Profiling of Amatoxins and Phallotoxins in the Genus Lepiota by Liquid Chromatography Combined with UV Absorbance and Mass Spectrometry
Abstract
:1. Introduction
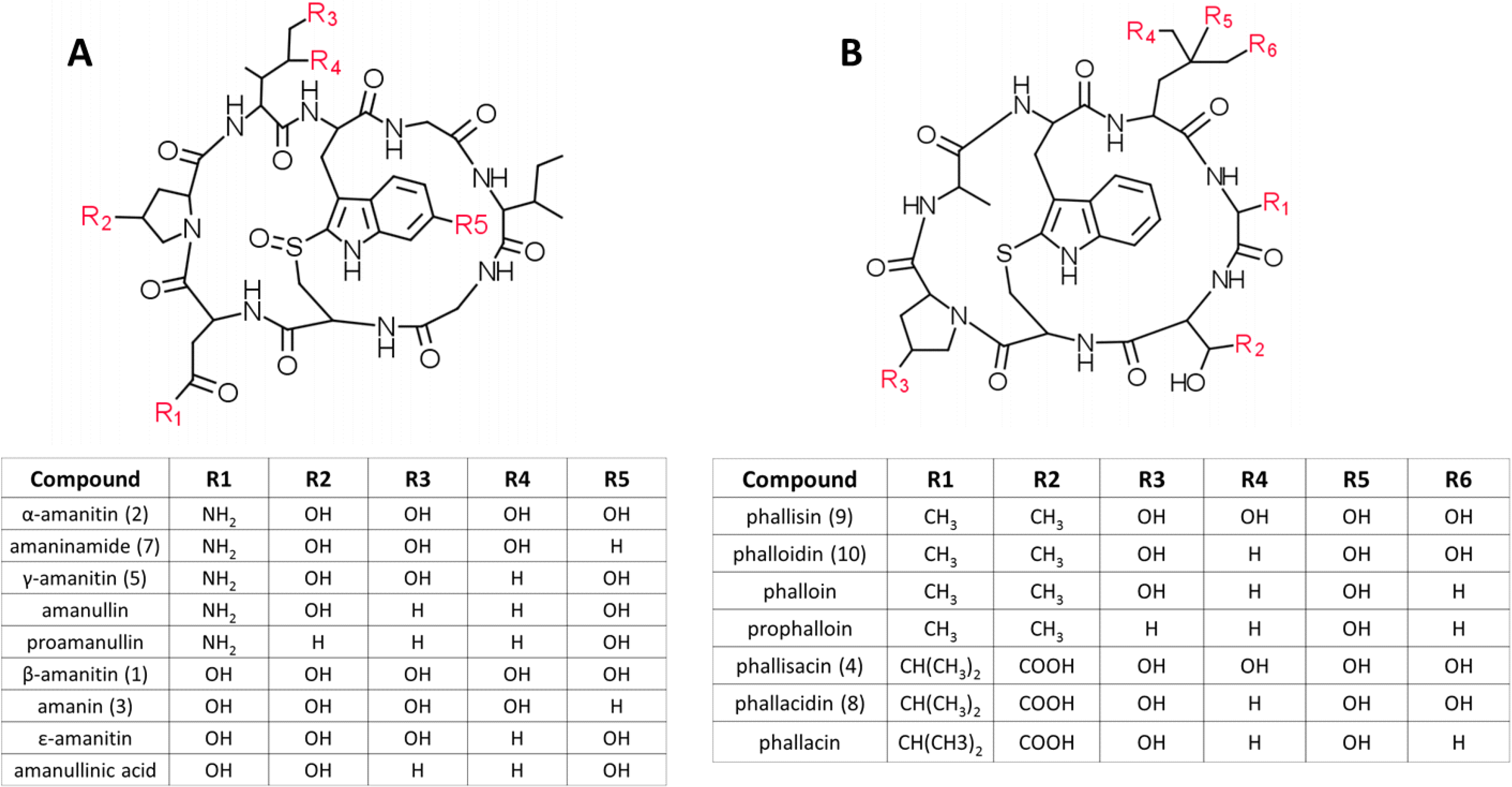
2. Results
2.1. Mushroom Identification
2.2. Toxin Analysis
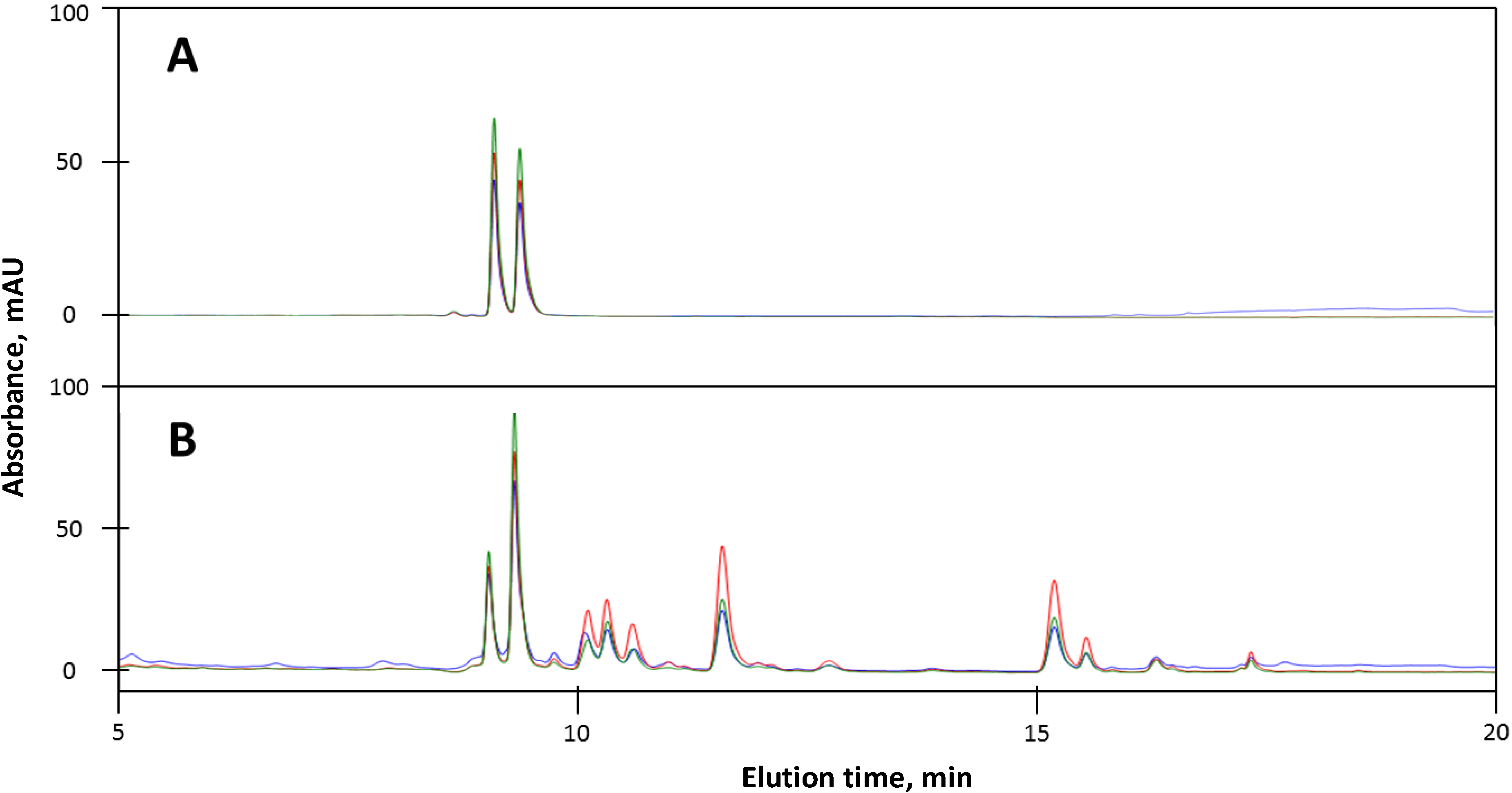
2.2.1. Standards and A. phalloides
| Peak Number | Compound | True Mass (Da) | Observed Masses (m/z) |
|---|---|---|---|
| 1 | β-amanitin | 919.338182 | 920.3 [M+H+], 942.4 [M+Na+], 958.4 [M+K+] |
| 2 | α-amanitin | 918.354170 | 919.3 [M+H+], 941.2 [M+Na+], 957.2 [M+K+] |
| 3 | amanin | 903.343267 | 904.3 [M+H+], 926.3 [M+Na+], 942.2 [M+K+] |
| 4 | phallisacin | 862.316720 | 863.3 [M+H+], 885.3 [M+Na+], 901.2 [M+K+], 925.3 |
| 5 | γ-amanitin | 902.359252 | 903.4 [M+H+], 925.4 [M+Na+], 941.3 [M+K+] |
| 6 | phallisin II | 804.311240 | 805.3 [M+H+], 827.3 [M+Na+], 843.2 [M+K+], 740.5 |
| 7 | amaninamide | 902.359252 | 903.3 [M+H+], 925.3 [M+Na+], 941.2 [M+K+] |
| 8 | phallacidin | 846.321804 | 847.3 [M+H+], 869.3 [M+Na+], 885.3 [M+K+] |
| 9 | phallisin I | 804.311240 | 805.4 [M+H+], 827.3 [M+Na+], 843.3 [M+K+], 864.3, 905.3, 927.1, 942.9 |
| 10 | phalloidin | 788.316330 | 789.3 [M+H+], 811.3 [M+Na+], 827.3 [M+K+], 848.3 |
| 11 | unknown | - | 889.3 [M+H+], 911.3 [M+Na+], 927.2 [M+K+], 789.2, 811.3, 827.2, 848.3 |
| 12 | unknown | - | 872.5 [M+H+], 893.4 [M+Na+], 914.5 |
| 13 | unknown | - | 915.4, 937.4, 953.3, 960.6, 974.4 |
| 14 | unknown | - | 755.3, 795.3, 811.2, 832.4, 869.5, 891.5 |
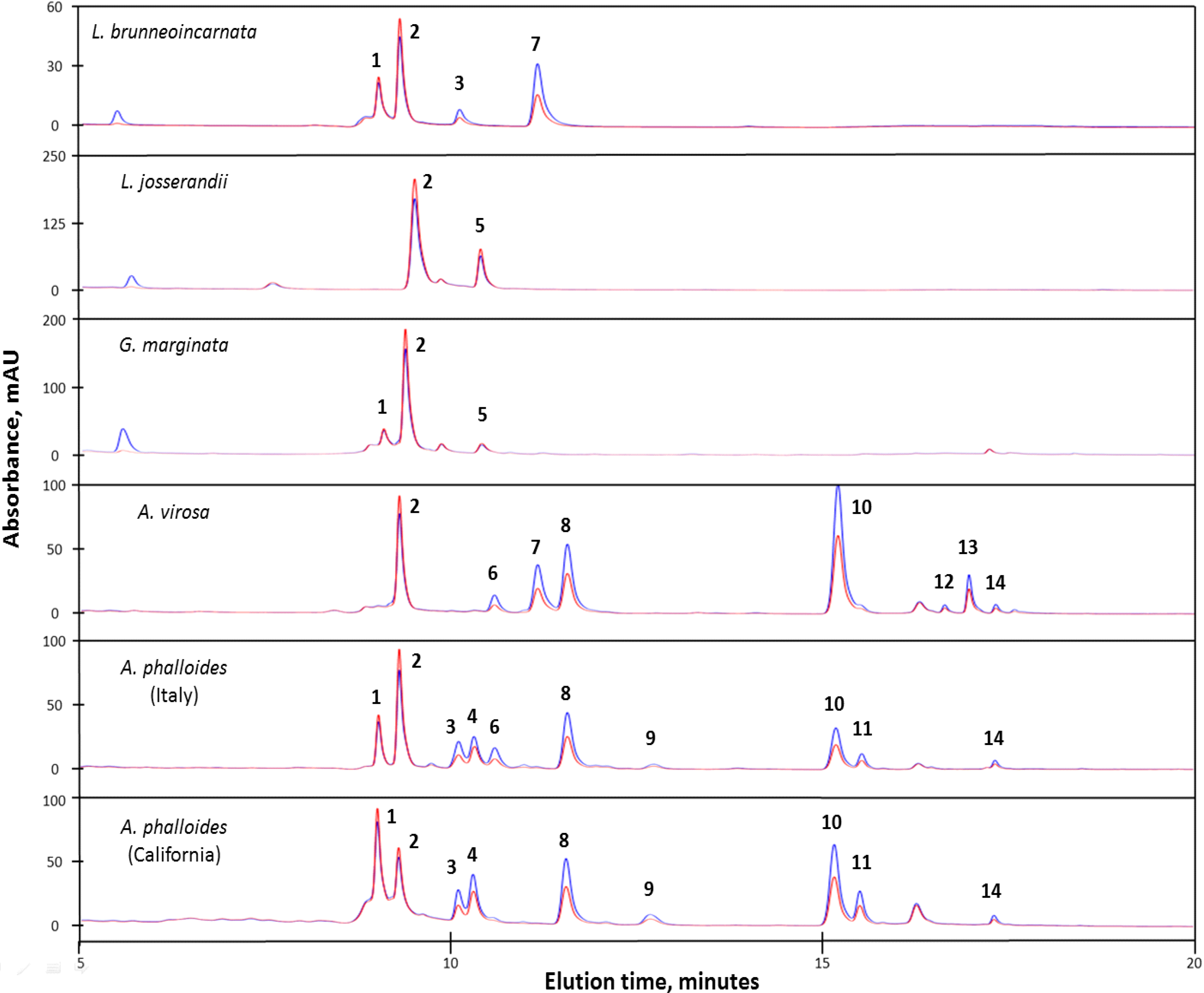
2.2.2. Lepiota Species
| Species | α-Amanitin Content (mg/g dry weight) |
|---|---|
| A. phalloides (Italy) | 1.33 |
| A. phalloides (USA) | 0.88 |
| A. virosa | 1.39 |
| G. marginata (mycelium) | 0.57 |
| L. josserandii Sample #1 | 4.24 |
| L. josserandii Sample #2 | 4.39 |
