Gold-Poly(methyl methacrylate) Nanocomposite Films for Plasmonic Biosensing Applications
Abstract
: Gold-poly(methyl methacrylate) nanocomposites are prepared by an in situ method, by irradiating spin-coated films containing the polymer and the gold precursor dissolved in acetone. The reduction of gold ions results in the formation of Au that nucleates and grows within the polymer film. It is shown that, depending on the energy source, gold nanoparticles with different shapes can be formed. Nanocomposites prepared through UV-, thermal-, and MW-irradiation, respectively, show a low sensitivity toward the environment. However, by annealing the samples at temperatures well above the glass transition temperature of the polymer, the response to dielectric environment appears to be enhanced significantly. The sensitivity of samples synthesized through the three different methods is found to be comparable, around 100 nm/RIU. The increased sensitivity of the annealed sample is accounted for by the increased mobility of both polymer chains and gold nanoparticles in the rubbery state of the material and the presence of the monomer. Gold nanoparticles “freed” from the strong interaction with the polymer are now able to feel the molecules from the surrounding environment. The results show that, by using adequate post-synthesis heat treatments, gold-polymer nanocomposites can be used as plasmonic sensing platforms.1. Introduction
Metal-polymer nanocomposite (or nanometal-polymer composite) films are hybrid materials with inorganic nanoparticles immobilized and integrated into a polymer matrix. Upon the combination of the properties of the two fundamentally different materials, novel functions can be generated, leading to a wide range of interesting applications. The polymer host provides processability and, in many cases, transparency, and, by immobilizing the metal nanoparticles, protects them against aggregation or segregation. On the other hand, metal nanoparticles such as Au or Ag, significantly contribute with their distinctive optical properties. In order to emphasize their major contribution to the overall optical properties of the hybrid material, they are generally called “optically effective additives” [1]. Nanocomposites may show optical nonlinearities and/or ultralow or ultrahigh refractive indices and are suitable for applications such as color filters, optical sensors, data storages, waveguides, etc.
There are two general approaches to the preparation of nanocomposite materials. Either, a suitable metal precursor is dissolved in the polymer solution and the ions are subsequently reduced to nanoparticles chemically, sono-, or photochemically (by using X or γ-rays, UV or microwaves) or, the corresponding monomer is polymerized around the metal nanoparticles.
Poly(methyl methacrylate) (PMMA) is a commercially available polymer having a high transparency and a relatively low glass-transition temperature, suitable for lower post synthesis temperature treatments [2]. Different methods have already been used to prepare Au (Ag)-PMMA nanocomposites. Among them, the reduction of Au ions by sodium borohydride, leading to non-uniform spherical nanoparticles, the ultrasonic reduction within an emulsion containing the monomer that results in silver nanoparticles of about 20 nm [3] or the reduction of metal ions by the solvent [4]. The UV photochemical reduction of Au3+ followed by annealing yielded Au nanoparticles that were shown to grow during the annealing process [5,6]. The optical properties of Au-PMMA nanocomposites depend significantly on the size of Au nanoparticles which in turn, depends on the fraction of gold precursor in the reaction mixture and the irradiation time [7]. UV irradiation of the PMMA film containing uniformly distributed AuCl4− ions has also been used for photopatterning [8]. Microwave (MW)-assisted coating of PMMA beads by silver nanoparticles in solution was found to be a new and efficient technique [9]. It has been shown that the main advantages of MW are the uniform heating and, most importantly, the short thermal induction period leading to a homogeneous nucleation [10]. The synthesis of silver nanoparticles based on a polyol process and variable frequency microwave has also been described [11] but microwave-assisted synthesis of Au-PMMA nanocomposites has not been reported so far. However, it has been shown that thin PVA films with in-situ grown silver nanoparticles are good microwave absorbers in the 8–12 GHz range [12].
Because of their strong plasmon band in the visible spectrum, arising from the excitation of plasmons by the incident light, gold nanoparticles are particularly suited for sensing applications. The plasmon band is shifted to longer wavelengths when the dielectric constant (refractive index) in the vicinity of the particle changes and this fact is used for sensing applications. The shift, generally accompanied by a broadening of the band, is dependent both on the size and shape of gold nanoparticles and the mass of the bio-molecules surrounding them. All the nanocomposites containing Au and Ag display the strong Localized Surface Plasmon Resonance (LSPR) band that characterize the metal and, for this reason, UV-Visible spectroscopy became a powerful tool for the study, not only of metal nanostructures, but of nanocomposites as well.
In a previous work, we have prepared Au(Ag)-poly(dimethyl siloxane) (Au(Ag)-PDMS) free standing films and have studied their suitability for bio-sensing applications [13]. More specifically, we attempted to compare the sensing properties of polymer-gold nanocomposites with those of pure gold nanostructures and assess the importance of nanoparticle-polymer interactions in this context. The results of our preliminary study have shown that, generally, the performance of Ag and Au-PDMS nanocomposites for sensing bio-entities was poor.
It is well established that the decay length of the electromagnetic field for LSPR sensors depends on the size, shape, and composition of nanoparticles and it is not more than 5–15 nm [14-17]. The direct implication of this short decay length is the ability to detect very thin layers of adsorbate molecules. However, if the gold nanoparticles are not on the surface but embedded deeply into the polymer, the biomolecules may be very well too far away from the sensing volume of the particle. On the other hand, the same thing may happen if the interaction between the particles and the polymer chains is too strong and the nanoparticles are entangled in the polymer network. The strong interactions between the chain segments of the polymer and nanoparticles contribute to the improvement of mechanical properties but, at the same time, they may decrease the mobility of the particles and interfere with the interaction with the biomolecule. Since the biosensing properties are determined by the spatial distribution of the Au nanoparticles, it is important to be able to control this distribution. We have explored some promising ways to increase the mobility of Au nanoparticles in order to concentrate most of them on the surface of the film and thus, make them accessible to the surrounding environment. Among them, annealing at moderate temperatures seems a promising method to improve considerably the sensitivity of Au-PMMA nanocomposite platforms.
To prepare Au-PMMA for bio-sensing applications, the UV photochemical and MW-assisted reduction, as well as the thermal reduction methods were investigated. In order to have a good sensitivity towards the bio-entities, nanocomposites with a low content of Au were synthesized through the above-mentioned methods. The samples were prepared in acetone, a common solvent for PMMA and the Au precursor. The mixture was spin-coated on glass and irradiated (UV or MW) for performing the reduction to Au nanoparticles. As far as we are aware, microwave-assisted reactions have been carried out and studied only in solutions by using water because of its high dipole moment and high value of dielectric loss [9,10]. This is the first work on MW-assisted synthesis of Au-PMMA through exposing to the microwaves a spin-coated film containing only traces of acetone. The sensitivity of non-spherical gold nanoparticles towards bio-molecules is investigated as well.
