated TRPA1 = TRPV1 > TRPA1V1 and 7 compounds that activated TRPA1V1 > TRPA1 or TRPV1 < TRPA1V1 (Table 1). These findings demonstrated that compounds can behave uniquely when they interact with TRPA1 and TRPV1 compared to the TRPA1V1.
- Compounds that activate either TRPA1 or TRPV1 or TRPA1V1 individually: We identified 1 compound that activated TRPA1 but not TRPA1V1 or TRPV1, 1 compound that activated TRPA1V1 but not TRPA1 or TRPV1 and 5 compounds that activated TRPV1 but not TRPA1V1 or TRPA1 (Table 2). This finding indicates that co-expression can change receptor activation and compound specificity.


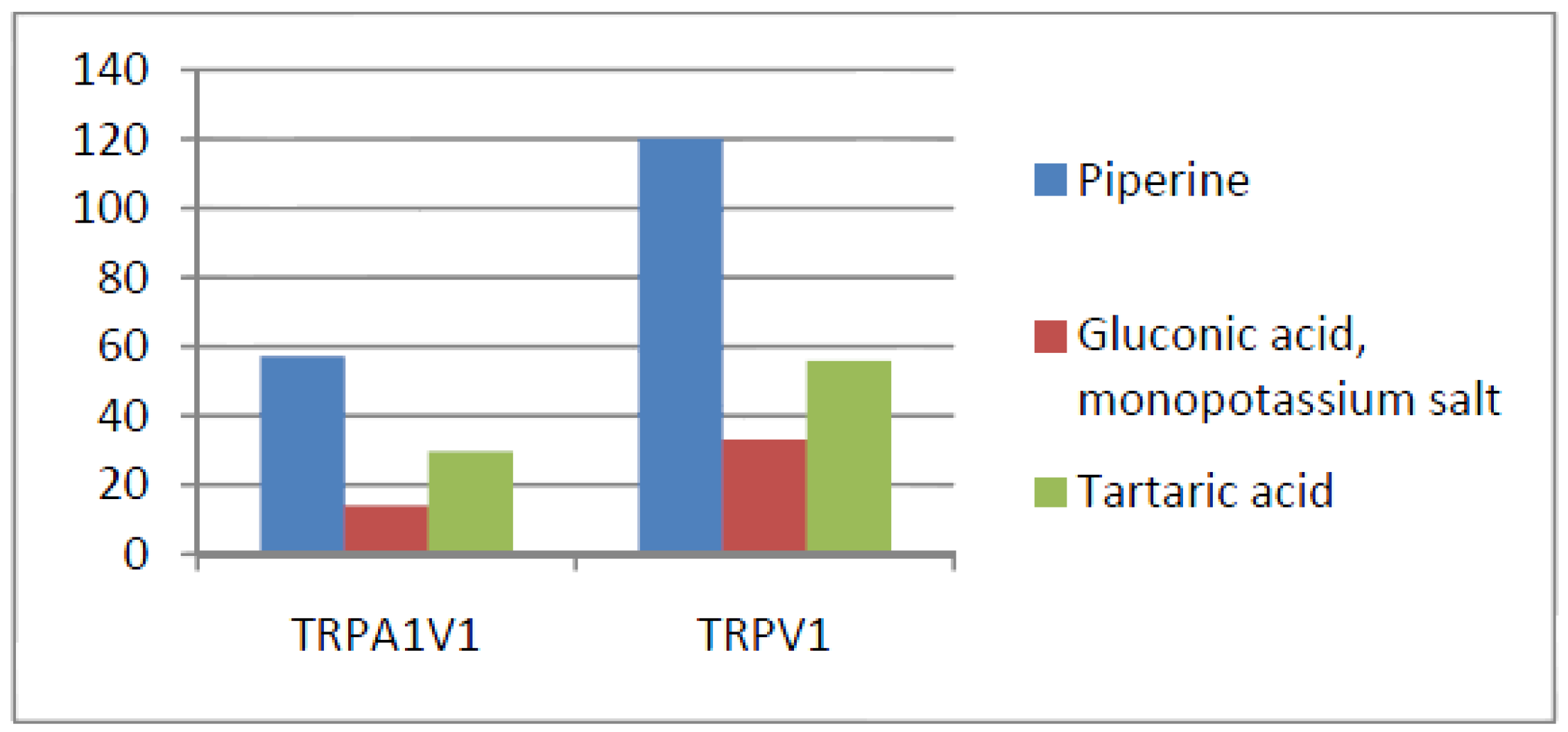

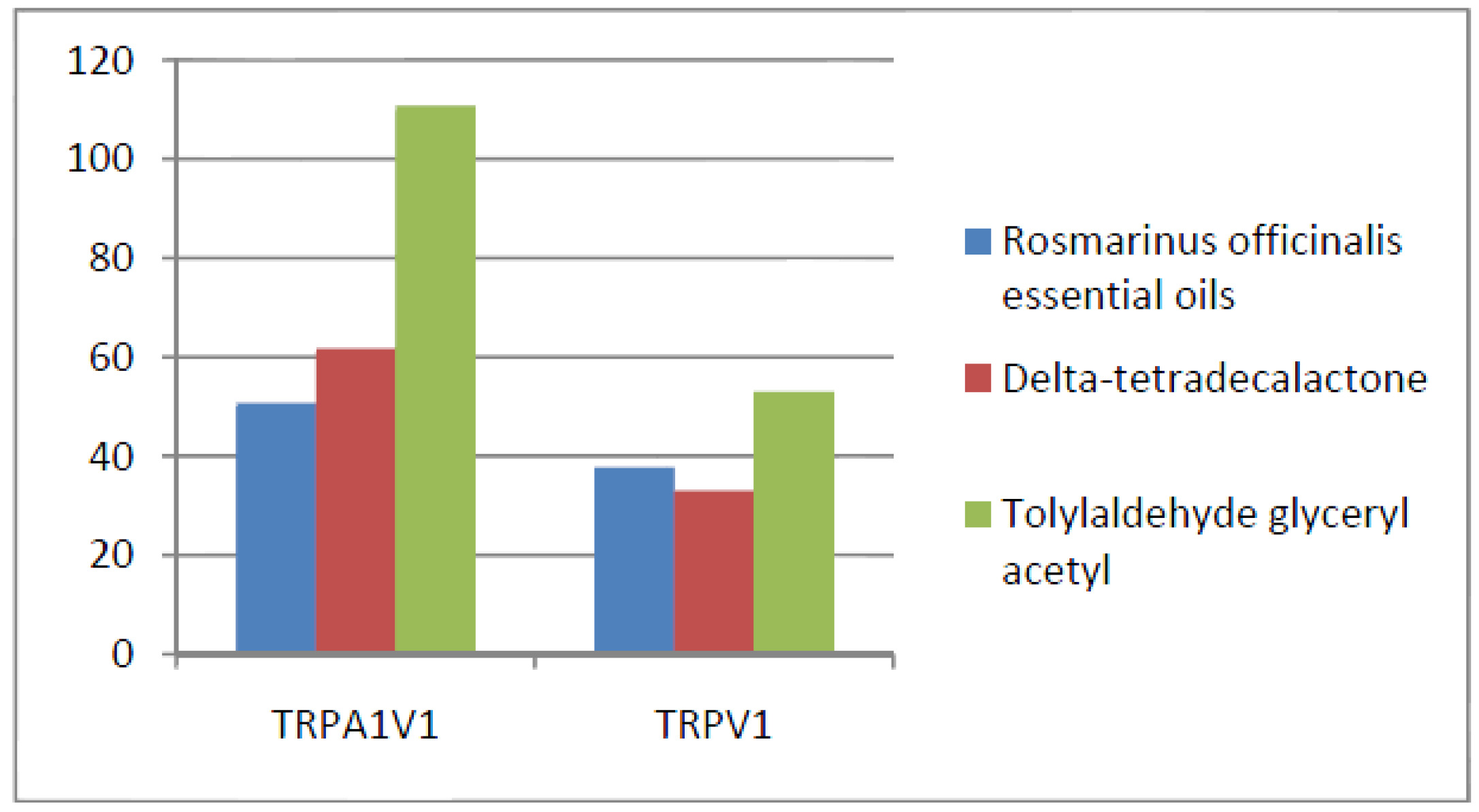
| Compound | TRPA1 | TRPA1V1 | TRPV1 | Remarks |
|---|---|---|---|---|
| Ethyl 3-hydroxyoctanoate | 97.29 | 76.90 | 28.05 | Negative effect of V1 |
| Geraniol | 82.52 | 63.93 | 42.16 | Negative effect of V1 |
| 5-Oxodecanoic acid | 28.57 | 39.34 | 53.38 | Negative effect of A1 |
| Vanillylacetone | 22.26 | 37.75 | 60.05 | Negative effect of A1 |
| 2-Napphlaenthiol | 66.3 | 85.2 | 120.3 | Negative effect of A1 |
| 3-Methyl-5-propyl-2-cyclohexen-1-one | 69.00 | 101.0 | 59.22 | A1 and V1 additive |
| Benzaldehyde propylene glycol acetal | 48.03 | 78.33 | 34.38 | A1 and V1 additive |
| Methyl cyclohexanecarboxylate | 45.50 | 85.17 | 69.51 | A1 and V1 additive |
| 4-Hydroxybenzaldehyde | 80.4 | 163.2 | 123.1 | A1 and V1 additive |
| Compound | TRPA1 | TRPA1V1 | TRPV1 |
|---|---|---|---|
| Propylparaben | 93.2 | 2.1 | 1.6 |
| Quercinitol | 1.3 | 57.6 | 2.1 |
| Malic acid | 1.9 | 2.4 | 67.5 |
| 2-Methylbutyl isovalerate | 1.7 | 2.1 | 75.3 |
| 2,4,5-Trimethyl-3-oxazoline | 2.1 | 4.1 | 100.7 |
