Neuroantigen-Specific CD4 Cells Expressing Interferon-γ (IFN-γ), Interleukin (IL)-2 and IL-3 in a Mutually Exclusive Manner Prevail in Experimental Allergic Encephalomyelitis (EAE)
Abstract
:Abbreviations
| ELISPOT | enzyme-linked immunospot |
| EAE | experimental autoimmune encephalomyelitis |
| PLP | proteolipid protein |
| CNS | central nervous system |
| TCR | T cell receptor |
| PLP | proteolipid protein |
| PBST | phosphate buffer saline with tween-20 |
| dLN | draining lymph nodes |
| HRP | horseradish peroxidase |
| ALPH | alkaline phosphatase |
| SFU | spot-forming units |
| MS | multiple sclerosis |
| IL | interleukin |
| Th | T helper |
| IFN | interferon |
| IFA | incomplete Freund's adjuvant |
| TNF | tumor necrosis factor |
| CNS | central nerve system |
| APC | antigen presenting cell |
| CFA | complete Freund's adjuvant |
| ND | not defined |
1. Introduction
2. Results
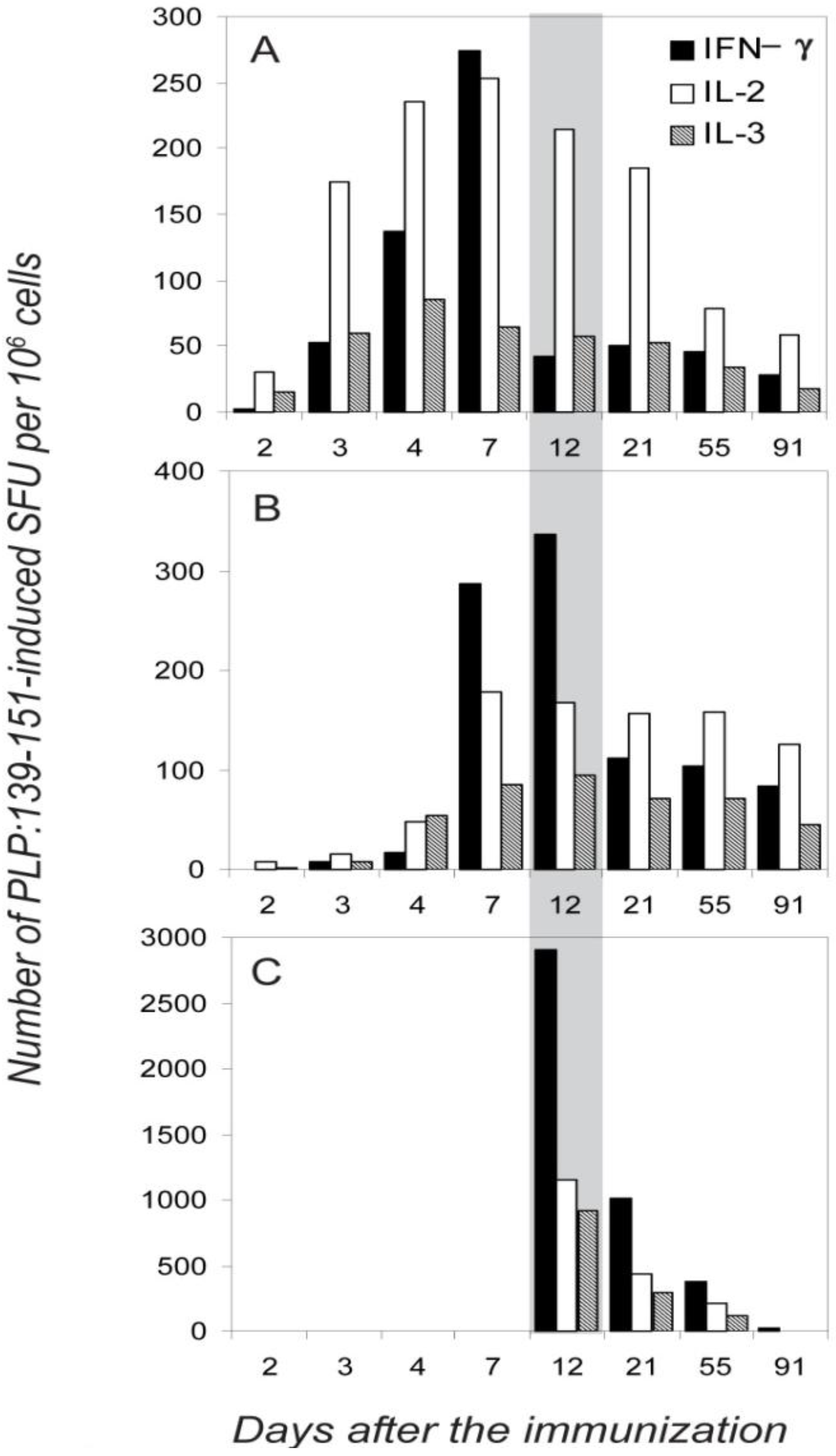
2.1. The Type 1 Cytokine Signature of the PLP: 139-151-Specific CD4 Cell Population; IL-4 Bystander Reaction in Splenic APC

| Time | Disease | Organ | IFN-γ/IL-2 assay | IFN-γ /IL-3 assay | IFN-γ/IL-4 assay | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (days) | state | IFN-γ | IL-2 | DP | %DP | IFN-γ | IL-3 | DP | %DP | IFN-γ | IL-4 | DP | %DP | |
| 2 | No disease | LN | 4 | 29 | 3 | 8.3 | 5 | 6 | 1 | 8.3 | 5 | 9 | 0 | 0.0 |
| Spleen | 2 | 6 | 0 | 0.0 | 4 | 2 | 0 | 0.0 | 3 | 47 ** | 0 | 0.0 | ||
| CNS * | 0 | 0 | 0 | 0.0 | 0 | 0 | 0 | 0.0 | 0 | 0 | 0 | 0.0 | ||
| 3 | No disease | LN | 41 | 104 | 12 | 7.6 | 59 | 62 | 8 | 6.2 | 51 | 14 | 0 | 0.0 |
| Spleen | 6 | 23 | 1 | 3.3 | 4 | 7 | 0 | 0.0 | 9 | 127 ** | 0 | 0.0 | ||
| CNS * | 0 | 0 | 0 | 0.0 | 0 | 0 | 0 | 0.0 | 0 | 0 | 0 | 0.0 | ||
| 7 | No disease | LN | 224 | 213 | 21 | 4.6 | 232 | 62 | 8 | 2.6 | 240 | 9 | 1 | 0.0 |
| Spleen | 356 | 217 | 23 | 3.9 | 335 | 86 | 9 | 2.1 | 349 | 218 ** | 7 | 0.0 | ||
| CNS * | 1 | 2 | 0 | 0.0 | 3 | 0 | 0 | 0.0 | 1 | 0 | 0 | 0.0 | ||
| 12 | First paralysis | LN | 46 | 183 | 7 | 3.0 | 43 | 36 | 5 | 5.9 | 52 | 7 | 0 | 0.0 |
| Spleen | 408 | 210 | 22 | 3.4 | 397 | 114 | 13 | 2.5 | 429 | 236 ** | 4 | 0.0 | ||
| CNS* | 932 | 374 | 49 | 3.6 | 971 | 372 | 47 | 3.4 | 938 | 8 | 1 | 0.0 | ||
| 21 | Second paralysis | LN | 45 | 139 | 4 | 2.1 | 52 | 53 | 6 | 5.4 | 47 | 6 | 0 | 0.0 |
| Spleen | 116 | 202 | 7 | 2.2 | 115 | 71 | 9 | 4.6 | 121 | 223 ** | 2 | 0.0 | ||
