Sedimentation Patterns of Toxin-Producing Microcystis Morphospecies in Freshwater Reservoirs
Abstract
:1. Introduction
2. Results and Discussion
2.1. Microcystis and MCs in Water
| Reservoir | Watershed | River | Depth (m) | Water uses b | |
|---|---|---|---|---|---|
| Mean | Maximum | ||||
| Cogotas | Duero | Adaja | 14.9 a | 60 a | D, I |
| Santillana | Tajo | Manzanares | 8.7 | 36 | D |
| Valmayor | Tajo | Aulencia | 16.4 | 51 | D, R |
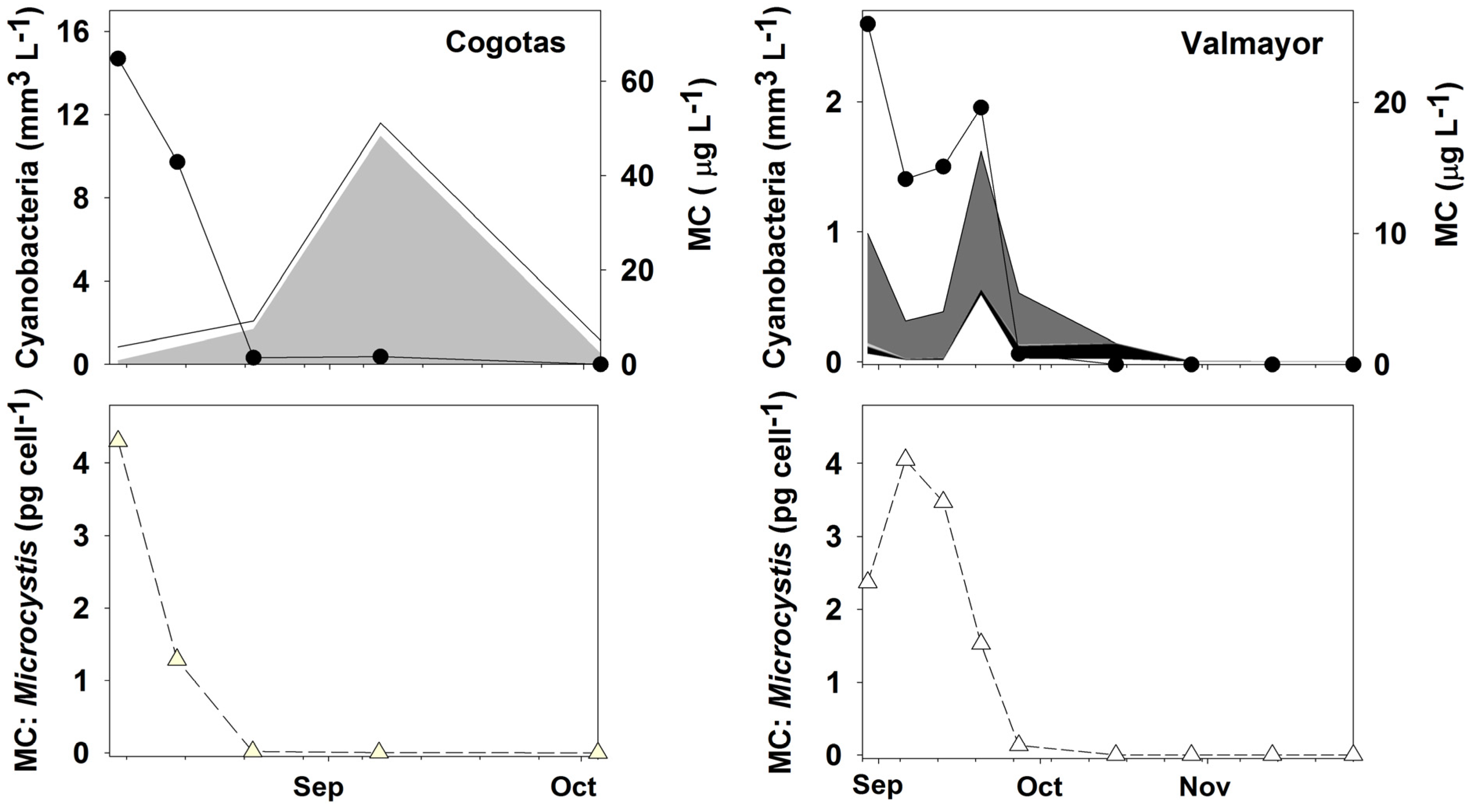
2.2. Settling Dynamics of Microcystis
2.2.1. Spatiotemporal Patterns
| Reservoir | Date | Total Microcystis settled | Dominant species b | |||
|---|---|---|---|---|---|---|
| 109 cells m−2 d−1 | mm3 m−2 d−1 | Mic: OM (%) a | ||||
| Cogotas | Epilimnetic | 14/08/2006 | 13.4 ± 3.1 | 300.4 ± 69.2 | 3.8 | M. flos-aquae (100%) |
| 23/08/2006 | 18.2 ± 0.9 | 409.3 ± 21.7 | 4.2 | M. flos-aquae (100%) | ||
| 07/09/2006 | 36.7 ± 0.7 | 823.4 ± 16.8 | 7.6 | M. flos-aquae (100%) | ||
| 03/10/2006 | 17.3 ± 9.1 | 389.2 ± 203.7 | 5.3 | M. flos-aquae (100%) | ||
| Santillana | Epilimnetic | 01/08/2007 | 0.07 ± 0.04 | 1.5 ± 0.8 | 0.1 | M. flos-aquae (100%) |
