Nano-Array Integrated Structured Catalysts: A New Paradigm upon Conventional Wash-Coated Monolithic Catalysts?
Abstract
:1. Introduction
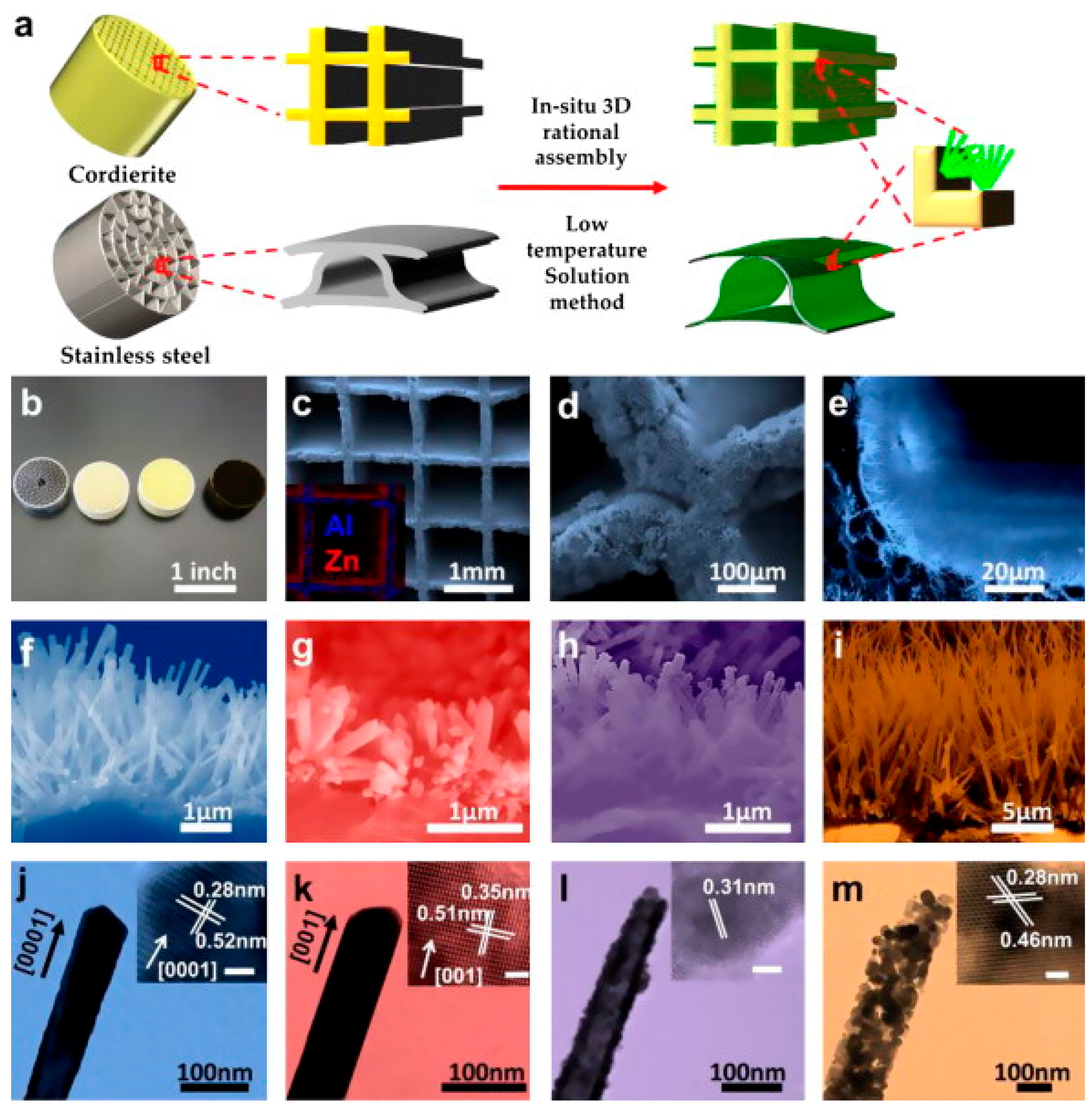
2. Preparation and Fabrication
2.1. Conventional Wash-Coating Procedure
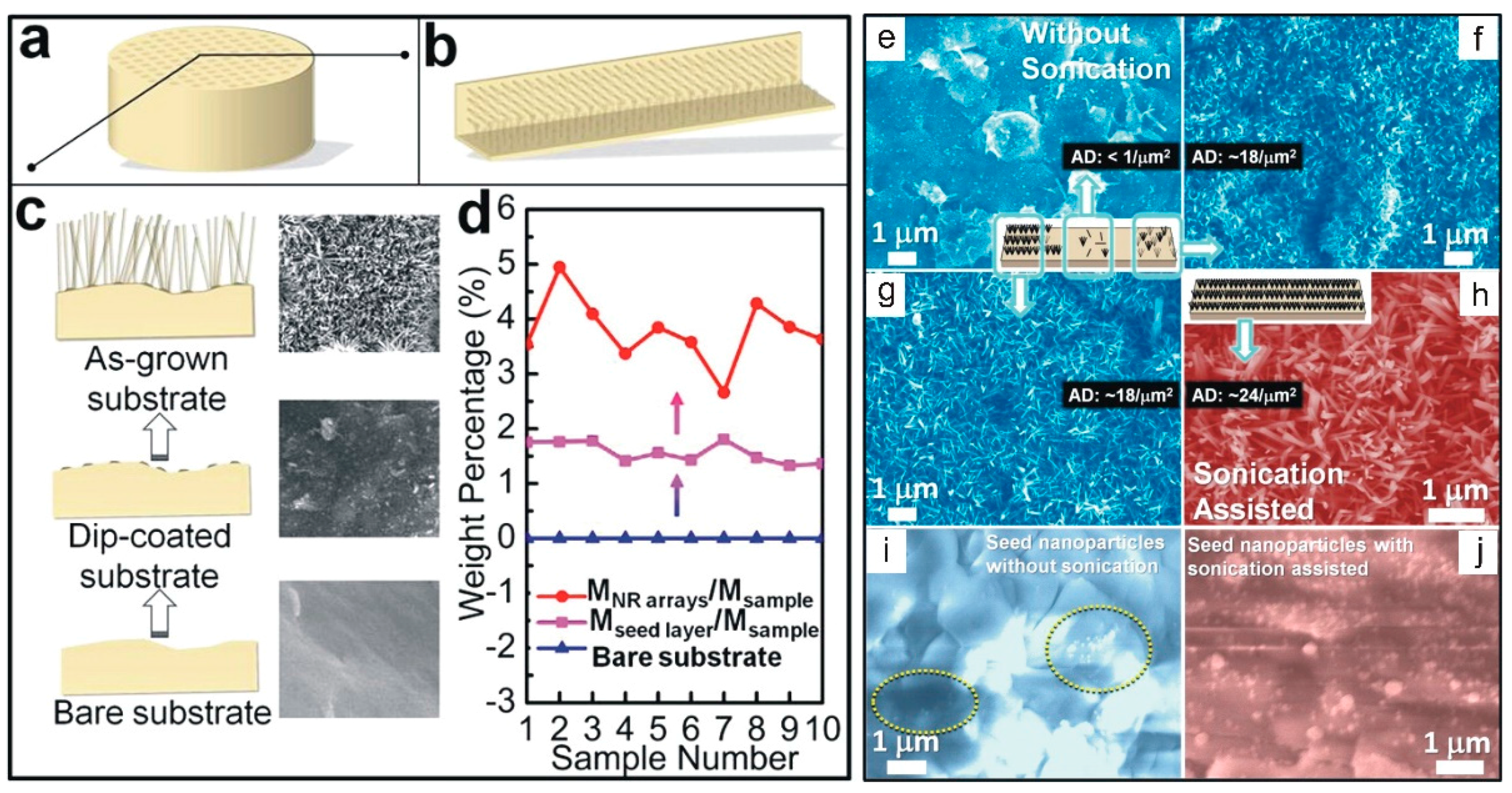
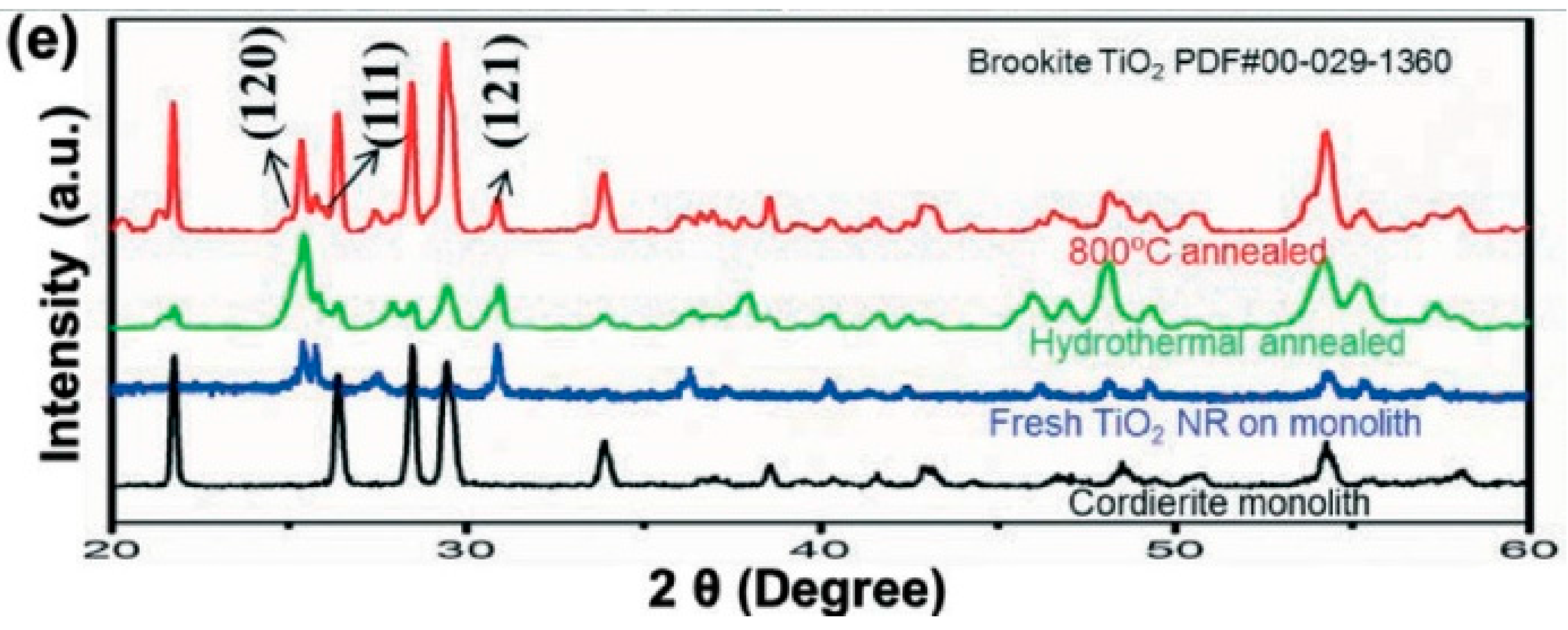
2.2. Hydrothermal Synthesis
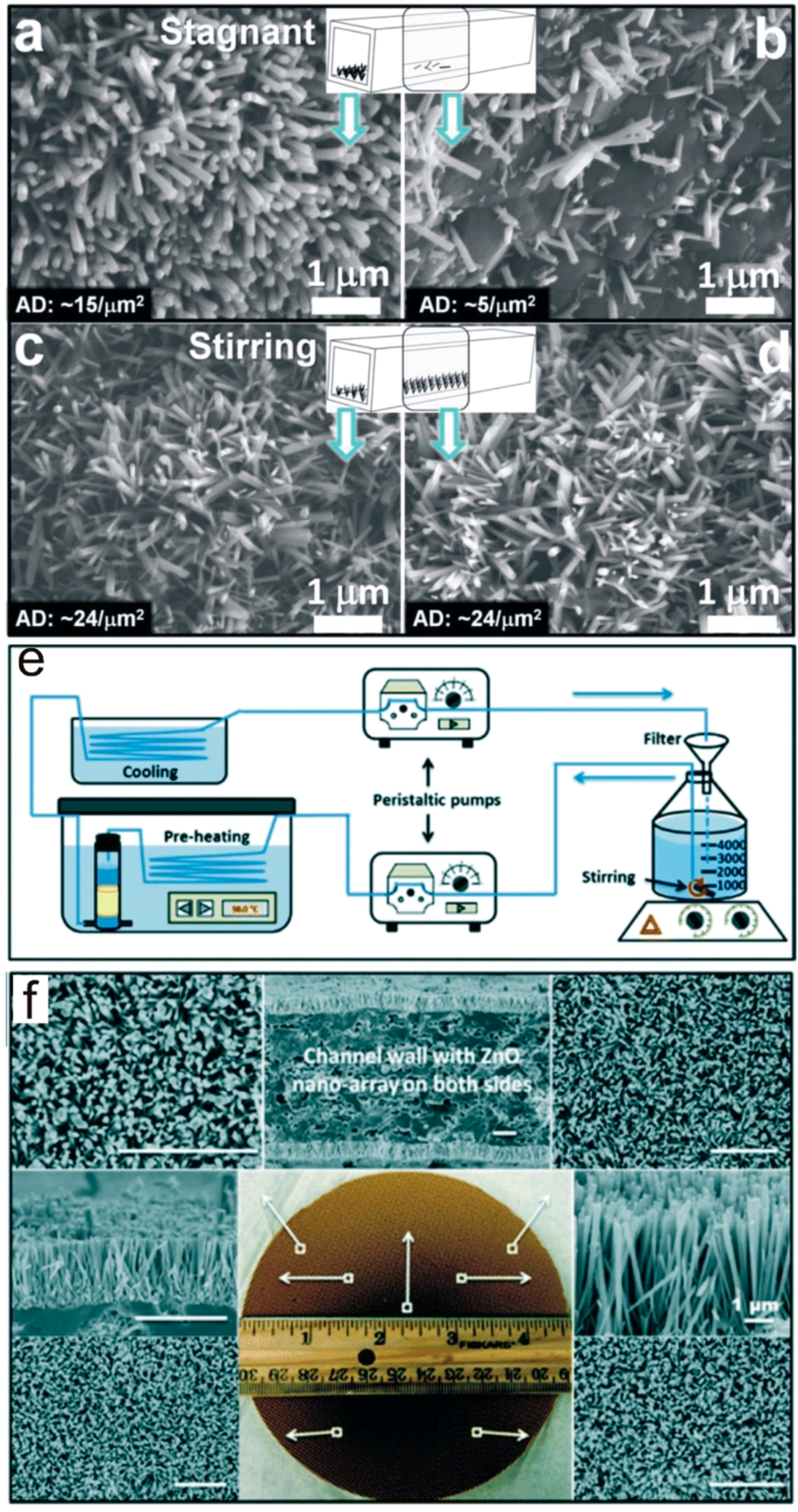
2.3. Scalable Fabrication
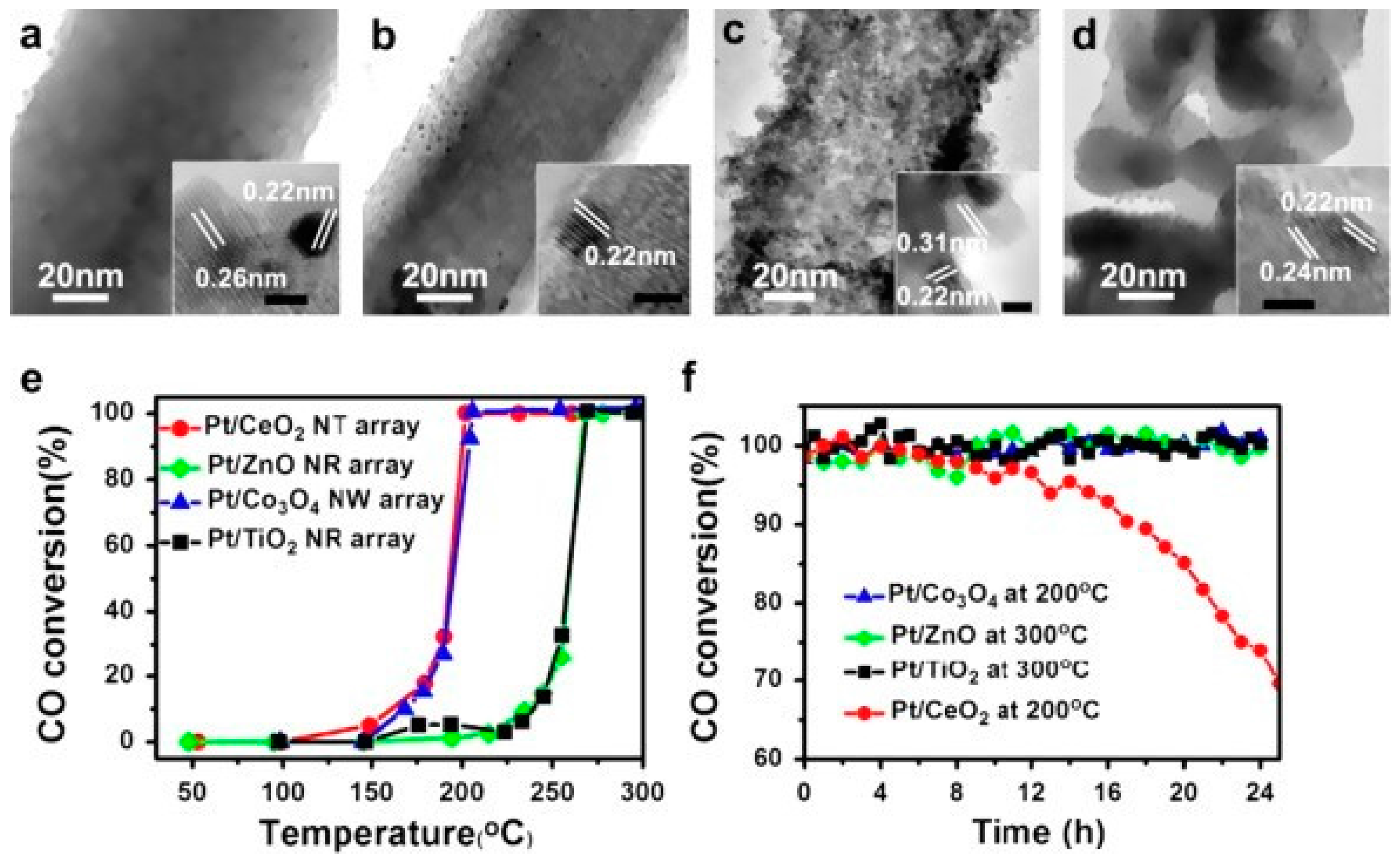
3. Tunable Structural and Catalytic Performance
3.1. General Catalytic Performance of Nano-Array Based Monolithic Catalysts
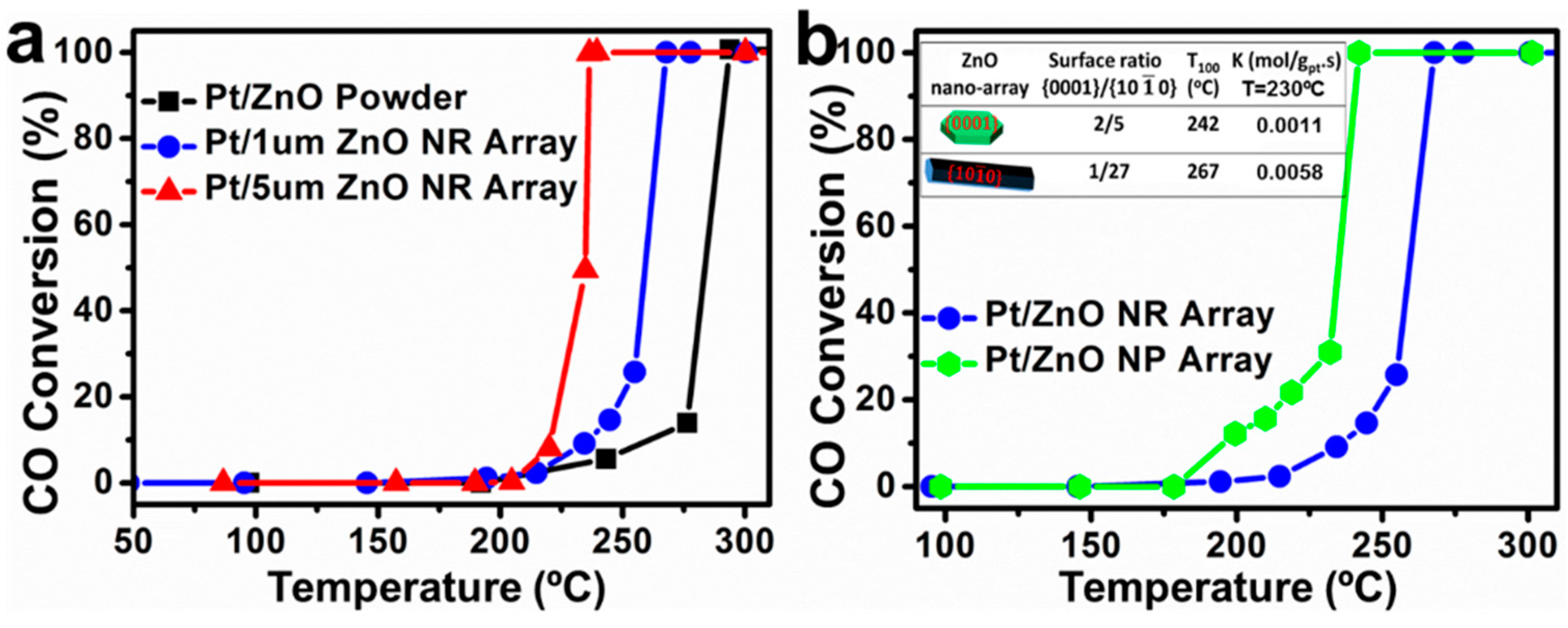
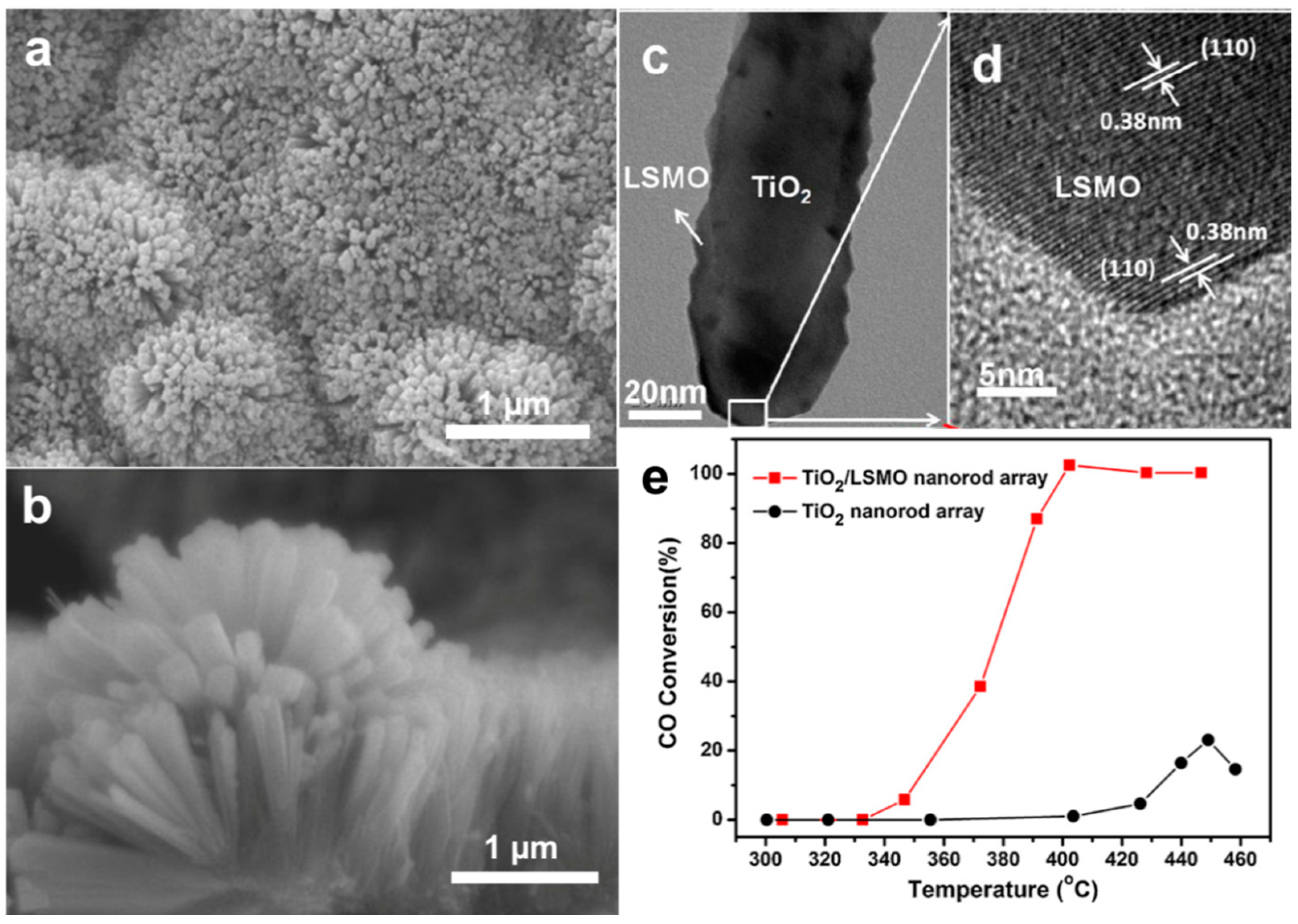
3.2. Morphology and Geometry Adjustment
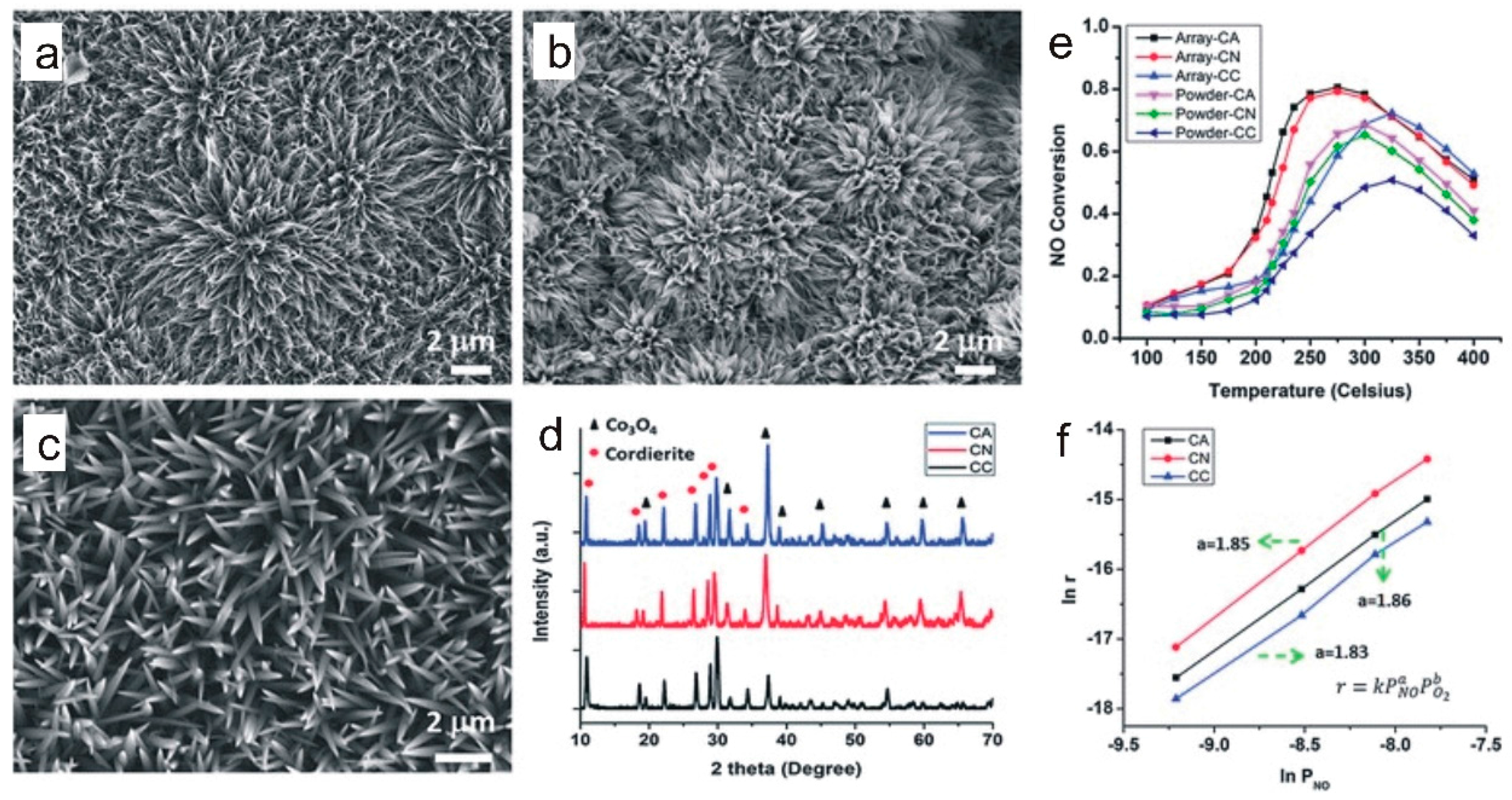
3.3. Guest Atoms and Defects Manipulation
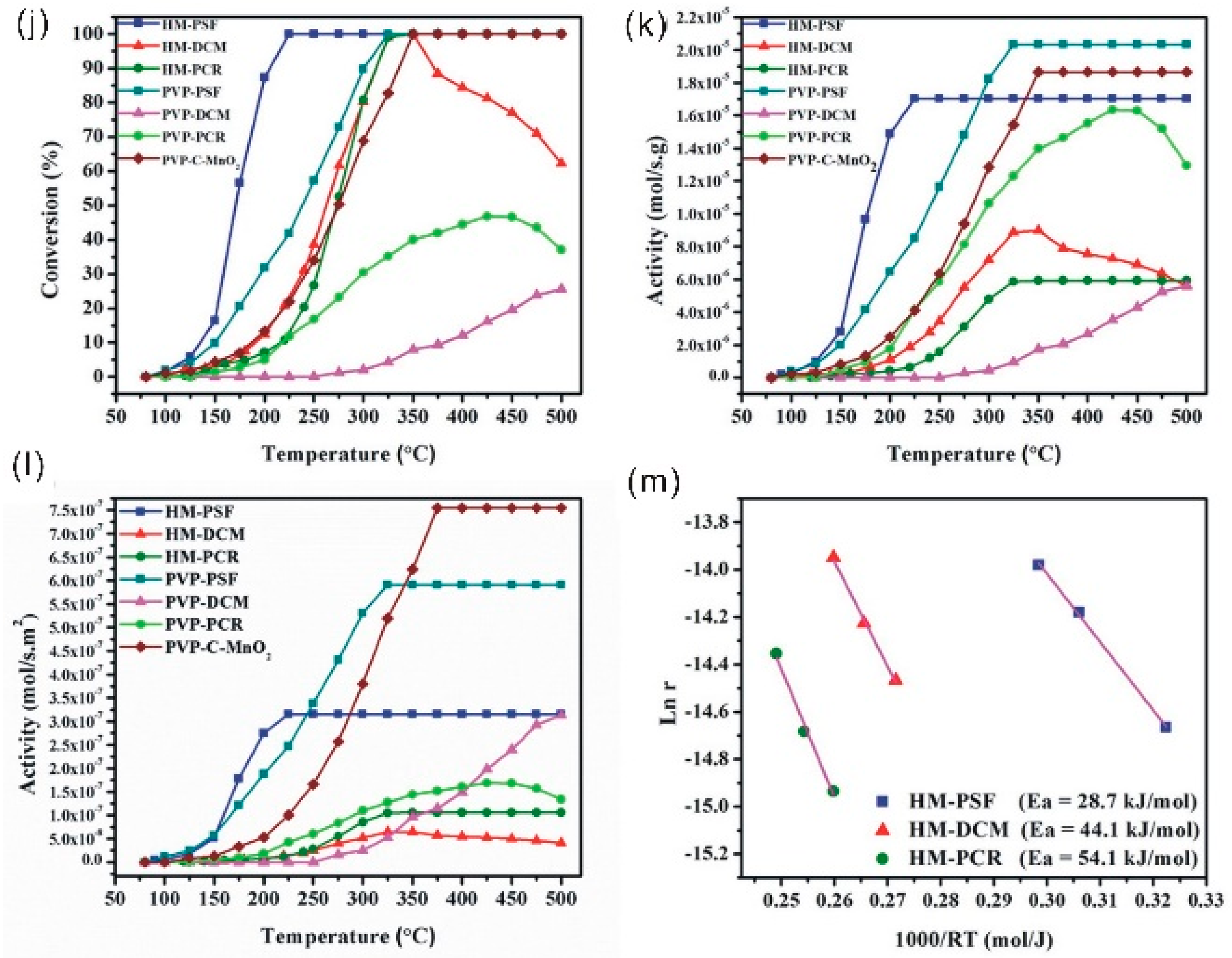
3.4. Composite Nano-Array Catalysts
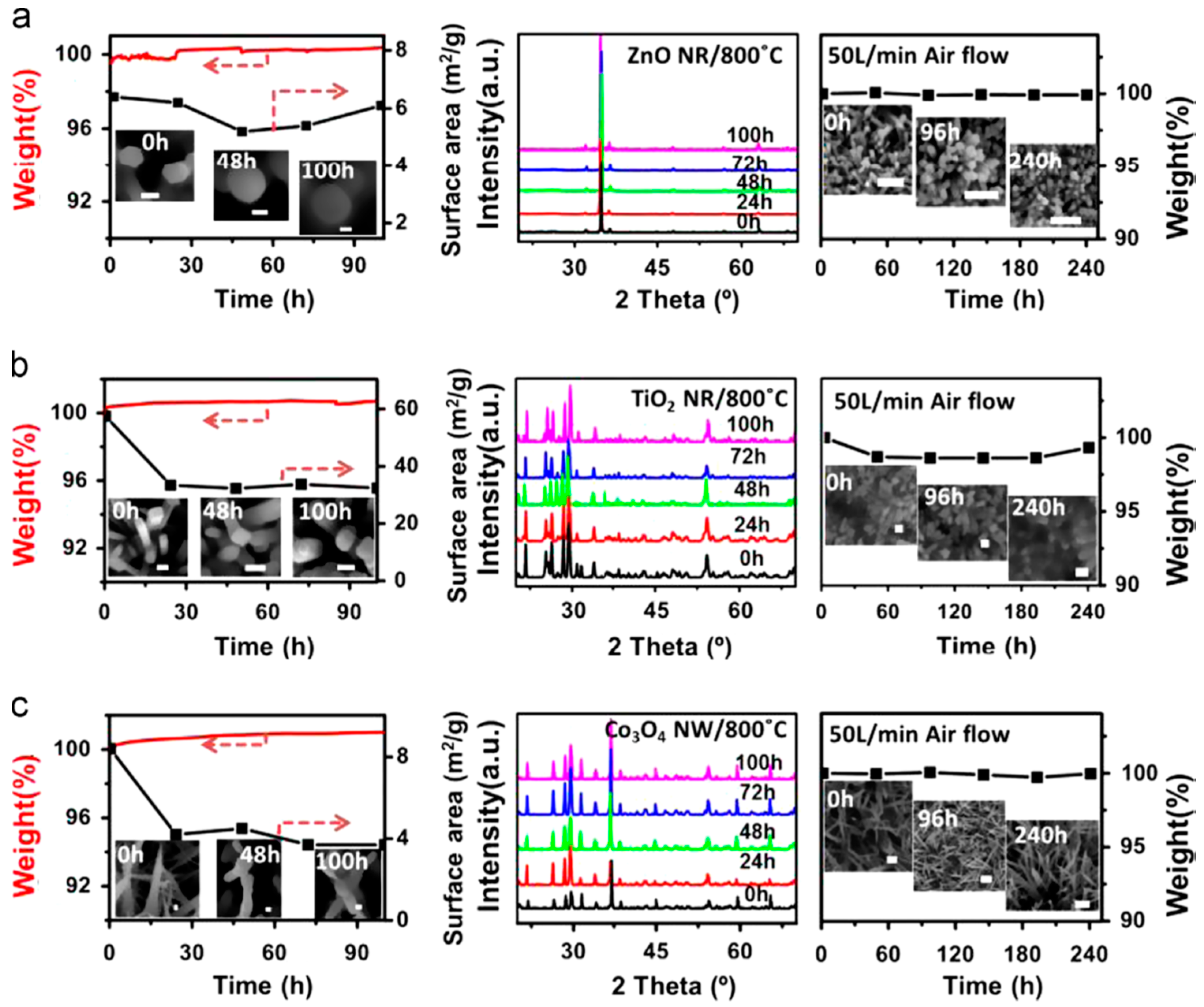
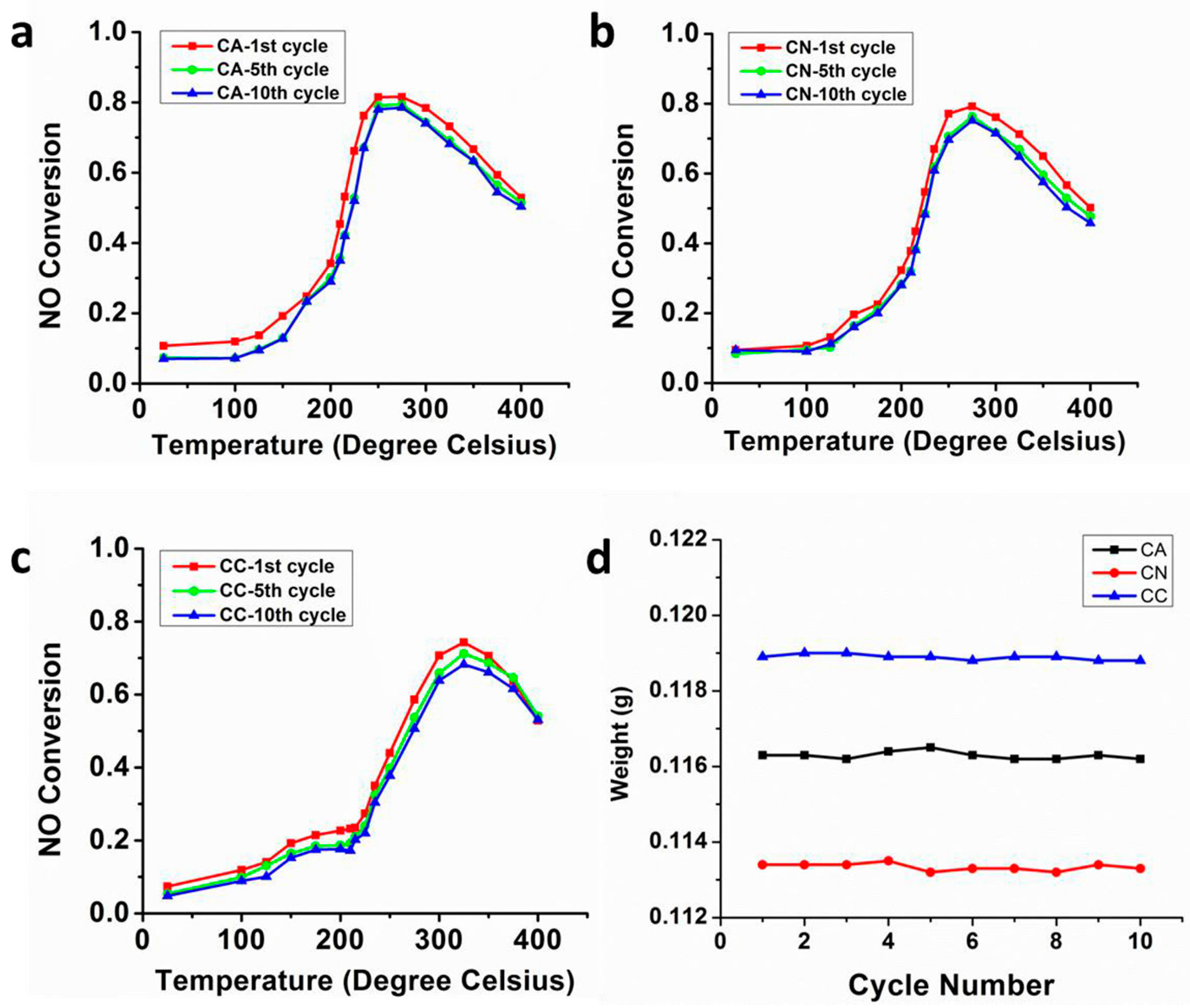
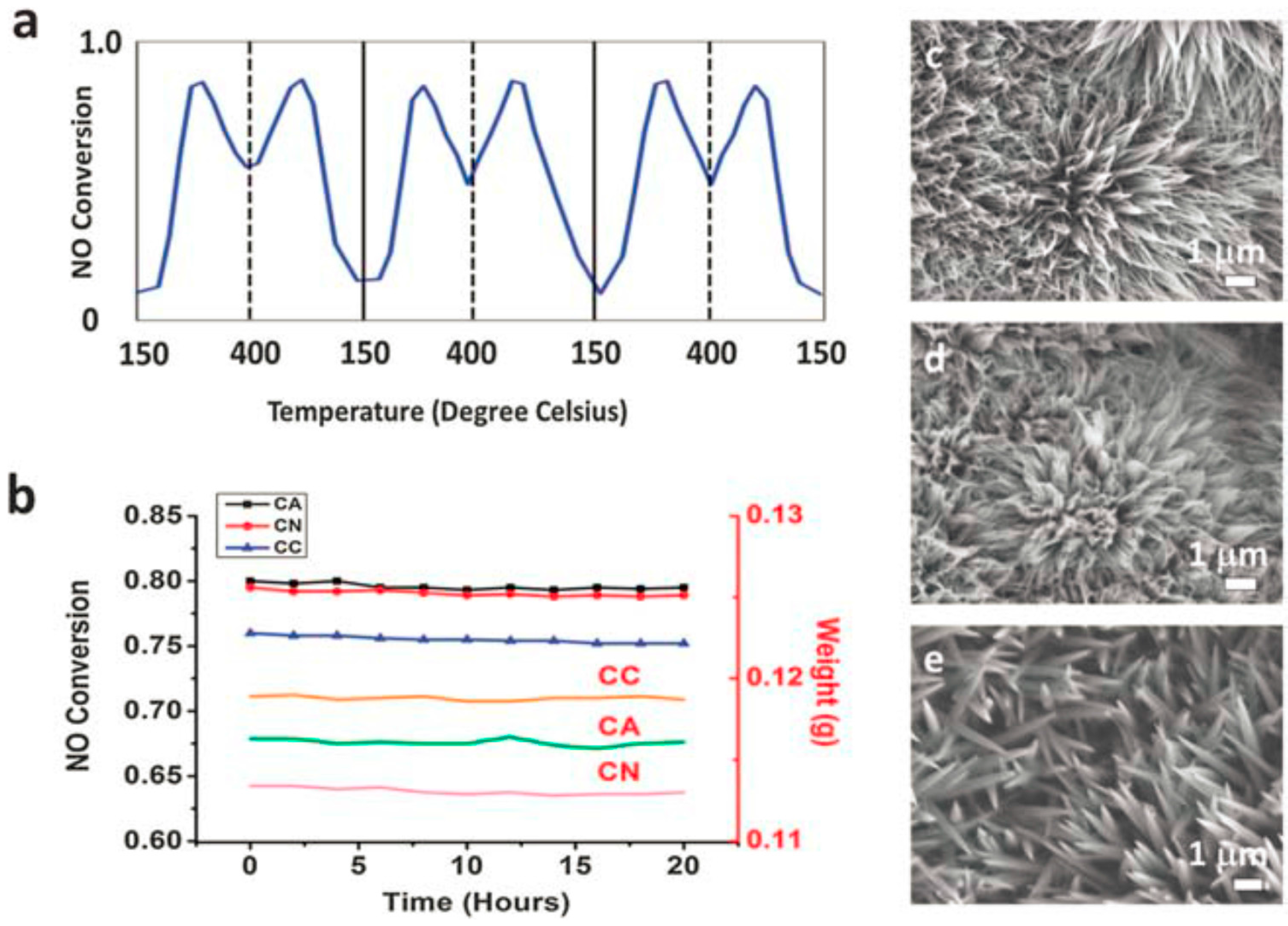
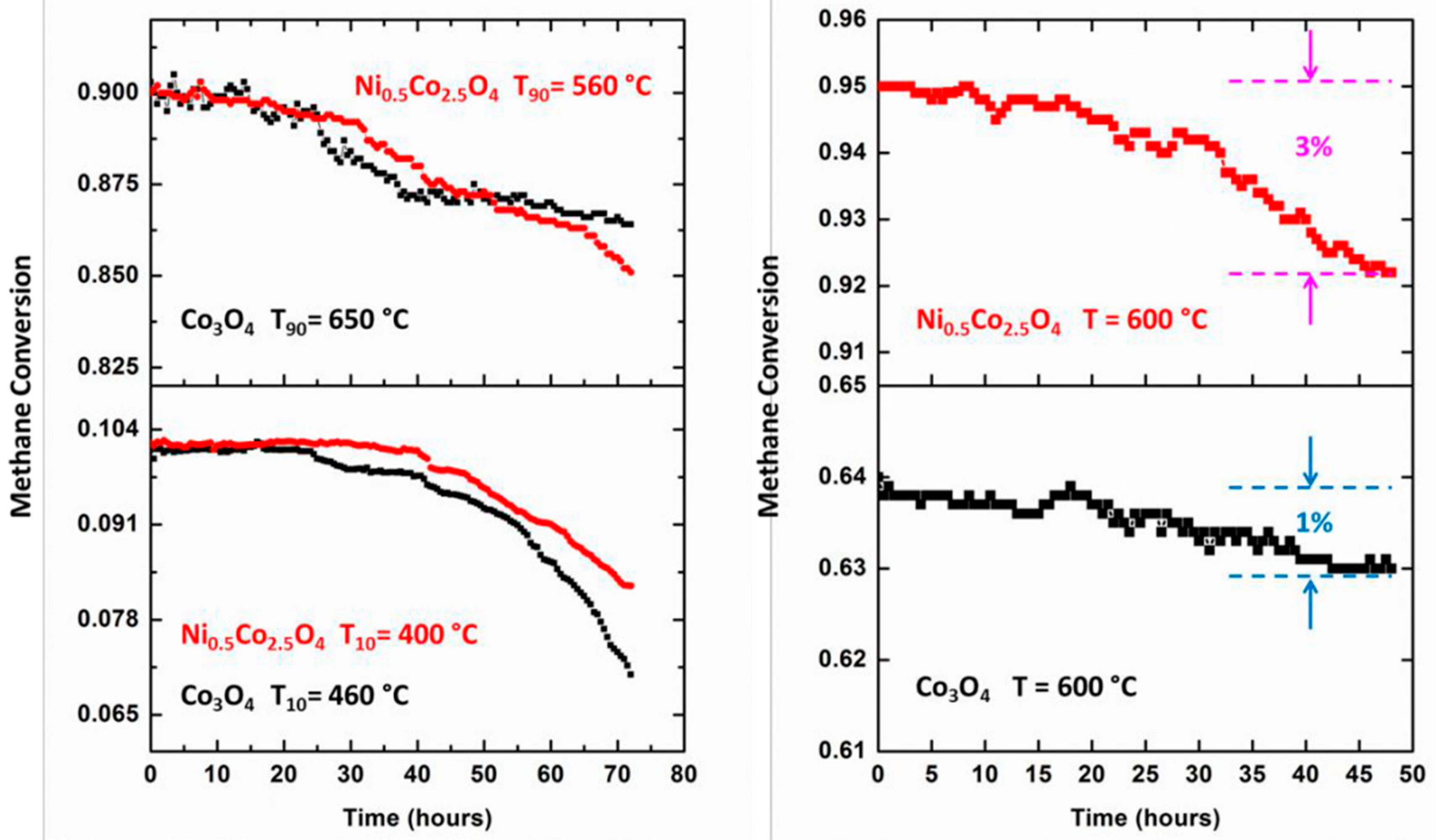
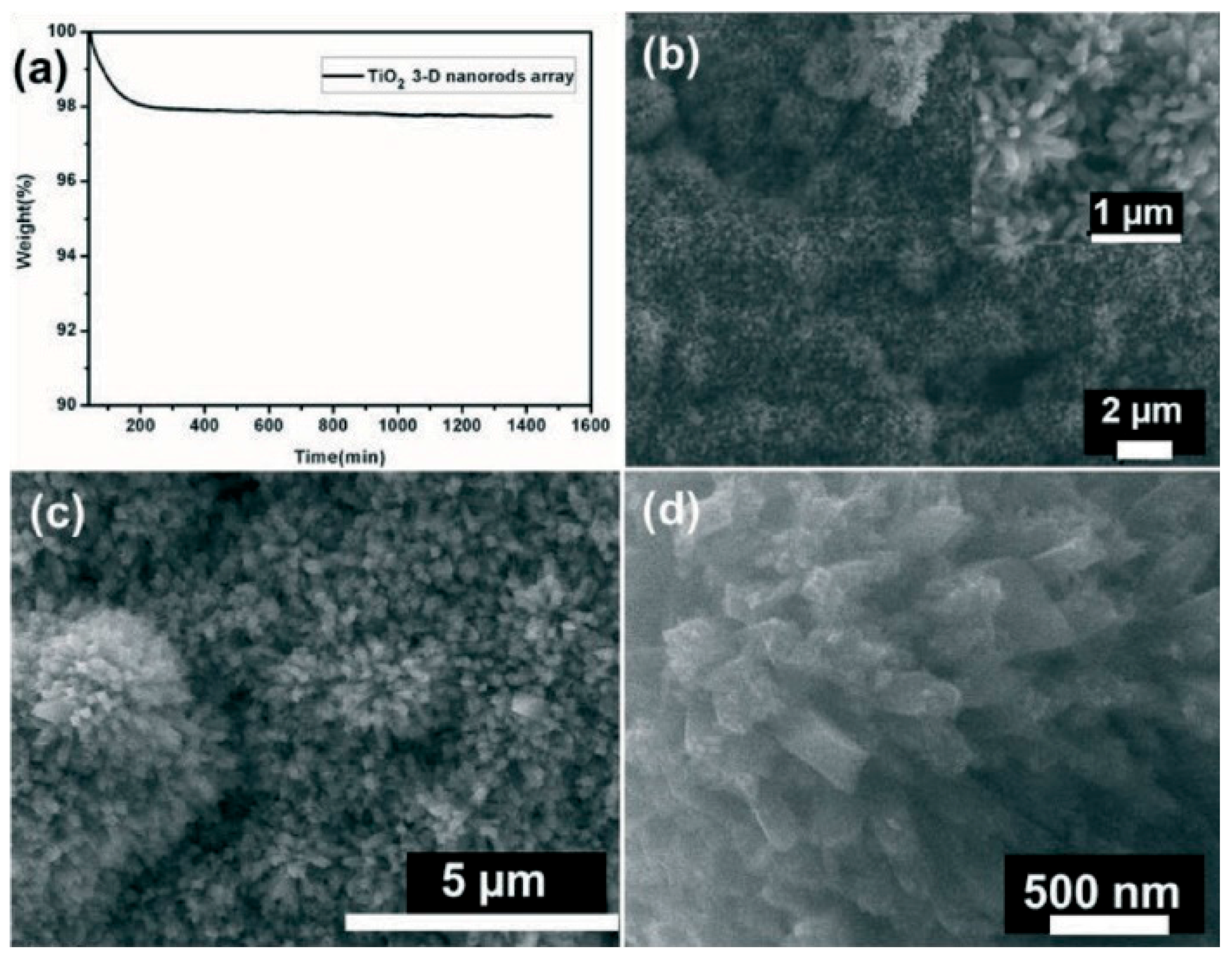
4. Stability Properties
5. Conclusions and Outlook
- (a)
- Cost-effective fabrication methods without multi-step complex processes and binders or additives involved;
- (b)
- Well-defined structure with good uniformity on the monolithic substrates to control and predict the desired structure;
