The Critical Impact of HIF-1a on Gastric Cancer Biology
Abstract
:1. Introduction

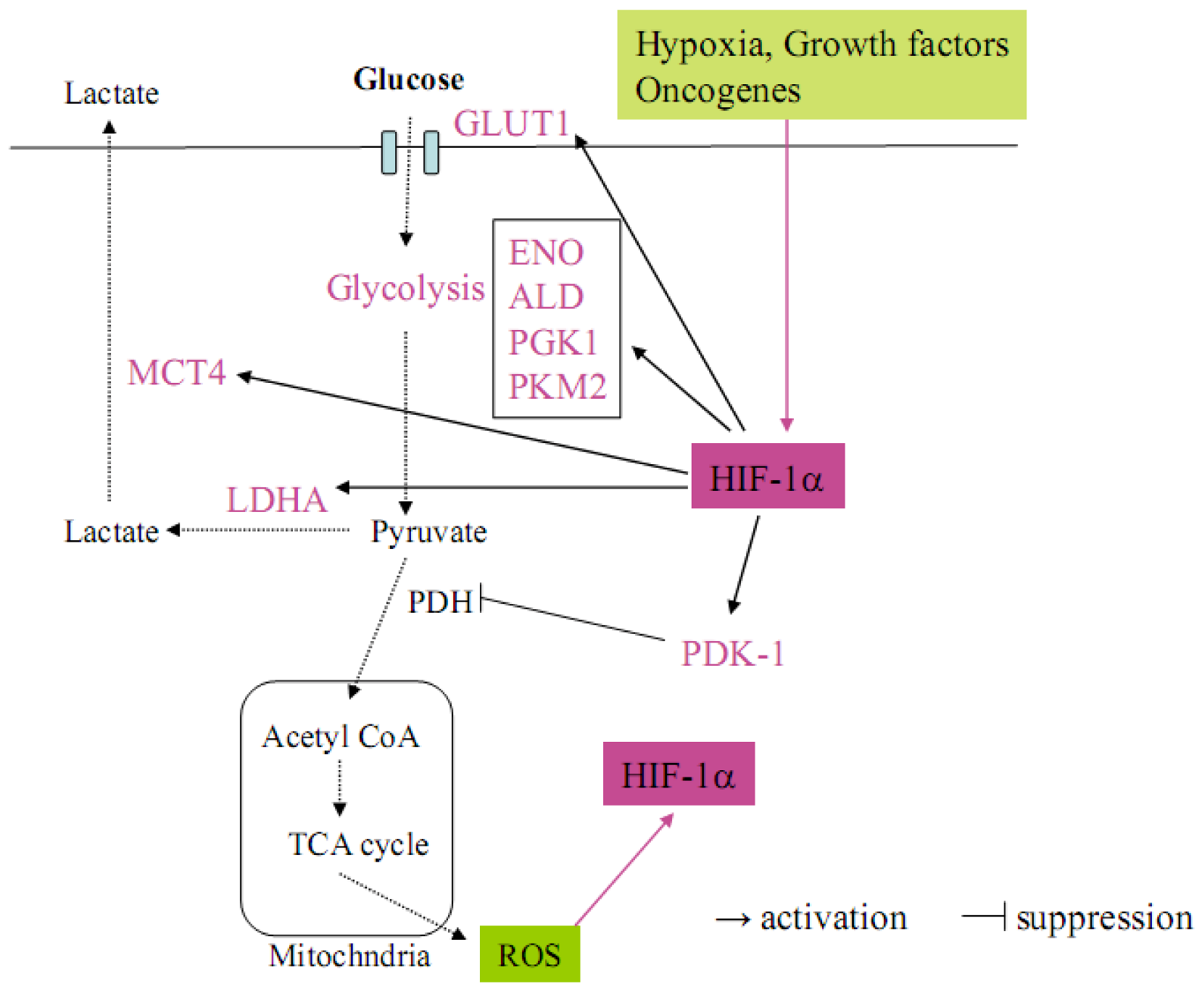
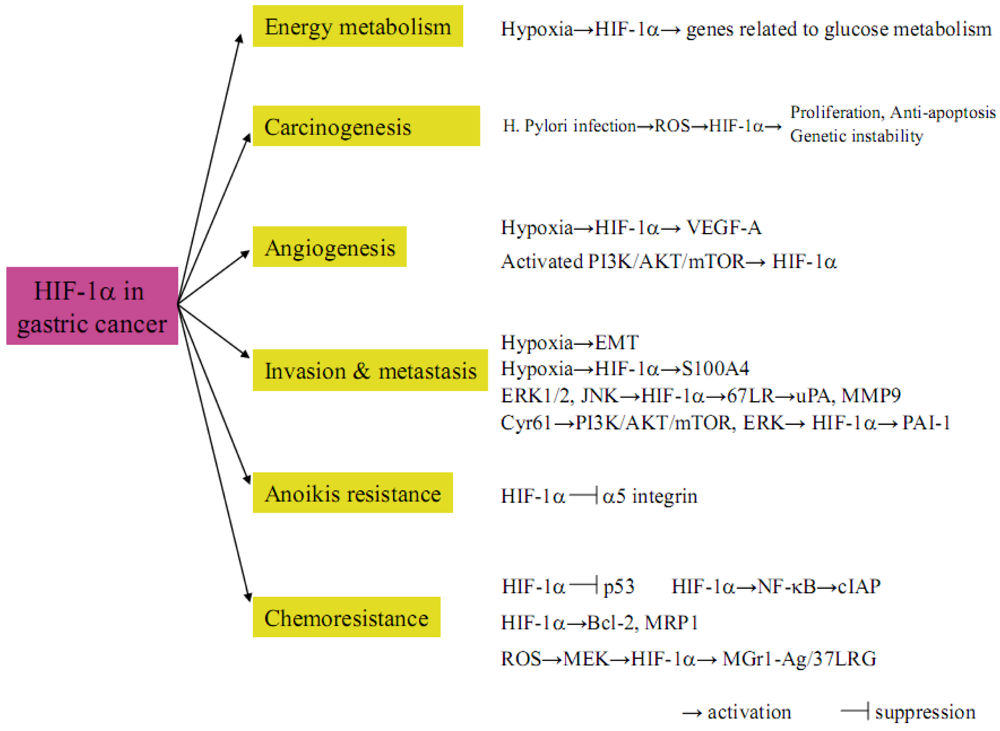
2. Glucose Metabolism
3. Carcinogenesis
4. Angiogenesis
5. Cancer Invasion and Metastasis
6. Anoikis Resistance
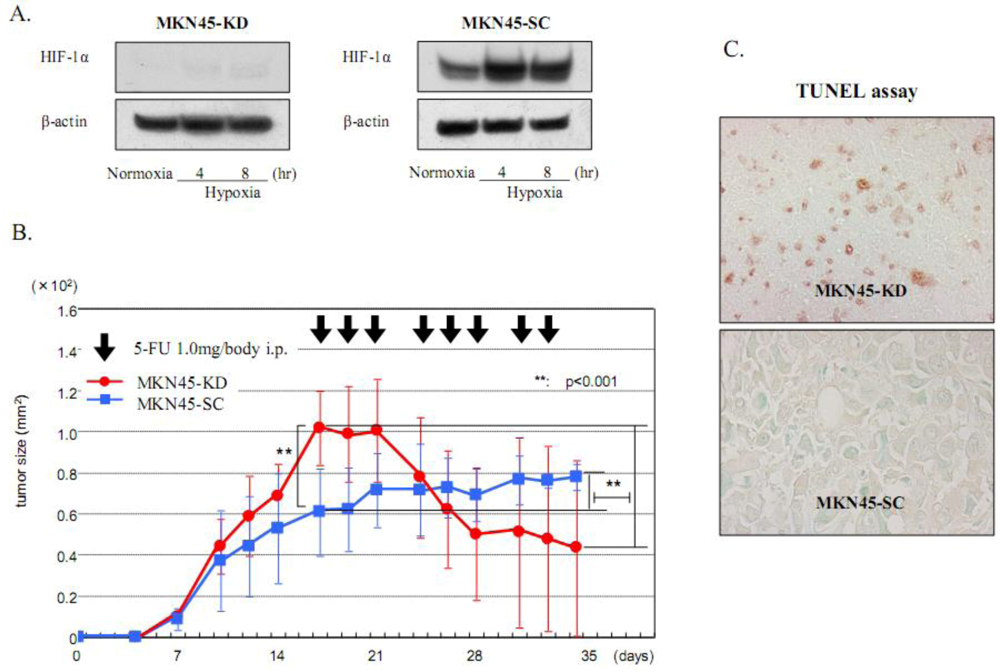
7. Chemoresistance

8. Conclusions
Conflict of Interest
References
- Hallis, A.L. Hypoxia, a key regulatory factor in tumour growth. Nat. Rev. Cancer 2002, 2, 38–47. [Google Scholar]
- Semenza, G.L. HIF-1 and tumor progression: Pathophysiology and therapeutics. Trends Mol. Med. 2002, 8, S62–S67. [Google Scholar] [CrossRef]
- Lee, B.L.; Kim, W.H.; Jung, J.; Cho, S.J.; Park, J.W.; Kim, J.; Chung, H.Y.; Chang, M.S.; Nam, S.Y. A hypoxia-independent up-regulation of hypoxia-inducible factor-1 by AKT contributes to angiogenesis in human gastric cancer. Carcinogenesis 2008, 29, 44–51. [Google Scholar]
- Chun, S.Y.; Johnson, C.; Washbum, J.G.; Cruz-Correa, M.; Dang, D.T.; Dang, L.H. Oncogenic KRAS modurates mitochondrial metabolism in human colon cancer cells by inducing HIF-1α and HIF-2α target genes. Mol. Cancer 2010, 9, 293. [Google Scholar]
- Lu, X.; Kang, Y. Hypoxia and hypoxia-inducible factors (HIFs): Master regulators of metastasis. Clin. Cancer Res. 2010, 16, 5928–5935. [Google Scholar]
- Mole, D.R.; Blancher, C.; Copley, R.R.; Pollard, P.J.; Gleadle, J.M.; Ragoussis, J.; Ratcliffe, P.J. Genome-wide association of hypoxia-inducible factor (HIF)-1α and HIF-2α DNA binding with expression profiling of hypoxia-inducible transcripts. J. Biol. Chem. 2009, 284, 16767–16775. [Google Scholar]
- Xia, X.; Lemieux, M.E.; Li, W.; Carroll, J.S.; Brown, M.; Liu, X.S.; Kung, A.L. Integrative analysis of HIF binding and transactivation reveals its role in maintaining histone methylation homeostasis. Proc. Natl. Acad. Sci. USA 2009, 106, 4260–4265. [Google Scholar]
- Rohwer, N.; Cramer, T. HIFs as central regulators of gastric cancer pathogenesis. Cancer Biol. Ther. 2010, 10, 383–385. [Google Scholar]