| 13/08/2007 | 0.07 ± 0.008 | 1.4 ± 0.2 | 0.1 | M. flos-aquae (100%) | ||
| 29/08/2007 | - | - | - | - | ||
| 11/09/2007 | 0.001 ± 0.001 | 0.01 ± 0.008 | 0.002 | M. flos-aquae (100%) | ||
| 05/10/2007 | 0.001 ± 0.001 | 0.03 ± 0.005 | 0.003 | M. flos-aquae (100%) | ||
| 22/10/2007 | 0.001 ± 0.001 | 0.02 ± 0.008 | 0.002 | M. flos-aquae (100%) | ||
| Hypolimnetic | 01/08/2007 | 0.1 ± 0.04 | 2.3 ± 0.8 | 0.1 | M. flos-aquae (100%) | |
| 13/08/2007 | 0.2 ± 0.08 | 4.7 ± 1.9 | 0.3 | M. flos-aquae (87%) | ||
| 29/08/2007 | - | - | - | - | ||
| 11/09/2007 | - | - | - | - | ||
| 05/10/2007 | 0.03 ± 0.004 | 0.6 ± 0.1 | 0.05 | M. flos-aquae (100%) | ||
| 22/10/2007 | 0.008 ± 0.001 | 0.2 ± 0.02 | 0.005 | M. flos-aquae (100%) | ||
| Valmayor | Epilimnetic | 06/09/2007 | 2.9 ± 0.5 | 209.0 ± 37.8 | 2.2 | M. aeruginosa (98%) |
| 13/09/2007 | 0.7 ± 0.06 | 50.3 ± 4.1 | 0.5 | M. aeruginosa (99%) | ||
| 27/09/2007 | 3.4 ± 0.3 | 190.3 ± 19.6 | 2.8 | M. aeruginosa (86%) | ||
| 15/10/2007 | 0.4 ± 0.2 | 27.0 ± 13.6 | 0.3 | M. aeruginosa (84%) | ||
| 29/10/2007 | 1.2 ± 0.2 | 52.4 ± 6.5 | 1.2 | M. novacekii (55%) | ||
| 28/11/2007 | 0.05 ± 0.008 | 3.0 ± 0.5 | 0.02 | M. aeruginosa (81%) | ||