- (c)
- Capability to tune catalysts performance and potential to rationally design better catalysts with excellent stable properties.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lundberg, B.; Sjöblom, J.; Johansson, Å.; Westerberg, B.; Creaser, D. Doc modeling combining kinetics and mass transfer using inert washcoat layers. Appl. Catal. B Environ. 2016, 191, 116–129. [Google Scholar] [CrossRef]
- Rhys-Tyler, G.A.; Legassick, W.; Bell, M.C. The significance of vehicle emissions standards for levels of exhaust pollution from light vehicles in an urban area. Atmos. Environ. 2011, 45, 3286–3293. [Google Scholar] [CrossRef]
- Bielaczyc, P.; Woodburn, J.; Szczotka, A. An assessment of regulated emissions and CO2 emissions from a European light-duty CNG-fueled vehicle in the context of Euro 6 emissions regulations. Appl. Energy 2014, 117, 134–141. [Google Scholar] [CrossRef]
- Williams, J.L. Monolith structures, materials, properties and uses. Catal. Today 2001, 69, 3–9. [Google Scholar] [CrossRef]
- Elmer, T.H. Ultra-Low Expansion Ceramic Articles. U.S. Patent 3958058 A, 18 May 1976. [Google Scholar]
- Avila, P.; Montes, M.; Miró, E.E. Monolithic reactors for environmental applications: A review on preparation technologies. Chem. Eng. J. 2005, 109, 11–36. [Google Scholar] [CrossRef]