| Heptaldehyde | 4.3 | 2.4 | 65.3 |
| 2-Ethylfuran | 1.2 | 1.8 | 58.7 |
3.1.3. Activity of TRPA1, TRPAV1 and TRPV1 Agonists towards TRPV3 and TRPM8
| Compound or Extract | TRPA1 | TRPA1V1 | TRPV1 | TRPV3 | TRPM8 |
|---|---|---|---|---|---|
| L-Piperitone | 45 | 68.4 | 0 | 0 | 81.6 |
| (+/−)-Piperitone | 90.1 | 109.1 | 53.3 | 0 | 45.6 |
| DL-Menthol | 54 | 43 | 0 | 0 | 78.4 |
| Tannic acid | 0 | 69.3 | 52.6 | 0 | 67.4 |
| Eugenol | 79 | 91.3 | 0 | 0 | 70.4 |
| Beta-cyclohomocitral | 81.4 | 66.8 | 61.7 | 76.5 | 70.7 |
| Rosemarinus officinalis essential oils | 37.7 | 50.7 | 0 | 85.6 | 0 |
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bautista, D.M.; Jordt, S.E.; Nikai, T.; Tsuruda, P.R.; Read, A.J.; Poblete, J.; Yamoah, E.N.; Basbaum, A.I.; Julius, D. TRPA1 mediates the inflammatory actions of environmental irritants and proalgesic agents. Cell 2006, 124, 1269–1282. [Google Scholar] [CrossRef]
- Birrell, M.A.; Belvisi, M.G.; Grace, M.; Sadofsky, L.; Faruqi, S.; Hele, D.J.; Maher, S.A.; Freund-Michel, V.; Morice, A.H. TRPA1 agonists evoke coughing in guinea-pig and human volunteers. Am. J. Resp. Crit. Care Med. 2009, 180, 1024. [Google Scholar]
- Caterina, M.J.; Schumacher, M.A.; Tominaga, M.; Rosen, T.A.; Levine, J.D.; Julius, D. The capsaicin receptor: A heat-activated ion channel in the pain pathway. Nature 1997, 389, 816–824. [Google Scholar] [CrossRef]
- Bandell, M.; Story, G.M.; Hwang, S.W.; Viswanath, V.; Eid, S.R.; Petrus, M.J.; Earley, T.J.; Patapoutian, A. Noxious cold ion channel TRPA1 is activated by pungent compounds and bradykinin. Neuron 2004, 41, 849–857. [Google Scholar] [CrossRef]