| Hypolimnetic | 06/09/2007 | 1.6 ± 0.6 | 101.8 ± 34.8 | 0.9 | M. aeruginosa (98%) | |
| 13/09/2007 | 1.5 ± 0.6 | 91.0 ± 37.5 | 1.2 | M. aeruginosa (99%) | ||
| 27/09/2007 | 4.2 ± 0.1 | 295.4 ± 2.5 | 1.2 | M. aeruginosa (98%) | ||
| 15/10/2007 | 2.6 ± 0.4 | 129.7 ± 0.02 | 1.0 | M. aeruginosa (69%) | ||
| 29/10/2007 | 1.1 ± 0.01 | 50.9 ± 4.5 | 0.7 | M. aeruginosa (58%) | ||
| 28/11/2007 | 0.08 ± 0.02 | 5.2 ± 1.3 | 0.05 | M. aeruginosa (96%) | ||
| Layer | Period | |||||
|---|---|---|---|---|---|---|
| 1–6 September | 6–13 September | 13–27 September | 27 September–15 October | 15–29 October | 29 October–28 November | |
| Epilimnion | 0.05 ± 0.009 | 0.03 ± 0.002 | 0.04 ± 0.004 | 0.01 ± 0.006 | 0.07 ± 0.009 | 0.04 ± 0.006 |
| Entire water column | 0.02 ± 0.009 | 0.04 ± 0.02 | 0.06 ± 0.001 | 0.05 ± 0.01 | 0.05 ± 0.004 | 0.05 ± 0.003 |
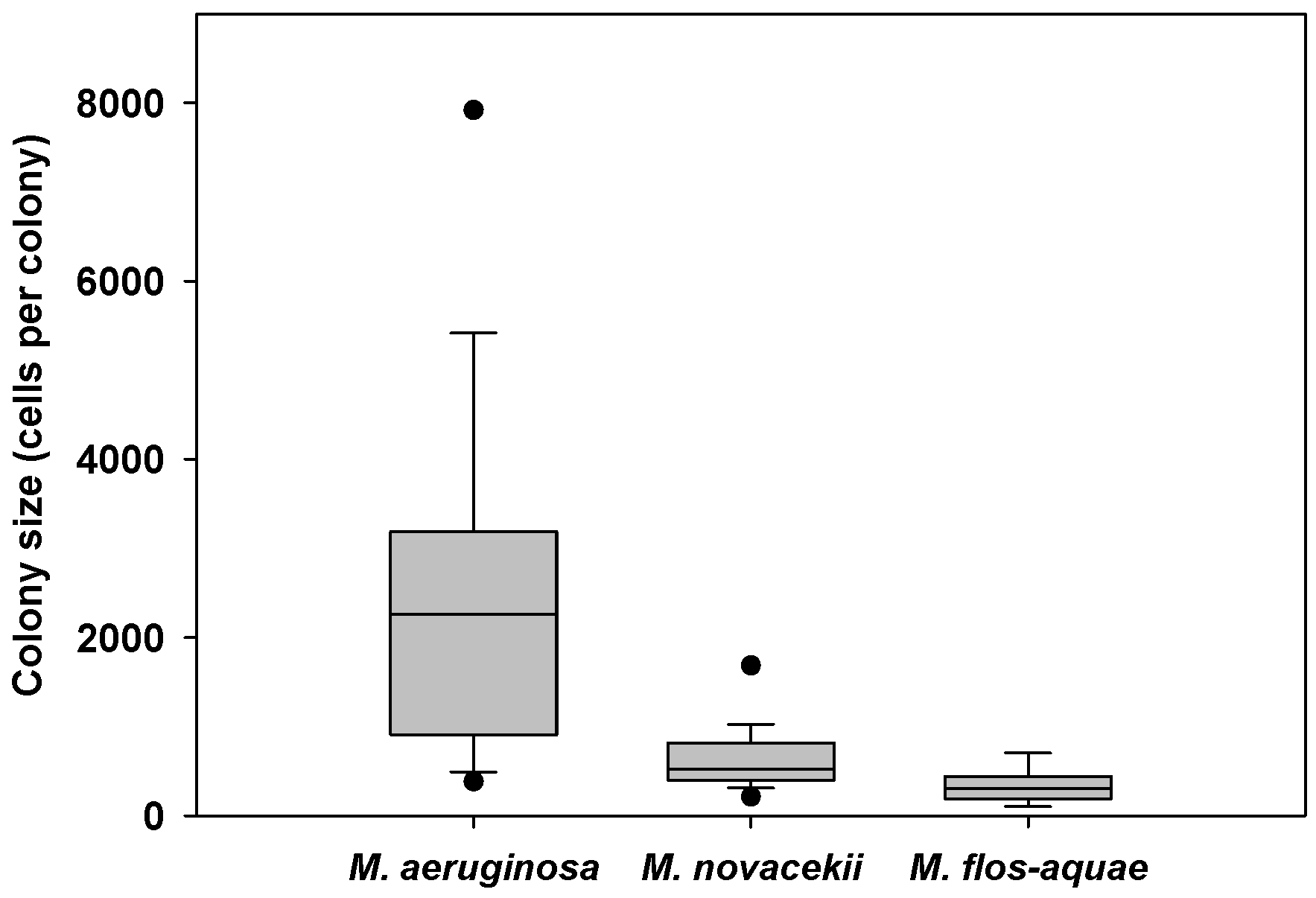
2.2.2. Microcystis Morphospecies
| Layer | Morphospecies | Estimated settling rate (d−1) | Estimated settling velocity (m d−1) | ||
|---|---|---|---|---|---|
| Average ± SD | Range | Average ± SD | Range | ||
