Dynamic Energy Balance: An Integrated Framework for Discussing Diet and Physical Activity in Obesity Prevention—Is it More than Eating Less and Exercising More?
Abstract
:1. Introduction
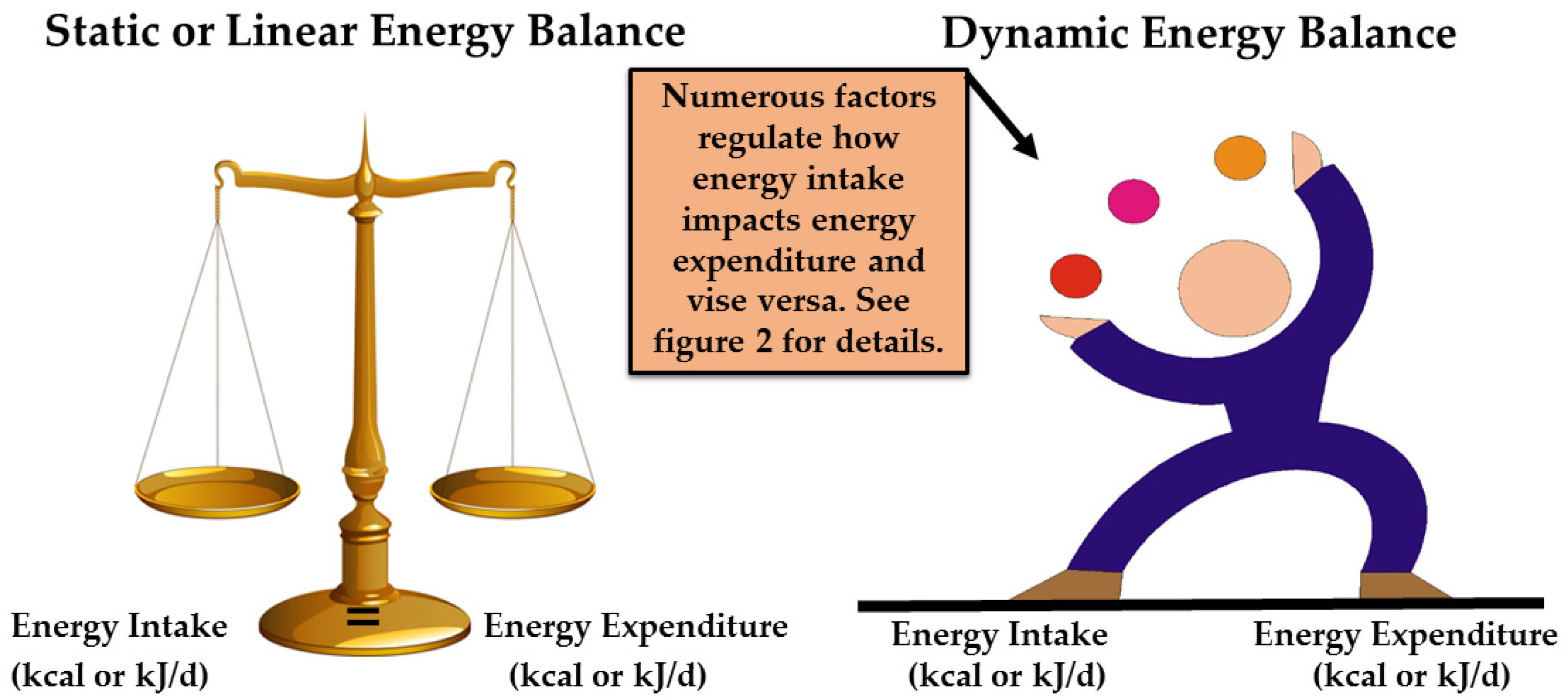
2. Dynamic vs. Static Energy Balance
3. Role of Physical Activity in Body Weight Management
3.1. Effect of Physical Activity Quantity on Body Weight and Size
3.2. Effect of Health-Related Fitness Level on Metabolic Rate
3.3. Effect of Physical Activity on Muscle and Bone Mass
3.4. Effect of Physical Activity and Energy Restriction on Metabolic Rate
3.5. Effect of Weight Loss or Gain on Energy Expenditure
4. Role of Physical Activity in Appetite Regulation
4.1. Effect of Type and Intensity of Physical Activity on Appetite and Energy Intake
4.2. Effect of Environmental Temperature on Appetite and Energy Intake
4.3. Factors that Drive Hunger and Desire to Eat after Exercise
4.4. Differences Between Men and Women
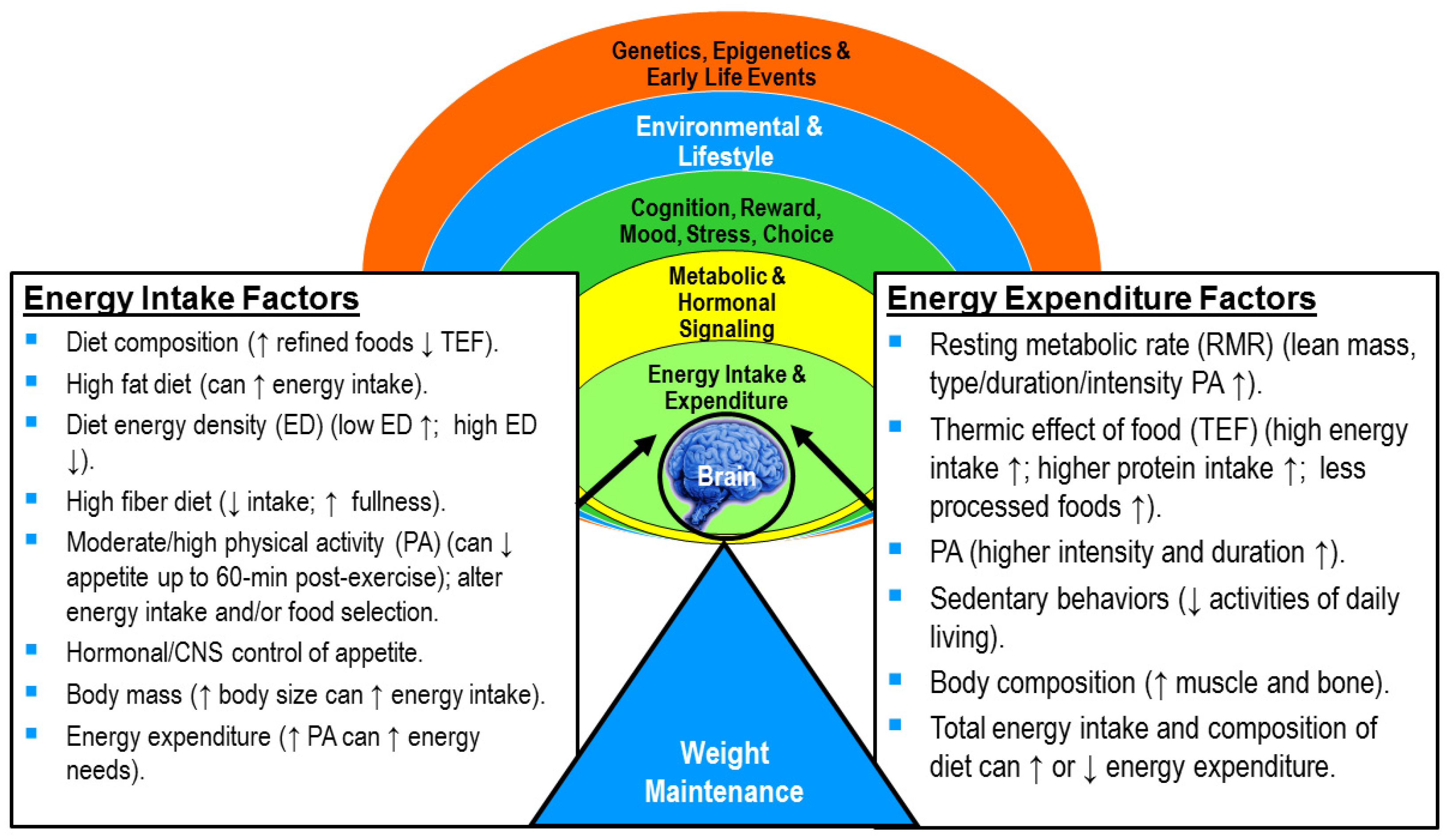
5. Energy Flux: Putting It All Together
- Maintains overall higher energy expenditure by maintaining muscle mass, thermic effect of food, and a higher RMR (e.g., in high energy flux), in addition to the energy expended in PA. A person in high energy flux will expend more energy in PA and need to eat more food to cover their energy needs.
- Heightens sensitivity to appetite control through its impact on appetite-regulatory hormones and food preferences. Thus, the desire to overconsume food is dampened and the total energy intake modified.
- Allows for more appropriate energy intake or volume of food consumed, thus, reducing the probability of overeating. Sedentary individuals (e.g., in low energy flux) can have daily energy needs that are so low that it is easy to consume more food (e.g., kcals) than needed in our current obesogenic environment.
6. Integrating Dynamic Energy Balance into Obesity Prevention Programs
- Diet and PA are both important for effective obesity prevention. Action: Incorporate both into obesity prevention programs.
