The Role of the Immune Response in Merkel Cell Carcinoma
Abstract
:1. Introduction
2. Incidence
3. Clinical Course
3.1. Tumor Infiltrating Cells
| Study | N | TIL Characterization | Survival | Comment |
|---|---|---|---|---|
| Mott et al. 2004 [33]. | 25 | Minimal/moderate = 17 (68%) | “Heavy” infiltration was associated with poor prognosis in multivariate analysis. | Depth of invasion was associated with poor prognosis in multivariate analysis. |
| Heavy = 8 (32%) | ||||
| Llombart et al. 2005 [28]. | 20 | Absent = 8 (40%) | “Absent” was an independent prognostic factor of DFI in multivariate analysis. | Tumor size>30 mm, stage II, and the presence of >50% of Ki67+ tumor cells were found to be prognostic indicators of disease free interval in univariate analysis. |
| Discontinuous = 12 (60%) | ||||
| Continuous = 0 | ||||
| Andea et al. 2008 [27]. | 156 | Absent = 81 (53%) | “Present nondense” plus “Dense” was associated with longer survival in univariate analysis. | Nodular growth pattern, low tumor depth, and absence of lymphovascular invasion were associated with longer survival on multivariate analysis. |
| Present nondense = 55 (36%) | ||||
| Dense = 17 (11%) | ||||
| Paulson et al. 2011 [30]. | 130 | Not identified = 44 (34%) | TILs were associates with better prognosis on univariate but not multivariate analyses. | Intratumoral CD8+ was independently associated with improved survival in multivariate analysis. |
| Non-brisk/Brisk = 86 (66%) | ||||
| CD8+ infiltrates (IHC) scored on a 0 to 5 scale | ||||
| Sihto et al. 2012 [29]. | 116 | Numbers of intratumoral CD3+, CD8+, CD16+, FoxP3+, and CD68+ cells (IHC) per 1 high power field | High CD3+, CD8+, or FoxP3+ cells, and high CD8+/CD4+ or FoxP3+/CD4+ ratios, were significantly associated with favorable overall survival. | Although the numbers of T cells are generally higher in MCPyV-positive than in MCPyV-negative MCC, high intratumoral T-cell counts are also associated with favorable survival in MCPyV-negative MCC. |
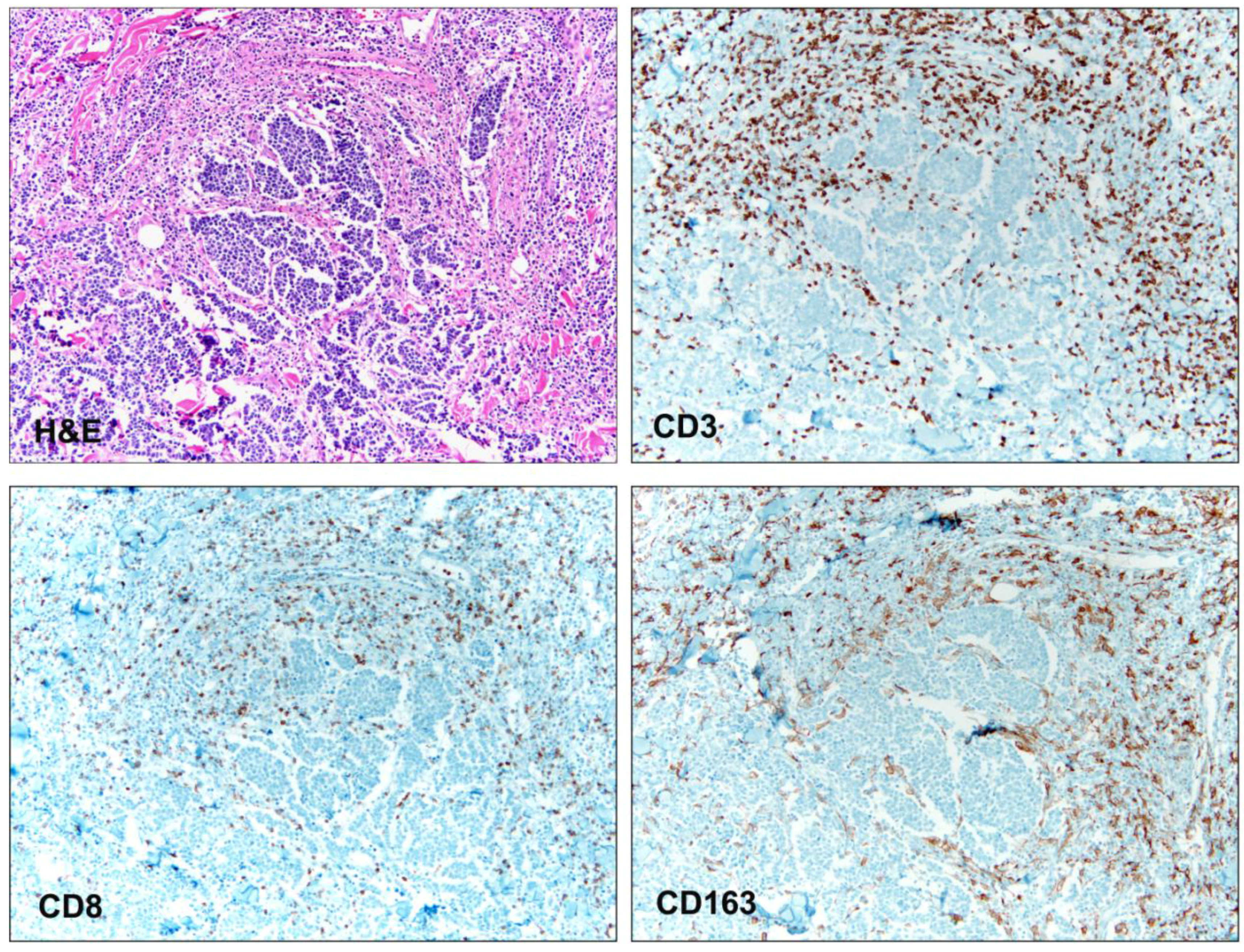
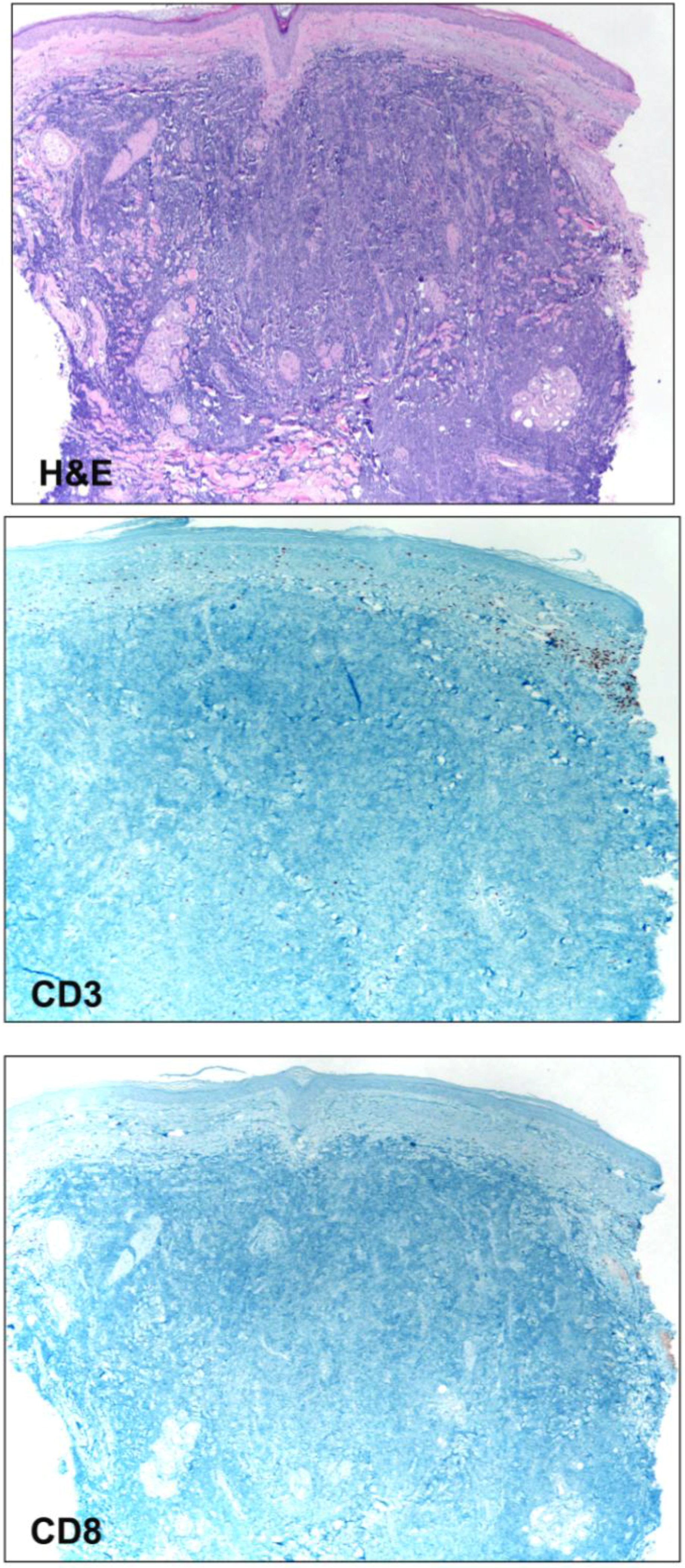
3.2. Tumor Immune Transcripts
3.3. Spontaneous Regression
3.4. Paraneoplastic Autoimmune Syndromes
3.5. Reduction of Immune Suppression
4. MCPyV Immune Response
4.1. Humoral
4.2. Cellular
5. Immunotherapy
5.1. Cytokines
5.2. Adoptive Cellular
5.3. Topical
5.4. Vaccines
6. Conclusions and Future Directions
References
- Rockville Merkel Cell Carcinoma Group. Merkel cell carcinoma: Recent progress and current priorities on etiology, pathogenesis, and clinical management. J. Clin. Oncol. 2009, 27, 4021–4026. [CrossRef]
- Feng, H.; Shuda, M.; Chang, Y.; Moore, P.S. Clonal integration of a polyomavirus in human merkel cell carcinoma. Science 2008, 319, 1096–1100. [Google Scholar]
- Medina-Franco, H.; Urist, M.M.; Fiveash, J.; Heslin, M.J.; Bland, K.I.; Beenken, S.W. Multimodality treatment of Merkel cell carcinoma: Case series and literature review of 1024 cases. Ann. Surg. Oncol. 2001, 8, 204–208. [Google Scholar] [CrossRef]
- Voog, E.; Biron, P.; Martin, J.P.; Blay, J.Y. Chemotherapy for patients with locally advanced or metastatic Merkel cell carcinoma. Cancer 1999, 85, 2589–2595. [Google Scholar] [CrossRef]
- Tai, P. Merkel cell cancer: Update on biology and treatment. Curr. Opin. Oncol. 2008, 20, 196–200. [Google Scholar] [CrossRef]
- Garneski, K.M.; Nghiem, P. Merkel cell carcinoma adjuvant therapy: Current data support radiation but not chemotherapy. J. Am. Acad. Dermatol. 2007, 57, 166–169. [Google Scholar] [CrossRef]
- Poulsen, M.G.; Rischin, D.; Porter, I.; Walpole, E.; Harvey, J.; Hamilton, C.; Keller, J.; Tripcony, L. Does chemotherapy improve survival in high-risk stage I and II Merkel cell carcinoma of the skin? Int. J. Radiat. Oncol. Biol. Phys. 2006, 64, 114–119. [Google Scholar]
- Foulongne, V.; Kluger, N.; Dereure, O.; Mercier, G.; Molès, J.P.; Guillot, B.; Segondy, M. Merkel cell polyomavirus in cutaneous swabs. Emerging Infect. Dis. 2010, 16, 685–687. [Google Scholar]
