Enhanced Transdermal Permeability via Constructing the Porous Structure of Poloxamer-Based Hydrogel
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
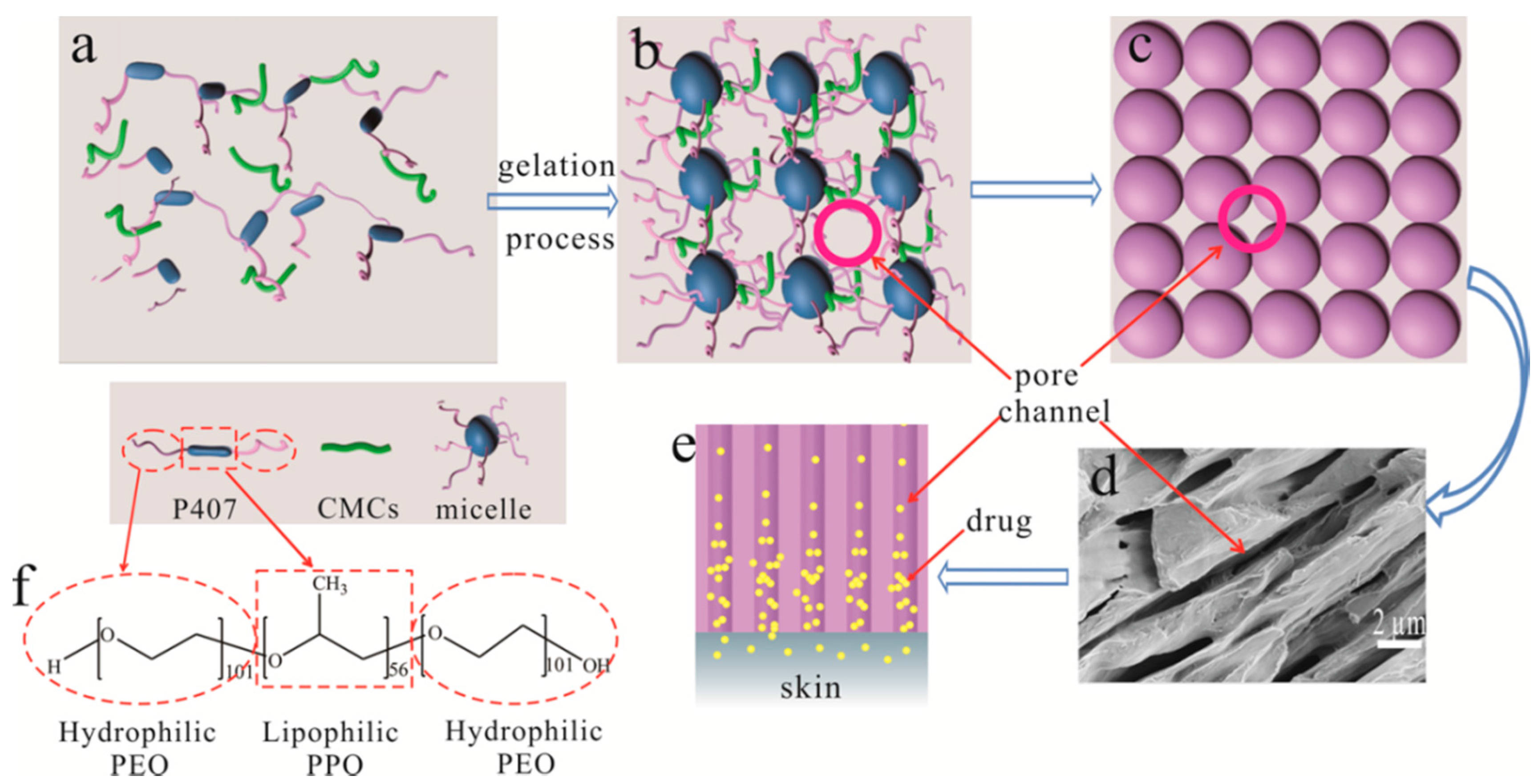
2.2. Preparation of P407/CMCs Composite Hydrogel
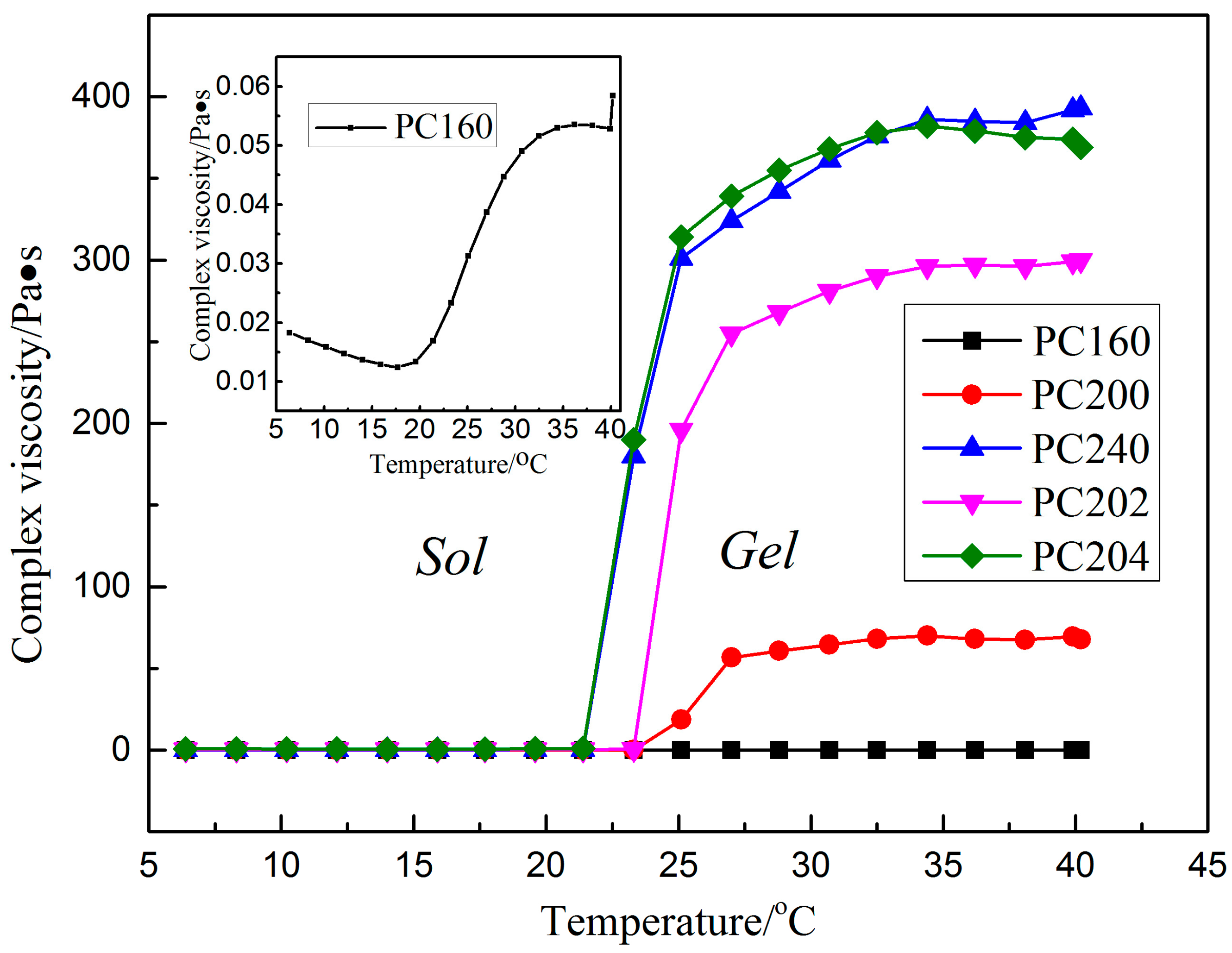
2.3. Viscosity Analysis
2.4. Observation of Porous Structure
2.5. Porosity Analysis
2.6. Transdermal Behaviour Analysis
- V: the volume of the receiving chamber (6.5 cm3);
- A: the area of the skin surface exposed to the receiving chamber (2.8 cm2);
- Ci: the initial concentration of GA in the hydrogel formulation (μg/cm3);
- dCa/dt: the change in the concentration of GA in the receiving chamber.