- Physical activity improves one’s ability to attain and maintain a desirable body weight and body composition (i.e., lower body fat mass). Action: Incorporate the key points in section 3.0 to obesity prevention programing. Provide examples to help illustrate how PA works to help manage weight and improve body composition beyond just energy expenditure.
- Physical activity promotes appetite regulation through the body’s appetite-regulatory hormones. Action: Explain effects of type and intensity of PA and environmental temperature on appetite and energy intake. Recommend consuming food slowly to allow appetite hormones to positively impact satiety and decrease hunger, consuming foods higher in fiber (whole fruit, vegetables, and grains) to slow eating and increase a sense of fullness. Encourage PA and foods that positively affect appetite and amount of energy intake. Discourage food rewards after exercising because the energy expended can be easily “eaten back”.
- Physical activity promotes higher energy flux, which may make it easier to match energy needs with expenditure. Action: Explain how high energy flux is key to successful weight maintenance, preventing excess weight gain, or maintaining weight loss. Demonstrate how to design a personal diet and PA program that helps an individual attain a higher level of energy flux. An effective way to prevent obesity is to maintain a higher level of PA.
- Dynamic energy balance tools can help nutrition educators understand how changes in energy intake and expenditure affect body weight and composition over time. Action: Provide examples from these tools to help clients better understand the dynamic energy balance concept.
7. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sparling, P.B.; Franklin, B.A.; Hill, J.O. Energy balance: The key to a unified message on diet and physical activity. J. Cardiopulm. Rehabil. Prev. 2013, 33, 12–15. [Google Scholar] [CrossRef] [PubMed]
- Hall, K.D.; Heymsfield, S.B.; Kemnitz, J.W.; Klein, S.; Schoeller, D.A.; Speakman, J.R. Energy balance and its components: Implications for body weight regulation. Am. J. Clin. Nutr. 2012, 95, 989–994. [Google Scholar] [CrossRef] [PubMed]
- Shook, R.P.; Hand, G.A.; Blair, S.N. Top 10 research questions related to energy balance. Res. Q. Exerc. Sport 2014, 85, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Pagoto, S.L.; Appelhans, B.M. A call for an end to the diet debates. JAMA 2013, 310, 687–688. [Google Scholar] [CrossRef] [PubMed]
- Flatt, J.P. Issues and misconceptions about obesity. Obesity 2011, 19, 676–686. [Google Scholar] [CrossRef] [PubMed]
- Casazza, K.; Fontaine, K.R.; Astrup, A.; Birch, L.L.; Brown, A.W.; Bohan Brown, M.M.; Durant, N.; Dutton, G.; Foster, E.M.; Heymsfield, S.B.; et al. Myths, presumptions, and facts about obesity. New Engl. J. Med. 2013, 368, 446–454. [Google Scholar] [CrossRef] [PubMed]
- Pate, R.R.; Taverno Ross, S.E.; Liese, A.D.; Dowda, M. Associations among physical activity, diet quality, and weight status in us adults. Med. Sci. Sports Exerc. 2015, 47, 743–750. [Google Scholar] [CrossRef] [PubMed]
- Malhotra, A.; Noakes, T.; Phinney, S. It is time to bust the myth of physical inactivity and obesity: You cannot outrun a bad diet. Br. J. Sports Med. 2015, 49, 967–968. [Google Scholar] [CrossRef] [PubMed]
- Blair, S.N.; Hand, G.A.; Hill, J.O. Energy balance: A crucial issue for exercise and sports medicine. Br. J. Sports Med. 2015. [Google Scholar] [CrossRef] [PubMed]
- Galgani, J.; Ravussin, E. Energy metabolism, fuel selection and body weight regulation. Int. J. Obes. 2008, 32, S109–S119. [Google Scholar] [CrossRef] [PubMed]
- Acheson, K.J.; Blondel-Lubrano, A.; Oguey-Araymon, S.; Beaumont, M.; Emady-Azar, S.; Ammon-Zufferey, C.; Monnard, I.; Pinaud, S.; Nielsen-Moennoz, C.; Bovetto, L. Protein choices targeting thermogenesis and metabolism. Am. J. Clin. Nutr. 2011, 93, 525–534. [Google Scholar] [CrossRef] [PubMed]
- Rolls, B.J.; Roe, L.S.; Meengs, J.S. Reductions in portion size and energy density of foods are additive and lead to sustained decreases in energy intake. Am. J. Clin. Nutr. 2006, 83, 11–17. [Google Scholar] [PubMed]
