Ultrathin g-C3N4 Nanosheet-Modified BiOCl Hierarchical Flower-Like Plate Heterostructure with Enhanced Photostability and Photocatalytic Performance
Abstract
:1. Introduction
2. Results and Discussion
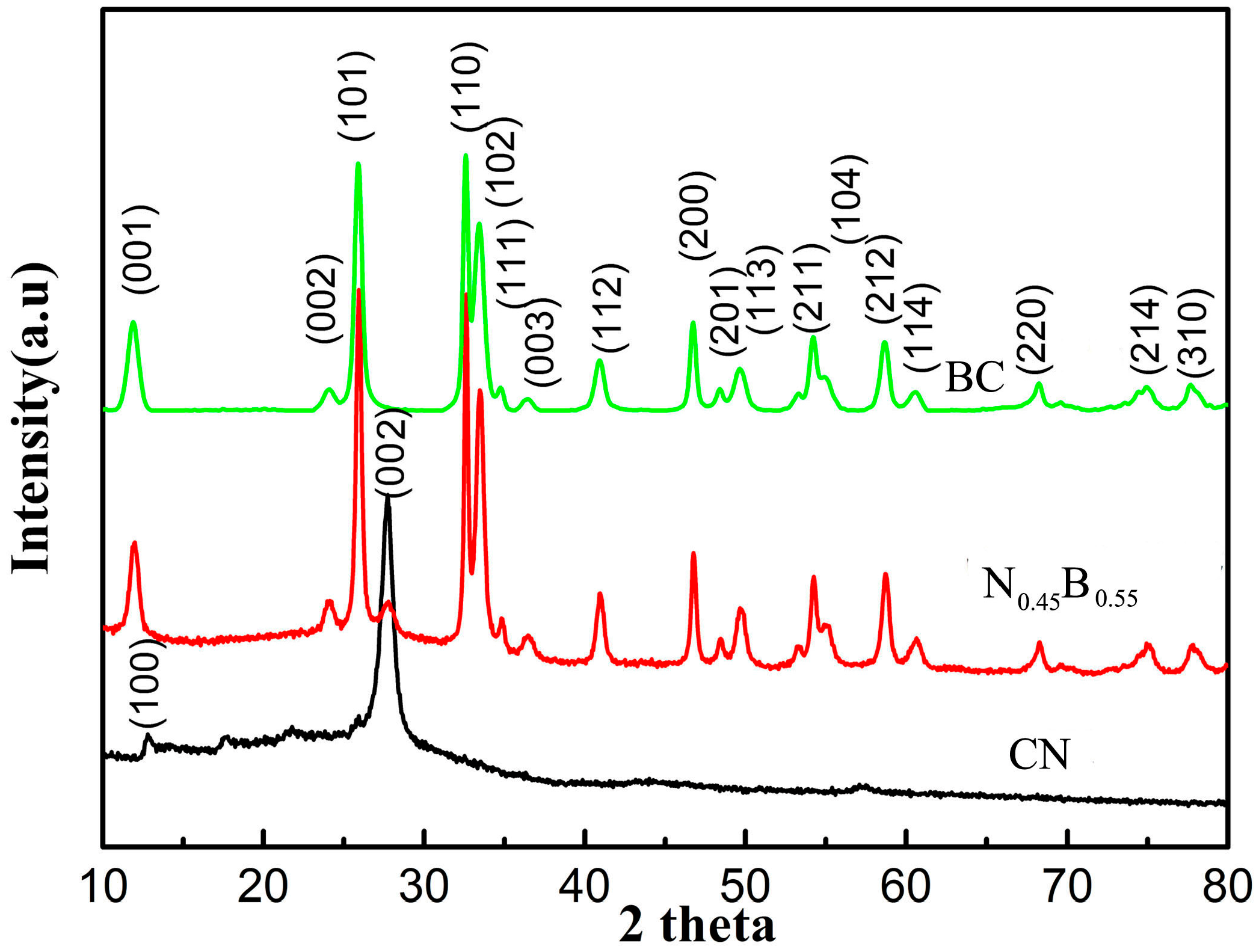
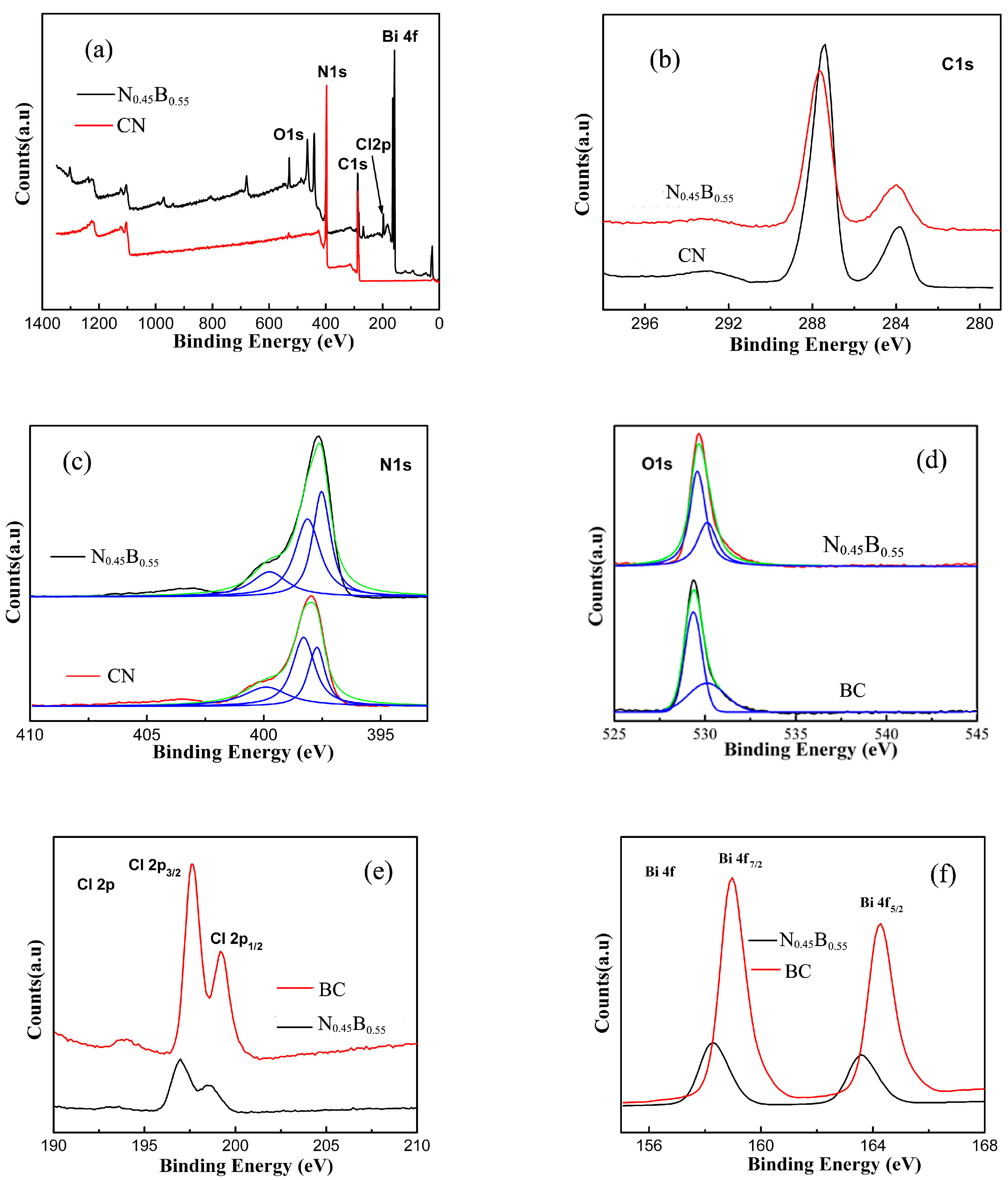
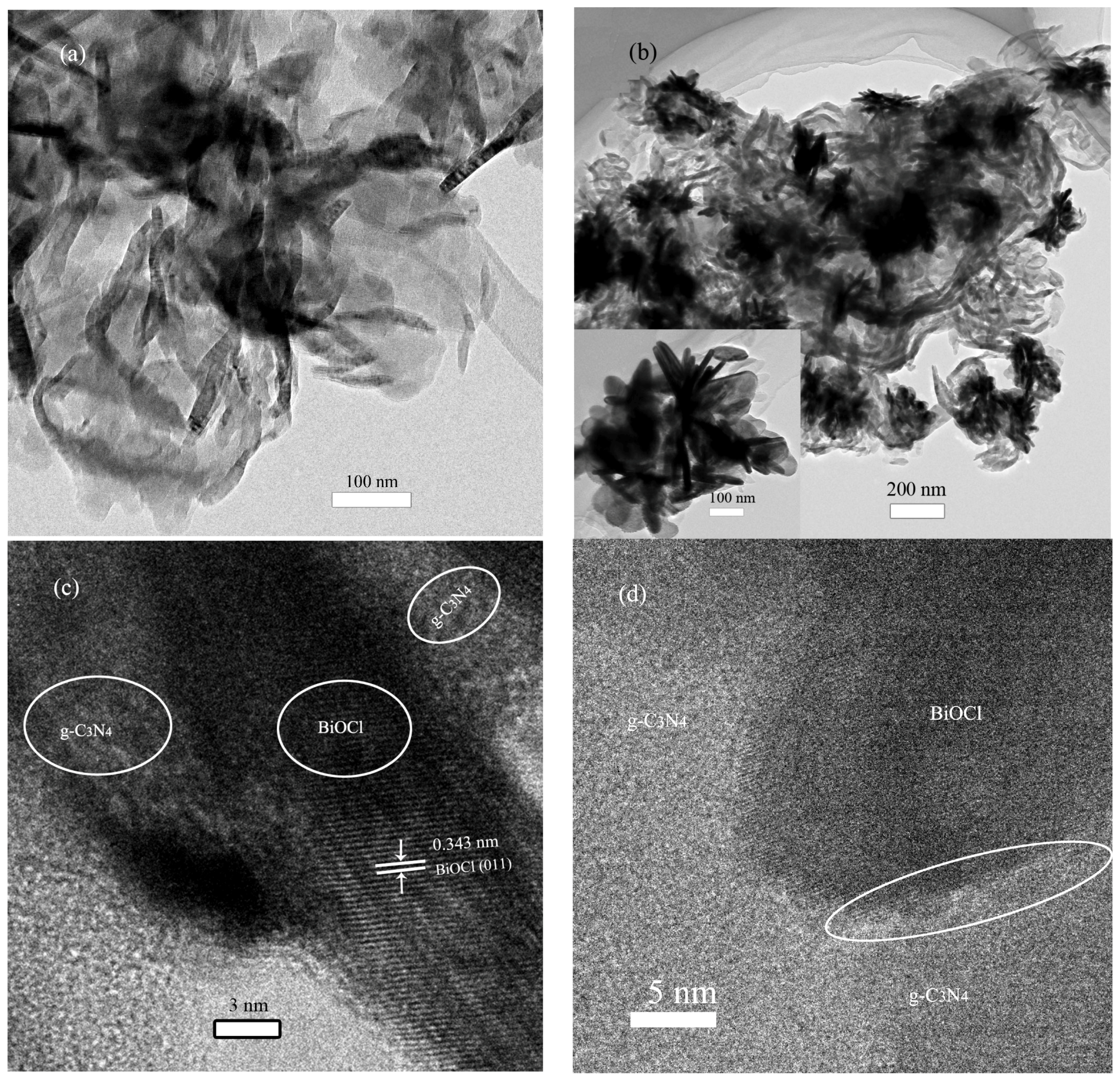
2.1. Phase Structure and Morphology
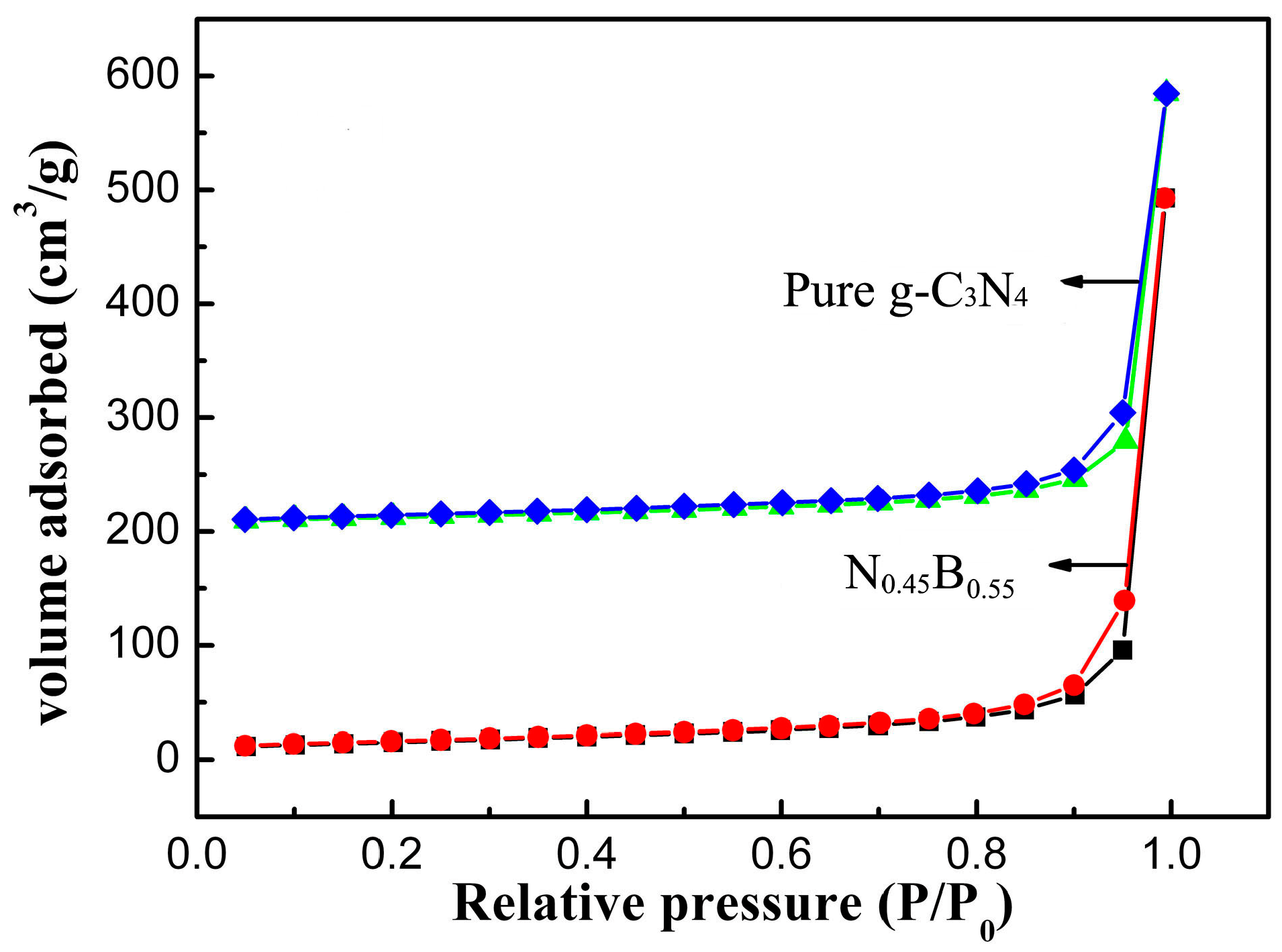
2.2. BET Surface Area
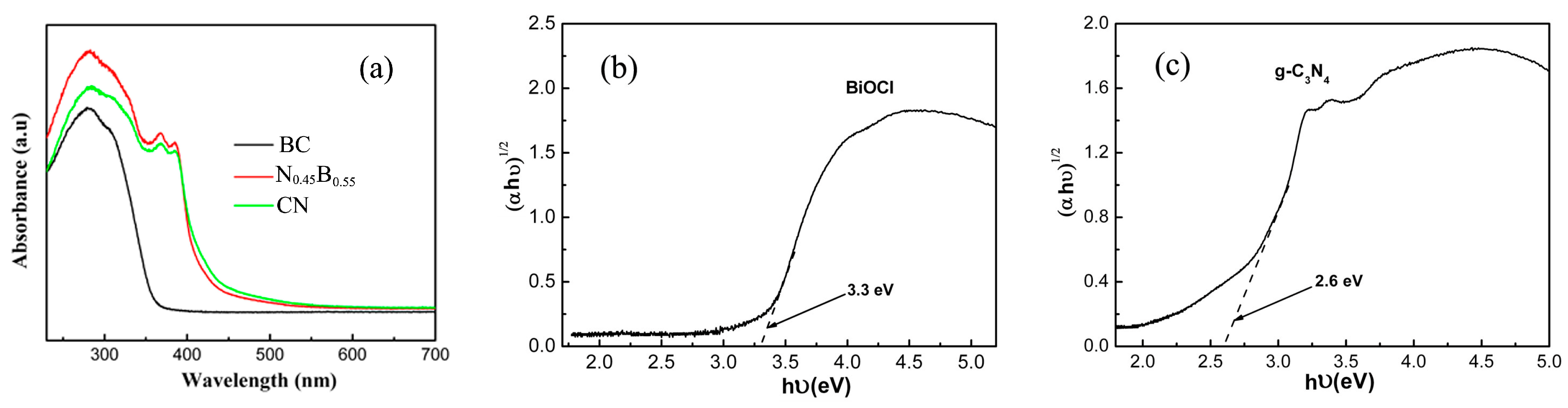
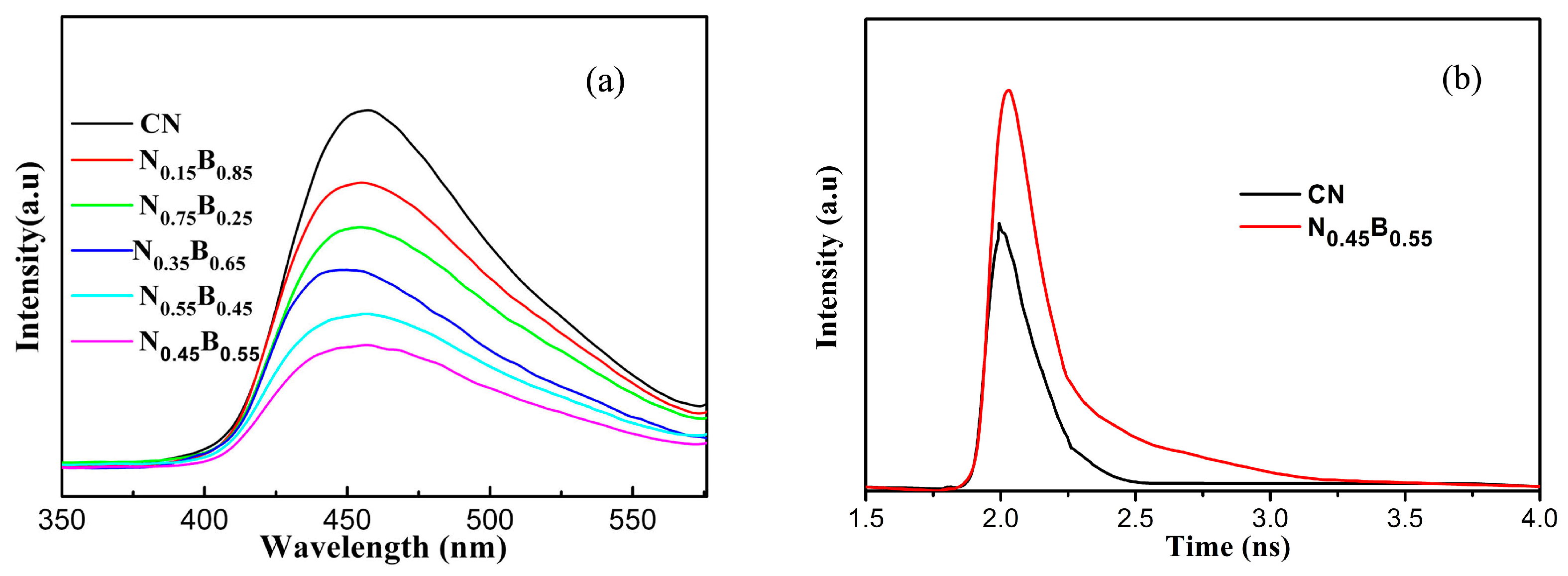
2.3. Optical and Electronic Properties
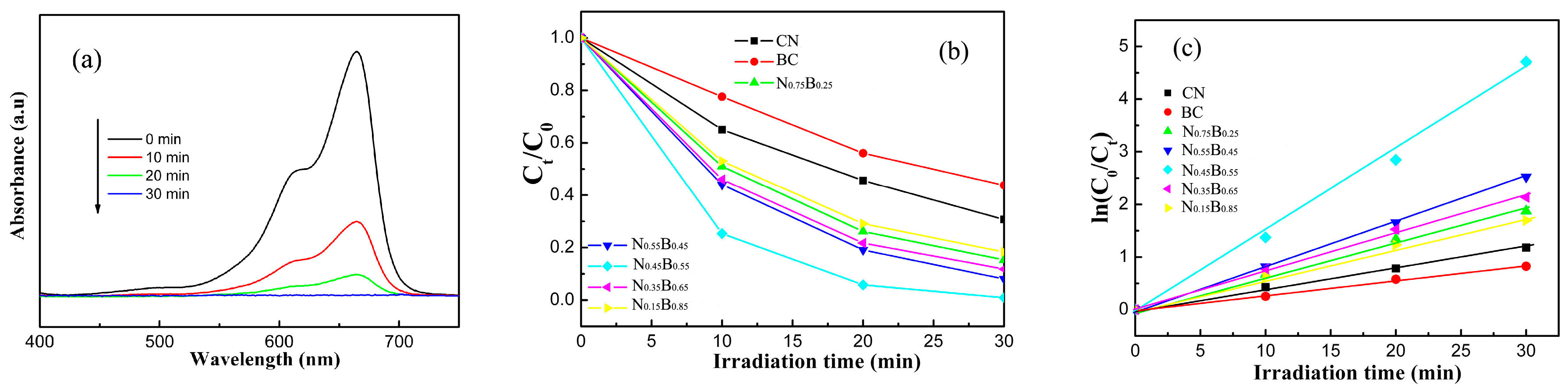
2.4. Photocatalytic Performance
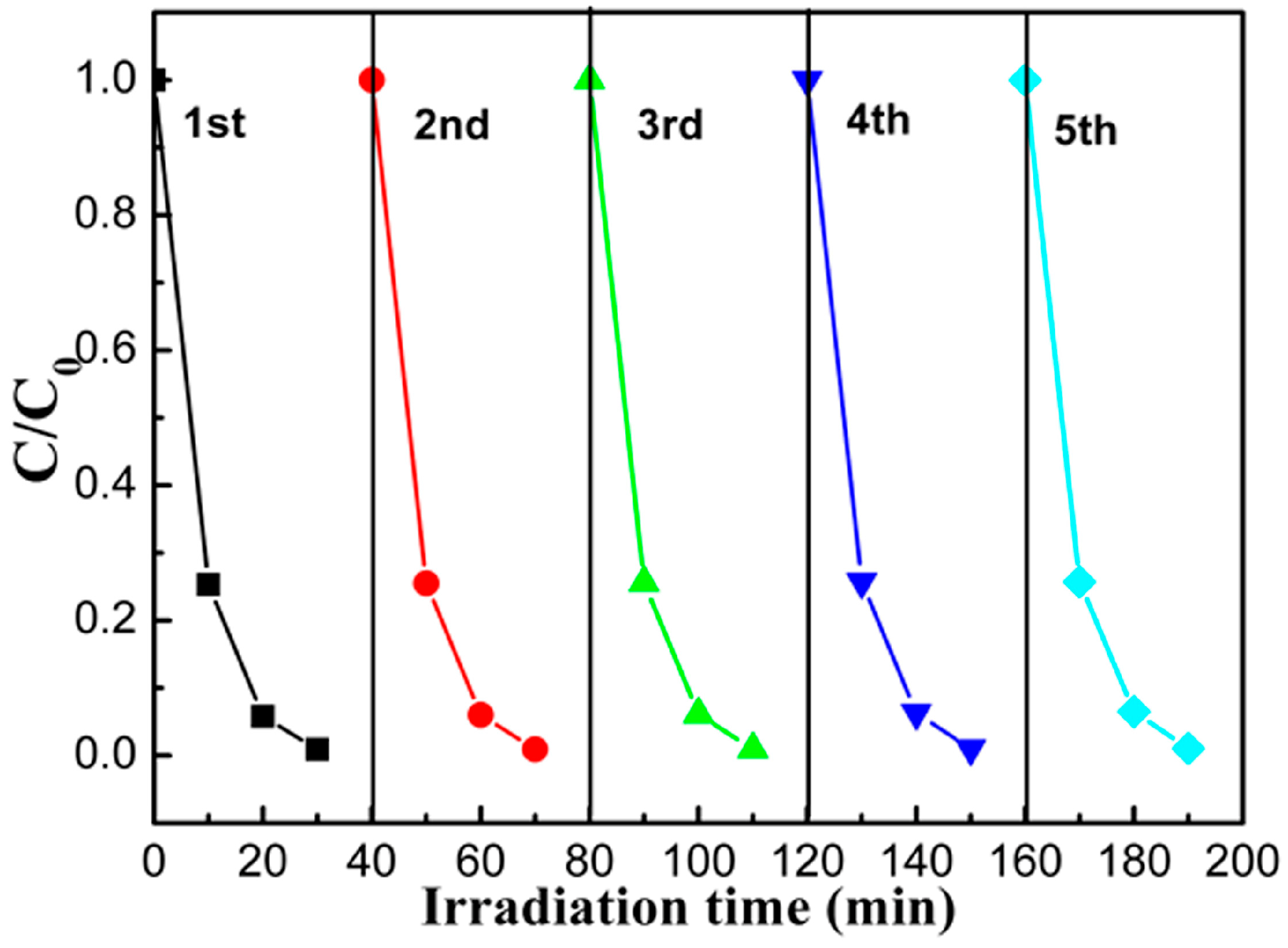
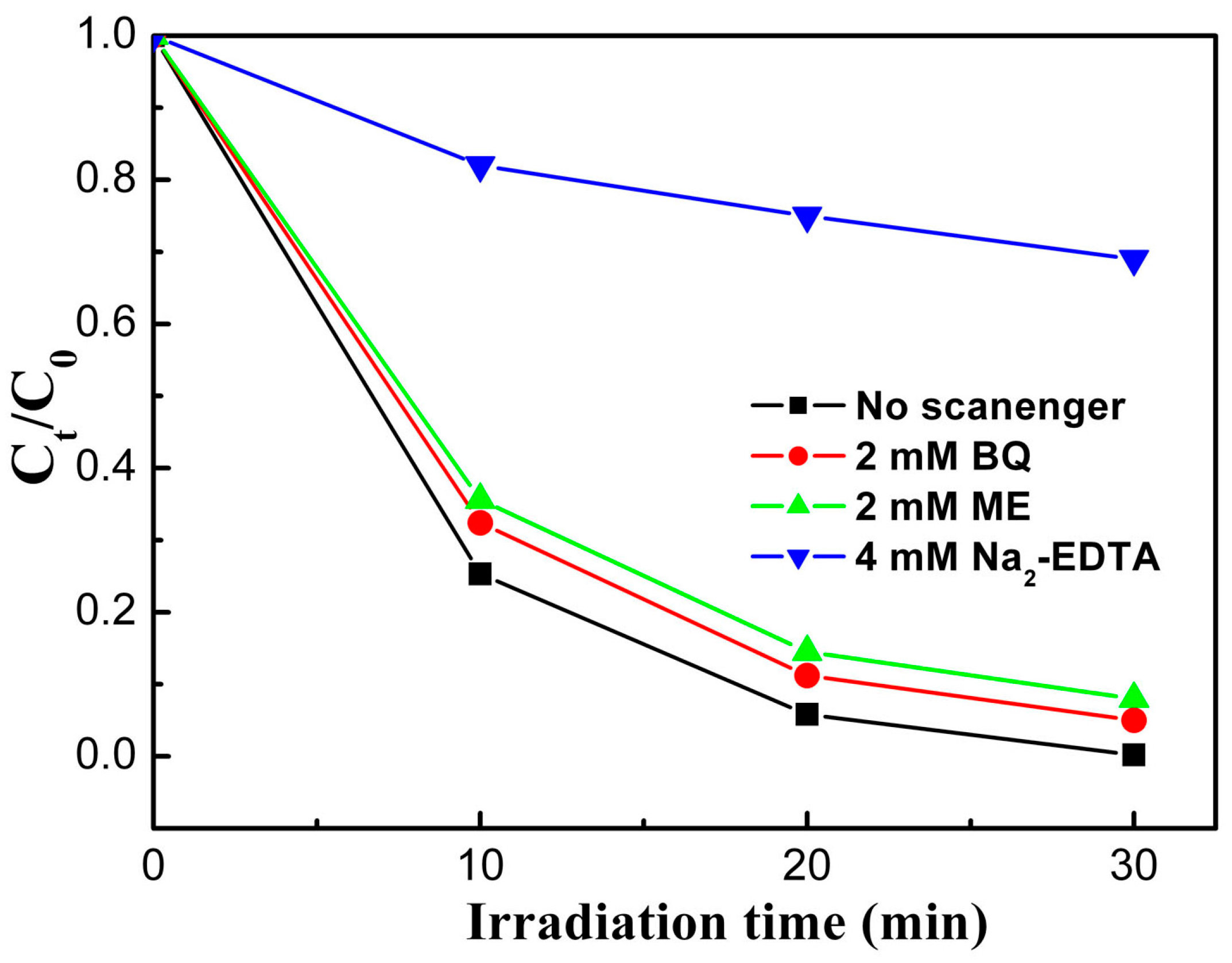
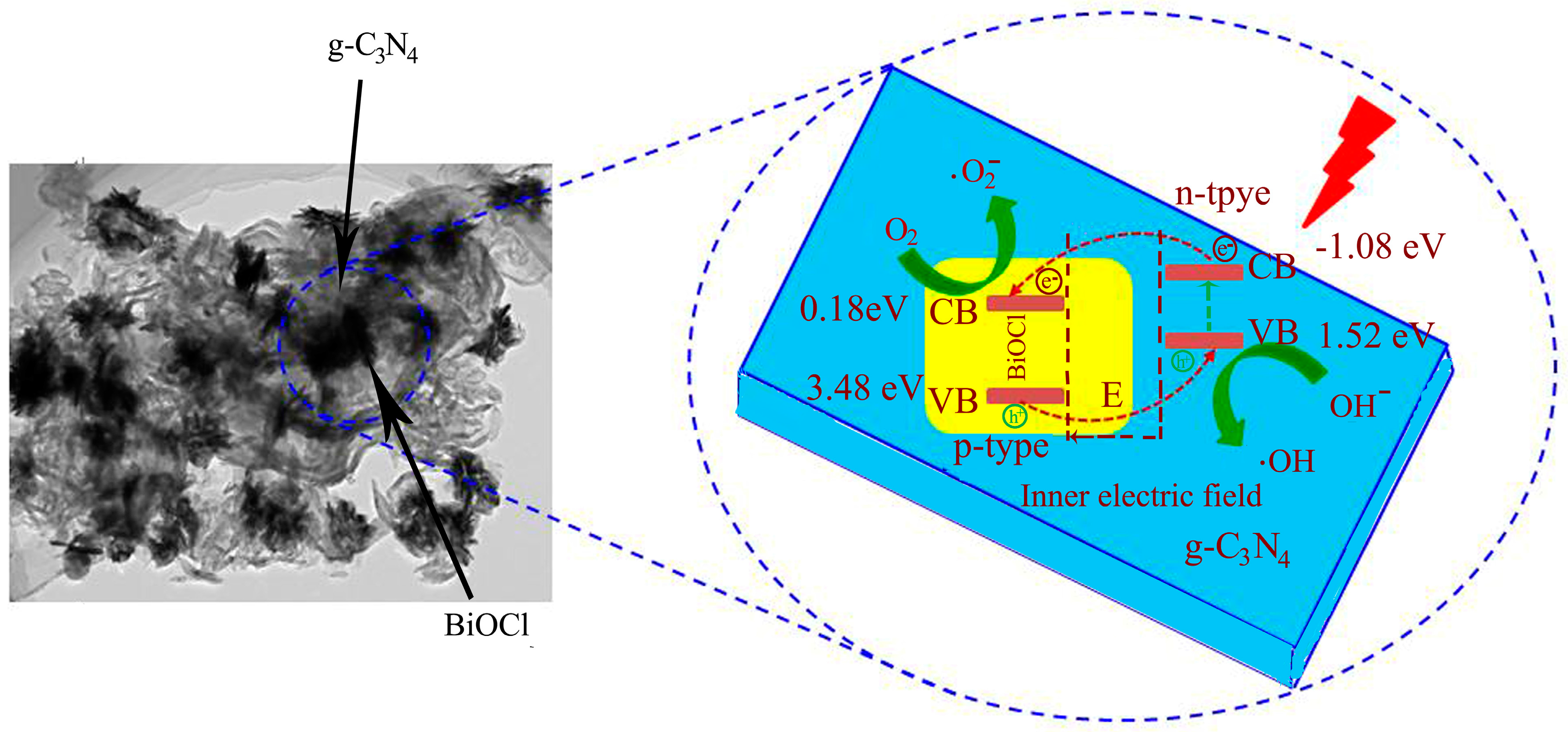
2.5. Catalyst Photostability and Photocatalytic Mechanism
3. Experimental Section
3.1. Materials
3.2. Synthesis of g-C3N4/ BiOCl Heterostructure
3.2.1. Synthesis of Ultrathin g-C3N4 Nanosheets
3.2.2. Synthesis of g-C3N4 /BiOCl Heterostructure
3.3. Characterization
3.4. Photocatalytic Experiments
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| XRD | X-ray diffraction |
| SEM | Scanning electron microscopy |
| TEM | Transmission electron microscopy |
| HRTEM | High resolution transmission electron microscopy |
| XPS | X-ray photoelectron spectroscopy |
| BET | Brunauer–Emmett–Teller |
| DRS | Diffuse reflectance spectrum |
| PL | Photoluminescence |
| MB | Methylene blue |
References
- Liu, S.W.; Yu, J.G.; Jaroniec, M. Tuable photocatalytic selectivity of hollow TiO2 microspheres composed of anatase polyhedra with exposed {001} facets. J. Am. Chem. Soc. 2010, 132, 11914–11916. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.Y.; Wang, Z.F.; Xie, L.Y.; Fang, Z.B.; Feng, W.H.; Huang, M.L.; Liu, P. Synthesis of single-crystal-like TiO2 hierarchical spheres with exposed {101} and {111} facets via lysine-inspired method. Appl. Surf. Sci. 2015, 353, 714–722. [Google Scholar] [CrossRef]
