Transition of Treatment for Patients with Extra-Abdominal Desmoid Tumors: Nagoya University Modality
Abstract
:1. Introduction
2. Surgical Treatment with Intended Wide Surgical Margin (From 1991 to 2003)
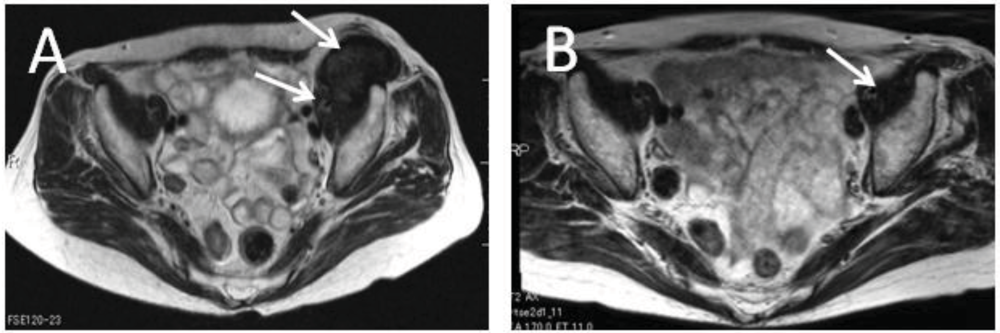
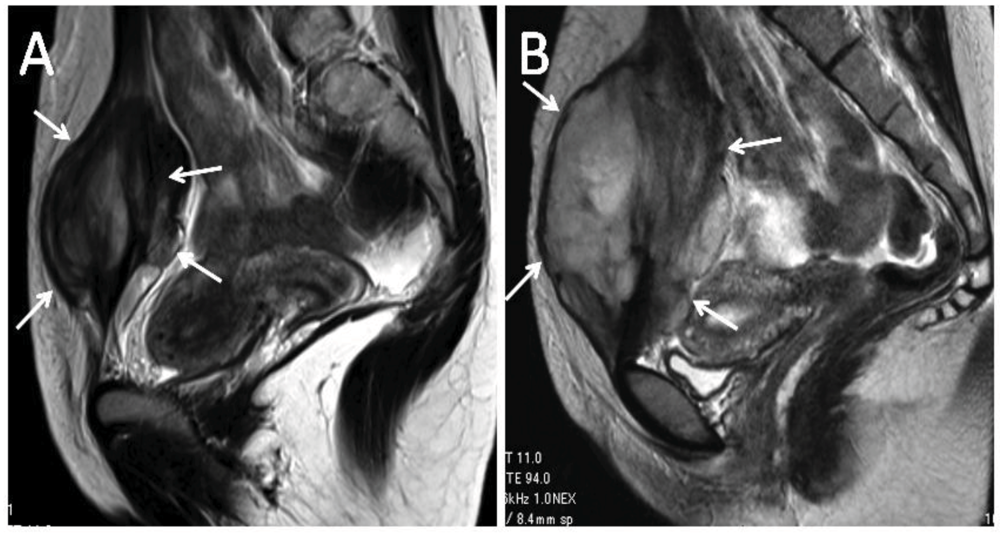
3. Conservative Therapy with Meloxicam (Since 2003)
| No. | Age/48 (mean) | Sex | Location | Follow Up/44 (median) | Recist |
|---|---|---|---|---|---|
| 1 | 20 | F | abdominal wall | 27 | PD |
| 2 | 20 | M | shoulder | 43 | SD |
| 3 | 20 | F | thigh | 65 | SD |
| 4 | 21 | M | thigh | 50 | SD |
| 5 | 27 | F | thigh | 41 | SD |
| 6 | 32 | M | thigh | 44 | SD |
| 7 | 32 | F | chest wall | 24 | PR |
| 8 | 34 | F | neck | 81 | PR |
| 9 | 36 | M | abdominal wall | 8 | SD |
| 10 | 39 | F | back | 52 | SD |
| 11 | 43 | M | back | 48 | SD |
| 12 | 55 | F | calf | 59 | PR |
| 13 | 55 | F | back | 82 | PR |
| 14 | 56 | M | abdominal wall | 52 | ND |
| 15 | 56 | M | groin | 27 | PR |
| 16 | 59 | F | forearm | 75 | PR |
| 17 | 71 | F | groin | 60 | CR |
| 18 | 73 | F | shoulder | 27 | PR |
| 19 | 73 | F | neck | 41 | PR |
| 20 | 74 | M | back | 30 | PR |
| 21 | 75 | F | foot | 31 | PR |
| 22 | 86 | M | back | 31 | ND |
4. Discussion
5. Conclusions
Conflict of Interest
Acknowledgements
References
- Fletcher, J.A.; Naeem, R.; Xiao, S.; Corson, J.M. Chromosome aberrations in desmoid tumors Trisomy 8 may be a predictor of recurrence. Cancer Genet. Cytogenet. 1995, 79, 139–143. [Google Scholar] [CrossRef]
- Li, M.; Cordon-Cardo, C.; Gerald, W.L.; Rosai, J. Desmoid fibromatosis is a clonal process. Hum. Pathol. 1996, 27, 939–943. [Google Scholar] [CrossRef]
- Posner, M.C.; Shiu, M.H.; Newsome, J.L.; Hajdu, S.I.; Gaynor, J.J.; Brennan, M.F. The desmoid tumor: Not a benign disease. Arch. Surg. 1989, 124, 191–196. [Google Scholar] [CrossRef]
- Giarola, M.; Wells, D.; Mondini, P.; Pilotti, S.; Sala, P.; Azzarelli, A.; Bertario, L.; Pierotti, M.A.; Delhanty, J.D.; Radice, P. Mutations of adenomatous polyposis coli (APC) gene are uncommon in sporadic desmoid tumours. Br. J. Cancer 1998, 78, 582–587. [Google Scholar] [CrossRef]
- Jones, I.T.; Jagelman, D.G.; Fazio, V.W.; Lavery, I.C.; Weakley, F.L.; McGannon, E. Desmoid tumors in familial polyposis coli. Ann. Surg. 1986, 204, 94–97. [Google Scholar] [CrossRef]
- Ballo, M.T.; Zagars, G.K.; Pollack, A.; Pisters, P.W.; Pollack, R.A. Desmoid tumor: Prognostic factors and outcome after surgery, radiation therapy, or combined surgery and radiation therapy. J. Clin. Oncol. 1999, 17, 158–167. [Google Scholar]