| CNS * | 307 | 108 | 11 | 2.6 | 288 | 103 | 10 | 2.5 | 296 | 14 | 1 | 0.0 | ||
| 55 | After three relapses | LN | 47 | 76 | 5 | 3.9 | 49 | 33 | 2 | 2.4 | 41 | 3 | 0 | 0.0 |
| Spleen | 140 | 201 | 12 | 3.4 | 151 | 74 | 11 | 4.7 | 148 | 192 ** | 2 | 0.1 | ||
| CNS * | 119 | 62 | 8 | 4.2 | 107 | 42 | 5 | 3.2 | 111 | 4 | 0 | 0.0 | ||

2.2. Dissociated Expression of IL-2,-3,-4 and IFN-γ in Individual PLP: 139-151-Specific CD4 Cells Prevails in the Inductive Phase of the Autoimmune Response
2.3. Segregated Expression of Type-1 Cytokines Occurs as Early as 2 Days after the Immunization
2.4. Dissociated Expression of IL-2,-3,-4 and IFN-γ in Individual PLP: 139-151-Specific T Cells is Maintained during Relapsing EAE

2.5. Dissociated Cytokine Expression in PLP: 139-151-Specific CD4 Cells is Independent of the Antigen Dose

| Day 7 | Day 12 | Day 21 | Day 55 | ||
|---|---|---|---|---|---|
| No clinical disease | During the first paralysis | During second paralysis | After three relapses of EAE | ||
| dLN | IL-2 | 6 ± 2 | 5 ± 1 | 6 ± 3 | 5 ± 1 |
| IL-3 | 5 ± 1 | 8 ± 2 | 5 ± 1 | 6 ± 2 | |
| IFN-g | 4 ± 1 | 7 ± 2 | 9 ± 3 | 7 ± 3 | |
| Spleen | IL-2 | 4 ± 2 | 4 ± 2 | 5 ± 2 | 6 ± 2 |
| IL-3 | 3 ± 1 | 6 ± 2 | 6 ± 3 | 5 ± 3 | |
| IFN-g | 3 ± 1 | 3 ± 1 | 5 ± 2 | 6 ± 2 | |
| CNS | IL-2 | ND | 7 ± 2 | 8 ± 4 | ND |
| IL-3 | ND | 7 ± 2 | 8 ± 3 | ND | |
| IFN-g | ND | 5 ± 2 | 7 ± 4 | ND | |
| CNS plus APC | IL-2 | ND | 7 ± 2 | 6 ± 3 | ND |
| IL-3 | ND | 7 ± 3 | 7 ± 2 | ND | |
| IFN-g | ND | 4 ± 1 | 6 ± 2 | ND | |
2.6. The Functional Avidity of PLP: 139-151-Specific T Cells is Constant over the Course of EAE
2.7. Different Distribution of the IL-2, IL-3 and IFN-γ -Producing Cells between the Immune Periphery and CNS
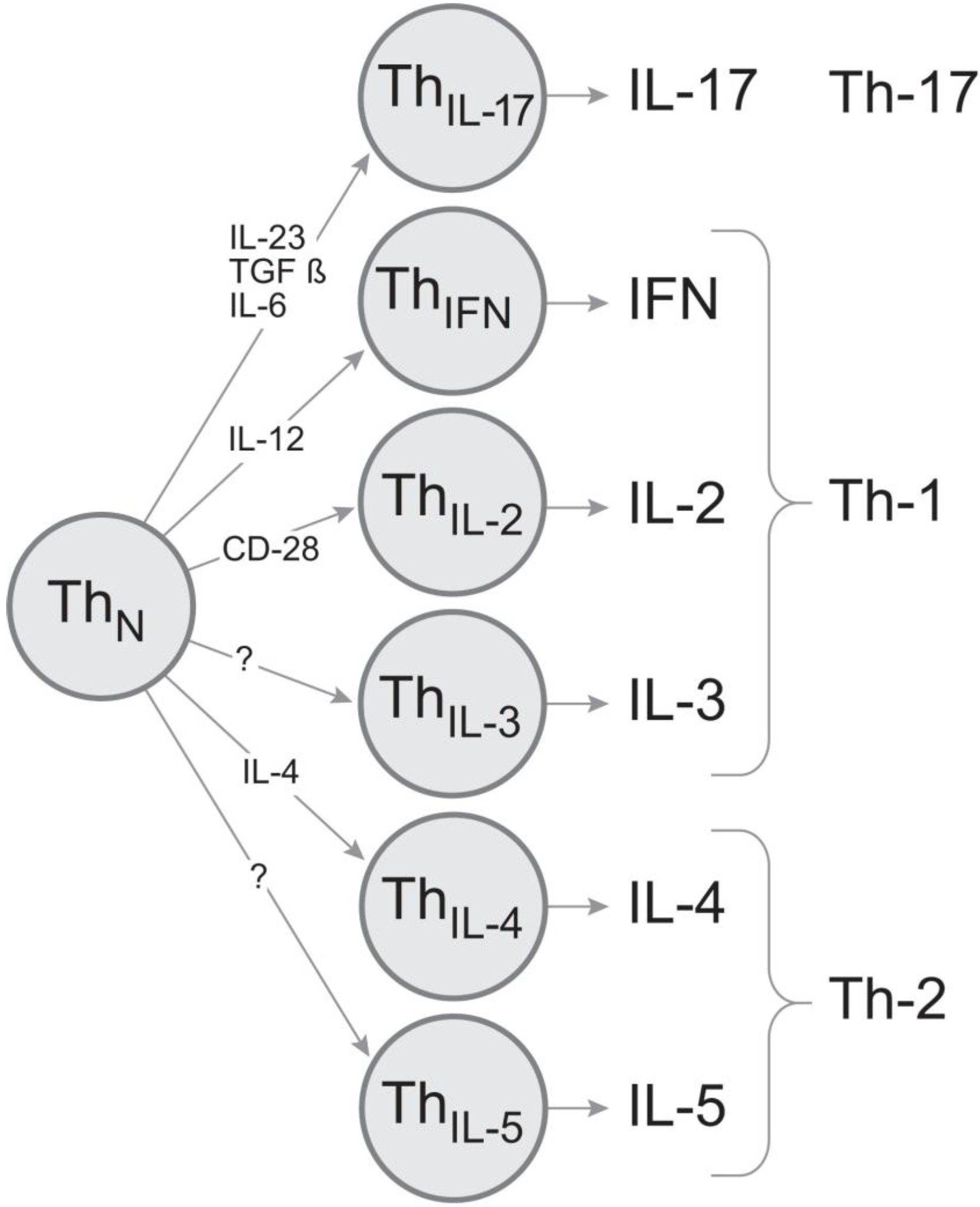
3. Discussion
4. Experimental Section
4.1. Animals and Immunizations
4.2. Cell Preparations
4.3. Two-Color Cytokine ELISPOT Assays
4.4. Measurements of Functional Avidity
5. Conclusions
References
- Murphy, K.; Ouyang, W.; Farrar, J.; Yang, J.; Ranganath, S.; Asnagli, H.; Afkarian, M.; Murphy, T. Signaling and transcription in T helper development. Annu. Rev. Immunol. 2000, 18, 451–494. [Google Scholar] [CrossRef]
- Han, Q.; Bagheri, N.; Bradshaw, E.; Hafler, D.; Lauffenburger, D.; Love, J. Polyfunctional responses by human T cells result from sequential release of cytokines. Proc. Natl. Acad. Sci. USA 2012, 109, 1607–1612. [Google Scholar]