- Vinu, R.; Madras, G. Kinetics of simultaneous photocatalytic degradation of phenolic compounds and reduction of metal ions with nano-TiO2. Environ. Sci. Technol. 2008, 42, 913–919. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Liu, J.; Lan, J.; Li, G.; Chen, Q.; Jiang, G. 3D hierarchical anatase TiO2 superstructures constructed by “nanobricks” built nanosheets with exposed{001}facets: Facile synthesis, formation mechanism and superior photocatalyticactivity. CrystEngComm 2014, 16, 10547–10552. [Google Scholar] [CrossRef]
- Zhu, X.F.; Cheng, B.; Yu, J.G.; Ho, W.K. Halogen poisoning effect of Pt-TiO2 for formaldehyde catalytic oxidation performance at room temperature. Appl. Surf. Sci. 2016, 364, 808–814. [Google Scholar] [CrossRef]
- Cheng, H.F.; Huang, B.B.; Dai, Y. Engineering BiOX (X = Cl, Br, I) nanostructures for highly efficient photocatalytic applications. Nanoscale 2014, 6, 2009–2026. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.J.; Wang, C.; Cui, Y.F. Controllable growth of BiOCl film with high percentage of exposed {001} facets. Appl. Surf. Sci. 2014, 289, 266–273. [Google Scholar] [CrossRef]
- Guan, L.M.; Xiao, C.; Zhang, J.; Fan, S.J.; An, R.; Cheng, Q.M.; Xie, J.F.; Zhou, M.; Ye, B.J.; Xie, Y. Vacancy associates promoting solar-driven photocatalytic activity of ultrathin bismuth oxychloride microspheres in degradation of bisphenol A. Environ. Sci. Technol. 2015, 49, 6240–6248. [Google Scholar]
- Liu, W.W.; Shang, Y.Y.; Zhu, A.Q.; Tan, P.F.; Liu, Y.; Qiao, L.L.; Chu, D.W.; Xiong, X.; Pan, J. Enhanced performance of doped BiOCl nanoplates for photocatalysis: Understanding from doping insight into improved spatial carrier separation. J. Mater. Chem. A 2017, 5, 12542–12549. [Google Scholar] [CrossRef]
- Chen, L.; Yin, S.-F.; Huang, R.; Zhou, Y.; Luo, S.-L.; Au, C.-T. Facile synthesis of BiOCl nano-flowers of narrow band gap and their visible-light-induced photocatalytic property. Catal. Commun. 2012, 23, 54–57. [Google Scholar] [CrossRef]
- Jiang, J.; Zhao, K.; Xiao, X.Y.; Zhang, L.Z. Synthesis and faceted-dependent photoreactivity of BiOCl single-crystalline nanosheets. J. Am. Chem. Soc. 2012, 134, 4473–4476. [Google Scholar] [CrossRef] [PubMed]
- Shenawi-Khalil, S.; Uvarov, V.; Menes, E.; Popov, I.; Sasson, Y. New efficient visible light photocatalyst based on heterojunction of BiOCl-bismuth oxyhydrate. Appl. Catal. A 2012, 413, 1–9. [Google Scholar] [CrossRef]