| L. josserandii Sample #3 | 3.99 |
| L. brunneoincarnata Sample #1 | 0.82 |
| L. brunneoincarnata Sample #2 | 0.69 |
2.2.3. A. phalloides from Italy and California
2.2.4. Amanita virosa and Galerina marginata
2.2.5. Unidentified Compounds
3. Discussion
4. Experimental Section
4.1. Biological Material
4.2. ITS Sequencing
4.3. Extraction
4.4. Liquid Chromatography-Mass Spectrometry (LC-MS)
5. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflict of Interest
References
- Bresinsky, A.; Besl, H. A Colour Atlas of Poisonous Fungi; Bisset, N.G., Translator; Wolfe: London, UK, 1990. [Google Scholar]
- Novello, F.; Fiume, L.; Stirpe, F. Inhibition by α-amanitin of ribonucleic acid polymerase solubilized from rat liver nuclei. Biochem. J. 1970, 116, 177–180. [Google Scholar]
- Hallen, H.E.; Luo, H.; Scott-Craig, J.S.; Walton, J.D. Gene family encoding the major toxins of lethal Amanita mushrooms. Proc. Natl. Acad. Sci. USA 2007, 104, 19097–19101. [Google Scholar] [CrossRef]
- Luo, H.; Hallen-Adams, H.E.; Scott-Craig, J.S.; Walton, J.D. Ribosomal biosynthesis of α-amanitin in Galerina marginata. Fung. Genet. Biol. 2012, 49, 123–129. [Google Scholar] [CrossRef]
- Arnison, P.G.; Bibb, M.J.; Bierbaum, G.; Bowers, A.A.; Bugni, T.S.; Bulaj, G.; Camarero, J.A.; Campopiano, D.J.; Challis, G.L.; Clardy, J.; et al. Ribosomally synthesized and post-translationally modified peptide natural products: Overview and recommendations for a universal nomenclature. Nat. Prod. Rep. 2013, 30, 108–160. [Google Scholar] [CrossRef]
- Camus, C.M.; Gerault, A.M.; Houssin, D.P.; Launois, B.G.; Messner, M.J.M.; Meunier, B.C. Liver transplantation after severe poisoning due to amatoxin-containing Lepiota—Report of three cases. J. Toxicol. Clin. Toxicol. 1995, 33, 165–171. [Google Scholar] [CrossRef]