- Ledikwe, J.H.; Rolls, B.J.; Smiciklas-Wright, H.; Mitchell, D.C.; Ard, J.D.; Champagne, C.; Karanja, N.; Lin, P.-H.; Stevens, V.J.; Appel, L.J. Reductions in dietary energy density are associated with weight loss in overweight and obese participants in the premier trial. Am. J. Clin. Nutr. 2007, 85, 1212–1221. [Google Scholar] [PubMed]
- Madjd, A.; Taylor, M.A.; Delavari, A.; Malekzadeh, R.; Macdonald, I.A.; Farshchi, H.R. Beneficial effects of replacing diet beverages with water on type 2 diabetic obese women following a hypo-energetic diet: A randomized, 24-week clinical trial. Diabetes Obes. Metab. 2016, 19, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Hawley, J.A.; Burke, L.M.; Phillips, S.M.; Spriet, L.L. Nutritional modulation of training-induced skeletal muscle adaptations. J. Appl. Physiol. 2011, 110, 834–845. [Google Scholar] [CrossRef] [PubMed]
- Hawley, J.A.; Burke, L.M. Carbohydrate availability and training adaptation: Effects on cell metabolism. Exerc. Sport Sci. Rev. 2010, 38, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Manore, M.M.; Meyer, N.L.; Thompson, J. Sport Nutrition for Health and Performance, 2nd ed.; Human Kinetics: Champaign, IL, USA, 2009; pp. 25–108. [Google Scholar]
- Manore, M.M. Rethinking energy balance: Facts you need to know about weight loss and management. ACSM’s Health Fit. J. 2015, 19, 9–15. [Google Scholar]
- Manore, M.M. Weight management for athletes and active individuals: A brief review. Sports Med. 2015, 45, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Hand, G.A.; Shook, R.P.; Hill, J.O.; Giacobbi, P.R.; Blair, S.N. Energy flux: Staying in energy balance at a high level is necessary to prevent weight gain for most people. Expert Rev. Endocrinol. Metab. 2015, 10, 599–605. [Google Scholar] [CrossRef]
- United States Department of Health and Human Services (DHHS). 2008 Physical Activity Guidelines for Americans; Office of Disease Prevention and Health Promotion, U.S. DHHS: Washington, DC, USA, 2008; pp. 1–14. Available online: http://health.gov/paguidelines/guidelines2008 (accessed on 18 August 2017).
- United States Department of Health and Human Services. Bone Health and Osteoporosis: A Report of the Surgeon General; Office of the Surgeon General: Rockville, MD, USA, 2004; p. 87.
- Ackland, T.R.; Lohman, T.G.; Sundgot-Borgen, J.; Maughan, R.J.; Meyer, N.L.; Stewart, A.D.; Müller, W. Current status of body composition assessment in sport. Sports Med. 2012, 42, 227–249. [Google Scholar] [CrossRef] [PubMed]
- Stensel, D. Exercise, appetite and appetite-regulating hormones: Implications for food intake and weight control. Ann. Nutr. Metab. 2010, 2, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Hagobian, T.A.; Braun, B. Physical activity and hormonal regulation of appetite: Sex differences and weight control. Exerc. Sport Sci. Rev. 2010, 38, 25–30. [Google Scholar] [CrossRef] [PubMed]
- King, J.A.; Garnham, J.O.; Jackson, A.P.; Kelly, B.M.; Xenophontos, S.; Nimmo, M.A. Appetite-regulatory hormone responses on the day following a prolonged bout of moderate-intensity exercise. Physiol. Behave. 2015, 141, 23–31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thackray, A.E.; Deighton, K.; King, J.A.; Stensel, D.J. Exercise, appetite and weight control: Are there differences between men and women? Nutrients 2016, 8, 583. [Google Scholar] [CrossRef] [PubMed]
- Hill, J.O.; Wyatt, H.R.; Peters, J.C. Energy balance and obesity. Circulation 2012, 126, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Melby, C.L.; Paris, H.L.; Foright, R.M.; Peth, J. Attenuating the biologic drive for weight regain following weight loss: Must what goes down always go back up? Nutrients 2017, 9, 468. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Vélez, R.; Correa-Bautista, J.E.; Lobelo, F.; Izquierdo, M.; Alonso-Martínez, A.; Rodríguez-Rodríguez, F.; Cristi-Montero, C. High muscular fitness has a powerful protective cardiometabolic effect in adults: Influence of weight status. BMC Public Health 2016, 16, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Kohrt, W.M.; Bloomfield, S.A.; Little, K.D.; Nelson, M.E.; Yingling, V.R. American college of sports medicine position stand: Physical activity and bone health. Med. Sci. Sports Exerc. 2004, 36, 1985–1996. [Google Scholar] [CrossRef] [PubMed]
