Solvent-Dependent Thermoelectric Properties of PTB7 and Effect of 1,8-Diiodooctane Additive
Abstract
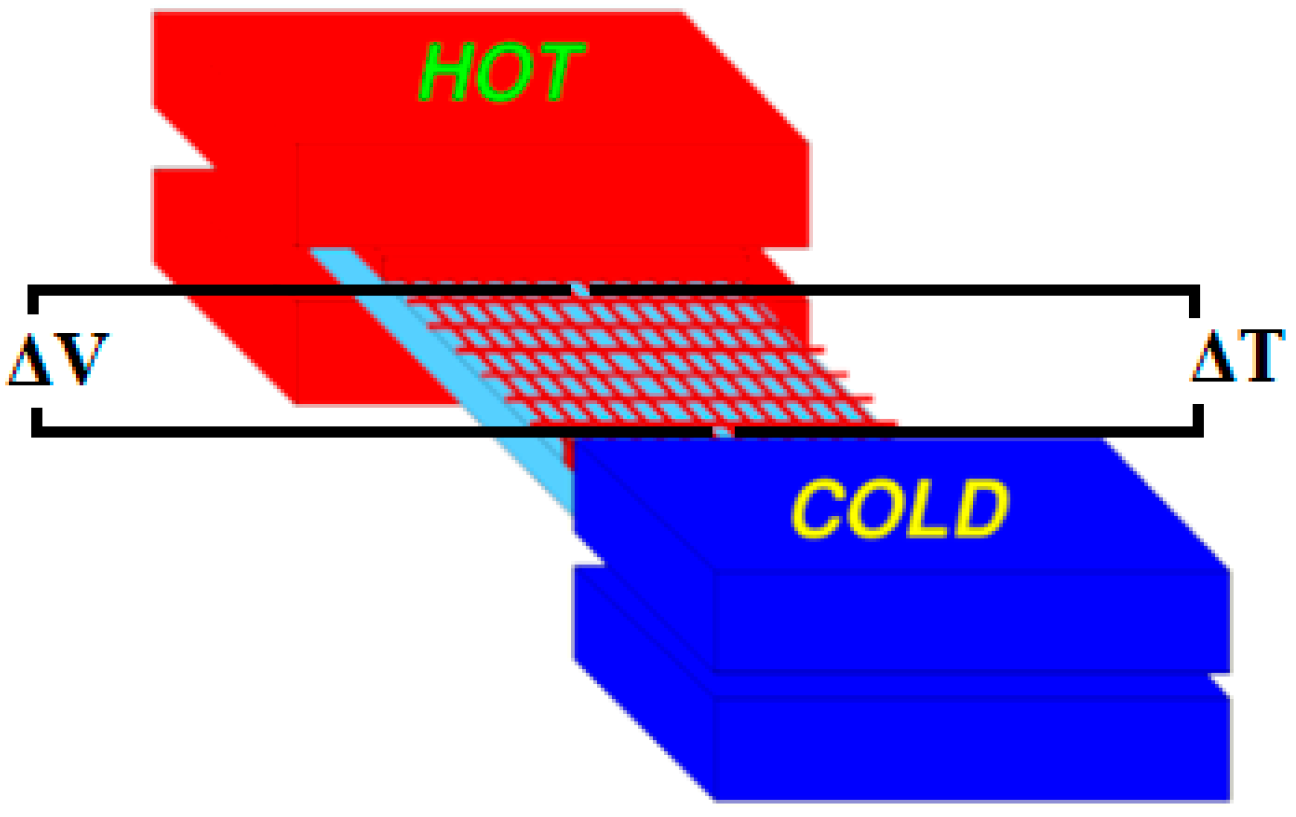
:1. Introduction
2. Materials and Methods
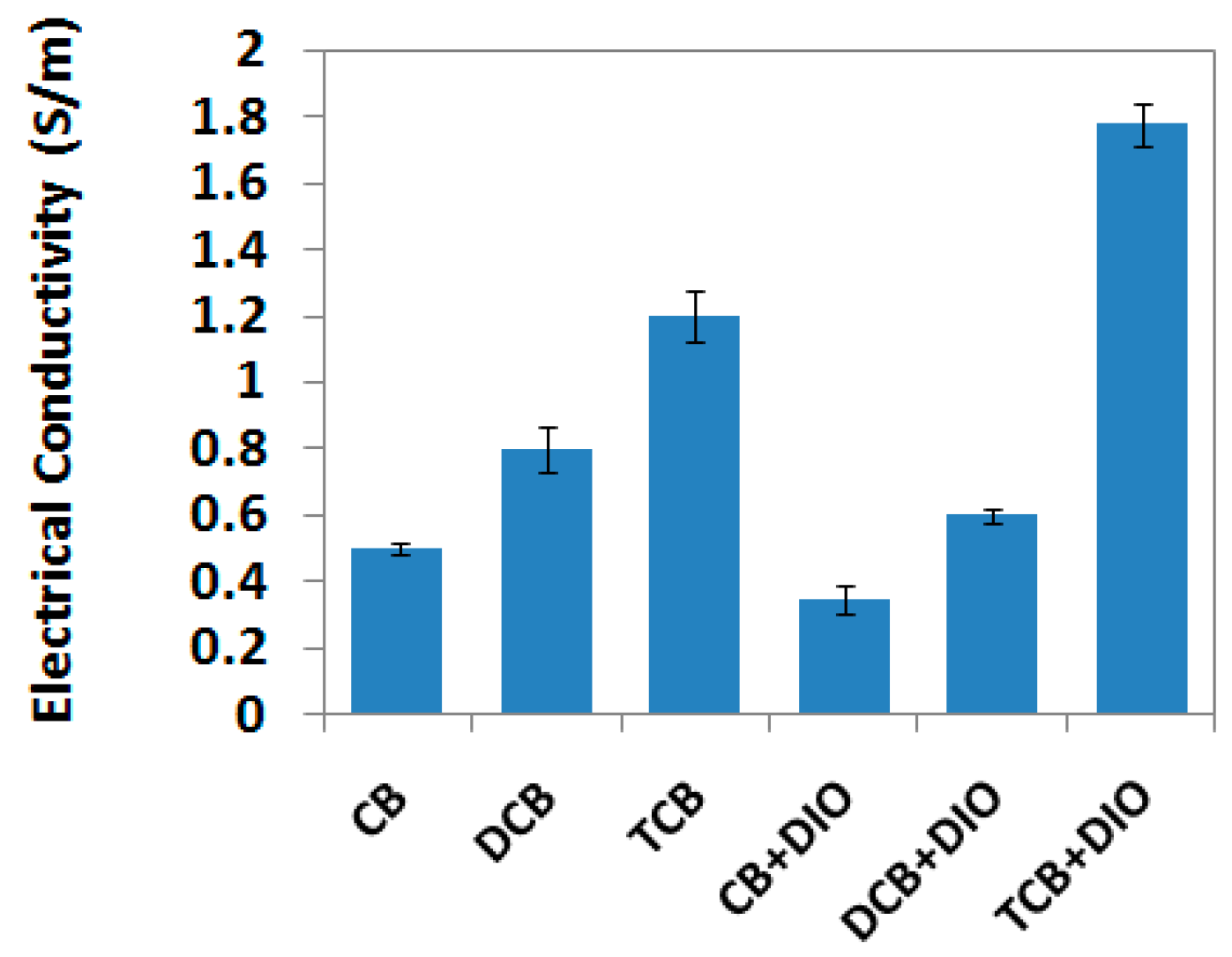
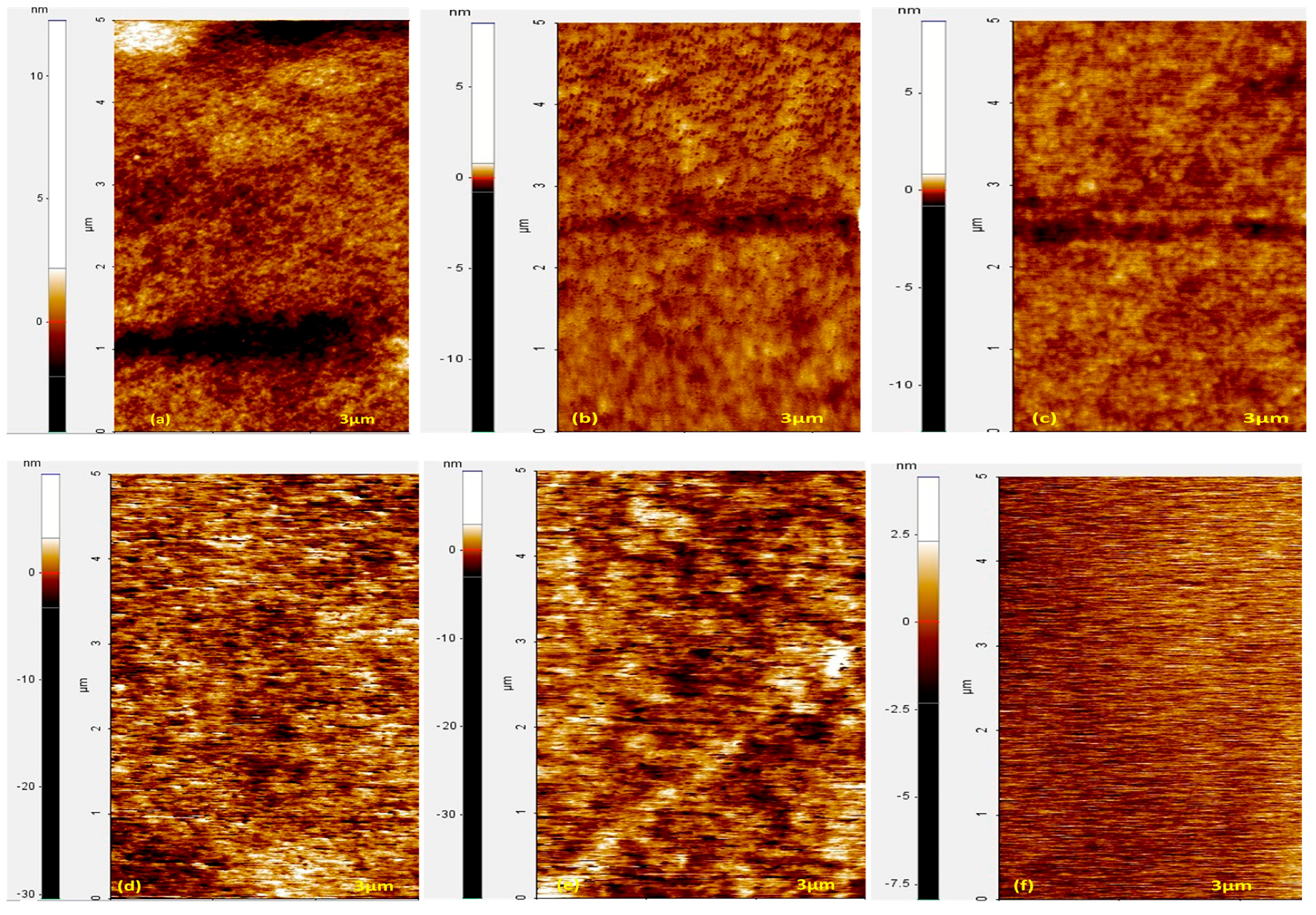
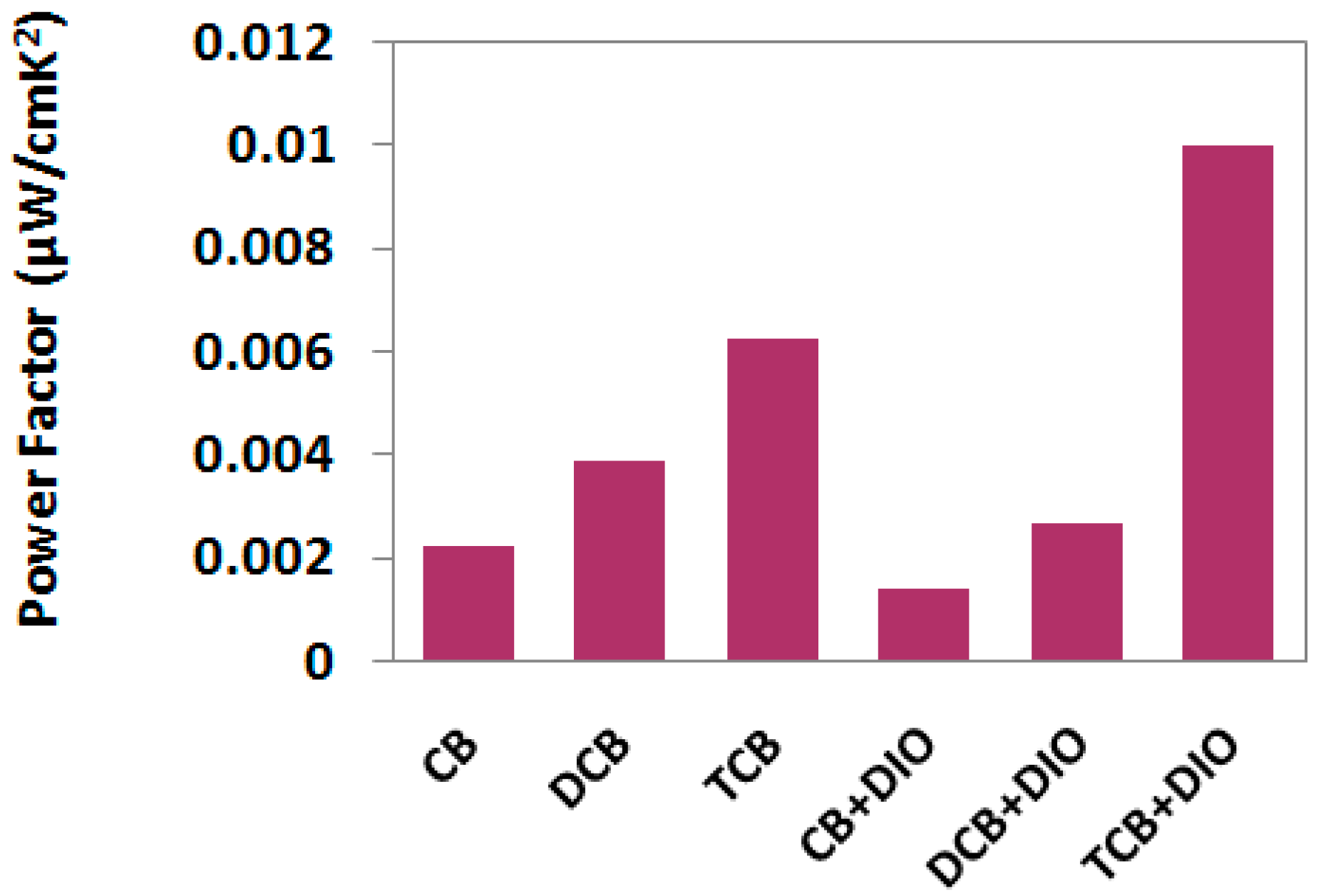
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Poudel, B.; Hao, Q.; Ma, Y.; Lan, Y.; Minnich, A.; Yu, B.; Yan, X.; Wang, D.; Muto, A.; Vashaee, D.; et al. High-Thermoelectric Performance of Nanostructured Bismuth Antimony Telluride Bulk Alloys. Science 2008, 320, 634–638. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Fan, S.; Xiao, N.; Liu, D.; Tay, Y.Y.; Yu, C.; Sim, D.; Hng, H.H.; Zhang, Q.; Boey, F.; et al. Flexible Carbon Nanotube Papers with Improved Thermoelectric Properties. Energy Environ. Sci. 2012, 5, 5364–5369. [Google Scholar] [CrossRef]