- Cao, J.; Xu, B.Y.; Lin, H.L.; Luo, B.D.; Chen, S.F. Novel Bi2S3-sensitized BiOCl with highly visisible light photocatalytic activity for the removal of rhodamine B. Catal. Commun. 2012, 26, 204–208. [Google Scholar] [CrossRef]
- Cao, Q.W.; Zheng, Y.F.; Song, X.C. The enhanced visible light photocatalytic activity of Bi2WxMo1−xO6- BiOCl heterojunctions with adjustable energy band. Ceram. Int. 2016, 42, 14533–14542. [Google Scholar] [CrossRef]
- Xiao, X.R.; Liang, H.; Zuo, M.X.; Nan, J.; Li, L.; Zhang, W. One-pot solvothermal synthesis of three-dimensional (3D) BiOI/BiOCl composites with enhanced visible-light photocatalytic activities for the degradation of bisphenol-A. J. Hazard. Mater. 2012, 233–234, 122–130. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Shi, Y.; Gao, C.; Wen, L.; Chen, J.; Song, S. BiOCl/BiVO4 p–n heterojunction with enhanced photocatalytic activity under visible-light irradiation. J. Phys. Chem. C 2014, 118, 389–398. [Google Scholar] [CrossRef]
- Shamaila, S.; Sajjad, A.K.; Chen, F.; Zhang, J. WO3/BiOCl, a novel heterojunction as visible light photocatalyst. J. Colloid Interface Sci. 2011, 356, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Xu, L.; Wei, B.; Zhang, M.; Gao, H.; Sun, W. Enhanced photosensitization process induced by the p–n junction of Bi2O2CO3/BiOCl heterojunctions on the degradation of rhodamine B. Appl. Surf. Sci. 2014, 303, 360–366. [Google Scholar] [CrossRef]
- Chai, S.Y.; Kim, Y.J.; Jung, M.H.; Chakraborty, A.K.; Jung, D.; Lee, W.L. Heterojunctioned BiOCl/Bi2O3, a new visible light photocatalyst. J. Catal. 2009, 262, 144–149. [Google Scholar] [CrossRef]
- Cao, S.W.; Low, X.J.; Yu, J.G.; Jaroniec, M. Polymeric photocatalysts based on graphitic carbon nitride. Adv. Mater. 2015, 27, 2150–2176. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, X.; Antonietti, M. Polymeric Graphitic carbon nitride as a heterogeneous organocatalyst: From photochemistry to multipurpose catalysis to sustainable chemistry. Angew. Chem. 2012, 51, 68–89. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Zhang, L.W.; Shi, R.; Zhu, Y.F. Chemical exfoliation of graphitic carbon nitride for efficient heterogeneous photocatalysis. J. Mater. Chem. A 2013, 1, 14766–14772. [Google Scholar] [CrossRef]
