CSF1R-Expressing Tumor-Associated Macrophages, Smoking and Survival in Lung Adenocarcinoma: Analyses Using Quantitative Phosphor-Integrated Dot Staining
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Immunostaining
2.3. Measurement of CSF1R Expression in TAMs
2.4. Detection of EGFR and KRAS Mutations and ALK Fusions
2.5. Statistical Analysis
3. Results
3.1. CSF1R Expression in TAMs
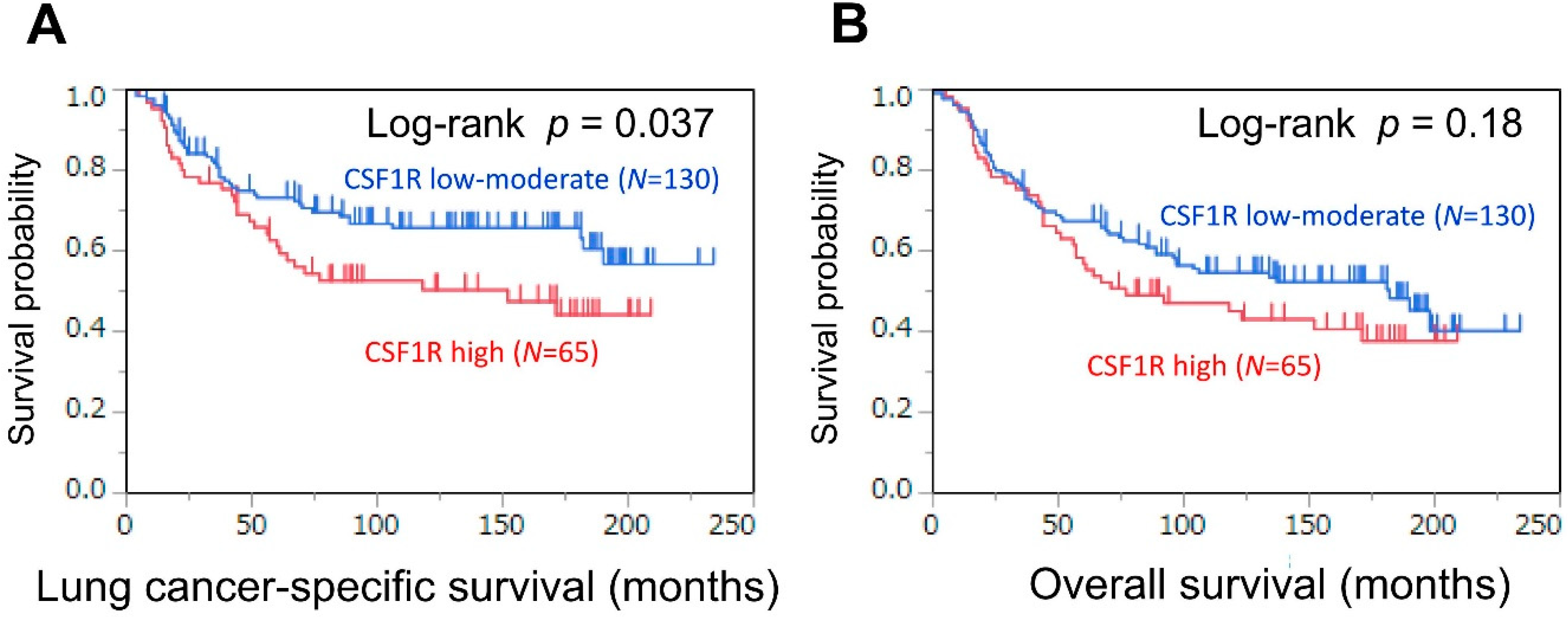
3.2. Association of CSF1R Expression in TAMs with Survival in Patients with Lung Adenocarcinoma
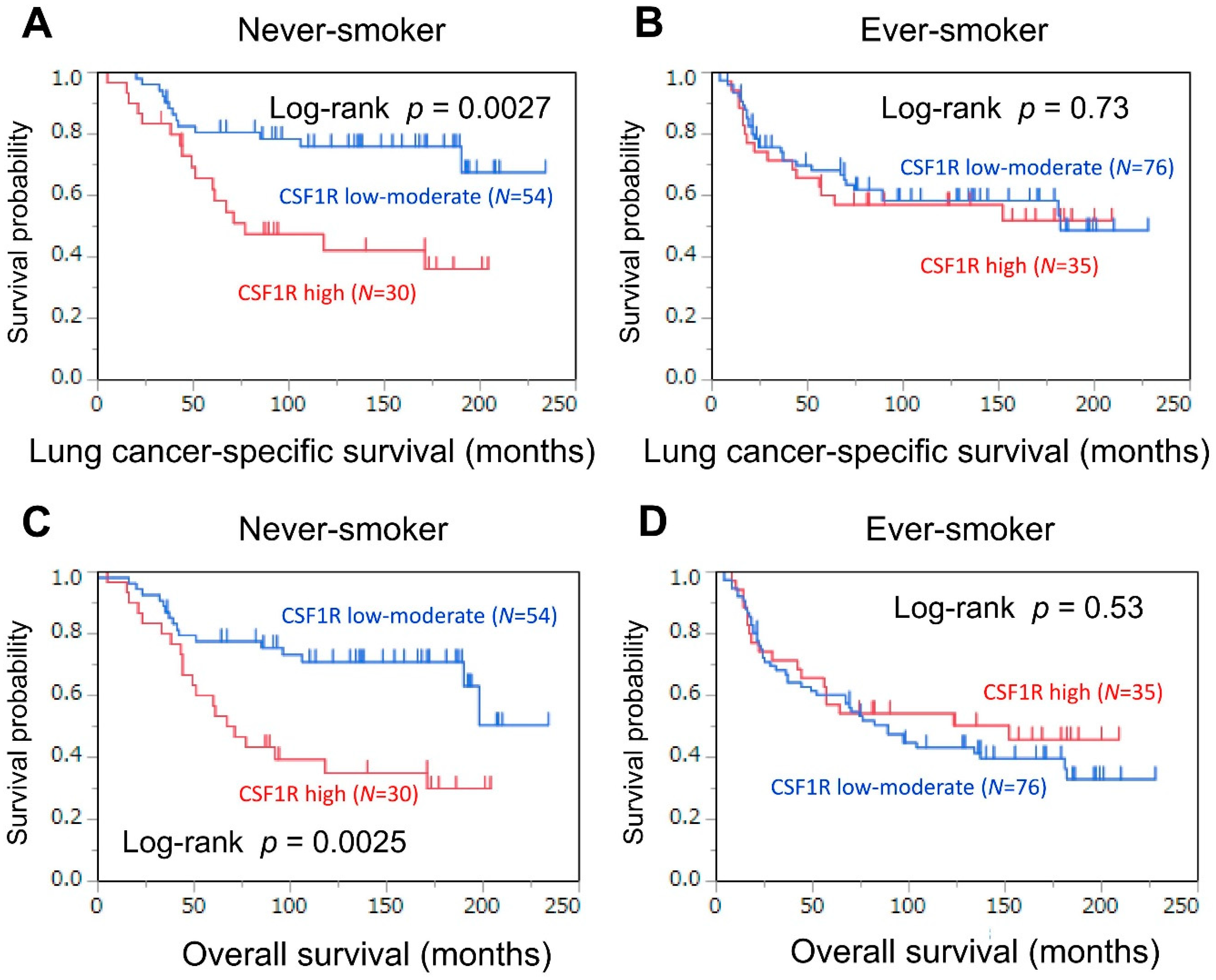
3.3. Association of CSF1R Expression in TAMs with Survival, Stratified by Smoking Status
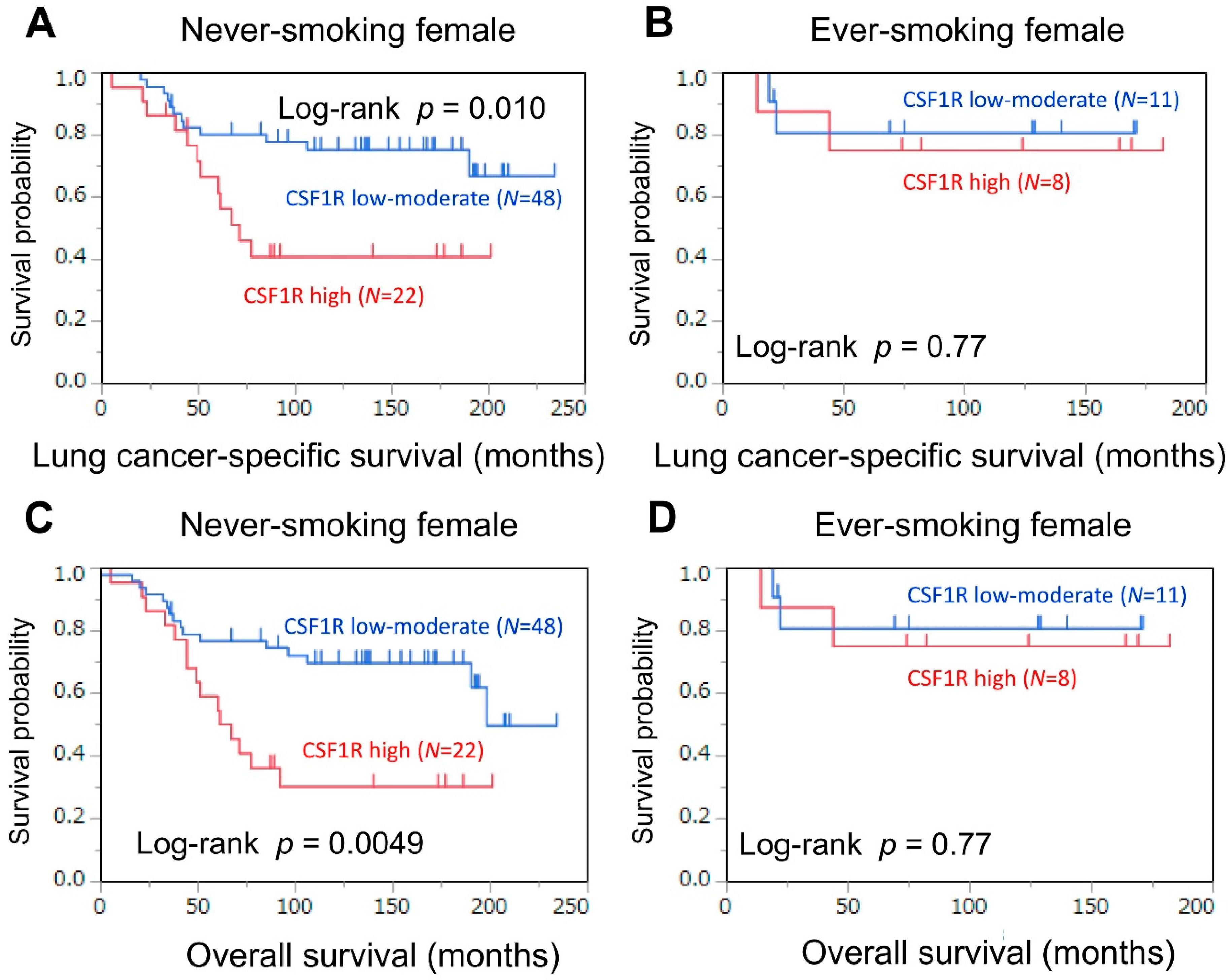
3.4. Association of CSF1R Expression in TAMs with Survival in Female Patients, Stratified by Smoking Status
4. Discussion
5. Conclusion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BSA | bovine serum albumin |
| CI | confidence interval |
| DAB | diaminobenzidine |
| HR | hazard ratio |
| JFCR | Japanese Foundation for Cancer Research |
| PBS | phosphate-buffered saline |
| PID | phosphor-integrated dot |
| SNP | single nucleotide polymorphism |
| TAM | tumor-associated macrophage |
References
- Herbst, R.S.; Soria, J.C.; Kowanetz, M.; Fine, G.D.; Hamid, O.; Gordon, M.S.; Sosman, J.A.; McDermott, D.F.; Powderly, J.D.; Gettinger, S.N.; et al. Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature 2014, 515, 563–567. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rizvi, N.A.; Hellmann, M.D.; Snyder, A.; Kvistborg, P.; Makarov, V.; Havel, J.J.; Lee, W.; Yuan, J.; Wong, P.; Ho, T.S.; et al. Cancer immunology. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science 2015, 348, 124–128. [Google Scholar] [CrossRef] [PubMed]
- Rooney, M.S.; Shukla, S.A.; Wu, C.J.; Getz, G.; Hacohen, N. Molecular and genetic properties of tumors associated with local immune cytolytic activity. Cell 2015, 160, 48–61. [Google Scholar] [CrossRef] [PubMed]
- Desrichard, A.; Kuo, F.; Chowell, D.; Lee, K.-W.; Riaz, N.; Wong, R.J.; Chan, T.A.; Morris, L.G.T. Tobacco Smoking-Associated Alterations in the Immune Microenvironment of Squamous Cell Carcinomas. J. Natl. Cancer Inst. 2018. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, T.; Kudo-Saito, C.; Muramatsu, R.; Fujita, T.; Saito, M.; Nagumo, H.; Sakurai, T.; Noji, S.; Takahata, E.; Yaguchi, T.; et al. Determination of poor prognostic immune features of tumour microenvironment in non-smoking patients with lung adenocarcinoma. Eur. J. Cancer 2017, 86, 15–27. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, T.; Muramatsu, R.; Fujita, T.; Nagumo, H.; Sakurai, T.; Noji, S.; Takahata, E.; Yaguchi, T.; Tsukamoto, N.; Kudo-Saito, C.; et al. Prognostic value of tumor-infiltrating lymphocytes differs depending on histological type and smoking habit in completely resected non-small-cell lung cancer. Ann. Oncol. 2016, 27, 2117–2123. [Google Scholar] [CrossRef] [PubMed]
- Haratani, K.; Hayashi, H.; Tanaka, T.; Kaneda, H.; Togashi, Y.; Sakai, K.; Hayashi, K.; Tomida, S.; Chiba, Y.; Yonesaka, K.; et al. Tumor immune microenvironment and nivolumab efficacy in EGFR mutation-positive non-small-cell lung cancer based on T790M status after disease progression during EGFR-TKI treatment. Ann. Oncol. 2017, 28, 1532–1539. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cannarile, M.A.; Weisser, M.; Jacob, W.; Jegg, A.M.; Ries, C.H.; Ruttinger, D. Colony-stimulating factor 1 receptor (CSF1R) inhibitors in cancer therapy. J. Immunother. Cancer 2017, 5, 53. [Google Scholar] [CrossRef] [PubMed]
- Mantovani, A.; Marchesi, F.; Malesci, A.; Laghi, L.; Allavena, P. Tumour-associated macrophages as treatment targets in oncology. Nat. Rev. Clin. Oncol. 2017, 14, 399–416. [Google Scholar] [CrossRef] [PubMed]
- Hume, D.A.; MacDonald, K.P. Therapeutic applications of macrophage colony-stimulating factor-1 (CSF-1) and antagonists of CSF-1 receptor (CSF-1R) signaling. Blood 2012, 119, 1810–1820. [Google Scholar] [CrossRef] [PubMed]
- Koh, Y.W.; Park, C.; Yoon, D.H.; Suh, C.; Huh, J. CSF-1R expression in tumor-associated macrophages is associated with worse prognosis in classical Hodgkin lymphoma. Am. J. Clin. Pathol. 2014, 141, 573–583. [Google Scholar] [CrossRef] [PubMed]
- Ries, C.H.; Cannarile, M.A.; Hoves, S.; Benz, J.; Wartha, K.; Runza, V.; Rey-Giraud, F.; Pradel, L.P.; Feuerhake, F.; Klaman, I.; et al. Targeting tumor-associated macrophages with anti-CSF-1R antibody reveals a strategy for cancer therapy. Cancer Cell 2014, 25, 846–859. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulos, K.P.; Gluck, L.; Martin, L.P.; Olszanski, A.J.; Tolcher, A.W.; Ngarmchamnanrith, G.; Rasmussen, E.; Amore, B.M.; Nagorsen, D.; Hill, J.S.; et al. First-in-Human Study of AMG 820, a Monoclonal Anti-Colony-Stimulating Factor 1 Receptor Antibody, in Patients with Advanced Solid Tumors. Clin. Cancer Res. 2017, 23, 5703–5710. [Google Scholar] [CrossRef] [PubMed]