- Semenza, G.L. HIF-1: Upstream and downstream of cancer metabolism. Curr. Opin. Genet. Dev. 2010, 20, 51. [Google Scholar] [CrossRef]
- Warburg, O. On the origin of cancer cells. Science 1956, 123, 309–314. [Google Scholar]
- Papandreu, I.; Cairns, R.A.; Fontana, L.; Lim, A.L.; Denko, N.C. HIF-1 mediates adaptation to hypoxia by actively downregulating mitochondrial oxygen consumption. Cell Metab. 2006, 3, 187–197. [Google Scholar] [CrossRef]
- Song, I.S.; Wang, A.G.; Yoon, S.Y.; Kim, J.M.; Kim, J.H.; Lee, D.S.; Kim, N.S. Regulation of glucose metabolism-related genes anf VEGF by HIF-1α and HIF-1β, but not HIF-2α, in gastric cancer. Exp. Mol. Med. 2009, 41, 51–58. [Google Scholar] [CrossRef]
- Griffiths, E.A.; Pritchard, S.A.; Valentine, H.R.; Whitchelo, N.; Bishop, P.W.; Ebert, M.P.; Price, P.M.; Welch, I.M.; West, C.M.L. Hypoxia-inducible factor-1α expression in the gastric carcinogenesis sequence and its prognostic role in gastric and gastro-esophageal adenocarcinoma. Br. J. Cancer 2007, 96, 95–103. [Google Scholar]
- Koshiji, M.; To, K.K.; Hammer, S.; Kumamoto, K.; Harris, A.L.; Modrich, P.; Huang, L.E. HIF-1 alpha induces genetic instability by transcriptionally downregulating MutSalpha expression. Mol. Cell. 2005, 17, 793–803. [Google Scholar]
- Kondo, Y.; Arii, S.; Mori, A.; Furutani, M.; Chiba, T.; Imamura, M. Enhancement of angiogenesis, tumor growth, and metastasis by transfection of vascular endothelial growth factor into LoVo human colon cancer cell line. Clin. Cancer Res. 2000, 6, 622–630. [Google Scholar]
- Maeda, K.; Chung, Y.S.; Ogawa, Y.; Takatsuka, S.; Kang, S.M.; Ogawa, M.; Sawada, T.; Sowa, M. Prognostic value of vascular endothelial growth factor expression in gastric cancer. Cancer 1996, 77, 858–863. [Google Scholar] [CrossRef]
- Saito, H.; Tsujitani, S.; Kondo, A.; Ikeguchi, M.; Maeda, M.; Kaibara, N. Expression of vascular endothelial growth factor correlates with hematogenous recurrence in gastric carcinoma. Surgery 1999, 125, 195–201. [Google Scholar] [CrossRef]