| Epilimnion | M. aeruginosa | 0.033 ± 0.014 | 0.019–0.078 | 0.3 ± 0.1 | 0.2–0.8 |
| M. flos-aquae | 0.010 ± 0.005 | 0.004–0.017 | 0.1 ± 0.05 | 0.0–0.2 | |
| M. novacekii | 0.030 ± 0.028 | 0.005–0.080 | 0.3 ± 0.3 | 0.1–0.8 | |
| Entire column | M. aeruginosa | 0.053 ± 0.021 | 0.024–0.071 | 0.8 ± 0.3 | 0.4–1.1 |
| M. flos-aquae | 0.024 ± 0.015 | 0.010–0.042 | 0.4 ± 0.2 | 0.1–0.7 | |
| M. novacekii | 0.012 ± 0.011 | 0.006–0.029 | 0.2 ± 0.2 | 0.1–0.5 | |
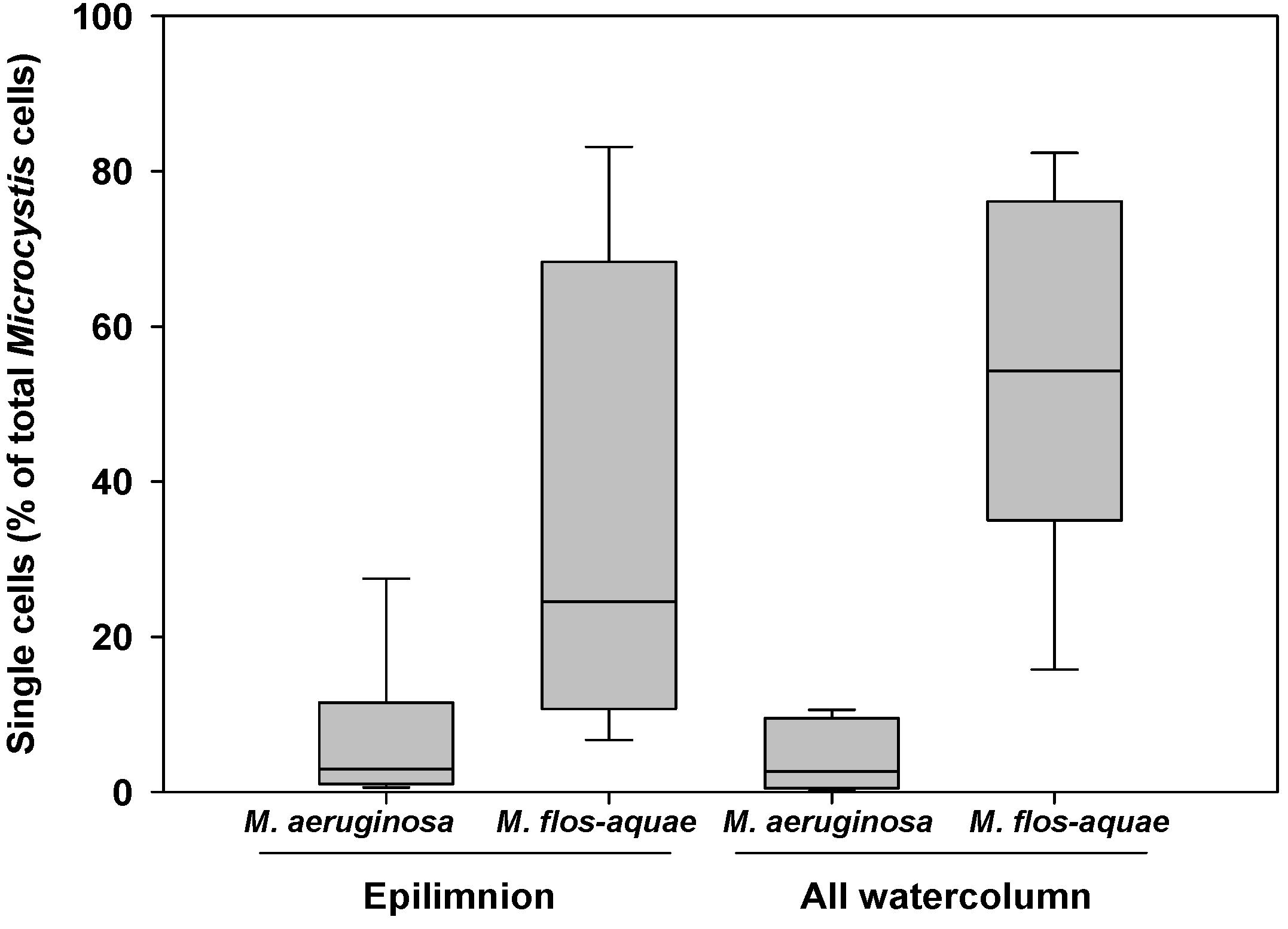
2.2.3. Colony Sizes
| Layer | Size class (cells per colony) | Estimated settling rate (d−1) | Estimated settling velocity (m d−1) | ||
|---|---|---|---|---|---|
| Average ± SD | Range | Average ± SD | Range | ||
| Epilimnion | <1000 | 0.034 ± 0.034 | 0.010–0.093 | 0.3 ± 0.3 | 0.1–0.9 |
| 1000–5000 | 0.037 ± 0.030 | 0.005–0.080 | 0.4 ± 0.3 | 0.1–0.8 | |
| >5000 | 0.043 ± 0.032 | 0.010–0.078 | 0.4 ± 0.3 | 0.1–0.8 | |
| Entire watercolumn | <1000 | 0.043 ± 0.020 | 0.006–0.064 | 0.7 ± 0.3 | 0.1–1.0 |
| 1000–5000 | 0.036 ± 0.022 | 0.018–0.067 | 0.6 ± 0.4 | 0.3–1.1 | |
| >5000 | 0.039 ± 0.024 | 0.021–0.074 | 0.6 ± 0.4 | 0.4–1.2 | |
2.3. Shifts in MC Cell Quota during Settling