- Kryczek, I.; Banerjee, M.; Cheng, P.; Vatan, L.; Szeliga, W.; Wei, S.; Huang, E.; Finlayson, E.; Simeone, D.; Welling, T.; et al. Phenotype, distribution, generation, and functional and clinical relevance of Th17 cells in the human tumor environments. Blood 2009, 114, 1141–1149. [Google Scholar]
- Bucy, R.; Karr, L.; Huang, G.; Li, J.; Carter, D.; Honjo, K.; Lemons, J.; Murphy, K.; Weaver, C. Single cell analysis of cytokine gene coexpression during CD4+ T-cell phenotype development. Proc. Natl. Acad. Sci. USA 1995, 92, 7565–7569. [Google Scholar]
- Saparov, A.; Elson, C.; Devore-Carter, D.; Bucy, R.; Weaver, C. Single-cell analyses of CD4+ T cells from alpha beta T cell receptor- transgenic mice: A distinct mucosal cytokine phenotype in the absence of transgene-specific antigen. Eur. J. Immunol. 1997, 27, 1774–1781. [Google Scholar] [CrossRef]
- Karulin, A.; Hesse, M.; Tary-Lehmann, M.; Lehmann, P. Single-cytokine-producing CD4 memory cells predominate in type 1 and type 2 immunity. J. Immunol. 2000, 164, 1862–1872. [Google Scholar]
- Veiga-Fernandes, H.; Walter, U.; Bourgeois, A.; McLean, A.; Rocha, B. Response of naive and memory CD8+ T cells to antigen stimulation in vivo. Nat. Immunol. 2000, 1, 47–53. [Google Scholar] [CrossRef]
- Effros, R.; Pawelec, G. Replicative senescence of T cells: Does the Hayflick Limit lead to immune exhaustion? Immunol. Today 1997, 18, 450–454. [Google Scholar] [CrossRef]
- Bradl, M.; Schmid, M.; Wekerle, H. Clonotypic chromosomal aberrations in long-term lines of myelin- specific rat T lymphocytes. J. Neuroimmunol. 1989, 24, 95–104. [Google Scholar] [CrossRef]
- Betts, M.; Nason, M.; West, S.; de Rosa, S.; Migueles, S.; Abraham, J.; Lederman, M.; Benito, J.; Goepfert, P.; Connors, M.; et al. HIV nonprogressors preferentially maintain highly functional HIV-specific CD8+ T cells. Blood 2006, 107, 4781–4789. [Google Scholar] [CrossRef]
- Duvall, M.; Precopio, M.; Ambrozak, D.; Jaye, A.; McMichael, A.; Whittle, H.; Roederer, M.; Rowland-Jones, S.; Koup, R. Polyfunctional T cell responses are a hallmark of HIV-2 infection. Eur. J. Immunol. 2008, 38, 350–363. [Google Scholar] [CrossRef]
- Krol, L.; Stuchly, J.; Hubacek, P.; Keslova, P.; Sedlacek, P.; Stary, J.; Hrusak, O.; Kalina, T. Signature profiles of CMV-specific T-cells in patients with CMV reactivation after hematopoietic SCT. Bone Marrow Transplant. 2011, 46, 1089–1098. [Google Scholar]