- Liu, J.H.; Zhang, T.K.; Wang, Z.C.; Dawson, G.; Chen, W. Simple pyrolysis of urea into graphitic carbon nitride with recyclable adsorption and photocatalytic activity. J. Mater. Chem. 2011, 21, 14398–14401. [Google Scholar] [CrossRef]
- Yu, Y.Z.; Wang, J.G. Direct microwave synthesis of graphitic C3N4 with improved visible-light photocatalytic activity. Ceram. Int. 2016, 42, 4063–4071. [Google Scholar] [CrossRef]
- Xiang, Q.; Yu, J.; Jaroniec, M. Preparation and enhanced visible-Light photocatalytic H2-production activity of graphene/C3N4 composites. J. Phys. Chem. C 2011, 115, 7355–7363. [Google Scholar] [CrossRef]
- Groenewolt, M.; Antonietti, M. Synthesis of g-C3N4 nanoparticles in mesoporous silica host matrices. Adv. Mater. 2005, 17, 1789–1792. [Google Scholar] [CrossRef]
- Chen, W.; Liu, T.Y.; Huang, T.; Liu, X.H.; Zhu, J.W.; Duan, G.R.; Yang, X.J. In situ fabrication of novel Z-scheme Bi2WO6 quantum dots/g-C3N4 ultrathin nanosheets heterostructures with improved photocatalytic activity. Appl. Surf. Sci. 2015, 355, 379–387. [Google Scholar] [CrossRef]
- Zhao, W.; Wei, Z.B.; He, H.; Xu, J.; Li, J.H.; Yang, S.G.; Sun, C. Supporting 1-D AgVO3 nanoribbons on single layer 2-D graphitic carbon nitride ultrathin nanosheets and their excellent photocatalytic activities. Appl. Catal. A Gen. 2015, 501, 74–82. [Google Scholar] [CrossRef]
- Li, Z.; Yang, S.; Zhou, J.; Li, D.; Zhou, X.; Ge, C.; Fang, Y. Novel mesoporous g-C3N4 and BiPO4 nanorods hybrid architectures and their enhanced visible-light driven photocatalytic performances. Chem. Eng. J. 2014, 241, 344–351. [Google Scholar] [CrossRef]
- Yao, Y.J.; Lu, F.; Zhu, Y.P.; Wei, F.Y.; Liu, X.T.; Lian, C.; Wang, S.B. Magnetic core-shell CuFe2O4@C3N4 hybrids for visible light photocatalysis of Orange II. J. Hazard. Mater. 2015, 297, 224–233. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Yan, Q.; Yan, T.; Li, M.M.; Wei, D.; Wang, Z.P.; Wei, Q.; Du, B. Facile fabrication of 3D flower-like heterostructured g-C3N4/SnS2 composite with efficient photocatalytic activity under visible light. RSC Adv. 2014, 4, 31019–31027. [Google Scholar] [CrossRef]