In this work, the sensitivity of the Au-PMMA nanocomposites prepared through different ways toward to dielectric environment was studied. We used PMMA-Au composites to evidence the antibody-antigen interactions as well. LSPR in gold nanoparticles is used to assess the characteristics of the interaction and to obtain information on the binding. The present study is focused on development of a method for fast and efficient covalent immobilization of antibodies, based on a carboxymethyl dextran hydrogel matrix. Hydrogels are known to provide increased immobilization capacity and, most importantly, to preserve the structure and properties of biomolecules after immobilization. In addition to the dextran derivative, an alkanethiol self-assembled monolayer (SAM) linker was used to modify the surface of gold nanoparticles.
The bio-sensing protocol used to evaluate the quality of the samples is described in detail in the experimental part.
Many computational tools have been developed to simulate the optical properties of the metallic nanostructures. One of them is the Discrete Dipole Approximation (DDA). DDA [18-20] is a computational tool to simulate the optical response of the metallic nanoparticles of different shape, size, and composition with a complex dielectric function. It can be used for a single nanoparticle or for assemblies of particles that are surrounded by a medium with a complex dielectric function. The exact solution of the Maxwell's equation corresponding to the interaction of the incident electromagnetic radiation with the spherical particle is represented by the Mie theory [21]. It is used to calculate the scattering and absorption cross section for a homogenous spherical particle with an arbitrary size and a complex dielectric function. There is no restriction on the size parameters value which can be chosen. The reason why Mie's theory is still important for so long is that it is the only exact solution to Maxwell's equations that is relevant to spherical particles. Most of the standard methods to prepare metal colloidal solutions yield particles of different shapes but the majority of particles are, generally, spherical. The optical properties of Au-PMMA nanocomposite were simulated here, by using both the Mie theory and the DDA method.
2. Experimental
The preparation of Au-PMMA nanocomposites was carried out by three different methods: (i) an in-situ photo-reduction using UV irradiation; (ii) micro-wave-assisted reduction; and (iii) thermal reduction assisted by the polymer. Au-PMMA nanocomposites were prepared as follows: 1 g of PMMA (average MW = 15,000) was first dissolved in 5 mL of acetone and different amounts of hydrogen tetrachloroaurate trihydrate (HAuCl4·3H2O) were dissolved in the polymer solution to obtain a nanocomposite with 1.0–3.0% (w) Au. This percentage represents the weight of gold with respect to the weight of the composite. Subsequently, the solution was spin-coated on glass substrates using 1,500 rpm for 40 s.
UV Photo-Reduction: The spin-coated samples were irradiated in a Stratagene Strainlinker 2,400 unit equipped with five 25 W UV lamps having the wavelength maximum at 254 nm and providing non-collimated radiation with a power of 7 mW/cm2. The samples are kept at a distance of 8 cm from the lamp for durations ranging between 90 min and 10 h. The irradiated samples were annealed for 30 min at temperatures ranging between 70 and 90 °C.
Micro-Wave-Assisted Reduction: The synthesis is carried out by keeping the spin-coated sample in a commercial MW oven with a power of 1,100 W. Short irradiating times (between 60 s and 3 min) are used. As in the case of UV irradiated samples, the reaction is completed when the color of the sample changed from yellow to red.
Thermal Reduction: Spin-coated samples were kept in the oven for short times and the color of the samples turned to red even in the absence of UV irradiation.
In all the cases, the completion of the reaction is indicated by the change of the color, from yellow, corresponding to the gold anions, to wine red, the color of gold nanoparticles.
UV-Visible spectra were measured using a Perkin-Elmer (Model Lambda 650) spectrophotometer and a scanning Electron Microscope (Model JEOL JSM-7600 TFE) was used for morphological studies. The AFM instrument Model NSCRIPTORTM DPN® System DS006 (NanoINK) was used for some of the measurements. Additional annealing experiments were conducted on both UV and MW- irradiated samples. The samples were kept in an oven at 300 °C for 30 min. The surface of the annealed samples was activated by keeping them for 40 s in a Plasma Cleaner apparatus (Harrick Plasma).
Bio-Sensing Protocol
Prior to biosensing, gold nanoparticles were functionalized by using a Nanothink 18 (1-octadecanethiol) or carboxymethyl dextran solution, by immersing the annealed sample in the solution of the linker for 8–10 h. The polyclonal antibody, Anti-Human Serum produced in rabbit (Sigma), was dissolved in PBS and the functionalized sample was immersed in the antibody solution for several hours. Finally, the sample holding the antibody was kept in the antigen solution for several hours.
Simulation of the Au-PMMA Nanostructures
DDA is an analytical technique for calculating the extinction and absorption efficiencies for nanoparticles. The mathematical formulation of DDA is out of scope of this study and it can found elsewhere. The basic idea of DDA method is to represent the target under investigation by a three-dimensional array of N polarizable points (the points acquire dipole moments in response to the incident electric field) which are situated on a periodic square lattice of side length d which represents the inter-dipole spacing (the lattice spacing). The lattice spacing should be smaller than the incident wavelength and any structural parameters. A large number of the dipoles is required (typically N > 104) to provide an accurate description of the target's geometry. DDA involves solving a linear equation of the form A·P = E, where A is a square matrix of the order 3N, P is the induced polarization due to the interaction with the incident field and E is the sum of the incident electric field and the internal field attributed to the other dipoles in the same particle. Once the polarization is obtained from the solution of the last 3N complex linear equations, the optical properties can be calculated. The outputs of the DDA are the extinction, absorption and scattering cross sections of the nanostructure normalized to its geometrical cross section. In the calculations presented here, the open-source Fortran-90 software package (DDSCAT 7.1) was used.
3. Results and Discussion
3.1. Simulation Results
The observed absorbance spectrum of the gold spherical nanoparticle of a size of 30 nm (size measured in the SEM images) is compared with the simulated spectra (Figure 1). In the DDA approximation, the inter-dipole separation is chosen to be a few tenths of nanometer, giving a total of about 72,000 dipoles, sufficient to mimic the structure of the spherical particle, and to achieve a proper convergence of the simulation. The band position of both the simulated spectra shows an excellent agreement with the observed one. For particles with a size much smaller than the wavelength of the incident radiation, the quasi-static approximation is used. In this approximation, the observed surface plasmon resonance (SPR) is attributed to the excitation of the dipolar resonance. The observed SPR band centered at 540 nm is attributed to the dipolar distribution of the polarization charges. The Full Width at Half Maximum (FWHM) in the simulated spectra is comparable. Because of the size distribution of the particles in the sample, the observed band width is larger than the modeled ones.
