Polyester Dendrimers
Abstract
:Abbreviations
| ADH | alcohol dehydrogenase |
| ADH-LB | alcohol dehydrogenase from Lactobacillus brevis |
| ADH-T | alcohol dehydrogenase from Thermoanaerobacter sp. |
| ATRP | atom transfer radical polymerization |
| bis-HMPA | 2,2-bis(hydroxymethyl)propanoic acid |
| BOP | benzotriazole-1-yl-oxy-tris-(dimethylamino)-phosphonium hexafluorophosphate |
| CCS | core cross-linked star |
| COMU | 1-[(1-(cyano-2-ethoxy-2-oxoethylideneaminooxy)dimethylaminomorpho-linomethylene)] methanaminium hexafluorophosphate |
| DBU | 1,8-diazabicyclo[5.4.0]undec-7-ene |
| DCC | dicyclohexylcarbodiimide |
| DEAD | diethyl azodicarboxylate |
| DIAD | diisopropyl azodicarboxylate |
| DIEA | diisopropylethylamine |
| DMAP | 4-dimethylaminopyridine |
| DMPA | 2,2-dimethoxy-1,2-diphenylacetophenone |
| DPPA | diphenylphosphoryl azide |
| DPTS | 4-(dimethylamino)pyridinium p-toluenesulfonate |
| EDCI | N-(3-dimethylaminopropyl)-N'-ethylcarbodiimide hydrochloride |
| FDLCP | ferroelectric dendritic liquid crystalline polymer |
| FLCPs | ferroelectric liquid crystalline polymers |
