Application of the Hyperspectral Imager for the Coastal Ocean to Phytoplankton Ecology Studies in Monterey Bay, CA, USA
Abstract
:1. Introduction
2. Materials and Methods
2.1. HICO Image Acquisition and Processing
2.2. Algorithms for Phytoplankton Characterization
2.3. Satellite Observations
2.4. In Situ Observations
3. Results and Discussion
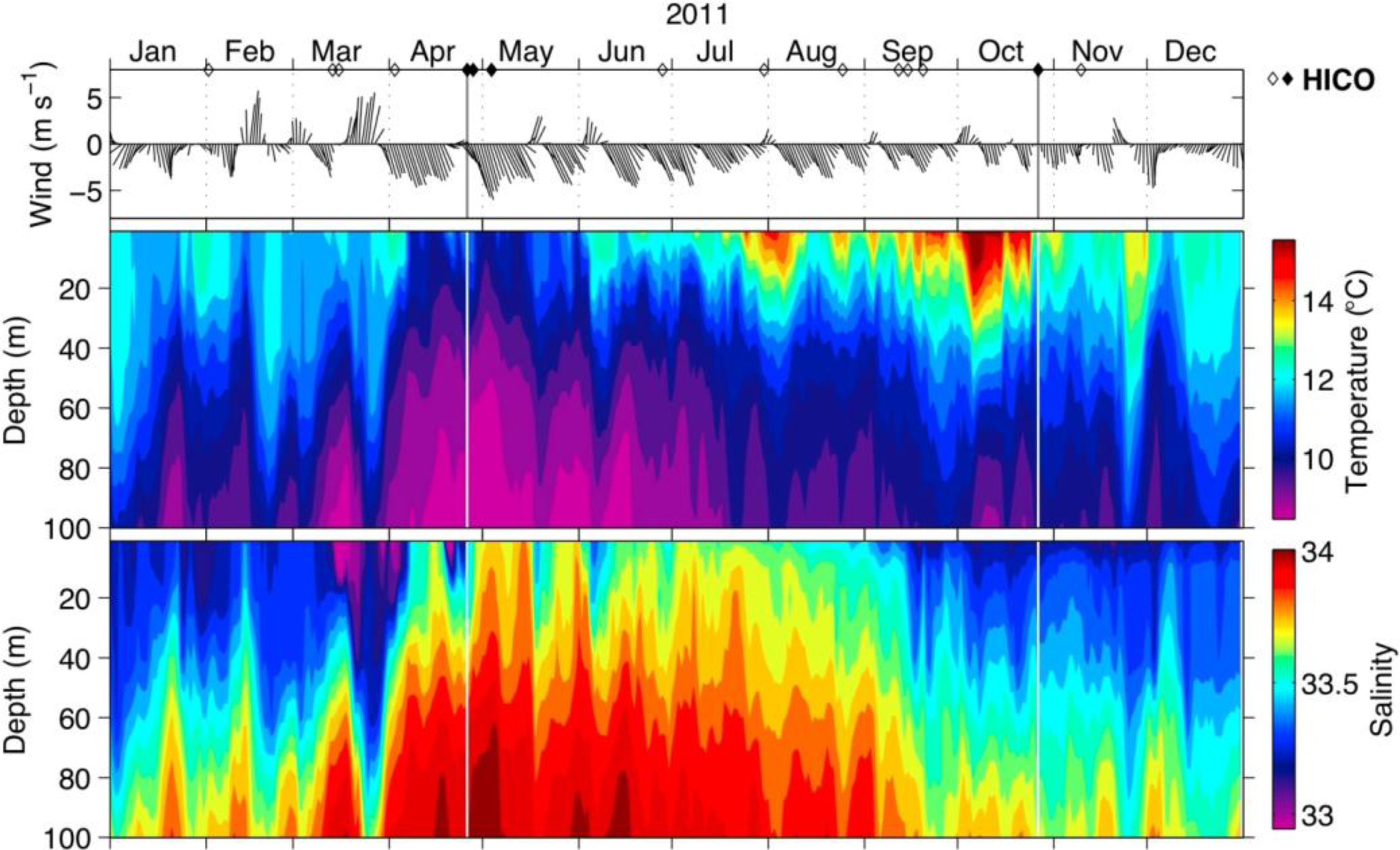
3.1. Environmental Overview for 2011
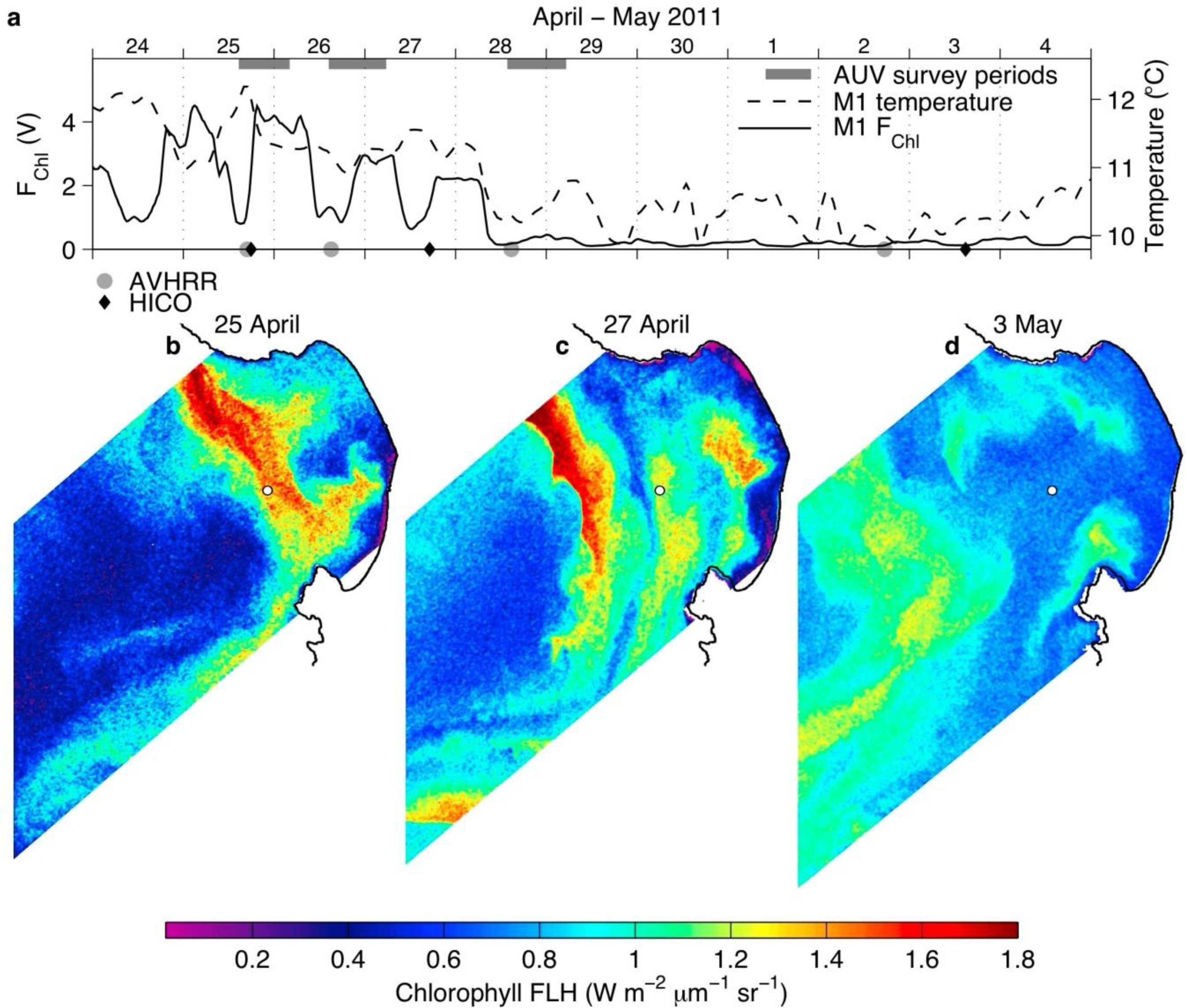
3.2. Spring 2011 Study
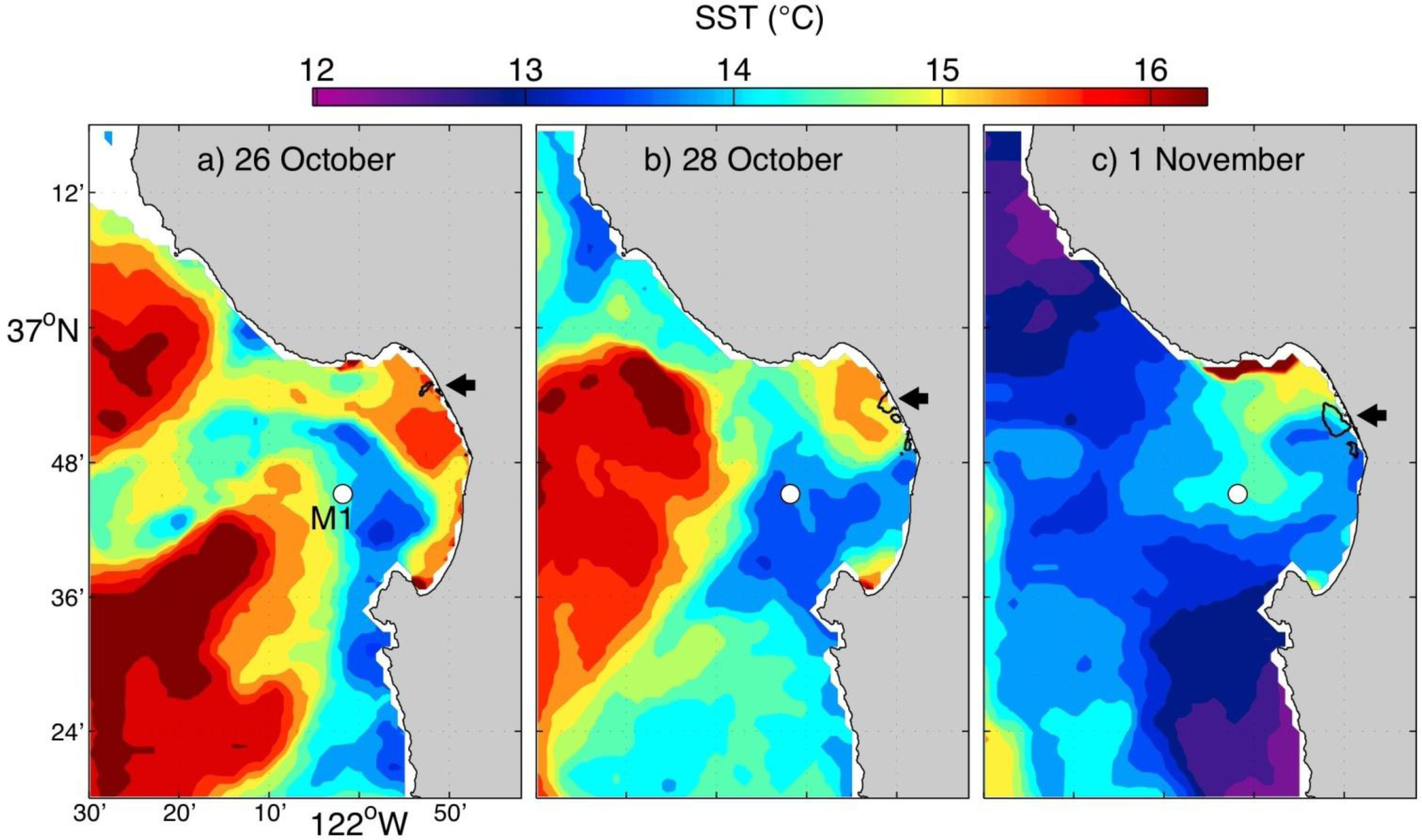
3.3. Fall 2011 Study
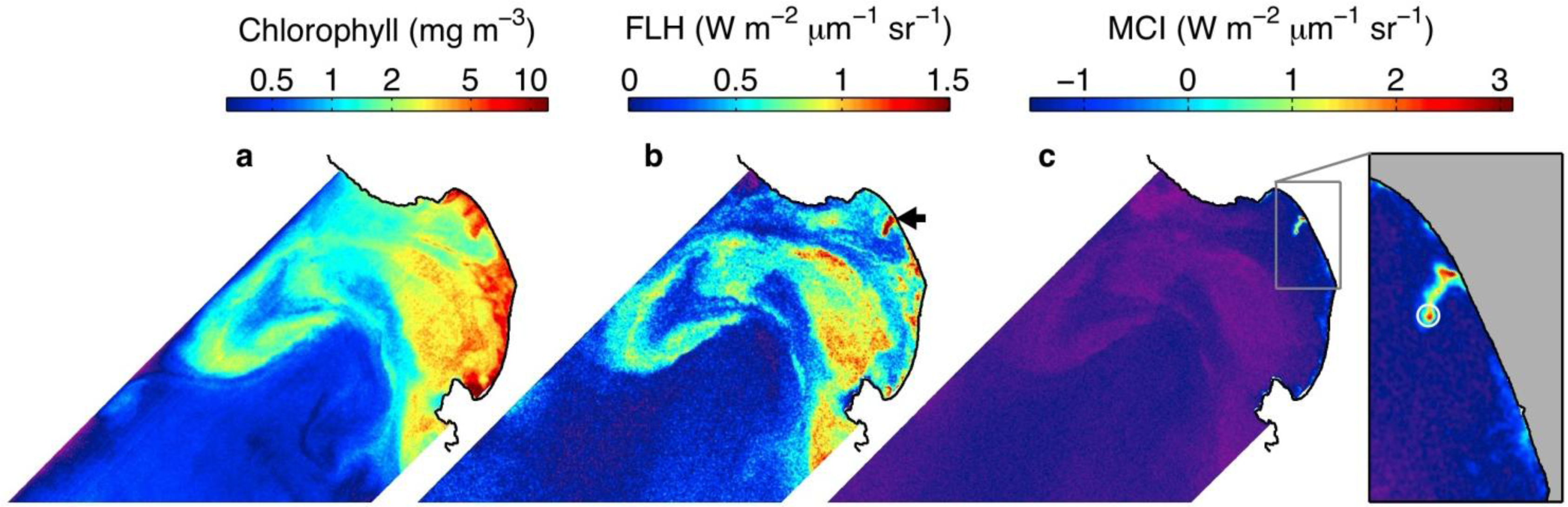
3.4. Examination of Algorithms for Characterization of Phytoplankton
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lucke, R.L.; Corson, M.; McGlothlin, N.R.; Butcher, S.D.; Wood, D.L.; Korwan, D.R.; Li, R.R.; Snyder, W.A.; Davis, C.O.; Chen, D.T. Hyperspectral imager for the coastal ocean: Instrument description and first images. Appl. Opt 2011, 50, 1501–1516. [Google Scholar]