- Adroja, N.; Mehta, S.B.; Shah, P. Review of thermoelectricity to improve energy quality. Int. J. Emerg. Technol. Innov. Res. 2015, 2, 847–850. [Google Scholar]
- Sharp, J.; Bierschenk, J.; Lyon, H.B. Overview of Solid-State Thermoelectric Refrigerators and Possible Applications to On-Chip Thermal Management. Proc. IEEE 2006, 94, 1602–1612. [Google Scholar] [CrossRef]
- Bell, L.E. Cooling, heating, generating power, and recovering waste heat with thermoelectric systems. Science 2008, 321, 1457–1461. [Google Scholar] [CrossRef] [PubMed]
- Snyder, G.J.; Toberer, E.S. Complex thermoelectric materials. Nat. Mater. 2008, 7, 105–114. [Google Scholar] [PubMed]
- Karni, J. Solar energy: The thermoelectric alternative. Nat. Mater. 2011, 10, 481–482. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Liu, Y.; Chen, B.; Zhao, C.; Lu, K. Enhancement in thermoelectric properties using a P-type and N-type thin-film device structure. Polym. Compos. 2013, 34, 1728–1734. [Google Scholar] [CrossRef]
- Petsagkourakis, I.; Pavlopoulou, E.; Portale, G.; Kuropatwa, B.A.; Dilhaire, S.; Fleury, G.; Hadziioannou, G. Structurally-driven enhancement of thermoelectric properties within poly 3,4-ethylenedioxythiophene) thin films. Sci. Rep. 2016, 6, 30501. [Google Scholar] [CrossRef] [PubMed]
- Dubey, N.; Leclerc, M. Conducting Polymers: Efficient Thermoelectric Materials. J. Polym. Sci. B Polym. Phys. 2011, 49, 467–475. [Google Scholar]
- Lodha, A.; Singh, R. Prospects of manufacturing organic semiconductor- based integrated circuits. IEEE Trans. Semicond. Manuf. 2001, 14, 281–296. [Google Scholar] [CrossRef]
- Goodson, K.E.; Ju, Y.S. Heat conduction in novel electronic films. Annu. Rev. Mater. Sci. 1999, 29, 261–293. [Google Scholar] [CrossRef]
