Polyoxometalate (POM)-Layered Double Hydroxides (LDH) Composite Materials: Design and Catalytic Applications
Abstract
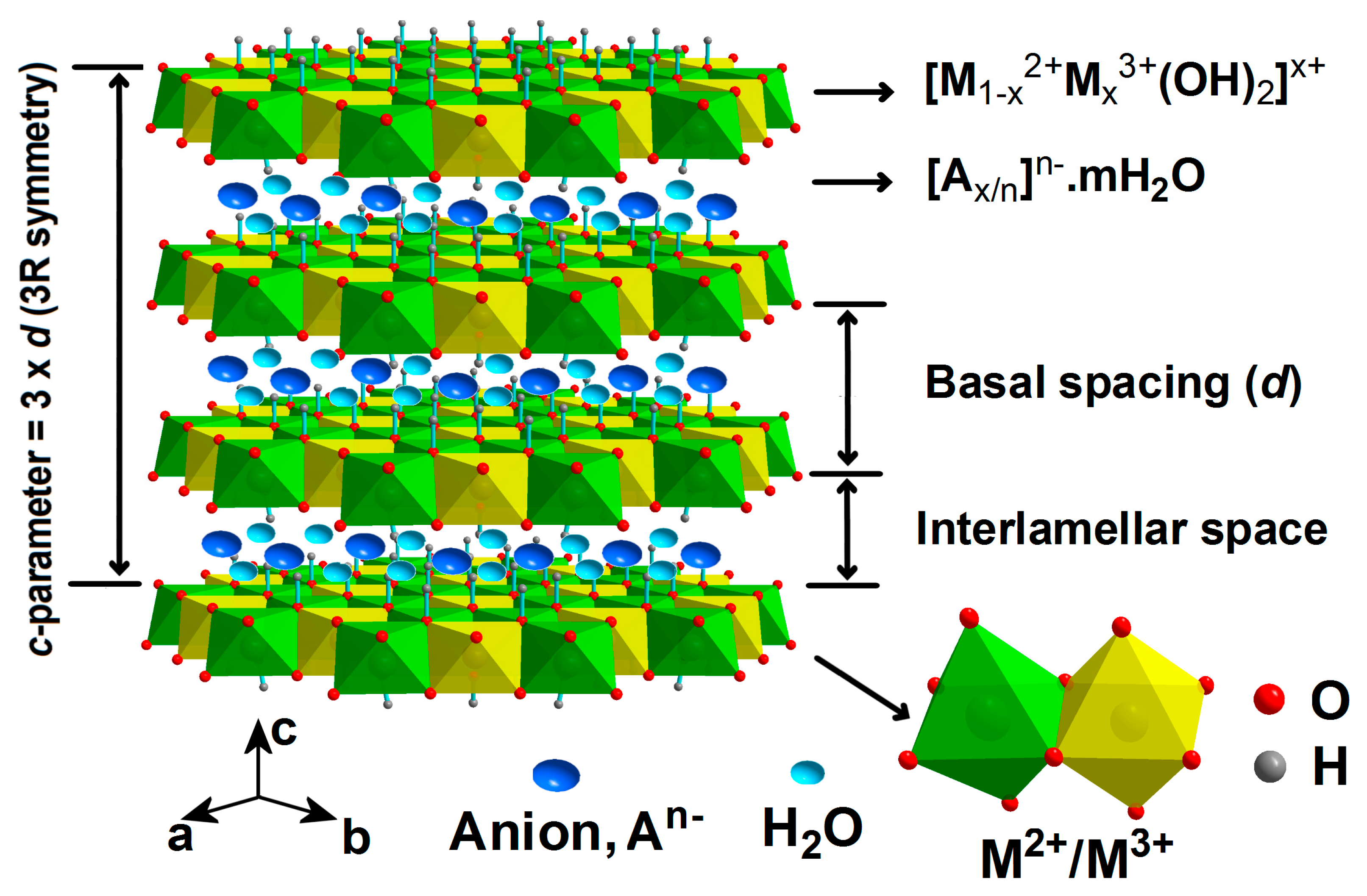
:1. Layered Double Hydroxides (LDHs)-Intro Background
2. LDH-Based Composite Materials
3. POM-LDH Composite Materials
3.1. Synthesis of POM/LDH Composites
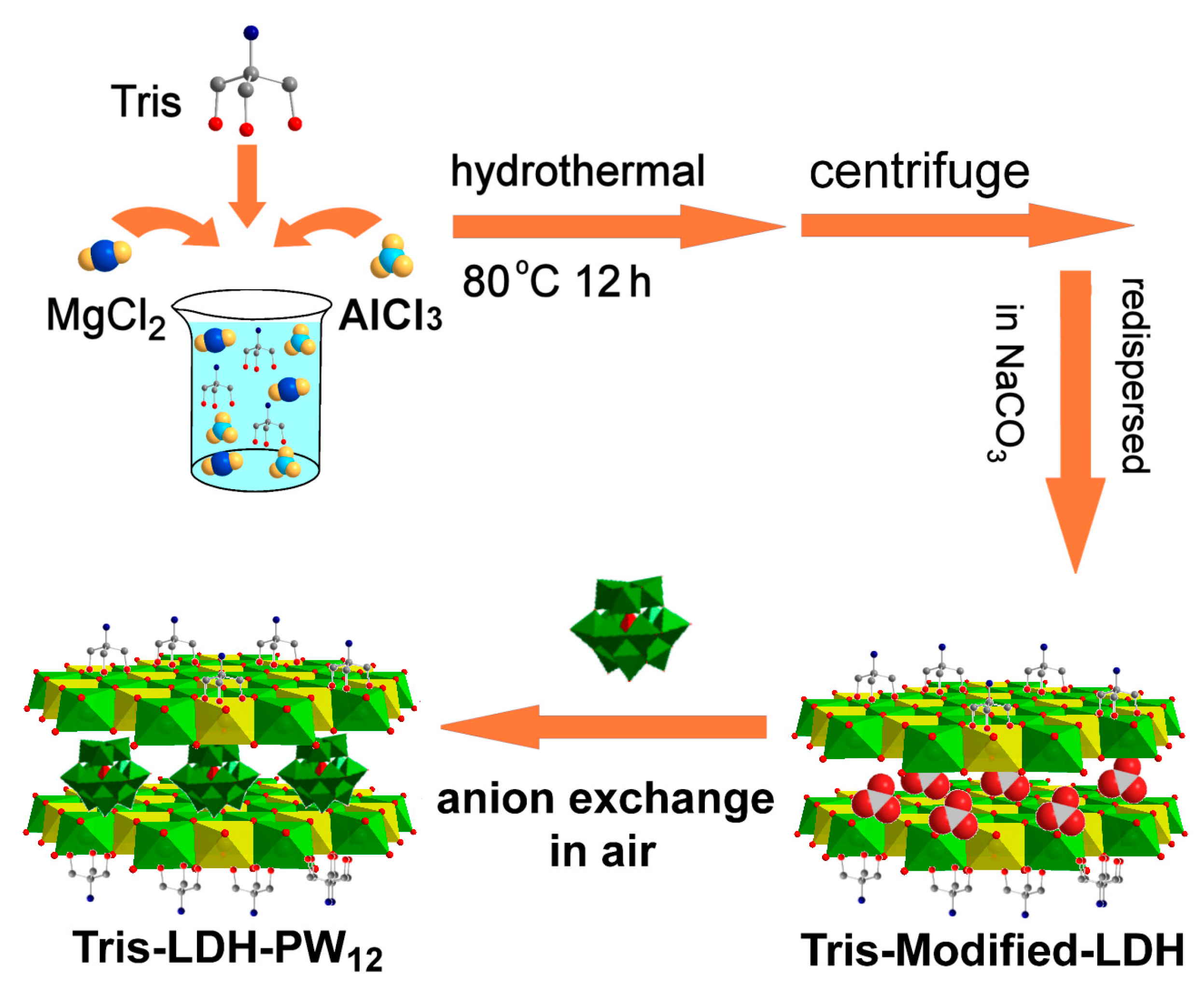
3.1.1. Host Layer Modification Method
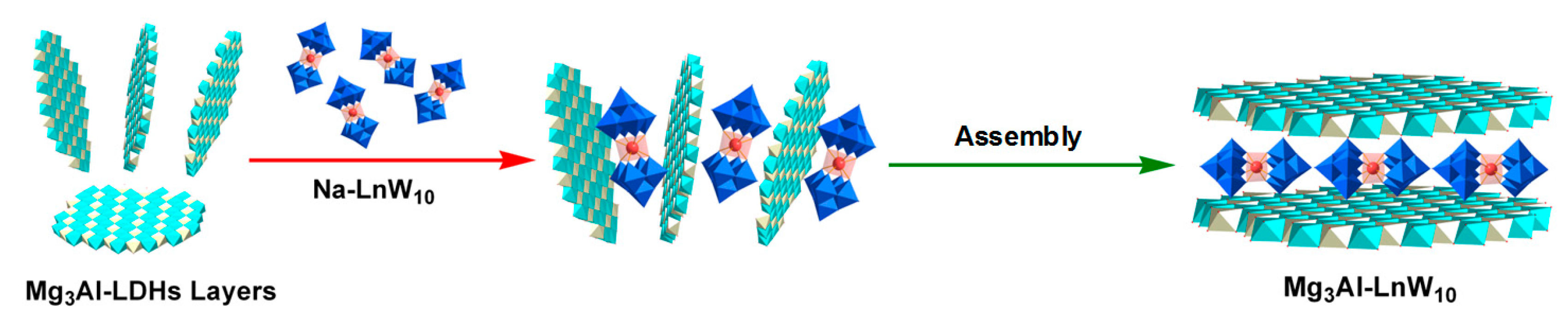
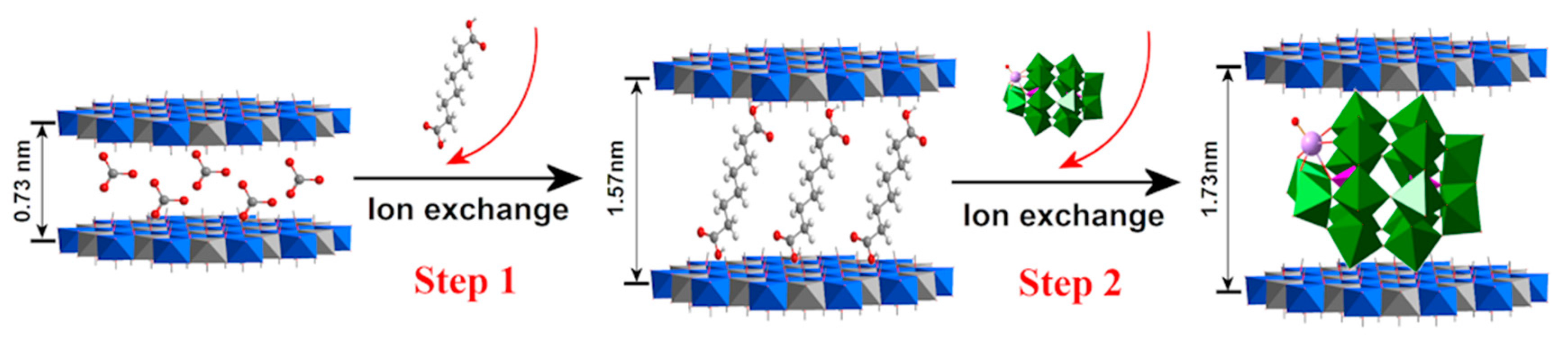
3.1.2. Exfoliation Assembly Method
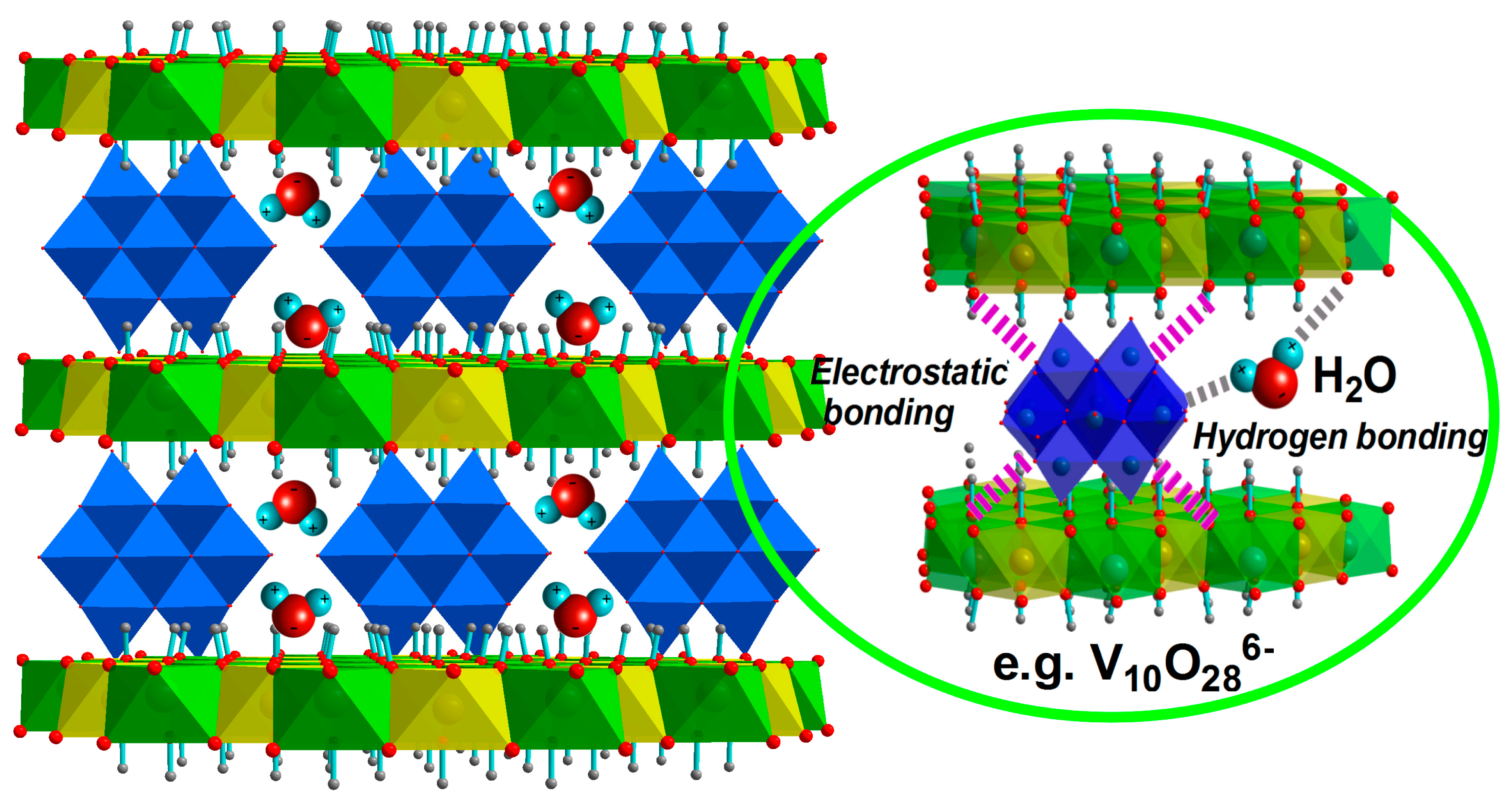
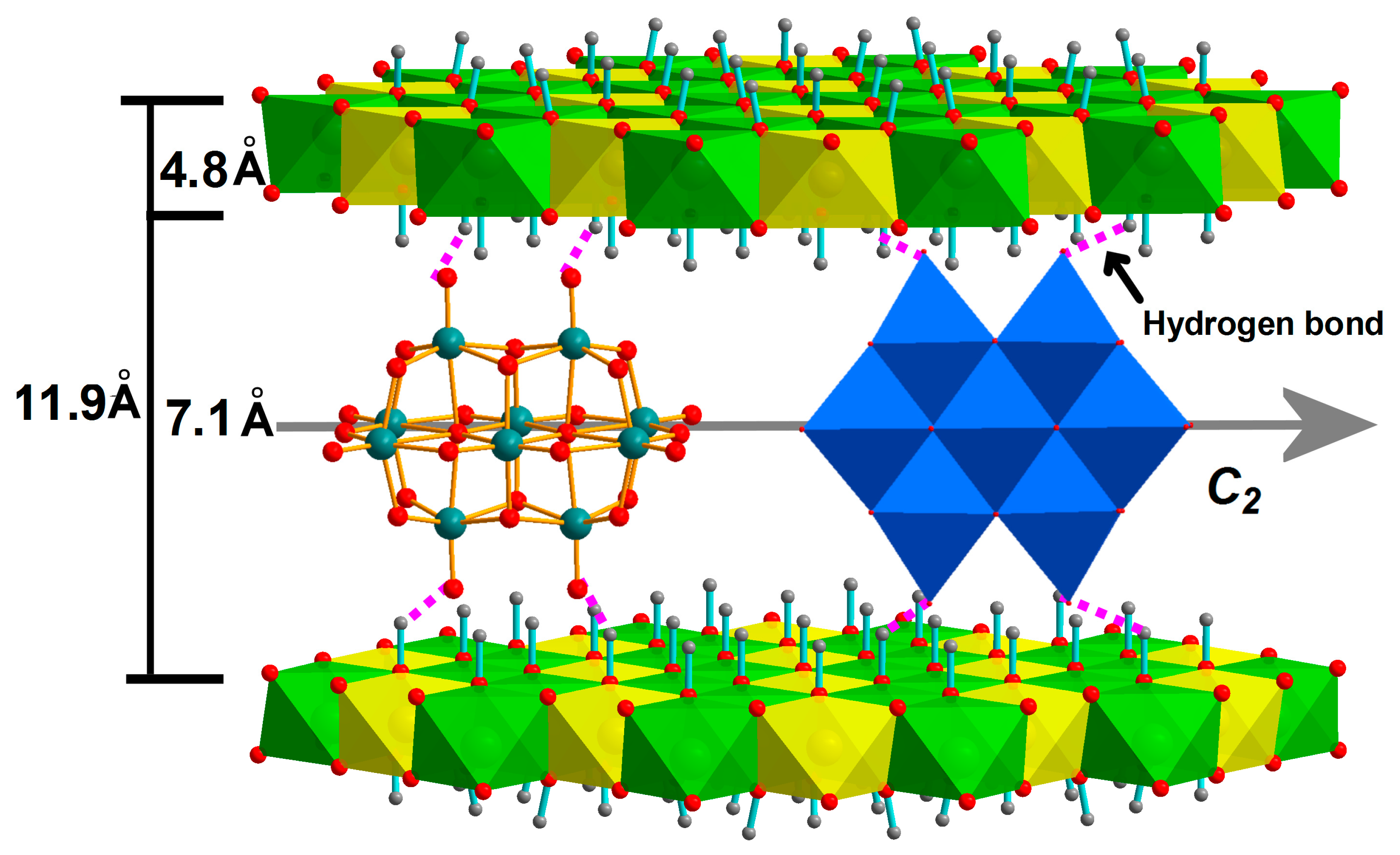
3.2. Structural Features
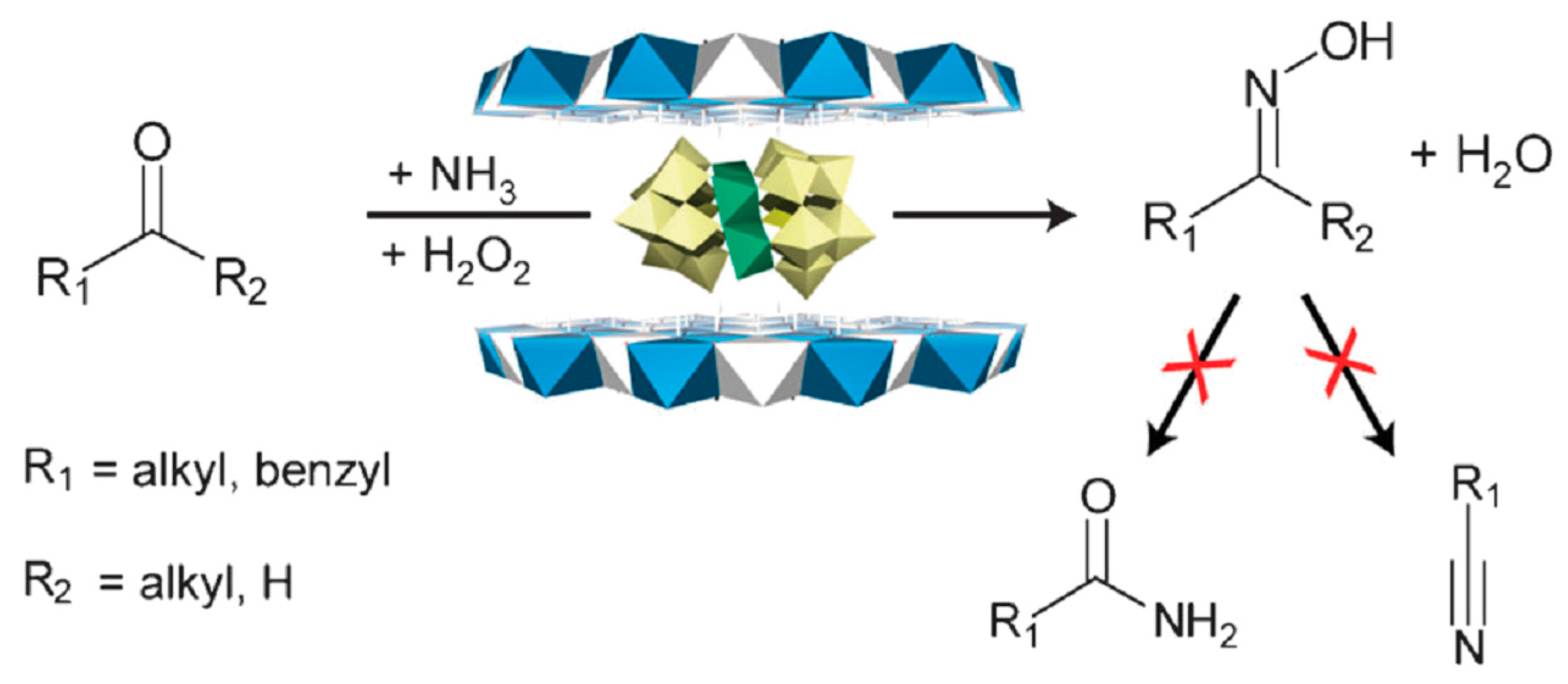
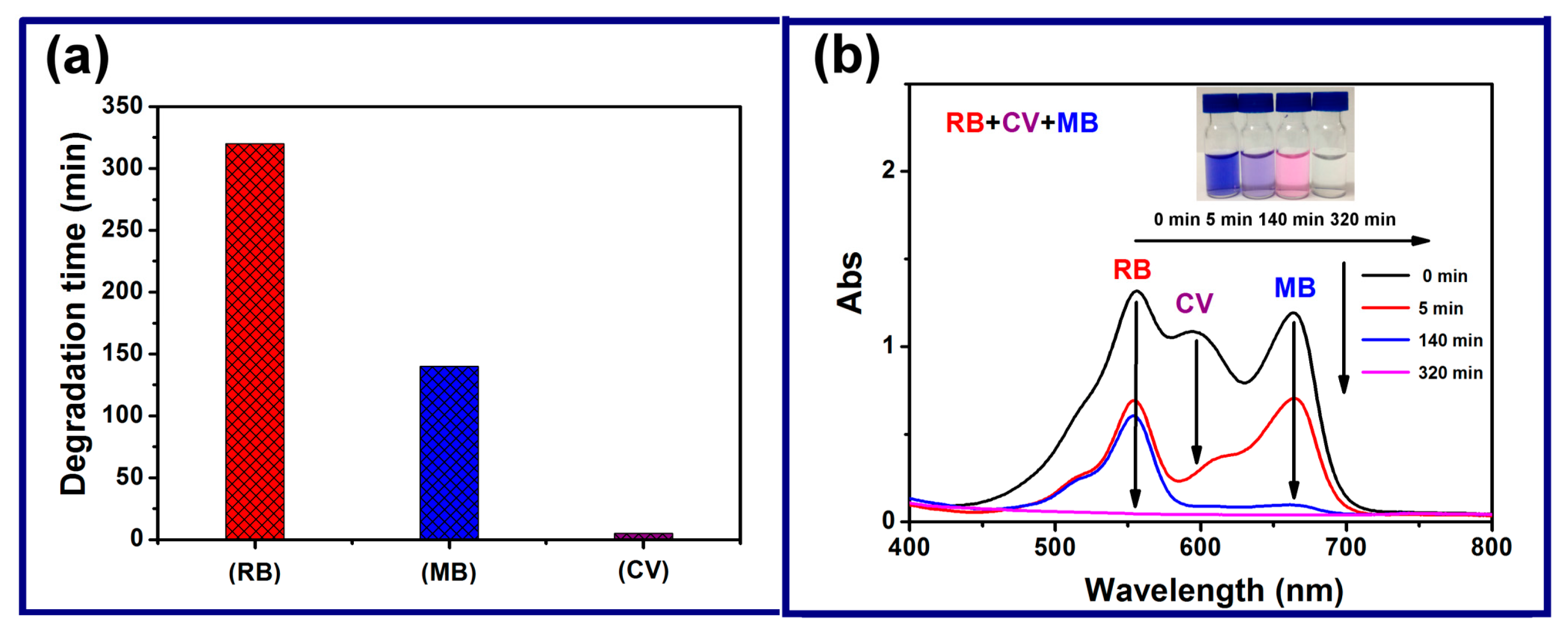
3.3. Oxidation Catalyst
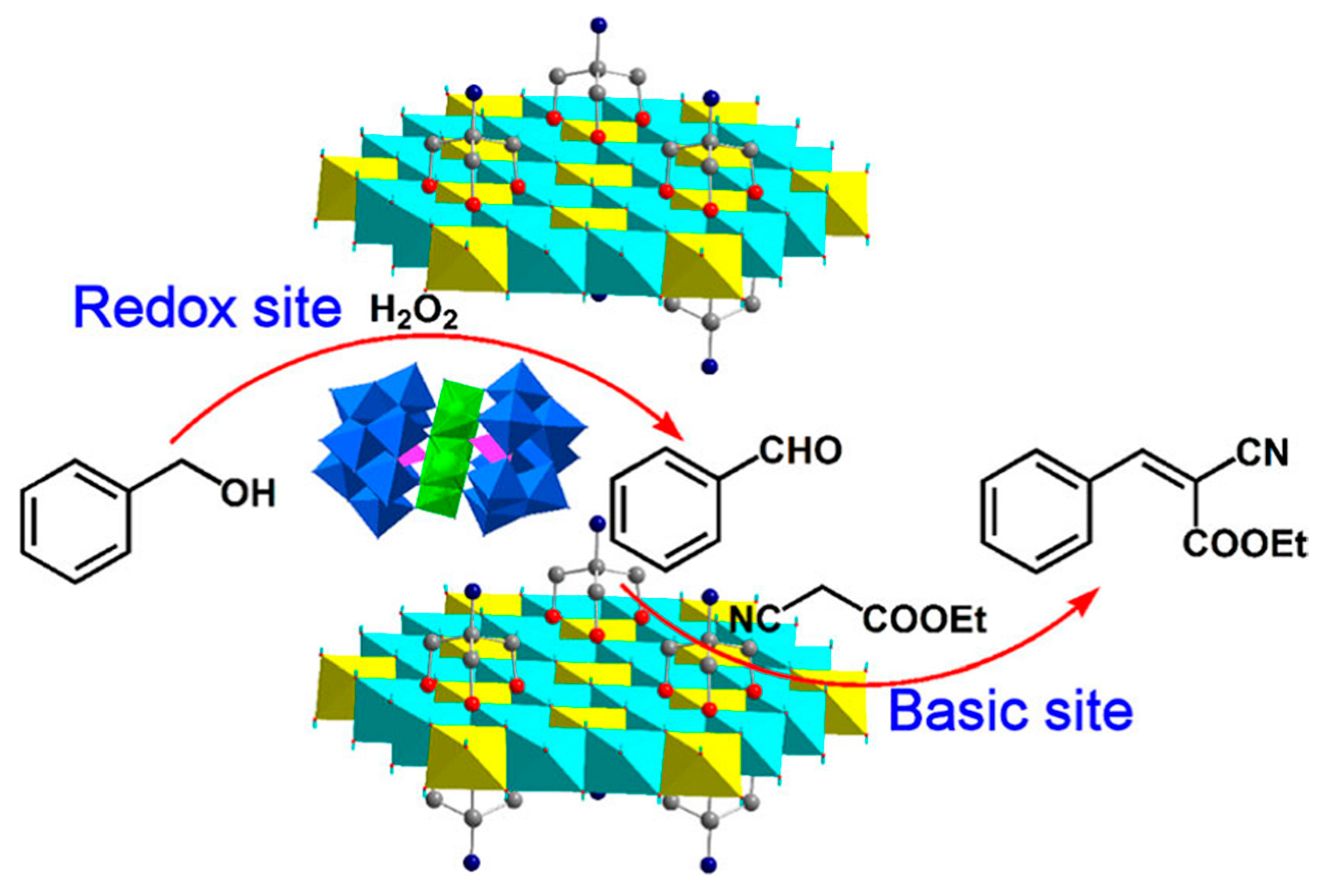
3.4. Acid-Base Synergistic Catalyst
4. Conclusions-Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fan, G.L.; Li, F.; Evans, D.G.; Duan, X. Catalytic applications of layered double hydroxides: Recent advances and perspectives. Chem. Soc. Rev. 2014, 43, 7040–7066. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; O’Hare, D. Recent Advances in the Synthesis and Application of Layered Double Hydroxide (LDH) Nanosheets. Chem. Rev. 2012, 112, 4124–4155. [Google Scholar] [CrossRef] [PubMed]
- Sideris, P.J.; Nielsen, U.G.; Gan, Z.; Grey, C.P. Mg/Al Ordering in Layered Double Hydroxides Revealed by Multinuclear NMR Spectroscopy. Science 2008, 321, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.J.; Tang, Z.Y. Ultrathin two-dimensional layered metal hydroxides: An emerging platform for advanced catalysis, energy conversion and storage. Chem. Soc. Rev. 2016, 45, 4873–4891. [Google Scholar] [CrossRef] [PubMed]
- Williams, G.R.; Khan, A.I.; O’Hare, D. Mechanistic and Kinetic Studies of Guest Ion Intercalation into Layered Double Hydroxides Using Time-resolved, in situ X-ray Powder Diffraction. Struct. Bond. 2006, 119, 161–192. [Google Scholar]
- Evans, D.G.; Slade, R.C.T. Structural Aspects of Layered Double Hydroxides. Struct. Bond. 2006, 119, 1–87. [Google Scholar]
- Zhao, S.; Xu, J.H.; Wei, M.; Song, Y.-F. Synergistic catalysis by polyoxometalate-intercalated layered double hydroxides: Oximation of aromatic aldehydes with large enhancement of selectivity. Green Chem. 2011, 13, 384–389. [Google Scholar] [CrossRef]