2.7. Statistical Analysis
3. Results
3.1. Viscosity Analysis
3.2. Porosity Analysis for P407 Matrix
3.3. Porosity Analysis for P407/CMCs
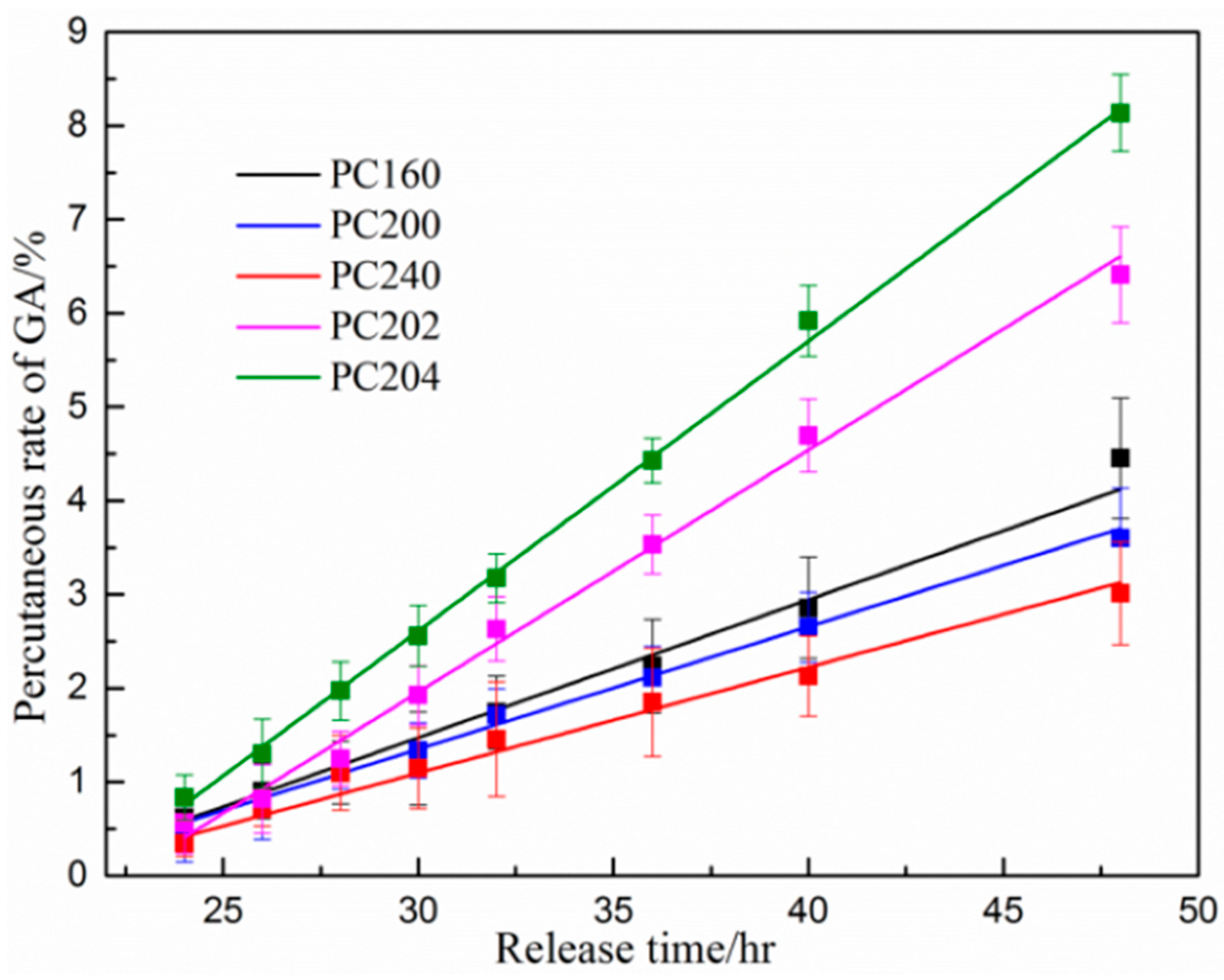
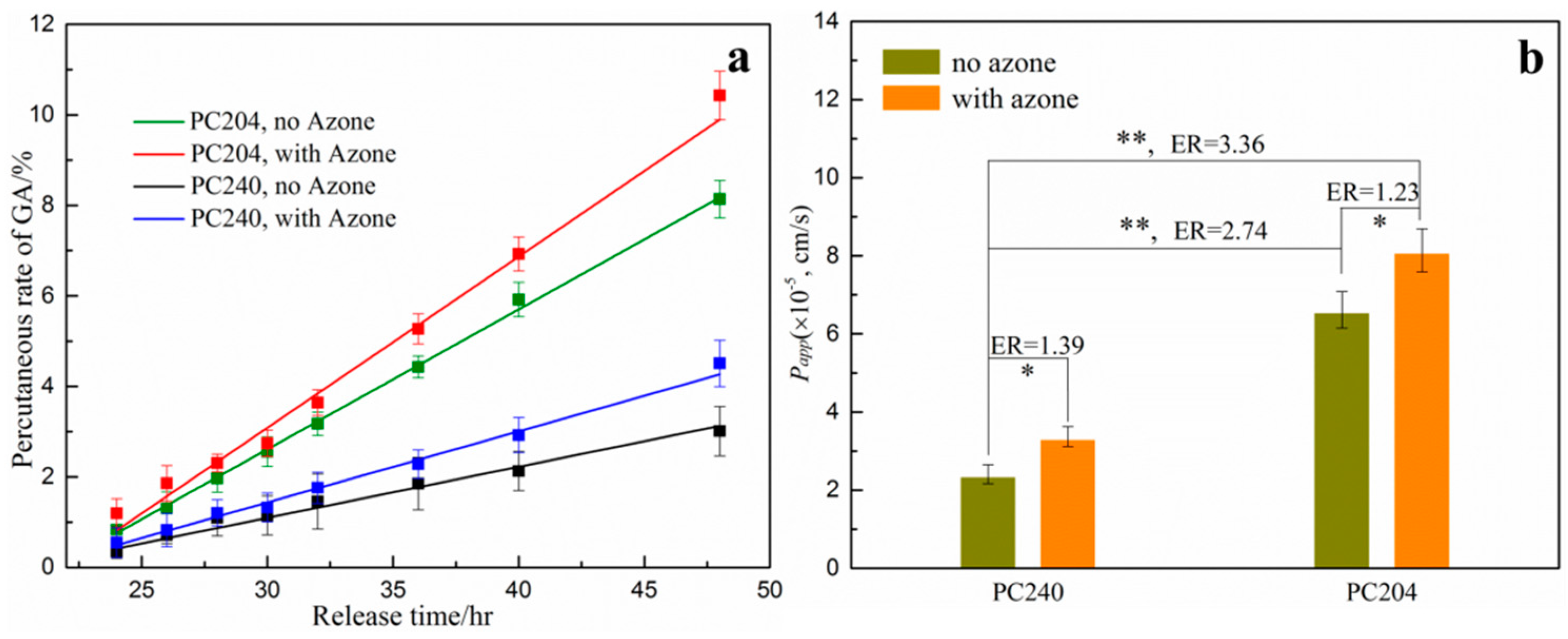
3.4. Percutaneous Analysis
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Prausnitz, M.R.; Langer, R. Transdermal drug delivery. Nat. Biotechnol. 2008, 26, 1261–1268. [Google Scholar] [CrossRef] [PubMed]
- Alexander, A.; Dwivedi, S.; Ajazuddin; Giri, T.K.; Saraf, S.; Saraf, S.; Tripathi, D.K. Approaches for breaking the barriers of drug permeation through transdermal drug delivery. J. Control. Release 2012, 164, 26–40. [Google Scholar] [CrossRef] [PubMed]
- Aktar, B.; Erdal, M.S.; Sagirli, O.; Güngör, S.; Özsoy, Y. Optimization of biopolymer based transdermal films of metoclopramide as an alternative delivery approach. Polymers 2014, 6, 1350–1365. [Google Scholar] [CrossRef]
- Cevc, G.; Vierl, U. Nanotechnology and the transdermal route: A state of the art review and critical appraisal. J. Control Release 2010, 141, 277–299. [Google Scholar] [CrossRef] [PubMed]
- Van der Maaden, K.; Jiskoot, W.; Bouwstra, J. Microneedle technologies for (trans)dermal drug and vaccine delivery. J. Control. Release 2012, 161, 645–655. [Google Scholar] [CrossRef] [PubMed]
- Liao, A.-H.; Ho, H.C.; Lin, Y.C.; Chen, H.K.; Wang, C.H. Effects of microbubble size on ultrasound-induced transdermal delivery of high-molecular-weight drugs. PLoS ONE 2015, 10, e0138500. [Google Scholar] [CrossRef] [PubMed]
- Kalia, Y.N.; Naik, A.; Garrison, J.; Guy, R.H. Iontophoretic drug delivery. Adv. Drug Deliv. Rev. 2004, 56, 619–658. [Google Scholar] [CrossRef] [PubMed]
- Aihara, H.; Miyazaki, J.-I. Gene transfer into muscle by electroporation in vivo. Nat. Biotechnol. 1998, 16, 867–870. [Google Scholar] [CrossRef] [PubMed]
- Peppas, N.A.; Hilt, J.Z.; Khademhosseini, A.; Langer, R. Hydrogels in biology and medicine: From molecular principles to bionanotechnology. Adv. Mater. 2006, 18, 1345–1360. [Google Scholar] [CrossRef]