3.2. UV Photo- and Thermal Reduction
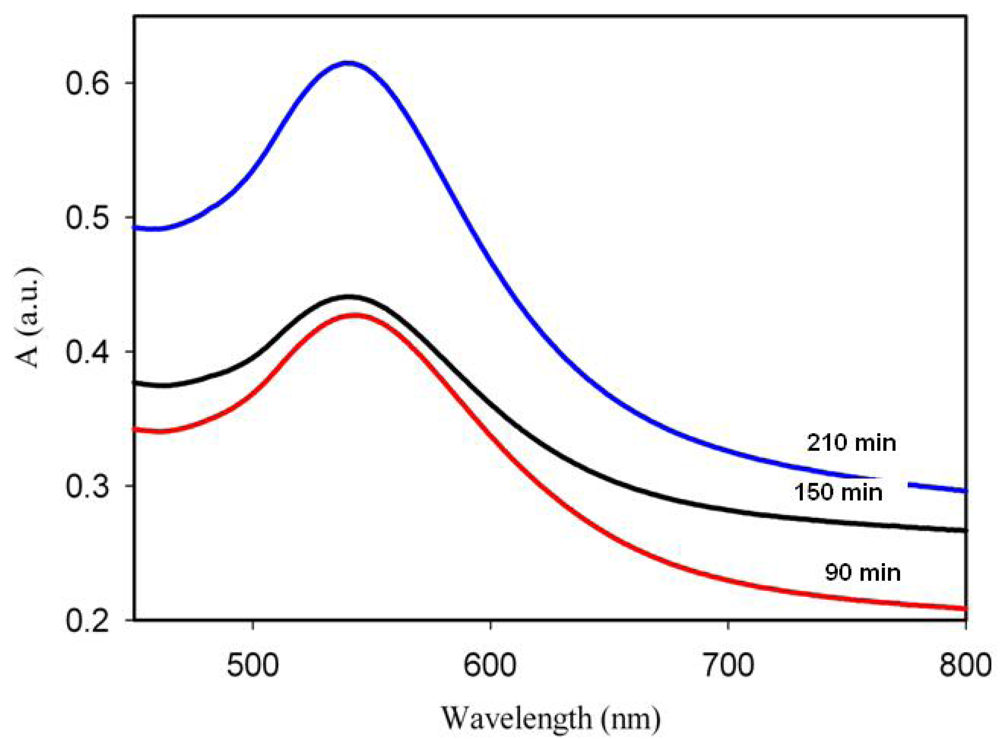
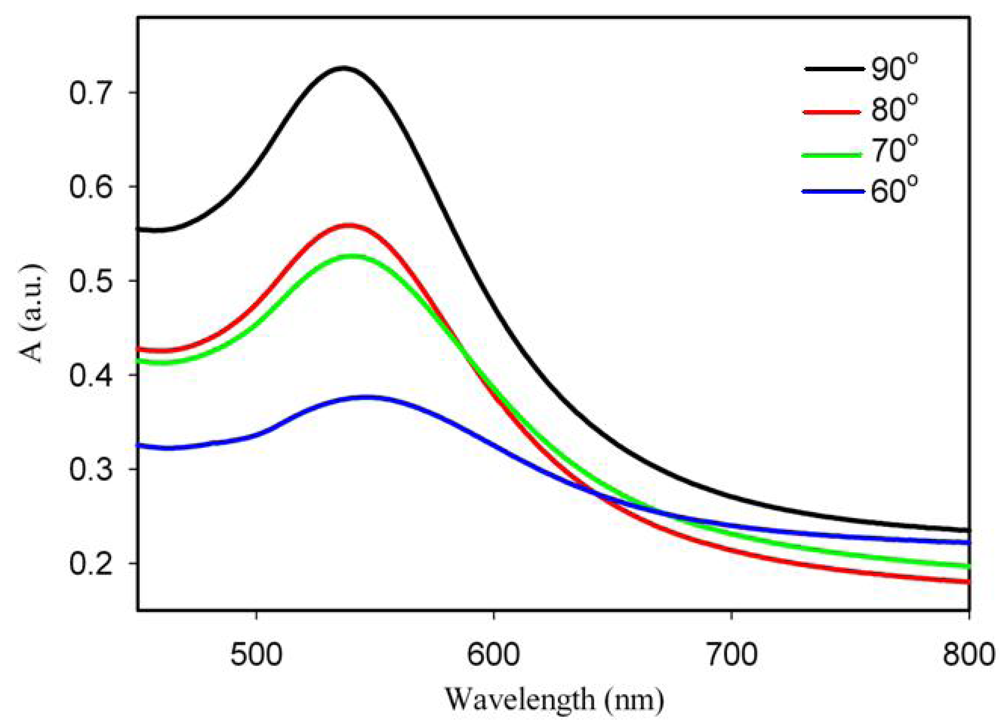
In this work, the concentration of gold in the composite material was kept constant at around 1–2% Au. Several samples were prepared with different irradiation times and post-synthesis annealing temperatures and they were used subsequently for bio-sensing experiments. The spectra are shown in Figures 2 and 3 and the corresponding SEM images in Figure 4. The spectra show that, under the conditions of irradiations in our experiments, gold nanoparticles are formed even upon irradiation for quite short times and that the amount of nanoparticles increases (although not linearly), when longer times are used. The irradiated samples were further annealed for 30 min at 90 °C. The effect of the annealing temperature on the Au coverage is shown in Figure 3. It can be seen that annealing at a temperature near to the glass transition temperature of the polymer (90 °C) yields the highest amount of gold nanoparticles. Figure 3 also shows that the size of Au nanoparticles changes when the annealing is done at different temperatures. Indeed, increasing the temperature, the LSPR band appears to be shifted by 10 nm (from 547 to 537 nm) towards lower wavelengths (blue shift), meaning that smaller particles are formed when the annealing temperature is higher. It is interesting to note that the UV-Visible absorption spectra do not show an absorption in the 600–800 nm range that would indicate the presence of non-spherical particles in the UV-irradiated and annealed samples. However, the SEM images show that, in addition to spherical nanoparticles, triangular particles are formed as well but, probably, their concentration is not high enough to generate the characteristic band.