- Lachmann, R.; Bajwa, M.; Vita, S.; Smith, H.; Cheek, E.; Akbar, A.; Kern, F. Polyfunctional T cells accumulate in large human cytomegalovirus-specific T cell responses. J. Virol. 2012, 86, 1001–1009. [Google Scholar]
- Caccamo, N.; Guggino, G.; Joosten, S.; Gelsomino, G.; di Carlo, P.; Titone, L.; Galati, D.; Bocchino, M.; Matarese, A.; Salerno, A.; et al. Multifunctional CD4(+) T cells correlate with active Mycobacterium tuberculosis infection. Eur. J. Immunol. 2010, 40, 2211–2220. [Google Scholar]
- El Fenniri, L.; Toossi, Z.; Aung, H.; El Iraki, G.; Bourkkadi, J.; Benamor, J.; Laskri, A.; Berrada, N.; Benjouad, A.; Mayanja-Kizza, H.; et al. Polyfunctional Mycobacterium tuberculosis-specific effector memory CD4+ T cells at sites of pleural TB. Tuberculosis (Edinb) 2011, 91, 224–230. [Google Scholar] [CrossRef]
- Wilkinson, K.; Wilkinson, R. Polyfunctional T cells in human tuberculosis. Eur. J. Immunol. 2010, 40, 2139–2142. [Google Scholar] [CrossRef]
- Hilchey, S.; Rosenberg, A.; Hyrien, O.; Secor-Socha, S.; Cochran, M.; Brady, M.; Wang, J.; Sanz, I.; Burack, W.; Quataert, S.; et al. Follicular lymphoma tumor-infiltrating T-helper (T(H)) cells have the same polyfunctional potential as normal nodal T(H) cells despite skewed differentiation. Blood 2011, 118, 3591–3602. [Google Scholar] [CrossRef]
- Darrah, P.; Patel, D.; de Luca, P.; Lindsay, R.; Davey, D.; Flynn, B.; Hoff, S.; Andersen, P.; Reed, S.; Morris, S.; et al. Multifunctional TH1 cells define a correlate of vaccine-mediated protection against Leishmania major. Nat. Med. 2007, 13, 843–850. [Google Scholar]
- Gomez, C.; Najera, J.; Perdiguero, B.; Garcia-Arriaza, J.; Sorzano, C.; Jimenez, V.; Gonzalez-Sanz, R.; Jimenez, J.; Munoz-Fernandez, M.; Lopez Bernaldo de Quiros, J.C.; et al. The HIV/AIDS vaccine candidate MVA-B administered as a single immunogen in humans triggers robust, polyfunctional, and selective effector memory T cell responses to HIV-1 antigens. J. Virol. 2011, 85, 11468–11478. [Google Scholar]
- Lindenstrom, T.; Agger, E.; Korsholm, K.; Darrah, P.; Aagaard, C.; Seder, R.; Rosenkrands, I.; Andersen, P. Tuberculosis subunit vaccination provides long-term protective immunity characterized by multifunctional CD4 memory T cells. J. Immunol. 2009, 182, 8047–8055. [Google Scholar] [CrossRef]