- Joo, V.; Ramasamy, T.; Haidar, Z.S. A novel self-assembled liposome-based polymeric hydrogel for cranio-maxillofacial applications: Preliminary findings. Polymers 2011, 3, 967–974. [Google Scholar] [CrossRef]
- Dayan, N.; Touitou, E. Carriers for skin delivery of trihexyphenidyl HCl: Ethosomes vs. liposomes. Biomaterials 2000, 21, 1879–1885. [Google Scholar] [CrossRef]
- Kong, M.; Hou, L.; Wang, J.; Feng, C.; Liu, Y.; Cheng, X.; Chen, X. Enhanced transdermal lymphatic drug delivery of hyaluronic acid modified transfersomes for tumor metastasis therapy. Chem. Commun. 2015, 51, 1453–1456. [Google Scholar] [CrossRef] [PubMed]
- Vashist, A.; Kaushik, A.; Vashist, A.; Jayant, R.D.; Tomitaka, A.; Ahmad, S.; Guptad, Y.K. Madhavan nair recent trends on hydrogel based drug delivery systems for infectious diseases. Biomater. Sci. 2016, 4, 1535–1553. [Google Scholar] [CrossRef] [PubMed]
- Vashist, A.; Vashist, A.; Gupta, Y.K. Sharif Ahmad Recent advances in hydrogel based drug delivery systems for the human body. J. Mater. Chem. B 2014, 2, 147–166. [Google Scholar] [CrossRef]
- Kotsuchibashi, Y.; Ebara, M.; Aoyagi, T.; Narain, R. Recent advances in dual temperature responsive block copolymers and their potential as biomedical applications. Polymers 2016, 8, 380. [Google Scholar] [CrossRef]
- Klouda, L.; Mikos, A.G. Thermoresponsive hydrogels in biomedical applications. Eur. J. Pharm. Biopharm. 2008, 68, 34–45. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Wu, X.; Li, J.; Hao, X.; Lin, J.; Cheng, D.; Lu, Y. Thermosensitive behavior and antibacterial activity of cotton fabric modified with a chitosan-poly(n-isopropylacrylamide) interpenetrating polymer network hydrogel. Polymers 2016, 8, 110. [Google Scholar] [CrossRef]
- Trong, L.C.; Djabourov, M.; Ponton, A. Mechanisms of micellization and rheology of PEO-PPO-PEO triblock copolymers with various architectures. J. Colloid Interface Sci. 2008, 328, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Sosnik, A.; Cohn, D. Ethoxysilane-capped PEO–PPO–PEO triblocks: A new family of reverse thermo-responsive polymers. Biomaterials 2004, 25, 2851–2858. [Google Scholar] [CrossRef] [PubMed]