- Spear, M.A.; Jennings, L.C.; Mankin, H.J.; Spiro, I.J.; Springfield, D.S.; Gebhardt, M.C.; Rosenberg, A.E.; Efird, J.T.; Suit, H.D. Individualizing management of aggressive fibromatoses. Int. J. Radiat. Oncol. Biol. Phys. 1998, 40, 637–645. [Google Scholar] [CrossRef]
- Stoeckle, E.; Coindre, J.M.; Longy, M.; Binh, M.B.; Kantor, G.; Kind, M.; de Lara, C.T.; Avril, A.; Bonichon, F.; Bui, B.N. A critical analysis of treatment strategies in desmoid tumours: A review of a series of 106 cases. Eur. J. Surg. Oncol. 2009, 35, 129–134. [Google Scholar] [CrossRef]
- Gronchi, A.; Casali, P.G.; Mariani, L.; Lo Vullo, S.; Colecchia, M.; Lozza, L.; Bertulli, R.; Fiore, M.; Olmi, P.; Santinami, M.; et al. Quality of surgery and outcome in extra-abdominal aggressive fibromatosis: A series of patients surgically treated at a single institution. J. Clin. Oncol. 2003, 21, 1390–1397. [Google Scholar]
- Lev, D.; Kotilingam, D.; Wei, C.; Ballo, M.T.; Zagars, G.K.; Pisters, P.W.; Lazar, A.A.; Patel, S.R.; Benjamin, R.S.; Pollock, R.E. Optimizing treatment of desmoid tumors. J. Clin. Oncol. 2007, 25, 1785–1791. [Google Scholar]
- Lewis, J.J.; Boland, P.J.; Leung, D.H.; Woodruff, J.M.; Brennan, M.F. The enigma of desmoid tumors. Ann. Surg. 1999, 229, 866–873. [Google Scholar] [CrossRef]
- Merchant, N.B.; Lewis, J.J.; Woodruff, J.M.; Leung, D.H.; Brennan, M.F. Extremity and trunk desmoid tumors: A multifactorial analysis of outcome. Cancer 1999, 86, 2045–2052. [Google Scholar] [CrossRef]
- Shido, Y.; Nishida, Y.; Nakashima, H.; Katagiri, H.; Sugiura, H.; Yamada, Y.; Ishiguro, N. Surgical treatment for local control of extremity and trunk desmoid tumors. Arch. Orthop. Trauma. Surg. 2009, 129, 929–933. [Google Scholar] [CrossRef]
- Gega, M.; Yanagi, H.; Yoshikawa, R.; Noda, M.; Ikeuchi, H.; Tsukamoto, K.; Oshima, T.; Fujiwara, Y.; Gondo, N.; Tamura, M.; et al. Successful chemotherapeutic modality of doxorubicin plus dacarbazine for the treatment of desmoid tumors in association with familial adenomatous polyposis. J. Clin. Oncol. 2006, 24, 102–105. [Google Scholar]
- Francis, W.P.; Zippel, D.; Mack, L.A.; DiFrancesco, L.M.; Kurien, E.; Schachar, N.S.; Temple, W.J. Desmoids: A revelation in biology and treatment. Ann. Surg. Oncol. 2009, 16, 1650–1654. [Google Scholar] [CrossRef]
- Poon, R.; Smits, R.; Li, C.; Jagmohan-Changur, S.; Kong, M.; Cheon, S.; Yu, C.; Fodde, R.; Alman, B.A. Cyclooxygenase-two (COX-2) modulates proliferation in aggressive fibromatosis (desmoid tumor). Oncogene 2001, 20, 451–460. [Google Scholar]