- Niedergethmann, M.; Hildenbrand, R.; Wostbrock, B.; Hartel, M.; Sturm, J.W.; Richter, A.; Post, S. High expression of vascular endothelial growth factor predicts early recurrence and poor prognosis after curative resection for ductal adenocarcinoma of the pancreas. Pancreas 2002, 25, 122–129. [Google Scholar]
- Stoeltzing, O.; McCarty, M.F.; Wey, J.S.; Fan, F.; Liu, W.; Belcheva, A.; Bucana, C.D.; Semenza, G.L.; Ellis, L.M. Role of hypoxia-inducible factor 1α in gastric cancer cell growth, angiogenesis, and vessel maturation. J. Natl. Cancer Inst. 2004, 96, 946–956. [Google Scholar]
- Lang, S.A.; Gaumann, A.; Koehl, G.E.; Seidel, U.; Bataille, F.; Klein, D.; Ellis, L.M.; Bolder, U.; Hofstaedter, F.; Schlitt, H.J.; et al. Mammalian target of rapamycin is activated in human gastric cancer and serves as a target for therapy in an experimental model. Int. J. Cancer 2007, 120, 1803–1810. [Google Scholar]
- Miyoshi, A.; Kitajima, Y.; Ide, T.; Nagasawa, H.; Uto, Y.; Hori, H.; Miyazaki, K. Hypoxia accerelates cancer invasion of hepatoma cells by upregulating MMP expression in HIF-1α independent manner. Int. J. Oncol. 2006, 29, 1533–1539. [Google Scholar]
- Ide, T.; Kitajima, Y.; Miyoshi, A.; Ohtsuka, T.; Mitsuno, M.; Ohtaka, K.; Koga, Y.; Miyazaki, K. Tumor-stromal cell interaction under hypoxia increases the invasiveness of pancreatic cancer cells through the hepatocyte growth factor/c-Met pathway. Int. J. Cancer 2006, 119, 2750–2759. [Google Scholar]
- Kitajima, Y.; Ide, T.; Ohtsuka, T.; Miyazaki, K. Induction of hepatocyte growth factor activator gene expression under hypoxia activates the hepatocyte growth factor/c-Met system via hypoxia inducible factor-1 in pancreatic cancer. Cancer Sci. 2008, 99, 1341–1347. [Google Scholar]
- Kato, Y.; Yashiro, M.; Noda, S.; Tendo, M.; Kashiwagi, S.; Doi, Y.; Nishii, T.; Matsuoka, J.; Fuyuhiro, Y.; Shinto, O.; et al. Establishment and characterization of a new hypoxia-resistant cancer cell line, OCUM-12/Hypo, derived from a scirrhous gastric carcinoma. Br. J. Cancer 2010, 102, 898–907. [Google Scholar]