| Reservoir | Period | Sample | MC content (pg MC Microcystis cell−1) |
|---|---|---|---|
| Cogotas | 7/08/2006–7/09/2006 | Subsurface water | 1.4 (0.01–4.3) |
| Epilimnetic trap | 0.1 (0.01–0.2) | ||
| Santillana | 1/08/2007–13/08/2007 | Subsurface water | Nd |
| Epilimnetic trap | 0.15 (0.14–0.15) | ||
| Hypolimnetic trap | 0.10 (0.09–0.10) | ||
| Valmayor | 30/08/2007–27/09/2007 | Subsurface water | 2.3 (0.1–4.0) |
| Epilimnetic trap | 0.6 (0.8–1.2) | ||
| Hypolimnetic trap | 0.4 (0.3–0.6) |
3. Experimental Section
3.1. Sampling Setup
3.2. Water Column Sampling
3.3. Sediment Trap Sampling
3.4. Identification and Quantification of Microcystis in Water and Sediment Traps
3.5. Estimation of Settling Rates in the Valmayor Reservoir
3.6. MC Analysis
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Sivonen, K.; Jones, G. Cyanobacterial Toxins. In Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring and Management; Chorus, I., Bartram, J., Eds.; E and FN Spon: London, UK, 1999; pp. 41–111. [Google Scholar]
- Harmful Cyanobacteria; Huisman, J.; Matthijs, H.C.P.; Visser, P.M. (Eds.) Springer-Verlag: Berlin, Germany, 2005.