- Hupin, D.; Roche, F.; Gremeaux, V.; Chatard, J.-C.; Oriol, M.; Gaspoz, J.-M.; Barthélémy, J.-C.; Edouard, P. Even a low-dose of moderate-to-vigorous physical activity reduces mortality by 22% in adults aged ≥ 60 years: A systematic review and meta-analysis. Br. J. Sports Med. 2015, 49, 1262–1267. [Google Scholar] [CrossRef] [PubMed]
- Wishnofsky, M. Caloric equivalents of gained or lost weight. Am. J. Clin. Nutr. 1958, 6, 542–546. [Google Scholar] [CrossRef] [PubMed]
- Rosenbaum, M.; Leibel, R.L. Models of energy homeostasis in response to maintenance of reduced body weight. Obesity 2016, 24, 1620–1629. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.M.; Gonzalez, M.C.; Pereira, A.Z.; Redman, L.M.; Heymsfield, S.B. Time to correctly predict the amount of weight loss with dieting. J. the Acad. Nutr. Diet. 2014, 114, 857–861. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.M.; Martin, C.K.; Lettieri, S.; Bredlau, C.; Kaiser, K.; Church, T.; Bouchard, C.; Heymsfield, S.B. Can a weight loss of one pound a week be achieved with a 3500-kcal deficit? Commentary on a commonly accepted rule. Int. J. Obes. 2013, 37, 1611–1613. [Google Scholar] [CrossRef] [PubMed]
- Manore, M.M.; Brown, K.; Houtkooper, L.; Jakicic, J.; Peters, J.C.; Edge, M.S.; Steiber, A.; Going, S.; Gable, L.G.; Krautheim, A.M. Energy balance at a crossroads: Translating the science into action. Med. Sci. Sports Exerc. 2014, 46, 1466–1473. [Google Scholar] [CrossRef] [PubMed]
- Hall, K.D.; Sacks, G.; Chandramohan, D.; Chow, C.C.; Wang, Y.C.; Gortmaker, S.L.; Swinburn, B.A. Quantification of the effect of energy imbalance on bodyweight. Lancet 2011, 378, 826–837. [Google Scholar] [CrossRef]
- Hall, K.D. Modeling metabolic adaptations and energy regulation in humans. Ann. Rev. Nutr. 2012, 32, 35–54. [Google Scholar] [CrossRef] [PubMed]
- Swinburn, B.; Ravussin, E. Energy balance or fat balance? Am. J. Clin. Nutr. 1993, 57, 770S–771S. [Google Scholar]
- Westerterp-Plantenga, M.S.; Nieuwenhuizen, A.; Tomé, D.; Soenen, S.; Westerterp, K.R. Dietary protein, weight loss, and weight maintenance. Ann. Rev. Nutr. 2009, 29, 21–41. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.M.; Ciesla, A.; Levine, J.A.; Stevens, J.G.; Martin, C.K. A mathematical model of weight change with adaptation. Math. Biosci. Eng. 2009, 6, 873–887. [Google Scholar] [CrossRef] [PubMed]
- National Institutes of Diabetes and Digestive and Kidney Diseases (NIDDK) Body Weight Planner. Available online: https://www.niddk.nih.gov/health-information/weight-management/body-weight-planner (accessed on 18 August 2017).
- Pennington Biomedical Research Center (PBRC). Weight Loss Predictor. Available online: http://www.pbrc.edu/research-and-faculty/calculators/weight-loss-predictor/ (accessed on 18 August 2017).
- Webb, D. Farewell to the 3,500-calorie rule. Today’s Diet. 2014, 26, 36–39. [Google Scholar]
- Hughes, V.A.; Frontera, W.R.; Roubenoff, R.; Evans, W.J.; Singh, M.A.F. Longitudinal changes in body composition in older men and women: Role of body weight change and physical activity. Am. J. Clin. Nutr. 2002, 76, 473–481. [Google Scholar] [PubMed]
- Ekelund, U.; Besson, H.; Luan, J.a.; May, A.M.; Sharp, S.J.; Brage, S.; Travier, N.; Agudo, A.; Slimani, N.; Rinaldi, S.; et al. Physical activity and gain in abdominal adiposity and body weight: Prospective cohort study in 288,498 men and women. Am. J. Clin. Nutr. 2011, 93, 826–835. [Google Scholar] [CrossRef] [PubMed]
- Shook, R.P.; Hand, G.A.; Drenowatz, C.; Hebert, J.R.; Paluch, A.E.; Blundell, J.E.; Hill, J.O.; Katzmarzyk, P.T.; Church, T.S.; Blair, S.N. Low levels of physical activity are associated with dysregulation of energy intake and fat mass gain over 1 year. Am. J. Clin. Nutr. 2015, 102, 1332–1338. [Google Scholar] [CrossRef] [PubMed]
- Tudor-Locke, C.; Schuna, J.M., Jr.; Han, H.; Aguiar, E.J.; Green, M.A.; Busa, M.A.; Larrivee, S.; Johnson, W.D. Step-based physical activity metrics and cardiometabolic risk: Nhanes 2005–2006. Med. Sci. Sports Exerc. 2017, 49, 283–291. [Google Scholar] [CrossRef] [PubMed]