- Story, G.M.; Peier, A.M.; Reeve, A.J.; Eid, S.R.; Mosbacher, J.; Hricik, T.R.; Earley, T.J.; Hergarden, A.C.; Andersson, D.A.; Hwang, S.W.; et al. ANKTM1, a TRP-like channel expressed in nociceptive neurons, is activated by cold temperatures. Cell 2003, 112, 819–829. [Google Scholar] [CrossRef]
- Jordt, S.E.; Bautista, D.M.; Chuang, H.H.; McKemy, D.D.; Zygmunt, P.M.; Högestätt, E.D.; Meng, I.D.; Julius, D. Mustard oils and cannabinoids excite sensory nerve fibres through the TRP channel ANKTM1. Nature 2004, 427, 260–268. [Google Scholar] [CrossRef]
- McNamara, C.R.; Mandel-Brehm, J.; Bautista, D.M.; Siemens, J.; Deranian, K.L.; Zhao, M.; Hayward, N.J.; Chong, J.A.; Julius, D.; Moran, M.M.; et al. TRPA1 mediates formalin-induced pain. Proc. Natl. Acad. Sci. 2007, 104, 13525–13530. [Google Scholar] [CrossRef]
- Kedei, N.; Szabo, T.; Lile, J.D.; Treanor, J.J.; Olah, Z.; Iadarola, M.J.; Blumberg, P.M. Analysis of the native quaternary structure of vanilloid receptor 1. J. Biol. Chem. 2001, 276, 28613–28619. [Google Scholar]
- Akopian, A.N.; Ruparel, N.B.; Jeske, N.A.; Hargreaves, K.M. Transient receptor potential TRPA1 channel desensitization in sensory neurons is agonist dependent and regulated by TRPV1-directed internalization. J. Physiol. 2007, 583, 175–193. [Google Scholar] [CrossRef]
- Akopian, A.N.; Ruparel, N.B.; Patwardhan, A.; Hargreaves, K.M. Cannabinoids desensitize capsaicin and mustard oil responses in sensory neurons via TRPA1 activation. J. Neurosci. 2008, 28, 1064–1075. [Google Scholar] [CrossRef]
- Macpherson, L.J.; Dubin, A.E.; Evans, M.J.; Marr, F.; Schultz, P.G.; Cravatt, B.F.; Patapoutian, A. Noxious compounds activate TRPA1 ion channels through covalent modification of cysteines. Nature 2007, 445, 541–545. [Google Scholar] [CrossRef]