- Foulongne, V.; Dereure, O.; Kluger, N.; Molès, J.P.; Guillot, B.; Segondy, M. Merkel cell polyomavirus DNA detection in lesional and nonlesional skin from patients with Merkel cell carcinoma or other skin diseases. Br. J. Dermatol. 2010, 162, 59–63. [Google Scholar] [CrossRef]
- Houben, R.; Shuda, M.; Weinkam, R.; Schrama, D.; Feng, H.; Chang, Y.; Moore, P.S.; Becker, J.C. Merkel cell polyomavirus infected Merkel cell carcinoma cells require expression of viral T antigens. J. Virol. 2010, 84, 7064–7072. [Google Scholar]
- Shuda, M.; Kwun, H.J.; Feng, H.; Chang, Y.; Moore, P.S. Human Merkel cell polyomavirus small T antigen is an oncoprotein targeting the 4E-BP1 translation regulator. J. Clin. Invest. 2011, 9, 3623–3634. [Google Scholar]
- Pastrana, D.V.; Tolstov, Y.L.; Becker, J.C.; Moore, P.S.; Chang, Y.; Buck, C.B. Quantitation of human seroresponsiveness to Merkel cell polyomavirus. PLoS Pathog. 2009, 5, e1000578. [Google Scholar] [CrossRef] [Green Version]
- Sihto, H.; Kukko, H.; Koljonen, V.; Sankila, R.; Böhling, T.; Joensuu, H. Merkel cell polyomavirus infection, large T antigen, retinoblastoma protein and outcome in Merkel cell carcinoma. Clin. Cancer Res. 2011, 17, 4806–4813. [Google Scholar] [CrossRef]
- Heath, M.; Jaimes, N.; Lemos, B.; Mostaghimi, A.; Wang, LC.; Peñas, PF.; Nghiem, P. Clinical characteristics of Merkel cell carcinoma at diagnosis in 195 patients: The AEIOU features. J. Am. Acad. Dermatol. 2008, 58, 375–381. [Google Scholar] [CrossRef]
- Buell, J.F.; Trofe, J.; Hanaway, M.J.; Beebe, T.M.; Gross, T.G.; Alloway, R.R.; First, M.R.; Woodle, E.S. Immunosuppression and Merkelcell cancer. Transplant. Proc. 2002, 34, 1780–1781. [Google Scholar]
- Penn, I.; First, M.R. Merkel’s cell carcinoma in organrecipients: Report of 41 cases. Transplantation 1999, 68, 1717–1721. [Google Scholar] [CrossRef]
- Koljonen, V.; Kukko, H.; Tukiainen, E.; Böhling, T.; Sankila, R.; Pukkala, E.; Sihto, H.; Joensuu, H.; Kyllönen, L.; Mäkisalo, H. Incidence of Merkel cell carcinoma in renal transplant recipients. Nephrol. Dial. Transplant. 2009, 24, 3231–3235. [Google Scholar] [CrossRef]
- Tadmor, T.; Aviv, A.; Polliack, A. Merkel cell carcinoma, chronic lymphocytic leukemia and other lymphoproliferative disorders: An old bond with possible new viral ties. Ann. Oncol. 2011, 22, 250–256. [Google Scholar] [CrossRef]
- Brewer, J.D.; Shanafelt, T.D.; Otley, C.C.; Roenigk, R.K.; Cerhan, J.R.; Kay, N.E.; Weaver, A.L.; Call, T.G. Chronic lymphocytic leukemia is associated with decreased survival of patients with malignant melanoma and Merkel cell carcinoma in a SEER population-based study. J. Clin. Oncol. 2012, 30, 843–849. [Google Scholar]
- Shuda, M.; Arora, R.; Kwun, H.J.; Feng, H.; Sarid, R.; Fernández-Figueras, M.T.; Tolstov, Y.; Gjoerup, O.; Mansukhani, M.M.; Swerdlow, S.H.; et al. Human Merkel cell polyomavirus infection I. MCV T antigen expression in Merkel cell carcinoma, lymphoid tissues and lymphoid tumors. Int. J. Cancer 2009, 125, 1243–1249. [Google Scholar] [CrossRef]
- Cimino, P.J., Jr.; Bahler, D.W.; Duncavage, E.J. Detection of Merkelcell polyomavirus in chronic lymphocytic leukemia T-cells. Exp. Mol. Pathol. 2013, 94, 40–44. [Google Scholar] [CrossRef]
- Hemminki, K.; Liu, X.; Ji, J.; Sundquist, J.; Sundquist, K. Kaposi sarcoma and Merkel cell carcinoma after autoimmune disease. Int. J. Cancer 2012, 131, E326–E328. [Google Scholar] [CrossRef]
- Engels, E.A.; Frisch, M.; Goedert, J.J.; Biggar, R.J.; Miller, R.W. Merkel cell carcinoma and HIV infection. Lancet 2001, 359, 497–498. [Google Scholar]
- Tuthill, R.J.; Unger, J.M.; Liu, P.Y.; Flaherty, L.E.; Sondak, V.K. Southwest Oncology Group. Risk assessment in localized primary cutaneous melanoma: A Southwest Oncology Group study evaluating nine factors and a test of the Clark logistic regression prediction model. Am. J. Clin. Pathol. 2002, 118, 504–511. [Google Scholar] [CrossRef]