- Sun, J.; Yeh, M.L.; Jung, B.J.; Zhang, B.; Feser, J.; Majumdar, A.; Katz, H.E. Simultaneous increase in Seebeck coefficient and conductivity in a doped poly (alkylthiophene) blend with defined density of states. Macromolecules 2010, 43, 2897–2903. [Google Scholar] [CrossRef]
- Hewitt, C.A.; Kaiser, A.B.; Roth, S.; Craps, M.; Czerw, R.; Carroll, D.L. Multilayered carbon nanotube/polymer composite based thermoelectric fabrics. Nano Lett. 2012, 12, 1307–1310. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.; Shi, K.; Wu, Y.; Lu, Z.Y.; Liu, H.Y.; Wang, J.Y.; Pei, J. Enhanced molecular packing of a conjugated polymer with high organic thermoelectric power factor. ACS Appl. Mater. Interfaces 2016, 8, 24737–24743. [Google Scholar] [CrossRef] [PubMed]
- Maiz, J.; Rojo, M.M.; Abad, B.; Wilson, A.A.; Nogales, A.; Borca-Tasciuc, D.A.; Borca-Tasciuc, T.; Martín-González, M. Enhancement of thermoelectric efficiency of doped PCDTBT polymer films. RSC Adv. 2015, 5, 66687–66694. [Google Scholar] [CrossRef]