- Candido, J.B.; Morton, J.P.; Bailey, P.; Campbell, A.D.; Karim, S.A.; Jamieson, T.; Lapienyte, L.; Gopinathan, A.; Clark, W.; McGhee, E.J.; et al. CSF1R(+) Macrophages Sustain Pancreatic Tumor Growth through T Cell Suppression and Maintenance of Key Gene Programs That Define the Squamous Subtype. Cell Rep. 2018, 23, 1448–1460. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.G.; Lee, S.Y.; Jeon, H.S.; Choi, Y.Y.; Kim, S.; Lee, W.K.; Lee, H.C.; Choi, J.E.; Bae, E.Y.; Yoo, S.S.; et al. A functional polymorphism in CSF1R gene is a novel susceptibility marker for lung cancer among never-smoking females. J. Thorac. Oncol. 2014, 9, 1647–1655. [Google Scholar] [CrossRef] [PubMed]
- Yoo, S.S.; Kang, H.G.; Choi, J.E.; Do, S.K.; Lee, W.K.; Choi, S.H.; Lee, S.Y.; Lee, S.Y.; Lee, J.; Cha, S.I.; et al. Effects of polymorphisms identified in genome-wide association studies of never-smoking females on the prognosis of non-small cell lung cancer. Cancer Genet. 2017, 212–213, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Szymanowska-Narloch, A.; Jassem, E.; Skrzypski, M.; Muley, T.; Meister, M.; Dienemann, H.; Taron, M.; Rosell, R.; Rzepko, R.; Jarzab, M.; et al. Molecular profiles of non-small cell lung cancers in cigarette smoking and never-smoking patients. Adv. Med. Sci. 2013, 58, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Gonda, K.; Watanabe, M.; Tada, H.; Miyashita, M.; Takahashi-Aoyama, Y.; Kamei, T.; Ishida, T.; Usami, S.; Hirakawa, H.; Kakugawa, Y.; et al. Quantitative diagnostic imaging of cancer tissues by using phosphor-integrated dots with ultra-high brightness. Sci. Rep. 2017, 7, 7509. [Google Scholar] [CrossRef] [PubMed]
- Yamaki, S.; Yanagimoto, H.; Tsuta, K.; Ryota, H.; Kon, M. PD-L1 expression in pancreatic ductal adenocarcinoma is a poor prognostic factor in patients with high CD8(+) tumor-infiltrating lymphocytes: Highly sensitive detection using phosphor-integrated dot staining. Int. J. Clin. Oncol. 2017, 22, 726–733. [Google Scholar] [CrossRef] [PubMed]
- Inamura, K.; Yokouchi, Y.; Kobayashi, M.; Ninomiya, H.; Sakakibara, R.; Nishio, M.; Okumura, S.; Ishikawa, Y. Relationship of tumor PD-L1 (CD274) expression with lower mortality in lung high-grade neuroendocrine tumor. Cancer Med. 2017, 6, 2347–2356. [Google Scholar] [CrossRef] [PubMed]
- Inamura, K.; Yokouchi, Y.; Kobayashi, M.; Ninomiya, H.; Sakakibara, R.; Subat, S.; Nagano, H.; Nomura, K.; Okumura, S.; Shibutani, T.; et al. Association of tumor TROP2 expression with prognosis varies among lung cancer subtypes. Oncotarget 2017, 8, 28725–28735. [Google Scholar] [CrossRef] [PubMed]
