Dietary Zinc Deficiency Affects Blood Linoleic Acid: Dihomo-γ-linolenic Acid (LA:DGLA) Ratio; a Sensitive Physiological Marker of Zinc Status in Vivo (Gallus gallus)
Abstract
:1. Introduction
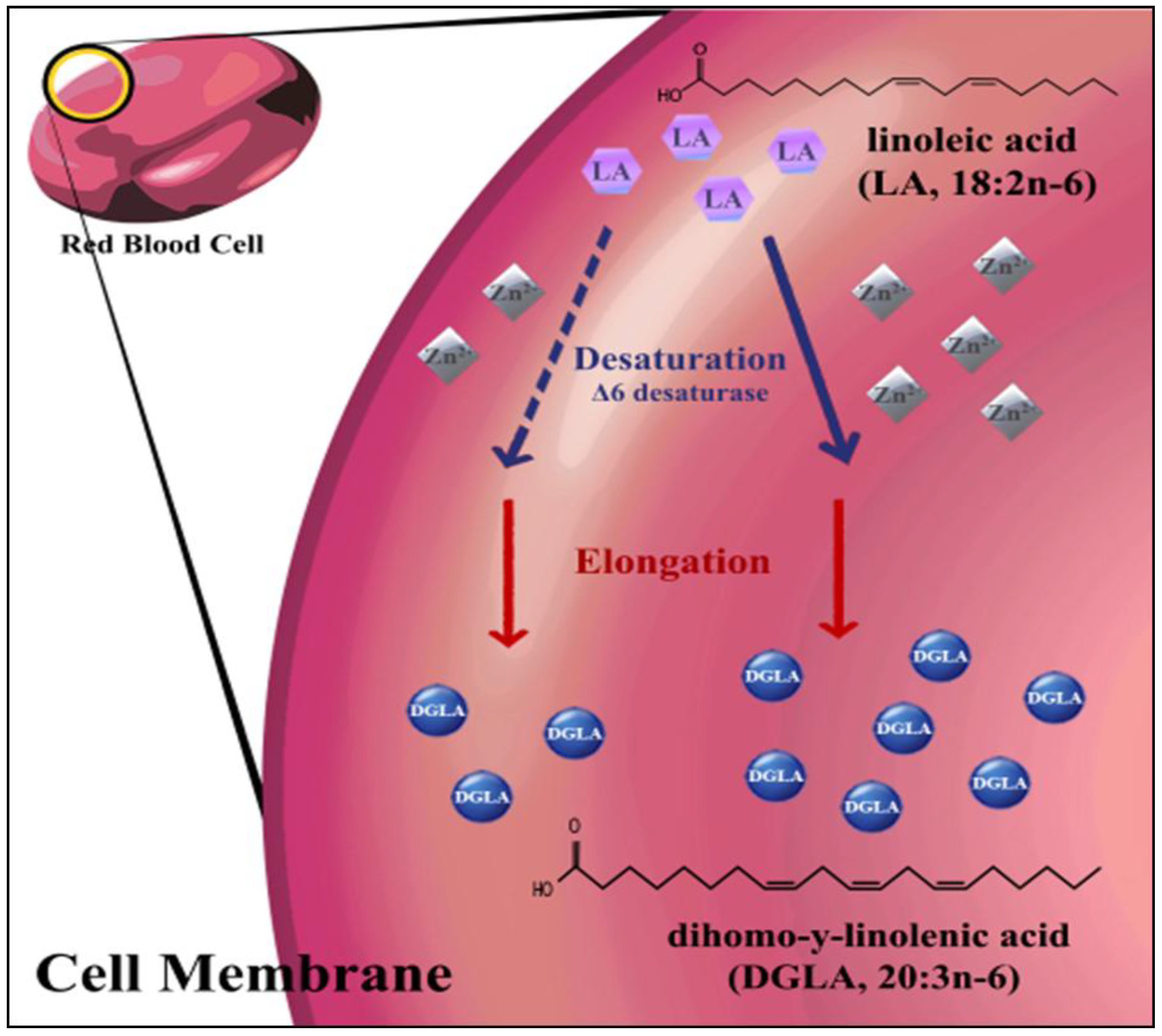
2. Experimental Section
2.1. Animals, Diets and Study Design
| Ingredient | Zn(+) Control Diet | Zn(−) Diet |
|---|---|---|
| g/kg (by Formulation) | ||
| Egg whites | 200 | 200 |
| dl-Methionine | 3 | 3 |
| Cornstarch | 318.2 | 318.2 |
| Dyetrose | 105 | 105 |
| Dextrose | 200.0 | 190.8 |
| Cellulose | 50 | 50 |
| Corn oil | 50 | 50 |
| Salt mix (no Zn) | 60 | 60 |
| Vitamin mix | 10 | 10 |
| Biotin (1 mg/g) | 1.8 | 1.8 |
| Choline bitartrate | 2 | 2 |
| Zinc carbonate (5 mg/g) | 9.2 | − |
| Total (g) | 1000 | 1000 |
| Concentrations of Selected Components (means ± SEM, n = 5) | ||
| Zinc concentration (ppm) ** | 42.29 a ± 0.25 | 2.55 b ± 0.02 |
| Iron concentration (ppm) | 98.75 ± 2.04 | 102.19 ± 5.21 |
| Phytic acid *** | <dL | <dL |
| Fatty Acid | Zn(+) Control Diet (% w/w) | Zn(−) Diet (% w/w) |
|---|---|---|
| 16:0 | 11.20 ± 0.11 | 11.57 ± 0.34 |
| 16:1 | 0.17 ± 0.01 | 0.18 ± 0.02 |
| 17:0 | 0.10 ± 0.01 | 0.11 ± 0.04 |
| 17:1 | 0.08 ± 0.03 | 0.07 ± 0.01 |
| 18:0 | 2.26 ± 0.04 | 2.31 ± 0.05 |
| 18:1 n-9 | 28.31 ± 0.15 | 28.42 ± 0.16 |
| 18:2 n-6 | 55.55 ± 0.22 | 55.19 ± 0.18 |
| 18:3 n-3 | 1.25 ± 0.03 | 1.28 ± 0.02 |
| 20:3 n-6 | 0.49 ± 0.06 | 0.43 ± 0.01 |
| 20:1 n-9 | 0.41 ± 0.01 | 0.42 ± 0.02 |
2.2. Determination of Serum, Nail and Feather Zinc Content
2.3. Isolation of Total RNA
2.4. Gene Expression Analysis
2.5. Fatty Acid Analysis of Erythrocytes and Experimental Diets
| Analyte | Organ | Forward Primer (5′→3′) (Nucleotide Position) | Reverse Primer (5′→3′) (Nucleotide Position) | Length | GI Identifier |
|---|---|---|---|---|---|
| (Base Pairs) | |||||
| ZnT1 | Intestine | CCTCCAGACAACCTTTGGTG (64–83) | TACTGATCTGCAAACCTTGCCA (133–112) | 69 | 54109718 |
| ZnT5 | Intestine | TCGTGGAGGCTGTCATTCAC (1657–1676) | TGCAGATCTTTCTCCTGTTCGT (2016–1995) | 359 | 56555150 |
| ZnT7 | Intestine | GGCGTCTGGAGTAACAGCTT (166–185) | GTGAATGCCCATGACCTCCA (502–483) | 336 | 56555152 |
| ZIP6 | Intestine | TTGTGGAATCATCCCAGGGC (549–568) | GCTCATTCGCATCTCTCCGA (929–909) | 380 | 66735072 |
| ZIP9 | Intestine | TTATTCCCCTGGCCGTGAAC (68–87) | CCAATGCGAAGACCAGCAAG (643–624) | 575 | 237874618 |