- Feinfeld, D.A.; Mofenson, H.C.; Caraccio, T.; Kee, M. Poisoning by amatoxin-containing mushrooms in suburban New York—Report of four cases. J. Toxicol. Clin. Toxicol. 1994, 32, 715–721. [Google Scholar] [CrossRef]
- Haines, J.H.; Lichsteine, E.; Glickerman, D. A fatal poisoning from an amatoxin containing Lepiota. Mycopathologia 1986, 93, 15–17. [Google Scholar] [CrossRef]
- Kervègant, M.; de Haro, L.; Patat, A.M.; Pons, C.; Thomachot, L.; Minodier, P. Phalloides syndrome poisoning after ingestion of Lepiota mushrooms. Wilderness Environ. Med. 2013, 24, 170–172. [Google Scholar] [CrossRef]
- Mottram, A.R.; Lazio, M.P.; Bryant, S.M. Lepiota subincarnata J.E. Lange induced fulminant hepatic failure presenting with pancreatitis. J. Med. Toxicol. 2010, 6, 155–157. [Google Scholar] [CrossRef]
- Paydas, C.; Kocak, R.; Erturk, F.; Erken, E.; Zaksu, H.S.; Gurcay, A. Poisoning due to amatoxin-containing Lepiota species. Br. J. Clin. Pract. 1990, 44, 450–453. [Google Scholar]
- Ramirez, P.; Parrilla, P.; Bueno, F.S.; Robles, R.; Pons, J.A.; Bixquert, V.; Nicolas, S.; Nunez, R.; Alegria, M.S.; Miras, M.; et al. Fulminant hepatic failure after Lepiota mushroom poisoning. J. Hepatol. 1993, 19, 51–54. [Google Scholar] [CrossRef]
- Besl, H.; Mack, P.; Schmid-Heckel, H. Giftpilze in den gattungen Galerina und Lepiota. Z. Mykol. 1984, 50, 183–192. (in German). [Google Scholar]
- Beutler, J.A.; Vergeer, P.P. Amatoxins in American mushrooms: Evaluation of the Meixner test. Mycologia 1980, 72, 1142–1149. [Google Scholar] [CrossRef]
- Gérault, A.; Girre, L. Recherches toxicologiques sur le genre Lepiota Fries. C. R. Acad. Sci. Paris Sér. D 1975, 280, 2841–2843. (in French). [Google Scholar]
- Andres, R.Y.; Frei, W.; Gautschi, K.; Vonderschmitt, D.J. Radioimmunoassay for amatoxins by use of a rapid, 125I-tracer-based system. Clin. Chem. 1986, 32, 1751–1755. [Google Scholar]