- Inamura, K.; Yokouchi, Y.; Kobayashi, M.; Sakakibara, R.; Ninomiya, H.; Subat, S.; Nagano, H.; Nomura, K.; Okumura, S.; Shibutani, T.; et al. Tumor B7-H3 (CD276) expression and smoking history in relation to lung adenocarcinoma prognosis. Lung Cancer 2017, 103, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Travis, W.D.; Brambilla, E.; Burke, A.P.; Marx, A.; Nicholson, A.G. WHO Classification of Tumours of the Lung, Pleura, Thymus and Heart, 4th ed.; IARC Press: Lyon, France, 2015. [Google Scholar]
- Edge, S.B.; Byrd, D.R.; Compton, C.C.; Fritz, A.G.; Greene, F.L.; Trotti, A. AJCC Cancer Staging Manual, 7th ed.; Springer: New York, NY, USA, 2010. [Google Scholar]
- Jung, K.Y.; Cho, S.W.; Kim, Y.A.; Kim, D.; Oh, B.C.; Park, D.J.; Park, Y.J. Cancers with Higher Density of Tumor-Associated Macrophages Were Associated with Poor Survival Rates. J. Pathol. Transl. Med. 2015, 49, 318–324. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, K.; Soda, M.; Togashi, Y.; Suzuki, R.; Sakata, S.; Hatano, S.; Asaka, R.; Hamanaka, W.; Ninomiya, H.; Uehara, H.; et al. RET, ROS1 and ALK fusions in lung cancer. Nat. Med. 2012, 18, 378–381. [Google Scholar] [CrossRef] [PubMed]
- The Cancer Genome Atlas Research Network. Comprehensive molecular profiling of lung adenocarcinoma. Nature 2014, 511, 543–550. [Google Scholar] [Green Version]
- Chen, Z.; Fillmore, C.M.; Hammerman, P.S.; Kim, C.F.; Wong, K.K. Non-small-cell lung cancers: A heterogeneous set of diseases. Nat. Rev. Cancer 2014, 14, 535–546. [Google Scholar] [CrossRef] [PubMed]
- Varn, F.S.; Tafe, L.J.; Amos, C.I.; Cheng, C. Computational immune profiling in lung adenocarcinoma reveals reproducible prognostic associations with implications for immunotherapy. Oncoimmunology 2018, 7, e1431084. [Google Scholar] [CrossRef] [PubMed]
- Jamal-Hanjani, M.; Wilson, G.A.; McGranahan, N.; Birkbak, N.J.; Watkins, T.B.K.; Veeriah, S.; Shafi, S.; Johnson, D.H.; Mitter, R.; Rosenthal, R.; et al. Tracking the Evolution of Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2017, 376, 2109–2121. [Google Scholar] [CrossRef] [PubMed]
- Vargas, A.J.; Harris, C.C. Biomarker development in the precision medicine era: Lung cancer as a case study. Nat. Rev. Cancer 2016, 16, 525–537. [Google Scholar] [CrossRef] [PubMed]