- Verspagen, J.M.H.; Passarge, J.; Johnk, K.D.; Visser, P.M.; Peperzak, L.; Boers, P.; Laanbroek, H.J.; Huisman, J. Water management strategies against toxic Microcystis blooms in the Dutch delta. Ecol. Appl. 2006, 16, 313–327. [Google Scholar] [CrossRef]
- Reynolds, C.S.; Jaworski, G.H.M.; Cmiech, H.A.; Leedale, G.F. On the annual cycle of the blue-green alga Microcystis aeruginosa Kutz emend. Elenkin. Philos. Trans. R. Soc. London Ser. B 1981, 293, 419–477. [Google Scholar] [CrossRef]
- Ihle, T.; Jahnichen, S.; Benndorf, J. Wax and wane of Microcystis (Cyanophyceae) and microcystins in lake sediments: A case study in Quitzdorf reservoir (Germany). J. Phycol. 2005, 41, 479–488. [Google Scholar] [CrossRef]
- Verspagen, J.M.H.; Snelder, E.; Visser, P.M.; Johnk, K.D.; Ibelings, B.W.; Mur, L.R.; Huisman, J. Benthic-pelagic coupling in the population dynamics of the harmful cyanobacterium Microcystis. Freshwater Biol. 2005, 50, 854–867. [Google Scholar] [CrossRef]
- Preston, T.; Stewart, W.D.P.; Reynolds, C.S. Bloom-forming cyanobacterium Microcystis aeruginosa overwinters on sediment surface. Nature 1980, 288, 365–367. [Google Scholar] [CrossRef]
- Oliver, R.L.; Thomas, R.H.; Reynolds, C.S.; Walsby, A.E. The sedimentation of buoyant Microcystis colonies caused by precipitation with an iron-containing colloid. Proc. R. Soc. London B 1985, 223, 511–528. [Google Scholar] [CrossRef]
- Visser, P.M.; Ibelings, B.W.; Mur, L.R. Autumnal sedimentation of Microcystis spp. as result of an increase in carbohydrate ballast at reduced temperature. J. Plankton Res. 1995, 17, 919–933. [Google Scholar] [CrossRef]
- Brunberg, A.K.; Blomqvist, P. Recruitment of Microcystis (cyanophyceae) from lake sediments: The importance of littoral inocula. J. Phycol. 2003, 39, 58–63. [Google Scholar] [CrossRef]
- Roelke, D.; Buyukates, Y. Dynamics of phytoplankton succession coupled to species diversity as a system-level tool for study of microcystis population dynamics in eutrophic lakes. Limnol. Oceanogr. 2002, 47, 1109–1118. [Google Scholar] [CrossRef]
- Verspagen, J.M.H.; Visser, P.M.; Huisman, J. Aggregation with clay causes sedimentation of the buoyant cyanobacteria Microcystis spp. Aquat. Microb. Ecol. 2006, 44, 165–174. [Google Scholar] [CrossRef]
- Smayda, T.J. The suspension and sinking of phytoplakton in the sea. Oceanogr. Mar. Biol. Annu. Rev. 1970, 8, 353–414. [Google Scholar]
- Reynolds, C.S.; Wiseman, S.W. Sinking losses of phytoplankton in closed limnetic systems. J. Plankton Res. 1982, 4, 489–522. [Google Scholar] [CrossRef]
- Walsby, A.E. Gas vesicles. Microbiol. Rev. 1994, 58, 94–144. [Google Scholar]
- Fallon, R.D.; Brock, T.D. Planktonic blue-green algae: Production, sedimentation and decomposition in lake Mendota, Wisconsin. Limnol. Oceanogr. 1980, 25, 72–88. [Google Scholar] [CrossRef]
- Takamura, N.; Yasuno, M. Sedimentation of phytoplankton populations dominated by Microcystis in a shallow lake. J. Plankton Res. 1988, 10, 283–299. [Google Scholar] [CrossRef]
- Reynolds, C.S. Sinking movements of phytoplankton indicated by a simple trapping method 2. Vertical activity ranges in a stratified lake. Brit. Phycol. J. 1976, 11, 293–303. [Google Scholar] [CrossRef]