- Faulstich, H.; Zobeley, S.; Trischmann, H. A rapid radioimmunoassay, using a nylon support, for amatoxins from Amanita mushrooms. Toxicon 1982, 20, 913–924. [Google Scholar] [CrossRef]
- Epis, S.; Matinato, C.; Gentili, G.; Varotto, F.; Bandi, C.; Sassera, D. Molecular detection of poisonous mushrooms in different matrices. Mycologia 2010, 102, 747–754. [Google Scholar] [CrossRef]
- Wieland, T. Peptides of Poisonous Amanita Mushrooms; Springer: New York, NY, USA, 1986. [Google Scholar]
- Clarke, D.B.; Lloyd, A.S.; Robb, P. Application of liquid chromatography coupled to time-of-flight mass spectrometry separation for rapid assessment of toxins in Amanita mushrooms. Anal. Meth. 2012, 4, 1298–1309. [Google Scholar] [CrossRef]
- Enjalbert, F.; Gallion, C.; Jehl, F.; Monsteil, H.; Faulstich, H.J. Simultaneous assay for amatoxins and phallotoxins in Amanita phalloides Fr., by high-performance liquid chromatography. J. Chromatogr. 1992, 598, 227–236. [Google Scholar] [CrossRef]
- Jansson, D.; Fredriksson, S.A.; Hermann, A.; Nilsson, C. A concept study on identification and attribution profiling of chemical threat agents using liquid chromatography-mass spectrometry applied to Amanita toxins in food. Forensic Sci. Int. 2012, 221, 44–49. [Google Scholar] [CrossRef]
- Pringle, A.; Adams, R.I.; Cross, H.B.; Bruns, T.D. The ectomycorrhizal fungus Amanita phalloides was introduced and is expanding its range on the west coast of North America. Mol. Ecol. 2009, 18, 817–833. [Google Scholar] [CrossRef]
- Yocum, R.R.; Simons, D.M. Amatoxins and phallotoxins in Amanita species of the northeastern United States. Lloydia 1977, 40, 178–190. [Google Scholar]
- Buku, A.; Wieland, T.; Bodenmuller, H.; Faulstich, H. Amaninamide, a new toxin of Amanita virosa mushrooms. Experientia 2008, 36, 33–34. [Google Scholar]