- Nijhuis, T.A.; Beers, A.E.; Vergunst, T.; Hoek, I.; Kapteijn, F.; Moulijn, J.A. Preparation of monolithic catalysts. Catal. Rev. 2001, 43, 345–380. [Google Scholar] [CrossRef]
- Ren, Z.; Guo, Y.; Liu, C.-H.; Gao, P.-X. Hierarchically nanostructured materials for sustainable environmental applications. Front. Chem. 2013, 1, 18. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Liu, G.; Ren, Z.; Piyadasa, A.; Gao, P.-X. Single crystalline brookite titanium dioxide nanorod arrays rooted on ceramic monoliths: A hybrid nanocatalyst support with ultra-high surface area and thermal stability. CrystEngComm 2013, 15, 8345–8352. [Google Scholar] [CrossRef]
- Chen, H.; Rui, Z.; Ji, H. Monolith-like TiO2 nanotube array supported Pt catalyst for HCHO removal under mild conditions. Ind. Eng. Chem. Res. 2014, 53, 7629–7636. [Google Scholar] [CrossRef]
- Ren, Z.; Guo, Y.; Gao, P.-X. Nano-array based monolithic catalysts: Concept, rational materials design and tunable catalytic performance. Catal. Today 2015, 258, 441–453. [Google Scholar] [CrossRef]
- Wang, S.; Ren, Z.; Guo, Y.; Gao, P.-X. Nano-array integrated monolithic devices: Toward rational materials design and multi-functional performance by scalable nanostructures assembly. CrystEngComm 2016, 18, 2980–2993. [Google Scholar] [CrossRef]
- Xu, S.; Wang, Z.L. One-dimensional zno nanostructures: Solution growth and functional properties. Nano Res. 2011, 4, 1013–1098. [Google Scholar] [CrossRef]