| HBTU | 2-(1H-benzotriazole-1-yl)-1,1,3,3-tetramethyluronium hexafluorophosphate |
| HOBT | 1-hydroxybenzotriazole |
| LCPs | linear crystalline polymers |
| MTBD | 7-methyl-1,5,7-triazabicyclo[4.4.0]dec-5-ene |
| MOMCl | methoxymethyl chloride |
| NADPH | nicotinamide adenine dinucleotide phosphate |
| PAMAM | polyamidoamine |
| PEO-NH2 | amine functionalized polyethylene oxide |
| PTP | proton-transfer polymerization |
| ROP | ring opening polymerization |
| SCROP | self-condensing ring-opening polymerization |
| SCVP | self-condensing vinyl polymerization |
| SEC | size exclusion chromatography |
| SET-LRP | single electron transfer living radical polymerization |
| SPECT | single photon emission computed tomography |
| TATU | 2-(1H-7-azabenzotriazole-1-yl)-1,1,3,3-tetramethyluronium tetrafluoroborate |
| TBAF | tetrabutylammonium fluoride |
| TBDMSCl | tert-butyldimethylsilyl chloride |
| TBTU | 2-(1H-benzotriazole-1-yl)-1,1,3,3-tetramethyluronium tetrafluoroborate |
| TEA | triethylamine |
| TPP | triphenylphosphine |
| TPPH2 | tetraphenylporphyrin |