- Al-Shibli, K.I.; Donnem, T.; Al-Saad, S.; Persson, M.; Bremnes, R.M.; Busund, L.T. Prognostic effect of epithelial and stromal lymphocyte infiltration in non-small cell lung cancer. Clin. Cancer Res. 2008, 14, 5220–5227. [Google Scholar]
- Zhang, L.; Conejo-Garcia, J.R.; Katsaros, D.; Gimotty, P.A.; Massobrio, M.; Regnani, G.; Makrigiannakis, A.; Gray, H.; Schlienger, K.; Liebman, M.N.; Rubin, S.C.; Coukos, G. Intratumoral T cells, recurrence, and survival in epithelial ovarian cancer. N. Engl. J. Med. 2003, 348, 203–213. [Google Scholar] [CrossRef]
- Andea, A.A.; Coit, D.G.; Amin, B.; Busam, K.J. Merkel cell carcinoma: Histologic features and prognosis. Cancer 2008, 113, 2549–2558. [Google Scholar] [CrossRef]
- Llombart, B.; Monteagudo, C.; Lopez-Guerrero, J.A.; Carda, C.; Jorda, E.; Sanmartin, O.; Almenar, S.; Molina, I.; Martin, J.M.; Llombart-Bosch, A. Clinicopathological and immunohistochemical analysis of 20 cases of Merkel cell carcinoma in search of prognostic markers. Histopathology 2005, 46, 622–634. [Google Scholar] [CrossRef]
- Sihto, H.; Böhling, T.; Kavola, H.; Koljonen, V.; Salmi, M.; Jalkanen, S.; Joensuu, H. Tumor infiltrating immune cells and outcome of Merkel cell carcinoma: A population-based study. Clin. Cancer Res. 2012, 18, 2872–2881. [Google Scholar] [CrossRef]
- Paulson, K.G.; Iyer, J.G.; Tegeder, A.R.; Thibodeau, R.; Schelter, J.; Koba, S.; Schrama, D.; Simonson, W.T.; Lemos, B.D.; Byrd, D.R.; et al. Transcriptome-wide studies of Merkel cell carcinoma and validation of intratumoral CD8+ lymphocyte invasion as an independent predictor of survival. J. Clin. Oncol. 2011, 12, 1539–1546. [Google Scholar]
- Zhang, L.; Conejo-Garcia, J.R.; Katsaros, D.; Gimotty, P.A.; Massobrio, M.; Regnani, G.; Makrigiannakis, A.; Gray, H.; Schlienger, K.; Liebman, M.N.; et al. Intratumoral T cells, recurrence, and survival in epithelial ovarian cancer. N. Engl. J. Med. 2003, 348, 203–213. [Google Scholar] [CrossRef]
- Galon, J.; Costes, A.; Sanchez-Cabo, F.; Kirilovsky, A.; Mlecnik, B.; Lagorce-Pagès, C.; Tosolini, M.; Camus, M.; Berger, A.; Wind, P.; et al. Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 2006, 313, 1960–1964. [Google Scholar]
- Mott, R.T.; Smoller, B.R.; Morgan, M.B. Merkel cell carcinoma: A clinicopathologic study with prognostic implications. J. Cutan. Pathol. 2004, 31, 217–223. [Google Scholar] [CrossRef]
- Wang, W.; Hodkinson, P.; McLaren, F.; MacKinnon, A.; Wallace, W.; Howie, S.; Sethi, T. Small cell lung cancer tumour cells induce regulatory T lymphocytes, and patient survival correlates negatively with FOXP3+ cells in tumour infiltrate. Int. J. Cancer 2012, 131, E928–E937. [Google Scholar] [CrossRef]
- Gooden, M.J.M.; de Bock, G.H.; Leffers, N.; Daemen, T.; Nijman, H.W. The prognostic influence of tumor-infiltrating lymphocytes in cancer: A systematic review with meta-analysis. Br. J. Cancer 2011, 105, 93–103. [Google Scholar]
- Jensen, T.O.; Schmidt, H.; Møller, H.J.; Høyer, M.; Maniecki, M.B.; Sjoegren, P.; Christensen, I.J.; Steiniche, T. Macrophage markers in serum and tumor have prognostic impact in American Joint Committee on Cancer stage I/II melanoma. J. Clin. Oncol. 2009, 27, 3330–3337. [Google Scholar]
- Montero, A.J.; Diaz-Montero, C.M.; Kyriakopoulos, C.E.; Bronte, V.; Mandruzzato, S. Myeloid-derivedsuppressorcells in cancer patients: A clinical perspective. J. Immunother. 2012, 35, 107–115. [Google Scholar] [CrossRef]
- Zitvogel, L.; Kepp, O.; Aymeric, L.; Ma, Y.; Locher, C.; Delahaye, N.F.; André, F.; Kroemer, G. Integration of host-related signatures with cancer cell-derived predictors for the optimal management of anticancer chemotherapy. Cancer Res. 2010, 70, 9538–9543. [Google Scholar]
- Arany, I.; Tyring, S.K. Status of cytokine and antigen presentation genes in Merkel cell carcinoma of the skin. J. Cutan. Med. Surg. 1998, 2, 138–141. [Google Scholar]