- Xiao, W.; Guo, Y.; Ren, Z.; Wrobel, G.; Ren, Z.; Lu, T.; Gao, P.-X. Mechanical-agitation-assisted growth of large-scale and uniform zno nanorod arrays within 3D multichannel monolithic substrates. Cryst. Growth Des. 2013, 13, 3657–3664. [Google Scholar] [CrossRef]
- Wang, S.; Ren, Z.; Song, W.; Guo, Y.; Zhang, M.; Suib, S.L.; Gao, P.-X. ZnO/perovskite core-shell nanorod array based monolithic catalysts with enhanced propane oxidation and material utilization efficiency at low temperature. Catal. Today 2015, 258, 549–555. [Google Scholar] [CrossRef]
- Ren, Z.; Guo, Y.; Zhang, Z.; Liu, C.; Gao, P.-X. Nonprecious catalytic honeycombs structured with three dimensional hierarchical Co3O4 nano-arrays for high performance nitric oxide oxidation. J. Mater. Chem. A 2013, 1, 9897–9906. [Google Scholar] [CrossRef]
- Ren, Z.; Botu, V.; Wang, S.; Meng, Y.; Song, W.; Guo, Y.; Ramprasad, R.; Suib, S.L.; Gao, P.-X. Monolithically integrated spinel MxCo3−xO4 (M = Co, Ni, Zn) nanoarray catalysts: Scalable synthesis and cation manipulation for tunable low-temperature CH4 and CO oxidation. Angew. Chem. 2014, 53, 7223–7227. [Google Scholar] [CrossRef] [PubMed]
- Ren, Z.; Wu, Z.; Song, W.; Xiao, W.; Guo, Y.; Ding, J.; Suib, S.L.; Gao, P.-X. Low temperature propane oxidation over Co3O4 based nano-array catalysts: Ni dopant effect, reaction mechanism and structural stability. Appl. Catal. B Environ. 2016, 180, 150–160. [Google Scholar] [CrossRef]