- Makedonas, G.; Betts, M. Polyfunctional analysis of human t cell responses: Importance in vaccine immunogenicity and natural infection. Springer Semin. Immunopathol. 2006, 28, 209–219. [Google Scholar] [CrossRef]
- Precopio, M.; Betts, M.; Parrino, J.; Price, D.; Gostick, E.; Ambrozak, D.; Asher, T.; Douek, D.; Harari, A.; Pantaleo, G.; et al. Immunization with vaccinia virus induces polyfunctional and phenotypically distinctive CD8(+) T cell responses. J. Exp. Med. 2007, 204, 1405–1416. [Google Scholar]
- Rosa, D.; Ribeiro, S.; Almeida, R.; Mairena, E.; Postol, E.; Kalil, J.; Cunha-Neto, E. A DNA vaccine encoding multiple HIV CD4 epitopes elicits vigorous polyfunctional, long-lived CD4+ and CD8+ T cell responses. PLoS One 2011, 6, e16921. [Google Scholar]
- Hofstetter, H.; Toyka, K.; Tary-Lehmann, M.; Lehmann, P. Kinetics and organ distribution of IL-17-producing CD4 cells in proteolipid protein 139-151 peptide-induced experimental autoimmune encephalomyelitis of SJL mice. J. Immunol. 2007, 178, 1372–1378. [Google Scholar]
- Jager, A.; Dardalhon, V.; Sobel, R.; Bettelli, E.; Kuchroo, V. Th1, Th17, and Th9 effector cells induce experimental autoimmune encephalomyelitis with different pathological phenotypes. J. Immunol. 2009, 183, 7169–7177. [Google Scholar]
- Komiyama, Y.; Nakae, S.; Matsuki, T.; Nambu, A.; Ishigame, H.; Kakuta, S.; Sudo, K.; Iwakura, Y. IL-17 plays an important role in the development of experimental autoimmune encephalomyelitis. J. Immunol. 2006, 177, 566–573. [Google Scholar]
- Kroenke, M.; Segal, B. IL-23 modulated myelin-specific T cells induce EAE via an IFNgamma driven, IL-17 independent pathway. Brain Behav. Immunol. 2011, 25, 932–937. [Google Scholar] [CrossRef]
- Lovett-Racke, A.; Yang, Y.; Racke, M. Th1 versus Th17: Are T cell cytokines relevant in multiple sclerosis? Biochim. Biophys. Acta 2011, 1812, 246–251. [Google Scholar]
- Pollinger, B. IL-17 producing T cells in mouse models of multiple sclerosis and rheumatoid arthritis. J. Mol. Med. (Berl) 2012, 90, 613–624. [Google Scholar] [CrossRef]
- Harrington, L.; Hatton, R.; Mangan, P.; Turner, H.; Murphy, T.; Murphy, K.; Weaver, C. Interleukin 17-producing CD4+ effector T cells develop via a lineage distinct from the T helper type 1 and 2 lineages. Nat. Immunol. 2005, 6, 1123–1132. [Google Scholar] [CrossRef]