Figure 4(a) shows the close proximity of a large triangular plate like- and several smaller spherical nanoparticles. It can be inferred that the three spherical particles seen in the image blocked the growth of the facet where they are adsorbed. The formation of non-spherical nanoparticles, specifically of triangular plates has been reported in the literature however, under quite different conditions. Triangular and hexagonal gold nanoparticles have been obtained in the presence of PVA by changing the time of irradiation and the concentration of gold in the composite [22] and their formation, favored by long irradiation times, was accounted for by the slow reduction process. Triangular prismatic nanoparticles protected by PVP were prepared by MW-assisted reduction as well [23]. The formation of nanoprisms was explained by the different growth rates at the different planes of the particles and by a competition between the growth of the nanoparticles and the surface capping process by the polymer [24]. Under the conditions of our experiment, that is, low gold concentration, low irradiation time and the presence of the PMMA matrix, it is possible that the polymer, through its carbonyl groups, would attach to Au nanoparticles non-uniformly, most probably, during the nucleation stage and the initial stage of growing. As a result, the growth rate will be different for the different facets, depending on the concentration of the stabilizing molecules. The large size of the triangular particles (around 100 nm) suggests a rapid growth, most probably, after the UV irradiation, during the subsequent annealing process.
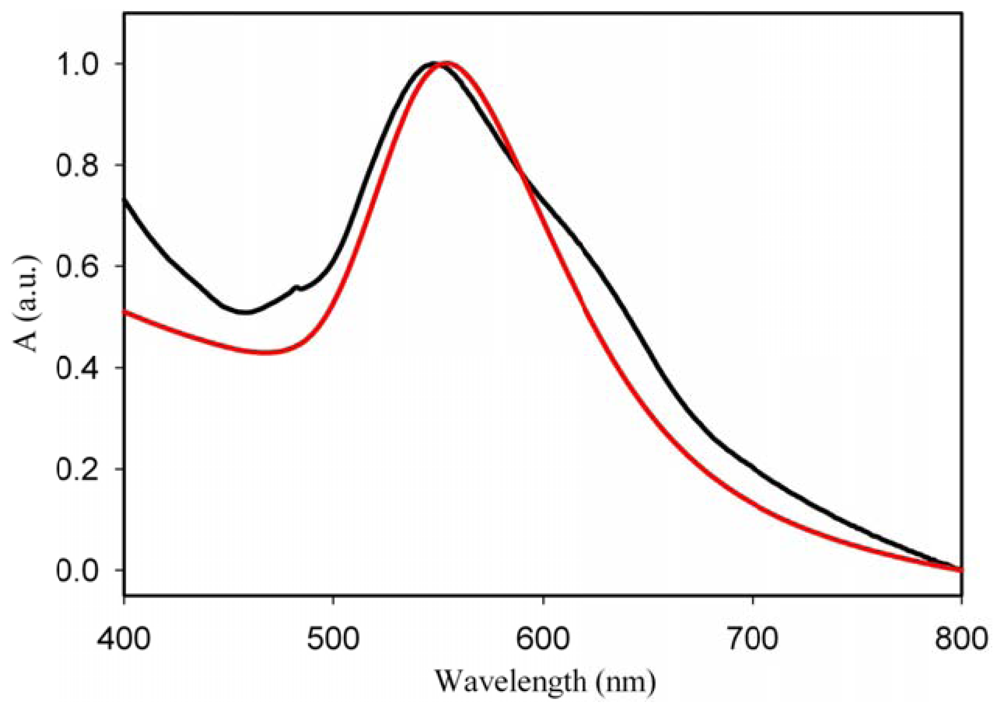
Although it is generally accepted that the formation of nanoparticles is triggered by photo-irradiation, we have demonstrated the possibility of the reduction of the gold ions without UV irradiation, by only heating the HAuCl4-PMMA spin-coated film. Figure 5 shows the spectrum of the film annealed at 90 °C.
It can be seen that heating the film immediately after the deposition, when the film still contains some acetone, results in the formation of nanoparticles with a well-defined LSPR band at 544 nm. If the heating is delayed by 30 min, the band appears to be broader and its intensity is lower. In this spectrum (red trace), a weak absorption in the longer wavelength region can be seen as well. It can be inferred that heating the film in the presence of only traces of acetone, triggers the formation of anisotropic nanoparticles as well. In this case, the reduction process of Au ions takes place on the spots containing acetone but the main route seems to be the thermo-reduction. Further work is needed to elucidate the mechanism of the reduction in this particular case, most specifically, the role of acetone.
3.3. Microwave-Assisted Reduction
The SEM image corresponding to the sample MW-irradiated for 2 min is shown in Figure 7. The image reveals a very good coverage of the surface with spherical (or quasi-spherical) nanoparticles with an average size around 30 nm. No triangular or other anisotropic nanoparticles can be seen in the SEM image corresponding to the MW-assisted reduced sample. MW-assisted synthesis of gold nanoparticles reported until now was carried out in solution and resulted in the formation of a mixture of triangular, hexagonal and other shapes. To the best of our knowledge, this is the first report on the MW irradiation of a thin film with the formation of well-distributed spherical gold nanoparticles, in the absence of any surfactant. It has been shown that MW provides rapid and uniform heating that accelerates the reduction of the metal precursor and the nucleation of gold [10]. Although, generally, the high heating rate accelerates the growing of nanoparticles as well, under the conditions of our experiment, the short duration of irradiation prevents the formation of large particles.
We suggest that, due to the presence of acetone in spin-coated sample, localized high temperatures are generated that enhance the rate of reduction of the metal precursor. Because of the quite uniform distribution of acetone in the film, hot spots are created all over the surface, ensuing in the uniformity of the particle distribution.
3.4. Bio-Sensing Experiments
3.4.1. Annealing of the Au-PMMA Nanocomposite Films
Before performing the biosensing experiments, the nanocomposite films were annealed at temperatures in the range of 250–300 °C to increase the mobility of both the Au nanoparticles and the polymer chains. Thermo-gravimetric analysis measurements carried out on Au-PMMA composites have shown that the presence of silver nanoparticles increases the thermal stability of PMMA. Indeed, it has been reported that, for composites with a low content of silver, the thermal oxidative decomposition of the composites starts only around 350 °C and that the influence of small amounts is pronounced [25]. In order to study the annealing process, the AFM images of the samples were measured, before and after annealing. The AFM images of the samples before and after annealing, are shown in Figure 7(a,b), respectively. The thickness of the film as measured from the 3D AFM image was found to be 2.3 μm. The images shown in Figure 7 clearly prove that the density of gold nanoparticles on the surface increased noticeably after annealing the sample at 300 °C for 30 min. Heating the nanocomposite samples above the glass transition temperature, transforms the polymer from a glassy state to a rubbery state that would increase the mobility of the polymer chains and, implicitly, that of Au nanoparticles. In addition, depolymerization may occur to some extent and, the presence of the monomer may contribute to the increased mobility of Au nanoparticles. Figure 8 shows the spectra of the Au-PMMA nanocomposite, before and after the annealing process.
The broad LSPR band between 500 and 700 nm in the spectrum of the irradiated sample (before annealing) shows the presence of gold aggregates in the composite. After annealing, the structure is improved as shown by the presence of the strong band at 557 nm. This result is in agreement with the AFM images that show a high density of individual gold nanoparticles on the surface of the annealed film.