- Lam, Y.-M.; Grigorieff, N.; Goldbeck-Wood, G. Direct visualisation of micelles of Pluronic block copolymers in aqueous solution by cryo-TEM. Phys. Chem. Chem. Phys. 1999, 1, 3331–3334. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, J.; Zhang, X.; Lu, W.; Zhang, Q. A novel mixed micelle gel with thermo-sensitive property for the local delivery of docetaxel. J. Control. Release 2009, 135, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Hyun, K.; Nam, J.G.; Wilhellm, M.; Ahn, K.H.; Lee, S.J. Large amplitude oscillatory shear behavior of PEO-PPO-PEO triblock copolymer solutions. Rheol. Acta 2006, 45, 239–249. [Google Scholar] [CrossRef]
- Jeong, B.; Kim, S.W.; Bae, Y.H. Thermosensitive sol-gel reversible hydrogels. Adv. Drug Deliv. Rev. 2012, 64, 154–162. [Google Scholar] [CrossRef]
- Schmolka, I.R. Artificial skin I. Preparation and properties of pluronic F-127 gels for treatment of burns. J. Biomed. Mater. Res. 1972, 6, 571–582. [Google Scholar] [CrossRef] [PubMed]
- Chan, B.C.; Li, L.F.; Hu, S.Q.; Wat, E.; Wong, E.C.W.; Zhang, V.X.; Lau, C.B.S.; Wong, C.K.; Hon, K.L.E.; Hui, P.C.L.; et al. Leung gallic acid is the major active component of cortex moutan in inhibiting immune maturation of human monocyte-derived dendritic cells. Molecules 2015, 20, 16388–16403. [Google Scholar] [CrossRef] [PubMed]
- Lombardi Borgia, S.; Schlupp, P.; Mehnert, W.; Schäfer-Korting, M. In vitro skin absorption and drug release—A comparison of six commercial prednicarbate preparations for topical use. Eur. J. Pharm. Biopharm. 2008, 68, 380–389. [Google Scholar] [CrossRef] [PubMed]
- Schafer-Korting, M.; Bock, U.; Gamer, A.; Haberland, A.; Haltner-Ukomadu, E.; Kaca, M.; Kamp, H.; Kietzmann, M.; Korting, H.C.; Krächter, H.U.; et al. Reconstructed human epidermis for skin absorption testing: Results of the German prevalidation study. Altern. Lab. Anim. 2006, 34, 283–294. [Google Scholar] [PubMed]