| TNF-α | Liver | CATTTGGAAGCAGCGTTCGG (48–67) | GACAGGGTAGGGGTGAGGAT (249–230) | 202 | 53854909 |
| IL-1β | Liver | CCTCCAGCCAGAAAGTGAGG (431–450) | TTGTAGCCCTTGATGCCCAG (539–520) | 109 | 88702685 |
| IL-6 | Liver | AACAACCTCAACCTGCCCAA (338–357) | AGGTCTGAAAGGCGAACAGG (449–430) | 112 | 302315692 |
| NF-κB | Liver | GGATGGTCTGTTCCTGAAGA (1682–1702) | ACCTCTGCCTGCTTTGTGAT (1981–1961) | 300 | 2130627 |
| AP | Intestine | GAATGAGGGCTTTGCCTCCT (1245–1264) | GAAGTTGCTGTTGGTGGCTG (1854–1835) | 610 | 45382360 |
| SI | Intestine | CAGATCTCAGCCCGTCTTCC (237–256) | CCAGAATGCCACCGGTAACT (519–500) | 282 | 2246388 |
| Na + K + ATPase | Intestine | CTGAGGGCAACGAAACAGTG (104–123) | ATCCCTCGGGTTGACCTCC (177–159) | 74 | 14330321 |
| SGLT-1 | Intestine | GTGGAATGCCTTGGAGGGTA (3–22) | GCTTCCTCAGATACTCCGGC (123–104) | 121 | 8346783 |
| MT4 | Intestine | ACCCGAACTGAACCATGGAC (36–55) | TTTTCGTGGTCCCTGTCACC (312–293) | 277 | 46048710 |
| Δ6 desaturase | Liver | ACATGAACAGAGGAAGCGGG (780–799) | TCTGGATCTCCTCCCAGGTG (1754–1735) | 975 | 261865208 |
| DMT-1 | Intestine | TTCCTCCTCAACAACGTCGG (1755–1774) | TCCCAATGCCATCCCAGTTC (1908–1889) | 154 | 206597489 |
| 18S rRNA | Intestine, Liver | CGATGCTCTTAACTGAGT (1251–1269) | CAGCTTTGCAACCATACTC (1550–1531) | 300 | 7262899 |
2.6. Determination of Phytic Acid Concentration in the Diet Samples
2.7. Statistical Analysis
3. Results
| Treatment | Day 0 | Day 7 | Day 14 | Day 21 | Day 28 |
|---|---|---|---|---|---|
| Body weight (g) | |||||
| Zn(+) | 39.5 a ± 1.1 | 83.3 a ± 2.7 | 154.6 a ± 8.9 | 293.1 a ± 24.9 | 482.5 a ± 44.2 |
| Zn(−) | 38.8 a ± 1.2 | 72.1 b ± 2.7 | 106.9 b ± 5.3 | 132.8 b ± 6.6 | 147.7 b ± 14.4 |
| Feed consumption (kg/day) ** | |||||
| Zn(+) | − | 0.152 a ± 0.007 | 0.194 a ± 0.009 | 0.253 a ± 0.013 | 0.329 a ± 0.016 |
| Zn(−) | − | 0.131 b ± 0.007 | 0.143 b ± 0.007 | 0.130 b ± 0.007 | 0.122 b ± 0.006 |
| Zinc intake (g) *** | |||||
| Zn(+) | − | 7.4 a ± 0.5 | 19.1 a ± 0.95 | 37.2 a ± 1.9 | 64.6 a ± 3.2 |
| Zn(−) | − | 0.40 b ± 0.03 | 0.84 b ± 0.04 | 1.1 b ± 0.06 | 1.4 b ± 0.07 |
| Serum zinc concentration (μg/g) | |||||
| Zn(+) | − | 3.42 a ± 0.19 | 4.66 a ± 0.35 | 4.73 a ± 0.25 | 4.49 a ± 0.23 |
| Zn(−) | − | 2.87 b ± 0.17 | 3.59 b ± 0.21 | 3.45 b ± 0.11 | 3.39 b ± 0.11 |
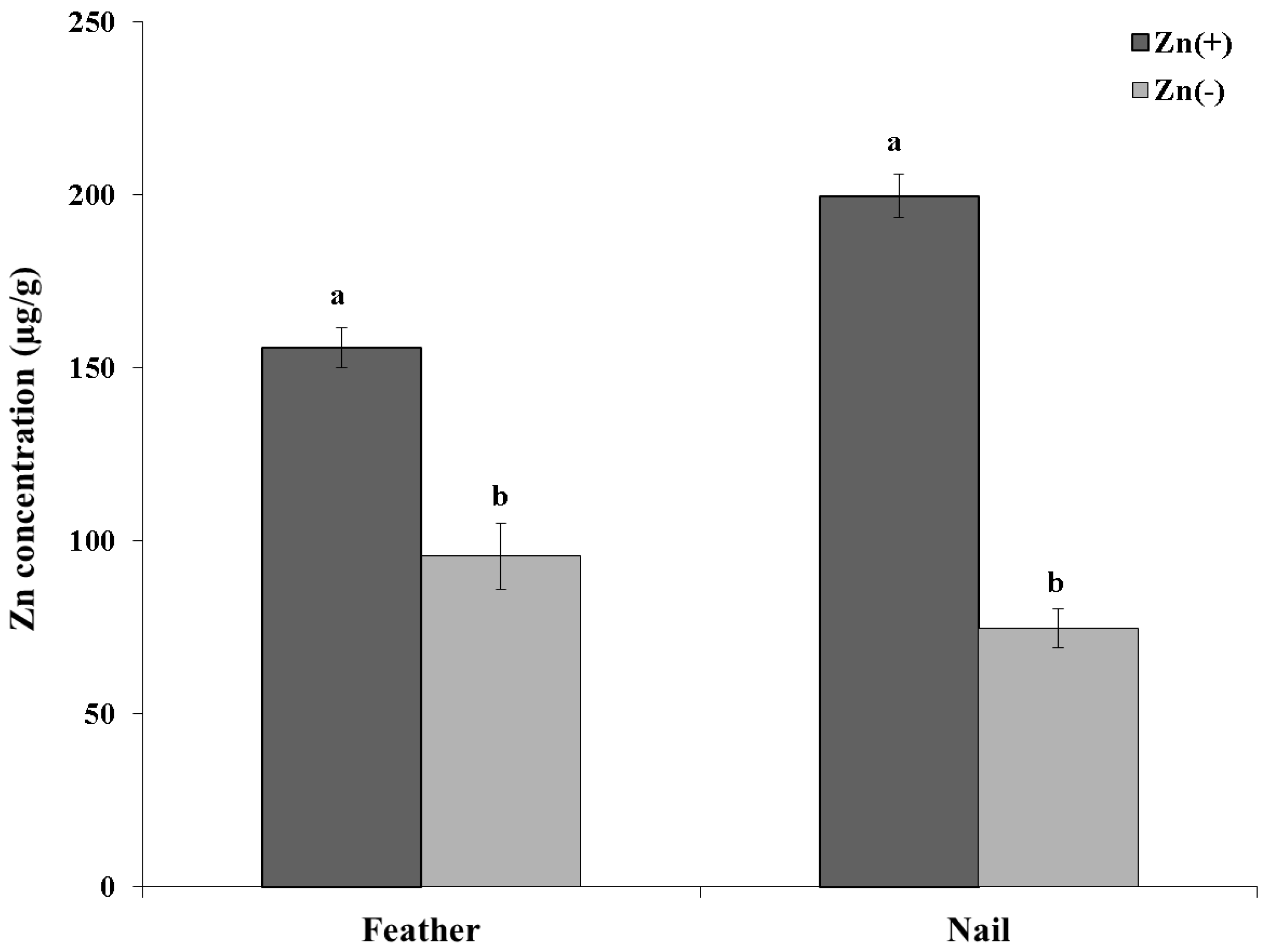