- Rohwer, N.; Lobitz, S.; Daskalow, K.; Jons, T.; Vieth, M.; Schlag, P.M.; Kemmner, W.; Cramer, T.; Hocker, M. HIF-1α determines the metastatic potential of gastric cancer cells. Br. J. Cancer 2009, 100, 772–781. [Google Scholar]
- Zimmer, D.B.; Cornwall, E.H.; Landar, A.; Song, W. The S100 protein family: History, function, and expression. Brain Res. Bull. 1995, 37, 417–429. [Google Scholar] [CrossRef]
- Kohya, N.; Kitajima, Y.; Jiao, W.; Miyazaki, K. Effects of E-cadherin transfection on gene expression of a ballbladder carcinoma cell line: Repression of MTS1/S100A4 gene expression. Int. J. Cancer 2003, 104, 44–53. [Google Scholar]
- Zhang, R.; Fu, H.; Chen, D.; Hua, J.; Hu, Y.; Sun, K.; Sun, X. Subcellular distribution of S100A4 and its transcriptional regulation under hypoxic conditions in gastric cancer cell line BGC823. Cancer Sci. 2010, 101, 1141–1146. [Google Scholar] [CrossRef]
- Liu, L.; Sun, L.; Zhao, P.; Yao, L.; Jin, H.; Liang, S.; Wang, Y.; Zhang, D.; Pang, Y.; Shi, Y.; et al. Hypoxia promotes metastasis in human gastric cancer by up-regulating the 67-lDa laminin receptor. Cancer Sci. 2010, 101, 1653–1660. [Google Scholar] [CrossRef]
- Lin, M.T.; Kuo, I.H.; Chang, C.C.; Chu, C.Y.; Chen, H.Y.; Lin, B.R.; Sureshbabu, M.; Shih, H.J.; Kuo, M.L. Involvement of hypoxia-inducing factor-1α-dependent plasminogen activator-1 up-regulation in Cyr61/CCN1-induced gastric cancer cell invasion. J. Biol. Chem. 2008, 282, 15807–15815. [Google Scholar]
- Rohwer, N.; Welzel, M.; Daskalow, K.; Pfander, D.; Wiedenmann, B.; Detjen, K.; Cramer, T. Hypoxia-inducible factor 1a mediates anoikis resistance via suppression of α5 integrin. Cancer Res. 2008, 68, 10113–10120. [Google Scholar] [CrossRef]
- Nakamura, J.; Kitajima, Y.; Kai, K.; Hashiguchi, K.; Hiraki, M.; Noshiro, H.; Miyazaki, K. HIF-1alpha is an unfavorable determinant of relapse in gastric cancer patients who underwent curative surgery followed by adjuvant 5-FU chemotherapy. Int. J. Cancer 2010, 127, 1158–1171. [Google Scholar]