- Muraoka, S.; Shinozawa, T. Effective production of amanitins by two-step cultivation of the basidiomycete, Galerina fasciculata GF-060. J. Biosci. Bioeng. 1999, 889, 73–76. [Google Scholar]
- Ahmed, W.H.A.; Gonmori, K.; Suzuki, M.; Watanabe, K.; Suzuki, O. Simultaneous analysis of α-amanitin, β-amanitin, and phalloidin in toxic mushrooms by liquid chromatography coupled to time-of-flight mass spectrometry. Forensic Toxicol. 2010, 28, 69–76. [Google Scholar] [CrossRef]
- Faulstich, H.; Buku, A.; Bodenmuller, H.; Wieland, T. Virotoxins: Actin-binding cyclic peptides of Amanita virosa mushrooms. Biochemistry 1980, 19, 3334–3343. [Google Scholar] [CrossRef]
- Mazza, R. I Funghi—Guida al Riconoscimento; Manuali Sonzogno: Milan, Italy, 1995. (In Italian) [Google Scholar]
- Moser, M. Guida alla Determinazione dei Funghi; Saturnia: Trento, Italy, 1993; Volume 1. (In Italian) [Google Scholar]
- Candusso, M.; Lanzoni, G. Fungi Europaei, Lepiota; Candusso Edizioni: Alassio, Italy, 1990; Volume 4, p. 743. (In Italian) [Google Scholar]
- Lange, J.E. Flora Agaricina Danica; Recato: Copenhagen, Denmark, 1935–1940; Volumes 1–5. [Google Scholar]
- Vellinga, E.C. Macrolepiota Sing., Leucocoprinus Pat., Leucoagaricus (Locq. ex) Sing., Lepiota (Pers.: Fr.) S.F. Gray, Cystolepiota Sing., and Melanophyllum Velen. In Flora Agaricina Neerlandica; Noordeloos, M.E., Kuyper, T.W., Vellinga, E.C., Eds.; A.A. Balkema: Rotterdam, The Netherlands, 2001; Volume 5, pp. 64–162. [Google Scholar]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenies. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic: San Diego, CA, USA, 1990; pp. 315–322. [Google Scholar]
- Al Shahni, M.M.; Makimura, K.; Yamada, T.; Satoh, K.; Ishihara, K.; Sawada, T. Direct colony PCR of several medically important fungi using Ampdirect Plus. Jpn. J. Infect. Dis. 2009, 62, 164–167. [Google Scholar]