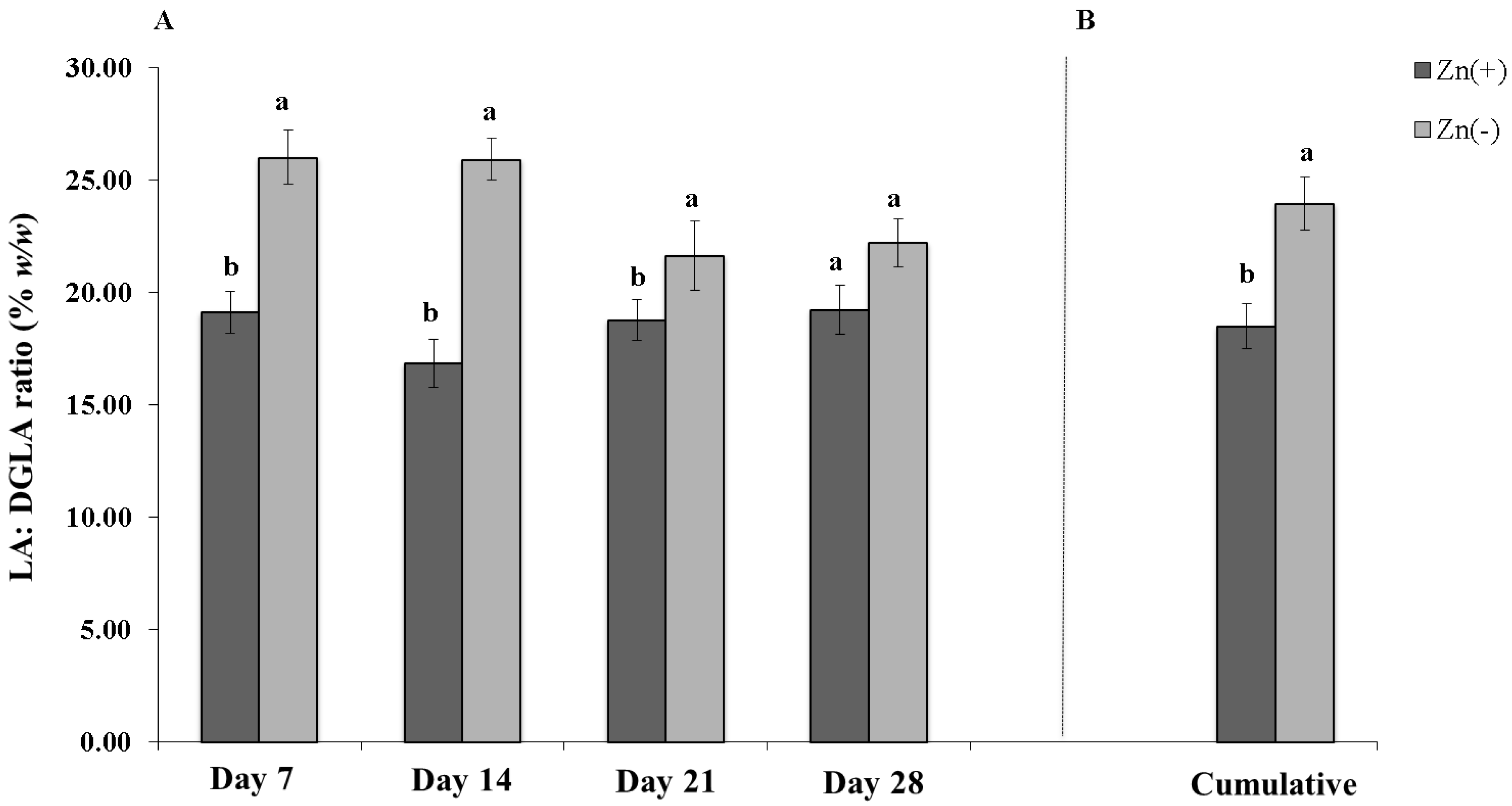
4. Discussion
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Brown, K.H.; Peerson, J.M.; Rivera, J.; Allen, L.H. Effect of supplemental zinc on the growth and serum zinc concentrations of prepubertal children: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2002, 75, 1062–1071. [Google Scholar]
- King, J.C.; Cousins, R.J. Zinc. In Modern Nutrition in Health and Disease, 11th ed.; Shils, M.E., Shike, M., Ross, A.C., Caballero, B., Cousins, R.J., Eds.; Lippincott Williams & Wilkins Press: Philadelphia, PA, USA, 2006; pp. 271–528. [Google Scholar]
- McBean, L.D.; Dove, J.T.; Halstead, J.A.; Smith, J.C., Jr. Zinc concentration in human tissues. Am. J. Clin. Nutr. 1972, 25, 672–676. [Google Scholar]
- Gaither, L.A.; Eide, D.J. Eukaryotic zinc transporters and their regulation. Biometals 2001, 14, 251–270. [Google Scholar] [CrossRef]
- Sharif, R.; Thomas, P.; Zalewski, P.; Fenech, M. The role of zinc in genomic stability. Mutat. Res. 2012, 733, 111–121. [Google Scholar] [CrossRef]
- Johnston, M. Genetic evidence that zinc is an essential co-factor in the DNA binding domain of GAL4 protein. Nature 1987, 328, 353–355. [Google Scholar] [CrossRef]
- Haase, H.; Rink, L. Signal transduction in monocytes: The role of zinc ions. Biometals 2007, 20, 579–585. [Google Scholar] [CrossRef]
- Prasad, A.S. Impact of the discovery of human zinc deficiency on health. J. Am. Coll. Nutr. 2009, 28, 257–265. [Google Scholar] [CrossRef]
- Beyersmann, D.; Haase, H. Functions of zinc in signaling, proliferation and differentiation of mammalian cells. Biometals 2001, 14, 331–141. [Google Scholar] [CrossRef]
- Kruse-Jarres, J.D. The significance of zinc for humoral and cellular immunity. J. Trace Elem. Electrolytes Health Dis. 1989, 3, 1–8. [Google Scholar]
- Ibs, K.H.; Rink, L. Zinc-altered immune function. J. Nutr. 2003, 133, S1452–S1456. [Google Scholar]
- Dardenne, M. Zinc and immune function. Eur. J. Clin. Nutr. 2002, 56, S20–S23. [Google Scholar] [CrossRef]
- Fraker, P.J.; King, L.E.; Laakko, T.; Vollmer, T.L. The dynamic link between the integrity of the immune system and zinc status. Am. Soc. Nutr. Sci. 2000, 130, S1399–S1406. [Google Scholar]
- Fukada, T.; Kambe, T. Molecular and genetic features of zinc transporters in physiology and Pathogenesis. Metallomics 2001, 3, 662–674. [Google Scholar] [CrossRef]