- Connelly, T. Regarding complete spontaneous regression of Merkel cell carcinoma. Dermatol. Surg. 2009, 35, 721. [Google Scholar] [CrossRef]
- Allen, P.J.; Bowne, W.B.; Jaques, D.P.; Brennan, M.F.; Busam, K.; Coit, D.G. Merkel cell carcinoma: Prognosis and treatment from a single institution. J. Clinl. Oncol. 2005, 23, 2300–2309. [Google Scholar] [CrossRef]
- Hui, A.; Stillie, A.; Seel, M.; Ainslie, J. Merkel cell carcinoma: 27 Year experience at the Peter MacCallum Cancer Centre. Int. J. Radiat. Oncol. Biol. Phys. 2011, 80, 1430–1435. [Google Scholar] [CrossRef]
- Foote, M.; Veness, M.; Zarate, D.; Poulsen, M. Merkel cell carcinoma: The prognostic implications of an occult primary in stage IIIB (nodal) disease. J. Am. Acad. Dermatol. 2012, 67, 395–399. [Google Scholar] [CrossRef]
- Deneve, J.L.; Messina, J.L.; Marzban, S.S.; Gonzalez, R.J.; Walls, B.M.; Fisher, K.J.; Chen, Y.A.; Cruse, C.W.; Sondak, V.K.; Zager, J.S. Merkel cell carcinoma of unknown primary origin. Ann. Surg. Oncol. 2012, 19, 2360–2366. [Google Scholar]
- Wooff, J.C.; Trites, J.R.; Walsh, N.M.; Bullock, M.J. Complete spontaneous regression of metastatic merkel cell carcinoma: A case report and review of the literature. Am. J. Dermatopathol. 2010, 32, 614–617. [Google Scholar] [CrossRef]
- Cotter, S.E.; Dunn, G.P.; Collins, K.M.; Sahni, D.; Zukotynski, K.A.; Hansen, J.L.; O’Farrell, D.A.; Ng, A.K.; Devlin, P.M.; Wang, L.C. Abscopal effect in a patient with metastatic Merkel cell carcinoma following radiation therapy: Potential role of induced antitumor immunity. Arch. Dermatol. 2011, 147, 870–872. [Google Scholar] [CrossRef]
- Kayashima, K.; Ono, T.; Johno, M.; Kojo, Y.; Yamashita, N.; Matsunaga, W. Spontaneous regression in Merkel cell (neuroendocrine) carcinoma of the skin. Arch. Dermatol. 1991, 127, 550–553. [Google Scholar] [CrossRef]
- Maruo, K.; Kayashima, K.I.; Ono, T. Regressing Merkel cel carcinoma-a case showing replacement of tumour cells by foamy cells. Br. J. Dermatol. 2000, 142, 1184–1189. [Google Scholar] [CrossRef]
- Vesely, M.J.; Murray, DJ.; Neligan, P.C.; Novak, C.B.; Gullane, P.J.; Ghazarian, D. Complete spontaneous regression in Merkel cell carcinoma. J. Plast. Reconstr. Aesthet. Surg. 2008, 61, 165–171. [Google Scholar] [CrossRef]
- Inoue, T.; Yoneda, K.; Manabe, M.; Demitsu, T. Spontaneous regression of merkel cell carcinoma: A comparative study of TUNEL index and tumor-infiltrating lymphocytes between spontaneous regression and non-regression group. J. Dermatol. Sci. 2000, 24, 203–211. [Google Scholar] [CrossRef]
- Koba, S.; Paulson, K.G.; Nagase, K.; Tegeder, A.; Thibodeau, R.; Iyer, J.G.; Narisawa, Y.; Nghiem, P. Diagnostic biopsy does not commonly induce intratumoral CD8 T cell infiltration in merkel cell carcinoma. PLoS One 2012, 7, e41465. [Google Scholar]
- Richetta, A.G.; Mancini, M.; Torroni, A.; Lorè, B.; Iannetti, G.; Sardella, B.; Calvieri, S. Total spontaneous regression of advanced merkel cell carcinoma after biopsy: Review and a new case. Dermatol. Surg. 2008, 34, 815–822. [Google Scholar] [CrossRef]
- Junquera, L.; Torre, A.; Vicente, J.C.; García-Consuegra, L.; Fresno, M.F. Complete spontaneous regression of Merkel cell carcinoma. Ann. Otol. Rhinol. Laryngol. 2005, 114, 376–380. [Google Scholar]
- Chiu, L.S.; Choi, P.C.; Luk, N.M.; Chang, M.; Tang, W.Y. Spontaneous regression of primary cutaneous Epstein-Barr virus-positive, CD30-positive anaplastic large T-cell lymphoma in a heart-transplant recipient. Clin. Exp. Dermatol. 2009, 34, e21–e24. [Google Scholar] [CrossRef]
- Adiguzel, C.; Bozkurt, S.U.; Kaygusuz, I.; Uzay, A.; Tecimer, T.; Bayik, M. Human herpes virus 8-unrelated primary effusion lymphoma-like lymphoma: Report of a rare case and review of the literature. APMIS 2009, 117, 222–229. [Google Scholar] [CrossRef]