- Devarakonda, S.; Morgensztern, D.; Govindan, R. Genomic alterations in lung adenocarcinoma. Lancet Oncol. 2015, 16, e342–e351. [Google Scholar] [CrossRef]
- Barlesi, F.; Mazieres, J.; Merlio, J.P.; Debieuvre, D.; Mosser, J.; Lena, H.; Ouafik, L.; Besse, B.; Rouquette, I.; Westeel, V.; et al. Routine molecular profiling of patients with advanced non-small-cell lung cancer: Results of a 1-year nationwide programme of the French Cooperative Thoracic Intergroup (IFCT). Lancet 2016, 387, 1415–1426. [Google Scholar] [CrossRef]
- Campbell, B.B.; Light, N.; Fabrizio, D.; Zatzman, M.; Fuligni, F.; de Borja, R.; Davidson, S.; Edwards, M.; Elvin, J.A.; Hodel, K.P.; et al. Comprehensive Analysis of Hypermutation in Human Cancer. Cell 2017, 171, 1042–1056. [Google Scholar] [CrossRef] [PubMed]
- Lavin, Y.; Kobayashi, S.; Leader, A.; Amir, E.D.; Elefant, N.; Bigenwald, C.; Remark, R.; Sweeney, R.; Becker, C.D.; Levine, J.H.; et al. Innate Immune Landscape in Early Lung Adenocarcinoma by Paired Single-Cell Analyses. Cell 2017, 169, 750–765. [Google Scholar] [CrossRef] [PubMed]
- Zamay, T.N.; Zamay, G.S.; Kolovskaya, O.S.; Zukov, R.A.; Petrova, M.M.; Gargaun, A.; Berezovski, M.V.; Kichkailo, A.S. Current and Prospective Protein Biomarkers of Lung Cancer. Cancers (Basel) 2017, 9, 155. [Google Scholar] [CrossRef] [PubMed]
- Legras, A.; Pecuchet, N.; Imbeaud, S.; Pallier, K.; Didelot, A.; Roussel, H.; Gibault, L.; Fabre, E.; Le Pimpec-Barthes, F.; Laurent-Puig, P.; et al. Epithelial-to-Mesenchymal Transition and MicroRNAs in Lung Cancer. Cancers (Basel) 2017, 9, 101. [Google Scholar] [CrossRef] [PubMed]
- Hofman, V.; Lassalle, S.; Bence, C.; Long-Mira, E.; Nahon-Esteve, S.; Heeke, S.; Lespinet-Fabre, V.; Butori, C.; Ilie, M.; Hofman, P. Any Place for Immunohistochemistry within the Predictive Biomarkers of Treatment in Lung Cancer Patients? Cancers (Basel) 2018, 10, 70. [Google Scholar] [CrossRef] [PubMed]
- Heusinkveld, M.; van der Burg, S.H. Identification and manipulation of tumor associated macrophages in human cancers. J. Transl. Med. 2011, 9, 216. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Condeelis, J.; Pollard, J.W. Macrophages: Obligate partners for tumor cell migration, invasion, and metastasis. Cell 2006, 124, 263–266. [Google Scholar] [CrossRef] [PubMed]