- Zhang, F.; Xie, F.; Zhu, S.; Liu, J.; Zhang, J.; Mei, S.; Zhao, W. A novel photofunctional g-C3N4/Ag3PO4 bulk heterojunction for decolorization of RhB. Chem. Eng. J. 2013, 228, 435–441. [Google Scholar] [CrossRef]
- Chen, J.; Shen, S.H.; Guo, P.H.; Wang, M.; Wu, P.; Wang, X.X.; Guo, L.J. In-situ reduction synthesis of nano-sized Cu2O particles modifyingg-C3N4 for enhanced photocatalytic hydrogen production. Appl. Catal. B Environ. 2014, 152–153, 335–341. [Google Scholar] [CrossRef]
- Zhang, X.; Hu, J.; Jiang, H. Facile modification of a graphitic carbon nitride catalyst to improve its photo-reactivity under visible light irradiation. Chem. Eng. J. 2014, 256, 230–237. [Google Scholar] [CrossRef]
- Zhang, L.H.; Li, L.; Sun, X.M.; Liu, P.; Yang, D.F.; Zhao, X.S. ZnO-Layered double hydroxide@graphitic carbon nitride composite for consecutive adsorption and photodegradation of dyes under UV and visible lights. Materials 2016, 9, 927. [Google Scholar] [CrossRef] [PubMed]
- Leong, K.H.; Liu, S.L.; Sim, L.C.; Saravanana, P.; Jang, M.; Ibrahim, S. Surface reconstruction of titania with g-C3N4 and Ag for promoting efficient electrons migration and enhanced visible light photocatalysis. Appl. Surf. Sci. 2015, 358, 370–376. [Google Scholar] [CrossRef]
- Xu, H.; Wang, C.; Song, Y.; Zhu, J.; Xu, Y.; Yan, J.; Song, Y.; Li, H. CNT/Ag3PO4 composites with highly enhanced visible light photocatalytic activity and stability. Chem. Eng. J. 2014, 241, 35–42. [Google Scholar] [CrossRef]
- Jia, T.K.; Zhao, J.W.; Fu, F.; Deng, Z.; Wang, W.M.; Fu, Z.Y.; Meng, F.C. Synthesis, characterization and photcatalytic activity Zn-doped SnO2/Zn2SnO4 coupled nanocomposites. Int. J. Photoenergy 2014, 2014, 197824. [Google Scholar] [CrossRef]
- Feng, W.; Fang, J.; Zhou, G.; Zhang, L.; Lu, S.; Wu, S.; Chen, Y.; Ling, Y.; Fang, Z. Rationally designed Bi@BiOCl/g-C3N4 heterostructure with exceptional solar-driven photocatalytic activity. Mol. Catal. 2017, 434, 69–79. [Google Scholar] [CrossRef]