- Abe, R.; Ogawa, K.; Maruyama, Y.; Nakamura, N.; Abe, M. Spontaneous regresión of diffuse large B-cell lymphoma harbouring Epstein-Barr virus: A case report and review of the literature. J. Clin. Exp. Hematop. 2007, 47, 23–26. [Google Scholar] [CrossRef]
- Yagi, Y.; Fujisawa, A.; Makiura, M.; Morita, K. Spontaneous regression of Merkel cell carcinoma after biopsy. J. Dermatol. 2009, 36, 312–313. [Google Scholar] [CrossRef]
- Maddison, P.; Lang, B. Paraneoplastic neurological autoimmunity and survival in small-cell lung cancer. J. Neuroimmunol. 2008, 201–202, 159–162. [Google Scholar] [CrossRef]
- Balegno, S.; Ceroni, M.; Corato, M.; Franciotta, D.; Giometto, B.; Marinu-Aktipu, K.; Nano, R. Antibodies to cerebellar nerve fibres in two patients with paraneoplastic cerebellar ataxia. Anticancer Res. 2005, 25, 3211–3214. [Google Scholar]
- Lopez, M.C.; Pericay, C.; Agustí, M.; Martinez-Gallo, M.; Bordes, R.; Gelpí, C. Merkel cell carcinoma associated with a paraneoplastic neurological syndrome. Histopathology 2004, 44, 628–629. [Google Scholar] [CrossRef]
- Greenlee, J.E.; Steffens, J.D.; Clawson, S.A.; Hill, K.; Dalmau, J. Anti-Hu antibodies in Merkel cell carcinoma. Ann. Neurol. 2002, 52, 111–115. [Google Scholar] [CrossRef]
- Cher, L.M.; Hochberg, F.H.; Teruya, J.; Nitschke, M.; Valenzuela, R.F.; Schmahmann, J.D.; Herbert, M.; Rosas, H.D.; Stowell, C. Therapy for paraneoplastic neurologic syndromes in sixpatients with protein A column immunoadsorption. Cancer 1995, 75, 1678–1683. [Google Scholar] [CrossRef]
- Eggers, S.D.; Salomao, D.R.; Dinapoli, R.P.; Vernino, S. Paraneoplastic and metastatic neurologic complications of Merkel cell carcinoma. Mayo Clin. Proc. 2001, 76, 327–330. [Google Scholar] [CrossRef]
- Muirhead, R.; Ritchie, D.M. Partial regression of Merkel cell carcinoma in response to withdrawal of azathioprine in an immunosuppression-induced case of metastatic Merkel cell carcinoma. Clin. Oncol. (R. Coll. Radiol.) 2007, 19, 96. [Google Scholar] [CrossRef]
- Friedlaender, M.M.; Rubinger, D.; Rosenbaum, E.; Amir, G.; Siguencia, E. Temporary regression of Merkel cell carcinoma metastases after cessation of cyclosporine. Transplantation 2002, 73, 1849–1850. [Google Scholar] [CrossRef]
- Kauffman, H.M.; Cherikh, W.S.; Cheng, Y.; Hanto, D.W.; Kahan, B.D. Maintenance immunosuppression with target-of-rapamycin inhibitors is associated with a reduced incidence of de novo malignancies. Transplantation 2005, 80, 883–889. [Google Scholar] [CrossRef]
- Tolstov, Y.L.; Pastrana, D.V.; Feng, H.; Becker, J.C.; Jenkins, F.J.; Moschos, S.; Chang, Y.; Buck, C.B.; Moore, P.S. Human Merkel cell polyomavirus infection II. MCV is a common human infection that can be detected by conformational capsid epitope immunoassays. Int. J. Cancer 2009, 125, 1250–1256. [Google Scholar] [CrossRef]
- Chen, T.; Hedman, L.; Mattila, P.S.; Jartti, T.; Ruuskanen, O.; Söderlund-Venermo, M.; Hedman, K. Serological evidence of Merkel cell polyomavirus primary infections in childhood. J. Clin. Virol. 2010, 50, 125–129. [Google Scholar]
- Kean, J.M.; Rao, S.; Wang, M.; Garcea, R.L. Seroepidemiology of human polyomaviruses. PLoS Pathog. 2009, 5, e1000363. [Google Scholar] [CrossRef]
- Carter, J.J.; Paulson, K.G.; Wipf, G.C.; Miranda, D.; Madeleine, M.M.; Johnson, L.G.; Lemos, B.D.; Lee, S.; Warcola, A.H.; Iyer, J.G.; et al. Association of Merkel cell polyomavirus-specific antibodies with Merkel cell carcinoma. J. Natl. Cancer Inst. 2009, 101, 1510–1522. [Google Scholar] [CrossRef]
- Paulson, K.G.; Carter, J.J.; Johnson, L.G.; Cahill, K.W.; Iyer, J.G.; Schrama, D.; Becker, J.C.; Madeleine, M.M.; Nghiem, P.; Galloway, D.A. Antibodies to Merkel cell polyomavirus T-antigen oncoproteins reflect tumor burden in Merkel cell carcinoma patients. Cancer Res. 2010, 70, 8388–8397. [Google Scholar]