- Nishida, Y.; Tsukushi, S.; Shido, Y.; Wasa, J.; Ishiguro, N.; Yamada, Y. Successful treatment with meloxicam, a cyclooxygenase-2 inhibitor, of patients with extra-abdominal desmoid tumors: A pilot study. J. Clin. Oncol. 2010, 28, e107–e109. [Google Scholar] [CrossRef]
- Therasse, P.; Arbuck, S.G.; Eisenhauer, E.A.; Wanders, J.; Kaplan, R.S.; Rubinstein, L.; Verweij, J.; van Glabbeke, M.; van Oosterom, A.T.; Christian, M.C.; et al. New guidelines to evaluate the response to treatment in solid tumors. J. Natl. Cancer Inst. 2000, 92, 205–216. [Google Scholar] [CrossRef]
- Salas, S.; Dufresne, A.; Bui, B.; Blay, J.Y.; Terrier, P.; Ranchere-Vince, D.; Bonvalot, S.; Stoeckle, E.; Guillou, L.; Le Cesne, A.; et al. Prognostic factors influencing progression-free survival determined from a series of sporadic desmoid tumors: A wait-and-see policy according to tumor presentation. J. Clin. Oncol. 2011, 29, 3553–3558. [Google Scholar]
- Anthony, T.; Rodriguez-Bigas, M.A.; Weber, T.K.; Petrelli, N.J. Desmoid tumors. J. Am. Coll. Surg. 1996, 182, 369–377. [Google Scholar]
- Nuyttens, J.J.; Rust, P.F.; Thomas, C.R., Jr.; Turrisi, A.T., 3rd. Surgery versus radiation therapy for patients with aggressive fibromatosis or desmoid tumors: A comparative review of 22 articles. Cancer 2000, 88, 1517–1523. [Google Scholar] [CrossRef]
- Janinis, J.; Patriki, M.; Vini, L.; Aravantinos, G.; Whelan, J.S. The pharmacological treatment of aggressive fibromatosis: A systematic review. Ann. Oncol. 2003, 14, 181–190. [Google Scholar] [CrossRef]
- Lackner, H.; Urban, C.; Kerbl, R.; Schwinger, W.; Beham, A. Noncytotoxic drug therapy in children with unresectable desmoid tumors. Cancer 1997, 80, 334–340. [Google Scholar] [CrossRef]
- Thomas, S.; Datta-Gupta, S.; Kapur, B.M. Treatment of recurrent desmoid tumour with tamoxifen. Aust. N. Z. J. Surg. 1990, 60, 919–921. [Google Scholar] [CrossRef]
- Wilcken, N.; Tattersall, M.H. Endocrine therapy for desmoid tumors. Cancer 1991, 68, 1384–1388. [Google Scholar] [CrossRef]
- Klein, W.A.; Miller, H.H.; Anderson, M.; DeCosse, J.J. The use of indomethacin, sulindac, and tamoxifen for the treatment of desmoid tumors associated with familial polyposis. Cancer 1987, 60, 2863–2868. [Google Scholar] [CrossRef]
- Tsukada, K.; Church, J.M.; Jagelman, D.G.; Fazio, V.W.; McGannon, E.; George, C.R.; Schroeder, T.; Lavery, I.; Oakley, J. Noncytotoxic drug therapy for intra-abdominal desmoid tumor in patients with familial adenomatous polyposis. Dis. Colon Rectum 1992, 35, 29–33. [Google Scholar]