- Guo, X.X.; Zhang, F.Z.; Xu, S.L.; Evans, D.G.; Duan, X. Preparation of layered double hydroxide films with different orientations on the opposite sides of a glass substrate by in situ hydrothermal crystallization. Chem. Commun. 2009, 28, 6836–6838. [Google Scholar] [CrossRef] [PubMed]
- Evans, D.G.; Duan, X. Preparation of layered double hydroxides and their applications as additives in polymers, as precursors to magnetic materials and in biology and medicine. Chem. Commun. 2006, 485–496. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.P.; Stevenson, G.S.; Lu, C.Q.; Lu, G.Q.; Bartlett, P.F.; Gray, P.P. Stable Suspension of Layered Double Hydroxide Nanoparticles in Aqueous Solution. J. Am. Chem. Soc. 2006, 128, 36–37. [Google Scholar] [CrossRef] [PubMed]
- Rives, V.; Ulibarri, M.A. Layered double hydroxides (LDH) intercalated with metal coordination compounds and oxometalates. Coord. Chem. Rev. 1999, 181, 61–120. [Google Scholar] [CrossRef]
- Omwoma, S.; Chen, W.; Tsunashima, R.; Song, Y.-F. Recent advances on polyoxometalates intercalated layered double hydroxides: From synthetic approaches to functional material applications. Coord. Chem. Rev. 2014, 258–259, 58–71. [Google Scholar] [CrossRef]
- Duan, X.; Zhang, F.Z. Intercalation Assembly and Functional Materials; Chemical Industry Press: Beijing, China, 2007. [Google Scholar]
- Theiss, F.L.; Ayoko, G.A.; Frost, R.L. Synthesis of layered double hydroxides containing MgII, ZnII, CaII and AlIII layer cations by co-precipitation methods—A review. Appl. Surf. Sci. 2016, 383, 200–213. [Google Scholar] [CrossRef]
- Li, K.W.; Kumada, N.; Yonesaki, Y.; Takei, T.; Kinomura, N.; Wang, H.; Wang, C. The pH effects on the formation of Ni/Al nitrate form layered double hydroxides (LDHs) by chemical precipitation and hydrothermal method. Mater. Chem. Phys. 2010, 121, 223–229. [Google Scholar] [CrossRef]
- Inayat, A.; Klumpp, M.; Schwieger, W. The urea method for the direct synthesis of ZnAl layered double hydroxides with nitrate as the interlayer anion. Appl. Clay Sci. 2011, 51, 452–459. [Google Scholar] [CrossRef]
- Qu, J.; Zhang, Q.W.; Li, X.W.; He, X.M.; Song, S.X. Mechanochemical approaches to synthesize layered double hydroxides: A review. Appl. Clay Sci. 2016, 119, 185–192. [Google Scholar] [CrossRef]
- Zubaira, M.; Daud, M.; McKay, G.; Shehzad, F.; Al-Harthi, M.A. Recent progress in layered double hydroxides (LDH)-containing hybrids as adsorbents for water remediation. Appl. Clay Sci. 2017, 143, 279–292. [Google Scholar] [CrossRef]