- Chung, A.E.; Skinner, A.C.; Steiner, M.J.; Perrin, E.M. Physical activity and BMI in a nationally representative sample of children and adolescents. Clin. Pediatr. 2012, 51, 122–129. [Google Scholar] [CrossRef] [PubMed]
- Carson, V.; Staiano, A.E.; Katzmarzyk, P.T. Physical activity, screen time, and sitting among US adolescents. Pediatr. Exerc. Sci. 2015, 27, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Carson, V.; Ridgers, N.D.; Howard, B.J.; Winkler, E.A.; Healy, G.N.; Owen, N.; Dunstan, D.W.; Salmon, J. Light-intensity physical activity and cardiometabolic biomarkers in us adolescents. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Shook, R.P.; Hand, G.A.; Paluch, A.E.; Wang, X.; Moran, R.; Hébert, J.R.; Lavie, C.J.; Blair, S.N. Moderate cardiorespiratory fitness is positively associated with resting metabolic rate in young adults. Mayo Clin. Proc. 2014, 89, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Romijn, J.; Coyle, E.; Sidossis, L.; Gastaldelli, A.; Horowitz, J.; Endert, E.; Wolfe, R. Regulation of endogenous fat and carbohydrate metabolism in relation to exercise intensity and duration. Am. J. Physiol.-Endocrinol. Metab. 1993, 265, E380–E391. [Google Scholar]
- Srikanthan, P.; Karlamangla, A.S. Relative muscle mass is inversely associated with insulin resistance and prediabetes. Findings from the third national health and nutrition examination survey. J. Clin. Endocrinol. Metab. 2011, 96, 2898–2903. [Google Scholar] [CrossRef] [PubMed]
- Redman, L.M.; Heilbronn, L.K.; Martin, C.K.; de Jonge, L.; Williamson, D.A.; Delany, J.P.; Ravussin, E. Metabolic and behavioral compensations in response to caloric restriction: Implications for the maintenance of weight loss. PLoS ONE 2009, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ryan, A.S.; Pratley, R.E.; Elahi, D.; Goldberg, A.P. Resistive training increases fat-free mass and maintains rmr despite weight loss in postmenopausal women. J. Appl. Physiol. 1995, 79, 818–823. [Google Scholar] [PubMed]
- St-Onge, M.-P.; Gallagher, D. Body composition changes with aging: The cause or the result of alterations in metabolic rate and macronutrient oxidation? Nutrition 2010, 26, 152–155. [Google Scholar] [CrossRef] [PubMed]
- National Intitutes of Health Osteoporosis and Related Bone Diseases National Resource Center. Available online: https://www.niams.nih.gov/Health_Info/Bone/default.asp (accessed on 18 August 2017).
- Heymsfield, S.B.; Gonzalez, M.C.; Shen, W.; Redman, L.; Thomas, D. Weight loss composition is one-fourth fat-free mass: A critical review and critique of this widely cited rule. Obes. Rev. 2014, 15, 310–321. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, J.E.; Blair, S.N.; Jakicic, J.M.; Manore, M.M.; Rankin, J.W.; Smith, B.K. Appropriate physical activity intervention strategies for weight loss and prevention of weight regain for adults. Med. Sci. Sports Exerc. 2009, 41, 459–471. [Google Scholar] [CrossRef] [PubMed]
- Rosenbaum, M.; Vandenborne, K.; Goldsmith, R.; Simoneau, J.A.; Heymsfield, S.; Joanisse, D.R.; Hirsch, J.; Murphy, E.; Matthews, D.; Segal, K.R.; et al. Effects of experimental weight perturbation on skeletal muscle work efficiency in human subjects. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2003, 285, R183–R192. [Google Scholar] [CrossRef] [PubMed]
- Melanson, E.L.; Keadle, S.K.; Donnelly, J.E.; Braun, B.; King, N.A. Resistance to exercise-induced weight loss: Compensatory behavioral adaptations. Med. Sci. Sports Exerc. 2013, 45, 1600–1609. [Google Scholar] [CrossRef] [PubMed]
- King, N.A.; Hopkins, M.; Caudwell, P.; Stubbs, R.J.; Blundell, J.E. Individual variability following 12 weeks of supervised exercise: Identification and characterization of compensation for exercise-induced weight loss. Int. J. Obes. 2008, 32, 177–184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ueda, S.Y.; Yoshikawa, T.; Katsura, Y.; Usui, T.; Fujimoto, S. Comparable effects of moderate intensity exercise on changes in anorectic gut hormone levels and energy intake to high intensity exercise. J. Endocrinol. 2009, 203, 357–364. [Google Scholar] [CrossRef] [PubMed]
