Advanced Capillary Soft Valves for Flow Control in Self-Driven Microfluidics
Abstract
:1. Introduction
2. Experimental Section
3. Results and Discussion
3.1. Working principle of CSVs
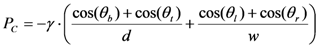
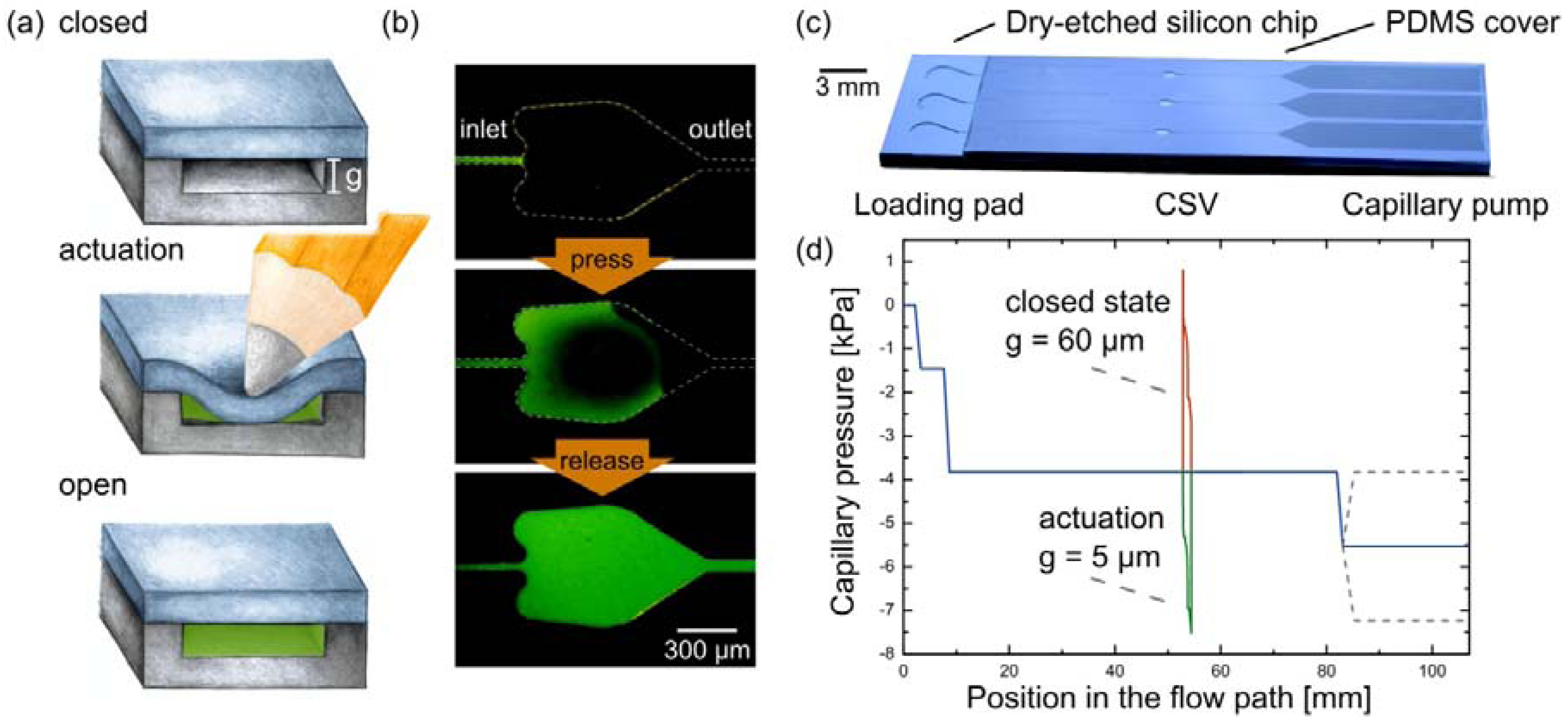
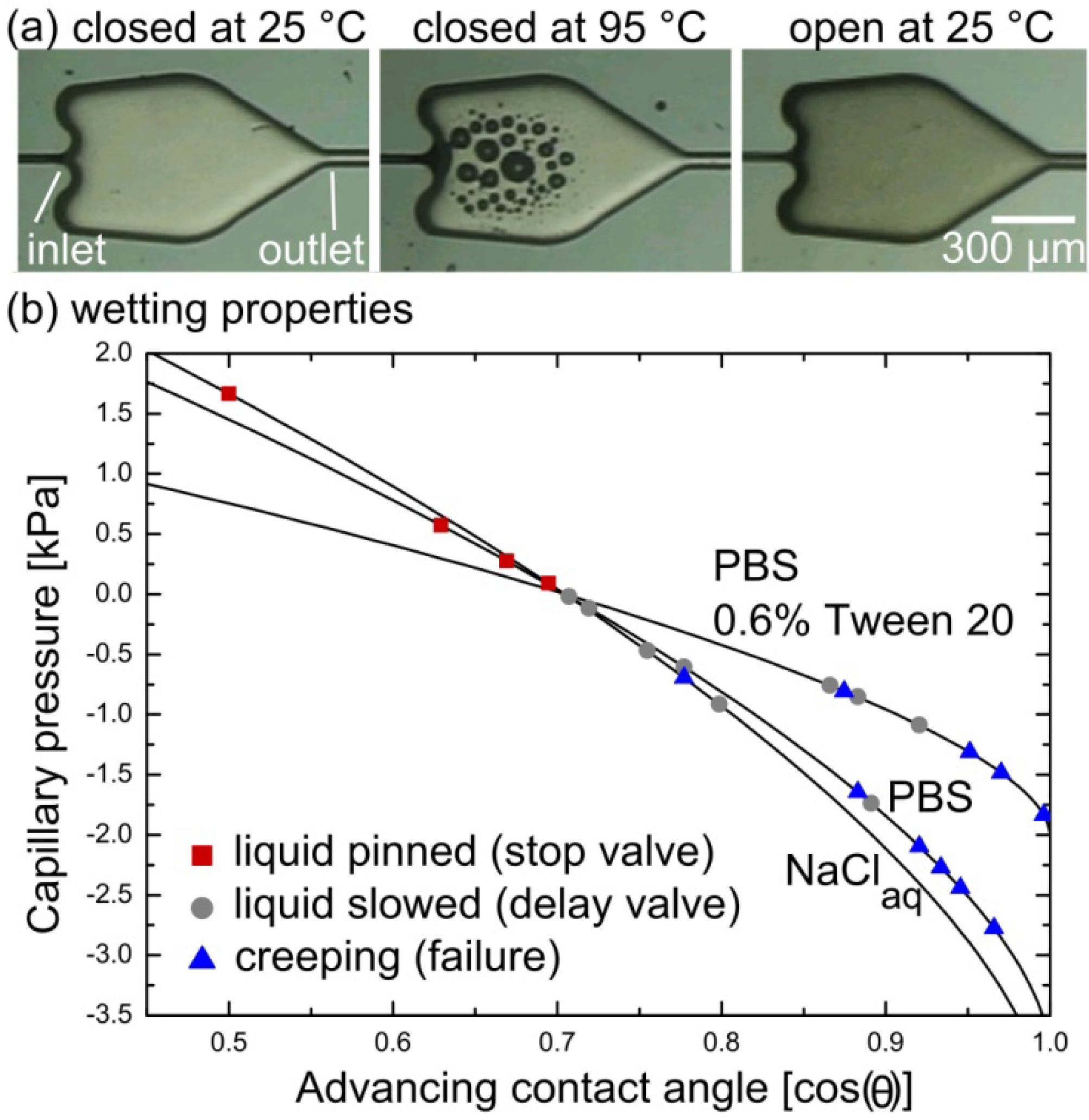
3.2. Performance of CSVs at Elevated Temperatures and with Different Liquids

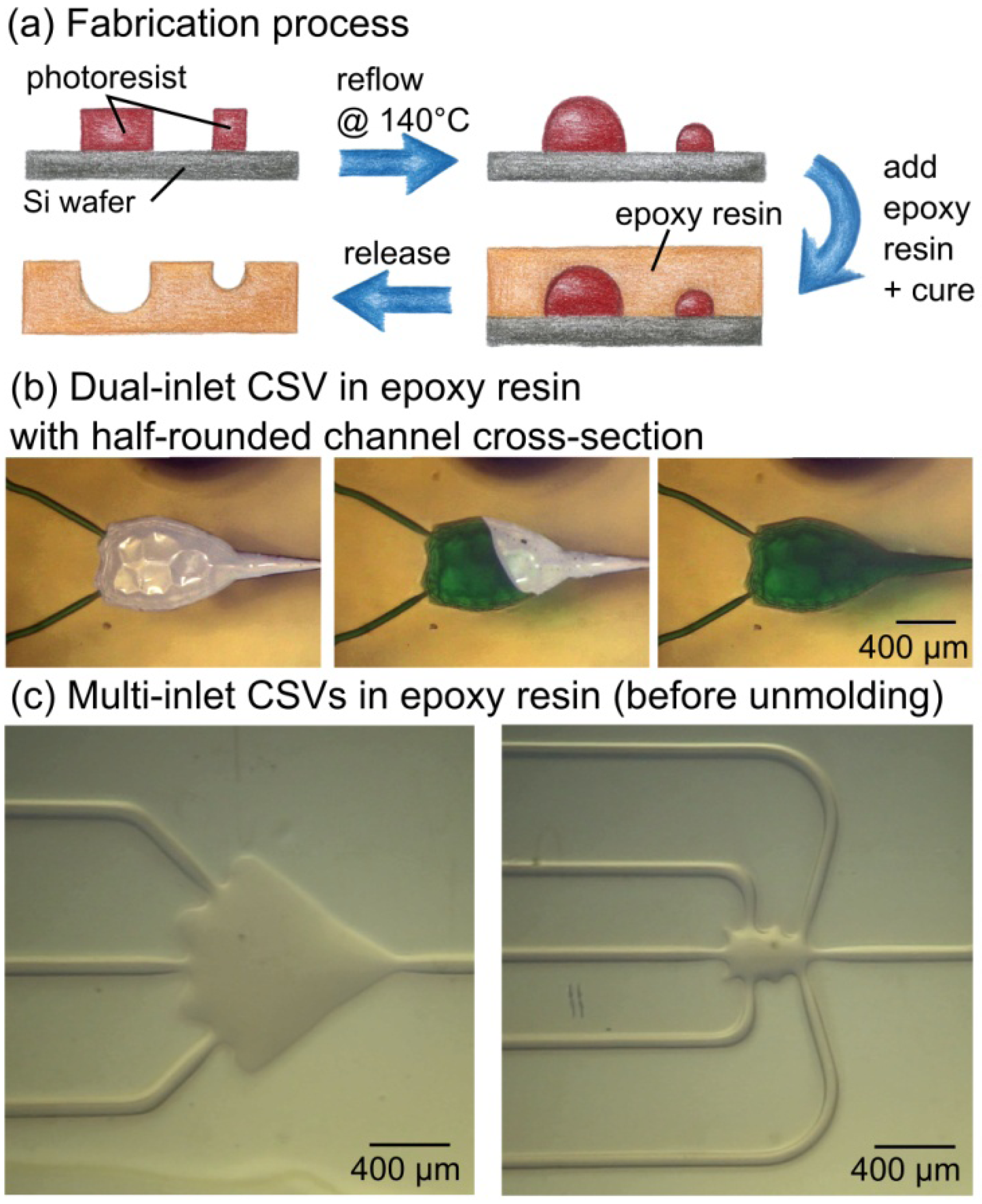
3.3. Advanced CSVs
4. Conclusions
Acknowledgments
References
- Weibel, D.; Whitesides, G. Applications of microfluidics in chemical biology. Curr. Opin. Chem. Biol. 2006, 10, 584–591. [Google Scholar] [CrossRef]
- Gervais, L.; de Rooij, N.; Delamarche, E. Microfluidics chips for point-of-care immunodiagnostics. Adv. Mater. 2011, 23, H151–H176. [Google Scholar] [CrossRef]
- Oh, K.W.; Ahn, C.H. A review of microvalves. J. Micromech. Microeng. 2006, 16, R13–R35. [Google Scholar] [CrossRef]