- Bi, X.; Zhang, H.; Dou, L.G. Layered Double Hydroxide-Based Nanocarriers for Drug Delivery. Pharmaceutics 2014, 6, 298–332. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.J.; Li, D.Q.; Wang, Y.; Evans, D.G.; Duan, X. Synthesis and characterization of a UV absorbent-intercalated Zn-Al layered double hydroxide. Polym. Degrad. Stab. 2006, 91, 789–794. [Google Scholar] [CrossRef]
- Wang, X.; Sumboja, A.; Lin, M.F.; Yan, J.; Lee, P.S. Enhancing electrochemical reaction sites in nickel–cobalt layered double hydroxides on zinc tin oxide nanowires: A hybrid material for an asymmetric supercapacitor device. Nanoscale 2012, 4, 7266–7272. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Song, Y.-F. Highly Selective and Efficient Removal of Cr(VI) and Cu(II) by the Chromotropic Acid-Intercalated Zn–Al Layered Double Hydroxides. Ind. Eng. Chem. Res. 2013, 52, 4436–4442. [Google Scholar] [CrossRef]
- Kuroda, Y.; Miyamoto, Y.; Hibino, M.; Yamaguchi, K.; Mizuno, N. Tripodal Ligand-Stabilized Layered Double Hydroxide Nanoparticles with Highly Exchangeable CO32−. Chem. Mater. 2013, 25, 2291–2296. [Google Scholar] [CrossRef]
- Kuroda, Y.; Koichi, T.; Muramatsu, K.; Yamaguchi, K.; Mizuno, N.; Shimojima, A.; Wada, H.; Kuroda, K. Direct Synthesis of Highly Designable Hybrid Metal Hydroxide Nanosheets by Using Tripodal Ligands as One-Size-Fits-All Modifiers. Chem. Eur. J. 2017, 23, 5023–5032. [Google Scholar] [CrossRef] [PubMed]
- Oka, Y.; Kuroda, Y.; Matsuno, T.; Kamata, K.; Wada, H.; Shimojima, A.; Kuroda, K. Preparation of Mesoporous Basic Oxides through Assembly of Monodispersed Mg–Al Layered Double Hydroxide Nanoparticles. Chem. Eur. J. 2017, 23, 9362–9368. [Google Scholar] [CrossRef] [PubMed]
- Pinnavaia, T.J.; Chibwe, M.; Constantino, V.R.L.; Yun, S.K. Organic chemical conversions catalyzed by intercalated layered double hydroxides (LDHs). Appl. Clay Sci. 1995, 10, 117–129. [Google Scholar] [CrossRef]
- Tatsumi, T.; Yamamoto, K.; Tajima, H.; Tominaga, H.-O. Shape selective epoxidation of alkenes catalysed by polyoxometalate-intercalated hydrotalcite. Chem. Lett. 1992, 21, 815–818. [Google Scholar] [CrossRef]
- Song, Y.-F.; Tsunashima, R. Recent advances on polyoxometalate-based molecular and composite materials. Chem. Soc. Rev. 2012, 41, 7384–7402. [Google Scholar] [CrossRef] [PubMed]