- Imbeault, P.; Saint-Pierre, S.; Almeras, N.; Tremblay, A. Acute effects of exercise on energy intake and feeding behaviour. Br. J. Nutr. 1997, 77, 511–521. [Google Scholar] [CrossRef] [PubMed]
- King, N.A.; Burley, V.J.; Blundell, J.E. Exercise-induced suppression of appetite: Effects on food intake and implications for energy balance. Eur. J. Clin. Nutr. 1994, 48, 715–724. [Google Scholar] [PubMed]
- Thompson, D.A.; Wolfe, L.A.; Eikelboom, R. Acute effects of exercise intensity on appetite in young men. Med. Sci. Sports Exerc. 1988, 20, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Reger, W.E.; Allison, T.G. Exercise, Postexercise, Metabolic Rate, and Appetite; Human Kinetics Publishers, INC: Champaign, IL, USA, 1984; pp. 115–123. [Google Scholar]
- Deighton, K.; Karra, E.; Batterham, R.L.; Stensel, D.J. Appetite, energy intake, and pyy3-36 responses to energy-matched continuous exercise and submaximal high-intensity exercise. Appl. Physiol. Nutr. Metab. 2013, 38, 947–952. [Google Scholar] [CrossRef] [PubMed]
- Larson-Meyer, D.E.; Palm, S.; Bansal, A.; Austin, K.J.; Hart, A.M.; Alexander, B.M. Influence of running and walking on hormonal regulators of appetite in women. J. Obes. 2012. [Google Scholar] [CrossRef] [PubMed]
- Kawano, H.; Mineta, M.; Asaka, M.; Miyashita, M.; Numao, S.; Gando, Y.; Ando, T.; Sakamoto, S.; Higuchi, M. Effects of different modes of exercise on appetite and appetite-regulating hormones. Appetite 2013, 66, 26–33. [Google Scholar] [CrossRef] [PubMed]
- King, J.A.; Wasse, L.K.; Stensel, D.J. The acute effects of swimming on appetite, food intake, and plasma acylated ghrelin. J. Obes. 2011. [Google Scholar] [CrossRef] [PubMed]
- Verger, P.; Lanteaume, M.T.; Louis-Sylvestre, J. Human intake and choice of foods at intervals after exercise. Appetite 1992, 18, 93–99. [Google Scholar] [CrossRef]
- Broom, D.R.; Batterham, R.L.; King, J.A.; Stensel, D.J. Influence of resistance and aerobic exercise on hunger, circulating levels of acylated ghrelin, and peptide yy in healthy males. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2009, 296, R29–R35. [Google Scholar] [CrossRef] [PubMed]
- Crabtree, D.R.; Blannin, A.K. Effects of exercise in the cold on ghrelin, pyy, and food intake in overweight adults. Med. Sci. Sports Exerc. 2015, 47, 49–57. [Google Scholar] [CrossRef] [PubMed]
- White, L.J.; Dressendorfer, R.H.; Holland, E.; McCoy, S.C.; Ferguson, M.A. Increased caloric intake soon after exercise in cold water. Int. J. Sport Nutr. Exerc. Metab. 2005, 15, 38–47. [Google Scholar] [CrossRef] [PubMed]
- Halse, R.E.; Wallman, K.E.; Guelfi, K.J. Postexercise water immersion increases short-term food intake in trained men. Med. Sci. Sports Exerc. 2011, 43, 632–638. [Google Scholar] [CrossRef] [PubMed]
- Howe, S.M.; Hand, T.M.; Manore, M.M. Exercise-trained men and women: Role of exercise and diet on appetite and energy intake. Nutrients 2014, 6, 4935–4960. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, M.; Blundell, J.E.; King, N.A. Individual variability in compensatory eating following acute exercise in overweight and obese women. Br. J. Sports Med. 2013, 48, 1472–1476. [Google Scholar] [CrossRef] [PubMed]
- Weinheimer, E.M.; Sands, L.P.; Campbell, W.W. A systematic review of the separate and combined effects of energy restriction and exercise on fat-free mass in middle-aged and older adults: Implications for sarcopenic obesity. Nutr. Rev. 2010, 68, 375–388. [Google Scholar] [CrossRef] [PubMed]
- Raynor, H.A.; Champagne, C.M. Position of the academy of nutrition and dietetics: Interventions for the treatment of overweight and obesity in adults. J. Acad. Nutr. Diet. 2016, 116, 129–147. [Google Scholar] [CrossRef] [PubMed]
| Terms | Definition |
|---|---|
| Static (linear) energy balance a | Assumes that a change in one side of the energy balance equation (e.g., energy intake) does not change or influence the other side of the equation (e.g., energy expenditure). |
| Dynamic (non-linear) energy balance a | Assumes that numerous biological and behavior factors regulate and influence both sides of the energy balance equation. Thus, a change in factors on one side of the equation (e.g., energy intake) can and does influence factors on the other side of the equation (e.g., energy expenditure). |