- Wang, W.; Wat, E.; Hui, P.C.L.; Chan, B.; Ng, F.S.F.; Kan, C.; Wang, X.; Hu, H.; Wong, E.C.W.; Lau, C.B.S.; et al. Dual-functional transdermal drug delivery system with controllable drug loading based on thermosensitive poloxamer hydrogel for atopic dermatitis treatment. Sci. Rep. 2016, 6, 24112. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Hui, P.C.; Wat, E.; Ng, F.S.; Kan, C.W.; Wang, X.; Wong, E.C.; Hu, H.; Chan, B.; Lau, C.B.; et al. In vitro drug release and percutaneous behavior of poloxamer-based hydrogel formulation containing traditional Chinese medicine. Colloid Surf. B 2016, 148, 526–532. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.-A.; Kim, E.S.; Kwon, J.H.; Kim, H.; Shin, J.H.; Yun, S.H.; Choi, K.Y.; Hahn, S.K. Transdermal delivery of hyaluronic acid—Human growth hormone conjugate. Biomaterials 2012, 33, 5947–5954. [Google Scholar] [CrossRef] [PubMed]
- Prausnitz, M.R.; Mitragotri, S.; Langer, R. Current status and future potential of transdermal drug delivery. Nat. Rev. Drug Discov. 2004, 3, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Shakeel, F.; Baboota, S.; Ahuja, A.; Ali, J.; Aqil, M.; Shafiq, S. Nanoemulsions as vehicles for transdermal delivery of aceclofenac. AAPS PharmSciTech 2007, 8, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Egli, S.; Schlaad, H.; Bruns, N.; Meier, W. Functionalization of block copolymer vesicle surfaces. Polymers 2011, 3, 252–280. [Google Scholar] [CrossRef] [Green Version]
- Santander-Ortega, M.; Stauner, T.; Loretz, B.; Ortega-Vinuesa, J.L.; Bastos-González, D.; Wenz, G.; Schaefer, U.F.; Lehr, C.M. Nanoparticles made from novel starch derivatives for transdermal drug delivery. J. Control. Release 2010, 141, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Hu, M.; Liu, W.; Xue, C.; Xu, H.; Yang, X. Investigation of the carbopol gel of solid lipid nanoparticles for the transdermal iontophoretic delivery of triamcinolone acetonide acetate. Int. J. Pharm. 2008, 364, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Mortensen, K. PEO-related block copolymer surfactants. Colloids Surf. A 2001, 183, 277–292. [Google Scholar] [CrossRef]