- Miras, H.N.; Vilà-Nadal, L.; Cronin, L. Polyoxometalate based open-frameworks (POMOFs). Chem. Soc. Rev. 2014, 43, 5679–5699. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.S.; Yang, G.Y. Recent advances in polyoxometalate-catalyzed reactions. Chem. Rev. 2015, 115, 4893–4962. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Guo, Z.J.; Hou, W.; Wang, Q.; Wang, J. Polyoxometalate-based phase transfer catalysis for liquid–solid organic reactions: A review. Catal. Sci. Technol. 2015, 5, 4324–4335. [Google Scholar] [CrossRef]
- Yamaguchi, K.; Yoshida, C.; Uchida, S.; Mizuno, N. Peroxotungstate immobilized on ionic liquid-modified silica as a heterogeneous epoxidation catalyst with hydrogen peroxide. J. Am. Chem. Soc. 2005, 127, 530–531. [Google Scholar] [CrossRef] [PubMed]
- Inumaru, K.; Ishihara, T.; Kamiya, Y.; Okuhara, T.; Yamanaka, S. Water-tolerant, highly active solid acid catalysts composed of the Keggin-type polyoxometalate H3PW12O40 ιmmobilized in hydrophobic nanospaces of organomodified mesoporous silica. Angew. Chem. Int. Ed. 2007, 46, 7625–7628. [Google Scholar] [CrossRef] [PubMed]
- Kwon, T.; Tsigdinos, G.A.; Pinnavaia, T.J. Pillaring of Layered Double Hydroxides (LDH’s) by Polyoxometalate Anions. J. Am. Chem. Soc. 1988, 110, 3653–3654. [Google Scholar] [CrossRef]
- Yun, S.K.; Pinnavaia, T.J. Layered Double Hydroxides Intercalated by Polyoxometalate Anions with Keggin (α-H2W12O406−), Dawson (α-P2W18O626−), and Finke (Co4(H2O)2(PW9O34)210−) Structures. Inorg. Chem. 1996, 35, 6853–6860. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Yao, Z.X.; Song, Y.-F. Polyoxometalates Hosted in Layered Double Hydroxides: Highly Enhanced Catalytic Activity and Selectivity in Sulfoxidation of Sulfides. Ind. Eng. Chem. Res. 2015, 54, 9133–9141. [Google Scholar] [CrossRef]
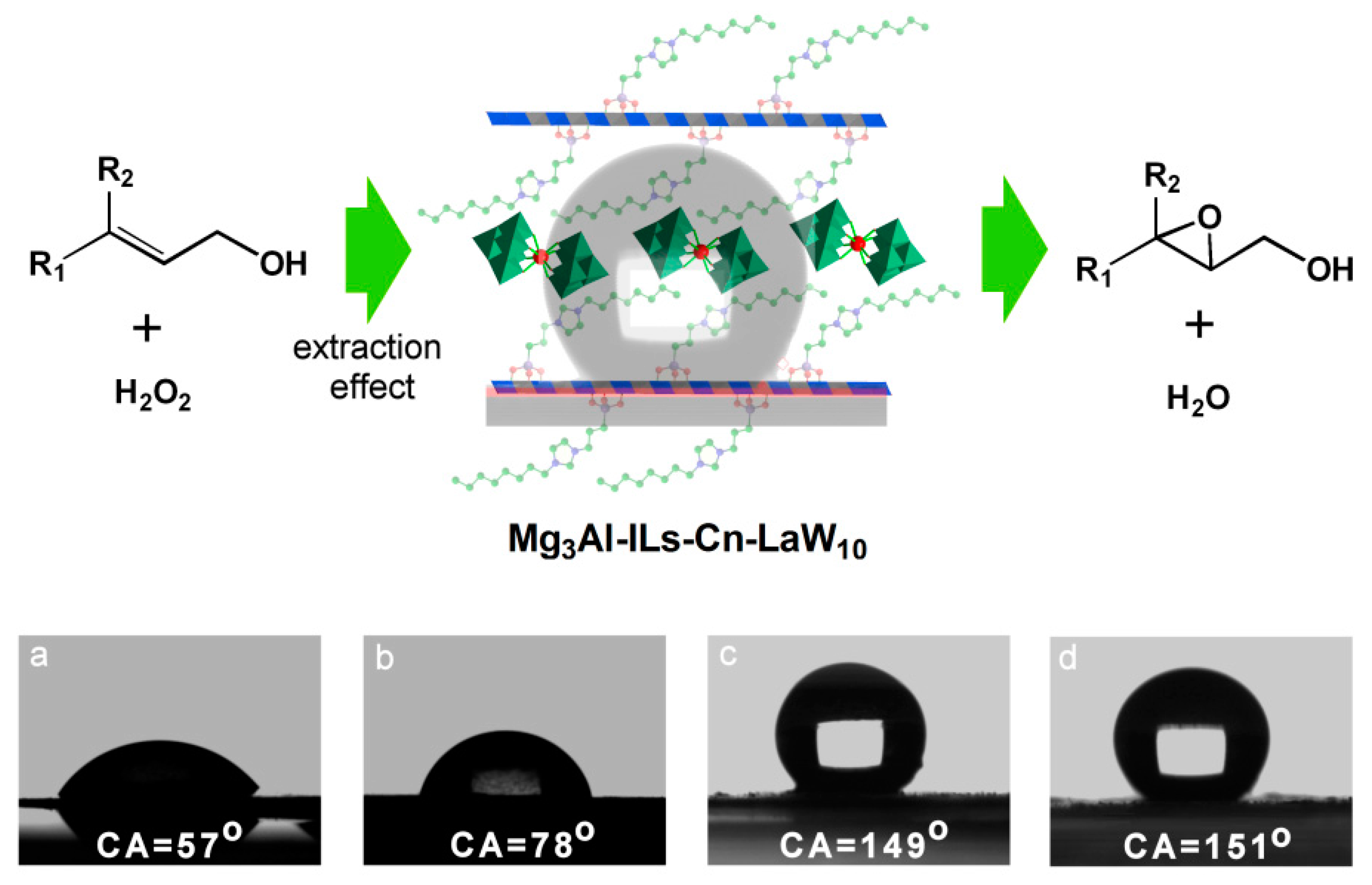
- Liu, P.; Wang, C.H.; Li, C. Epoxidation of allylic alcohols on self-assembled polyoxometalates hosted in layered double hydroxides with aqueous H2O2 as oxidant. J. Catal. 2009, 262, 159–168. [Google Scholar] [CrossRef]
- Narita, E.; Kaviratna, P.D.; Pinnavaia, T.J. Direct synthesis of a polyoxometallate-pillared layered double hydroxide by coprecipitation. J. Chem. Soc. Chem. Commun. 1993, 60–62. [Google Scholar] [CrossRef]
- Jana, S.K.; Kubota, Y.; Takashi, T. Cobalt-substituted polyoxometalate pillared hydrotalcite: Synthesis and catalysis in liquid-phase oxidation of cyclohexanol with molecular oxygen. J. Catal. 2008, 255, 40–47. [Google Scholar] [CrossRef]