- Shan, W.J.; Hu, Y.; Bai, Z.G.; Zheng, M.M.; Wei, C.H. In situ preparation of g-C3N4/bismuth-based oxide nanocomposites with enhanced photocatalytic activity. Appl. Catal. B 2016, 188, 1–12. [Google Scholar] [CrossRef]
- Aghdam, S.M.; Haghighi, M.; Allahyari, S.; Yosefi, L. Precipitation dispersion of various ratios of BiOI/BiOCl nanocomposite over g-C3N4 for promoted visible light nanophotocatalyst used in removal of acid orange 7 from water. J. Photochem. Photobiol. A Chem. 2017, 338, 201–212. [Google Scholar] [CrossRef]
- Li, Y.F.; Fang, L.; Jin, R.X.; Yang, Y.; Fang, X.; Xing, Y.; Song, S.Y. Preparation and enhanced visible light photocatalytic activity of novel g-C3N4 nanosheetsloaded with Ag2CO3 nanoparticles. Nanoscale 2015, 7, 758–764. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Yan, J.; Xu, Y.G.; Song, Y.H.; Li, H.M.; Xia, J.X.; Huang, C.J.; Wan, H.L. Novel visible-light-driven AgX/graphite-like C3N4 (X = Br, I) hybrid materials with synergistic photocatalytic activity. Appl. Catal. B 2013, 129, 182–193. [Google Scholar] [CrossRef]
- Zhao, H.; Dong, Y.M.; Jiang, P.P.; Miao, H.Y.; Wang, G.L.; Zhang, J.J. In situ light-assisted preparation of MoS2 on graphitic C3N4 nanosheets for enhanced photocatalytic H2 production from water. J. Mater. Chem. A 2015, 3, 7375–7381. [Google Scholar] [CrossRef]
- Bai, X.J.; Sun, C.P.; Wu, S.L.; Zhu, Y.F. Enhancement of photocatalytic performance via a P3HT-g-C3N4 heterojunction. J. Mater. Chem. A 2015, 3, 2741–2747. [Google Scholar] [CrossRef]
- Guo, Y.; Li, J.H.; Gao, Z.Q.; Zhu, X.; Liu, Y.; Wei, Z.B.; Zhao, W.; Sun, C. A simple and effective method for fabricating novel p-n heterojuction photocatalyst g-C3N4/Bi4Ti3O12 and its photocatalytic performances. Appl. Catal. B Environ. 2016, 192, 57–71. [Google Scholar] [CrossRef]
- Dang, X.M.; Zhang, X.F.; Chen, Y.T.; Dong, X.L.; Wang, G.W.; Ma, C.; Zhang, X.X.; Ma, H.C.; Xue, M. Preparation of β-Bi2O3/g-C3N4 nanosheet p–n junction for enhanced photocatalytic ability under visible light illumination. J. Nanopart. Res. 2015, 17, 93. [Google Scholar] [CrossRef]
- Fu, J.; Tian, Y.L.; Chang, B.B.; Xi, F.N.; Dong, X.P. BiOBr–carbon nitride heterojunctions: Synthesis, enhanced activity and photocatalytic mechanism. J. Mater. Chem. 2012, 22, 21159–21166. [Google Scholar] [CrossRef]