| TPPZn | zinc-cored tetraphenylporphyrin |
| TsEt | 2-p-toluenesulfonylethyl |
1. Introduction
2. Structure and Synthetic Strategies
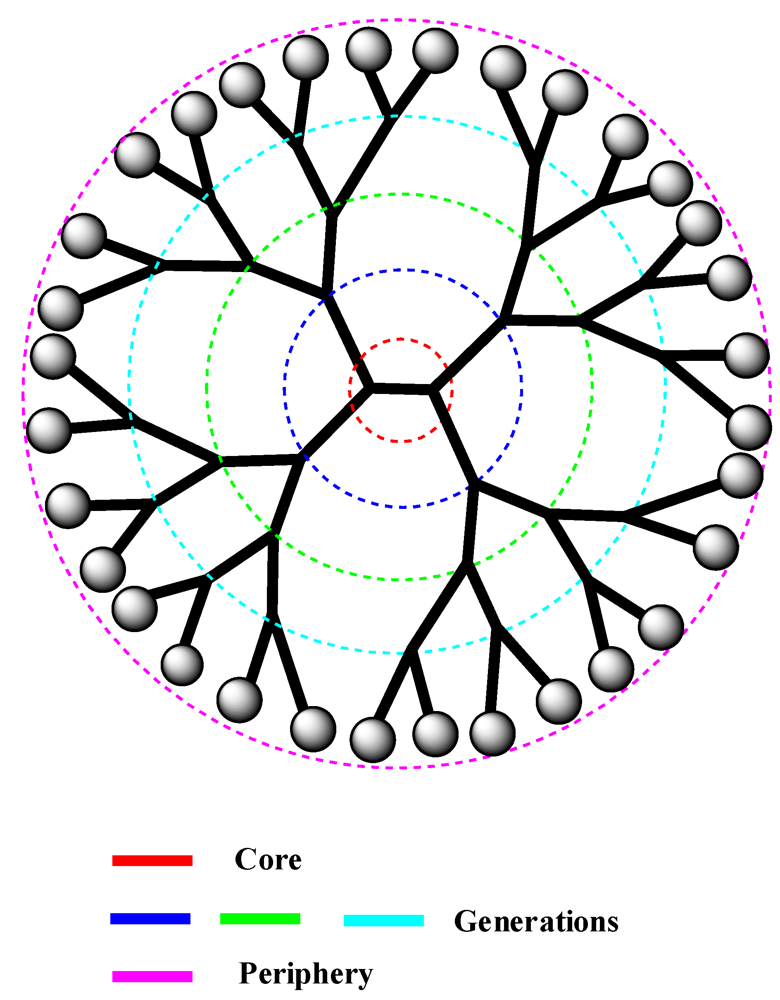
2.1. Dendrimers


2.2. Hyperbranched Polymers


3. Methods Used for Ester Bond Formation

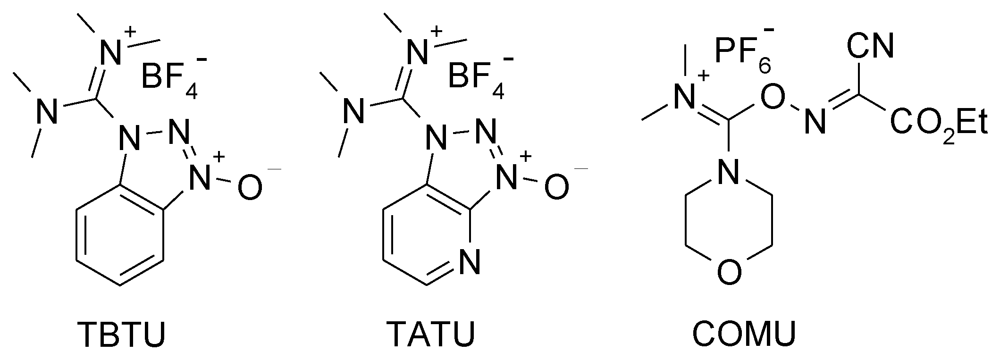

4. Advances in the Synthesis of Polyester Dendrons and Dendrimers
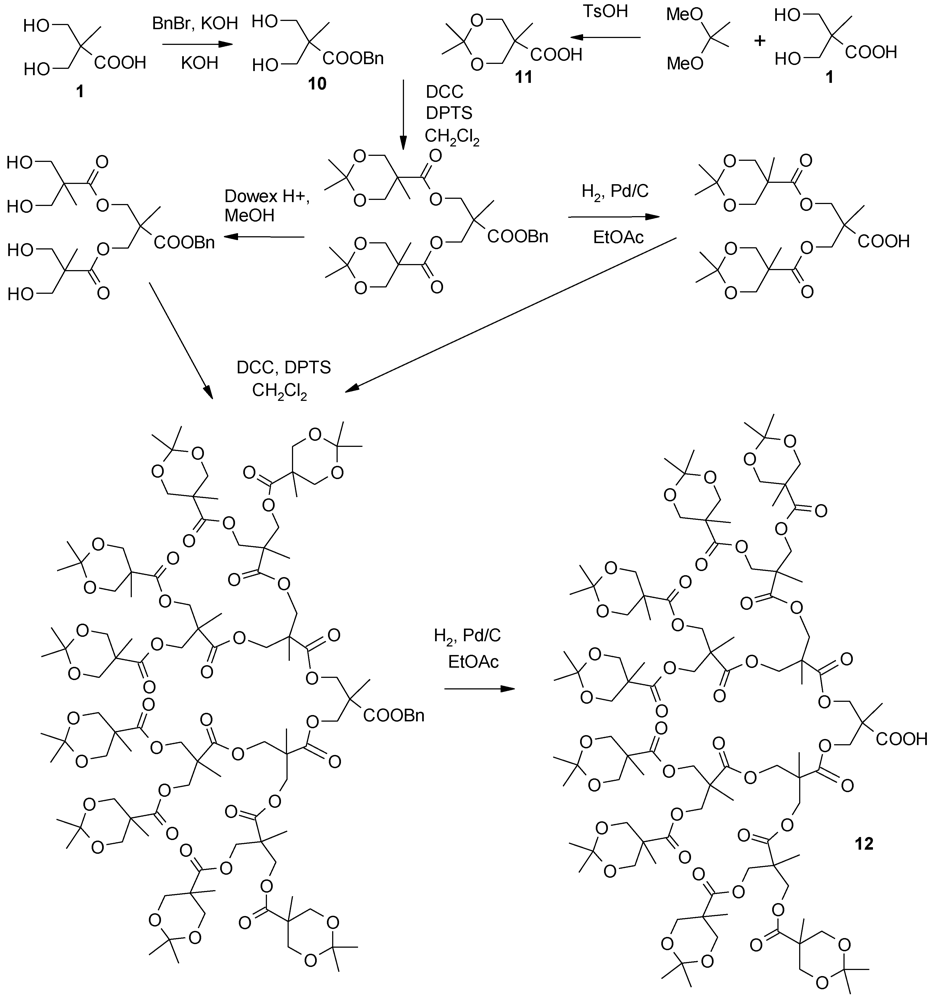
4.1. The Use of 2,2-Bis(Hydroxymethyl)Propanoic Acid-Derived Dendrons
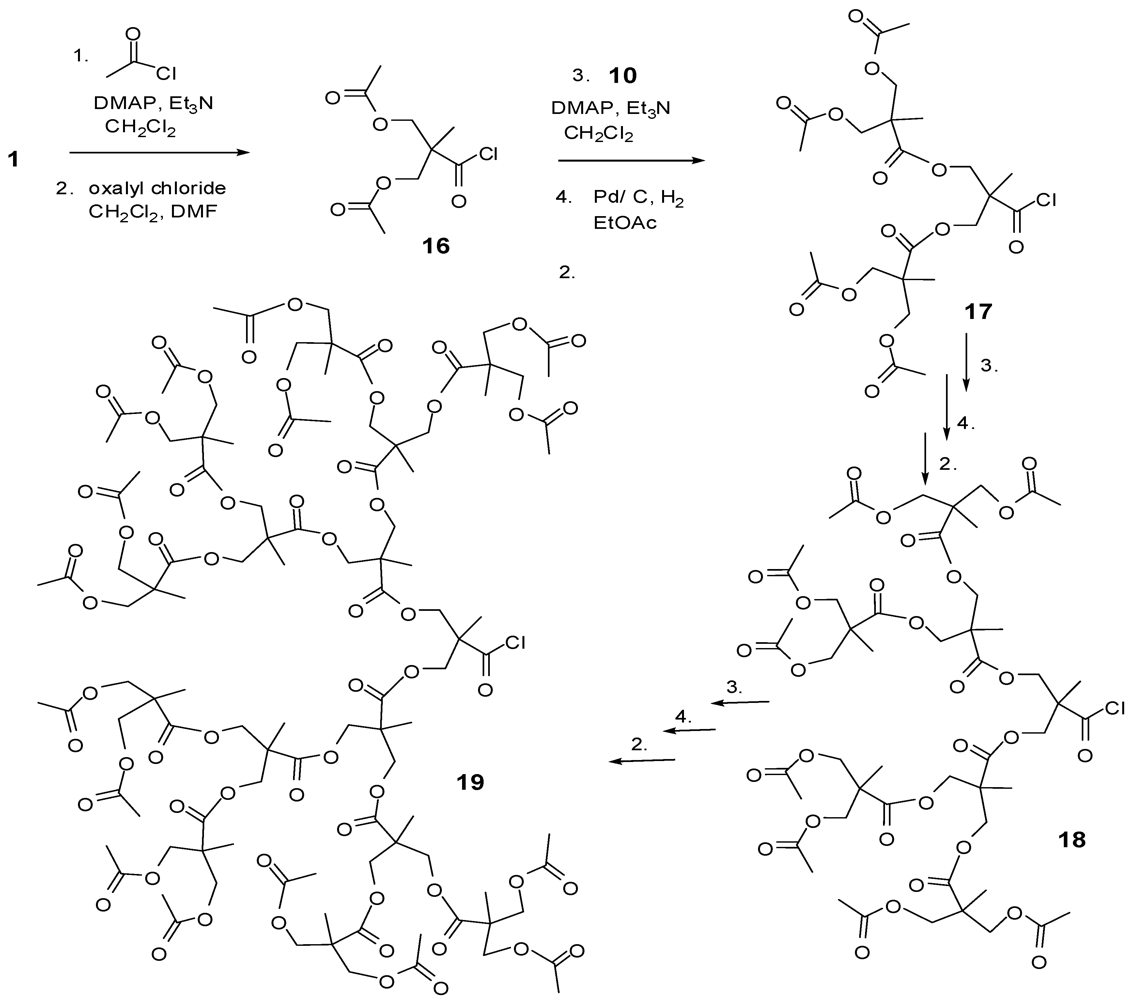
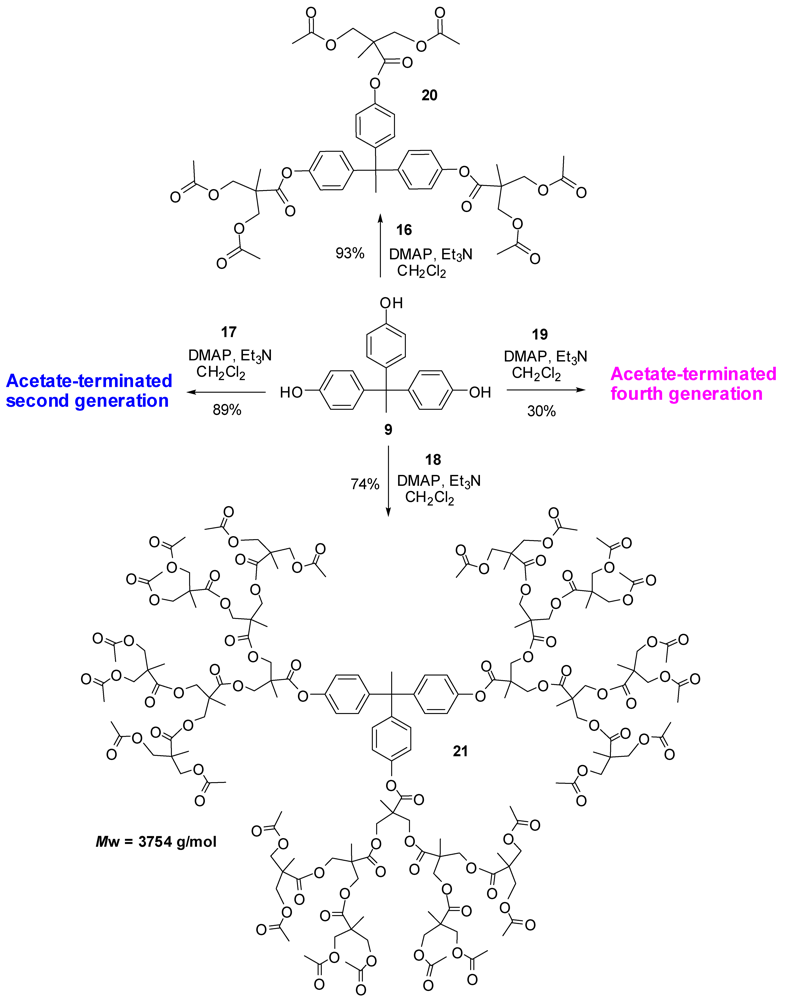
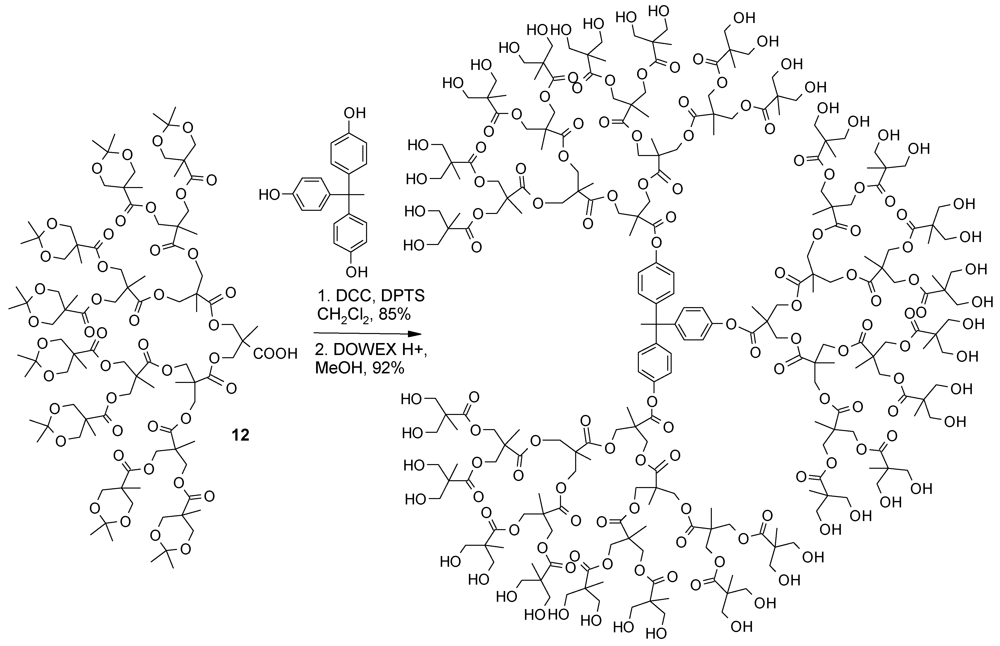
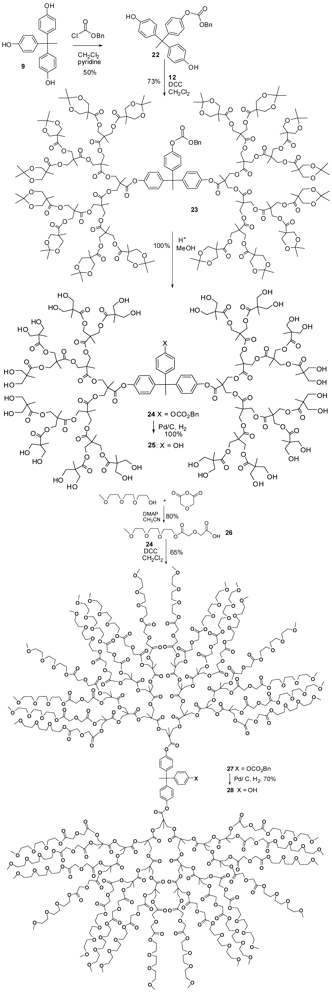
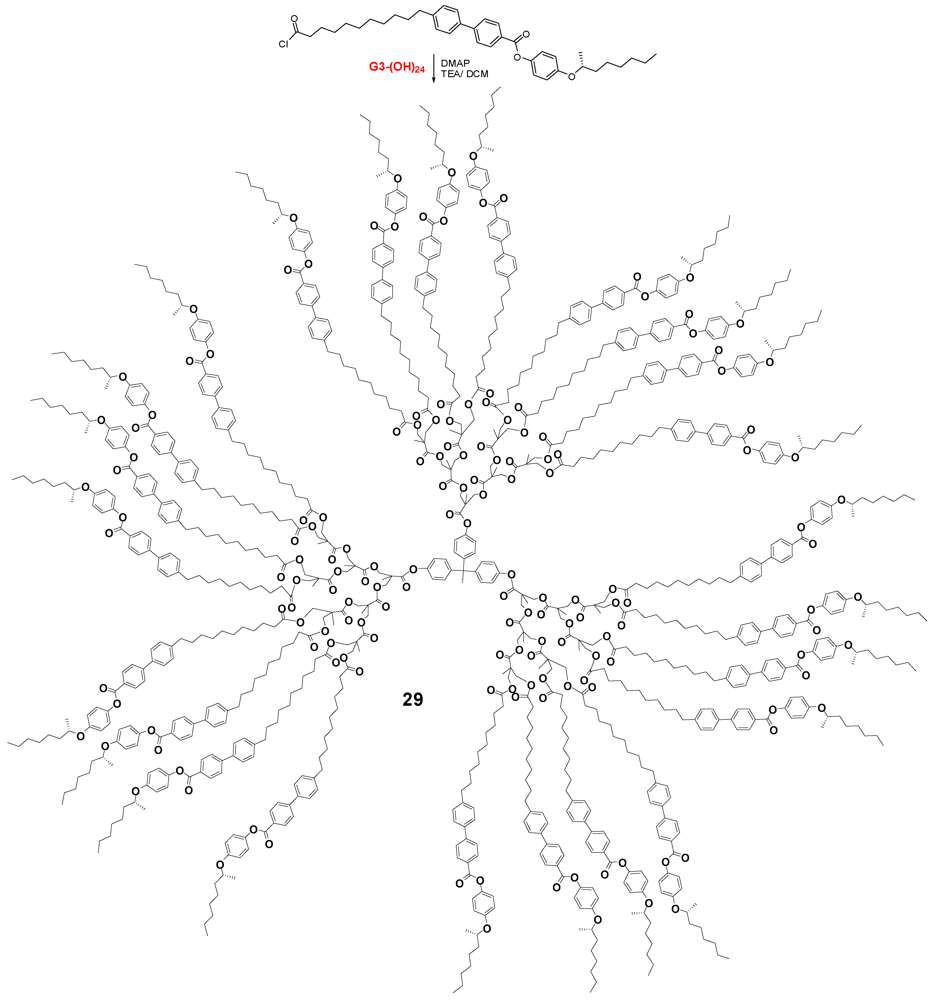
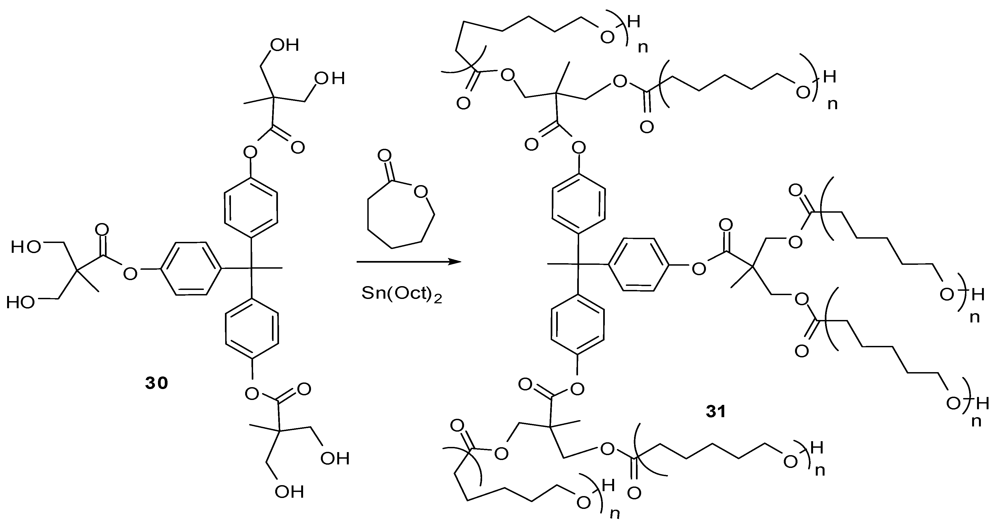
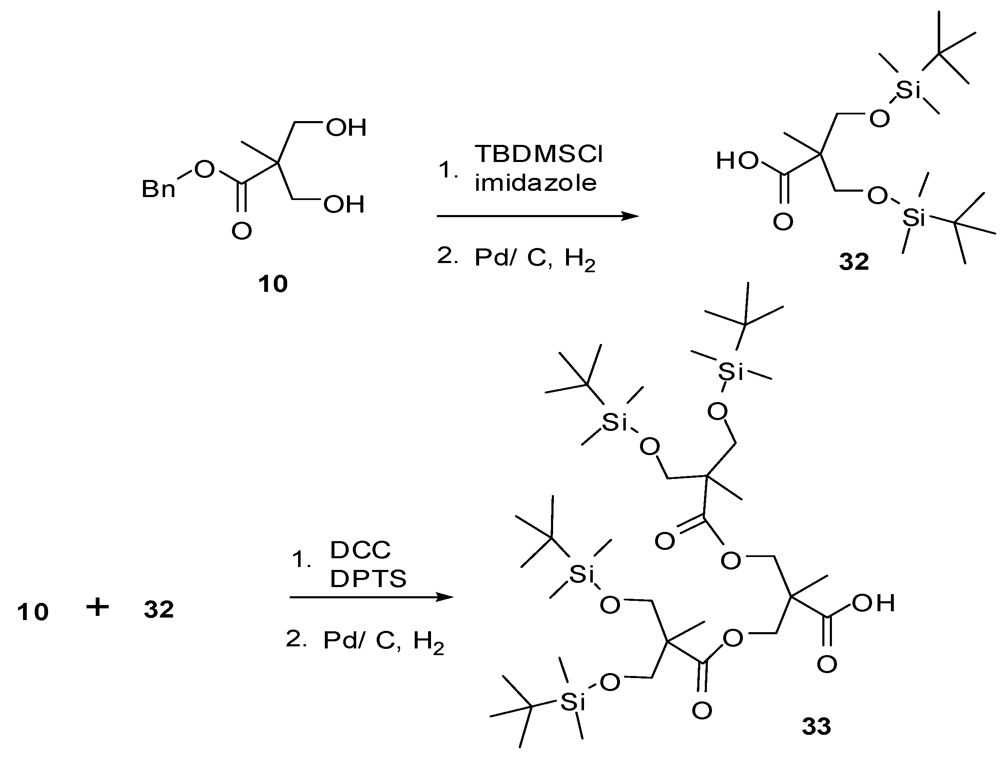
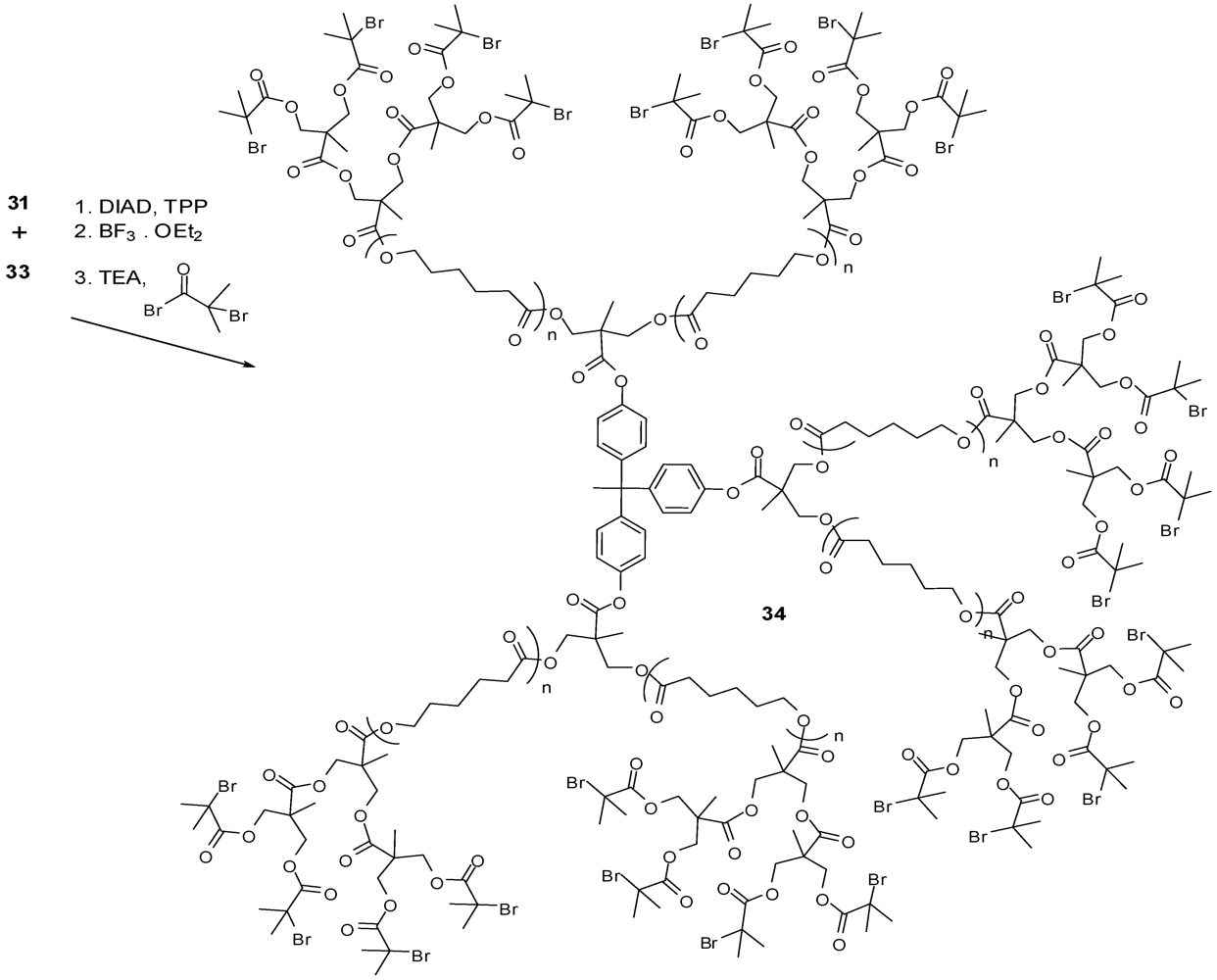
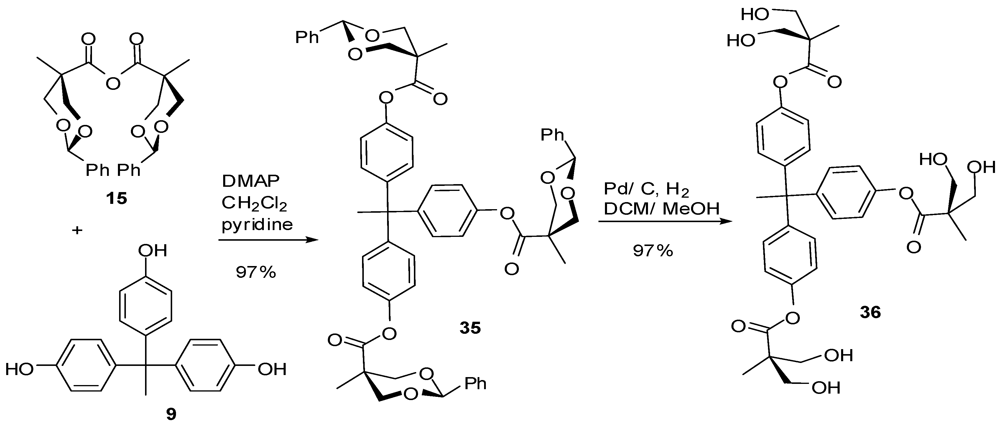
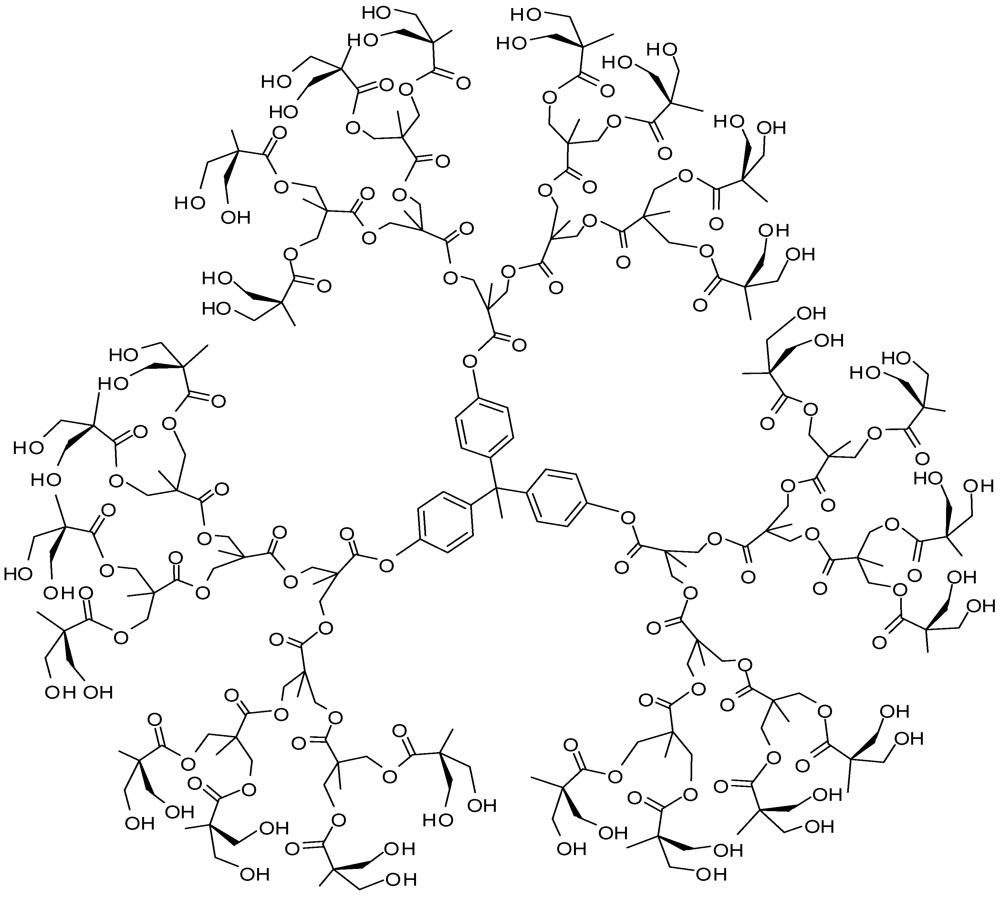
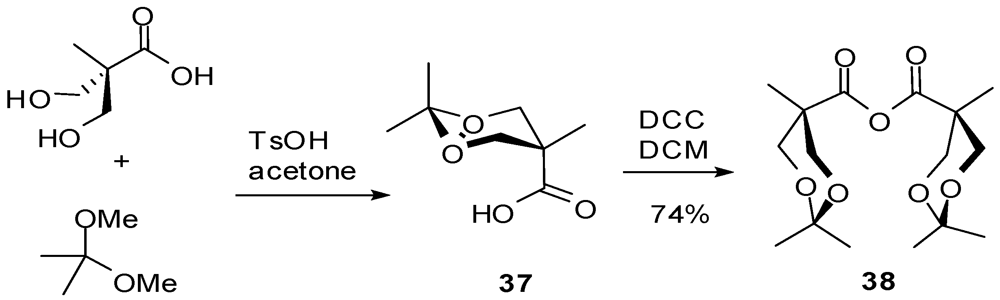

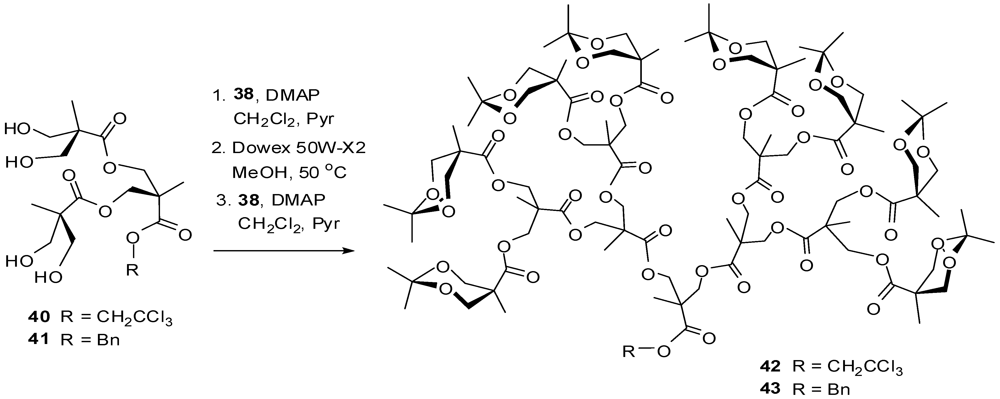
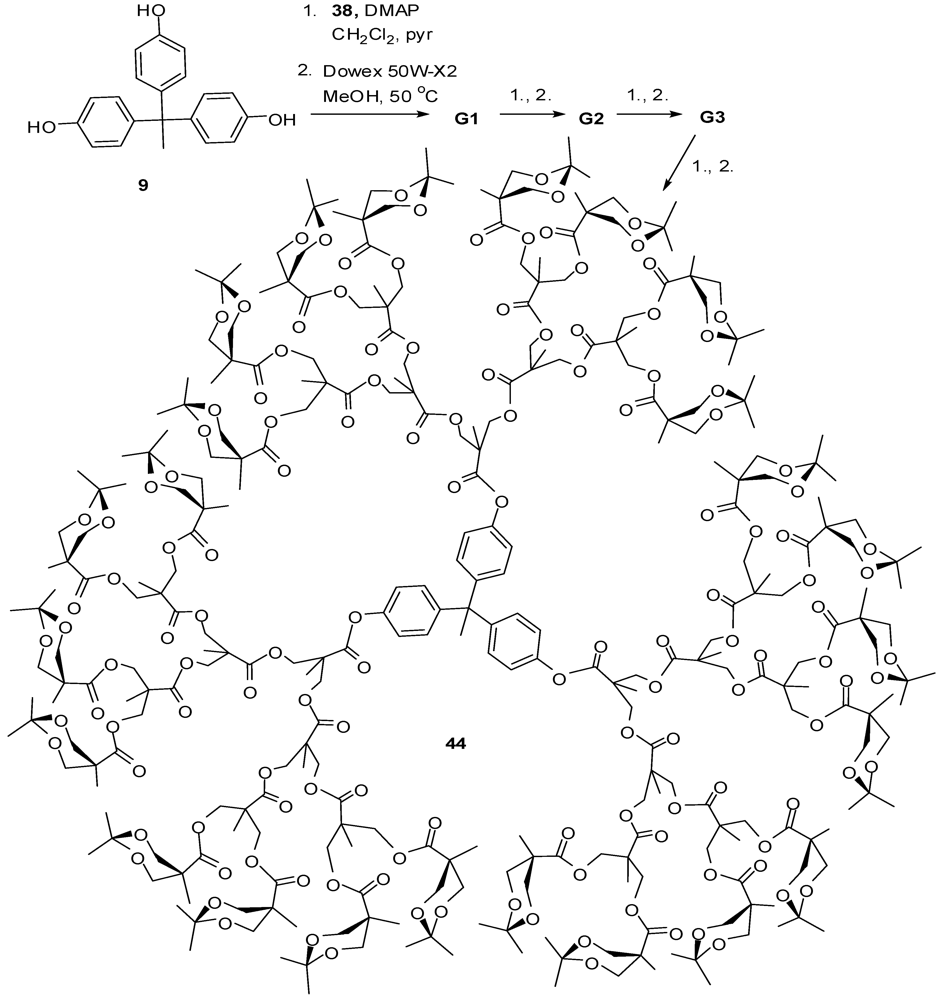
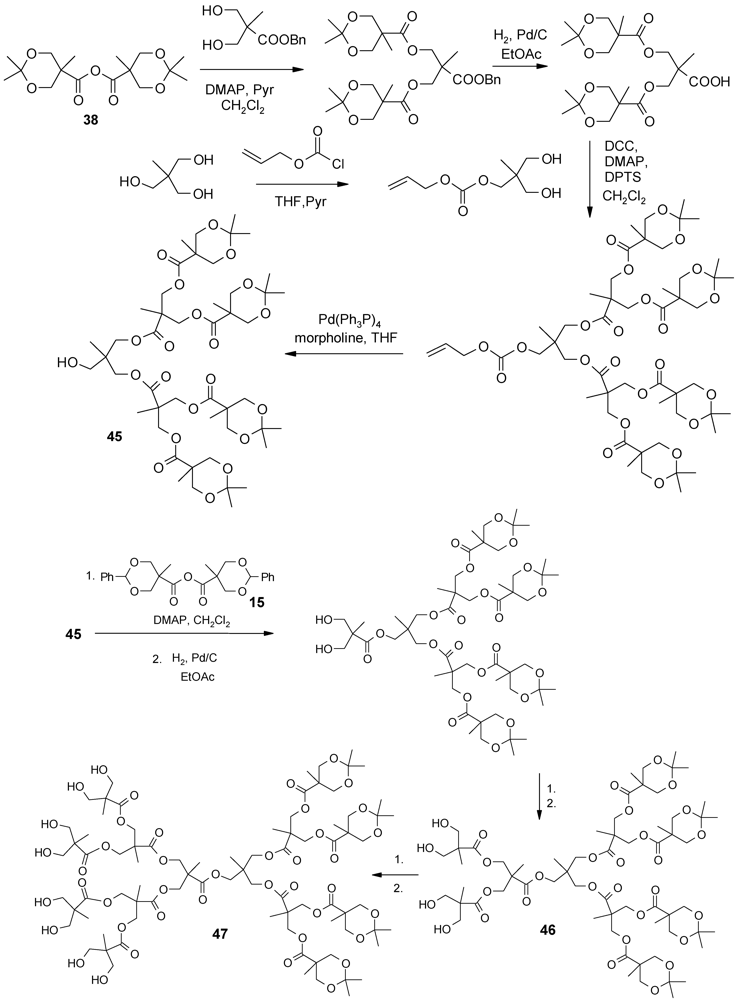