- Waddell, W.R.; Kirsch, W.M. Testolactone, sulindac, warfarin, and vitamin K1 for unresectable desmoid tumors. Am. J. Surg. 1991, 161, 416–421. [Google Scholar] [CrossRef]
- Izes, J.K.; Zinman, L.N.; Larsen, C.R. Regression of large pelvic desmoid tumor by tamoxifen and sulindac. Urology 1996, 47, 756–759. [Google Scholar] [CrossRef]
- Clark, S.K.; Phillips, R.K. Desmoids in familial adenomatous polyposis. Br. J. Surg. 1996, 83, 1494–1504. [Google Scholar] [CrossRef]
- Schnitzler, M.; Cohen, Z.; Blackstein, M.; Berk, T.; Gallinger, S.; Madlensky, L.; McLeod, R. Chemotherapy for desmoid tumors in association with familial adenomatous polyposis. Dis. Colon Rectum 1997, 40, 798–801. [Google Scholar] [CrossRef]
- Skapek, S.X.; Ferguson, W.S.; Granowetter, L.; Devidas, M.; Perez-Atayde, A.R.; Dehner, L.P.; Hoffer, F.A.; Speights, R.; Gebhardt, M.C.; Dahl, G.V.; et al. Vinblastine and methotrexate for desmoid fibromatosis in children: Results of a Pediatric Oncology Group Phase II Trial. J. Clin. Oncol. 2007, 25, 501–506. [Google Scholar]
- Patel, S.R.; Evans, H.L.; Benjamin, R.S. Combination chemotherapy in adult desmoid tumors. Cancer 1993, 72, 3244–3247. [Google Scholar] [CrossRef]
- Eberhart, C.E.; Coffey, R.J.; Radhika, A.; Giardiello, F.M.; Ferrenbach, S.; DuBois, R.N. Up-regulation of cyclooxygenase 2 gene expression in human colorectal adenomas and adenocarcinomas. Gastroenterology 1994, 107, 1183–1188. [Google Scholar]
- Gupta, S.; Srivastava, M.; Ahmad, N.; Bostwick, D.G.; Mukhtar, H. Over-expression of cyclooxygenase-2 in human prostate adenocarcinoma. Prostate 2000, 42, 73–78. [Google Scholar] [CrossRef]
- Hwang, D.; Scollard, D.; Byrne, J.; Levine, E. Expression of cyclooxygenase-1 and cyclooxygenase-2 in human breast cancer. J. Natl. Cancer Inst. 1998, 90, 455–460. [Google Scholar] [CrossRef]
- Urakawa, H.; Nishida, Y.; Naruse, T.; Nakashima, H.; Ishiguro, N. Cyclooxygenase-2 overexpression predicts poor survival in patients with high-grade extremity osteosarcoma: A pilot study. Clin. Orthop. Relat. Res. 2009, 467, 2932–2938. [Google Scholar] [CrossRef]
- Liu, X.H.; Yao, S.; Kirschenbaum, A.; Levine, A.C. NS398, a selective cyclooxygenase-2 inhibitor, induces apoptosis and down-regulates bcl-2 expression in LNCaP cells. Cancer Res. 1998, 58, 4245–4249. [Google Scholar]
- Naruse, T.; Nishida, Y.; Hosono, K.; Ishiguro, N. Meloxicam inhibits osteosarcoma growth, invasiveness and metastasis by COX-2-dependent and independent routes. Carcinogenesis 2006, 27, 584–592. [Google Scholar]