- Zhang, Z.; Gao, H.; Cai, W.; Liu, C.; Guo, Y.; Gao, P.-X. In situ TPR removal: A generic method for fabricating tubular array devices with mechanical and structural soundness, and functional robustness on various substrates. J. Mater. Chem. 2012, 22, 23098–23105. [Google Scholar] [CrossRef]
- Xie, X.; Li, Y.; Liu, Z.-Q.; Haruta, M.; Shen, W. Low-temperature oxidation of CO catalysed by Co3O4 nanorods. Nature 2009, 458, 746–749. [Google Scholar] [CrossRef]
- Hu, L.; Peng, Q.; Li, Y. Selective synthesis of Co3O4 nanocrystal with different shape and crystal plane effect on catalytic property for methane combustion. J. Am. Chem. Soc. 2008, 130, 16136–16137. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Sun, K.; Peng, Q.; Xu, B.; Li, Y. Surface active sites on Co3O4 nanobelt and nanocube model catalysts for CO oxidation. Nano Res. 2010, 3, 363–368. [Google Scholar] [CrossRef]
- Surendranath, Y.; Kanan, M.W.; Nocera, D.G. Mechanistic studies of the oxygen evolution reaction by a cobalt-phosphate catalyst at neutral ph. J. Am. Chem. Soc. 2010, 132, 16501–16509. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Dong, F.; Heinbuch, S.; Rocca, J.J.; Bernstein, E.R. Oxidation reactions on neutral cobalt oxide clusters: Experimental and theoretical studies. Phys. Chem. Chem. Phys. 2010, 12, 947–959. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.-Y.; Song, W.; Lin, H.-J.; Wang, S.; Biswas, S.; Mollahosseini, M.; Kuo, C.-H.; Gao, P.-X.; Suib, S.L. Manganese oxide nanoarray-based monolithic catalysts: Tunable morphology and high efficiency for CO oxidation. ACS Appl. Mater. Interfaces 2016, 8, 7834–7842. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Zhang, Z.; Ren, Z.; Gao, H.; Gao, P.-X. Synthesis, characterization and CO oxidation of TiO2/(La,Sr)MnO3 composite nanorod array. Catal. Today 2012, 184, 178–183. [Google Scholar] [CrossRef]
- Wu, D.; Zhang, H. Mechanical stability of monolithic catalysts: Scattering of washcoat adhesion and failure mechanism of active material. Ind. Eng. Chem. Res. 2013, 52, 14713–14721. [Google Scholar] [CrossRef]
- Wu, D.; Kong, S.; Zhang, H.; Li, Y. Mechanical stability of monolithic catalysts: Factors affecting washcoat adhesion and cohesion during preparation. AIChE J. 2014, 60, 2765–2773. [Google Scholar] [CrossRef]
- Hernández-Garrido, J.C.; Gaona, D.; Gómez, D.M.; Gatica, J.M.; Vidal, H.; Sanz, O.; Rebled, J.M.; Peiro, F.; Calvino, J.J. Comparative study of the catalytic performance and final surface structure of Co3O4/La-CeO2 washcoated ceramic and metallic honeycomb monoliths. Catal. Today 2015, 253, 190–198. [Google Scholar] [CrossRef]