3.4.2. Refractive Index Sensitivity Measurements
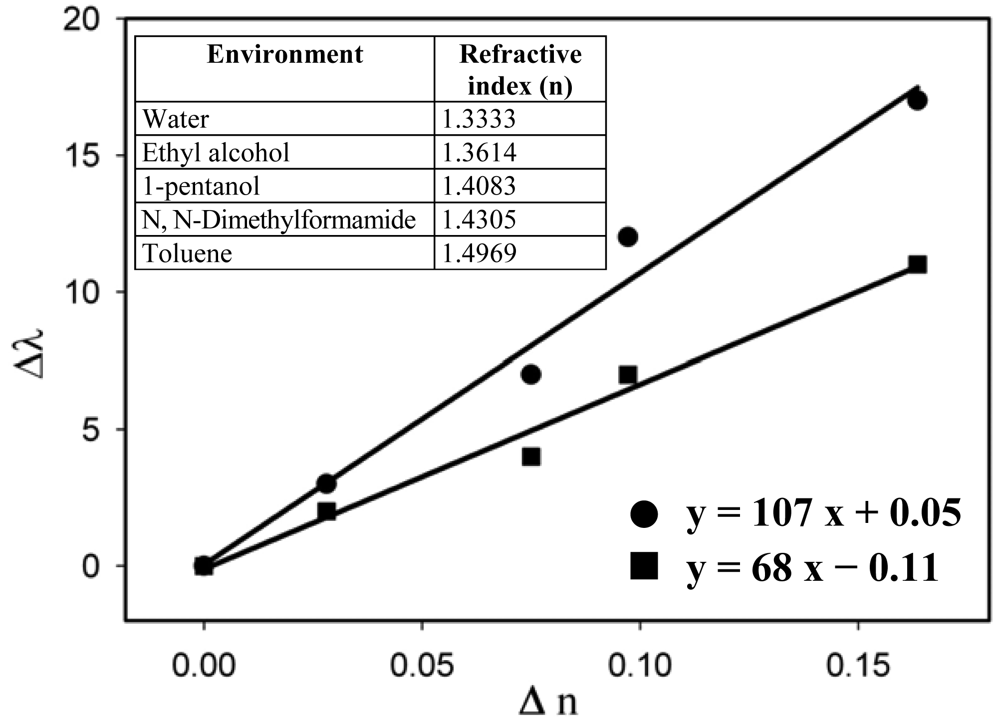
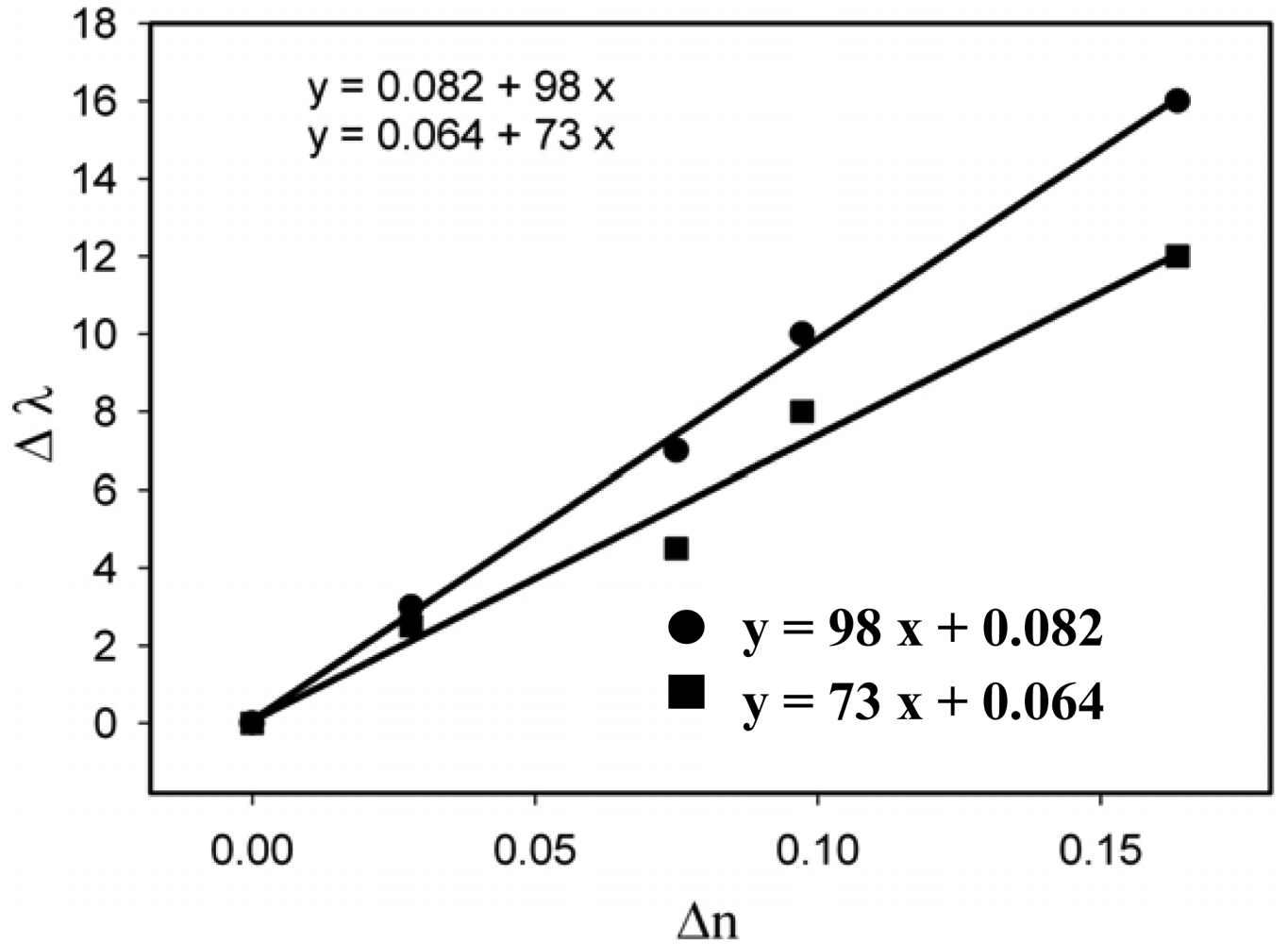
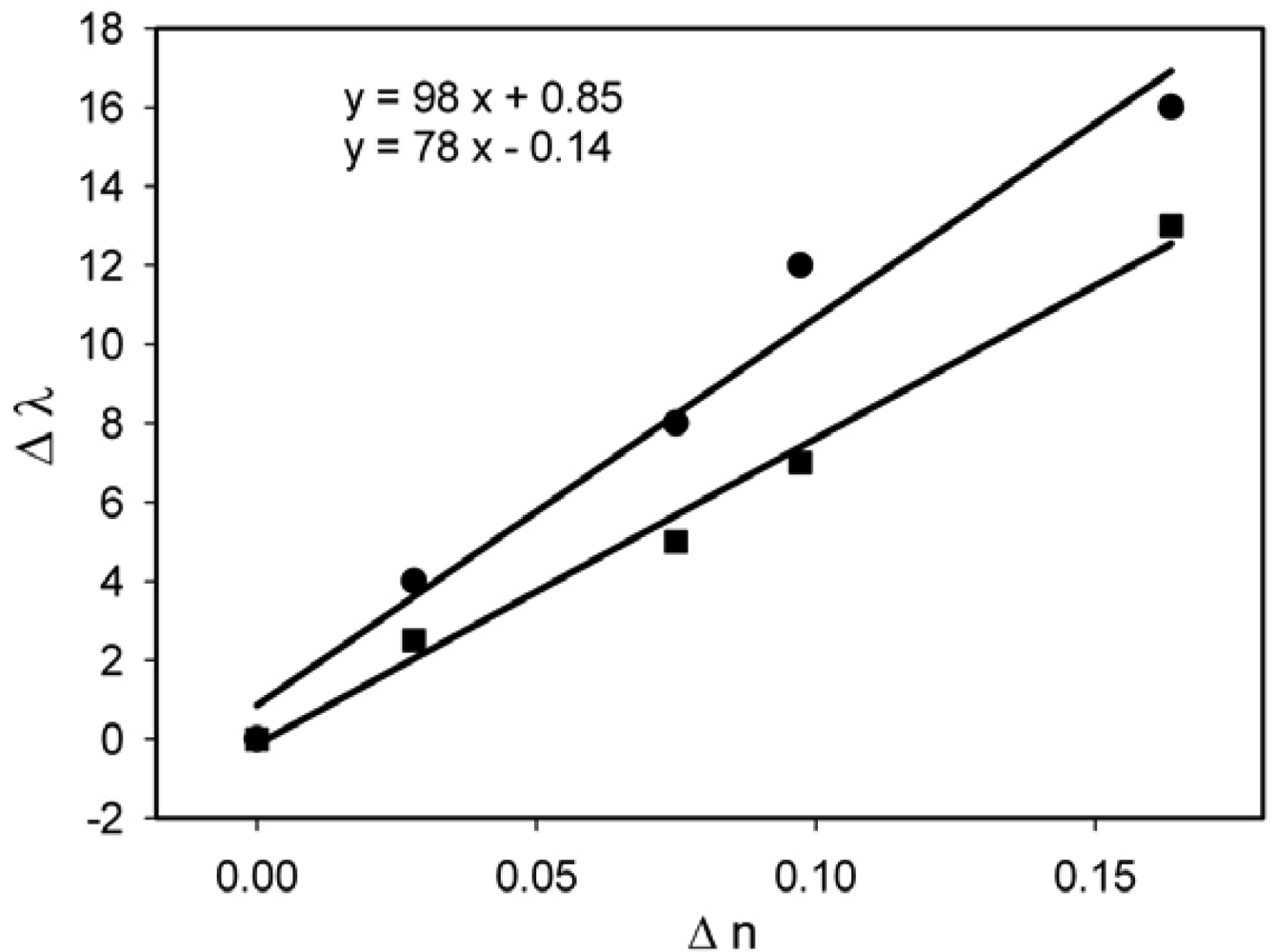
The refractive index sensitivity of the non-annealed and annealed samples was measured for the samples prepared through the three different methods (Figures 9–11).
The sensitivity results are summarized in Table 1.
The sensitivity results show that, as anticipated, the annealed samples are more sensitive than the as-prepared ones. However, even the annealed structures show a moderate sensitivity, probably because of the concentration of Au nanoparticles on the surface. To increase more the density of nanoparticles, the annealing temperature should be increased over 300 °C which would further enhance the rate of the depolymerization reaction. On the other hand, the results show that, in spite of the moderate sensitivity, nanocomposites containing gold can be used for sensing purposes. This result is important because it opens the door to lab-on-a-chip sensing by using polymer chips.
3.4.3. Biosensing Experiments
The sensitivity of the annealed nanocomposite samples to the surrounding environment was also investigated by immersing them into a solution of 1-octadecane thiol or in a carboxydextran solution for several hours. The surface-activated annealed samples have shown a significantly enhanced sensitivity toward the surrounding medium. Functionalization of gold nanoparticles with the 1-octadecane thiol molecule, by immersion into its ethanol solution, brought about a broadening of the LSPR band, together with a red shift of 12 nm, showing that they are now on the surface and can absorb and “see” the surrounding molecules.
The broadening may be due to the unexposed Au nanoparticles. However, as mentioned before, it is difficult to bring more Au np to the surface as the annealing temperature cannot be increased without provoking depolymerization of the polymer. In addition, other effects may also contribute to the broadening such as the polydispersity in nanoparticles size and shape. Indeed, nanoparticles having different sizes and/or shapes may interact in a different way with proteins yielding slightly different shifts. Possibly, some shape distortion or even denaturation of proteins may occur, making the local environment not homogeneous.