- Besson, C.; Amiel, C.; Le-Pendeven, C.; Brice, P.; Ferme, C.; Carde, P.; Hermine, O.; Raphael, M.; Abel, L.; Nicolas, J.C. Positive correlation between Epstein-Barr virus viral load and anti-viral capsid immunoglobulin G titers determined for Hodgkin’s lymphoma patients and their relatives. J. Clin. Microbiol. 2006, 44, 47–50. [Google Scholar]
- Morand-Joubert, L.; Mariotti, M.; Reed, D.; Petit, J.C.; Lefrere, J.J. Correlation between viral DNA load and serum anti p19 antibody concentration in symptomless human T-lymphotropic virus type-I (HTLV-I)-infected individuals. Int. J. Cancer 1995, 60, 156–159. [Google Scholar]
- Hung, I.F.; To, K.K.; Lee, C.K.; Lin, C.K.; Chan, J.F.; Tse, H.; Cheng, V.C.; Chen, H.; Ho, P.L.; Tse, C.W.; et al. Effect of clinical and virological parameters on the level of neutralizing antibody against pandemic influenza A virus H1N1 2009. Clin. Infect. Dis. 2010, 51, 274–279. [Google Scholar] [CrossRef] [Green Version]
- Randhawa, P.; Bohl, D.; Brennan, D.; Ruppert, K.; Ramaswami, B.; Storch, G.; March, J.; Shapiro, R.; Viscidi, R. longitudinal analysis of levels of immunoglobulins against BK virus capsid proteins in kidney transplant recipients. Clin. Vaccine Immunol. 2008, 15, 1564–1571. [Google Scholar] [CrossRef]
- Lundstig, A.; Stattin, P.; Persson, K.; Sasnauskas, K.; Viscidi, R.P.; Gislefoss, R.E.; Dillner, J. No excess risk for colorectal cancer among subjects seropositive for the JC polyomavirus. Int. J. Cancer 2007, 121, 1098–1102. [Google Scholar] [CrossRef]
- Faust, H.; Pastrana, D.V.; Buck, C.B.; Dillner, J.; Ekström, J. Antibodies to Merkel cell polyomavirus correlate to presence of viral DNA in the skin. J. Infect. Dis. 2011, 203, 1096–1100. [Google Scholar] [CrossRef]
- Pastrana, D.V.; Wieland, U.; Silling, S.; Buck, C.B.; Pfister, H. Positive correlation between Merkel cell polyomavirus viral load and capsid-specific antibody titer. Med. Microbiol. Immunol. 2012, 201, 17–23. [Google Scholar] [CrossRef]
- Nakamura, T.; Sato, Y.; Watanabe, D.; Ito, H.; Shimonohara, N.; Tsuji, T.; Nakajima, N.; Suzuki, Y.; Matsuo, K.; Nakagawa, H.; et al. Nuclear localization of Merkel cell polyomavirus large T-antigen in Merkel cell carcinoma. Virology 2010, 398, 273–279. [Google Scholar] [CrossRef]
- Touzé, A.; Le Bidre, E.; Laude, H.; Fleury, M.J.; Cazal, R.; Arnold, F.; Carlotti, A.; Maubec, E.; Aubin, F.; Avril, M.F.; et al. High levels of antibodies against merkel cell polyomavirus identify a subset of patients with merkel cell carcinoma with better clinical outcome. J. Clin. Oncol. 2011, 29, 1612–1619. [Google Scholar]
- Winter, S.F.; Sekido, Y.; Minna, J.D.; McIntire, D.; Johnson, B.E.; Gazdar, A.F.; Carbone, D.P. Antibodies against autologous tumor cell proteins in patients with small-cell lung cancer: Association with improved survival. J. Natl. Cancer Inst. 1993, 85, 2012–2018. [Google Scholar] [CrossRef]
- Gheuens, S.; Bord, E.; Kesari, S.; Simpson, D.M.; Gandhi, R.T.; Clifford, D.B.; Berger, J.R.; Ngo, L.; Koralnik, I.J. Role of CD4+ and CD8+ T-cell responses against JC virus in the outcome of patients with progressive multifocal leukoencephalopathy (PML) and PML with immune reconstitution inflammatory syndrome. J. Virol. 2011, 85, 7256–7263. [Google Scholar]
- Schneidawind, D.; Schmitt, A.; Wiesneth, M.; Mertens, T.; Bunjes, D.; Freund, M.; Schmitt, M. Polyomavirus BK-specific CD8+ T cell responses in patients after allogeneic stem cell transplant. Leuk. Lymphoma 2010, 51, 1055–1062. [Google Scholar] [CrossRef]
- Chan, P.K.; Liu, S.J.; Cheung, J.L.; Cheung, T.H.; Yeo, W.; Chong, P.; Man, S. T-cell response to human papillomavirus type 52 L1, E6, and E7 peptides in women with transient infection, cervical intraepithelial neoplasia, and invasive cancer. J. Med. Virol. 2011, 83, 1023–1030. [Google Scholar] [CrossRef]
- Rehermann, B.; Nascimbeni, M. Immunology of hepatitis B virus and hepatitis C virus infection. Nat. Rev. Immunol. 2005, 5, 215–229. [Google Scholar] [CrossRef]