- Weaver, B.P.; Dufner-Beattie, J.; Kambe, T.; Andrews, G.K. Novel zinc-responsive post-transcriptional mechanisms reciprocally regulate expression of the mouse Slc39a4 and Slc39a5 zinc transporters (Zip4 and Zip5). Biol. Chem. 2007, 388, 1301–1312. [Google Scholar]
- Sadhu, C.; Gedamu, L. Metal-specific posttranscriptional control of human metallothionein genes. Mol. Cell. Biol. 1989, 9, 5738–5741. [Google Scholar]
- Cousins, R.J.; Liuzzi, J.P.; Lichten, L.A. Mammalian zinc transport, trafficking, and signals. J. Biol. Chem. 2006, 281, 24085–24089. [Google Scholar] [CrossRef]
- Liuzzi, J.P.; Cousins, R.J. Mammalian zinc transporters. Annu. Rev. Nutr. 2004, 24, 151–172. [Google Scholar] [CrossRef]
- Tako, E.; Ferket, P.; Uni, Z. Changes in chicken intestinal zinc exporter mRNA expression and small intestinal functionality following intra-amniotic zinc-methionine administration. J. Nutr. Biochem. 2005, 16, 339–346. [Google Scholar] [CrossRef]
- Eide, D.J. The SLC39 family of metal ion transporters. Pflug. Arch. 2004, 447, 796–800. [Google Scholar]
- Palmiter, R.D.; Huang, L. Efflux and compartmentalization of zinc by members of the SLC30 family of solute carriers. Pflug. Arch. 2004, 447, 744–751. [Google Scholar]
- Lowe, N.M.; Fekete, K.; Decsi, T. Methods of assessment of zinc status in humans: A systematic review. Am. J. Clin. Nutr. 2009, 89, S2040–S2051. [Google Scholar] [CrossRef]
- Cousins, R.J. Zinc. In Present Knowledge in Nutrition, 7th ed.; Filer, L.J., Zieglerm, E.E., Eds.; International Life Sciences Institute—Nutrition Foundation Press: Washington, DC, USA, 1996; pp. 293–306. [Google Scholar]
- Young, V.R.; Erdman, J.W.; Standing Committee on the Scientific Evaluation of Dietary Reference Intakes; Institute of Medicine (US). Zinc. In Dietary Reference Intakes for Vitamin A, Vitamin K, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc; National Academy Press: Washington, DC, USA, 2000; pp. 442–501. [Google Scholar]
- Thomas, E.A.; Bailey, L.B.; Kauwell, G.A.; Lee, D.Y.; Cousins, R.J. Erythrocyte metallothionein response to dietary zinc in humans. J. Nutr. 1992, 122, 2408–2414. [Google Scholar]