- Liu, G.G.; Zhao, G.X.; Zhou, W.; Liu, Y.Y.; Pang, H.; Zhang, H.B.; Hao, D.; Meng, X.G.; Li, P.; Kako, T.; et al. In situ bond modulation of graphitic carbon nitride to construct p–n homojunctions for enhanced photocatalytic hydrogen production. Adv. Funct. Mater. 2016, 26, 6822–6829. [Google Scholar] [CrossRef]
- Hu, R.; Wang, X.; Dai, S.; Shao, D.; Hayat, T.A. Alsaedi, Application of graphitic carbon nitride for the removal of Pb(II) and aniline from aqueous solutions. Chem. Eng. J. 2015, 260, 469–477. [Google Scholar] [CrossRef]
- Zhu, T.T.; Song, Y.H.; Ji, H.J.; Xu, Y.G.; Song, Y.X.; Xia, J.X.; Yin, S.Y.; Li, Y.T.; Xu, H.; Zhang, Q.; et al. Synthesis of g-C3N4/Ag3VO4 composites with enhanced photocatalytic activity under visible light irradiation. Chem. Eng. J. 2015, 271, 96–105. [Google Scholar] [CrossRef]
- Li, Y.B.; Zhang, H.M.; Liu, P.R.; Wang, D.; Li, Y.; Zhao, H.J. Cross-linked g-C3N4/rGO nanocomposites with tunable band structure and enhanced visible light photocatalytic activity. Small 2013, 9, 3336–3344. [Google Scholar] [CrossRef] [PubMed]
- Li, C.J.; Wang, S.P.; Wang, T.; Wei, Y.J.; Zhang, P.; Gong, J.L. Monoclinic porous BiVO4 networks decorated by discrete g-C3N4 nano-islands with tunable coverage for highly efficient photocatalysis. Small 2014, 10, 2783–2790. [Google Scholar] [CrossRef] [PubMed]
- Niu, P.; Zhang, L.L.; Liu, G.; Cheng, H.M. Graphene-like carbon nitride nanosheets for improved photocatalytic activities. Adv. Funct. Mater. 2012, 22, 4763–4770. [Google Scholar] [CrossRef]
- Wang, F.; Li, S.F.; Yu, H.G.; Yu, J.G. In situ anion-exchange synthesis and photocatalytic activity of Ag8W4O16/AgCl–nanoparticle core–shell nanorods. J. Mol. Catal. A Chem. 2011, 334, 52–59. [Google Scholar] [CrossRef]
- Zhu, A.; Zhao, Q.; Li, X.; Shi, Y. BiFeO3/TiO2 nanotube arrays composite electrode: Construction, characterization, and enhanced photoelectrochemical properties. ACS Appl. Mater. Interfaces 2014, 6, 671–679. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Jin, R.; Fang, Y.; Yang, Y.; Yang, M.; Liu, X.; Xing, Y. In situ loading of Ag2WO4 on ultrathin nansheets with highly enhanced photocatalytic performance. J. Hazard. Mater. 