- Desai, P.R.; Sharma, R.K.; Bahadur, P. Effect of additives on the micellization of PEO/PPO/PEO block copolymer F127 in aqueous solution. Colloids Surf. A 2001, 178, 57–69. [Google Scholar] [CrossRef]
- Alexandridis, P.; Hatton, T.A. Poly(ethylene oxide) poly(propylene oxide) poly(ethylene oxide) block copolymer surfactants in aqueous solutions and at interfaces: Thermodynamics, structure, dynamics, and modeling. Colloids Surf. A 1995, 96, 1–46. [Google Scholar] [CrossRef]
- Nagarajan, R. Solubilization of hydrocarbons and resulting aggregate shape transitions in aqueous solutions of Pluronic®(PEO–PPO–PEO) block copolymers. Colloids Surf. B 1999, 16, 55–72. [Google Scholar] [CrossRef]
- Fromherz, P. Micelle structure: A surfactant-block model. Chem. Phys. Lett. 1981, 77, 460–466. [Google Scholar] [CrossRef]



| Sample | PC160 | PC200 | PC240 | PC202 | PC204 |
|---|---|---|---|---|---|
| The most probable pore-size/μm | 4.989 | 4.813 | 0.689 | 4.701 | 4.343 |
| Total surface area (m2/g) | 1.502 | 1.709 | 2.887 | 1.808 | 1.996 |
| Pore number fraction | 0.0177 | 0.0212 | 0.0211 | 0.213 | 0.277 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, W.-Y.; Hui, P.C.L.; Wat, E.; Ng, F.S.F.; Kan, C.-W.; Lau, C.B.S.; Leung, P.-C. Enhanced Transdermal Permeability via Constructing the Porous Structure of Poloxamer-Based Hydrogel. Polymers 2016, 8, 406. https://doi.org/10.3390/polym8110406
Wang W-Y, Hui PCL, Wat E, Ng FSF, Kan C-W, Lau CBS, Leung P-C. Enhanced Transdermal Permeability via Constructing the Porous Structure of Poloxamer-Based Hydrogel. Polymers. 2016; 8(11):406. https://doi.org/10.3390/polym8110406
Chicago/Turabian StyleWang, Wen-Yi, Patrick C. L. Hui, Elaine Wat, Frency S. F. Ng, Chi-Wai Kan, Clara B. S. Lau, and Ping-Chung Leung. 2016. "Enhanced Transdermal Permeability via Constructing the Porous Structure of Poloxamer-Based Hydrogel" Polymers 8, no. 11: 406. https://doi.org/10.3390/polym8110406