- Li, T.F.; Wang, Z.L.; Xu, Y.Q.; Wei, C.; Song, Y.-F. Recent progress in polyoxometalate-intercalated layered double hydroxides composite materials. Sci. China Chem. 2017, 47, 451–464. [Google Scholar] [CrossRef]
- Chen, Y.; Yan, D.P.; Song, Y.-F. Tris(hydroxymethyl)aminomethane modified layered double hydroxides greatly facilitate polyoxometalate intercalation. Dalton Trans. 2014, 43, 14570–14576. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.; Kwag, J.; Jeong, S.; Baek, Y.; Kim, S. Highly Fluorescent and Stable Quantum Dot-Polymer-Layered Double Hydroxide Composites. Chem. Mater. 2013, 25, 1071–1077. [Google Scholar] [CrossRef]
- Gunjakar, J.L.; Kim, T.W.; Kim, H.N.; Kim, I.Y.; Hwang, S.-J. Mesoporous Layer-by-Layer Ordered Nanohybrids of Layered Double Hydroxide and Layered Metal Oxide: Highly Active Visible Light Photocatalysts with Improved Chemical Stability. J. Am. Soc. Chem. 2011, 133, 14998–15007. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.Q.; Zhao, S.; Song, Y.-F. The application of spontaneous flocculation for the preparation of lanthanide-containing polyoxometalates intercalated layered double hydroxides: Highly efficient heterogeneous catalysts for cyanosilylation. Appl. Catal. A Gen. 2014, 487, 172–180. [Google Scholar] [CrossRef]
- Kuo, W.G. Decolorizing dye wastewater with Fenton’s reagent. Water Res. 1992, 26, 881–886. [Google Scholar] [CrossRef]
- Chen, Y.; Yao, Z.X.; Miras, H.N.; Song, Y.-F. Modular Polyoxometalate-Layered Double Hydroxide Composites as Efficient Oxidative Catalysts. Chem. Eur. J. 2015, 21, 10812–10820. [Google Scholar] [CrossRef] [PubMed]
- Jiang, B.; Yang, H.W.; Zhang, L.H.; Zhang, R.Y.; Sun, Y.L.; Huang, Y. Efficient oxidative desulfurization of diesel fuel using amide-based ionic liquids. Chem. Eng. J. 2016, 283, 89–96. [Google Scholar] [CrossRef]
- Xu, Y.Q.; Xuan, W.M.; Zhang, M.M.; Miras, H.N.; Song, Y.-F. A multicomponent assembly approach for the design of deep desulfurization heterogeneous catalysts. Dalton Trans. 2016, 45, 19511–19518. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.D.; Moulijn, J.A.; Ito, E.; Wagemans, R.; Makkee, M. Deep Desulfurization of Fossil Fuels by Air in the Absence of a Catalyst. ChemSusChem 2008, 1, 817–819. [Google Scholar] [CrossRef] [PubMed]
- Shannon, M.S.; Irvin, A.C.; Liu, H.Q.; Moon, J.D.; Hindman, M.S.; Turner, C.H.; Bara, J.E. Chemical and Physical Absorption of SO2 by N-Functionalized Imidazoles: Experimental Results and Molecular-level Insight. Ind. Eng. Chem. Res. 2015, 54, 462–471. [Google Scholar] [CrossRef]