- Corson, M.R.; Davis, C.O. A new view of coastal oceans from the space station. Eos Trans. Am. Geophys. Union 2011, 92, 161–162. [Google Scholar]
- Davis, C.O.; Bowles, J.; Leathers, R.A.; Korwan, D.; Downes, T.V.; Snyder, W.A.; Rhea, W.J.; Chen, W.; Fisher, J.; Bissett, W.P.; et al. Ocean PHILLS hyperspectral imager: Design, characterization, and calibration. Opt. Express 2002, 10, 210–221. [Google Scholar]
- Barber, R.T.; Smith, R.L. Coastal Upwelling Ecosystems. In Analysis of Marine Ecosystems; Longhurst, A.R., Ed.; Academic Press: New York, NY, USA, 1981; pp. 31–68. [Google Scholar]
- Breaker, L.C.; Broenkow, W.W. The circulation of Monterey Bay and related processes. Oceanogr. Mar. Biol. Annu. Rev 1994, 32, 1–64. [Google Scholar]
- Rosenfeld, L.K.; Schwing, F.B.; Garfield, N.; Tracy, D.E. Bifurcated flow from an upwelling center: A cold water source for Monterey Bay. Cont. Shelf Res 1994, 14, 931–964. [Google Scholar]
- Woodson, C.B.; Eerkes-Medrano, D.I.; Flores-Morales, A.; Foley, M.M.; Henkel, S.K.; Hessing-Lewis, M.; Jacinto, D.; Needles, L.; Nishizaki, M.T.; O’Leary, J.; et al. Local diurnal upwelling driven by sea breezes in northern Monterey Bay. Cont. Shelf Res 2007, 27, 2289–2302. [Google Scholar]
- Shea, R.E.; Broenkow, W.W. The role of internal tides in the nutrient enrichment of Monterey Bay, California. Estuar. Coast. Shelf Sci 1982, 15, 57–66. [Google Scholar]
- Ryan, J.P.; McManus, M.A.; Sullivan, J.M. Interacting physical, chemical and biological forcing of phytoplankton thin-layer variability in Monterey Bay, California. Cont. Shelf Res 2010, 30, 7–16. [Google Scholar]
- Kudela, R.; Pitcher, G.; Probyn, T.; Figueiras, F.; Moita, T.; Trainer, V. Harmful algal blooms in coastal upwelling systems. Oceanography 2005, 18, 184–197. [Google Scholar]
- Ryan, J.; Greenfield, D.; Marin, R., III; Preston, C.; Roman, B.; Jensen, S.; Pargett, D.; Birch, J.; Mikulski, C.; Doucette, G.; et al. Harmful phytoplankton ecology studies using an autonomous molecular analytical and ocean observing network. Limnol. Oceanogr 2011, 56, 1255–1272. [Google Scholar]
- Jessup, D.A.; Miller, M.A.; Ryan, J.P.; Nevins, H.M.; Kerkering, H.A.; Mekebri, A.; Crane, D.B.; Johnson, T.A.; Kudela, R.M. Mass stranding of marine birds caused by a surfactant-producing red tide. PLoS ONE 2009. [Google Scholar] [CrossRef]
- Kudela, R.M.; Lane, J.; Cochlan, W. The potential role of anthropogenically derived nitrogen in the growth of harmful algae in California, USA. Harmful Algae 2008, 8, 103–110. [Google Scholar]
- Kudela, R.M. University of California, Santa Cruz, CA, USA; Ryan, J.P. Monterey Bay Aquarium Research Institute, Moss Landing, CA, USA. Unpublished data, 2007–2009.