- Kumar, A.; Chen, T.; Pakkanen, S.; Kantele, A.; Söderlund-Venermo, M.; Hedman, K.; Franssila, R. T-helper cell-mediated proliferation and cytokine responses against recombinant Merkel cell polyomavirus-like particles. PLoS One 2011, 6, e25751. [Google Scholar]
- Iyer, J.G.; Afanasiev, O.K.; McClurkan, C.; Paulson, K.; Nagase, K.; Jing, L.; Marshak, J.O.; Dong, L.; Carter, J.; Lai, I.; et al. Merkelcell polyomavirus-specific CD8+ and CD4+ T-cell responses identified in Merkelcell carcinomas and blood. Clin. Cancer Res. 2011, 17, 6671–6680. [Google Scholar] [CrossRef]
- Mishra, R.; Chen, A.T.; Welsh, R.M.; Szmolanyi-Tsuda, E. NK cells and gammadelta T cells mediate resistance to polyomavirus-induced tumors. PLoS Pathog. 2010, 6, e1000924. [Google Scholar] [CrossRef]
- Co, J.K.; Verma, S.; Gurjav, U.; Sumibcay, L.; Nerurkar, V.R. Interferon-alpha and -beta restrict polyomavirus JC replication in primary human fetal glial cells: Implications for progressive multifocal leukoencephalopathy therapy. J. Infect. Dis. 2007, 96, 2–8. [Google Scholar]
- Abend, J.R.; Low, J.A.; Imperiale, M.J. Inhibitory effect of gamma interferon on BK virus gene expression and replication. J. Virol. 2007, 81, 272–279. [Google Scholar] [CrossRef]
- Krasagakis, K.; Kruger-Krasagakis, S.; Tzanakakis, G.N.; Darivianaki, K.; Stathopoulos, E.N.; Tosca, A.D. Interferon-alpha inhibits proliferation and induces apoptosis of merkel cell carcinoma in vitro. Cancer Invest. 2008, 26, 562–568. [Google Scholar] [CrossRef]
- Willmes, C.; Adam, C.; Alb, M.; Völkert, L.; Houben, R.; Becker, J.C.; Schrama, D. Type I and II IFNs inhibit Merkel cell carcinoma via modulation of the Merkelcell polyomavirus T antigens. Cancer Res. 2012, 72, 2120–2128. [Google Scholar] [CrossRef]
- Durand, J.M.; Weiller, C.; Richard, M.A.; Portal, I.; Mongin, M. Treatment of Merkel cell tumor with interferon-alpha-2b. Br. J. Dermatol. 1991, 124, 509. [Google Scholar]
- Nakajima, H.; Takaishi, M.; Yamamoto, M.; Kamijima, R.; Kodama, H.; Tarutani, M.; Sano, S. Screening of the specificpolyomavirus as diagnostic and prognostictools for Merkelcellcarcinoma. J. Dermatol. Sci. 2009, 56, 211–213. [Google Scholar]
- Bajetta, E.; Zilembo, N.; di Bartolomeo, M.; di Leo, A.; Pilotti, S.; Bochicchio, A.M.; Castellani, R.; Buzzoni, R.; Celio, L.; Dogliotti, L.; et al. Treatment of metastatic carcinoids and other neuroendocrine tumors with recombinant interferon-alpha-2a. A study by the Italian Trials in Medical Oncology Group. Cancer 1993, 72, 3099–3105. [Google Scholar] [CrossRef]
- Biver-Dalle, C.; Nguyen, T.; Touzé, A.; Saccomani, C.; Penz, S.; Cunat-Peultier, S.; Riou-Gotta, M.O.; Humbert, P.; Coursaget, P.; Aubin, F. Use of interferon-alpha in two patients with Merkel cell carcinoma positive for Merkel cell polyomavirus. Acta Oncol. 2011, 50, 479–480. [Google Scholar] [CrossRef]
- Matsushita, E.; Hayashi, N.; Fukushima, A.; Ueno, H. Evaluation of treatment and prognosis of Merkel cell carcinoma of the eyelid in Japan. Nippon Ganka Gakkai Zasshi 2007, 111, 459–462. [Google Scholar]
- Hata, Y.; Matsuka, K.; Ito, O.; Matsuda, H.; Furuichi, H.; Konstantinos, A.; Nuri, B. Two cases of Merkel cell carcinoma cured by intratumor injection of natural human tumor necrosis factor. Plast. Reconstr. Surg. 1997, 99, 547–553. [Google Scholar] [CrossRef]
- Olieman, A.F.; Liénard, D.; Eggermont, A.M.; Kroon, B.B.; Lejeune, F.J.; Hoekstra, H.J.; Koops, H.S. Hyperthermic isolated limb perfusion with tumor necrosis factor alpha, interferon gamma, and melphalan for locally advanced nonmelanoma skin tumors of the extremities: A multicenter study. Arch. Surg. 1999, 134, 303–307. [Google Scholar] [CrossRef]
- Burack, J.; Altschuler, E.L. Sustained remission of metastatic Merkel cell carcinoma with treatment of HIV infection. J. R. Soc. Med. 2003, 96, 238–239. [Google Scholar] [CrossRef]
- OMS-I100—Phase II Metastatic Melanoma Clinical Trial. Available online: http://oncosec.com/index.php/oms/clinical-trials/ (accessed on 30 January 2013).