- Kaernbach, W.; Kisielow, W.; Warzecha, L.; Miga, K.; Klecan, R. Influence of petroleum nitrogen compounds on hydrodesulphurization. Fuel 1990, 69, 221–224. [Google Scholar] [CrossRef]
- Macaud, M.; Sévignon, M.; Favre-Réguillon, A.; Lemaire, M. Novel Methodology toward Deep Desulfurization of Diesel Feed Based on the Selective Elimination of Nitrogen Compounds. Ind. Eng. Chem. Res. 2004, 43, 7843–7849. [Google Scholar] [CrossRef]
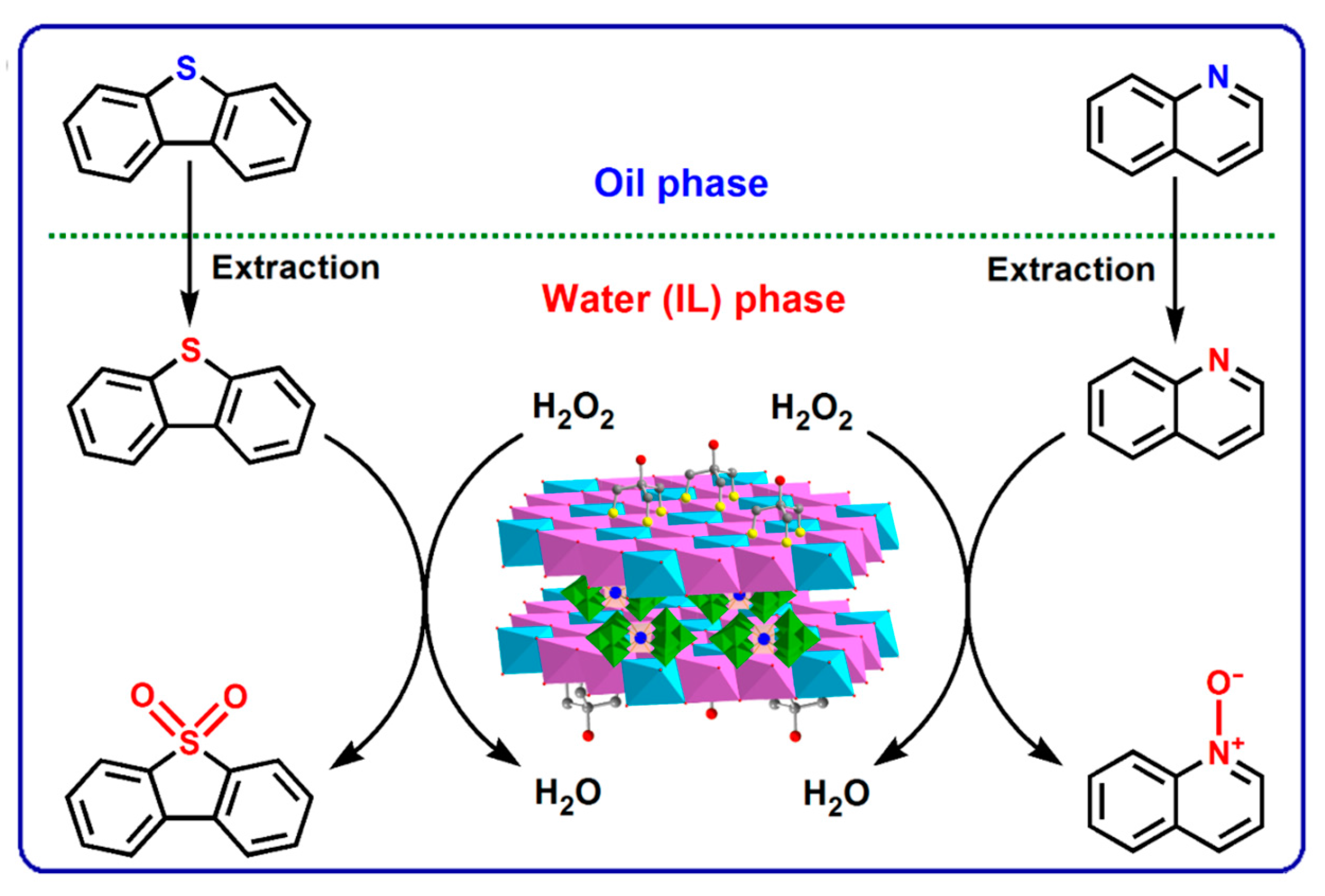
- Yao, Z.Y.; Miras, H.N.; Song, Y.-F. Efficient concurrent removal of sulfur and nitrogen contents from complex oil mixtures by using polyoxometalate-based composite materials. Inorg. Chem. Front. 2016, 3, 1007–1013. [Google Scholar] [CrossRef]
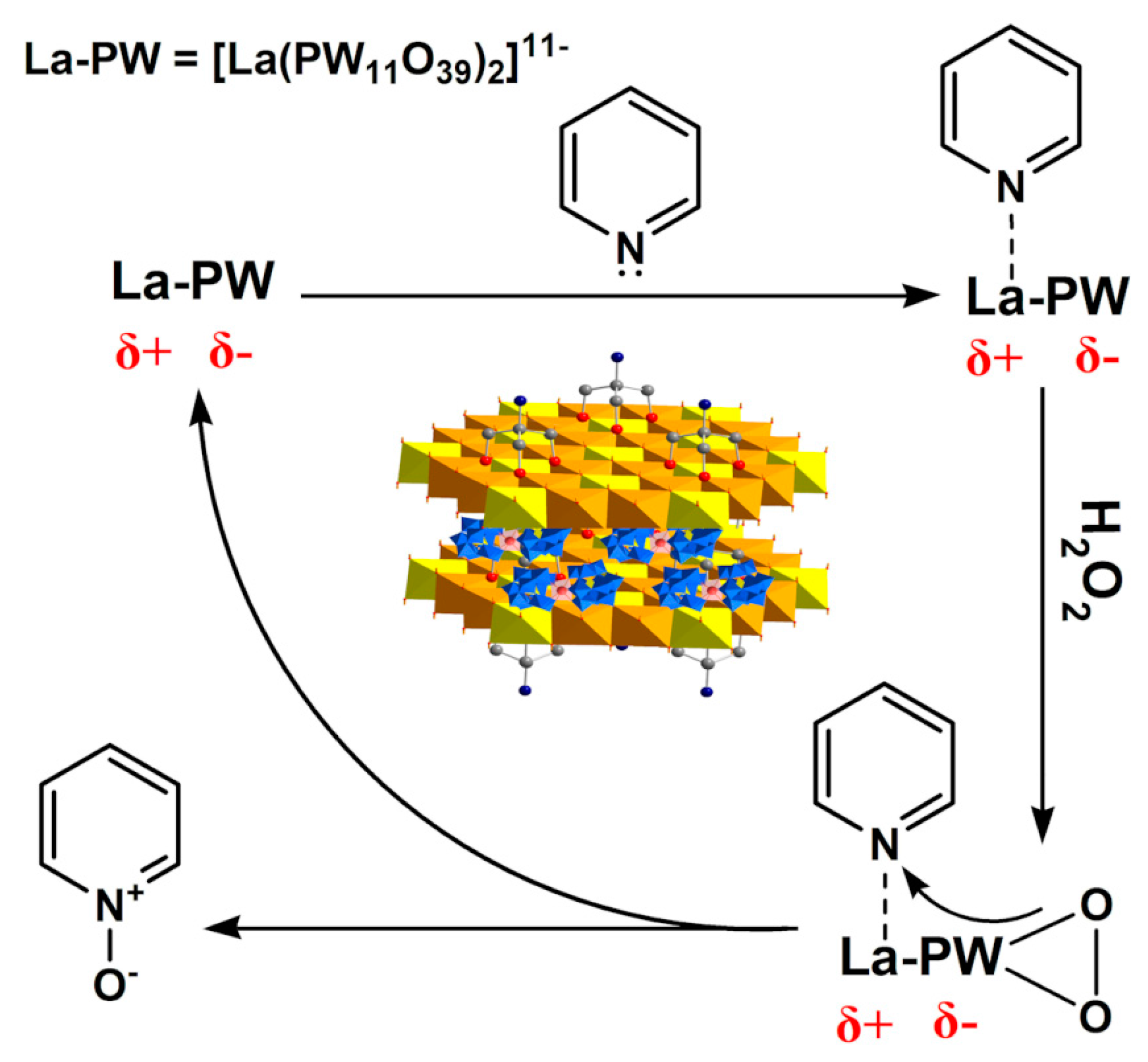
- Bamoharram, F.F.; Heravi, M.M.; Roshani, M.; Tavakoli, N. N-oxidation of pyridine carboxylic acids using hydrogen peroxide catalyzed by a green heteropolyacid catalyst: Preyssler’s anion, [NaP5W30O110]14−. J. Mol. Catal. A 2006, 252, 219–225. [Google Scholar] [CrossRef]
- Malkov, A.V.; Bell, M.; Castelluzzo, F.; Kočovský, P. METHOX: A New Pyridine N-Oxide Organocatalyst for the Asymmetric Allylation of Aldehydes with Allyltrichlorosilanes. Org. Lett. 2005, 7, 3219–3222. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Yao, Z.X.; Miras, H.N.; Song, Y.-F. Facile Immobilization of a Lewis Acid Polyoxometalate onto Layered Double Hydroxides for Highly Efficient N-Oxidation of Pyridine-Based Derivatives and Denitrogenation. ChemCatChem 2015, 7, 3903–3910. [Google Scholar] [CrossRef]
- Corma, A.; Ródenas, T.; Sabater, M.J. Monoalkylations with alcohols by a cascade reaction on bifunctional solid catalysts: Reaction kinetics and mechanism. J. Catal. 2011, 279, 319–327. [Google Scholar] [CrossRef]
- Liu, P.; Li, C.; Hensen, E.J. Efficient Tandem Synthesis of Methyl Esters and Imines by Using Versatile Hydrotalcite-Supported Gold Nanoparticles. Chem. Eur. J. 2012, 18, 12122–12129. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.X.; Wang, W.; Li, H.X. Water-Medium and Solvent-Free Organic Reactions over a Bifunctional Catalyst with Au Nanoparticles Covalently Bonded to HS/SO3H Functionalized Periodic Mesoporous Organosilica. J. Am. Chem. Soc. 2011, 133, 11632–11640. [Google Scholar] [CrossRef] [PubMed]
- Felpin, F.X.; Fouquet, E. Heterogeneous Multifunctional Catalysts for Tandem Processes: An Approach toward Sustainability. ChemSusChem 2008, 1, 718–724. [Google Scholar] [CrossRef] [PubMed]