- Hernández-Garrido, J.C.; Gómez, D.M.; Gaona, D.; Vidal, H.; Gatica, J.M.; Sanz, O.; Rebled, J.M.; Peiró, F.; Calvino, J.J. Combined (S)TEM-FIB insight into the influence of the preparation method on the final surface structure of a Co3O4/La-modified-CeO2 washcoated monolithic catalyst. J. Phys. Chem. C 2013, 117, 13028–13036. [Google Scholar] [CrossRef]
- Cao, A.; Veser, G. Exceptional high-temperature stability through distillation-like self-stabilization in bimetallic nanoparticles. Nat. Mater. 2010, 9, 75–81. [Google Scholar] [CrossRef]
- Gandhi, H.; Graham, G.; McCabe, R.W. Automotive exhaust catalysis. J. Catal. 2003, 216, 433–442. [Google Scholar] [CrossRef]
- Burton, P.D.; Peterson, E.J.; Boyle, T.J.; Datye, A.K. Synthesis of high surface area ZnO(0001) plates as novel oxide supports for heterogeneous catalysts. Catal. Lett. 2010, 139, 26–32. [Google Scholar] [CrossRef]
- Wang, Y.-Z.; Zhao, Y.-X.; Gao, C.-G.; Liu, D.-S. Origin of the high activity and stability of Co3O4 in low-temperature co oxidation. Catal. Lett. 2008, 125, 134–138. [Google Scholar] [CrossRef]
- Zamora, M.; López, T.; Asomoza, M.; Meléndrez, R.; Gómez, R. Alkaline doped TiO2 sol–gel catalysts: Effect of sintering on catalyst activity and selectivity for acetone condensation. Catal. Today 2006, 116, 234–238. [Google Scholar] [CrossRef]