- Ruparel, N.B.; Patwardhan, A.M.; Akopian, A.N.; Hargreaves, K.M. Homologous and heterologous desensitization of capsaicin and mustard oil responses utilize different cellular pathways in nociceptors. Pain 2008, 135, 271–279. [Google Scholar] [CrossRef]
- Staruschenko, A.; Jeske, N.A.; Akopian, A.N. Contribution of TRPV1-TRPA1 interaction to the single-channel properties of the TRPA1 channel. J. Biol. Chem. 2010, 285, 15167–15177. [Google Scholar] [CrossRef]
- Patil, M.J.; Jeske, N.A.; Akopian, A.N. TRPV1 regulates activation and modulation of TRPA1 by Ca2+. Neurosience 2010, 174, 1109–1119. [Google Scholar] [CrossRef]
- Sadofsky, L.R.; Campi, B.; Trevisani, M.; Compton, S.J.; Morice, A.H. Transient receptor potential vanilloid-1-mediated calcium responses are inhibited by the alkylamine antihistamines dexbrompheniramine and chlorpheniramine. Exp. Lung Res. 2008, 34, 681–693. [Google Scholar] [CrossRef]
- Mitchell, J.E.; Campbell, A.P.; New, N.E.; Sadofsky, L.R.; Kastelik, J.A.; Mulrennan, S.A.; Compton, S.J.; Morice, A.H. Expression and characterization of the intracellular vanilloid receptor (TRPV1) in bronchi from patients with chronic cough. Exp. Lung Res. 2005, 31, 295–306. [Google Scholar] [CrossRef]
- Sadofsky, L.R.; Sreekrishna, K.; Morice, A.H. Characterisation of a HEK293 cell line permanently co-expressing the cough receptors Transient Receptor Potential Ankyrin 1 and Vanilloid 1 (TRPA1 and TRPV1). In The Sixth London International Symposium on Cough: A translational approach, London, UK, 24–26 June 2010; Chung, K.F., Widdicombe, J., Eds.; Elsevier: London, UK.