- HICO. Available online: http://hico.coas.oregonstate.edu (accessed on 1 November 2013).
- Gao, B.-C.; Davis, C.O. Development of a line-by-line-based atmosphere removal algorithm for airborne and spaceborne imaging spectrometers. Proc. SPIE 1997. [Google Scholar] [CrossRef]
- Montes, M.J.; Gao, B.C. NRL Atmospheric Correction Algorithms for Oceans: Tafkaa User’s Guide; NRL Report; NRL: Washington, DC, USA, 2004; pp. 1–39. [Google Scholar]
- Bowles, J. The US Naval Research Laboratory: Washington, DC, USA, Private Communication; 2013.
- O’Reilly, J.E.; Maritorena, S.; Siegel, D.; O’Brien, M.C.; Toole, D.; Mitchell, B.G.; Kahru, M.; Chavez, F.P.; Strutton, P.; Cota, G.; et al. Ocean Color Chlorophyll A Algorithms for SeaWiFS, OC2, and OC4: Version 4. In SeaWiFS Postlaunch Calibration and Validation Analyses, Part 3; Hooker, S.B., Firestone, E.R., Eds.; NASA Goddard Space Flight Center: Greenbelt, MD, USA, 2000; Volume 11, pp. 9–23. [Google Scholar]
- Lee, Z.P.; Carder, K.L.; Arnone, R.A. Deriving inherent optical properties from water color: A multiband quasi-analytical algorithim for optically deep waters. Appl. Opt 2002, 41, 5755–5756. [Google Scholar]
- Lee, Z.P.; Carder, K.L. Absorption spectrum of phytoplankton pigments derived from hyperspectral remote-sensing reflectance. Remote Sens. Environ 2004, 89, 361–368. [Google Scholar]
- IOCCG. Available online: http://www.ioccg.org/groups/software.html (accessed on 1 November 2013).
- Letelier, R.M.; Abbott, M.R. An analysis of chlorophyll fluorescence algorithms for Moderate Resolution Imaging Spectrometer (MODIS). Remote Sens. Environ 1996, 58, 215–223. [Google Scholar]
- Gower, J.; King, S.; Borstad, G.; Brown, L. Detection of intense plankton blooms using the 709 nm band of the MERIS imaging spectrometer. Int. J. Remote Sens 2005, 26, 2005–2012. [Google Scholar]