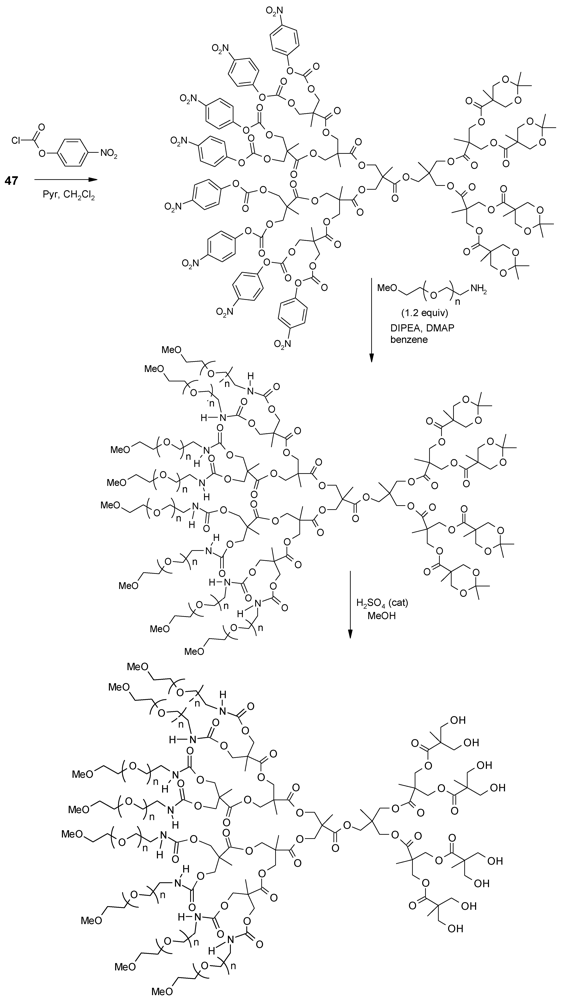
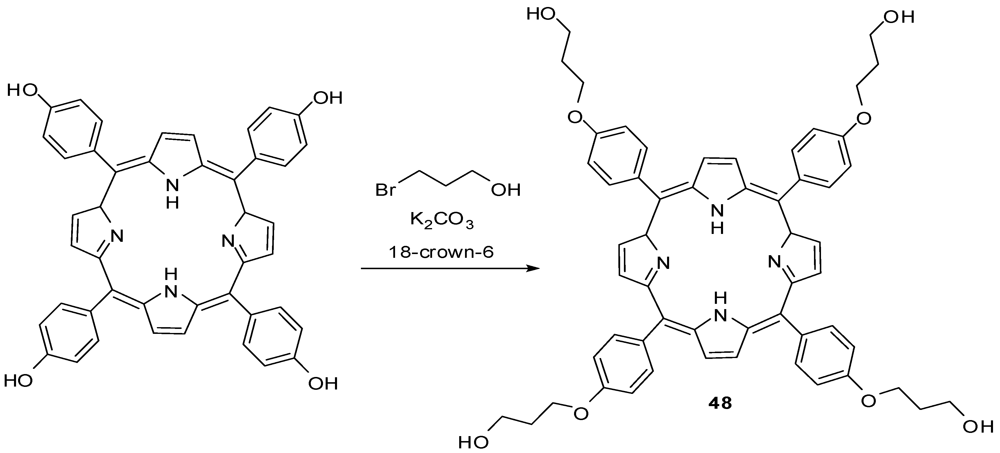
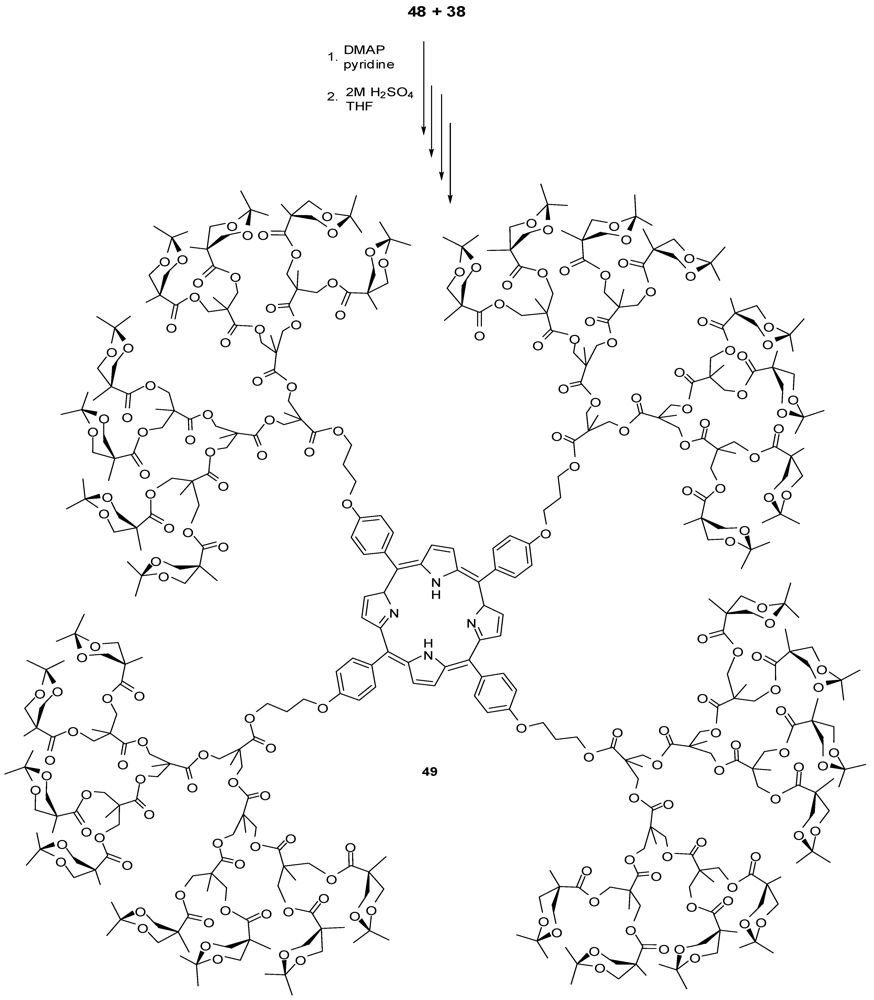
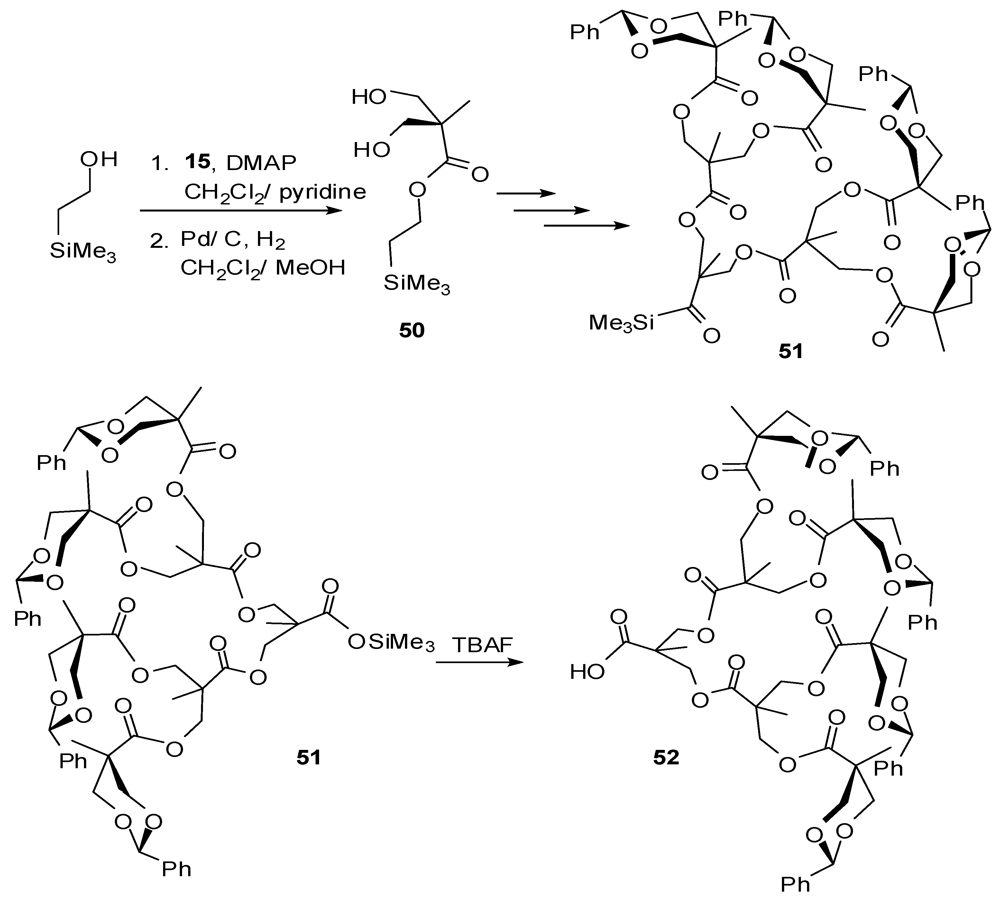
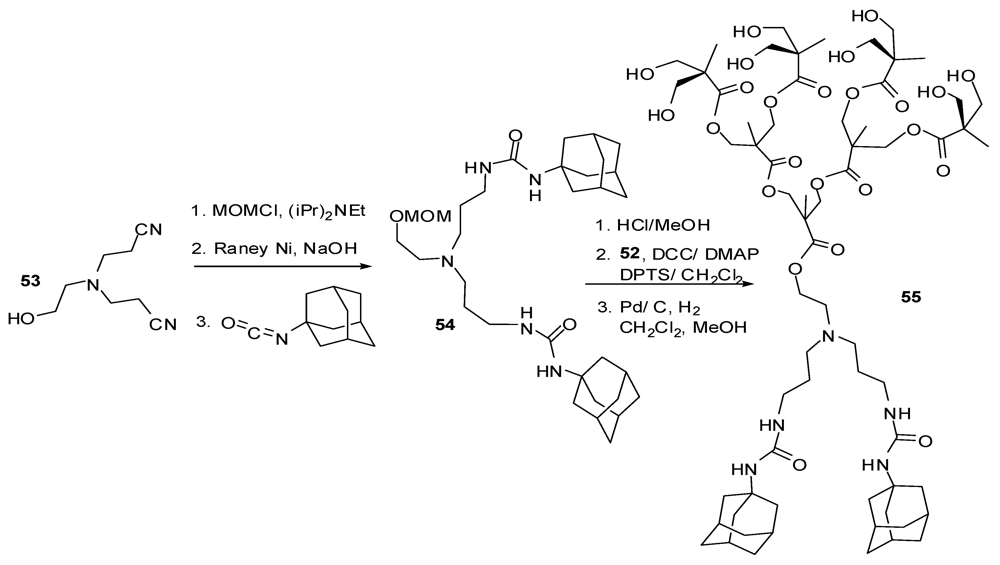
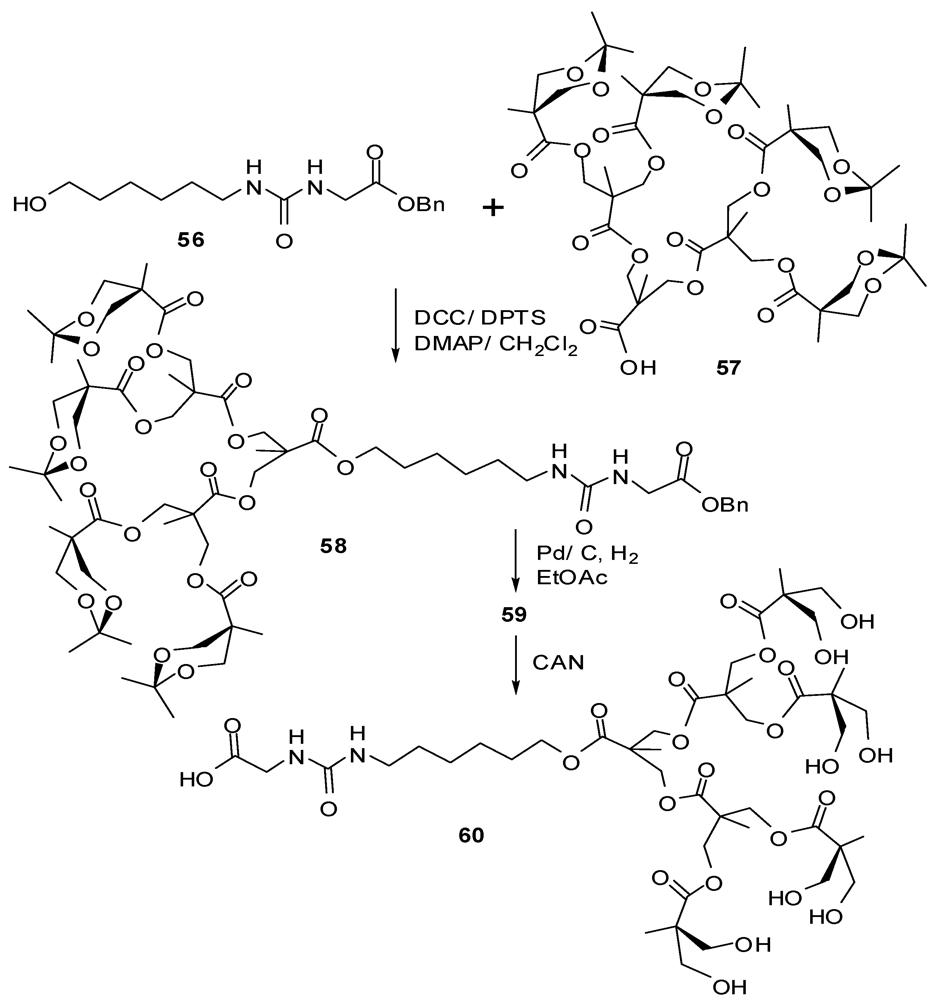

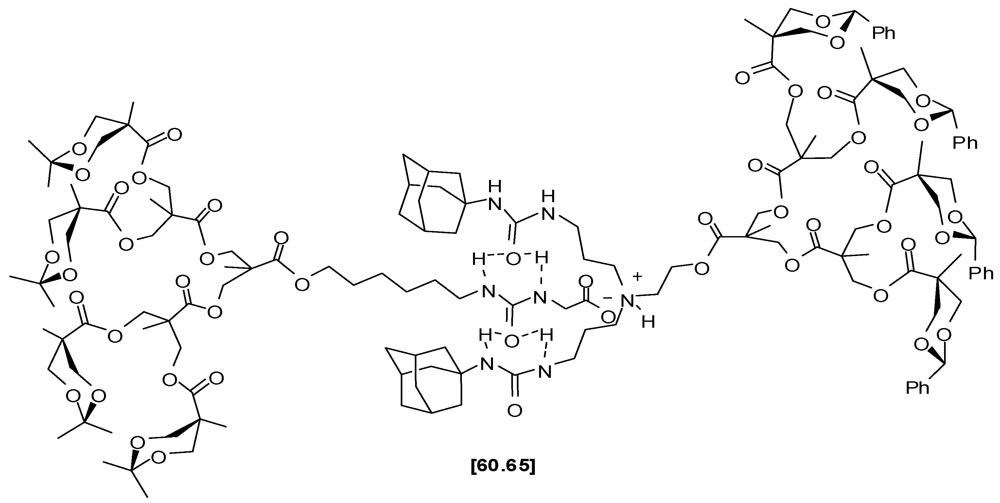
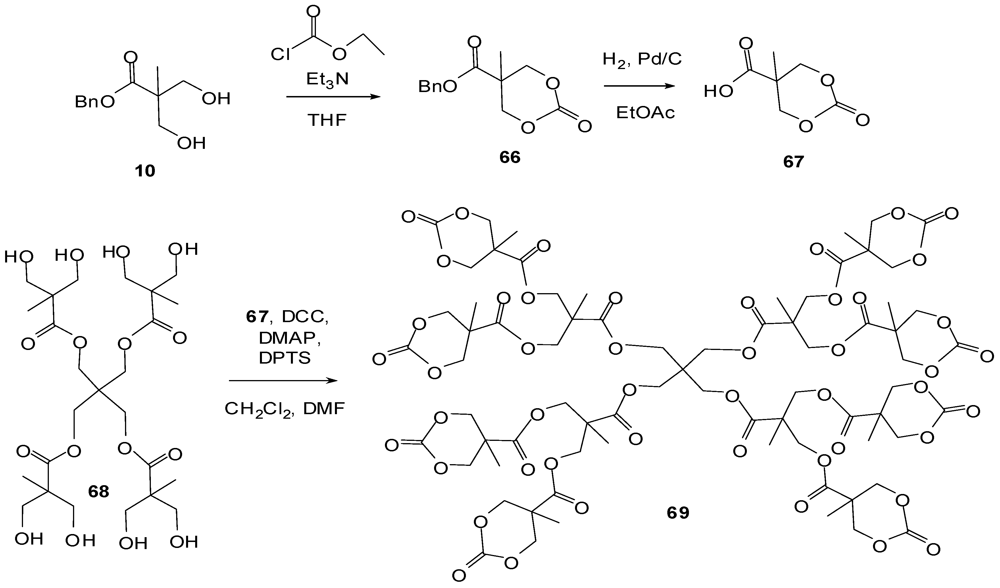

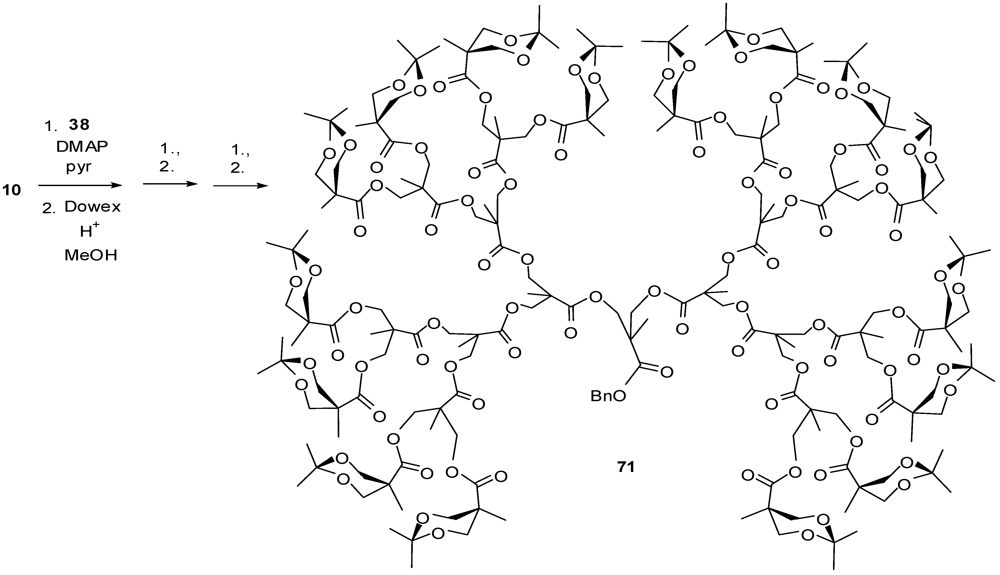
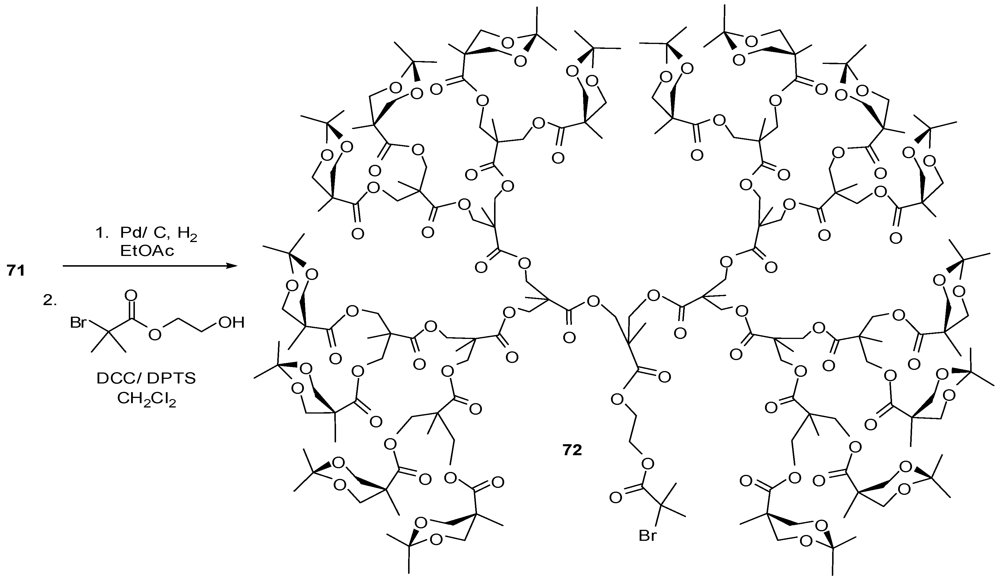
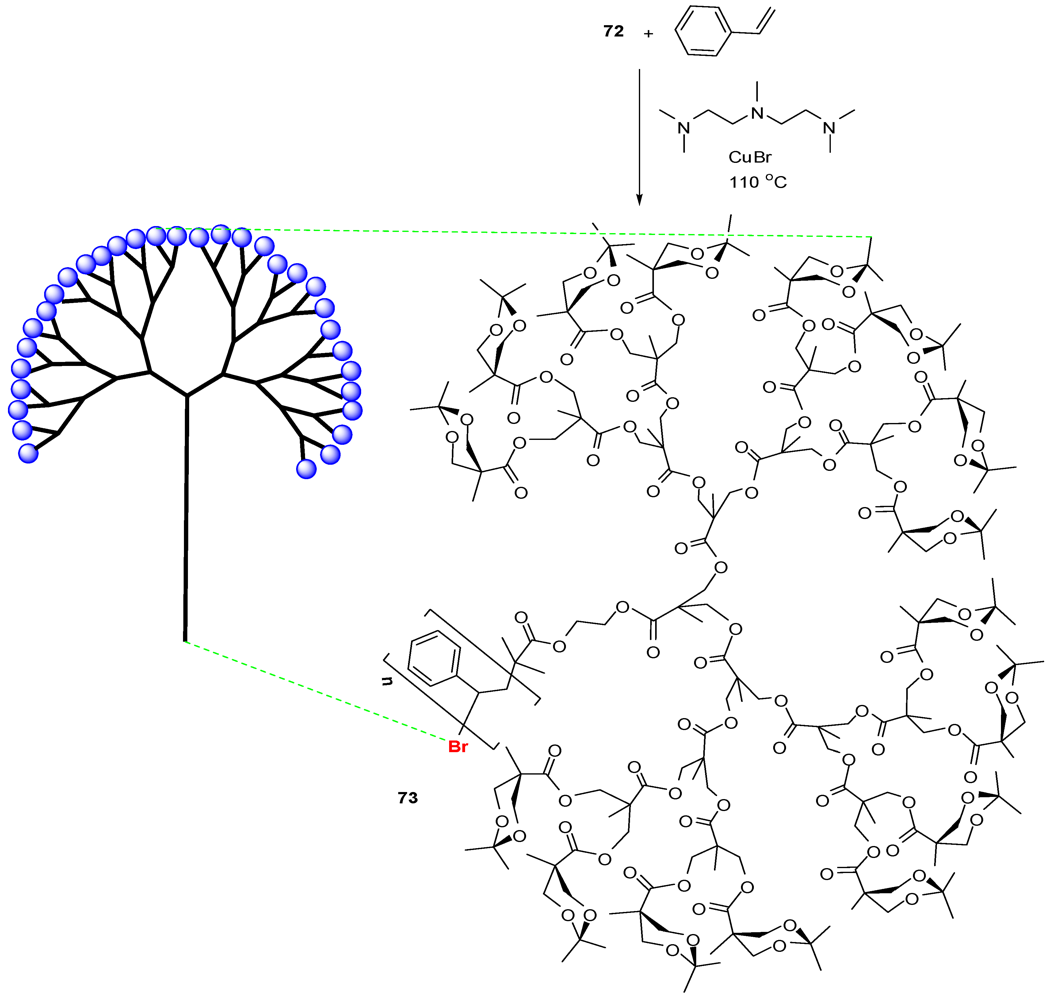

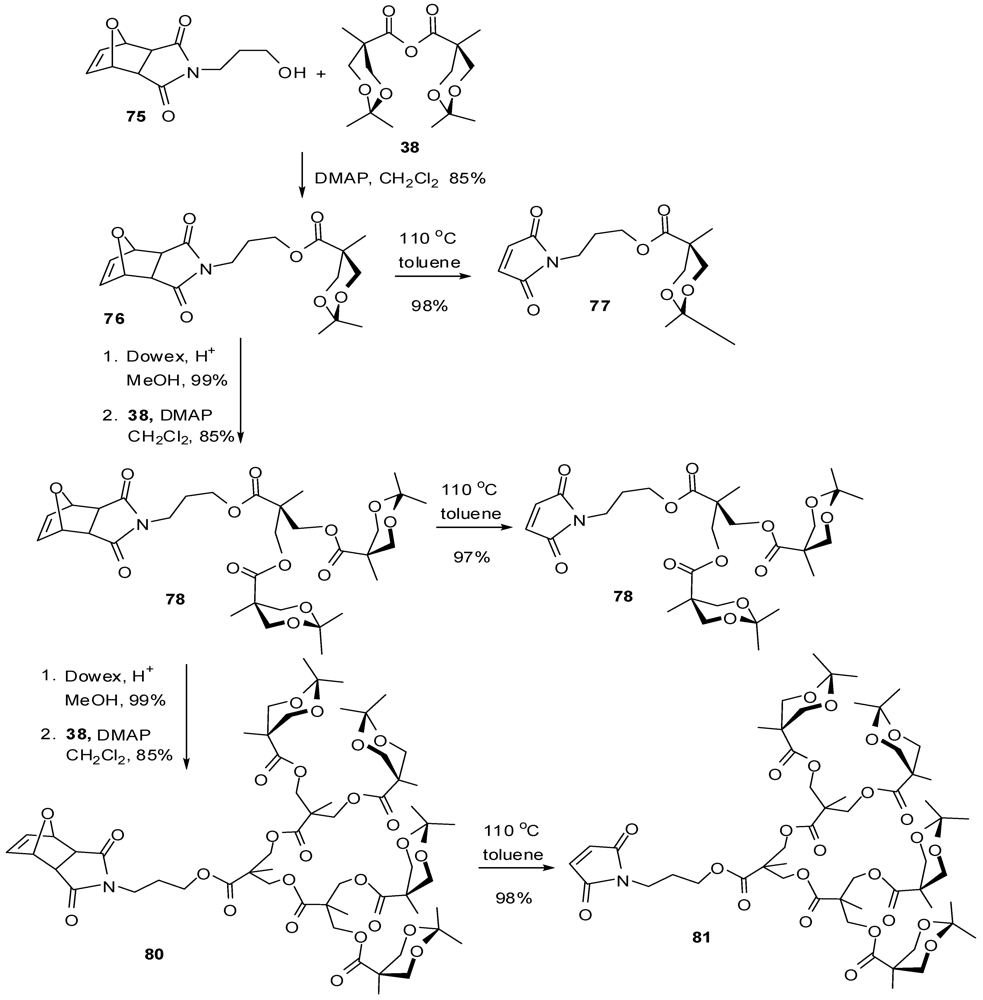
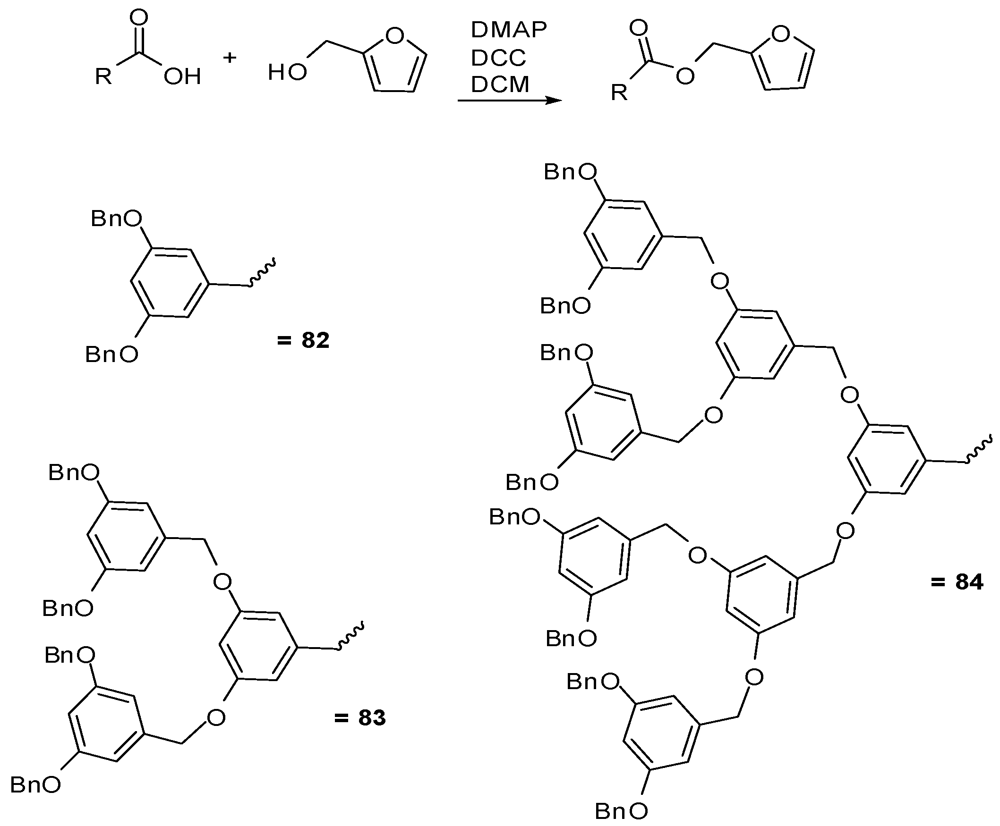
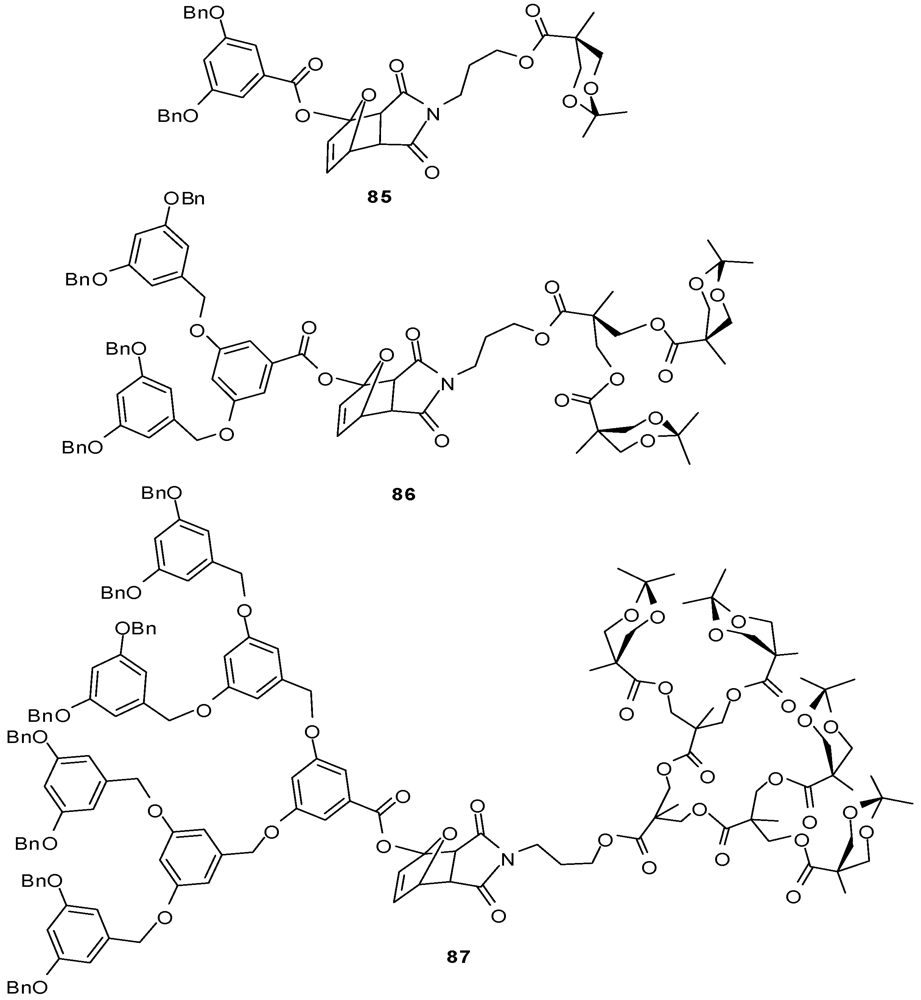

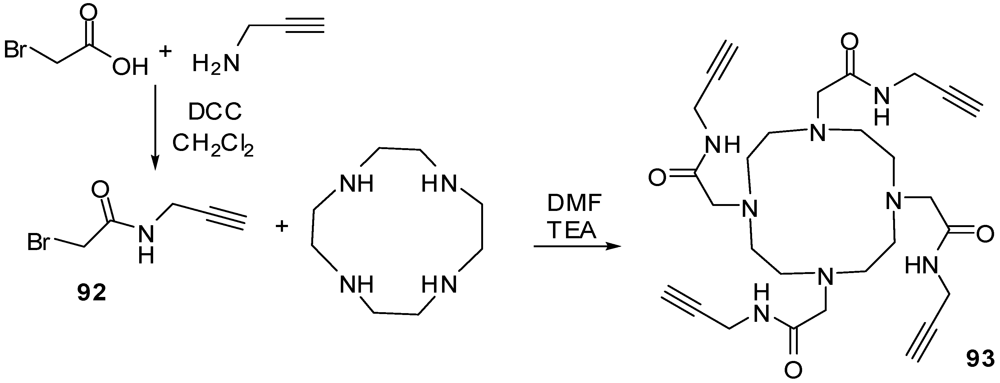
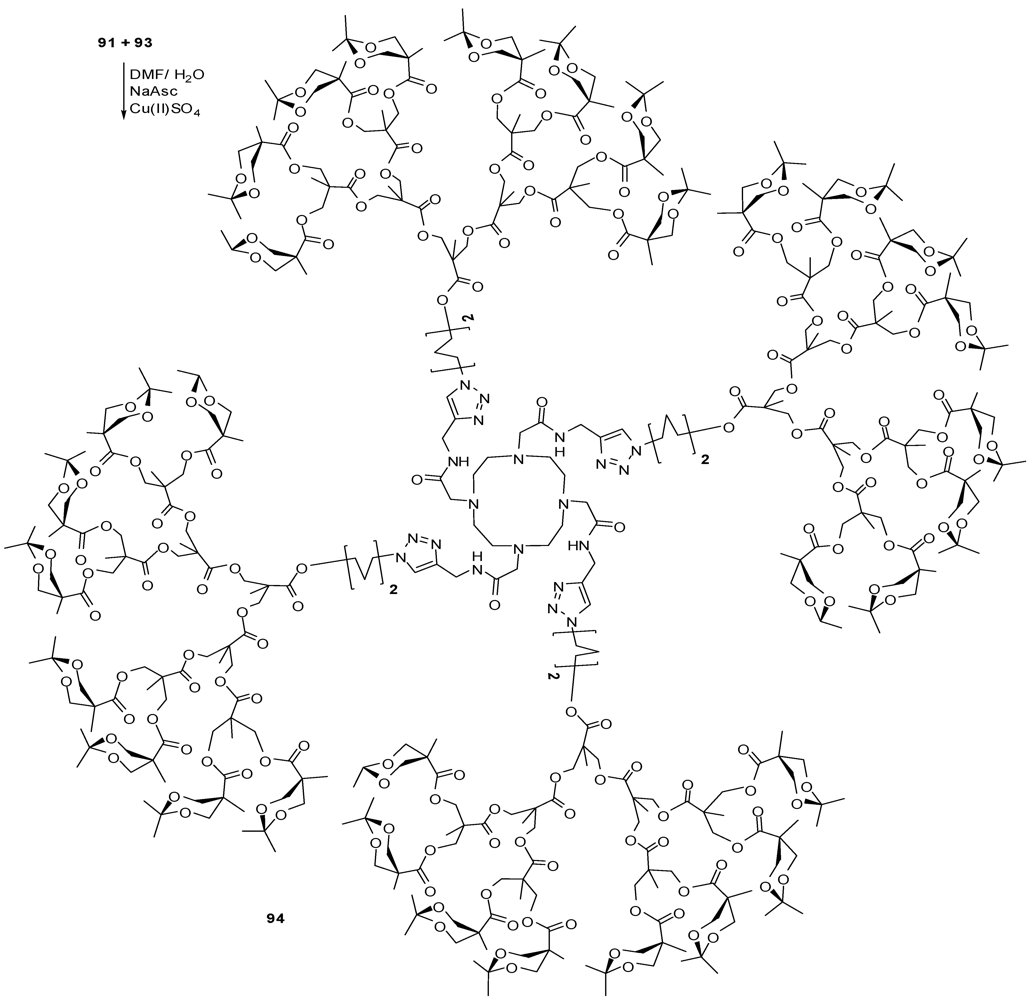



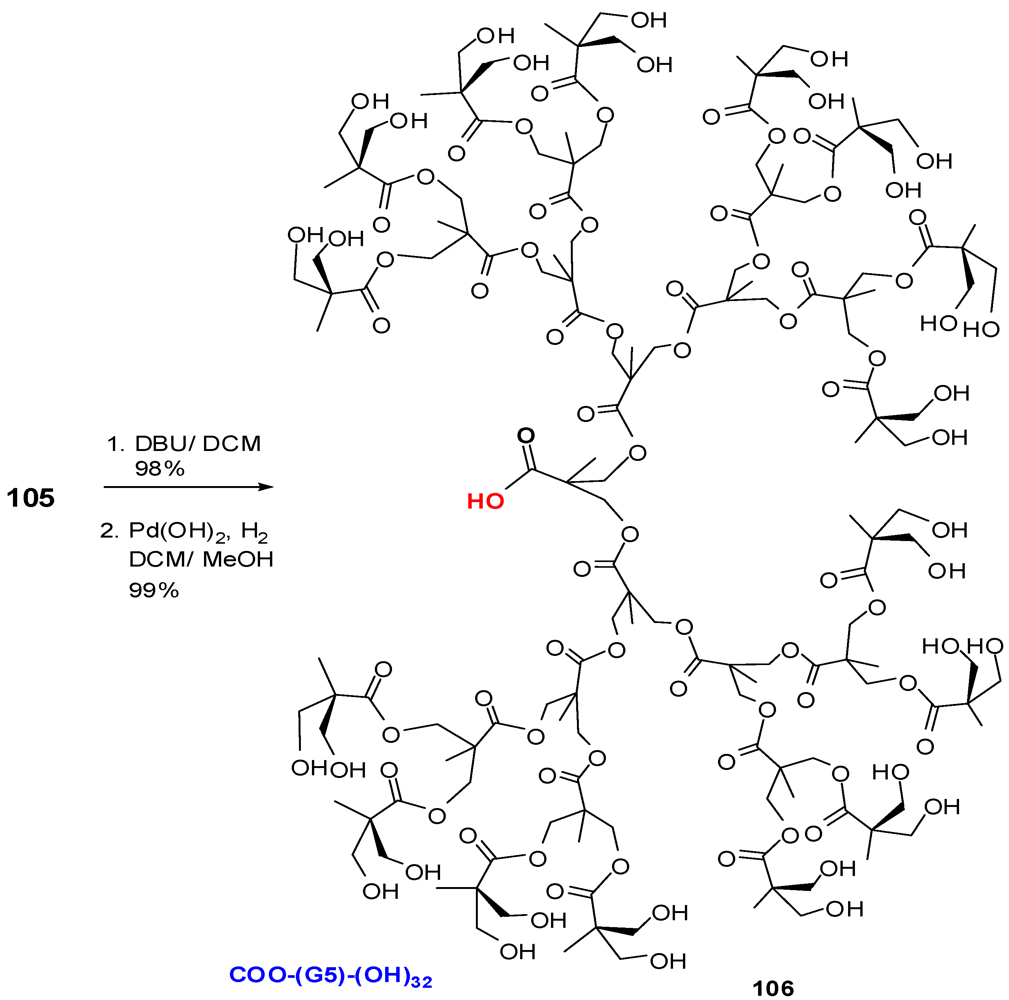



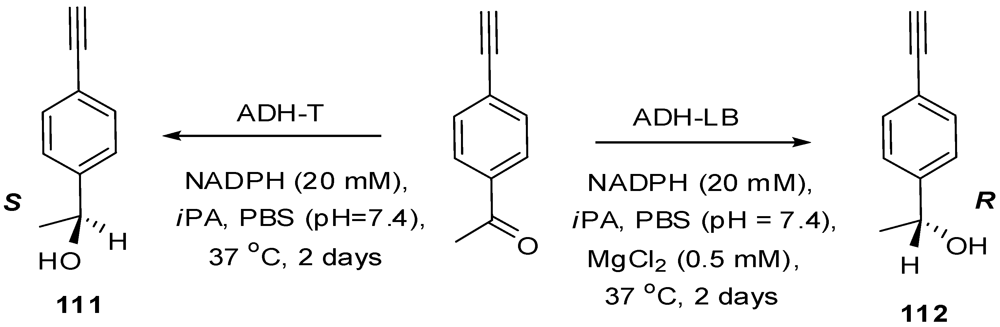
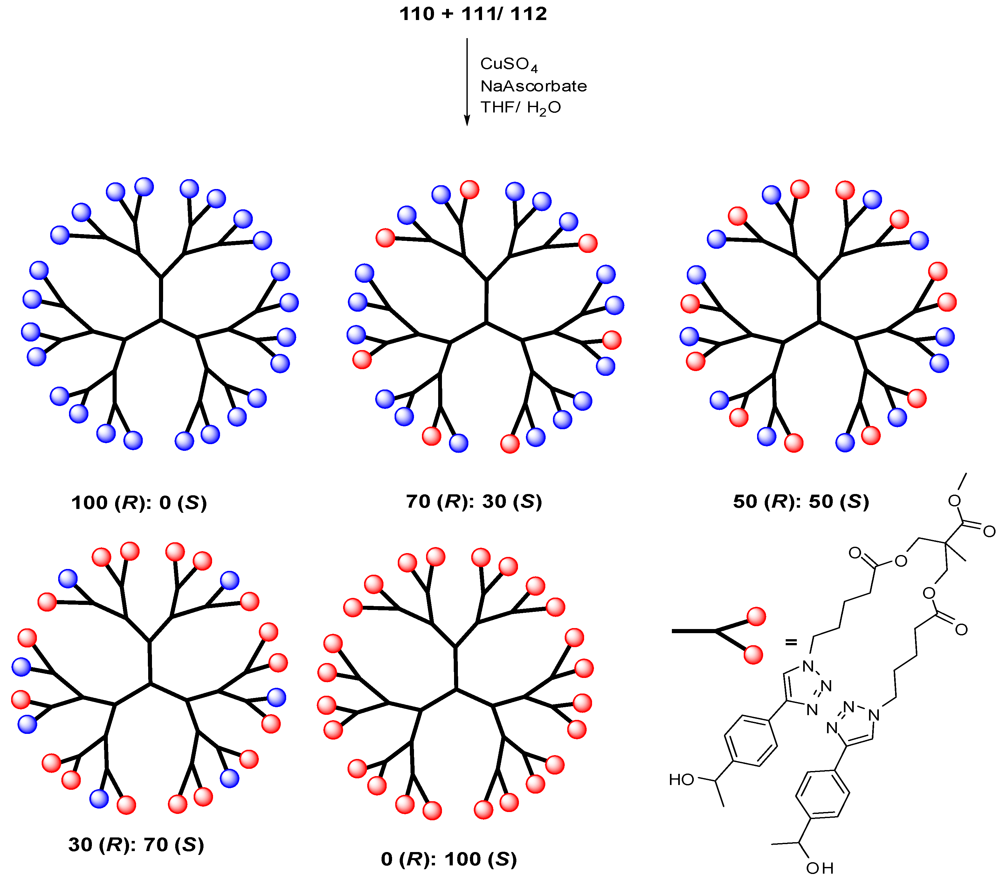
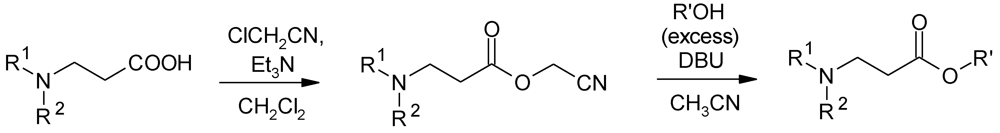
4.2. The Use of Other Aliphatic Dendrons
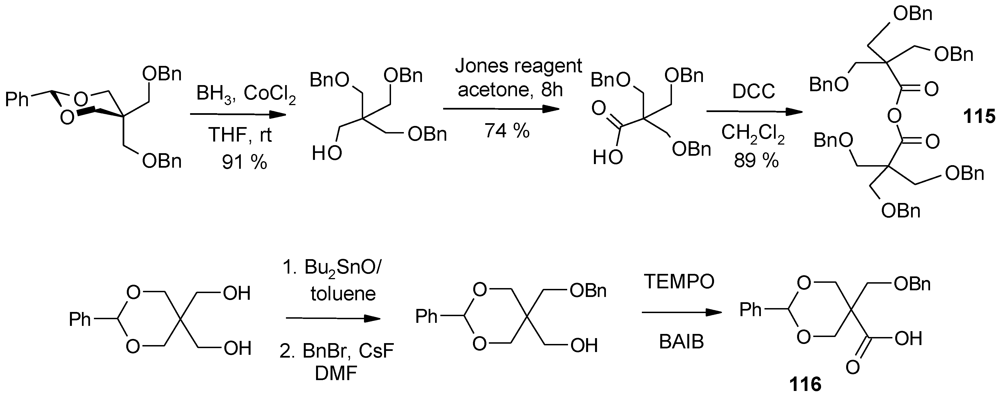
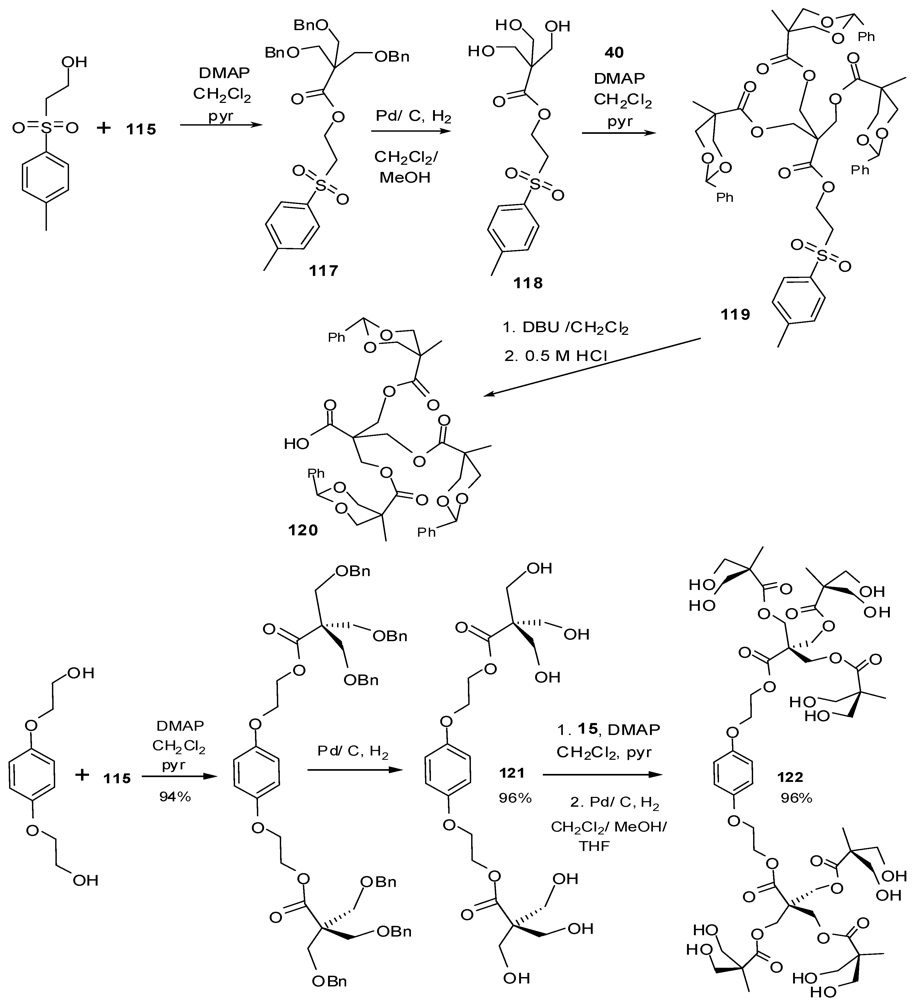
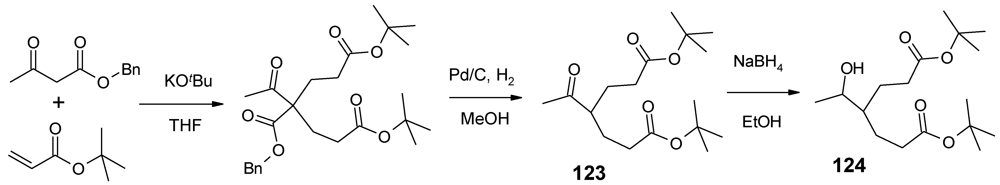


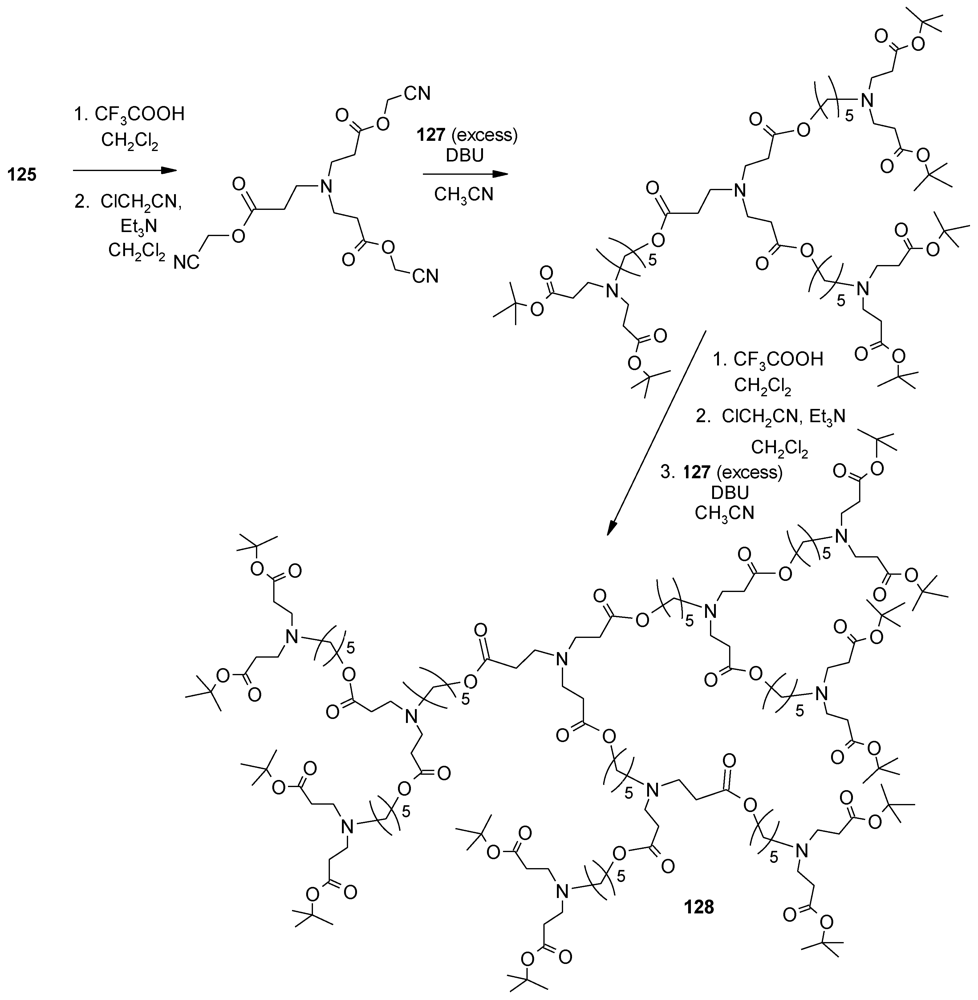
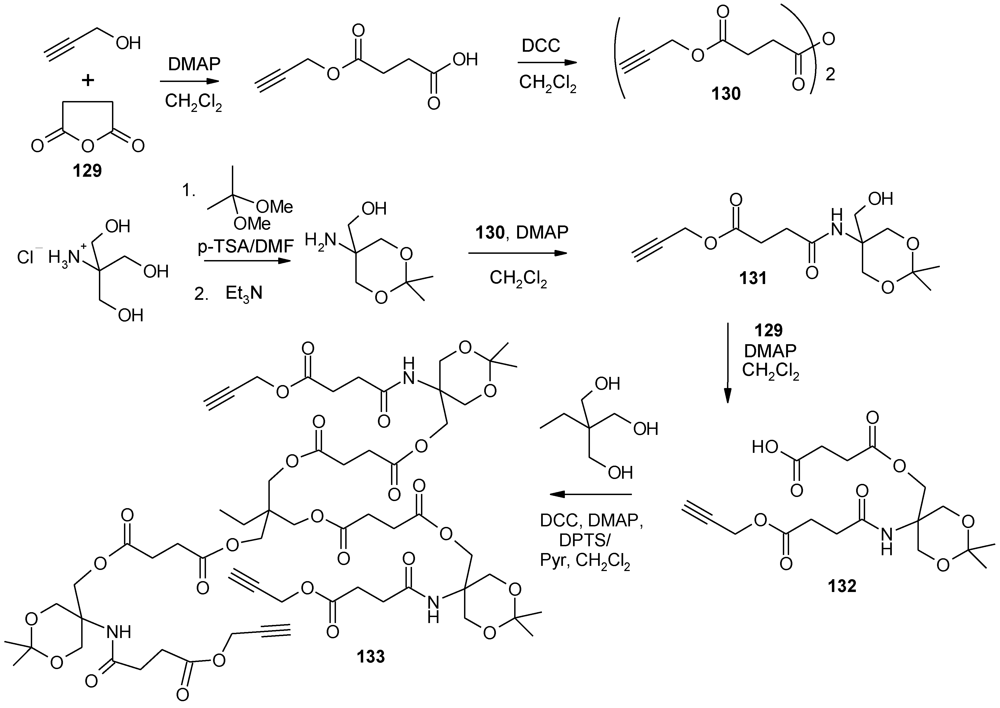