Further, by immersion of the sample with functionalized gold nanoparticles in the buffered solution of the antibody, the Au band is shifted significantly toward longer wavelengths (Δλ = 31 nm)
Figure 12 shows, step by step, the changes in the Au LSPR band, as a result of the changes in the immediate environment. Experiments performed with a dextran linker (not given here) have shown that the sensitivity is lower than with the thiol linker. The results reveal that Au-PMMA nanocomposites, annealed to increase the mobility of Au nanoparticles, can be successfully used as sensing platforms for detecting biomolecules. More quantitative experiments are necessary to determine the sensitivity of this platform and the detection limit for different antigens.
4. Conclusions
Au-PMMA nanocomposites have been prepared by UV and microwave irradiation, as well as by thermal reduction, specifically, for sensing applications. The synthesis was carried out by irradiation of a spin-coated film containing a mixture of gold precursor (HAuCl4) and poly(methyl methacrylate) dissolved in acetone. It is shown that the microwave-synthesized composite showed a uniform distribution of spherical nanoparticles, while a large number of triangular particles can be seen as a result of UV irradiation. As-prepared nanocomposites lacked sensitivity toward the surrounding environment. Annealing at 300 °C, temperature well above the glass transition temperature, brought about an important change in the structure and properties of the nanocomposite. It is inferred that in the rubbery state of the nanocomposite, the interactions between the gold and the polymer chains are weakened and, at the same time, the mobility of Au nanoparticles is augmented. The results show that, in the annealed samples, the surface density of gold nanoparticles is highly increased. Under these conditions, the nanocomposites show an enhanced sensitivity toward the surrounding molecules. The results also indicate that, by using an adequate post-synthesis heat treatment, gold-polymer nanocomposites can be successfully used as sensing platforms.