- Wilson, A.E.; Sarnelle, O.; Neilan, B.A.; Salmon, T.P.; Gehringer, M.M.; Hay, M.E. Genetic variation of the bloom-forming cyanobacterium Microcystis aeruginosa within and among lakes: Implications for harmful algal blooms. Appl. Environ. Microb. 2005, 71, 6126–6133. [Google Scholar]
- Welker, M.; Sejnohova, L.; Nemethova, D.; von Dohren, H.; Jarkovsky, J.; Marsalek, B. Seasonal shifts in chemotype composition of Microcystis sp. communities in the pelagial and the sediment of a shallow reservoir. Limnol. Oceanogr. 2007, 52, 609–619. [Google Scholar] [CrossRef]
- Visser, P.M.; Passarge, J.; Mur, L.R. Modelling vertical migration of the cyanobacterium Microcystis. Hydrobiologia 1997, 349, 99–109. [Google Scholar] [CrossRef]
- Rabouille, S.; Salencon, M.J. Functional analysis of microcystis vertical migration: A dynamic model as a prospecting tool. Ii. Influence of mixing, thermal stratification and colony diameter on biomass production. Aquat. Microb. Ecol. 2005, 39, 281–292. [Google Scholar] [CrossRef]
- Oliver, R.L. Floating and sinking in gas-vacuolate cyanobacteria. J. Phycol. 1994, 30, 161–173. [Google Scholar]
- Sigee, D.C.; Selwyn, A.; Gallois, P.; Dean, A.P. Patterns of cell death in freshwater colonial cyanobacteria during the late summer bloom. Phycologia 2007, 46, 284–292. [Google Scholar] [CrossRef]
- Latour, D.; Salencon, M.J.; Reyss, J.L.; Giraudet, H. Sedimentary imprint of Microcystis aeruginosa (cyanobacteria) blooms in Grangent reservoir (Loire, France). J. Phycol. 2007, 43, 417–425. [Google Scholar] [CrossRef]
- Wormer, L.; Cirés, S.; Quesada, A. Importance of natural sedimentation in the fate of microcystins. Chemosphere 2011, 82, 1141–1146. [Google Scholar] [CrossRef]
- De Hoyos, C.; Negro, A.; Aldasoro, J.J. Cyanobacteria distribution and abundance in the Spanish water reservoirs during thermal stratification. Limnetica 2004, 23, 119–132. [Google Scholar]
- Carrasco, D.; Moreno, E.; Sanchis, D.; Wormer, L.; Paniagua, T.; del Cueto, A.; Quesada, A. Cyanobacterial abundance and microcystin occurrence in Mediterranean water reservoirs in Central Spain: Microcystins in the Madrid area. Eur. J. Phycol. 2006, 41, 281–291. [Google Scholar] [CrossRef]
- Kardinaal, W.E.A.; Visser, P.M. Dynamics of Cyanobacterial Toxins: Sources of variability in microcystin concentrations. In Harmful Cyanobacteria; Huisman, J., Matthijs, H.C.P., Visser, P., Eds.; Springer: Berlin, Germany, 2005; pp. 41–63. [Google Scholar]
- Cook, C.M.; Vardaka, E.; Lanaras, T. Toxic cyanobacteria in Greek freshwaters, 1987–2000: Occurrence, toxicity, and impacts in the Mediterranean region. Acta Hydroch. Hydrob. 2004, 32, 107–124. [Google Scholar] [CrossRef]
- Demott, W.R.; Zhang, Q.X.; Carmichael, W.W. Effects of toxic cyanobacteria and purified toxins on the survival and feeding of a copepod and 3 species of Daphnia. Limnol. Oceanogr. 1991, 36, 1346–1357. [Google Scholar] [CrossRef]
- Komárek, J.; Komárkova, J. Review of the European Microcystis-morphospecies (Cyanoprokaryotes) from nature. Czech. Phycol. 2002, 2, 1–24. [Google Scholar]