- Helfer, A.G.; Meyer, M.R.; Michely, J.A.; Maurer, H.H. Direct analysis of the mushroom poisons α- and β-amanitin in human urine using a novel on-line turbulent flow chromatography mode coupled to liquid chromatography-high resolution-mass spectrometry/mass spectrometry. J. Chromatogr. 2014, 1325, 92–98. [Google Scholar]
- Leite, M.; Freitas, A.; Azul, A.M.; Barbosa, J.; Costa, S.; Ramos, F. Development, optimization and application of an analytical methodology by ultra performance liquid chromatography-tandem mass spectrometry for determination of amanitins in urine and liver samples. Anal. Chim. Acta 2013, 799, 77–87. [Google Scholar] [CrossRef]
- Nomura, M.; Suzuki, Y.; Kaneko, R.; Ogawa, T.; Hattori, H.; Seno, H.; Ishii, A. Simple and rapid analysis of amatoxins using UPLC-MS-MS. Forensic Toxicol. 2012, 30, 185–192. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sgambelluri, R.M.; Epis, S.; Sassera, D.; Luo, H.; Angelos, E.R.; Walton, J.D. Profiling of Amatoxins and Phallotoxins in the Genus Lepiota by Liquid Chromatography Combined with UV Absorbance and Mass Spectrometry. Toxins 2014, 6, 2336-2347. https://doi.org/10.3390/toxins6082336
Sgambelluri RM, Epis S, Sassera D, Luo H, Angelos ER, Walton JD. Profiling of Amatoxins and Phallotoxins in the Genus Lepiota by Liquid Chromatography Combined with UV Absorbance and Mass Spectrometry. Toxins. 2014; 6(8):2336-2347. https://doi.org/10.3390/toxins6082336
Chicago/Turabian StyleSgambelluri, R. Michael, Sara Epis, Davide Sassera, Hong Luo, Evan R. Angelos, and Jonathan D. Walton. 2014. "Profiling of Amatoxins and Phallotoxins in the Genus Lepiota by Liquid Chromatography Combined with UV Absorbance and Mass Spectrometry" Toxins 6, no. 8: 2336-2347. https://doi.org/10.3390/toxins6082336