- Ryan, J.P.; Fischer, A.M.; Kudela, R.M.; McManus, M.A.; Myers, J.S.; Paduan, J.D.; Ruhsam, C.M.; Woodson, C.B.; Zhang, Y. Recurrent frontal slicks of a coastal ocean upwelling shadow. J. Geophys. Res 2010. [Google Scholar] [CrossRef]
- Bellingham, J.G.; Streitlien, K.; Overland, J.; Rajan, S.; Stein, P.; Stannard, J.; Kirkwood, W.; Yoerger, D. An Arctic basin observational capability using AUVs. Oceanography 2000, 13, 64–71. [Google Scholar]
- Johnson, K.S.; Chavez, F.P.; Friederich, G.E. Continental-shelf sediment as a primary source of iron for coastal phytoplankton. Nature 1999, 398, 697–700. [Google Scholar]
- Bruland, K.W.; Rue, E.L.; Smith, G.J. The influence of iron and macronutrients in coastal upwelling regimes off central California: Implications for extensive blooms of large diatoms. Limnol. Oceanogr 2001, 46, 1661–1674. [Google Scholar]
- Rue, E.L.; Bruland, K.W. Domoic acid binds iron and copper: A possible role for the toxin produced by the marine diatom Pseudo-nitzchia. Mar. Chem 2001, 76, 127–134. [Google Scholar]
- Maldonado, M.T.; Hughes, M.P.; Rue, E.L.; Wells, M.L. The effect of Fe and Cu on growth and domoic acid production by Pseudo-nitzschia multiseries and Pseudo-nitzschia australis. Limnol. Oceanogr 2002, 47, 515–526. [Google Scholar]
- Rhodes, L.; Selwood, A.; McNabb, P.; Briggs, L.; Adamson, J.; van Ginkel, R.; Laczka, O. Trace metal effects on the production of biotoxins by microalgae. Afr. J. Mar. Sci 2006, 28, 393–397. [Google Scholar]
- Ryan, J.P.; McManus, M.A.; Kudela, R.M.; Lara Artigas, M.; Bellingham, J.G.; Chavez, F.P.; Doucette, G.; Foley, D.; Godin, M.; Harvey, J.B.J.; et al. Boundary influences on HAB phytoplankton ecology in a stratification-enhanced upwelling shadow. Deep Sea Res. Part II Top. Stud. Oceanogr 2013, in press. [Google Scholar]