| Dietary energy density | The energy content of food by weight (kcal or kJ/per gram). |
| Thermic effect of food | Energy required digesting, metabolizing, or storing energy as fat or glycogen. |
| Energy flux b | The rate of energy conversion after absorption from food into body tissues for use in metabolism or its conversion into energy stores. |
| Physical activity c | Bodily movement that enhances health such as walking, dancing, biking, and yoga. |
| Exercise c | Physical activity that is planned, structured, repetitive, and performed with the goal of improving health or fitness. |
| Health-related Fitness c | Cardiovascular or muscular fitness focused on the reduction of chronic disease risk. |
| Moderate-Vigorous PA c | Moderate PA is an intensity of exercise similar to walking at 3.0 miles per hour, while vigorous PA is an intensity of exercise equivalent to running a 10-minute mile. |
| Weight-bearing PA d | Physical activity such as walking, jogging, running, hiking, dancing, stair climbing, lifting weights, jumping, playing tennis, basketball, or soccer. |
| Body Composition e | The percentage or amount of fat and fat free (mineral, protein and water) in bone, muscle, and other tissues in the body. |
| Compensatory Behavior f | Partial or completely compensation, through diet, for the energy expended in exercise (e.g., eating back energy expended during exercise by increasing energy intake), thereby negating body weight loss due to increased PA. Decreasing PA could also be a compensatory behavior. |
| Program Name/Resource | Web Link |
|---|---|
| President’s Council on Fitness, Sports and Nutrition | https://www.hhs.gov/fitness/index.html |
| 2008 PA Guidelines for Americans | https://health.gov/paguidelines/ |
| 2015 Dietary Guidelines for Americans | https://health.gov/dietaryguidelines/2015/guidelines/ |
| USDA SuperTracker | https://www.supertracker.usda.gov/ |
| USDA SuperTracker My Plate | https://www.choosemyplate.gov/tools-supertracker |
| Centers for Disease Control & Prevention—Strategies to Prevent Obesity | https://www.cdc.gov/obesity/strategies/index.html |
| SNAP-Education Toolkit. Obesity Prevention Interventions & Evaluation Framework | https://snapedtoolkit.org/ |
| United Kingdom (UK) Eatwell Guide and Public Health England Government Dietary Recommendations | https://www.gov.uk/government/publications/the-eatwell-guide |
| UK Obesity Prevention | https://www.nice.org.uk/guidance/cg43 |
| Clinical Practice Guidelines for the Management of Overweight and Obesity Adults, Adolescents and Children in Australia (2013) | https://www.nhmrc.gov.au/guidelines-publications/n57 |
| Canadian Task Force on Prevention Heath Care Guidelines. Obesity in Children | http://canadiantaskforce.ca/guidelines/published-guidelines/obesity-in-children/ |
| Canadian Task Force on Prevention Heath Care Guidelines. Obesity in Adults | http://canadiantaskforce.ca/guidelines/published-guidelines/obesity-in-adults/ |
| World Health Organization. Obesity: Prevention and managing the global epidemic | www.who.int/nutrition/publications/obesity/WHO_TRS_894/en/ |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manore, M.M.; Larson-Meyer, D.E.; Lindsay, A.R.; Hongu, N.; Houtkooper, L. Dynamic Energy Balance: An Integrated Framework for Discussing Diet and Physical Activity in Obesity Prevention—Is it More than Eating Less and Exercising More? Nutrients 2017, 9, 905. https://doi.org/10.3390/nu9080905
Manore MM, Larson-Meyer DE, Lindsay AR, Hongu N, Houtkooper L. Dynamic Energy Balance: An Integrated Framework for Discussing Diet and Physical Activity in Obesity Prevention—Is it More than Eating Less and Exercising More? Nutrients. 2017; 9(8):905. https://doi.org/10.3390/nu9080905
Chicago/Turabian StyleManore, Melinda M., D. Enette Larson-Meyer, Anne R. Lindsay, Nobuko Hongu, and Linda Houtkooper. 2017. "Dynamic Energy Balance: An Integrated Framework for Discussing Diet and Physical Activity in Obesity Prevention—Is it More than Eating Less and Exercising More?" Nutrients 9, no. 8: 905. https://doi.org/10.3390/nu9080905