2016, 313, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.H.; Zhang, L.H.; Lin, H.J.; Nong, Q.Y.; Cui, M.; Wu, Y.; He, Y.M. Fabrication and characterization of hollow CdMoO4 coupled g-C3N4 heterojunction with enhanced photocatalytic activity. J. Harzard. Mater. 2015, 299, 333–342. [Google Scholar] [CrossRef] [PubMed]
- Ke, J.; Liu, J.; Sun, H.; Zhang, H.; Duan, X.; Liang, P.; Li, X.; Tade, M.O.; Liu, S.; Wang, S.B. Facile assembly of Bi2O3 and p-MoS2 for enhanced photocatalytic water oxidation and pollutant degradation. Appl. Catal. B Environ. 2017, 200, 47–55. [Google Scholar] [CrossRef]
- Jia, T.K.; Fu, F.; Long, F.; Min, Z.Y.; Zhao, J.W.; Chen, J.; Li, J.L. Synthesis, characterization and enhanced visible-light photocatalytic activity of Zn2SnO4-C nanocomposites with truncated octahedron morphology. Ceram. Int. 2016, 142, 13893–13899. [Google Scholar] [CrossRef]
- Jia, T.K.; Wang, X.F.; Long, F.; Li, J.L.; Kang, Z.F.; Fu, F.; Sun, G.; Chen, J. Visible-light photocatalytic activities of 3D hierarchical Bi2S3 architectures assembled by nanoplatelets. Crystals 2016, 6, 140. [Google Scholar] [CrossRef]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jia, T.; Li, J.; Long, F.; Fu, F.; Zhao, J.; Deng, Z.; Wang, X.; Zhang, Y. Ultrathin g-C3N4 Nanosheet-Modified BiOCl Hierarchical Flower-Like Plate Heterostructure with Enhanced Photostability and Photocatalytic Performance. Crystals 2017, 7, 266. https://doi.org/10.3390/cryst7090266
Jia T, Li J, Long F, Fu F, Zhao J, Deng Z, Wang X, Zhang Y. Ultrathin g-C3N4 Nanosheet-Modified BiOCl Hierarchical Flower-Like Plate Heterostructure with Enhanced Photostability and Photocatalytic Performance. Crystals. 2017; 7(9):266. https://doi.org/10.3390/cryst7090266
Chicago/Turabian StyleJia, Tiekun, Jili Li, Fei Long, Fang Fu, Junwei Zhao, Zhao Deng, Xiaohui Wang, and Ying Zhang. 2017. "Ultrathin g-C3N4 Nanosheet-Modified BiOCl Hierarchical Flower-Like Plate Heterostructure with Enhanced Photostability and Photocatalytic Performance" Crystals 7, no. 9: 266. https://doi.org/10.3390/cryst7090266