- Sheng, H.; Shao, J.; Kirkland, S.C.; Isakson, P.; Coffey, R.J.; Morrow, J.; Beauchamp, R.D.; DuBois, R.N. Inhibition of human colon cancer cell growth by selective inhibition of cyclooxygenase-2. J. Clin. Invest. 1997, 99, 2254–2259. [Google Scholar] [CrossRef]
- Yao, M.; Kargman, S.; Lam, E.C.; Kelly, C.R.; Zheng, Y.; Luk, P.; Kwong, E.; Evans, J.F.; Wolfe, M.M. Inhibition of cyclooxygenase-2 by rofecoxib attenuates the growth and metastatic potential of colorectal carcinoma in mice. Cancer Res. 2003, 63, 586–592. [Google Scholar]
- Oshima, M.; Dinchuk, J.E.; Kargman, S.L.; Oshima, H.; Hancock, B.; Kwong, E.; Trzaskos, J.M.; Evans, J.F.; Taketo, M.M. Suppression of intestinal polyposis in Apc delta716 knockout mice by inhibition of cyclooxygenase 2 (COX-2). Cell 1996, 87, 803–809. [Google Scholar] [CrossRef]
- His, L.C.; Angerman-Stewart, J.; Eling, T.E. Introduction of full-length APC modulates cyclooxygenase-2 expression in HT-29 human colorectal carcinoma cells at the translational level. Carcinogenesis 1999, 20, 2045–2049. [Google Scholar] [CrossRef]
- Mei, J.M.; Hord, N.G.; Winterstein, D.F.; Donald, S.P.; Phang, J.M. Differential expression of prostaglandin endoperoxide H synthase-2 and formation of activated beta-catenin-LEF-1 transcription complex in mouse colonic epithelial cells contrasting in Apc. Carcinogenesis 1999, 20, 737–740. [Google Scholar] [CrossRef]
- Signoroni, S.; Frattini, M.; Negri, T.; Pastore, E.; Tamborini, E.; Casieri, P.; Orsenigo, M.; Da Riva, L.; Radice, P.; Sala, P.; et al. Cyclooxygenase-2 and platelet-derived growth factor receptors as potential targets in treating aggressive fibromatosis. Clin. Cancer Res. 2007, 13, 5034–5040. [Google Scholar]
- Warner, T.D.; Giuliano, F.; Vojnovic, I.; Bukasa, A.; Mitchell, J.A.; Vane, J.R. Nonsteroid drug selectivities for cyclo-oxygenase-1 rather than cyclo-oxygenase-2 are associated with human gastrointestinal toxicity: A full in vitro analysis. Proc. Natl. Acad. Sci. USA 1999, 96, 7563–7568. [Google Scholar]
- Dequeker, J.; Hawkey, C.; Kahan, A.; Steinbruck, K.; Alegre, C.; Baumelou, E.; Bégaud, B.; Isomäki, H.; Littlejohn, G.; Mau, J.; et al. Improvement in gastrointestinal tolerability of the selective cyclooxygenase (COX)-2 inhibitor, meloxicam, compared with piroxicam: Results of the safety and efficacy large-scale evaluation of COX-inhibiting therapies (SELECT) trial in osteoarthritis. Br. J. Rheumatol. 1998, 37, 946–951. [Google Scholar] [CrossRef]