- Baer, M.T.; King, J.C. Tissue zinc levels and zinc excretion during experimental zinc depletion in young men. Am. J. Clin. Nutr. 1984, 39, 556–570. [Google Scholar]
- Ruz, M.; Cavan, K.R.; Bettger, W.J.; Gibson, R.S. Erythrocytes, erythrocyte membranes, neutrophils and platelets as biopsy materials for the assessment of zinc status in humans. Br. J. Nutr. 1992, 68, 515–527. [Google Scholar]
- Wang, X.B.; Fosmire, G.J.; Gay, C.V.; Leach, R.M., Jr. Short-term zinc deficiency inhibits chondrocyte proliferation and induces cell apoptosis in the epiphyseal growth plate of young chickens. J. Nutr. 2002, 132, 665–673. [Google Scholar]
- Burrell, A.L.; Dozier, W.A., III; Davis, A.J.; Compton, M.N.; Freeman, M.E.; Vendrell, P.F.; Ward, T.L. Responses of broilers to dietary zinc concentrations and sources in relation to environmental implications. Br. Poult. Sci. 2004, 45, 255–263. [Google Scholar]
- Bao, Y.M.; Choct, M.; Iji, P.A.; Bruerton, K. Trace mineral interactions in broiler chicken diets. Br. Poult. Sci. 2010, 51, 109–117. [Google Scholar] [CrossRef]
- Cui, H.; Xi, P.; Junliang, D.; Debing, L.; Guang, Y. Pathology of lymphoid organs in chickens fed a diet deficient in zinc. Avian Pathol. 2004, 33, 519–524. [Google Scholar] [CrossRef]
- Tako, E.; Rutzke, M.A.; Glahn, R. Using the domestic chicken (Gallus gallus) as an in vivo model for iron bioavailability. Poult. Sci. 2010, 89, 514–521. [Google Scholar] [CrossRef]
- Tako, E.; Glahn, R.P. White beans provide more bioavailable iron than red beans: Studies in poultry (Gallus gallus) and an in vitro digestion/Caco-2 model. Int. J. Vitam. Nutr. Res. 2010, 80, 416–429. [Google Scholar] [CrossRef]
- Tako, E.; Blair, M.; Glahn, R.P. Biofortified red mottled beans (Phaseolus vulgaris L.) in a maize and bean diet provide more bioavailable iron than standard red mottled beans: Studies in poultry (Gallus gallus) and an in vitro digestion/Caco-2 model. Nutr. J. 2011, 10, 113. [Google Scholar] [CrossRef]
- Tako, E.; Hoekenga, O.; Kochian, L.V.; Glahn, R.P. High bioavailability iron maize (Zea mays L.) developed through molecular breeding provides more absorbable iron in vitro (Caco-2 model) and in vivo (Gallus gallus). Nutr. J 2013, 12, 3. [Google Scholar] [CrossRef]
- Hulbert, A.J. Explaining longevity of different animals: Is membrane fatty acid composition the missing link? Age 2008, 30, 89–97. [Google Scholar] [CrossRef]