- Baghdadi, M.; Endo, H.; Takano, A.; Ishikawa, K.; Kameda, Y.; Wada, H.; Miyagi, Y.; Yokose, T.; Ito, H.; Nakayama, H.; et al. High co-expression of IL-34 and M-CSF correlates with tumor progression and poor survival in lung cancers. Sci. Rep. 2018, 8, 418. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Medrek, C.; Ponten, F.; Jirstrom, K.; Leandersson, K. The presence of tumor associated macrophages in tumor stroma as a prognostic marker for breast cancer patients. BMC Cancer 2012, 12, 306. [Google Scholar] [CrossRef] [PubMed]
- Mansuet-Lupo, A.; Alifano, M.; Pecuchet, N.; Biton, J.; Becht, E.; Goc, J.; Germain, C.; Ouakrim, H.; Regnard, J.F.; Cremer, I.; et al. Intratumoral Immune Cell Densities Are Associated with Lung Adenocarcinoma Gene Alterations. Am. J. Respir. Crit. Care Med. 2016, 194, 1403–1412. [Google Scholar] [CrossRef] [PubMed]
- Zdanov, S.; Mandapathil, M.; Abu Eid, R.; Adamson-Fadeyi, S.; Wilson, W.; Qian, J.; Carnie, A.; Tarasova, N.; Mkrtichyan, M.; Berzofsky, J.A.; et al. Mutant KRAS Conversion of Conventional T Cells into Regulatory T Cells. Cancer Immunol. Res. 2016, 4, 354–365. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, W.Q.; Liu, L.; Xu, H.X.; Wu, C.T.; Xiang, J.F.; Xu, J.; Liu, C.; Long, J.; Ni, Q.X.; Yu, X.J. Infiltrating immune cells and gene mutations in pancreatic ductal adenocarcinoma. Br. J. Surg. 2016, 103, 1189–1199. [Google Scholar] [CrossRef] [PubMed]
- Thunnissen, E.; Allen, T.C.; Adam, J.; Aisner, D.L.; Beasley, M.B.; Borczuk, A.C.; Cagle, P.T.; Capelozzi, V.L.; Cooper, W.; Hariri, L.P.; et al. Immunohistochemistry of Pulmonary Biomarkers: A Perspective From Members of the Pulmonary Pathology Society. Arch. Pathol. Lab. Med. 2018, 142, 408–419. [Google Scholar] [CrossRef] [PubMed]
- Forde, P.M.; Chaft, J.E.; Smith, K.N.; Anagnostou, V.; Cottrell, T.R.; Hellmann, M.D.; Zahurak, M.; Yang, S.C.; Jones, D.R.; Broderick, S.; et al. Neoadjuvant PD-1 Blockade in Resectable Lung Cancer. N. Engl. J. Med. 2018, 378, 1976–1986. [Google Scholar] [CrossRef] [PubMed]
- Gordon, S.R.; Maute, R.L.; Dulken, B.W.; Hutter, G.; George, B.M.; McCracken, M.N.; Gupta, R.; Tsai, J.M.; Sinha, R.; Corey, D.; et al. PD-1 expression by tumour-associated macrophages inhibits phagocytosis and tumour immunity. Nature 2017, 545, 495–499. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Choksi, S.; Liu, Z.G. Butylated hydroxyanisole blocks the occurrence of tumor associated macrophages in tobacco smoke carcinogen-induced lung tumorigenesis. Cancers (Basel) 2013, 5, 1643–1654. [Google Scholar] [CrossRef] [PubMed]
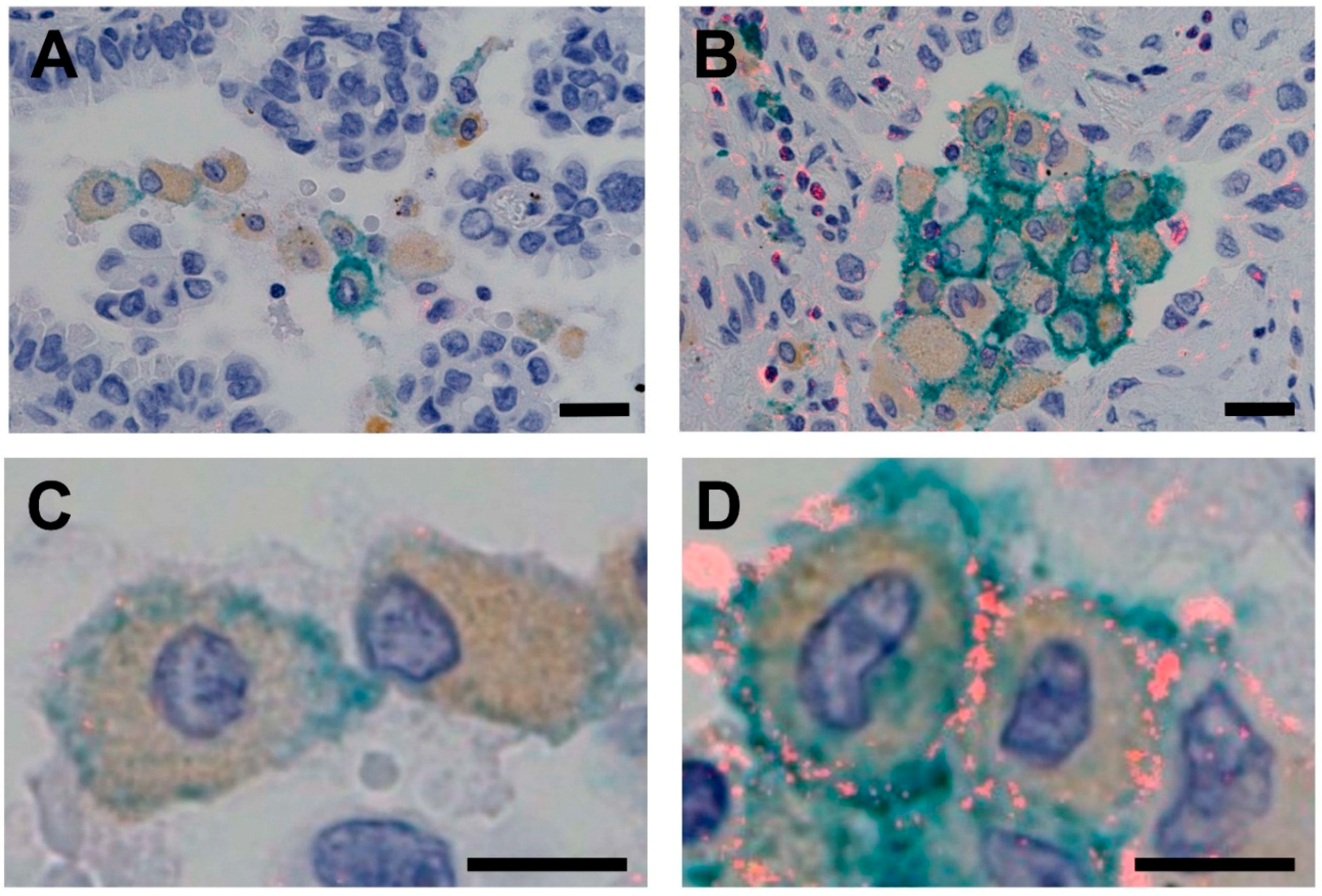
| Variables | N of Samples (%) | CSF1R Expression | ||
|---|---|---|---|---|