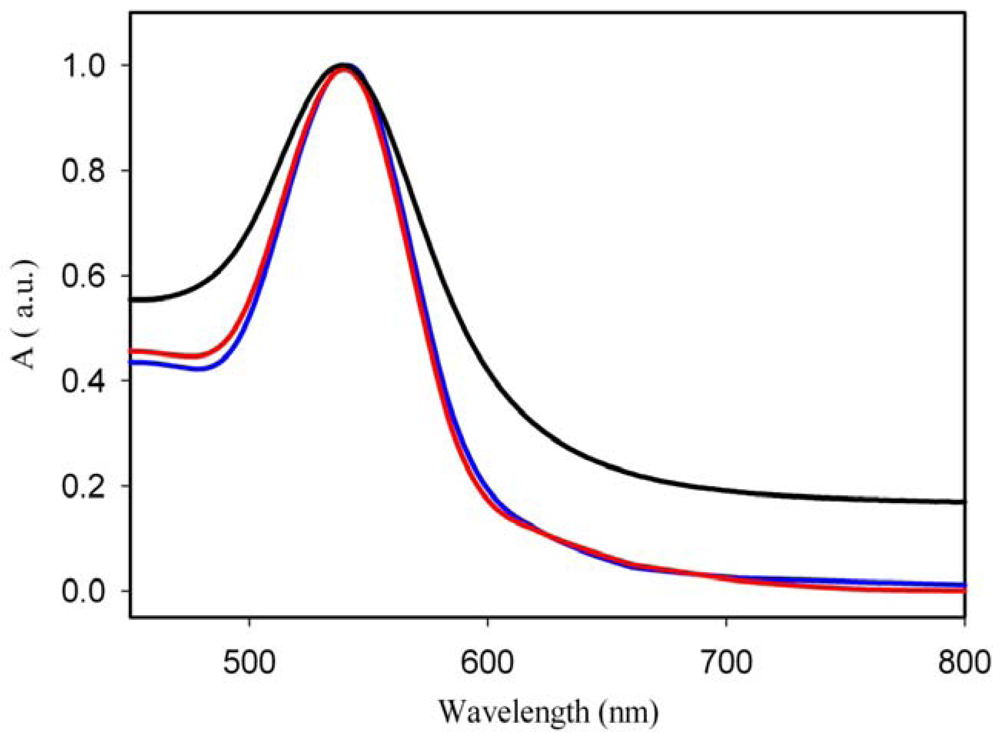

 the spin-coated sample is annealed after 30 min after the deposition, – the sample is annealed immediately).
the spin-coated sample is annealed after 30 min after the deposition, – the sample is annealed immediately).
 the spin-coated sample is annealed after 30 min after the deposition, – the sample is annealed immediately).
the spin-coated sample is annealed after 30 min after the deposition, – the sample is annealed immediately).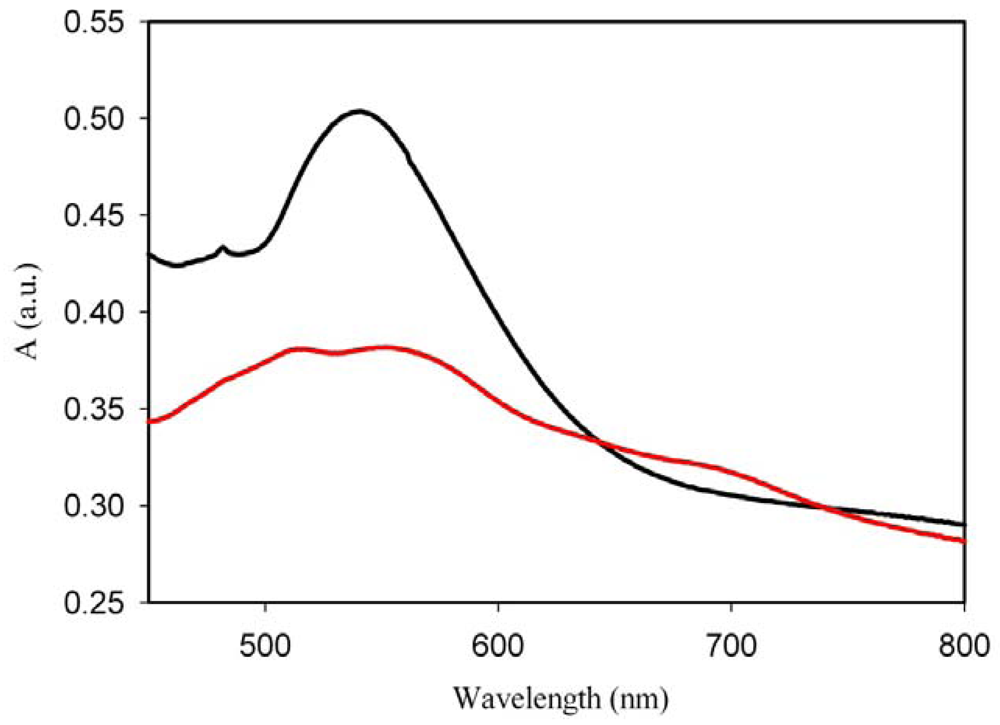



| Method of fabrication | S (nm/RIU) | |
|---|---|---|
| UV-Irradiation | Non-annealed | 77 |
| Annealed | 98 | |
| Thermal reduction | Non-annealed | 73 |
| Annealed | 98 | |
| MW-irradiation | Non-annealed | 68 |
| Annealed | 107 | |
References
- Caseri, W. Inorganic nanoparticles as optically effective additives for polymers. Chem. Eng. Comm. 2009, 196, 549–572. [Google Scholar]
- Liu, F.-K.; Hsieh, S.-Y.; Ko, F.-H.; Chu, T.-C. Synthesis of gold/poly(methyl methacrylate) hybrid nanocomposites. Colloids Surf. A: Physicochem. Eng. Asp. 2003, 231, 31–38. [Google Scholar]
- Xu, G.C.; Xiong, J.Y.; Ji, X.L.; Wang, Y.L. Synthesis of nanosilver/PMMA composites via ultrasonically Bi-in situ emulsion polymerization. J. Thermoplast. Compos. Mater. 2007, 20, 523–533. [Google Scholar]
- Singh, N.; Khanna, P.K. In situ synthesis of silver nano-particles in polymethylmethacrylate. Mater. Chem. Phys. 2007, 104, 367–372. [Google Scholar]
- Karadas, F.; Ertas, G.; Ozkaraouglu, E.; Suzer, S. X-ray induced production of gold nanoparticles on a SiO2/Si system and in a poly(methyl methacrylate) matrix. Langmuir 2005, 21, 437–442. [Google Scholar]
- Alexandrov, A.; Smirnova, L.; Yakimovich, N.; Sapogova, N.; Soustov, L.; Kirsanov, A.; Bityurin, N. UV-initiated growth of gold nanoparticles in PMMA matrix. Appl. Surf. Sci. 2005, 248, 181–184. [Google Scholar]
- Kazemian Abyaneh, M.; Paramanik, D.; Varma, S.; Gosavi, S.W.; Kulkarni, S.K. Formation of gold nanoparticles by UV irradiation. J. Phys. D: Appl. Phys. 2007, 40, 3771–3779. [Google Scholar]
- Yilmaz, E.; Ertas, G.; Bengu, E.; Suzer, S. Photopatterning of PMMA films with gold nanoparticles: Diffusion of AuCl4− ions. J. Phys. Chem. C 2010, 14, 18401–18406. [Google Scholar]
- Irzh, A.; Perkas, N.; Gedanken, A. Microwave-assisted coating of PMMA beads by silver nanoparticles. Langmuir 2007, 23, 9891–9897. [Google Scholar]
- Tsuji, M.; Hashimoto, Y.; Nishizawa, Y.; Kubokava, M.; Tsuji, T. Microwave-assisted synthesis of metallic nanostructures in solution. Chem. A Eur. J. 2005, 11, 440–452. [Google Scholar]
- Jiang, H.; Moon, K.; Zhang, Z.; Pothukuchi, S.; Wong, C.P. Variable frequency microwave synthesis of silver nanoparticles. J. Nanopart. Res. 2006, 8, 117–124. [Google Scholar]
- Ramesh, G.V.; Sudheendran, K.; Raju, K.C.J.; Streedhar, B.; Radhakrishnan, T.P. Microwave absorber based on silver nanoparticle-embedded polymer thin film. J. Nanosci. Nanotechnol. 2009, 9, 261–266. [Google Scholar]
- Devi, P.; Mahmoud, A.Y.; Badilescu, S.; Packirisamy, M.; Jeevanandam, P.; Truong, V.-V. Synthesis and Surface Modification of Poly (dimethylsiloxane)-Gold Nanocomposite Films for Biosensing Applications. Proceedings of 2010 International Conference on Biosciences (BIOSCIENCESWORLD), Cancun, Mexico, 7–13 March 2010.