- Huang, Y.S.; Cunnane, S.C.; Horrobin, D.F.; Davignon, J. Most biological effects of zinc deficiency corrected by gamma-linolenic acid (18:3 omega 6) but not by linoleic acid (18:2 omega 6). Atherosclerosis 1982, 41, 193–207. [Google Scholar] [CrossRef]
- Horrobin, D.F. Loss of delta-6-desaturase activity as a key factor in aging. Med. Hypotheses 1981, 7, 1211–1220. [Google Scholar] [CrossRef]
- Hanson, L.J.; Sorenson, D.K.; Kernkamp, H.C.H. Essential fatty acid deficiency—Its role in parakeratosis. Am. J. Vet. Res. 1958, 18, 921–930. [Google Scholar]
- Bettger, W.J.; Reeves, P.G.; Moscatelli, E.A.; Reynolds, G.; O’Dell, B.L. Interaction of zinc and essential fatty acids in the rat. J. Nutr. 1979, 109, 480–488. [Google Scholar]
- Cunnane, S.C. Differential regulation of essential fatty acid metabolism to the prostaglandins: Possible basis for the interaction of zinc and copper in biological systems. Prog. Lipid Res. 1982, 21, 73–90. [Google Scholar] [CrossRef]
- Maniongui, C.; Blond, J.P.; Ulmann, L.; Durand, G.; Poisson, J.P.; Bézard, J. Age-related changes in Δ5 and Δ6 desaturase activities in rat liver microsomes. Lipids 1993, 28, 291–297. [Google Scholar] [CrossRef]
- Tang, C.; Cho, H.P.; Nakamura, M.T.; Clarke, S.D. Regulation of human delta-6 desaturase gene transcription: Identification of a functional direct repeat-1 element. J. Lipid Res. 2003, 44, 686–695. [Google Scholar] [CrossRef]
- Sell, J.L.; Kratzer, F.H.; Latshaw, J.D.; Subcommittee on Poultry Nutrition; Committee on Animal Nutrition; Board on Agriculture; National Research Council. Nutrient Requirements of Poultry, 9th revised ed.; National Academy Press: Washington, DC, USA, 1994. [Google Scholar]
- Mahler, G.J.; Esch, M.B.; Tako, E.; Southard, T.L.; Archer, S.D.; Glahn, R.P.; Shuler, M.L. Oral exposure to polystyrene nanoparticles affects iron absorption. Nat. Nanotechnol. 2012, 7, 264–271. [Google Scholar] [CrossRef]
- Tako, E.; Glahn, R.P. Iron status of the late term broiler (Gallus gallus) embryo and hatchling. Int. J. Poult. Sci. 2011, 10, 42–48. [Google Scholar] [CrossRef]
- Tako, E.; Glahn, R.P.; Laparra, J.M.; Welch, R.M.; Lei, X.; Kelly, J.D.; Rutzke, M.A.; Miller, D.D. Iron and zinc bioavailabilities to pigs from red and white beans (Phaseolus vulgaris L.) are similar. J. Agric. Food Chem. 2009, 57, 3134–3140. [Google Scholar]