- Huot, Y.; Franz, B.A.; Fradette, M. Estimating variability in the quantum yield of Sun-induced chlorophyll fluorescence: A global analysis of oceanic waters. Remote Sens. Environ 2013, 132, 238–253. [Google Scholar]
- Abbott, M.R.; Brink, K.H.; Booth, C.R.; Blasco, D.; Swenson, M.S.; Davis, C.O.; Codispoti, L.A. Scales of variability of bio-optical properties as observed from near-surface drifters. J. Geophys. Res. Ocean 1995, 100, 13345–13367. [Google Scholar]
- Kudela, R.M.; Ryan, J.P.; Blakely, M.D.; Lane, J.Q.; Peterson, T.D. Linking the physiology and ecology of Cochlodinium to better understand harmful algal bloom events: A comparative approach. Harmful Algae 2008, 7, 278–292. [Google Scholar]
- Ryan, J.P.; Gower, J.F.R.; King, S.A.; Bissett, W.P.; Fischer, A.M.; Kudela, R.M.; Kolber, Z.; Mazzillo, F.; Rienecker, E.V.; Chavez, F.P. A coastal ocean extreme bloom incubator. Geophys. Res. Lett 2008. [Google Scholar] [CrossRef]
- Ryan, J.P.; Fischer, A.M.; Kudela, R.M.; Gower, J.F.R.; King, S.A.; Marin, R., III; Chavez, F.P. Influences of upwelling and downwelling winds on red tide bloom dynamics in Monterey Bay, California. Cont. Shelf Res 2009, 29, 785–795. [Google Scholar]
- Wynne, T.T.; Stumpf, R.P.; Tomlinson, M.C.; Warner, R.A.; Tester, P.A.; Dybie, J.; Fahnenstiel, G.L. Relating spectral shape to cyanobacterial blooms in the Laurentian Great Lakes. Int. J. Remote Sens 2008, 29, 3665–3672. [Google Scholar]
- Wynne, T.T.; Stumpf, R.P.; Briggs, T.O. Comparing MODIS and MERIS spectral shapes for cyanobacterial bloom detection. Int. J. Remote Sens 2013, 34, 6668–6678. [Google Scholar]
- Matthews, M.W.; Bernard, S.; Robertson, L. An algorithm for detecting trophic status (chlorophyll-a), cyanobacterial-dominance, surface scums and floating vegetation in inland and coastal waters. Remote Sens. Environ 2012, 124, 637–652. [Google Scholar]