- Park, H.; Li, Z.; Yang, X.; Chang, S.; Nurieva, R.; Wang, Y.; Wang, Y.; Hood, L.; Zhu, Z.; Tian, Q.; et al. A distinct lineage of CD4 T cells regulates tissue inflammation by producing interleukin 17. Nat. Immunol. 2005, 6, 1133–1141. [Google Scholar]
- Tuohy, V.; Lu, Z.; Sobel, R.; Laursen, R.; Lees, M. Identification of an encephalitogenic determinant of myelin proteolipid protein for SJL mice. J. Immunol. 1989, 142, 1523–1527. [Google Scholar]
- Kuerten, S.; Rottlaender, A.; Rodi, M.; Velasco, V., Jr.; Schroeter, M.; Kaiser, C.; Addicks, K.; Tary-Lehmann, M.; Lehmann, P. The clinical course of EAE is reflected by the dynamics of the neuroantigen-specific T cell compartment in the blood. Clin. Immunol. 2010, 137, 422–432. [Google Scholar]
- Mosmann, T.; Cherwinski, H.; Bond, M.; Giedlin, M.; Coffman, R. Two types of murine helper T cell clone. I. Definition according to profiles of lymphokine activities and secreted proteins. J. Immunol. 1986, 136, 2348–2357. [Google Scholar]
- Pelfrey, C.; Rudick, R.; Cotleur, A.; Lee, J.; Tary-Lehmann, M.; Lehmann, P. Quantification of self-recognition in multiple sclerosis by single-cell analysis of cytokine production. J. Immunol. 2000, 165, 1641–1651. [Google Scholar]
- Heeger, P.; Forsthuber, T.; Shive, C.; Biekert, E.; Genain, C.; Hofstetter, H.; Karulin, A.; Lehmann, P. Revisiting tolerance induced by autoantigen in incomplete Freund's adjuvant. J. Immunol. 2000, 164, 5771–5781. [Google Scholar]
- Karulin, A.; Hesse, M.; Yip, H.; Lehmann, P. Indirect IL-4 pathway in type 1 immunity. J. Immunol. 2002, 168, 545–553. [Google Scholar]
- Kersh, E.; Shaw, A.; Allen, P. Fidelity of T cell activation through multistep T cell receptor zeta phosphorylation. Science 1998, 281, 572–575. [Google Scholar]
- Cai, Z.; Sprent, J. Influence of antigen dose and costimulation on the primary response of CD8+ T cells in vitro. J. Exp. Med. 1996, 183, 2247–2257. [Google Scholar]
- Itoh, Y.; Germain, R. Single cell analysis reveals regulated hierarchical T cell antigen receptor signaling thresholds and intraclonal heterogeneity for individual cytokine responses of CD4+ T cells. J. Exp. Med. 1997, 186, 757–766. [Google Scholar] [CrossRef]
- Targoni, O.; Lehmann, P. Endogenous myelin basic protein inactivates the high avidity T cell repertoire. J. Exp. Med. 1998, 187, 2055–2063. [Google Scholar] [CrossRef]