| Low–Moderate 130 (67%) | High 65 (33%) | p Values | ||
| Age (years) | 0.40 | |||
| <60 | 73 (37%) | 46 (35%) | 27 (42%) | |
| ≥60 | 122 (63%) | 84 (65%) | 38 (58%) | |
| Gender | 0.92 | |||
| Male | 106 (54%) | 71 (55%) | 35 (54%) | |
| Female | 89 (47%) | 59 (45%) | 30 (46%) | |
| Smoking status | 0.54 | |||
| Never smoker | 84 (43%) | 54 (42%) | 30 (46%) | |
| Ever smoker | 111 (57%) | 76 (58%) | 35 (54%) | |
| Pathological stage | 0.68 | |||
| I | 112 (57%) | 76 (58%) | 36 (55%) | |
| II–IV | 83 (43%) | 54 (42%) | 29 (45%) | |
| Tumor differentiation | 0.012 | |||
| Well | 84 (43%) | 64 (50%) | 20 (31%) | |
| Moderate–poor | 110 (57%) | 65 (50%) | 45 (69%) | |
| EGFR status | 0.11 | |||
| Wild-type | 71 (54%) | 54 (59%) | 17 (44%) | |
| Mutant | 60 (46%) | 38 (41%) | 22 (56%) | |
| KRAS status | 0.22 | |||
| Wild-type | 114 (87%) | 78 (85%) | 36 (92%) | |
| Mutant | 17 (13%) | 14 (15%) | 3 (7.7%) | |
| ALK rearrangement | 0.43 | |||
| Negative | 187 (96%) | 124 (95%) | 64 (98%) | |
| Positive | 7 (3.6%) | 6 (4.6%) | 1 (1.5%) | |
| Number of TAMs | 0.83 | |||
| Low–moderate | 131 (67%) | 88 (68%) | 43 (66%) | |
| High | 64 (33%) | 42 (32%) | 22 (34%) | |
| Patients and CSF1R Expression Status | Lung Cancer-Specific Mortality | Overall Mortality | ||||||
|---|---|---|---|---|---|---|---|---|
| Univariable Analysis | Multivariable Analysis ** | Univariable Analysis | Multivariable Analysis ** | |||||
| HR (95% CI) | p Values | HR (95% CI) | p Values | HR (95% CI) | p Values | HR (95% CI) | p Values | |
| All patients | ||||||||
| CSF1R: low–moderate expression (N = 130) | 1 (referent) | 1 (referent) | 1 (referent) | 1 (referent) | ||||
| CSF1R: high expression (N = 65) | 1.61 (1.02–2.52) | 0.043 | 1.32 (1.00–2.49) | 0.048 | 1.31 (0.87–1.95) | 0.19 | 1.09 (0.72–1.65) | 0.68 |
| Never-smoking patients | ||||||||
| CSF1R: low–moderate expression (N = 54) | 1 (referent) | 1 (referent) | 1 (referent) | 1 (referent) | ||||
| CSF1R: high expression (N = 30) | 2.90 (1.41–6.11) | 0.0041 | 2.66 (1.28–5.66) | 0.0088 | 2.63 (1.37–5.09) | 0.0038 | 2.21 (1.05–4.77) | 0.037 |
| Ever-smoking patients | ||||||||
| CSF1R: low–moderate expression (N = 76) | 1 (referent) | 1 (referent) | 1 (referent) | 1 (referent) | ||||
| CSF1R: high expression (N = 35) | 1.11 (0.59–2.00) | 0.73 | 1.21 (0.65–2.19) | 0.54 | 0.84 (0.48–1.43) | 0.53 | 0.84 (0.47–1.43) | 0.52 |
| p values for interaction *** | 0.049 | 0.12 | 0.0078 | 0.062 | ||||