- Chapuis, A.G.; Afanasiev, O.; Iyer, J.; Parvathaneni, U.; Hwang, J.H.; Lai, I.; Roberts, I.; Farrar, E.; Koelle, D.; Nghiem, P.; et al. Anti-polyomavirus CTL targeting Merkel cell carcinoma combined with MHC class I upregulation induces tumor regression. Cancer Res. 2012, 72, Abstract LB-430. [Google Scholar]
- Herrmann, G.; Groth, W.; Krieg, T.; Mauch, C. Complete remission of Merkel cell carcinoma of the scalp with local and regional metastases after topical treatment with dinitrochlorbenzol. J. Am. Acad. Dermatol. 2004, 50, 965–969. [Google Scholar] [CrossRef]
- Scott, D.R. Apparent response of cutaneous Merkel cell tumor to topical imiquimod. Cutis 2006, 77, 109–110. [Google Scholar]
- Balducci, M.; de Bari, B.; Manfrida, S.; D’Agostino, G.R.; Valentini, V. Treatment of Merkel cell carcinoma with radiotherapy and imiquimod (Aldara): A case report. Tumori 2010, 96, 508–511. [Google Scholar]
- Zeng, Q.; Gomez, B.P.; Viscidi, R.P.; Peng, S.; He, L.; Ma, B.; Wu, T.C.; Hung, C.F. Development of a DNA vaccine targeting Merkelcell polyomavirus. Vaccine 2012, 30, 1322–1329. [Google Scholar]
- Gomez, B.P.; Wang, C.J.; Viscidi, R.P.; Peng, S.; He, L.; Wu, T.C.; Hung, C.F. Strategy for eliciting antigen-specific CD8+ T cell-mediated immune response against a cryptic CTL epitope of merkel cell polyomavirus large T antigen. Cell Biosci. 2012, 2, 36. [Google Scholar] [CrossRef]
- Kurzen, H.; Kaul, S.; Egner, U.; Deichmann, M.; Hartschuh, W. Expression of MUC 1 and Ep-CAM in Merkelcell carcinomas: Implications for immunotherapy. Arch. Dermatol. Res. 2003, 295, 146–154. [Google Scholar] [CrossRef]
- Erickson, K.D.; Garcea, R.L.; Tsai, B. Ganglioside GT1b is a putative host cell receptor for the Merkel cell polyomavirus. J. Virol. 2009, 83, 10275–10279. [Google Scholar] [CrossRef]
- Beatson, R.E.; Taylor-Papadimitriou, J.; Burchell, J.M. MUC1 immunotherapy. Immunotherapy 2010, 2, 305–327. [Google Scholar] [CrossRef]
- Sithambaram, D.; Palanivelu, S.; Subramanian, K.; Sahoo, S.; Verma, R.S. Specific targeting of Ep-CAM in various carcinomas by novel monoclonal antibodies. Hybridoma (Larchmt.) 2011, 30, 511–518. [Google Scholar] [CrossRef]
- Giaccone, G.; Debruyne, C.; Felip, E.; Chapman, P.B.; Grant, S.C.; Millward, M.; Thiberville, L.; D’addario, G.; Coens, C.; Rome, L.S.; et al. Phase III study of adjuvant vaccination with Bec2/Bacille Calmette-Guerin in responding patients with limited-disease small-cell lung cancer (European Organisation for Research and Treatment of Cancer 08971–08971B; Silva study). J. Clin. Oncol. 2005, 23, 6854–6864. [Google Scholar]
- Lee, S.; Paulson, K.G.; Murchison, E.P.; Afanasiev, O.K.; Alkan, C.; Leonard, J.H.; Byrd, D.R.; Hannon, G.J.; Nghiem, P. Identification and validation of a novel mature microRNA encoded by the Merkel cell polyomavirus in human Merkel cell carcinomas. J. Clin. Virol. 2011, 52, 272–275. [Google Scholar] [CrossRef]
- Jackson, A.; Linsley, P.S. The therapeutic potential of microRNA modulation. Discov. Med. 2010, 9, 311–348. [Google Scholar]
- Hodi, F.S.; O’Day, S.J.; McDermott, D.F.; Weber, R.W.; Sosman, J.A.; Haanen, J.B.; Gonzalez, R.; Robert, C.; Schadendorf, D.; Hassel, J.C.; et al. Improved survival with ipilimumab in patients with metastatic melanoma. N. Engl. J. Med. 2010, 363, 711–723. [Google Scholar]
- Reck, M.; Bondarenko, I.; Luft, A.; Serwatowski, P.; Barlesi, F.; Chacko, R.; Sebastian, M.; Lu, H.; Cuillerot, J.M.; Lynch, T.J. Ipilimumab in combination with paclitaxel and carboplatin as first-line therapy in extensive-disease-small-cell lung cancer: Results from a randomized, double-blind, multicenter phase 2 trial. Ann. Oncol. 2012. [Google Scholar] [CrossRef]
- Brahmer, J.R.; Tykodi, S.S.; Chow, L.Q.; Hwu, W.J.; Topalian, S.L.; Hwu, P.; Drake, C.G.; Camacho, L.H.; Kauh, J.; Odunsi, K.; et al. Safety and activity of anti-PD-L1 antibody in patients with advanced cancer. N. Engl. J. Med. 2012, 366, 2455–2465. [Google Scholar] [CrossRef]
- Topalian, S.L.; Hodi, F.S.; Brahmer, J.R.; Gettinger, S.N.; Smith, D.C.; McDermott, D.F.; Powderly, J.D.; Carvajal, R.D.; Sosman, J.A.; Atkins, M.B.; et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N. Engl. J. Med. 2012, 366, 2443–2454. [Google Scholar] [CrossRef]
- Wolchok, J.D.; Yang, A.S.; Weber, J.S. Immune regulatory antibodies: Are they the next advance? Cancer J. 2010, 16, 311–317. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Triozzi, P.L.; Fernandez, A.P. The Role of the Immune Response in Merkel Cell Carcinoma. Cancers 2013, 5, 234-254. https://doi.org/10.3390/cancers5010234
Triozzi PL, Fernandez AP. The Role of the Immune Response in Merkel Cell Carcinoma. Cancers. 2013; 5(1):234-254. https://doi.org/10.3390/cancers5010234
Chicago/Turabian StyleTriozzi, Pierre L., and Anthony P. Fernandez. 2013. "The Role of the Immune Response in Merkel Cell Carcinoma" Cancers 5, no. 1: 234-254. https://doi.org/10.3390/cancers5010234