- Unger, M.A.; Chou, H.-P.; Thorsen, T.; Scherer, A.; Quake, S.R. Monolithic microfabricated valves and pumps by multilayer soft lithography. Science 2000, 288, 113–116. [Google Scholar] [CrossRef]
- Garcia-Cordero, J.L.; Kurzbuch, D.; Benito-Lopez, F.; Diamond, D.; Lee, L.P.; Ricco, A.J. Optically addressable single-use microfluidic valves by laser printer lithography. Lab Chip 2010, 10, 2680–2687. [Google Scholar] [CrossRef] [Green Version]
- Riegger, L.; Mielnik, M.M.; Gulliksen, A.; Mark, D.; Steigert, J.; Lutz, S.; Clad, M.; Zengerle, R.; Koltay, P.; Hoffmann, J. Dye-based coatings for hydrophobic valves and their application to polymer labs-on-a-chip. J. Micromech. Microeng. 2010, 20. [Google Scholar] [CrossRef]
- Gui, L.; Liu, J. Ice valve for a mini/micro flow channel. J. Micromech. Microeng. 2004, 14, 242–246. [Google Scholar] [CrossRef]
- Beebe, D.J.; Moore, J.S.; Bauer, J.M.; Yu, Q.; Liu, R.H.; Devadoss, C.; Jo, B.-H. Functional hydrogel structures for autonomous flow control inside microfluidic channels. Nature 2000, 404, 588–590. [Google Scholar] [CrossRef]
- Mao, X.; Huang, T.J. Microfluidic diagnostics for the developing world. Lab Chip 2012, 12, 1412–1416. [Google Scholar] [CrossRef]
- Gervais, L.; Hitzbleck, M.; Delamarche, E. Capillary-driven multiparametric microfluidic chips for one-step immunoassays. Biosens. Bioelectron. 2011, 27, 64–70. [Google Scholar] [CrossRef]
- Hitzbleck, M.; Avrain, L.; Smekens, V.; Lovchik, R.D.; Mertens, P.; Delamarche, E. Capillary soft valves for microfluidics. Lab Chip 2012, 12, 1972–1978. [Google Scholar] [CrossRef]
- Zimmermann, M.; Schmid, H.; Hunziker, P.; Delamarche, E. Capillary pumps for autonomous capillary systems. Lab Chip 2007, 7, 119–125. [Google Scholar] [CrossRef]
- Seemann, R.; Brinkmann, M.; Kramer, E.J.; Lange, F.F.; Lipowsky, R. Wetting morphologies at microstructured surfaces. P. Natl. Acad. Sci. USA 2005, 102, 1848–1852. [Google Scholar] [CrossRef]
- Kusumaatmaja, H.; Pooley, C.M.; Girardo, S.; Pisignano, D.; Yeomans, J.M. Capillary filling in patterned channels. Phys. Rev. E 2008, 77. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hitzbleck, M.; Delamarche, E. Advanced Capillary Soft Valves for Flow Control in Self-Driven Microfluidics. Micromachines 2013, 4, 1-8. https://doi.org/10.3390/mi4010001
Hitzbleck M, Delamarche E. Advanced Capillary Soft Valves for Flow Control in Self-Driven Microfluidics. Micromachines. 2013; 4(1):1-8. https://doi.org/10.3390/mi4010001
Chicago/Turabian StyleHitzbleck, Martina, and Emmanuel Delamarche. 2013. "Advanced Capillary Soft Valves for Flow Control in Self-Driven Microfluidics" Micromachines 4, no. 1: 1-8. https://doi.org/10.3390/mi4010001