| Patients and CSF1R Expression Status | Lung Cancer-Specific Mortality | Overall Mortality | ||||||
|---|---|---|---|---|---|---|---|---|
| Univariable Analysis | Multivariable Analysis ** | Univariable Analysis | Multivariable Analysis ** | |||||
| HR (95% CI) | p Values | HR (95% CI) | p Values | HR (95% CI) | p Values | HR (95% CI) | p Values | |
| Female patients | ||||||||
| CSF1R: low–moderate expression (N = 59) | 1 (referent) | 1 (referent) | 1 (referent) | 1 (referent) | ||||
| CSF1R: high expression (N = 30) | 2.28 (1.07–4.88) | 0.033 | 4.16 (1.62–11.0) | 0.0034 | 2.23 (1.13–4.39) | 0.021 | 2.37 (1.19–4.73) | 0.015 |
| Never-smoking female patients | ||||||||
| CSF1R: low–moderate expression (N = 48) | 1 (referent) | 1 (referent) | 1 (referent) | 1 (referent) | ||||
| CSF1R: high expression (N = 22) | 2.78 (1.22–6.32) | 0.015 | 3.31 (1.42–7.80) | 0.0060 | 2.69 (1.30–5.53) | 0.0081 | 3.18 (1.50–6.76) | 0.0028 |
| Ever-smoking female patients | ||||||||
| CSF1R: low–moderate expression (N = 11) | 1 (referent) | 1 (referent) | 1 (referent) | 1 (referent) | ||||
| CSF1R: high expression (N = 8) | 1.33 (0.16–11.1) | 0.77 | 0.92 (0.11–7.86) | 0.93 | 1.33 (0.16–11.1) | 0.77 | 0.92 (0.11–7.86) | 0.93 |
| p values for interaction *** | 0.49 | 0.29 | 0.51 | 0.31 | ||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Inamura, K.; Shigematsu, Y.; Ninomiya, H.; Nakashima, Y.; Kobayashi, M.; Saito, H.; Takahashi, K.; Futaya, E.; Okumura, S.; Ishikawa, Y.; et al. CSF1R-Expressing Tumor-Associated Macrophages, Smoking and Survival in Lung Adenocarcinoma: Analyses Using Quantitative Phosphor-Integrated Dot Staining. Cancers 2018, 10, 252. https://doi.org/10.3390/cancers10080252
Inamura K, Shigematsu Y, Ninomiya H, Nakashima Y, Kobayashi M, Saito H, Takahashi K, Futaya E, Okumura S, Ishikawa Y, et al. CSF1R-Expressing Tumor-Associated Macrophages, Smoking and Survival in Lung Adenocarcinoma: Analyses Using Quantitative Phosphor-Integrated Dot Staining. Cancers. 2018; 10(8):252. https://doi.org/10.3390/cancers10080252
Chicago/Turabian StyleInamura, Kentaro, Yasuyuki Shigematsu, Hironori Ninomiya, Yasuhiro Nakashima, Maki Kobayashi, Haruyuki Saito, Katsuhiro Takahashi, Etsuko Futaya, Sakae Okumura, Yuichi Ishikawa, and et al. 2018. "CSF1R-Expressing Tumor-Associated Macrophages, Smoking and Survival in Lung Adenocarcinoma: Analyses Using Quantitative Phosphor-Integrated Dot Staining" Cancers 10, no. 8: 252. https://doi.org/10.3390/cancers10080252