- Tako, E.; Laparra, J.M.; Glahn, R.P.; Welch, R.M.; Lei, X.; Beebee, S.; Miller, D.D. Biofortified black beans in a maize and bean diet provide more bioavailable iron to piglets than standard black beans. J. Nutr. 2009, 139, 305–309. [Google Scholar]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef]
- Hess, S.Y.; Peerson, J.M.; King, J.C.; Brown, K.H. Use of serum zinc concentration as an indicator of population zinc status. Food Nutr. Bull. 2007, 28, S403–S429. [Google Scholar]
- Swanson, C.A.; Mansourian, R.; Dirren, H.; Rapin, C.H. Zinc status of healthy elderly adults: Response to supplementation. Am. J. Clin. Nutr. 1998, 48, 343–349. [Google Scholar]
- Chiplonkar, S.; Khadilkar, A.; Pandit-Agrawal, D.; Kawade, R.; Kadam, N.; Ekbote, V.; Sanwalka, N.; Khadilkar, V. Influence of micronutrient status and socioeconomic gradient on growth indices of 2–18-year-old Indian girls. J. Pediatr. Endocrinol. Metab. 2013, 26, 825–832. [Google Scholar]
- Gaetke, L.M.; McClain, C.J.; Talwalkar, R.T.; Shedlofsky, S.I. Effects of endotoxin on zinc metabolism in human volunteers. Am. J. Physiol. 1997, 272, E952–E956. [Google Scholar]
- Wieringa, F.T.; Dijkhuizen, M.A.; West, C.E.; van der Ven-Jongekrjg, J.; van der Meer, J.W.M. Reduced production of immunoregulatory cytokines in vitamin A- and zinc-deficient Indonesian infants. Eur. J. Clin. Nutr. 2004, 58, 1498–1504. [Google Scholar] [CrossRef]
- Koletzko, B.; Abiodun, P.O.; Laryea, M.D.; Bremer, H.J. Fatty acid composition of plasma lipids in Nigerian children with protein-energy malnutrition. Eur. J. Pediatr. 1986, 145, 109–115. [Google Scholar] [CrossRef]
- Ayala, S.; Brenner, R.R. Essential fatty acid status in zinc deficiency, effect on lipid and fatty acid composition, desaturation activity and structure of microsomal membranes of rat liver and testes. Acta Physiol. Lat. Am. 1983, 33, 193–204. [Google Scholar]
- Hennig, B.; Meerarani, P.; Toborek, M.; McClain, C.J. Antioxidant-like properties of zinc in activated endothelial cells. J. Am. Coll. Nutr. 1999, 18, 152–158. [Google Scholar] [CrossRef]
- Beck, F.W.J.; Prasad, A.S.; Kaplan, J.; Fitzgerlad, J.T.; Brewer, G.J. Changes in cytokine production and T cell subpopulations in experimentally induced zinc-deficient humans. Am. J. Physiol. 1997, 272, E1002–E1007. [Google Scholar]