- Smart, D.; Jerman, J.C.; Gunthorpe, M.J.; Brough, S.J.; Ranson, J.; Cairns, W.; Hayes, P.D.; Randall, A.D.; Davis, J.B. Characterization using FLIPR of human vanilloid VR1 receptor pharmacology. Eur. J. Pharmacol. 2001, 417, 51–58. [Google Scholar] [CrossRef]
- Zhang, Y.; Sreekrishna, K.; Lin, Y.; Huang, L.; Eickhoff, D.; Degenhardt, D.; Xu, T. Modulation of Transient Receptor Potential (TRP) Channels by Chinese Herbal Extracts. Phytother. Res. 2011, 25, 1666–1670. [Google Scholar] [CrossRef]
- Everaerts, W.; Gees, M.; Alpizar, Y.A.; Farre, R.; Leten, C.; Apetrei, A.; Dewachter, I.; van Leuven, F.; Vennekens, R.; de Ridder, D.; et al. The capsaicin receptor TRPV1 is a crucial mediator of the noxious effects of mustard oil. Curr. Biol. 2011, 21, 316–321. [Google Scholar] [CrossRef]
- Capasso, R.; Aviello, G.; Romano, B.; Borrelli, F.; de Petrocellis, L.; di Marzo, V.; Izzo, A.A. Modulation of mouse gastrointestinal motility by allyl isothiocyanate, a constituent of cruciferous vegetables (Brassicaceae): Evidence for TRPA1-independent effects. Br. J. Pharmacol. 2012, 165, 1966–1977. [Google Scholar] [CrossRef]
- Son, H.J.; Kim, Y.; Misaka, T.; Noh, B.S.; Rhyu, M.R. Activation of the chemosensory ion channels TRPA1 and TRPV1 by hydroalcohol extract of Kalopanax. pictus leaves. Biomol. Ther. 2012, 20, 550–555. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sadofsky, L.R.; Sreekrishna, K.T.; Lin, Y.; Schinaman, R.; Gorka, K.; Mantri, Y.; Haught, J.C.; Huggins, T.G.; Isfort, R.J.; Bascom, C.C.; et al. Unique Responses are Observed in Transient Receptor Potential Ankyrin 1 and Vanilloid 1 (TRPA1 and TRPV1) Co-Expressing Cells. Cells 2014, 3, 616-626. https://doi.org/10.3390/cells3020616
Sadofsky LR, Sreekrishna KT, Lin Y, Schinaman R, Gorka K, Mantri Y, Haught JC, Huggins TG, Isfort RJ, Bascom CC, et al. Unique Responses are Observed in Transient Receptor Potential Ankyrin 1 and Vanilloid 1 (TRPA1 and TRPV1) Co-Expressing Cells. Cells. 2014; 3(2):616-626. https://doi.org/10.3390/cells3020616
Chicago/Turabian StyleSadofsky, Laura R., Koti T. Sreekrishna, Yakang Lin, Renee Schinaman, Kate Gorka, Yogita Mantri, John Christian Haught, Thomas G. Huggins, Robert J. Isfort, Charles C. Bascom, and et al. 2014. "Unique Responses are Observed in Transient Receptor Potential Ankyrin 1 and Vanilloid 1 (TRPA1 and TRPV1) Co-Expressing Cells" Cells 3, no. 2: 616-626. https://doi.org/10.3390/cells3020616