- Van Wichelen, J.; van Gremberghe, I.; Vanormelingen, P.; Vyverman, W. The importance of morphological versus chemical defences for the bloom-forming cyanobacterium Microcystis against amoebae grazing. Aquat. Ecol. 2012, 46, 73–84. [Google Scholar] [CrossRef]
- Puente, S.; Carrasco, D.; Cirés, S.; Quesada, A. Utilización de MALDI-TOF para el estudio de quimiotipos dentro del género Microcystis en embalses españoles. In 1° Congresso Ibérico de Cianotoxinas; Porto University: Porto, Portugal, 2009. [Google Scholar]
- Jähnichen, S.; Ihle, T.; Petzoldt, T. Variability of microcystin cell quota: A small model explains dynamics and equilibria. Limnologica 2008, 38, 339–349. [Google Scholar] [CrossRef]
- Kotak, B.G.; Lam, A.K.Y.; Prepas, E.E.; Hrudey, S.E. Role of chemical and physical variables in regulating microcystin-LR concentration in phytoplankton of eutrophic lakes. Can. J. Fish. Aquat. Sci. 2000, 57, 1584–1593. [Google Scholar] [CrossRef]
- Maruyama, T.; Kato, K.; Yokoyama, A.; Tanaka, T.; Hiraishi, A.; Park, H.D. Dynamics of microcystin-degrading bacteria in mucilage of Microcystis. Microb. Ecol. 2003, 46, 279–288. [Google Scholar]
- Zilliges, Y.; Kehr, J.C.; Meissner, S.; Ishida, K.; Mikkat, S.; Hagemann, M.; Kaplan, A.; Borner, T.; Dittmann, E. The cyanobacterial hepatotoxin microcystin binds to proteins and increases the fitness of microcystis under oxidative stress conditions. PLoS One 2011, 6, e17615. [Google Scholar]
- Schöne, K.; Jahnichen, S.; Ihle, T.; Ludwig, F.; Benndorf, J. Arriving in better shape: Benthic Microcystis as inoculum for pelagic growth. Harmful Algae 2010, 9, 494–503. [Google Scholar] [CrossRef]
- Misson, B.; Sabart, M.; Amblard, C.; Latour, D. Involvement of microcystins and colony size in the benthic recruitment of the cyanobacterium Microcystis (Cyanophyceae). J. Phycol. 2011, 47, 42–51. [Google Scholar] [CrossRef]
- Utermöhl, H. Zur vervollkommung der quantitativen phytoplankton-methodik. Mitt. Int. Ver. Limnol. 1958, 9, 1–38. [Google Scholar]
- Komárek, J.; Anagnostidis, K. Modern approach to the classification system of Cyanophytes 4. Nostocales. Int. J. Phycol. Res. 1989, 56, 247–345. [Google Scholar]
- Komárek, J.; Anagnostidis, K. Cyanoprokaryota 1. Chroococcales; Gustav-Fisher: Stuttgart, Germany, 1999; p. 548. [Google Scholar]
- Komárek, J.; Anagnostidis, K. Cyanoprokaryota 2: Oscillatoriales; Elsevier/Spektrum: Heidelberg, Germany, 2005; p. 759. [Google Scholar]
Supplementary Files
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cirés, S.; Wörmer, L.; Carrasco, D.; Quesada, A. Sedimentation Patterns of Toxin-Producing Microcystis Morphospecies in Freshwater Reservoirs. Toxins 2013, 5, 939-957. https://doi.org/10.3390/toxins5050939
Cirés S, Wörmer L, Carrasco D, Quesada A. Sedimentation Patterns of Toxin-Producing Microcystis Morphospecies in Freshwater Reservoirs. Toxins. 2013; 5(5):939-957. https://doi.org/10.3390/toxins5050939
Chicago/Turabian StyleCirés, Samuel, Lars Wörmer, David Carrasco, and Antonio Quesada. 2013. "Sedimentation Patterns of Toxin-Producing Microcystis Morphospecies in Freshwater Reservoirs" Toxins 5, no. 5: 939-957. https://doi.org/10.3390/toxins5050939