- Hawkey, C.; Kahan, A.; Steinbruck, K.; Alegre, C.; Baumelou, E.; Begaud, B.; Dequeker, J.; Isomäki, H.; Littlejohn, G.; Mau, J.; et al. Gastrointestinal tolerability of meloxicam compared to diclofenac in osteoarthritis patients. International MELISSA Study Group. Meloxicam Large-scale international study safety assessment. Br. J. Rheumatol. 1998, 37, 937–945. [Google Scholar] [CrossRef]
- Nakayama, T.; Tsuboyama, T.; Toguchida, J.; Hosaka, T.; Nakamura, T. Natural course of desmoid-type fibromatosis. J. Orthop. Sci. 2008, 13, 51–55. [Google Scholar] [CrossRef]
- Rock, M.G.; Pritchard, D.J.; Reiman, H.M.; Soule, E.H.; Brewster, R.C. Extra-abdominal desmoid tumors. J. Bone Joint Surg. Am. 1984, 66, 1369–1374. [Google Scholar]
- Bonvalot, S.; Eldweny, H.; Haddad, V.; Rimareix, F.; Missenard, G.; Oberlin, O.; Vanel, D.; Terrier, P.; Blay, J.Y.; Le Cesne, A.; et al. Extra-abdominal primary fibromatosis: Aggressive management could be avoided in a subgroup of patients. Eur. J. Surg. Oncol. 2008, 34, 462–468. [Google Scholar] [CrossRef]
- Reitamo, J.J.; Scheinin, T.M.; Hayry, P. The desmoid syndrome. New aspects in the cause, pathogenesis and treatment of the desmoid tumor. Am. J. Surg. 1986, 151, 230–237. [Google Scholar] [CrossRef]
- Salas, S.; Chibon, F.; Noguchi, T.; Terrier, P.; Ranchere-Vince, D.; Lagarde, P.; Benard, J.; Forget, S.; Blanchard, C.; Dômont, J.; et al. Molecular characterization by array comparative genomic hybridization and DNA sequencing of 194 desmoid tumors. Genes Chromosomes Cancer 2010, 49, 560–568. [Google Scholar]
- de Bree, E.; Keus, R.; Melissas, J.; Tsiftsis, D.; van Coevorden, F. Desmoid tumors: Need for an individualized approach. Expert Rev. Anticancer Ther. 2009, 9, 525–535. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Nishida, Y.; Tsukushi, S.; Shido, Y.; Urakawa, H.; Arai, E.; Ishiguro, N. Transition of Treatment for Patients with Extra-Abdominal Desmoid Tumors: Nagoya University Modality. Cancers 2012, 4, 88-99. https://doi.org/10.3390/cancers4010088
Nishida Y, Tsukushi S, Shido Y, Urakawa H, Arai E, Ishiguro N. Transition of Treatment for Patients with Extra-Abdominal Desmoid Tumors: Nagoya University Modality. Cancers. 2012; 4(1):88-99. https://doi.org/10.3390/cancers4010088
Chicago/Turabian StyleNishida, Yoshihiro, Satoshi Tsukushi, Yoji Shido, Hiroshi Urakawa, Eisuke Arai, and Naoki Ishiguro. 2012. "Transition of Treatment for Patients with Extra-Abdominal Desmoid Tumors: Nagoya University Modality" Cancers 4, no. 1: 88-99. https://doi.org/10.3390/cancers4010088