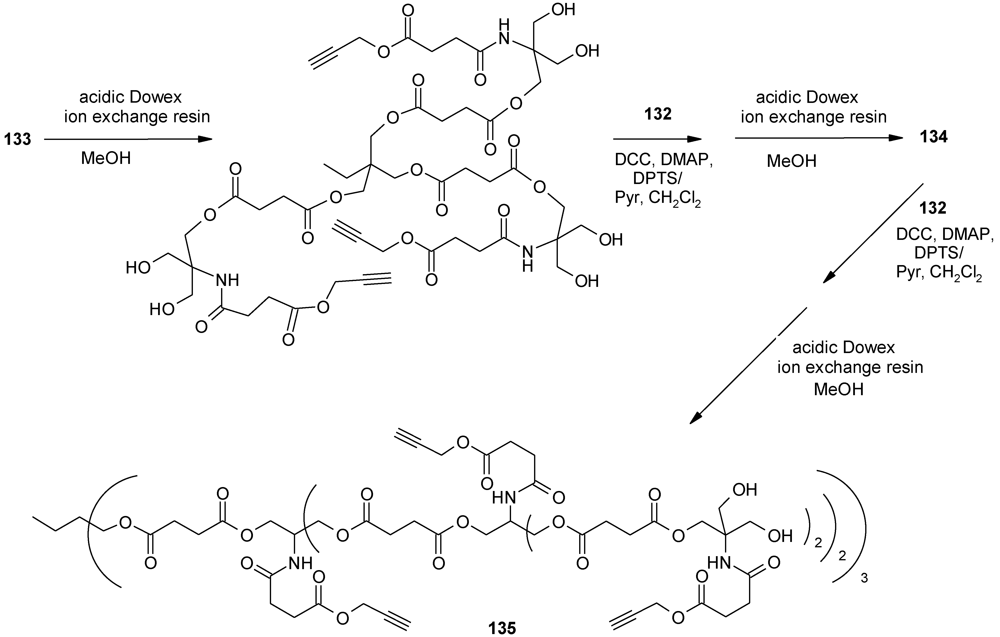
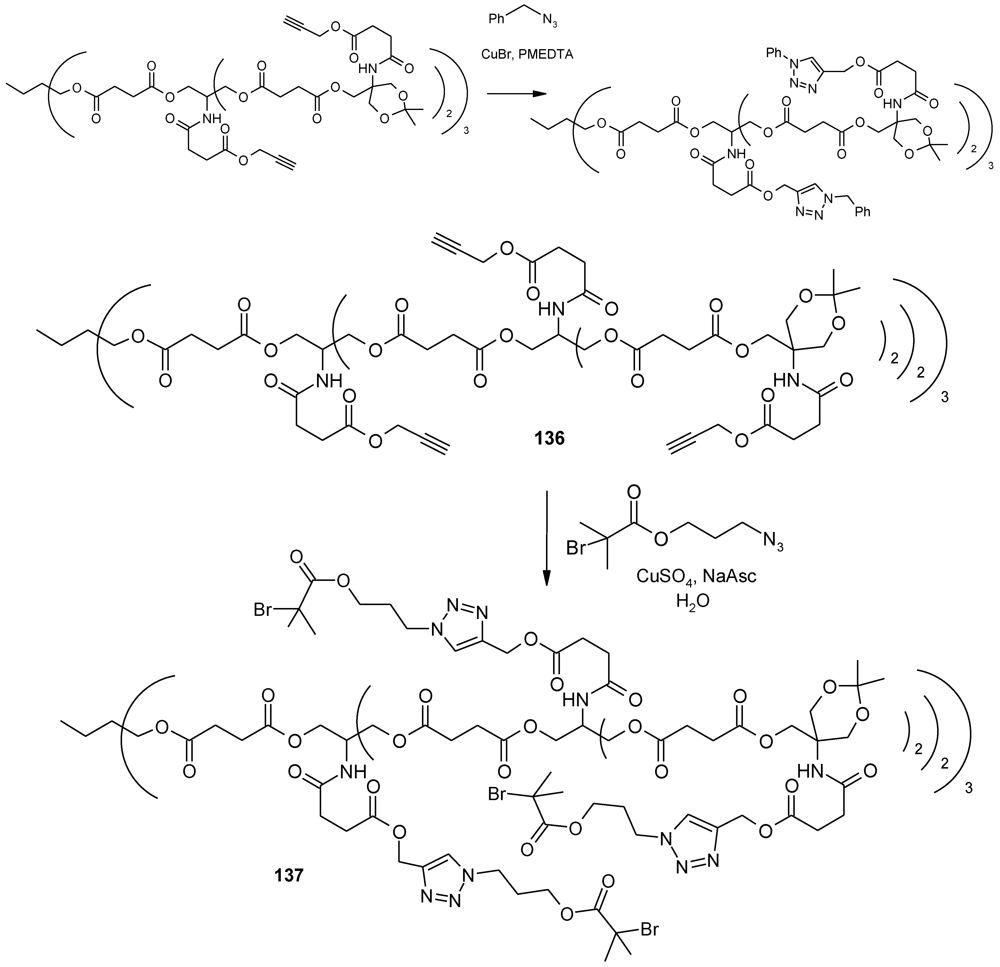
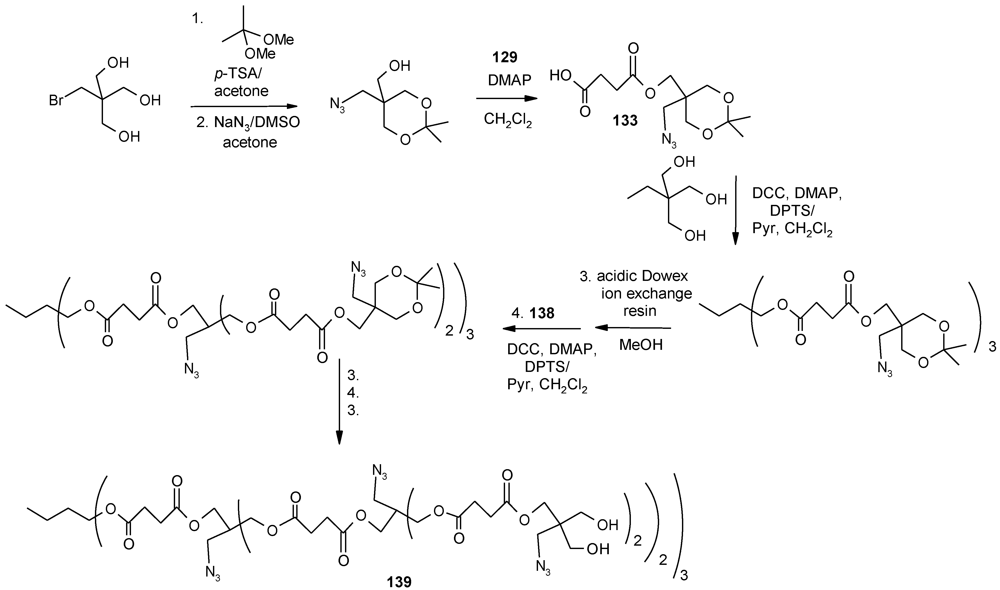
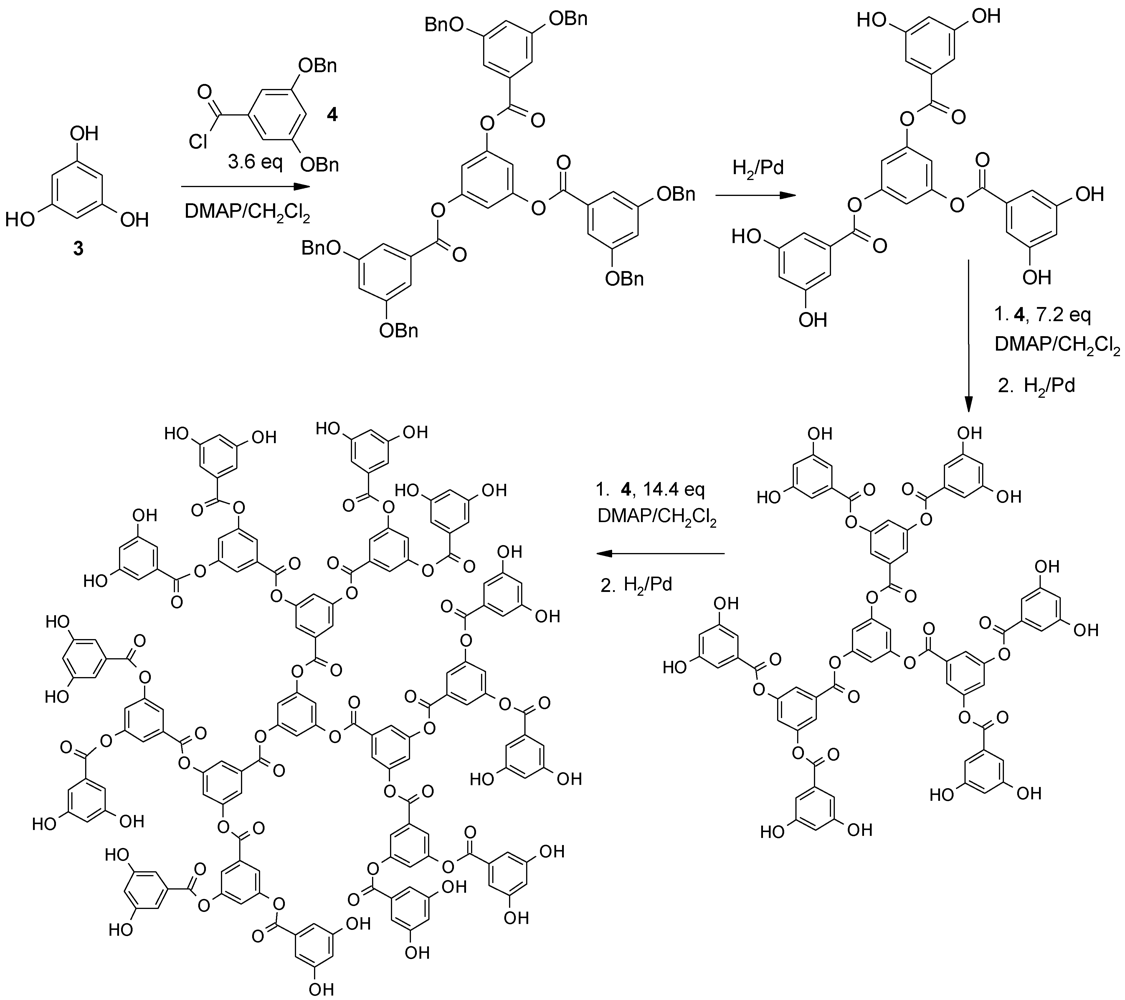
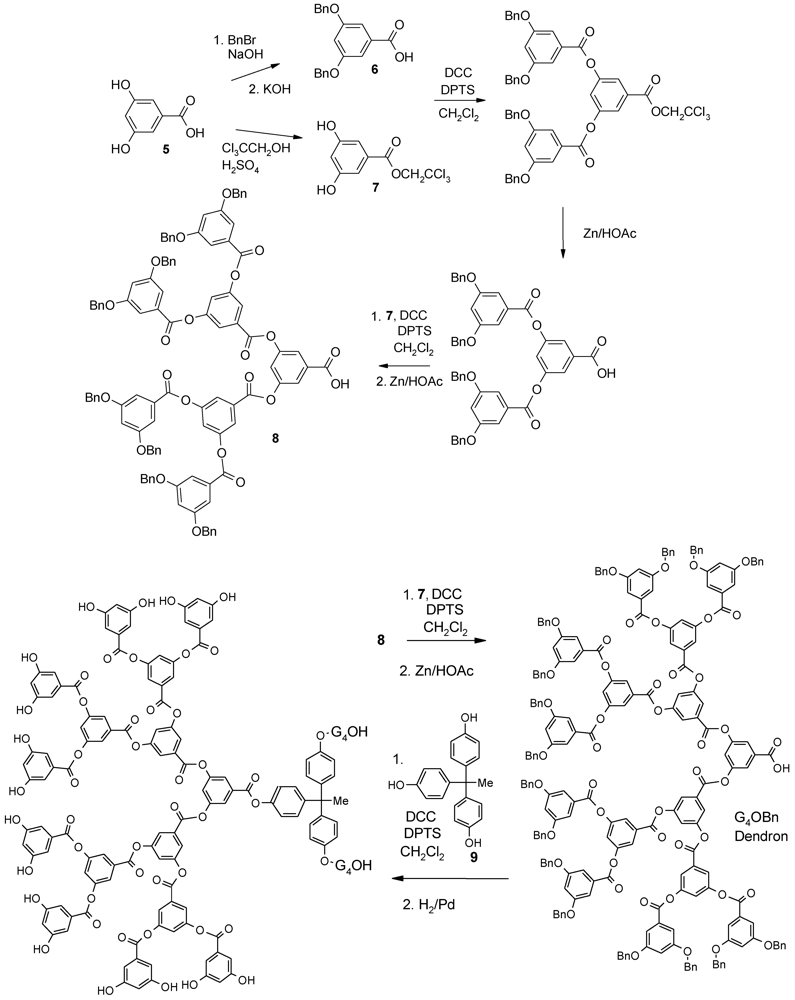
4.3. The Use of Aromatic Dendrons
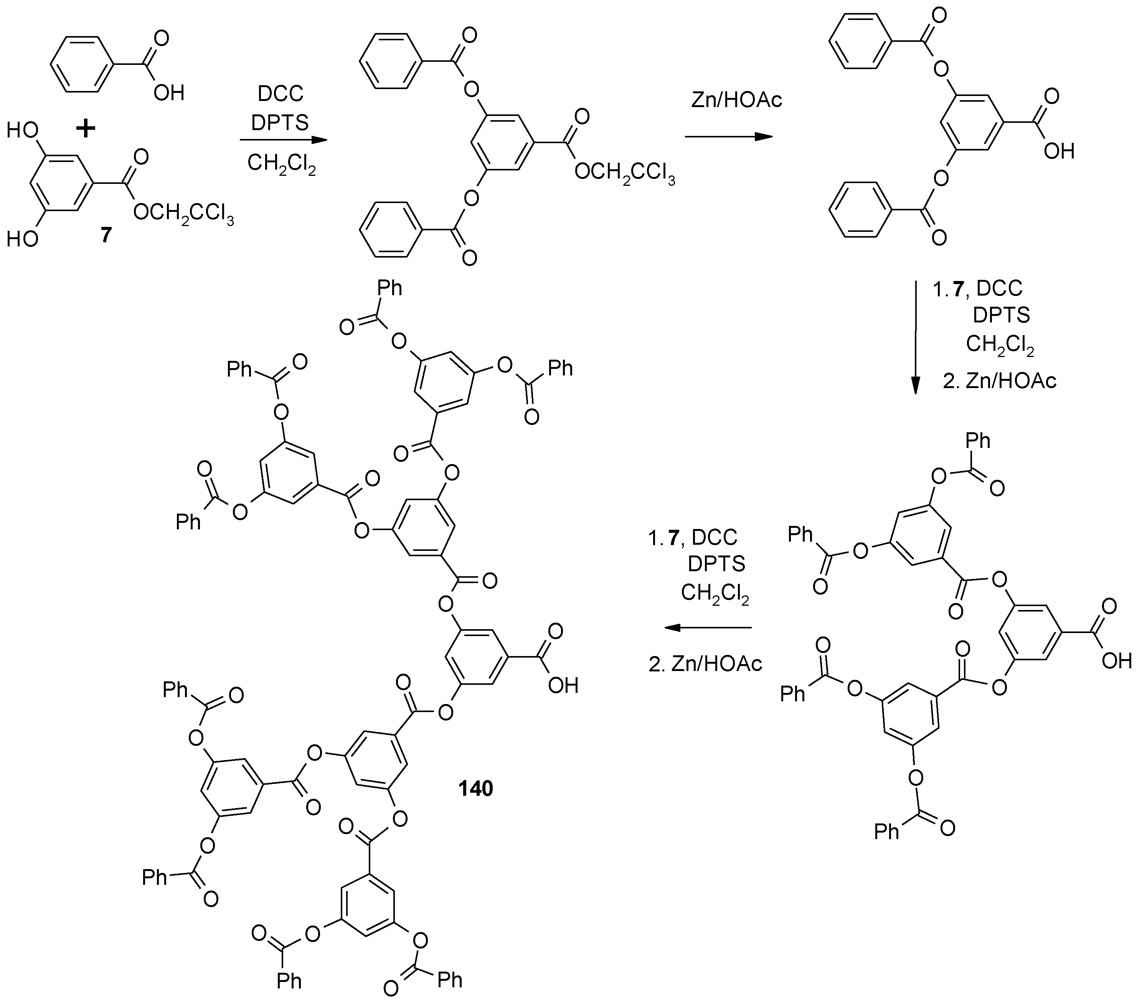
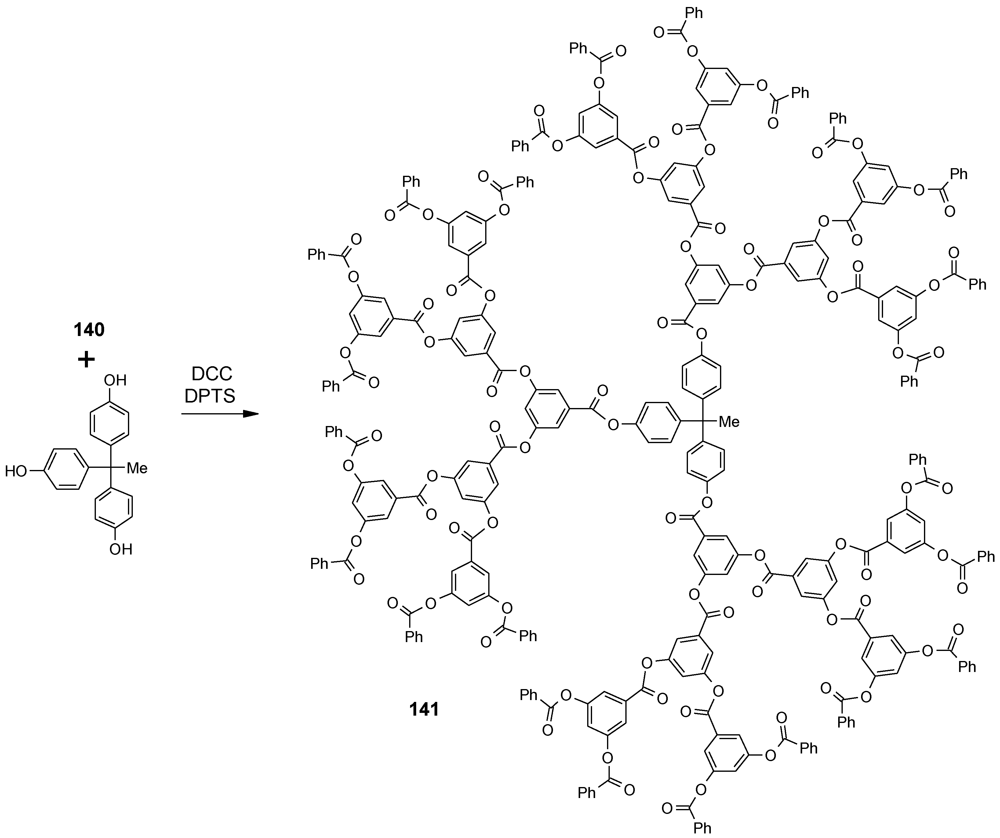
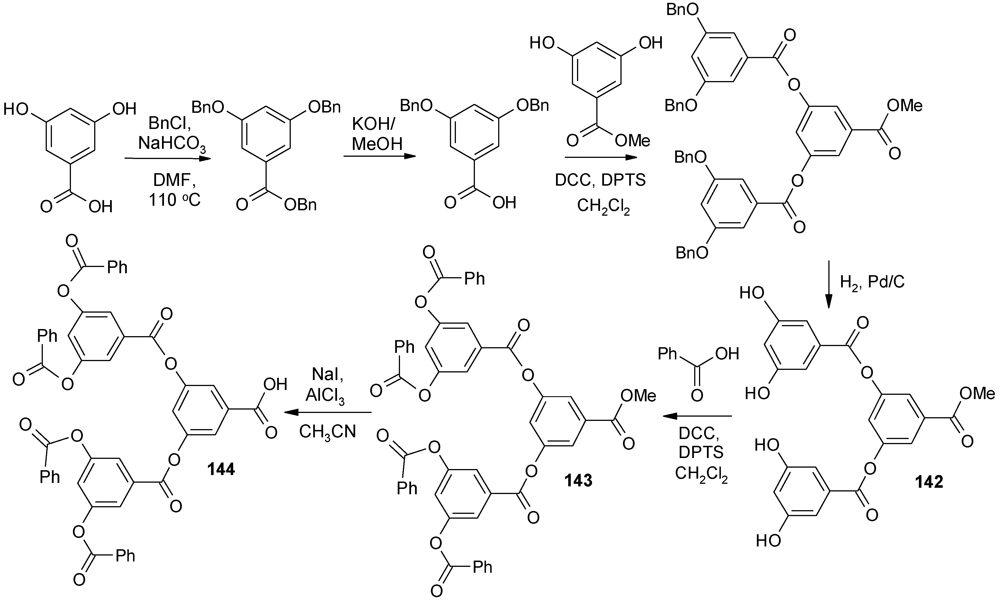


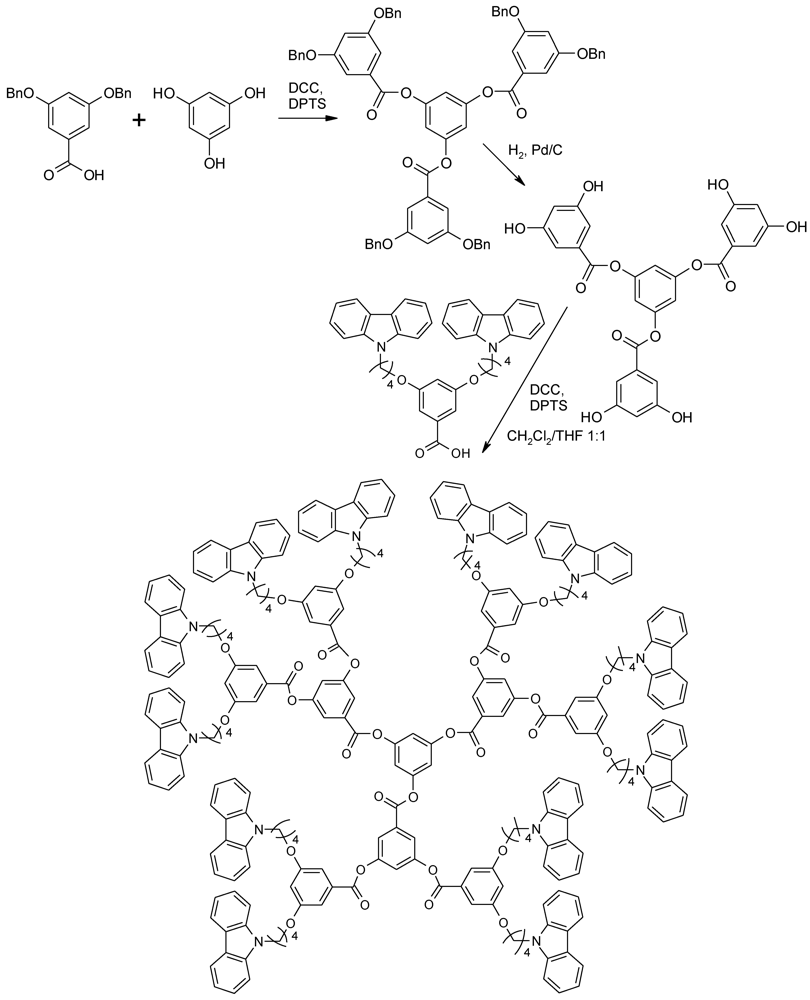
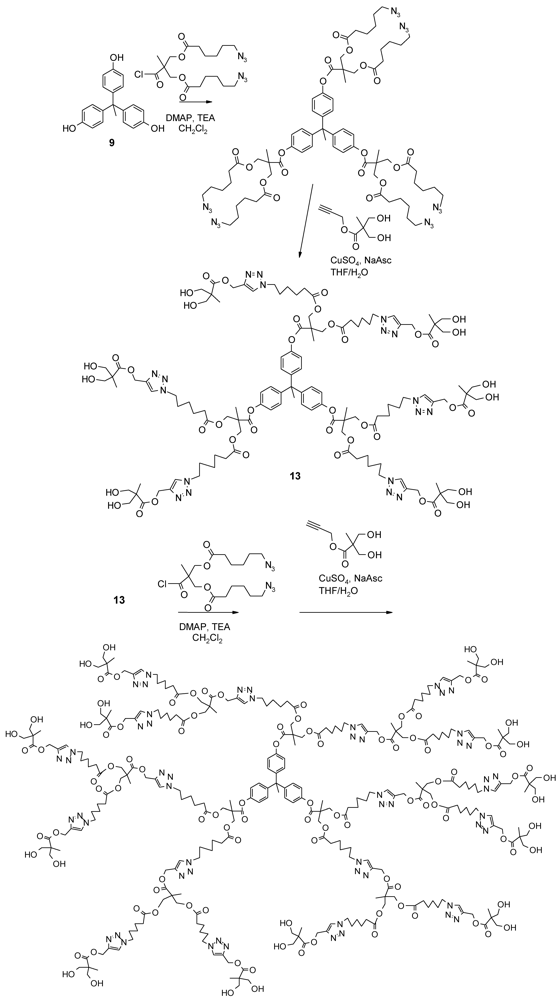
4.4. Alternating Polyester Dendrimers
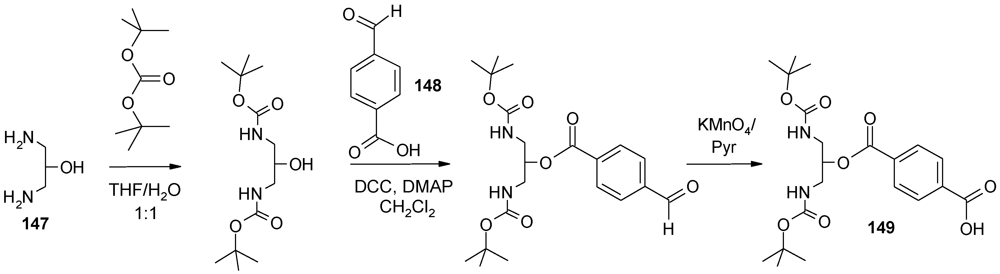








5. Conclusions
Acknowledgments
References
- Buhleier, E.; Wehner, W.; Vögtle, F. “Cascade”- and “nonskid-chain-like” syntheses of molecular cavity topologies. Synthesis 1978, 1978, 155–158. [Google Scholar]
- Denkewalter, R.G.; Kolc, J.; Kukasavage, W.J. Macromolecular highly branched homogeneous compound based on lysine units. U.S. Patent 4289872, 15 September 1981. [Google Scholar]
- Tomalia, D.A.; Dewald, J.R.; Hall, M.R.; Martin, S.J.; Smith, P.B. 1st International Polymer Conference, Kyoto, Japan, August 1984; Society of Polymer Science of Japan; p. 65.