- Elhamifar, D.; Kazempoor, S.; Karimi, B. Amine-functionalized ionic liquid-based mesoporous organosilica as a highly efficient nanocatalyst for the Knoevenagel condensation. Catal. Sci. Technol. 2016, 6, 4318–4326. [Google Scholar] [CrossRef]
- Zhang, L.N.; Wang, H.; Shen, W.Z.; Qin, Z.F.; Wang, J.G.; Fan, W.B. Controlled synthesis of graphitic carbon nitride and its catalytic properties in Knoevenagel condensations. J. Catal. 2016, 344, 293–302. [Google Scholar] [CrossRef]
- Zhao, S.; Chen, Y.; Song, Y.-F. Tri-lacunary polyoxometalates of Na8H[PW9O34] as heterogeneous Lewis base catalysts for Knoevenagel condensation, cyanosilylation and the synthesis of benzoxazole derivatives. Appl. Catal. A Gen. 2014, 475, 140–146. [Google Scholar] [CrossRef]
- Ezugwu, C.I.; Mousavi, B.; Asraf, M.A.; Luo, Z.X.; Verpoort, F. Post-synthetic modified MOF for Sonogashira cross-coupling and Knoevenagel condensation reactions. J. Catal. 2016, 344, 445–454. [Google Scholar] [CrossRef]
- Liu, K.; Xu, Y.Q.; Yao, Z.Y.; Miras, H.N.; Song, Y.-F. Polyoxometalate-Intercalated Layered Double Hydroxides as Efficient and Recyclable Bifunctional Catalysts for Cascade Reactions. ChemCatChem 2016, 8, 929–937. [Google Scholar] [CrossRef]
- Li, T.F.; Wang, Z.L.; Chen, W.; Miras, H.N.; Song, Y.-F. Rational Design of a Polyoxometalate Intercalated Layered Double Hydroxide: Highly Efficient Catalytic Epoxidation of Allylic Alcohols under Mild and Solvent-Free Conditions. Chem. Eur. J. 2017, 23, 1069–1077. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.D.; Henninger, T.; Abbanat, D.; Bush, K.; Foleno, B.; Hilliard, J.; Macielag, M. Synthesis and antibacterial activity of C2-fluoro, C6-carbamate ketolides, and their C9-oximes. Bioorg. Med. Chem. Lett. 2005, 15, 883–887. [Google Scholar] [CrossRef] [PubMed]
- Jain, N.; Kumar, A.; Chauhan, S.M.S. Metalloporphyrin and heteropoly acid catalyzed oxidation of C=NOH bonds in an ionic liquid: Biomimetic models of nitric oxide synthase. Tetrahedron Lett. 2005, 46, 2599–2602. [Google Scholar] [CrossRef]
- Bars, J.L.; Dakka, J.; Sheldon, R.A. Ammoximation of cyclohexanone and hydroxyaromatic ketones over titanium molecular sieves. Appl. Catal. A Gen. 1996, 136, 69–80. [Google Scholar] [CrossRef]
- Song, F.; Liu, Y.M.; Wu, H.H.; He, M.Y.; Wu, P.; Tatsumi, T. A novel titanosilicate with MWW structure: Highly effective liquid-phase ammoximation of cyclohexanone. J. Catal. 2006, 237, 359–367. [Google Scholar] [CrossRef]