- Rohwer, N.; Dame, C.; Haugstetter, A.; Wiedenmann, B.; Detjen, K.; Schmitt, C.A.; Cramer, T. Hypoxia-inducible factor 1α determines gastric cancer chemosensitivity via modulation of p53 and NF-kB. PLoS One 2010, 5, e12038. [Google Scholar]
- Liu, L.; Ning, X.; Zhang, H.; Shi, Y.; Guo, C.; Han, S.; Liu, J.; Sun, S.; Han, Z.; Wu, K.; et al. Hypoxia-inducible factor-1a contributes to hypoxia-induced chemoresistance in gastric cancer. Cancer Sci. 2008, 99, 121–128. [Google Scholar]
- Liu, L.; Zhang, H.; Sun, L.; Zhang, H.; Shi, Y.; Guo, C.; Han, S.; Liu, J.; Sun, S.; Han, Z.; et al. ERK/MAPK activation involves hypoxia-induced MGr1-Ag/37LRP expression and contributes to apoptosis resistance in gastric cancer. Int. J. Cancer 2009, 127, 820–829. [Google Scholar]
- Semenza, G.L. Hypoxia-inducible factor: Mediators of cancer progression and targets for cancer therapy. Trends Pharmacol. Sci. 2012, 33, 207–214. [Google Scholar] [CrossRef]
- Oommen, D.; Prise, K.M. KNK437, abrogates hypoxia-induced radioresistance by dual targeting of the AKT and HIF-1αsurvival pathways. Biochem. Biophys. Res. Commun. 2012, 421, 538–543. [Google Scholar] [CrossRef]
- Yu, J.; Mi, J.; Wang, Y.; Wang, A.; Tian, X. Regulation of radiosensitivity by HDAC inhibitor trichostatin A in the human cervical carcinoma cell line Hela. Eur. J. Gynaecol. Oncol. 2012, 33, 285–290. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kitajima, Y.; Miyazaki, K. The Critical Impact of HIF-1a on Gastric Cancer Biology. Cancers 2013, 5, 15-26. https://doi.org/10.3390/cancers5010015
Kitajima Y, Miyazaki K. The Critical Impact of HIF-1a on Gastric Cancer Biology. Cancers. 2013; 5(1):15-26. https://doi.org/10.3390/cancers5010015
Chicago/Turabian StyleKitajima, Yoshihiko, and Kohji Miyazaki. 2013. "The Critical Impact of HIF-1a on Gastric Cancer Biology" Cancers 5, no. 1: 15-26. https://doi.org/10.3390/cancers5010015