- Tomalia, D.A.; Baker, H.; Dewald, J.; Hall, M.; Kallos, G.; Martin, S.; Roeck, J.; Ryder, J.; Smith, P. A new class of polymers: Starburst-dendritic macromolecules. Polym. J. 1985, 17, 117–132. [Google Scholar] [CrossRef]
- Newkome, G.R.; Yao, Z.Q.; Baker, G.R.; Gupta, V.K. Micelles. 1. Cascade molecules—A new approach to micelles—A [27]-arborol. J. Org. Chem. 1985, 50, 2003–2004. [Google Scholar]
- Tomalia, D.A.; Naylor, A.M.; Goddard, W.A. Starburst dendrimers—Molecular-level control of size, shape, surface chemistry, topology, and flexibility from atoms to macroscopic matter. Angew. Chem. Int. Ed. 1990, 29, 138–175. [Google Scholar] [CrossRef]
- Hawker, C.J.; Fréchet, J.M.J. Unusual macromolecular architectures—The convergent growth approach to dendritic polyesters and novel block copolymers. J. Am. Chem. Soc. 1992, 114, 8405–8413. [Google Scholar] [CrossRef]
- Hawker, C.J.; Fréchet, J.M.J. Monodispersed dendritic polyesters with removable chain ends—A versatile approach to globular macromolecules with chemically reversible polarities. J. Chem. Soc. Perkin Trans. 1 1992, 2459–2469. [Google Scholar]
- Fréchet, J.M.J.; Tomalia, D.A. Dendrimers and Other Dendritic Polymers; Wiley: Hoboken, NJ, USA, 2001; p. 647. [Google Scholar]
- Newkome, G.R.; Morefield, C.N.; Vögtle, F. Dendritic Molecules: Concepts, Synthesis, Perspectives; Wiley-VCH: New York, NY, USA and Weinheim, Germany, 2001; p. 261. [Google Scholar]
- Vögtle, F.; Richardt, G.; Werner, N. Dendrimer Chemistry: Concepts, Syntheses, Properties, Applications; Wiley-VCH: Weinheim, Germany, 2009; p. 342. [Google Scholar]
- Nummelin, S.; Skrifvars, M.; Rissanen, K. Polyester and ester functionalized dendrimers. Topics Curr. Chem. 2000, 210, 1–67. [Google Scholar] [CrossRef]
- Newkome, G.R.; Shreiner, C. Dendrimers derived from 1 to 3 branching motifs. Chem. Rev. 2010, 110, 6338–6442. [Google Scholar] [CrossRef]
- Astruc, D.; Boisselier, E.; Ornelas, C. Dendrimers designed for functions: From physical, photophysical, and supramolecular properties to applications in sensing, catalysis, molecular electronics, photonics, and nanomedicine. Chem. Rev. 2010, 110, 1857–1959. [Google Scholar] [CrossRef]
- Boas, U.; Heegaard, P.M.H. Dendrimers in drug research. Chem. Soc. Rev. 2004, 33, 43–63. [Google Scholar]
- Heegaard, P.M.H.; Boas, U.; Sorensen, N.S. Dendrimers for vaccine and immunostimulatory uses. A review. Bioconjugate Chem. 2010, 21, 405–418. [Google Scholar] [CrossRef]
- Fréchet, J.M.J. Dendrimers and other dendritic macromolecules: From building blocks to functional assemblies in nanoscience and nanotechnology. J. Polym. Sci. Part A 2003, 41, 3713–3725. [Google Scholar] [CrossRef]
- Grayson, S.M.; Fréchet, J.M.J. Convergent dendrons and dendrimers: From synthesis to applications. Chem. Rev. 2001, 101, 3819–3867. [Google Scholar] [CrossRef]
- Menjoge, A.R.; Kannan, R.M.; Tomalia, D.A. Dendrimer-based drug and imaging conjugates: Design considerations for nanomedical applications. Drug Discov. Today 2010, 15, 171–185. [Google Scholar] [CrossRef]
- Tomalia, D.A. Birth of a new macromolecular architecture: Dendrimers as quantized building blocks for nanoscale synthetic polymer chemistry. Prog. Polym. Sci. 2005, 30, 294–324. [Google Scholar] [CrossRef]
- Medina, S.H.; El-Sayed, M.E.H. Dendrimers as carriers for delivery of chemotherapeutic agents. Chem. Rev. 2009, 109, 3141–3157. [Google Scholar] [CrossRef]
- Jang, W.D.; Selim, K.M.K.; Lee, C.H.; Kang, I.K. Bioinspired application of dendrimers: From bio-mimicry to biomedical applications. Prog. Polym. Sci. 2009, 34, 1–23. [Google Scholar] [CrossRef]
- Cameron, D.J.A.; Shaver, M.P. Aliphatic polyester polymer stars: Synthesis, properties and applications in biomedicine and nanotechnology. Chem. Soc. Rev. 2011, 40, 1761–1776. [Google Scholar]
- Scholl, M.; Kadlecova, Z.; Klok, H.A. Dendritic and hyperbranched polyamides. Prog. Polym. Sci. 2009, 34, 24–61. [Google Scholar] [CrossRef]
- Satija, J.; Sai, V.V.R.; Mukherji, S. Dendrimers in biosensors: Concept and applications. J. Mater. Chem. 2011, 21, 14367–14386. [Google Scholar] [CrossRef]
- Lin, Q.M.; Jiang, G.H.; Tong, K.K. Dendrimers in drug-delivery applications. Des. Monomers Polym. 2010, 13, 301–324. [Google Scholar] [CrossRef]
- Hourani, R.; Kakkar, A. Advances in the elegance of chemistry in designing dendrimers. Macromol. Rapid Commun. 2010, 31, 947–974. [Google Scholar] [CrossRef]
- Villalonga-Barber, C.; Micha-Screttas, M.; Steele, B.R.; Georgopoulos, A.; Demetzos, C. Dendrimers as biopharmaceuticals: Synthesis and properties. Curr. Top. Med. Chem. 2008, 8, 1294–1309. [Google Scholar] [CrossRef]
- Cheng, Y.Y.; Zhao, L.B.; Li, Y.W.; Xu, T.W. Design of biocompatible dendrimers for cancer diagnosis and therapy: Current status and future perspectives. Chem. Soc. Rev. 2011, 40, 2673–2703. [Google Scholar] [CrossRef]
- Soliman, G.M.; Sharma, A.; Maysinger, D.; Kakkar, A. Dendrimers and miktoarm polymers based multivalent nanocarriers for efficient and targeted drug delivery. Chem. Commun. 2011, 47, 9572–9587. [Google Scholar]
- Wolinsky, J.B.; Grinstaff, M.W. Therapeutic and diagnostic applications of dendrimers for cancer treatment. Adv. Drug Deliv. Rev. 2008, 60, 1037–1055. [Google Scholar] [CrossRef]
- Newkome, G.R.; Shreiner, C.D. Poly(amidoamine), polypropylenimine, and related dendrimers and dendrons possessing different 1 -> 2 branching motifs: An overview of the divergent procedures. Polymer 2008, 49, 1–173. [Google Scholar] [CrossRef]
- Jain, K.; Kesharwani, P.; Gupta, U.; Jain, N.K. Dendrimer toxicity: Let’s meet the challenge. Int. J. Pharm. 2010, 394, 122–142. [Google Scholar] [CrossRef]
- Nanjwade, B.K.; Bechra, H.M.; Derkar, G.K.; Manvi, F.V.; Nanjwade, V.K. Dendrimers: Emerging polymers for drug-delivery systems. Eur. J. Pharm. Sci. 2009, 38, 185–196. [Google Scholar] [CrossRef]
- Simanek, E.E.; Abdou, H.; Lalwani, S.; Lim, J.; Mintzer, M.; Venditto, V.J.; Vittur, B. The 8year thicket of triazine dendrimers: Strategies, targets and applications. Proc. R. Soc. A 2010, 466, 1445–1468. [Google Scholar] [CrossRef]
- Konkolewicz, D.; Monteiro, M.J.; Perrier, S. Dendritic and hyperbranched polymers from macromolecular units: Elegant approaches to the synthesis of functional polymers. Macromolecules 2011, 44, 7067–7087. [Google Scholar]
- Harvison, M.A.; Lowe, A.B. Combining RAFT radical polymerization and click/highly efficient coupling chemistries: A powerful strategy for the preparation of novel materials. Macromol. Rapid Commun. 2011, 32, 779–800. [Google Scholar] [CrossRef]
- Gillies, E.R.; Dy, E.; Fréchet, J.M.J.; Szoka, F.C. Biological evaluation of polyester dendrimer: Poly(ethylene oxide) “Bow-Tie” hybrids with tunable molecular weight and architecture. Mol. Pharm. 2005, 2, 129–138. [Google Scholar] [CrossRef]
- Morgan, M.T.; Carnahan, M.A.; Immoos, C.E.; Ribeiro, A.A.; Finkelstein, S.; Lee, S.J.; Grinstaff, M.W. Dendritic molecular capsules for hydrophobic compounds. J. Am. Chem. Soc. 2003, 125, 15485–15489. [Google Scholar]
- Antoni, P.; Hed, Y.; Nordberg, A.; Nyström, D.; von Holst, H.; Hult, A.; Malkoch, M. Bifunctional dendrimers: From robust synthesis and accelerated one-pot postfunctionalization strategy to potential applications. Angew. Chem. Int. Ed. 2009, 48, 2126–2130. [Google Scholar]
- Kienle, R.H.; Hovey, A.G. The polyhydric alcohol-polybasic acid reaction I Glycerol-phthalic anhydride. J. Am. Chem. Soc. 1929, 51, 509–519. [Google Scholar] [CrossRef]
- Smith, W. A new glyceride: Glycerine phthalate. J. Soc. Chem. Ind. 1901, 20, 1075–1076. [Google Scholar]
- Kienle, R.H.; van der Meulen, P.A.; Petke, F.E. Polyhydric alcohol-polybasic acid reaction. III. Further studies of the glycerol-phthalic anhydride reaction. J. Am. Chem. Soc. 1939, 61, 2258–2268. [Google Scholar]
- Kienle, R.H.; van der Meulen, P.A.; Petke, F.E. Polyhydric alcohol-polybasic acid reaction. IV. Glyceryl phthalate from phthalic acid. J. Am. Chem. Soc. 1939, 61, 2268–2271. [Google Scholar]
- Kienle, R.H.; Petke, F.E. The polyhydric alcohol-polybasic acid reaction. V. The glyceryl succinate and glyceryl maleate polyesters. J. Am. Chem. Soc. 1940, 62, 1053–1056. [Google Scholar] [CrossRef]
- Baekeland, L.H. The synthesis, constitution, and uses of bakelite. Ind. Eng. Chem. 1909, 1, 149–161. [Google Scholar] [CrossRef]
- Baekeland, L.H.; Bender, H.L. Phenol resins and resinoids. Ind. Eng. Chem. 1925, 17, 225–237. [Google Scholar] [CrossRef]
- Flory, P.J. Molecular size distribution in three dimensional polymers. I. Gelation. J. Am. Chem. Soc. 1941, 63, 3083–3090. [Google Scholar] [CrossRef]
- Flory, P.J. Molecular size distribution in three dimensional polymers. II. Trifunctional branching units. J. Am. Chem. Soc. 1941, 63, 3091–3096. [Google Scholar] [CrossRef]
- Flory, P.J. Molecular size distribution in three dimensional polymers. III. Tetrafunctional branching units. J. Am. Chem. Soc. 1941, 63, 3096–3100. [Google Scholar] [CrossRef]
- Stockmayer, W.H. Theory of molecular size distribution and gel formation in branched-chain polymers. J. Chem. Phys. 1943, 11, 45–55. [Google Scholar] [CrossRef]
- Flory, P.J. Molecular size distribution in three dimensional polymers. VI. Branched polymers containing A-R-Bf−1 type units. J. Am. Chem. Soc. 1952, 74, 2718–2723. [Google Scholar] [CrossRef]
- Schaefgen, J.R.; Flory, P.J. Synthesis of multichain polymers and investigation of their viscosities. J. Am. Chem. Soc. 1948, 70, 2709–2718. [Google Scholar] [CrossRef]
- Hawker, C.J.; Lee, R.; Fréchet, J.M.J. One-step synthesis of hyperbranched dendritic polyesters. J. Am. Chem. Soc. 1991, 113, 4583–4588. [Google Scholar] [CrossRef]
- Malmström, E.; Johansson, M.; Hult, A. Hyperbranched aliphatic polyesters. Macromolecules 1995, 28, 1698–1703. [Google Scholar] [CrossRef]
- Schlüter, A.D.; Rabe, J.P. Dendronized polymers: Synthesis, characterization, assembly at interfaces, and manipulation. Angew. Chem. Int. Ed. 2000, 39, 864–883. [Google Scholar] [CrossRef]
- Zhang, A.; Shu, L.; Bo, Z.; Schlüter, A.D. Dendronized polymers: Recent progress in synthesis. Macromol. Chem. Phys. 2003, 204, 328–339. [Google Scholar] [CrossRef]
- Frauenrath, H. Dendronized polymers—Building a new bridge from molecules to nanoscopic objects. Prog. Polym. Sci. 2005, 30, 325–384. [Google Scholar] [CrossRef]
- Zhang, A. Synthesis, characterization and applications of dendronized polymers. Prog. Chem. 2005, 17, 157–171. [Google Scholar]
- Carlmark, A.; Hawker, C.J.; Hult, A.; Malkoch, M. New methodologies in the construction of dendritic materials. Chem. Soc. Rev. 2009, 38, 352–362. [Google Scholar] [CrossRef]
- Gauthier, M. Arborescent polymers and other dendrigraft polymers: A journey into structural diversity. J. Polym. Sci. Part A 2007, 45, 3803–3810. [Google Scholar] [CrossRef]
- Wurm, F.; Frey, H. Linear-dendritic block copolymers: The state of the art and exciting perspectives. Prog. Polym. Sci. 2011, 36, 1–52. [Google Scholar] [CrossRef]
- Yan, D.; Gao, C.; Frey, H. Hyperbranched Polymers: Synthesis, Properties and Applications; John Wiley & Sons: New York, NY, USA, 2011; p. 480. [Google Scholar]
- Hult, A.; Johansson, M.; Malmström, E. Hyperbranched polymers. Adv. Polym. Sci. 1999, 143, 1–34. [Google Scholar] [CrossRef]
- Gao, C.; Yan, D. Hyperbranched polymers: From synthesis to applications. Prog. Polym. Sci. 2004, 29, 183–275. [Google Scholar] [CrossRef]
- Voit, B. Hyperbranched polymers—All problems solved after 15 years of research? J. Polym. Sci. Part A 2005, 43, 2679–2699. [Google Scholar] [CrossRef]
- Yates, C.R.; Hayes, W. Synthesis and applications of hyperbranched polymers. Eur. Polym. J. 2004, 40, 1257–1281. [Google Scholar] [CrossRef]
- McKee, M.G.; Unal, S.; Wilkes, G.L.; Long, T.E. Branched polyesters: Recent advances in synthesis and performance. Prog. Polym. Sci. 2005, 30, 507–539. [Google Scholar] [CrossRef]
- Voit, B.I.; Lederer, A. Hyperbranched and highly branched polymer architectures-synthetic strategies and major characterization aspects. Chem. Rev. 2009, 109, 5924–5973. [Google Scholar] [CrossRef]
- Zhang, X. Hyperbranched aromatic polyesters: From synthesis to applications. Prog. Org. Coat. 2010, 69, 295–309. [Google Scholar]
- Calderón, M.; Quadir, M.A.; Strumia, M.; Haag, R. Functional dendritic polymer architectures as stimuli-responsive nanocarriers. Biochimie 2010, 92, 1242–1251. [Google Scholar] [CrossRef]
- Žagar, E.; Žigon, M. Aliphatic hyperbranched polyesters based on 2,2-bis(methylol)propionic acid-Determination of structure, solution and bulk properties. Prog. Polym. Sci. 2011, 36, 53–88. [Google Scholar] [CrossRef]
- Binauld, S.; Damiron, D.; Connal, L.A.; Hawker, C.J.; Drockenmuller, E. Precise synthesis of molecularly defined oligomers and polymers by orthogonal iterative divergent/convergent approaches. Macromol. Rapid Commun. 2011, 32, 147–168. [Google Scholar] [CrossRef]
- Zhang, X. Modifications and applications of hyperbranched aliphatic polyesters based on dimethylolpropionic acid. Polym. Int. 2011, 60, 153–166. [Google Scholar] [CrossRef]
- Newkome, G.R.; Nayak, A.; Behera, R.K.; Moorefield, C.N.; Baker, G.R. Chemistry of micelles series. 22. Cascade polymers—synthesis and characterization of 4-directional spherical dendritic macromolecules based on adamantane. J. Org. Chem. 1992, 57, 358–362. [Google Scholar]
- De Brabander-van den Berg, E.M.M.; Meijer, E.W. Poly(propylene imine) dendrimers: Large-scale synthesis by hetereogeneously catalyzed hydrogenations. Angew. Chem. Int. Ed. 1993, 32, 1308–1311. [Google Scholar] [CrossRef]
- Hawker, C.J.; Fréchet, J.M.J. Preparation of polymers with controlled molecular architecture—A new convergent approach to dendritic macromolecules. J. Am. Chem. Soc. 1990, 112, 7638–7647. [Google Scholar]
- Gillies, E.R.; Fréchet, J.M.J. Dendrimers and dendritic polymers in drug delivery. Drug Discov. Today 2005, 10, 35–43. [Google Scholar] [CrossRef]
- Lee, C.C.; Gillies, E.R.; Fox, M.E.; Guillaudeu, S.J.; Fréchet, J.M.J.; Dy, E.E.; Szoka, F.C. A single dose of doxorubicin-functionalized bow-tie dendrimer cures mice bearing C-26 colon carcinomas. Proc. Nat. Acad. Sci. USA 2006, 103, 16649–16654. [Google Scholar]
- Haddleton, D.M.; Sahota, H.S.; Taylor, P.C.; Yeates, S.G. Synthesis of polyester dendrimers. J. Chem. Soc. Perkin Trans. 1 1996, 649–656. [Google Scholar]
- Kawaguchi, T.; Walker, K.L.; Wilkins, C.L.; Moore, J.S. Double exponential dendrimer growth. J. Am. Chem. Soc. 1995, 117, 2159–2165. [Google Scholar]
- Klopsch, R.; Franke, P.; Schlüter, A.-D. Repetitive strategy for exponential growth of hydroxy-functionalized dendrons. Chem. Eur. J. 1996, 2, 1330–1334. [Google Scholar] [CrossRef]
- Ashton, P.R.; Hounsell, E.F.; Jayaraman, N.; Nilsen, T.M.; Spencer, N.; Stoddart, J.F.; Young, M. Synthesis and biological evaluation of α-D-mannopyranoside-containing dendrimers. J. Org. Chem. 1998, 63, 3429–3437. [Google Scholar]
- Ihre, H.; Hult, A.; Fréchet, J.M.J.; Gitsov, I. Double-stage convergent approach for the synthesis of functionalized dendritic aliphatic polyesters based on 2,2-bis(hydroxymethyl)propionic acid. Macromolecules 1998, 31, 4061–4068. [Google Scholar]