- Li, Z.H.; Chen, R.Z.; Xing, W.H.; Jin, W.Q.; Xu, N.P. Continuous Acetone Ammoximation over TS-1 in a Tubular Membrane Reactor. Ind. Eng. Chem. Res. 2010, 49, 6309–6316. [Google Scholar] [CrossRef]
- Sloboda-Rozner, D.; Neumann, R. Aqueous biphasic catalysis with polyoxometalates: Oximation of ketones and aldehydes with aqueous ammonia and hydrogen peroxide. Green Chem. 2006, 8, 679–681. [Google Scholar] [CrossRef]
- Liu, P.; Wang, H.; Feng, Z.C.; Ying, P.L.; Li, C. Direct immobilization of self-assembled polyoxometalate catalyst in layered double hydroxide for heterogeneous epoxidation of olefins. J. Catal. 2008, 256, 345–348. [Google Scholar] [CrossRef]
- Brunel, J.M.; Holmes, I.P. Chemically Catalyzed Asymmetric Cyanohydrin Syntheses. Angew. Chem. Int. Ed. 2004, 43, 2752–2778. [Google Scholar] [CrossRef] [PubMed]
- Sreekanth, P.; Kim, S.-W.; Hyeon, T.; Kim, B.M. A Novel Mesoporous Silica-Supported Lewis Acid Catalyst for C-C Bond Formation Reactions in Water. Adv. Synth. Catal. 2003, 345, 936–938. [Google Scholar] [CrossRef]
- Procopio, A.; Das, G.; Nardi, M.; Oliverio, M.; Pasqua, L. A Mesoporous ErIII-MCM-41 Catalyst for the Cyanosilylation of Aldehydes and Ketones under Solvent-free Conditions. ChemSusChem 2008, 1, 916–919. [Google Scholar] [CrossRef] [PubMed]
- Song, J.J.; Gallou, F.; Reeves, J.T.; Tan, Z.L.; Yee, N.K.; Senanayake, C.H. Activation of TMSCN by N-Heterocyclic Carbenes for Facile Cyanosilylation of Carbonyl Compounds. J. Org. Chem. 2006, 71, 1273–1276. [Google Scholar] [CrossRef] [PubMed]
- Denmark, S.E.; Chung, W.J. Lewis Base Catalyzed Addition of Trimethylsilyl Cyanide to Aldehydes. J. Org. Chem. 2006, 71, 4002–4005. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, T.; Miras, H.N.; Song, Y.-F. Polyoxometalate (POM)-Layered Double Hydroxides (LDH) Composite Materials: Design and Catalytic Applications. Catalysts 2017, 7, 260. https://doi.org/10.3390/catal7090260
Li T, Miras HN, Song Y-F. Polyoxometalate (POM)-Layered Double Hydroxides (LDH) Composite Materials: Design and Catalytic Applications. Catalysts. 2017; 7(9):260. https://doi.org/10.3390/catal7090260
Chicago/Turabian StyleLi, Tengfei, Haralampos N. Miras, and Yu-Fei Song. 2017. "Polyoxometalate (POM)-Layered Double Hydroxides (LDH) Composite Materials: Design and Catalytic Applications" Catalysts 7, no. 9: 260. https://doi.org/10.3390/catal7090260