- Busch, D.; Pamer, E. T cell affinity maturation by selective expansion during infection. J. Exp. Med. 1999, 189, 701–710. [Google Scholar] [CrossRef]
- Savage, P.; Boniface, J.; Davis, M. A kinetic basis for T cell receptor repertoire selection during an immune response. Immunity. 1999, 10, 485–492. [Google Scholar]
- Padovan, E.; Giachino, C.; Cella, M.; Valitutti, S.; Acuto, O.; Lanzavecchia, A. Normal T lymphocytes can express two different T cell receptor beta chains: Implications for the mechanism of allelic exclusion. J. Exp. Med. 1995, 181, 1587–1591. [Google Scholar] [CrossRef]
- Roman, E.; Miller, E.; Harmsen, A.; Wiley, J.; von Andrian, U.; Huston, G.; Swain, S. CD4 effector T cell subsets in the response to influenza: Heterogeneity, migration, and function. J. Exp. Med. 2002, 196, 957–968. [Google Scholar] [CrossRef]
- Glickstein, L.; Huber, B. Karoushi--death by overwork in the immune system. J. Immunol. 1995, 155, 522–524. [Google Scholar]
- Renno, T.; Attinger, A.; Locatelli, S.; Bakker, T.; Vacheron, S.; MacDonald, H. Cutting edge: Apoptosis of superantigen-activated T cells occurs preferentially after a discrete number of cell divisions in vivo. J. Immunol. 1999, 162, 6312–6315. [Google Scholar]
- Lehmann, P. The fate of T cells in the brain: Veni, vidi, vici and veni, mori. Am. J. Pathol. 1998, 153, 677–680. [Google Scholar] [CrossRef]
- McKnight, A.; Perez, V.; Shea, C.; Gray, G.; Abbas, A. Costimulator dependence of lymphokine secretion by naive and activated CD4+ T lymphocytes from TCR transgenic mice. J. Immunol. 1994, 152, 5220–5225. [Google Scholar]
- Ahmadzadeh, M.; Hussain, S.; Farber, D. Heterogeneity of the memory CD4 T cell response: Persisting effectors and resting memory T cells. J. Immunol. 2001, 166, 926–935. [Google Scholar]
- Tuohy, V.; Lu, Z.; Sobel, R.; Laursen, R.; Lees, M. A synthetic peptide from myelin proteolipid protein induces experimental allergic encephalomyelitis. J. Immunol. 1988, 141, 1126–1130. [Google Scholar]
- Hofstetter, H.; Targoni, O.; Karulin, A.; Forsthuber, T.; Tary-Lehmann, M.; Lehmann, P. Does the frequency and avidity spectrum of the neuroantigen-specific T cells in the blood mirror the autoimmune process in the central nervous system of mice undergoing experimental allergic encephalomyelitis? J. Immunol. 2005, 174, 4598–4605. [Google Scholar]