- Allan, A.K.; Hawksworth, H.M.; Woodhouse, L.R.; Sutherland, B.; King, J.C.; Beattie, J.H. Lymphocyte metallothionein mRNA responds to marginal zinc intake in human volunteers. Br. J. Nutr. 2000, 84, 747–756. [Google Scholar]
- Sullivan, V.K.; Burnett, F.R.; Cousins, R.J. Metallothionein expression is increase in monocytes and erythrocytes of young men during zinc supplementation. J. Nutr. 2000, 128, 707–713. [Google Scholar]
- Andree, K.B.; Kim, J.; Kirschke, C.P.; Gregg, J.P.; Paik, H.; Joung, H.; Woodhouse, L.; King, J.C.; Huang, L. Investigation of lymphocyte gene expression for use as biomarkers for zinc status in humans. J. Nutr. 2004, 134, 1716–1723. [Google Scholar]
- Maret, W.; Sandstead, H.H. Zinc requirements and the risks and benefits of zinc supplementation. J. Trace Elem. Med. Biol. 2006, 20, 3–18. [Google Scholar] [CrossRef]
- Gibson, R.S.; Hess, S.Y.; Hotz, C.; Brown, K.H. Indicators of zinc status at the population level: A review of the evidence. Br. J. Nutr. 2008, 99, S14–S23. [Google Scholar]
- Hambidge, M. Biomarkers of trace mineral intake and status. J. Nutr. 2003, 133, 948S–955S. [Google Scholar]
- Sandstead, H.H. Zinc deficiency. A public health problem? Am. J. Dis. Child. 1991, 145, 853–859. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Reed, S.; Qin, X.; Ran-Ressler, R.; Brenna, J.T.; Glahn, R.P.; Tako, E. Dietary Zinc Deficiency Affects Blood Linoleic Acid: Dihomo-γ-linolenic Acid (LA:DGLA) Ratio; a Sensitive Physiological Marker of Zinc Status in Vivo (Gallus gallus). Nutrients 2014, 6, 1164-1180. https://doi.org/10.3390/nu6031164
Reed S, Qin X, Ran-Ressler R, Brenna JT, Glahn RP, Tako E. Dietary Zinc Deficiency Affects Blood Linoleic Acid: Dihomo-γ-linolenic Acid (LA:DGLA) Ratio; a Sensitive Physiological Marker of Zinc Status in Vivo (Gallus gallus). Nutrients. 2014; 6(3):1164-1180. https://doi.org/10.3390/nu6031164
Chicago/Turabian StyleReed, Spenser, Xia Qin, Rinat Ran-Ressler, James Thomas Brenna, Raymond P. Glahn, and Elad Tako. 2014. "Dietary Zinc Deficiency Affects Blood Linoleic Acid: Dihomo-γ-linolenic Acid (LA:DGLA) Ratio; a Sensitive Physiological Marker of Zinc Status in Vivo (Gallus gallus)" Nutrients 6, no. 3: 1164-1180. https://doi.org/10.3390/nu6031164