- Spindler, R.; Fréchet, J.M.J. 2-Step approach towards the accelerated synthesis of dendritic macromolecules. J. Chem. Soc. Perkin Trans. 1 1993, 913–918. [Google Scholar]
- Xu, Z.F.; Moore, J.S. Stiff dendritic macromolecules. 3. Rapid construction of large-size phenylacetylene dendrimers up to 12.5 nonometers in molecular diameter. Angew. Chem. Int. Ed. Engl. 1993, 32, 1354–1357. [Google Scholar]
- Zeng, F.W.; Zimmerman, S.C. Rapid synthesis of dendrimers by an orthogonal coupling strategy. J. Am. Chem. Soc. 1996, 118, 5326–5327. [Google Scholar]
- Deb, S.K.; Maddux, T.M.; Yu, L.P. A simple orthogonal approach to poly(phenylenevinylene) dendrimers. J. Am. Chem. Soc. 1997, 119, 9079–9080. [Google Scholar] [CrossRef]
- Klopsch, R.; Koch, S.; Schlüter, A.-D. Amino-functionalized, second-generation dendritic building blocks. Eur. J. Org. Chem. 1998, 1275–1283. [Google Scholar]
- Ingerl, A.; Neubert, I.; Klopsch, R.; Schlüter, A.D. Hydroxy-functionalized dendritic building blocks. Eur. J. Org. Chem. 1998, 2551–2556. [Google Scholar]
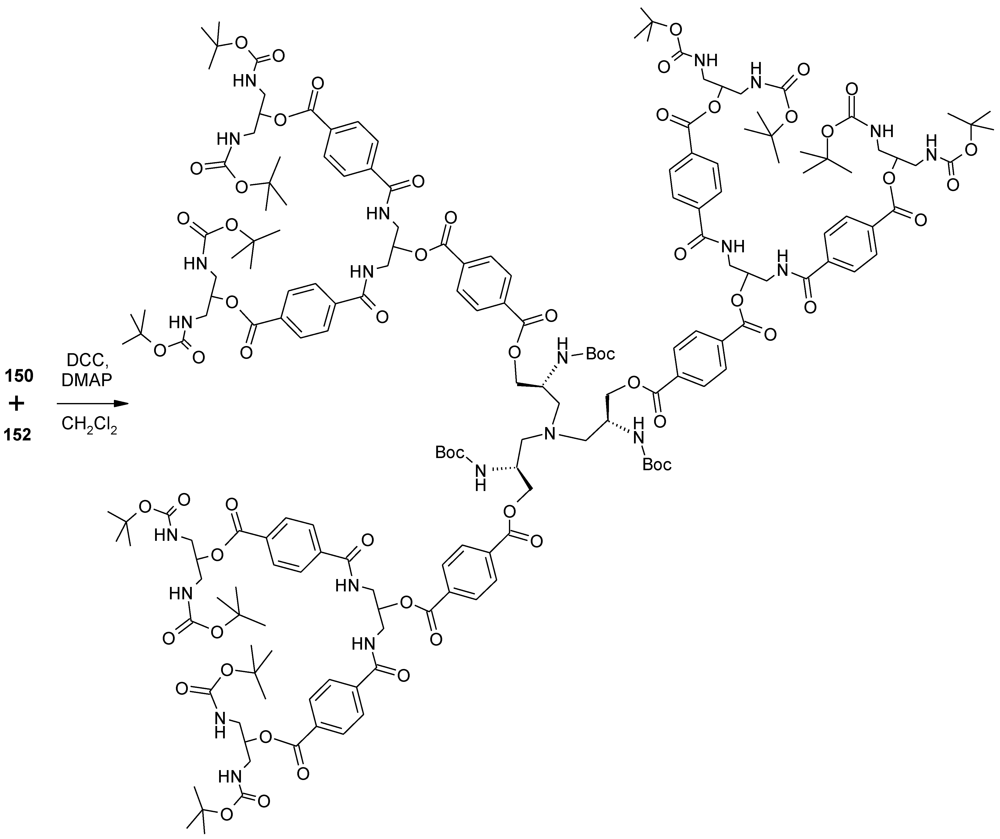
- Ishida, Y.; Jikei, M.; Kakimoto, M.A. Rapid synthesis of aromatic polyamide dendrimers by an orthogonal and a double-stage convergent approach. Macromolecules 2000, 33, 3202–3211. [Google Scholar] [CrossRef]
- Burai, R.; Chatwichien, J.; McNaughton, B.R. A programmable “build-couple” approach to the synthesis of heterofunctionalized polyvalent molecules. Org. Biomol. Chem. 2011, 9, 5056–5058. [Google Scholar]
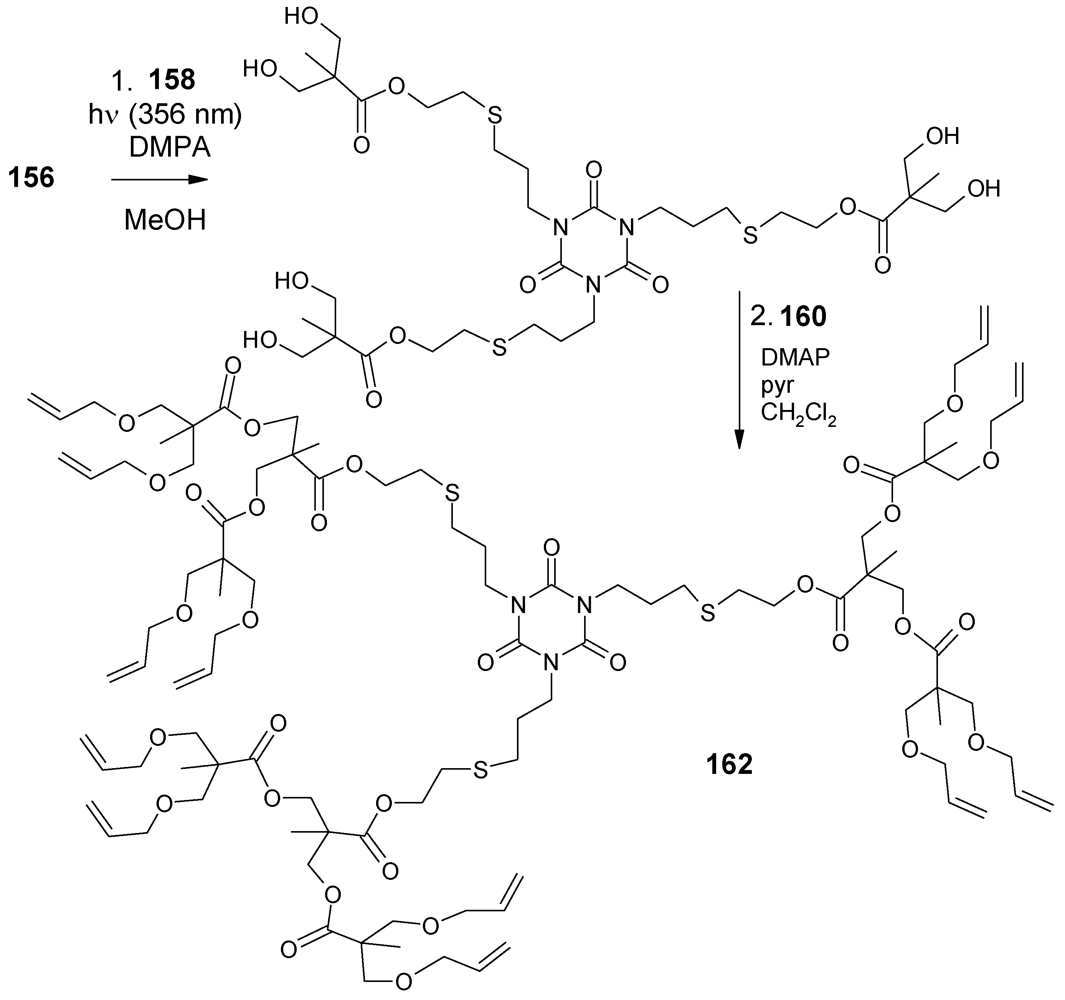
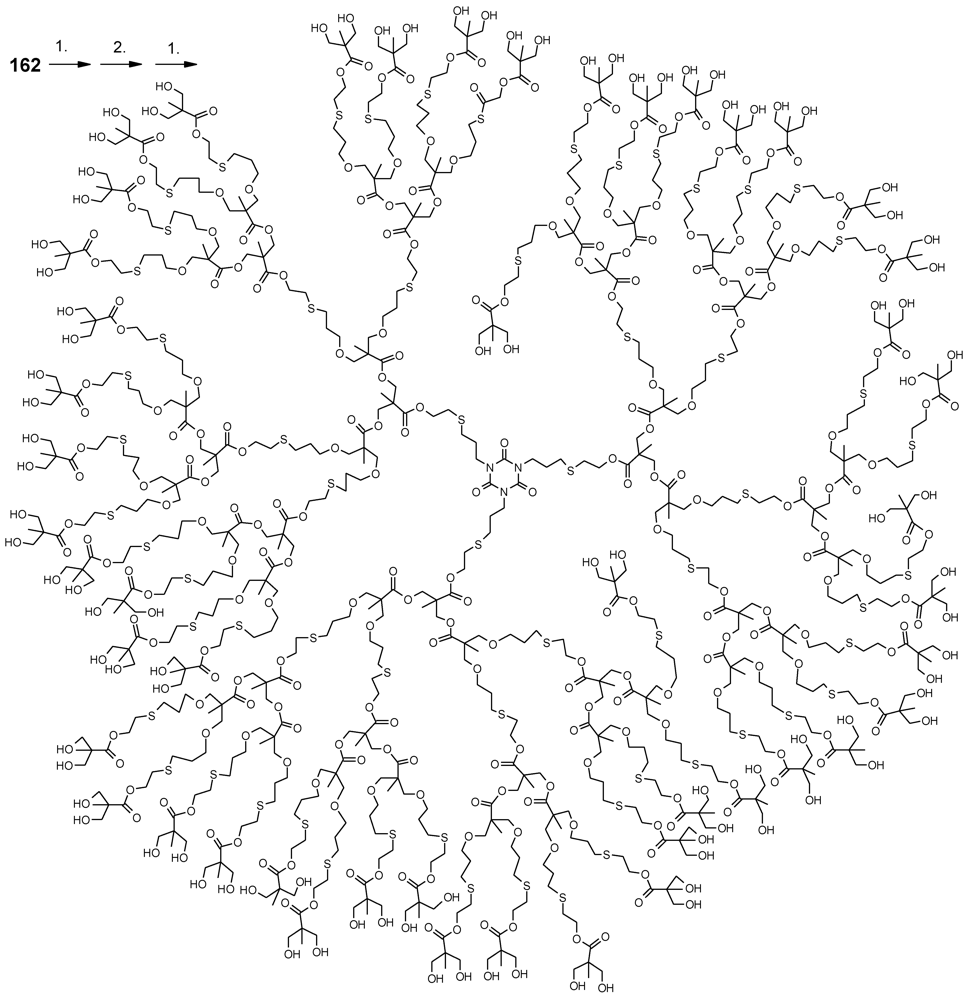
- Antoni, P.; Robb, M.J.; Campos, L.; Montanez, M.; Hult, A.; Malmström, E.; Malkoch, M.; Hawker, C.J. Pushing the limits for thiol-ene and CuAAC reactions: Synthesis of a 6th generation dendrimer in a single day. Macromolecules 2010, 43, 6625–6631. [Google Scholar]
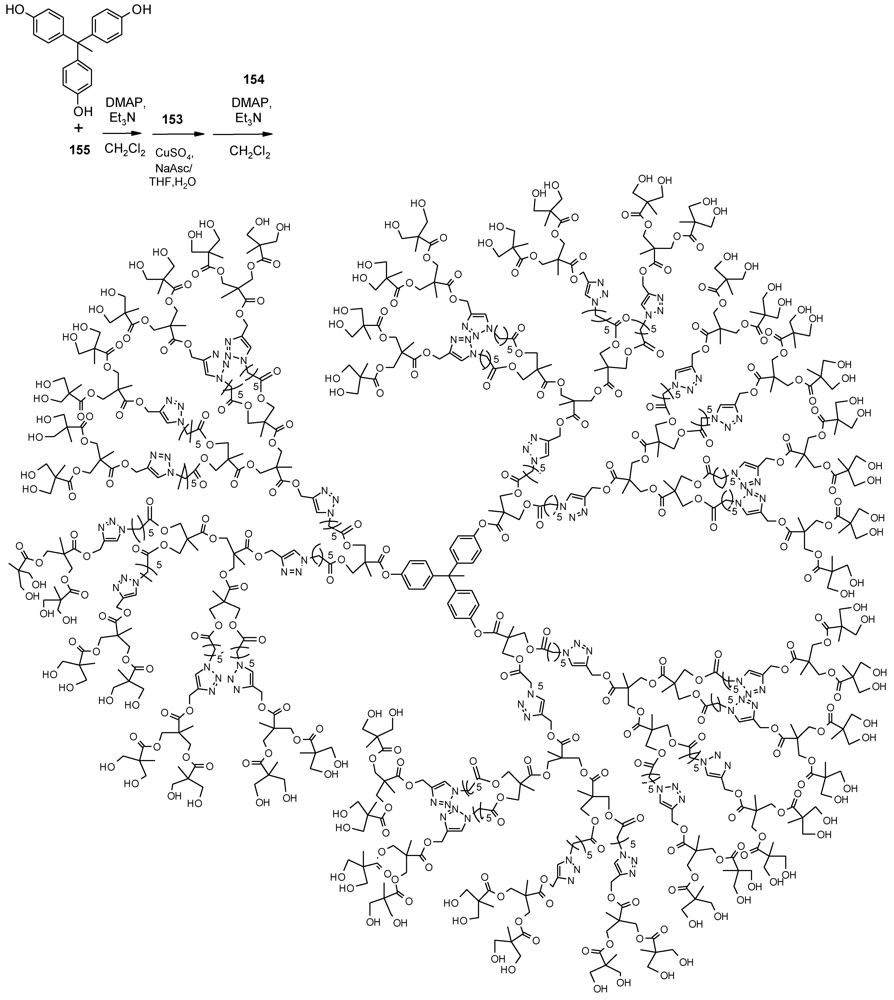
- Ma, X.; Tang, J.; Shen, Y.; Fan, M.; Tang, H.; Radosz, M. Facile synthesis of polyester dendrimers from sequential click coupling of asymmetrical monomers. J. Am. Chem. Soc. 2009, 131, 14795–14803. [Google Scholar]
- Rosen, B.M.; Lligadas, G.; Hahn, C.; Percec, V. Synthesis of dendrimers through divergent iterative thio-bromo “click” chemistry. J. Polym. Sci. Part A 2009, 47, 3931–3939. [Google Scholar] [CrossRef]
- In, I.; Kim, S.Y. Orthogonal synthesis of poly(aryl ether amide) dendrons. Macromolecules 2005, 38, 9399–9401. [Google Scholar] [CrossRef]
- Kozaki, M.; Okada, K. Snowflake-like dendrimers via site-selective synthesis of dendrons. Org. Lett. 2004, 6, 485–488. [Google Scholar] [CrossRef]
- Maraval, V.; Pyzowski, J.; Caminade, A.M.; Majoral, J.P. “Lego” chemistry for the straightforward synthesis of dendrimers. J. Org. Chem. 2003, 68, 6043–6046. [Google Scholar]
- Zhang, W.; Nowlan, D.T.; Thomson, L.M.; Lackowski, W.M.; Simanek, E.E. Orthogonal, convergent syntheses of dendrimers based on melamine with one or two unique surface sites for manipulation. J. Am. Chem. Soc. 2001, 123, 8914–8922. [Google Scholar]
- Leu, C.M.; Shu, C.F.; Teng, C.F.; Shiea, J. Dendritic poly(ether-imide)s: Synthesis, characterization, and modification. Polymer 2001, 42, 2339–2348. [Google Scholar]
- Wong, E.H.H.; Altintas, O.; Stenzel, M.H.; Barner-Kowollik, C.; Junkers, T. Nitrone-mediated radical coupling reactions: A new synthetic tool exemplified on dendrimer synthesis. Chem. Commun. 2011, 47, 5491–5493. [Google Scholar]
- Zeng, F.; Zimmerman, S.C. Rapid synthesis of dendrimers by an orthogonal coupling strategy. J. Am. Chem. Soc. 1996, 118, 5326–5327. [Google Scholar]
- Antoni, P.; Nyström, D.; Hawker, C.J.; Hult, A.; Malkoch, M. A chemoselective approach for the accelerated synthesis of well-defined dendritic architectures. Chem. Commun. 2007, 2249–2251. [Google Scholar]
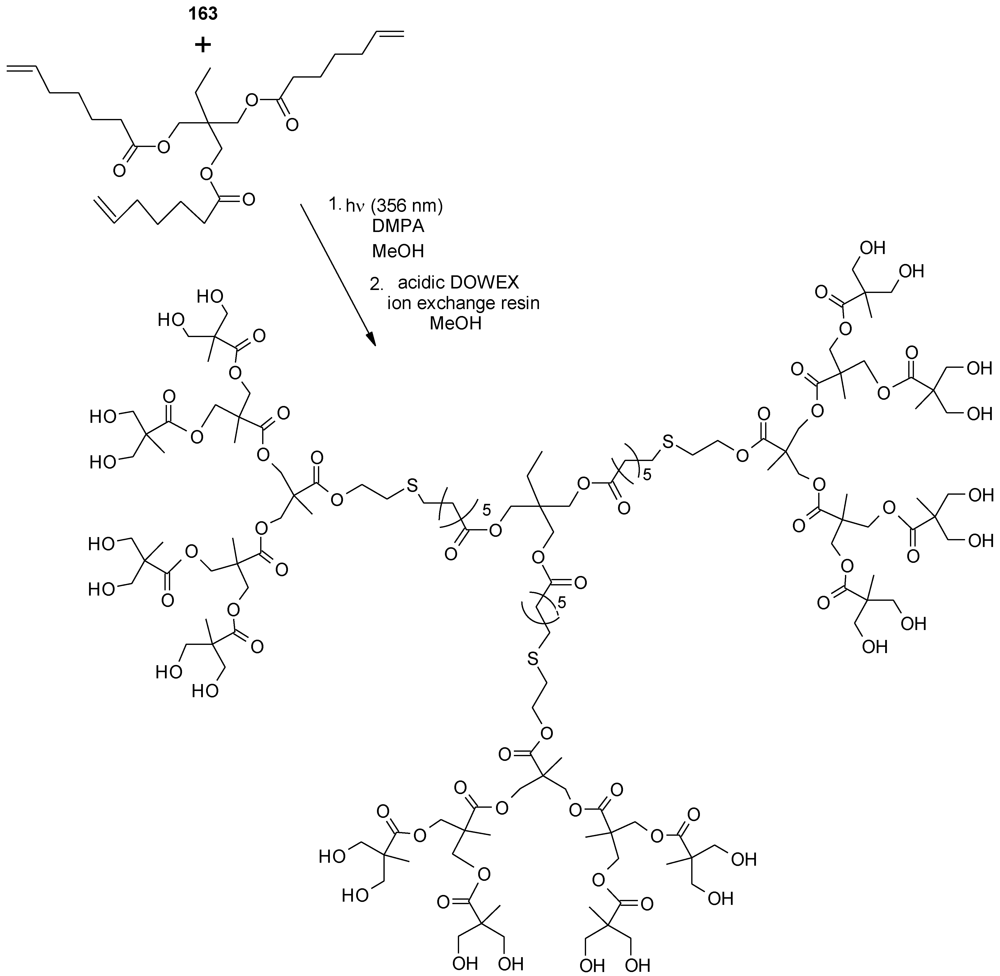
- Montañez, M.I.; Campos, L.M.; Antoni, P.; Hed, Y.; Walter, M.V.; Krull, B.T.; Khan, A.; Hult, A.; Hawker, C.J.; Malkoch, M. Accelerated growth of dendrimers via thiol-ene and esterification reactions. Macromolecules 2010, 43, 6004–6013. [Google Scholar]
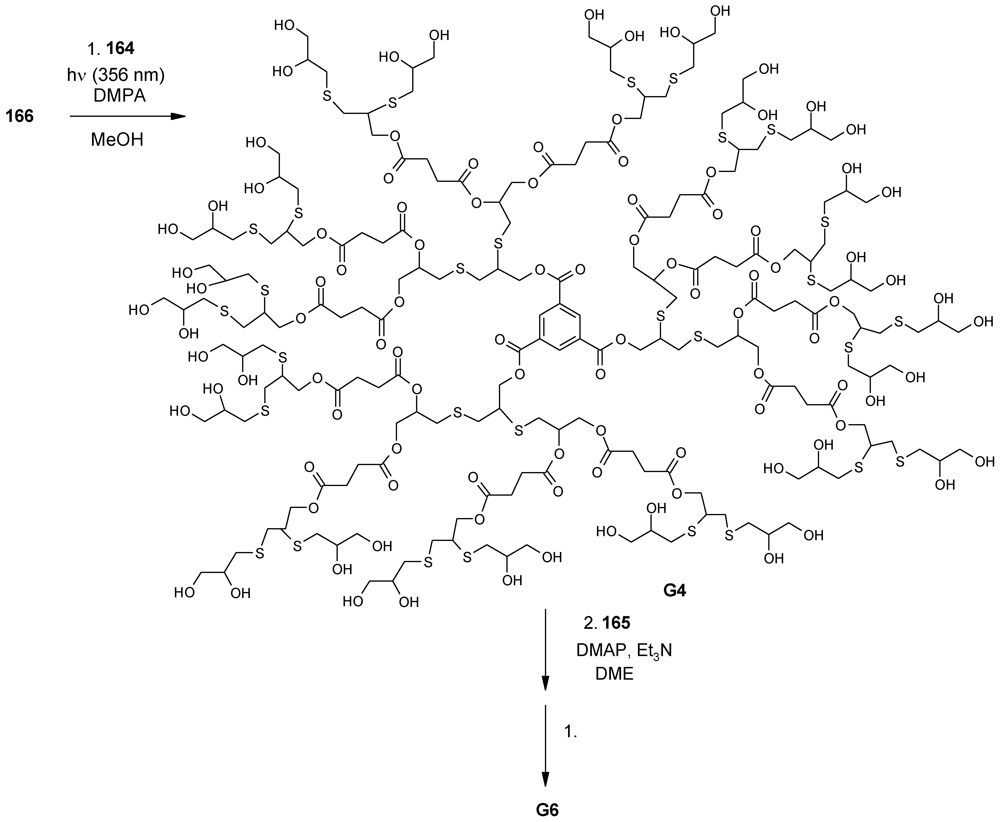
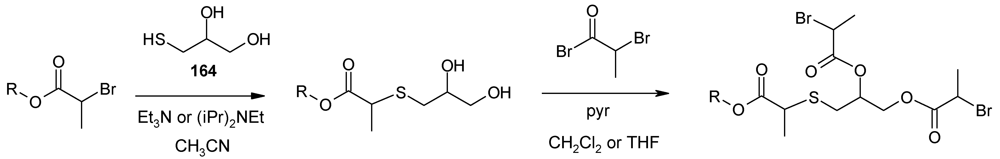
- Walter, M.V.; Lundberg, P.; Hult, A.; Malkoch, M. Novel macrothiols for the synthesis of a structurally comprehensive dendritic library using thiol-ene click chemistry. J. Polym. Sci. Part A 2011, 49, 2990–2995. [Google Scholar]
- Romagnoli, B.; Ashton, P.R.; Harwood, L.M.; Philp, D.; Price, D.W.; Smith, M.H.; Hayes, W. Synthesis and properties of polyaromatic dendrimers possessing a repetitive amide-ester coupling sequence. Tetrahedron 2003, 59, 3975–3988. [Google Scholar]
- Chen, G.J.; Kumar, J.; Gregory, A.; Stenzel, M.H. Efficient synthesis of dendrimers via a thiol-yne and esterification process and their potential application in the delivery of platinum anti-cancer drugs. Chem. Commun. 2009, 6291–6293. [Google Scholar]
- Maraval, V.; Caminade, A.M.; Majoral, J.P.; Blais, J.C. Dendrimer design: How to circumvent the dilemma of a reduction of steps or an increase of function multiplicity? Angew. Chem. Int. Ed. 2003, 42, 1822–1826. [Google Scholar]
- Servin, P.; Rebout, C.; Laurent, R.; Peruzzini, M.; Caminade, A.M.; Majoral, J.P. Reduced number of steps for the synthesis of dense and highly functionalized dendrimers. Tetrahedron Lett. 2007, 48, 579–583. [Google Scholar]
- Maraval, V.; Laurent, R.; Marchand, P.; Caminade, A.M.; Majoral, J.P. Accelerated methods of synthesis of phosphorus-containing dendrimers. J. Organomet. Chem. 2005, 690, 2458–2471. [Google Scholar] [CrossRef]
- De Gennes, P.G.; Hervet, H. Statistics of starburst polymers. J. Phys. Lett. 1983, 44, L351–L360. [Google Scholar] [CrossRef]
- Hecht, S.; Fréchet, J.M.J. Dendritic encapsulation of function: Applying nature’s site isolation principle from biomimetics to materials science. Angew. Chem. Int. Ed. 2001, 40, 74–91. [Google Scholar] [CrossRef]
- Bosman, A.W.; Janssen, H.M.; Meijer, E.W. About dendrimers: Structure, physical properties, and applications. Chem. Rev. 1999, 99, 1665–1688. [Google Scholar] [CrossRef]
- Moreno, K.X.; Simanek, E.E. Conformational analysis of triazine dendrimers: Using NMR spectroscopy to probe the choreography of a dendrimer’s dance. Macromolecules 2008, 41, 4108–4114. [Google Scholar] [CrossRef]
- Tanis, I.; Karatasos, K. Local dynamics and hydrogen bonding in hyperbranched aliphatic polyesters. Macromolecules 2009, 42, 9581–9591. [Google Scholar] [CrossRef]
- Tanis, I.; Karatasos, K.; Assimopoulou, A.N.; Papageorgiou, V.P. Modeling of hyperbranched polyesters as hosts for the multifunctional bioactive agent shikonin. Phys. Chem. Chem. Phys. 2011, 13, 10808–10817. [Google Scholar]
- Chang, H.T.; Fréchet, J.M.J. Proton-transfer polymerization: A new approach to hyperbranched polymers. J. Am. Chem. Soc. 1999, 121, 2313–2314. [Google Scholar]
- Fréchet, J.M.J.; Henmi, M.; Gitsov, I.; Aoshima, S.; Leduc, M.R.; Grubbs, R.B. Self-condensing vinyl polymerization: An approach to dendritic materials. Science 1995, 269, 1080–1083. [Google Scholar]
- Suzuki, M.; Ii, A.; Saegusa, T. Multibranching polymerization: Palladium-catalyzed ring-opening polymerization of cyclic carbamate to produce hyperbranched dendritic polyamine. Macromolecules 1992, 25, 7071–7072. [Google Scholar] [CrossRef]
- Flory, P.J. Kinetics of condensation polymerization: The reaction of ethylene glycol with succinic acid. J. Am. Chem. Soc. 1937, 59, 466–470. [Google Scholar] [CrossRef]
- Flory, P.J. Molecular size distribution in three dimensional polymers. 6 branched polymers containing A-R-BF-1 type units. J. Am. Chem. Soc. 1952, 74, 2718–2723. [Google Scholar] [CrossRef]
- Mathias, L.J.; Carothers, T.W. Hyperbranched poly(siloxysilanes). J. Am. Chem. Soc. 1991, 113, 4043–4044. [Google Scholar] [CrossRef]
- Hunter, W.H.; Woollett, G.H. A catalytic decomposition of certain phenol silver salts. IV. The constitution of the amorphous oxides. J. Am. Chem. Soc. 1921, 43, 135–151. [Google Scholar] [CrossRef]
- Yoon, K.; Son, D.Y. Syntheses of hyperbranched poly(carbosilarylenes). Macromolecules 1999, 32, 5210–5216. [Google Scholar] [CrossRef]
- Miravet, J.F.; Fréchet, J.M.J. New hyperbranched poly(siloxysilanes): Variation of the branching pattern and end-functionalization. Macromolecules 1998, 31, 3461–3468. [Google Scholar] [CrossRef]
- Ihre, H.; Hult, A.; Söderlind, E. Synthesis, characterization, and H-1 NMR self-diffusion studies of dendritic aliphatic polyesters based on 2,2-bis(hydroxymethyl)propionic acid and 1,1,1-tris(hydroxyphenyl)ethane. J. Am. Chem. Soc. 1996, 118, 6388–6395. [Google Scholar] [CrossRef]
- Ihre, H.; Padilla De Jesús, O.L.; Fréchet, J.M.J. Fast and convenient divergent synthesis of aliphatic ester dendrimers by anhydride coupling. J. Am. Chem. Soc. 2001, 123, 5908–5917. [Google Scholar]
- Malkoch, M.; Malmström, E.; Hult, A. Rapid and efficient synthesis of aliphatic ester dendrons and dendrimers. Macromolecules 2002, 35, 8307–8314. [Google Scholar] [CrossRef]
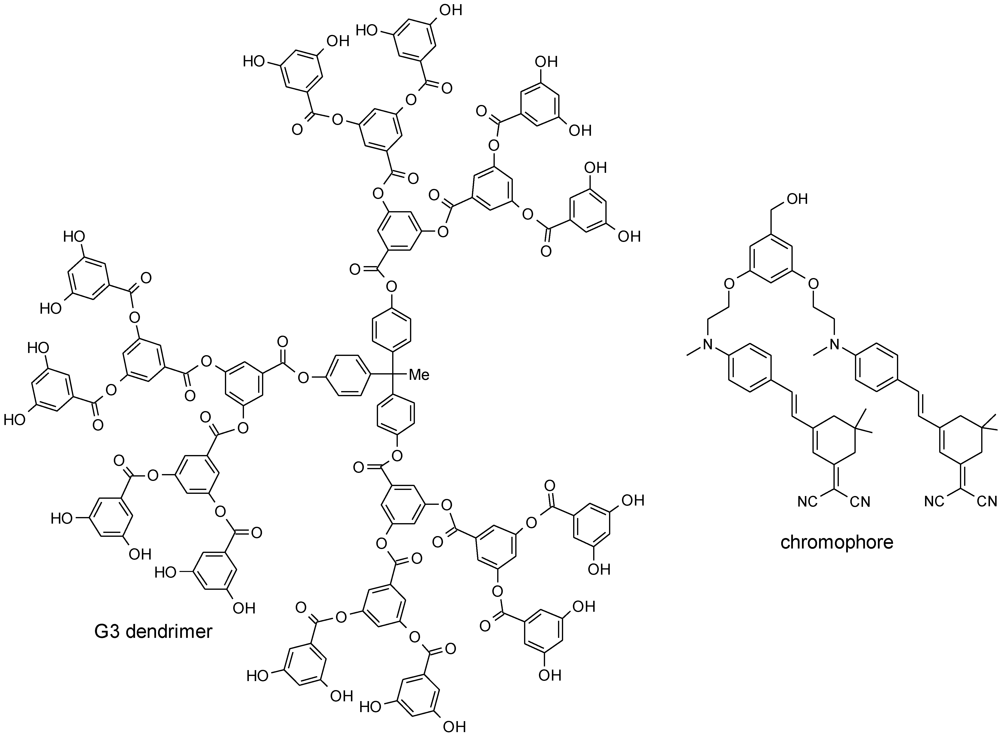
- Parrott, M.C.; Benhabbour, S.R.; Saab, C.; Lemon, J.A.; Parker, S.; Valliant, J.F.; Adronov, A. Synthesis, radiolabeling, and bio-imaging of high-generation polyester dendrimers. J. Am. Chem. Soc. 2009, 131, 2906–2916. [Google Scholar]
- Twibanire, J.K.; Al-Mughaid, H.; Grindley, T.B. Synthesis of new cores and their use in the preparation of polyester dendrimers. Tetrahedron 2010, 66, 9602–9609. [Google Scholar]
- Knorr, R.; Trzeciak, A.; Bannwarth, W.; Gillessen, D. New coupling reagents in peptide chemistry. Tetrahedron Lett. 1989, 30, 1927–1930. [Google Scholar]
- Carpino, L.A. 1-Hydroxy-7-azabenzotriazole—An efficient peptide coupling additive. J. Am. Chem. Soc. 1993, 115, 4397–4398. [Google Scholar] [CrossRef]
- El-Faham, A.; Subirós-Funosas, R.; Prohens, R.; Albericio, F. Comu: A safer and more effective replacement for benzotriazole-based uronium coupling reagents. Chem. Eur. J. 2009, 15, 9404–9416. [Google Scholar]
- Twibanire, J.K.; Grindley, T.B. Efficient and controllably selective preparation of esters using uronium-based coupling agents. Org. Lett. 2011, 13, 2988–2991. [Google Scholar] [CrossRef]
- Bouillon, C.; Quelever, G.; Peng, L. Efficient synthesis of esters containing tertiary amine functionalities via active cyanomethyl ester intermediates. Tetrahedron Lett. 2009, 50, 4346–4349. [Google Scholar] [CrossRef]
- Bouillon, C.; Tintaru, A.; Monnier, V.; Charles, L.; Quelever, G.; Peng, L. Synthesis of poly(amino)ester dendrimers via active cyanomethyl ester intermediates. J. Org. Chem. 2010, 75, 8685–8688. [Google Scholar]
- Robertson, S.A.; Ellman, J.A.; Schultz, P.G. A general and efficient route for chemical aminoacylation of transfer-RNAs. J. Am. Chem. Soc. 1991, 113, 2722–2729. [Google Scholar] [CrossRef]
- Ihre, H.R.; Padilla De Jesús, O.L.; Szoka, F.C.; Fréchet, J.M.J. Polyester dendritic systems for drug delivery applications: Design, synthesis, and characterization. Bioconjugate Chem. 2002, 13, 443–452. [Google Scholar] [CrossRef]
- Greenwald, R.B.; Conover, C.D.; Choe, Y.H. Poly(ethylene glycol) conjugated drugs and prodrugs: A comprehensive review. Crit. Rev. Ther. Drug Carr. Syst. 2000, 17, 101–161. [Google Scholar]
- Kapitza, H.; Zentel, R.; Twieg, R.J.; Nguyen, C.; Vallerien, S.U.; Kremer, F.; Willson, C.G. Ferroelectric liquid-crystalline polysiloxanes with high spontaneous polarization and possible applications in nonlinear optics. Adv. Mater. 1990, 2, 539–543. [Google Scholar]
- Keller, P.; Shao, R.F.; Walba, D.M.; Brunet, M. The first high polarization ferroelectric main-chain liquid-crystalline polymers. Liq. Cryst. 1995, 18, 915–918. [Google Scholar] [CrossRef]
- Walba, D.M.; Keller, P.; Shao, R.F.; Clark, N.A.; Hillmyer, M.; Grubbs, R.H. Main-chain ferroelectric liquid crystal oligomers by acyclic diene metathesis polymerization. J. Am. Chem. Soc. 1996, 118, 2740–2741. [Google Scholar]
- Hermann, D.S.; Hult, A.; Komitov, L.; Lagerwall, S.T.; Lindgren, M. Pyroelectric polymers for nonlinear optics. Ferroelectrics 1998, 213, 405–415. [Google Scholar]
- Hermann, D.S.; Hult, A.; Komitov, L.; Lagerwall, S.T.; Sahlen, F.; Trollsås, M. The influence of chiral strength on the spontaneous polarization and the second-order nonlinear optical susceptibility in ferroelectric liquid crystals. Ferroelectrics 1998, 213, 417–427. [Google Scholar]
- Busson, P.; Ihre, H.; Hult, A. Synthesis of a novel dendritic liquid crystalline polymer showing a ferroelectric SmC* phase. J. Am. Chem. Soc. 1998, 120, 9070–9071. [Google Scholar] [CrossRef]
- Hedrick, J.L.; Trollsås, M.; Hawker, C.J.; Atthoff, B.; Claesson, H.; Heise, A.; Miller, R.D.; Mecerreyes, D.; Jérôme, R.; Dubois, P. Dendrimer-like star block and amphiphilic copolymers by combination of ring opening and atom transfer radical polymerization. Macromolecules 1998, 31, 8691–8705. [Google Scholar]
- Kato, M.; Kamigaito, M.; Sawamoto, M.; Higashimura, T. Polymerization of methyl-methacrylate with the carbon-tetrachloride dichlorotris(triphenylphosphine) ruthenium(II) methylaluminum bis(2,6-di-tert-butylphenoxide) initiating system—Possibility of living radical polymerization. Macromolecules 1995, 28, 1721–1723. [Google Scholar] [CrossRef]
- Wang, J.S.; Matyjaszewski, K. Controlled living radical polymerization—Atom-transfer radical polymerization in the presence of transition-metal complexes. J. Am. Chem. Soc. 1995, 117, 5614–5615. [Google Scholar] [CrossRef]
- Annby, U.; Malmberg, M.; Pettersson, B.; Rehnberg, N. Benzylidene protected bis-MPA—A convenient dendrimer building block. Tetrahedron Lett. 1998, 39, 3217–3220. [Google Scholar]
- Trollsås, M.; Atthoff, B.; Claesson, H.; Hedrick, J.L. Hyperbranched poly(epsilon-caprolactone)s. Macromolecules 1998, 31, 3439–3445. [Google Scholar]
- Hao, X.J.; Nilsson, C.; Jesberger, M.; Stenzel, M.H.; Malmström, E.; Davis, T.P.; Östmark, E.; Barner-Kowollik, C. Dendrimers as scaffolds for multifunctional reversible addition-fragmentation chain transfer agents: Syntheses and polymerization. J. Polym. Sci. Part A 2004, 42, 5877–5890. [Google Scholar]
- Wang, L.L.; Meng, Z.L.; Yu, Y.L.; Meng, Q.W.; Chen, D.Z. Synthesis of hybrid linear-dendritic block copolymers with carboxylic functional groups for the biomimetic mineralization of calcium carbonate. Polymer 2008, 49, 1199–1210. [Google Scholar] [CrossRef]
- Parrott, M.C.; Marchington, E.B.; Valliant, J.F.; Adronov, A. Synthesis and properties of carborane-functionalized aliphatic polyester dendrimers. J. Am. Chem. Soc. 2005, 127, 12081–12089. [Google Scholar]
- Gillies, E.R.; Fréchet, J.M.J. Designing macromolecules for therapeutic applications: Polyester dendrimer-poly(ethylene oxide) “bow-tie” hybrids with tunable molecular weight and architecture. J. Am. Chem. Soc. 2002, 124, 14137–14146. [Google Scholar] [CrossRef]
- Vestberg, R.; Nystrom, A.; Lindgren, M.; Malmström, E.; Hult, A. Porphyrin-cored 2,2-bis(methylol)propionic acid dendrimers. Chem. Mater. 2004, 16, 2794–2804. [Google Scholar] [CrossRef]
- Nishiyama, N.; Stapert, H.R.; Zhang, G.D.; Takasu, D.; Jiang, D.L.; Nagano, T.; Aida, T.; Kataoka, K. Light-harvesting ionic dendrimer porphyrins as new photosensitizers for photodynamic therapy. Bioconjugate Chem. 2003, 14, 58–66. [Google Scholar] [CrossRef]