- Mateeva, N.; Niculescu, H.; Schlenoff, J.; Testardi, L.R. Correlation of Seebeck coefficient and electric conductivity in polyaniline and polypyrrole. J. Appl. Phys. 1998, 83, 3111–3117. [Google Scholar] [CrossRef]
- Toshima, N. Conductive polymers as a new type of thermoelectric material. Macromol. Symp. 2002, 186, 81–86. [Google Scholar] [CrossRef]
- Liang, Y.; Xu, Z.; Xia, J.; Tsai, S.; Wu, Y.; Li, G.; Ray, C.; Yu, L. For the bright future-bulk heterojunction polymer solar cells with power conversion efficiency of 7.4%. Adv. Mater. 2010, 22, E135–E138. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Herzig, E.M.; Naumann, A.; Tainter, G.; Perlich, J.; Muller-Buschbaum, P. Influence of solvent and solvent additive on the morphology of PTB7 films probed via X-ray scattering. J. Phys. Chem. B 2014, 118, 344–350. [Google Scholar] [CrossRef] [PubMed]
- Foote, A.L. Investigation of Solvent-Dependent Properties of Donor and Acceptor Materials for Photovoltaic Applications. Available online: http://corescholar.libraries.wright.edu/etd_all/1451/ (accessed on 29 July 2015).
- Xuan, Y.; Liu, X.; Desbief, S.; Leclère, P.; Fahlman, M.; Lazzaroni, R.; Berggren, M.; Cornil, J.; Emin, D.; Crispin, X. Thermoelectric properties of conducting polymers: The case of poly(3-hexylthiophene). Phys. Rev. B 2010, 82, 115454. [Google Scholar] [CrossRef]
- Gao, F.; Liu, Y.; Xiong, Y.; Wu, P.; Hu, B.; Xu, L. Fabricate organic thermoelectric modules use modified PCBM and PEDOT:PSS materials. Front. Optoelectron. 2017, 10, 117–123. [Google Scholar] [CrossRef]
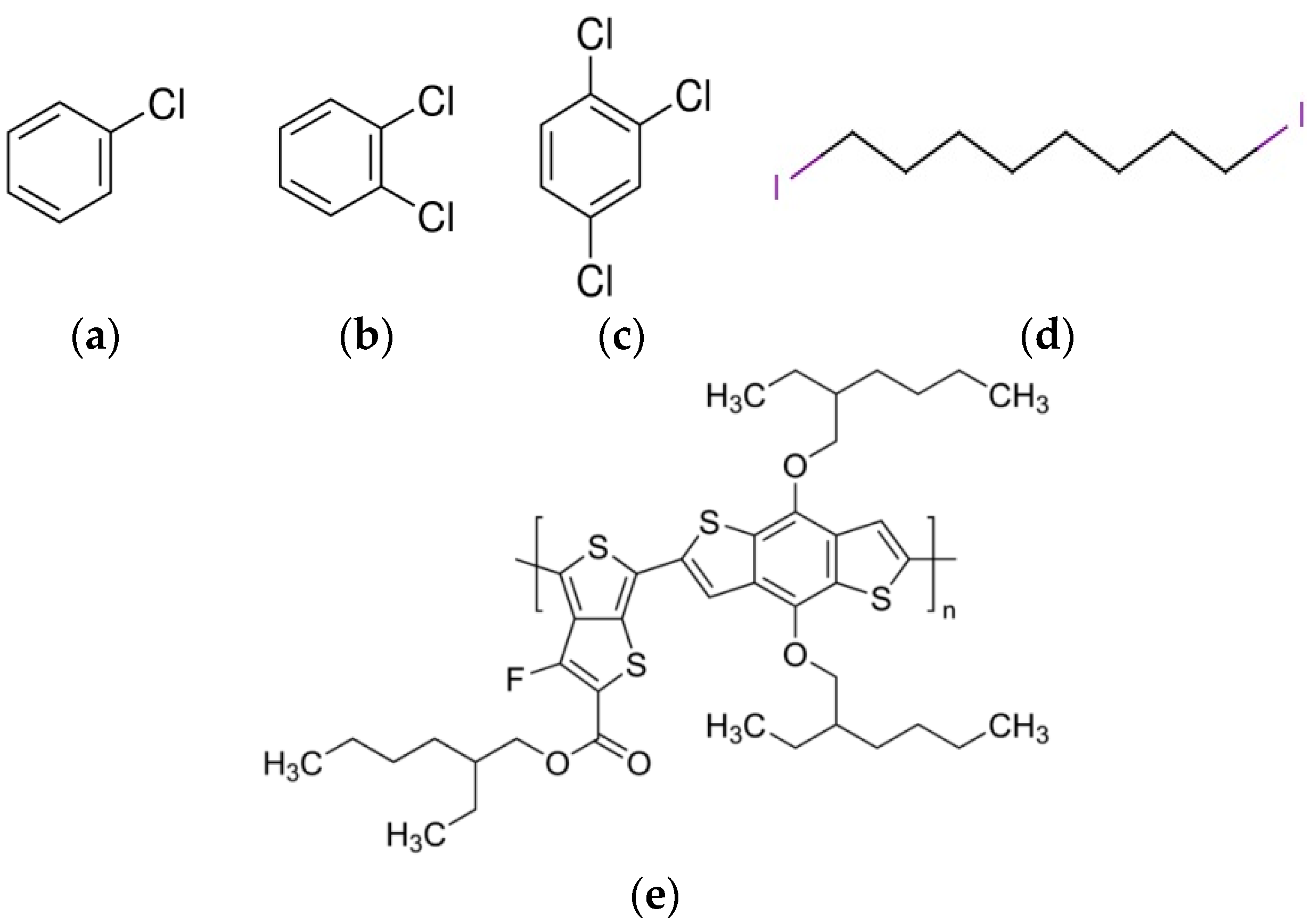

| Material | Boiling Point (°C) |
|---|---|
| 1,2,4-Trichlorobenzene (C6H3Cl3) | 214 |
| 1,2-Dichlorobenzene (C6H4Cl2) | 180 |
| Chlorobenzene (C6H5Cl) | 131 |
| 1,8-Diiodooctane(C8H16I2) | 167–169 |
| Sample | Mobility (cm2/Vs) |
|---|---|
| PTB7: TCB | 1.09 × 10−1 |
| PTB7: TCB + DIO | 1.69 × 10−1 |
| PTB7: DCB | 7.15 × 10−2 |
| PTB7: DCB + DIO | 5 × 10−2 |
| PTB7: CB | 3.9 × 10−2 |
| PTB7: CB + DIO | 2.66 × 10−2 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rastegaralam, M.; Lee, C.; Dettlaff-Weglikowska, U. Solvent-Dependent Thermoelectric Properties of PTB7 and Effect of 1,8-Diiodooctane Additive. Crystals 2017, 7, 292. https://doi.org/10.3390/cryst7100292
Rastegaralam M, Lee C, Dettlaff-Weglikowska U. Solvent-Dependent Thermoelectric Properties of PTB7 and Effect of 1,8-Diiodooctane Additive. Crystals. 2017; 7(10):292. https://doi.org/10.3390/cryst7100292
Chicago/Turabian StyleRastegaralam, Mina, Changhee Lee, and Urszula Dettlaff-Weglikowska. 2017. "Solvent-Dependent Thermoelectric Properties of PTB7 and Effect of 1,8-Diiodooctane Additive" Crystals 7, no. 10: 292. https://doi.org/10.3390/cryst7100292