- Barbillon, G.; Bijeon, J.-L.; Plain, J.; Lamy de la Chapelle, M.; Adam, P.-M.; Royer, P. Biological and chemical nanosensors based on localized surface plasmon resonance. Gold Bull. 2007, 40, 240–244. [Google Scholar]
- Stuart, D.A.; Haes, A.J.; Yonzon, C.R.; Hicks, E.M.; Van Duyne, R.P. Biological applications of localized surface plasmonic phenomenae. IEE Proc.-Nanobiotechnol. 2005, 152, 13–32. [Google Scholar]
- Haes, A.J.; Zou, S.; Schatz, G.C.; van Duyne, R.P. Nanoscale optical biosensor: Short range distance dependence of the localized surface plasmon resonance of noble metal nanoparticles. J. Phys. Chem. B 2004, 108, 6961–6968. [Google Scholar]
- Haes, A.J.; Zou, S.; Schatz, G.C.; Van Duyne, R.P. A nanoscale optical biosensor: The long range distance dependence of the localized surface plasmon resonance of noble metal nanoparticles. J. Phys. Chem. B 2004, 108, 109–116. [Google Scholar]
- Draine, B.T.; Flatau, P.J. Discrete-dipole approximation for periodic targets: Theory and tests. J. Opt. Soc. Am. A 1994, 11, 2693–2703. [Google Scholar]
- Collinge, M.J.; Draine, B.T. Discrete-dipole approximation with polarizabilities that account for both finite wavelength and target geometry. J. Opt. Soc. Am. A 2004, 21, 2023–2028. [Google Scholar]
- Draine, B.T.; Goodman, J.J. Beyond Clausius-Mossotti-Wave propagation on a polarizable point lattice and the discrete dipole approximation. Astrophys. J. 1993, 405, 685–697. [Google Scholar]
- Mie, G. Beitrage zur optik truber medien, speziell kolloida ler metallosungen. Ann. Phys. 1908, 25, 377–455. [Google Scholar]
- Zhou, Y.; Wang, C.Y.; Zhu, Y.R.; Chen, Z.Y. A novel ultraviolet irradiation technique for shape-controlled synthesis of gold nanoparticles at room temperature. Chem. Mater. 1999, 11, 2310–2312. [Google Scholar]
- Nirmala Grace, A.; Pandian, K. One pot synthesis of polymer protected gold nanoparticles and nanoprisms in glycerol. Colloid Surf. A: Physicochem. Eng. Asp. 2006, 290, 138–142. [Google Scholar]
- Petroski, J.M.; Wang, Z.L.; Green, T.C.; El-Sayed, M. Kinetically controlled growth and shape formation mechanism of platinum nanoparticles. J. Phys. Chem. B 1998, 102, 3316–3320. [Google Scholar]
- Vodnik, V.V.; Božanić, D.K.; Džunuzović, E.; Vuković, J.; Nedeljković, J.M. Thermal and optical properties of silver-poly(methylmethacrylate) nanocomposites prepared by in-situ radical polymerization. Eur. Polym. J. 2010, 46, 137–144. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license(http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Alsawafta, M.; Badilescu, S.; Paneri, A.; Truong, V.-V.; Packirisamy, M. Gold-Poly(methyl methacrylate) Nanocomposite Films for Plasmonic Biosensing Applications. Polymers 2011, 3, 1833-1848. https://doi.org/10.3390/polym3041833
Alsawafta M, Badilescu S, Paneri A, Truong V-V, Packirisamy M. Gold-Poly(methyl methacrylate) Nanocomposite Films for Plasmonic Biosensing Applications. Polymers. 2011; 3(4):1833-1848. https://doi.org/10.3390/polym3041833
Chicago/Turabian StyleAlsawafta, Mohammed, Simona Badilescu, Abhilash Paneri, Vo-Van Truong, and Muthukumaram Packirisamy. 2011. "Gold-Poly(methyl methacrylate) Nanocomposite Films for Plasmonic Biosensing Applications" Polymers 3, no. 4: 1833-1848. https://doi.org/10.3390/polym3041833