- Targoni, O.; Baus, J.; Hofstetter, H.; Hesse, M.; Karulin, A.; Boehm, B.; Forsthuber, T.; Lehmann, P. Frequencies of neuroantigen-specific T cells in the central nervous system versus the immune periphery during the course of experimental allergic encephalomyelitis. J. Immunol. 2001, 166, 4757–4764. [Google Scholar]
- Ishikawa, E.; Imagawa, M.; Hashida, S.; Yoshitake, S.; Hamaguchi, Y.; Ueno, T. Enzyme-labeling of antibodies and their fragments for enzyme immunoassay and immunohistochemical staining. J. Immunoass. 1983, 4, 209–327. [Google Scholar] [CrossRef]
- Hesse, M.; Karulin, A.; Boehm, B.; Lehmann, P.; Tary-Lehmann, M. A T cell clone’s avidity is a function of its activation state. J. Immunol. 2001, 167, 1353–1361. [Google Scholar]
- McKenzie, G.; Fallon, P.; Emson, C.; Grencis, R.; McKenzie, A. Simultaneous disruption of interleukin (IL)-4 and IL-13 defines individual roles in T helper cell type 2-mediated responses. J. Exp. Med. 1999, 189, 1565–1572. [Google Scholar] [CrossRef]
- Zhang, D.; Yang, L.; Ray, A. Differential responsiveness of the IL-5 and IL-4 genes to transcription factor GATA-3. J. Immunol. 1998, 161, 3817–3821. [Google Scholar]
- Zhang, D.; Cohn, L.; Ray, P.; Bottomly, K.; Ray, A. Transcription factor GATA-3 is differentially expressed in murine Th1 and Th2 cells and controls Th2-specific expression of the interleukin-5 gene. J. Biol. Chem. 1997, 272, 21597–21603. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Karulin, A.Y.; Quast, S.; Hesse, M.D.; Lehmann, P.V. Neuroantigen-Specific CD4 Cells Expressing Interferon-γ (IFN-γ), Interleukin (IL)-2 and IL-3 in a Mutually Exclusive Manner Prevail in Experimental Allergic Encephalomyelitis (EAE). Cells 2012, 1, 576-596. https://doi.org/10.3390/cells1030576
Karulin AY, Quast S, Hesse MD, Lehmann PV. Neuroantigen-Specific CD4 Cells Expressing Interferon-γ (IFN-γ), Interleukin (IL)-2 and IL-3 in a Mutually Exclusive Manner Prevail in Experimental Allergic Encephalomyelitis (EAE). Cells. 2012; 1(3):576-596. https://doi.org/10.3390/cells1030576
Chicago/Turabian StyleKarulin, Alexey Y., Stefan Quast, Maike D. Hesse, and Paul V. Lehmann. 2012. "Neuroantigen-Specific CD4 Cells Expressing Interferon-γ (IFN-γ), Interleukin (IL)-2 and IL-3 in a Mutually Exclusive Manner Prevail in Experimental Allergic Encephalomyelitis (EAE)" Cells 1, no. 3: 576-596. https://doi.org/10.3390/cells1030576