- Armstrong, N.R. Phthalocyanines and porphyrins as materials. J. Porphyrins Phthalocyanines 2000, 4, 414–417. [Google Scholar] [CrossRef]
- Krivokapic, A.; Anderson, H.L.; Bourhill, G.; Ives, R.; Clark, S.; McEwan, K.J. Meso-tetra-alkynyl porphyrins for optical limiting—A survey of group III and IV metal complexes. Adv. Mater. 2001, 13, 652–656. [Google Scholar] [CrossRef]
- Finikova, O.; Galkin, A.; Rozhkov, V.; Cordero, M.; Hagerhall, C.; Vinogradov, S. Porphyrin and tetrabenzoporphyrin dendrimers: Tunable membrane-impermeable fluorescent pH nanosensors. J. Am. Chem. Soc. 2003, 125, 4882–4893. [Google Scholar]
- Pollak, K.W.; Sanford, E.M.; Fréchet, J.M.J. Comparison of two convergent routes for the preparation of metalloporphyrin-core dendrimers: Direct condensation vs. chemical modification. J. Mater. Chem. 1998, 8, 519–527. [Google Scholar] [CrossRef]
- Pollak, K.W.; Leon, J.W.; Fréchet, J.M.J.; Maskus, M.; Abruna, H.D. Effects of dendrimer generation on site isolation of core moieties: Electrochemical and fluorescence quenching studies with metalloporphyrin core dendrimers. Chem. Mater. 1998, 10, 30–38. [Google Scholar] [CrossRef]
- Maes, W.; Amabilino, D.B.; Dehaen, W. Synthesis of novel dendrimers containing pyrimidine units. Tetrahedron 2003, 59, 3937–3943. [Google Scholar] [CrossRef]
- Capitosti, G.J.; Guerrero, C.D.; Binkley, D.E.; Rajesh, C.S.; Modarelli, D.A. Efficient synthesis of porphyrin-containing, benzoquinone-terminated, rigid polyphenylene dendrimers. J. Org. Chem. 2003, 68, 247–261. [Google Scholar]
- Yeow, E.K.L.; Ghiggino, K.P.; Reek, J.N.H.; Crossley, M.J.; Bosman, A.W.; Schenning, A.; Meijer, E.W. The dynamics of electronic energy transfer in novel multiporphyrin functionalized dendrimers: A time-resolved fluorescence anisotropy. J. Phys. Chem. B 2000, 104, 2596–2606. [Google Scholar]
- Rajesh, C.S.; Capitosti, G.J.; Cramer, S.J.; Modarelli, D.A. Photoinduced electron-transfer within free base and zinc porphyrin containing poly(amide) dendrimers. J. Phys. Chem. B 2001, 105, 10175–10188. [Google Scholar]
- Jiang, D.L.; Aida, T. Morphology-dependent photochemical events in aryl ether dendrimer porphyrins: Cooperation of dendron subunits for singlet energy transduction. J. Am. Chem. Soc. 1998, 120, 10895–10901. [Google Scholar] [CrossRef]
- Matos, M.S.; Hofkens, J.; Verheijen, W.; De Schryver, F.C.; Hecht, S.; Pollak, K.W.; Fréchet, J.M.J.; Forier, B.; Dehaen, W. Effect of core structure on photophysical and hydrodynamic properties of porphyrin dendrimers. Macromolecules 2000, 33, 2967–2973. [Google Scholar]
- Kimura, M.; Shiba, T.; Yamazaki, M.; Hanabusa, K.; Shirai, H.; Kobayashi, N. Construction of regulated nanospace around a porphyrin core. J. Am. Chem. Soc. 2001, 123, 5636–5642. [Google Scholar]
- Galie, K.M.; Mollard, A.; Zharov, I. Polyester-based carborane-containing dendrons. Inorg. Chem. 2006, 45, 7815–7820. [Google Scholar]
- Baars, M.; Meijer, E.W. Host-guest chemistry of dendritic molecules. Dendrimers Ii 2000, 210, 131–182. [Google Scholar] [CrossRef]
- Baars, M.; Karlsson, A.J.; Sorokin, V.; de Waal, B.F.W.; Meijer, E.W. Supramolecular modification of the periphery of dendrimers resulting in rigidity and functionality. Angew. Chem. Int. Ed. 2000, 39, 4262–4265. [Google Scholar]
- Gillies, E.R.; Fréchet, J.M.J. Synthesis and self-assembly of supramolecular dendritic “Bow-Ties”: Effect of peripheral functionality on association constants. J. Org. Chem. 2004, 69, 46–53. [Google Scholar] [CrossRef]
- Bell, J.A.; Kenworthy, C. Cyanoethylation of some alkanolamines. Synthesis 1971, 650–652. [Google Scholar]
- Goodwin, A.P.; Lam, S.S.; Fréchet, J.M.J. Rapid, efficient synthesis of heterobifunctional biodegradable dendrimers. J. Am. Chem. Soc. 2007, 129, 6994–6995. [Google Scholar]
- Burgel, T.; Fedtke, M.; Franzke, M. Reaction of cyclic carbonates with amines—Linear telechelic oligomers. Polym. Bull. 1993, 30, 155–162. [Google Scholar] [CrossRef]
- Ochiai, B.; Satoh, Y.; Endo, T. Nucleophilic polyaddition in water based on chemo-selective reaction of cyclic carbonate with amine. Green Chem. 2005, 7, 765–767. [Google Scholar] [CrossRef]
- Gurr, P.A.; Qiao, G.G.; Solomon, D.H.; Harton, S.E.; Spontak, R.J. Synthesis, characterization, and direct observation of star microgels. Macromolecules 2003, 36, 5650–5654. [Google Scholar]
- Bosman, A.W.; Vestberg, R.; Heumann, A.; Fréchet, J.M.J.; Hawker, C.J. A modular approach toward functionalized three-dimensional macromolecules: From synthetic concepts to practical applications. J. Am. Chem. Soc. 2003, 125, 715–728. [Google Scholar]
- Lord, H.T.; Quinn, J.F.; Angus, S.D.; Whittaker, M.R.; Stenzel, M.H.; Davis, T.P. Microgel stars via reversible addition fragmentation chain transfer (RAFT) polymerisation—A facile route to macroporous membranes, honeycomb patterned thin films and inverse opal substrates. J. Mater. Chem. 2003, 13, 2819–2824. [Google Scholar] [CrossRef]
- Wiltshire, J.T.; Qiao, G.G. Synthesis of core cross-linked star polymers with adjustable coronal properties. Macromolecules 2008, 41, 623–631. [Google Scholar] [CrossRef]
- Wiltshire, J.T.; Qiao, G.G. Selectively degradable core cross-linked star polymers. Macromolecules 2006, 39, 9018–9027. [Google Scholar] [CrossRef]
- Hawker, C.J.; Wooley, K.L. The convergence of synthetic organic and polymer chemistries. Science 2005, 309, 1200–1205. [Google Scholar]
- Abrol, S.; Kambouris, P.A.; Looney, M.G.; Solomon, D.H. Studies on microgels. 3. Synthesis using living free radical polymerization. Macromol. Rapid Commun. 1997, 18, 755–760. [Google Scholar] [CrossRef]
- Baek, K.Y.; Kamigaito, M.; Sawamoto, M. Star-shaped polymers by metal-catalyzed living radical polymerization. 1. Design of Ru(II)-based systems and divinyl linking agents. Macromolecules 2001, 34, 215–221. [Google Scholar] [CrossRef]
- Connal, L.A.; Vestberg, R.; Hawker, C.J.; Qiao, G.G. Synthesis of dendron functionalized core cross-linked star polymers. Macromolecules 2007, 40, 7855–7863. [Google Scholar]
- Kose, M.M.; Yesilbag, G.; Sanyal, A. Segment block dendrimers via Diels-Alder cycloaddition. Org. Lett. 2008, 10, 2353–2356. [Google Scholar] [CrossRef]
- McElhanon, J.R.; Wheeler, D.R. Thermally responsive dendrons and dendrimers based on reversible furan-maleimide Diels-Alder adducts. Org. Lett. 2001, 3, 2681–2683. [Google Scholar] [CrossRef]
- Szalai, M.L.; McGrath, D.V.; Wheeler, D.R.; Zifer, T.; McElhanon, J.R. Dendrimers based on thermally reversible furan-maleimide Diels-Alder adducts. Macromolecules 2007, 40, 818–823. [Google Scholar]
- Kawa, M.; Fréchet, J.M.J. Enhanced luminescence of lanthanide within lanthanide-cored dendrimer complexes. Thin Solid Films 1998, 331, 259–263. [Google Scholar] [CrossRef]
- Kawa, M.; Fréchet, J.M.J. Self-assembled lanthanide-cored dendrimer complexes: Enhancement of the luminescence properties of lanthanide ions through site-isolation and antenna effects. Chem. Mater. 1998, 10, 286–296. [Google Scholar] [CrossRef]
- Antoni, P.; Malkoch, M.; Vamvounis, G.; Nyström, D.; Nyström, A.; Lindgren, M.; Hult, A. Europium confined cyclen dendrimers with photophysically active triazoles. J. Mater. Chem. 2008, 18, 2545–2554. [Google Scholar] [CrossRef]
- Tornoe, C.W.; Christensen, C.; Meldal, M. Peptidotriazoles on solid phase: [1,2,3]-triazoles by regiospecific copper(I)-catalyzed 1,3-dipolar cycloadditions of terminal alkynes to azides. J. Org. Chem. 2002, 67, 3057–3064. [Google Scholar]
- Kolb, H.C.; Finn, M.G.; Sharpless, K.B. Click chemistry: Diverse chemical function from a few good reactions. Angew. Chem. Int. Ed. 2001, 40, 2004–2021. [Google Scholar]
- Mekelburger, H.B.; Rissanen, K.; Vögtle, F. Repetitive synthesis of bulky dendrimers—A reversibly photoactive dendrimer with six azobenzene side-chains. Chem. Berichte 1993, 126, 1161–1169. [Google Scholar] [CrossRef]
- Momotake, A.; Arai, T. Photochemistry and photophysics of stilbene dendrimers and related compounds. J. Photochem. Photobiol. C 2004, 5, 1–25. [Google Scholar] [CrossRef]
- Liao, L.X.; Stellacci, F.; McGrath, D.V. Photoswitchable flexible and shape-persistent dendrimers: Comparison of the interplay between a photochromic azobenzene core and dendrimer structure. J. Am. Chem. Soc. 2004, 126, 2181–2185. [Google Scholar]
- Puntoriero, F.; Ceroni, P.; Balzani, V.; Bergamini, G.; Vögtle, F. Photoswitchable dendritic hosts: A dendrimer with peripheral azobenzene groups. J. Am. Chem. Soc. 2007, 129, 10714–10719. [Google Scholar]
- Gabriel, C.J.; Parquette, J.R. Expanding dendrons. The photoisomerism of folded azobenzene dendrons. J. Am. Chem. Soc. 2006, 128, 13708–13709. [Google Scholar] [CrossRef]
- Kumar, G.S.; Neckers, D.C. Photochemistry of azobenzene-containing polymers. Chem. Rev. 1989, 89, 1915–1925. [Google Scholar] [CrossRef]
- Yagai, S.; Karatsu, T.; Kitamura, A. Photocontrollable self-assembly. Chem. Eur. J. 2005, 11, 4054–4063. [Google Scholar]
- Barrett, C.J.; Mamiya, J.I.; Yager, K.G.; Ikeda, T. Photo-mechanical effects in azobenzene-containing soft materials. Soft Matter 2007, 3, 1249–1261. [Google Scholar] [CrossRef]
- Yager, K.G.; Barrett, C.J. Photomechanical surface patterning in azo-polymer materials. Macromolecules 2006, 39, 9320–9326. [Google Scholar] [CrossRef]
- Tuuttila, T.; Lipsonen, J.; Lahtinen, M.; Huuskonen, J.; Rissanen, K. Synthesis and characterization of chiral azobenzene dye functionalized Janus dendrimers. Tetrahedron 2008, 64, 10590–10597. [Google Scholar] [CrossRef]
- Tuuttila, T.; Lipsonen, J.; Huuskonen, J.; Rissanen, K. Synthesis and characterization of polyene chromophores with hydroxyl functionalization. Dyes Pigment. 2008, 77, 357–362. [Google Scholar] [CrossRef]
- Yokoyama, S.; Nakahama, T.; Otomo, A.; Mashiko, S. Intermolecular coupling enhancement of the molecular hyperpolarizability in multichromophoric dipolar dendrons. J. Am. Chem. Soc. 2000, 122, 3174–3181. [Google Scholar] [CrossRef]
- Abram, U.; Alberto, R. Technetium and rhenium—Coordination chemistry and nuclear medical applications. J. Braz. Chem. Soc. 2006, 17, 1486–1500. [Google Scholar] [CrossRef]
- Kim, S.K.; Lee, S.B. Highly encoded one-dimensional nanostructures for rapid sensing. J. Mater. Chem. 2009, 19, 1381–1389. [Google Scholar] [CrossRef]
- Finkel, N.H.; Lou, X.H.; Wang, C.Y.; He, L. Barcoding the microworld. Anal. Chem. 2004, 76, 353A–359A. [Google Scholar]
- White, K.A.; Chengelis, D.A.; Gogick, K.A.; Stehman, J.; Rosi, N.L.; Petoud, S. Near-infrared luminescent lanthanide MOF barcodes. J. Am. Chem. Soc. 2009, 131, 18069–18071. [Google Scholar]
- Yeniad, B.; Naik, H.; Amir, R.J.; Koning, C.E.; Hawker, C.J.; Heise, A. Encoded dendrimers with defined chiral composition via ‘click’ reaction of enantiopure building blocks. Chem. Commun. 2011, 47, 9870–9872. [Google Scholar]
- Duxbury, C.J.; Hilker, I.; de Wildeman, S.M.A.; Heise, A. Enzyme-responsive materials: Chirality to program polymer reactivity. Angew. Chem. Int. Ed. 2007, 46, 8452–8454. [Google Scholar]
- Vestberg, R.; Malkoch, M.; Kade, M.; Wu, P.; Fokin, V.V.; Sharpless, K.B.; Drockenmuller, E.; Hawker, C.J. Role of architecture and molecular weight in the formation of tailor-made ultrathin multilayers using dendritic macromolecules and click chemistry. J. Polym. Sci. Part A 2007, 45, 2835–2846. [Google Scholar]
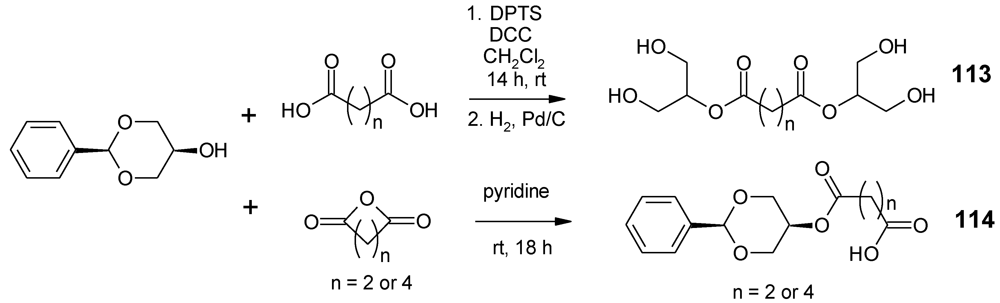
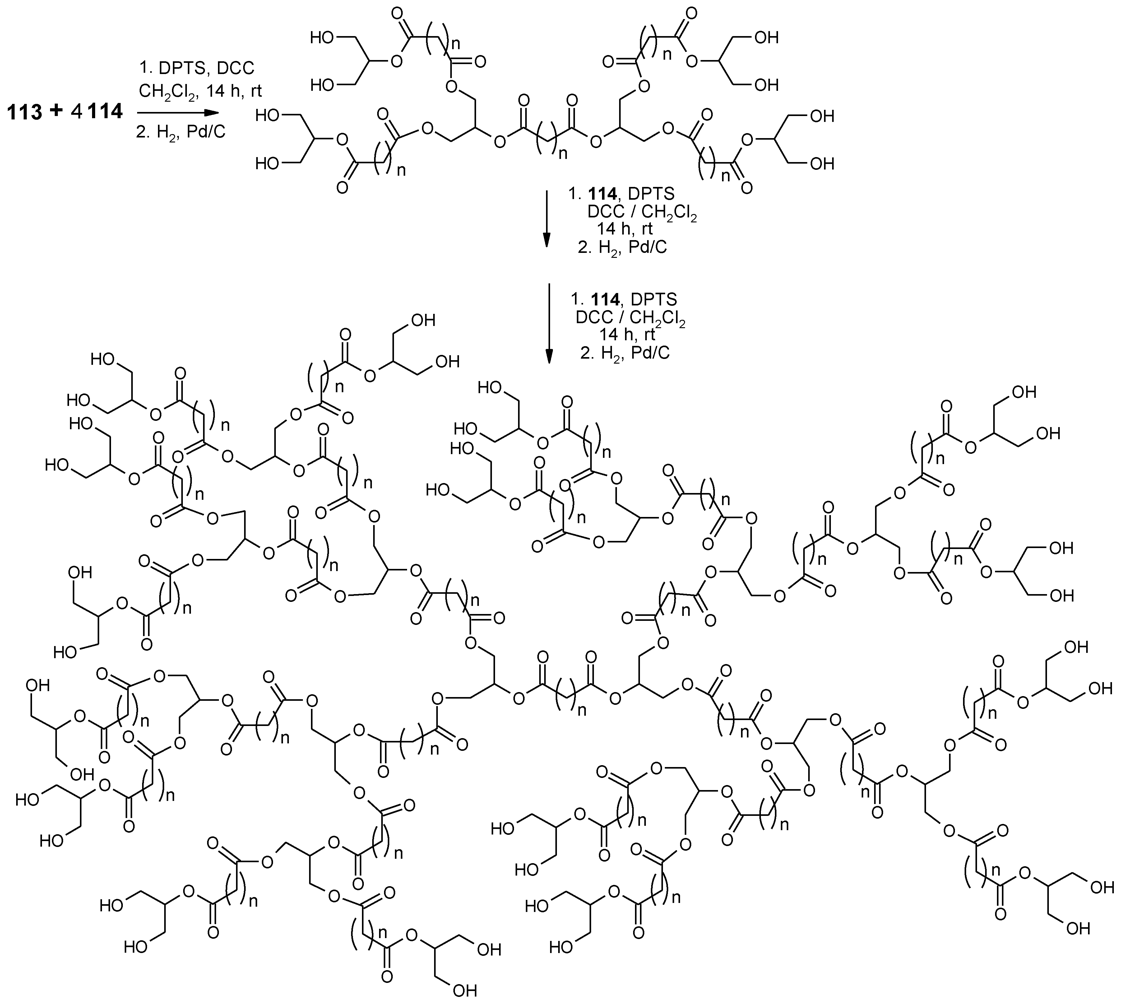
- Carnahan, M.A.; Grinstaff, M.W. Synthesis and characterization of poly(glycerol-succinic acid) dendrimers. Macromolecules 2001, 34, 7648–7655. [Google Scholar]
- Carnahan, M.A.; Grinstaff, M.W. Synthesis of generational polyester dendrimers derived from glycerol and succinic or adipic acid. Macromolecules 2006, 39, 609–616. [Google Scholar] [CrossRef]
- Oelker, A.M.; Grinstaff, M.W. Ophthalmic adhesives: A materials chemistry perspective. J. Mater. Chem. 2008, 18, 2521–2536. [Google Scholar] [CrossRef]
- Oelker, A.M.; Berlin, J.A.; Wathier, M.; Grinstaff, M.W. Synthesis and characterization of dendron cross-linked PEG hydrogels as corneal adhesives. Biomacromolecules 2011, 12, 1658–1665. [Google Scholar] [CrossRef]
- Söntjens, S.H.M.; Nettles, D.L.; Carnahan, M.A.; Setton, L.A.; Grinstaff, M.W. Biodendrimer-based hydrogel scaffolds for cartilage tissue repair. Biomacromolecules 2006, 7, 310–316. [Google Scholar] [CrossRef]
- Berdahl, J.P.; Johnson, C.S.; Proia, A.D.; Grinstaff, M.W.; Kim, T. Comparison of sutures and dendritic polymer adhesives for corneal laceration repair in an in vivo chicken model. Arch. Ophthalmol. 2009, 127, 442–447. [Google Scholar] [CrossRef]
- Twibanire, J.K.; Al-Mughaid, H.; MacIsaac, E.; Huesis, M.; Grindley, T.B. Unpublished results.
- Hirayama, Y.; Sakamoto, Y.; Yamaguchi, K.; Sakamoto, S.; Iwamura, M. Synthesis of polyester dendrimers and dendrons starting from Michael reaction of acrylates with 3-hydroxyacetophenone. Tetrahedron Lett. 2005, 46, 1027–1030. [Google Scholar] [CrossRef]
- Hirayama, Y.; Nakamura, T.; Uehara, S.; Sakamoto, Y.; Yamaguchi, K.; Sei, Y.; Iwamura, M. Synthesis and characterization of polyester dendrimers from acetoacetate and acrylate. Org. Lett. 2005, 7, 525–528. [Google Scholar] [CrossRef]
- Bo, Z.S.; Zhang, X.; Zhang, C.M.; Wang, Z.Q.; Yang, M.L.; Shen, J.C.; Ji, Y.P. Rapid synthesis of polyester dendrimers. J. Chem. Soc. Perkin Trans. 1 1997, 2931–2935. [Google Scholar]
- Bo, Z.S.; Zhang, W.K.; Zhang, X.; Zhang, C.M.; Shen, J.C. Synthesis and properties of polyester dendrimers bearing carbazole groups in their periphery. Macromol. Chem. Phys. 1998, 199, 1323–1327. [Google Scholar] [CrossRef]
- Do, J.Y.; Park, S.K.; Ju, J.J.; Park, S.; Lee, M.H. Electro-optic materials: Hyperbranched chromophores attached linear polyimide and dendritic polyesters. Polym. Adv. Technol. 2005, 16, 221–226. [Google Scholar] [CrossRef]
- Do, J.Y.; Jung, J.J. Polyester dendrimers carrying NLO chromophores: Synthesis and optical characterization. Macromol. Chem. Phys. 2005, 206, 1326–1331. [Google Scholar] [CrossRef]
- Romagnoli, B.; van Baal, I.; Price, D.W.; Harwood, L.M.; Hayes, W. Chiral poly(aromatic amide ester) dendrimers bearing an amino acid derived C-3-symmetric core—Synthesis and properties. Eur. J. Org. Chem. 2004, 4148–4157. [Google Scholar]
- Campbell, A.D.; Raynham, T.M.; Taylor, R.J.K. A simplified route to the (R)-Garner aldehyde and (S)-vinyl glycinol. Synthesis 1998, 1707–1709. [Google Scholar]
- Rosen, B.M.; Lligadas, G.; Hahn, C.; Percec, V. Synthesis of dendritic macromolecules through divergent iterative thio-bromo “click” chemistry and SET-LRP. J. Polym. Sci. Part A 2009, 47, 3940–3948. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Twibanire, J.K.; Grindley, T.B. Polyester Dendrimers. Polymers 2012, 4, 794-879. https://doi.org/10.3390/polym4010794
Twibanire JK, Grindley TB. Polyester Dendrimers. Polymers. 2012; 4(1):794-879. https://doi.org/10.3390/polym4010794
Chicago/Turabian StyleTwibanire, Jean–d’Amour K., and T. Bruce Grindley. 2012. "Polyester Dendrimers" Polymers 4, no. 1: 794-879. https://doi.org/10.3390/polym4010794