- Guo, Y.; Ren, Z.; Xiao, W.; Liu, C.; Sharma, H.; Gao, H.; Mhadeshwar, A.; Gao, P.-X. Robust 3-D configurated metal oxide nano-array based monolithic catalysts with ultrahigh materials usage efficiency and catalytic performance tunability. Nano Energy 2013, 2, 873–881. [Google Scholar] [CrossRef]






| Catalyst | Kinetics Parameters | XPS | Diffraction Planes | |||||
|---|---|---|---|---|---|---|---|---|
| k0 (mol·g−1·s−1) | Ea (kJ·mol−1) | Co3+/Co2+ | {111} | {220} | {311} | {222} | {400} | |
| CA | 5433 | 20.6 | 1.85 | 1.01 | 0.73 | 0.50 | 0.83 | 0.72 |
| CN | 5102 | 20.1 | 1.85 | 0.98 | 0.71 | 0.65 | 1.50 | 0.73 |
| CC | 3840 | 20.1 | 1.40 | 1.10 | 0.57 | 0.32 | 1.67 | 0.40 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Weng, J.; Lu, X.; Gao, P.-X. Nano-Array Integrated Structured Catalysts: A New Paradigm upon Conventional Wash-Coated Monolithic Catalysts? Catalysts 2017, 7, 253. https://doi.org/10.3390/catal7090253
Weng J, Lu X, Gao P-X. Nano-Array Integrated Structured Catalysts: A New Paradigm upon Conventional Wash-Coated Monolithic Catalysts? Catalysts. 2017; 7(9):253. https://doi.org/10.3390/catal7090253
Chicago/Turabian StyleWeng, Junfei, Xingxu Lu, and Pu-Xian Gao. 2017. "Nano-Array Integrated Structured Catalysts: A New Paradigm upon Conventional Wash-Coated Monolithic Catalysts?" Catalysts 7, no. 9: 253. https://doi.org/10.3390/catal7090253