- Tufillaro, N. The Shape of Ocean Color. In Topology and Dynamics of Chaos; Letellier, C., Gilmore, R., Eds.; World Scientific Publishing: Singapore, 2013; pp. 251–268. [Google Scholar]
- Raitsos, D.E.; Lavender, S.J.; Maravelias, C.D.; Haralabous, J.A.; Richardson, J.; Reid, P.C. Identifying four phytoplankton functional types from space: An ecological approach. Limnol. Oceanogr 2008, 53, 605–613. [Google Scholar] [Green Version]
- Sadeghi, A.; Dinter, T.; Vountas, M.; Taylor, B.; Altenburg-Soppa, M.; Bracher, A. Remote sensing of coccolithophore blooms in selected oceanic regions using the PhytoDOAS method applied to hyper-spectral satellite data. Biogeosciences 2012, 9, 2127–2143. [Google Scholar] [Green Version]
- Torrecilla, E.; Stramski, D.; Reynolds, R.A.; Millán-Núñez, E.; Piera, J. Cluster analysis of hyperspectral optical data for discriminating phytoplankton pigment assemblages in the open ocean. Remote Sens. Environ 2011, 115, 2578–2593. [Google Scholar] [Green Version]






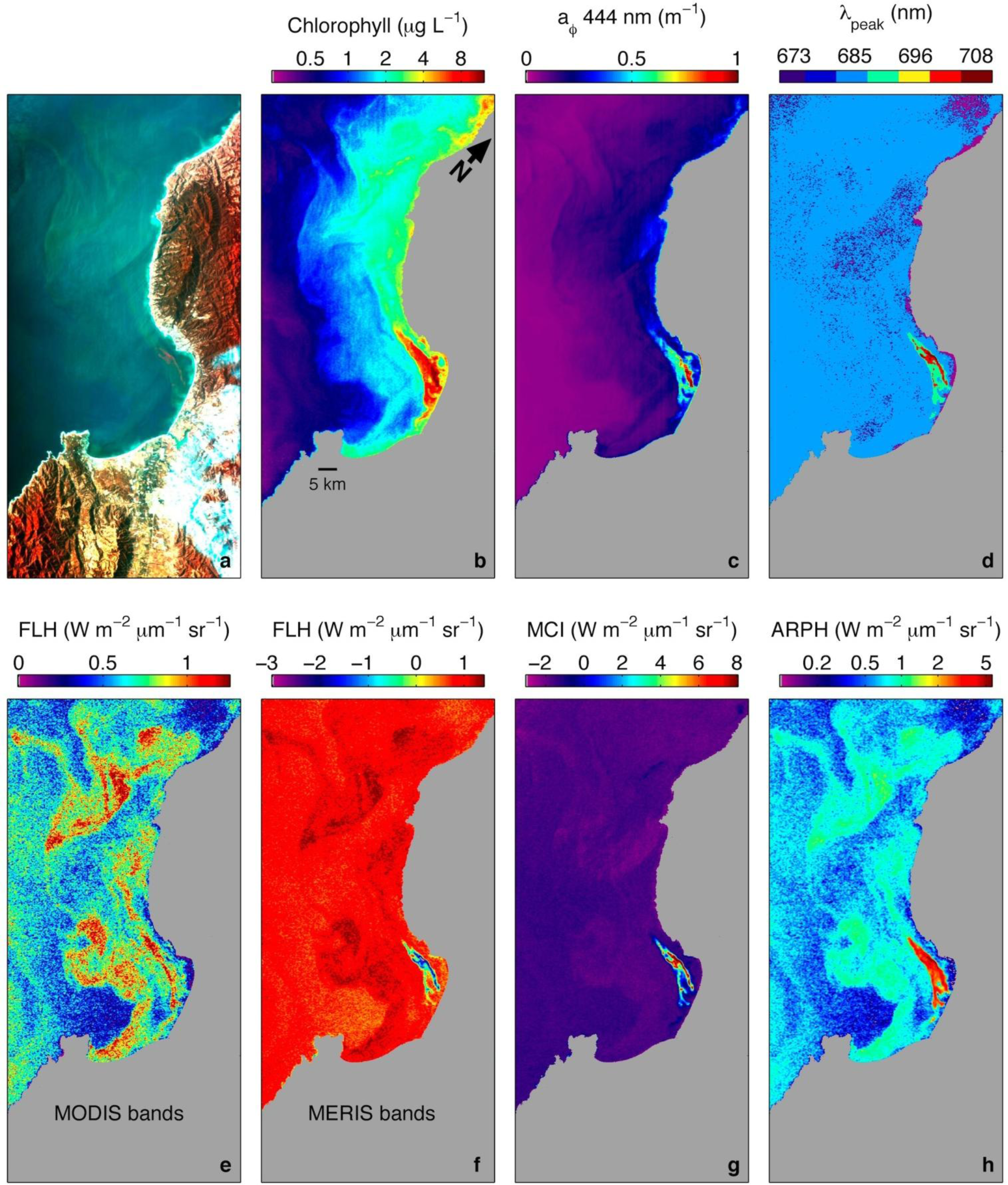
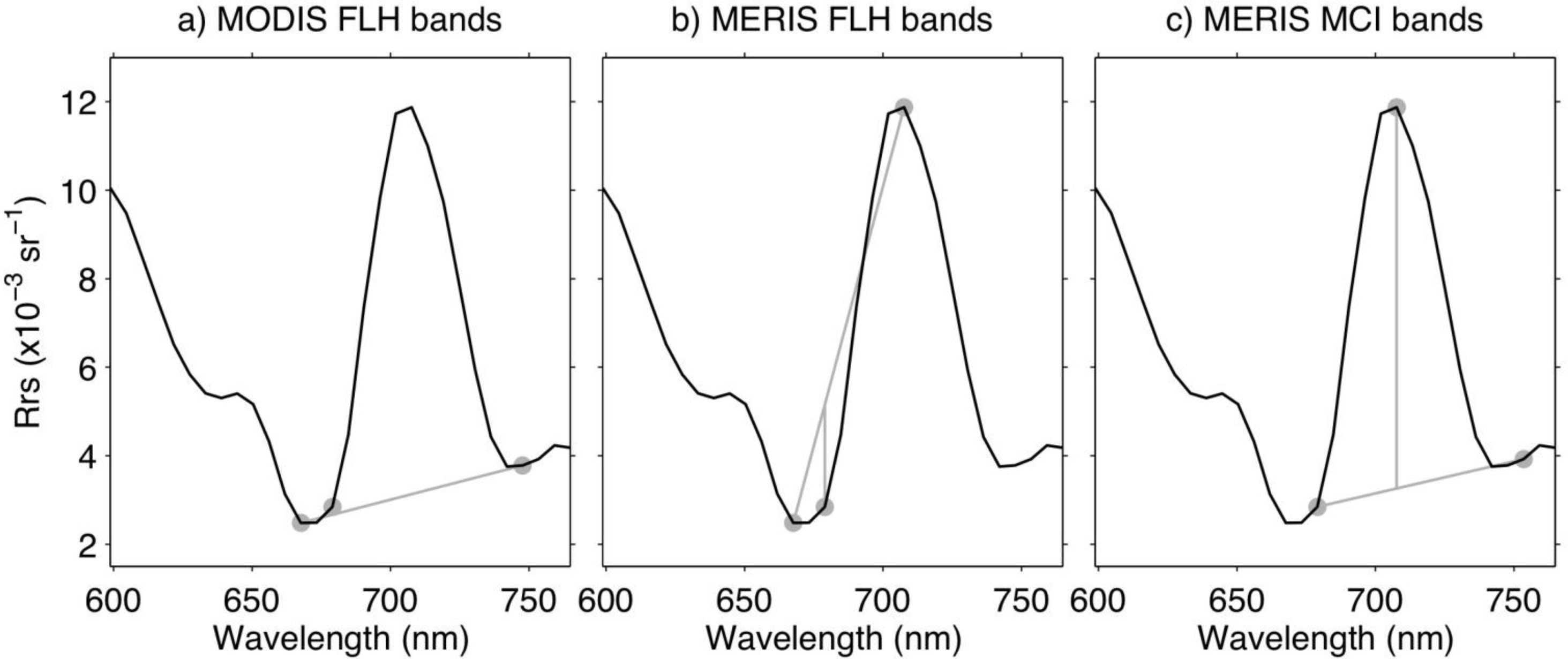
| Chlorophyll | QAA IOP | MODIS FLH | MERIS FLH | MERIS MCI | |
|---|---|---|---|---|---|
| Multispectral bands (nm) | 443, 490, 510, 555 | 411, 443, 490, 555, 667 | 665, 677, 746 | 665, 681, 709 | 681, 709, 754 |
| HICO bands (nm) | 444, 490, 507, 553 | 410, 444, 490, 553, 668 | 668, 679, 748 | 668, 679, 708 | 679, 708, 753 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ryan, J.P.; Davis, C.O.; Tufillaro, N.B.; Kudela, R.M.; Gao, B.-C. Application of the Hyperspectral Imager for the Coastal Ocean to Phytoplankton Ecology Studies in Monterey Bay, CA, USA. Remote Sens. 2014, 6, 1007-1025. https://doi.org/10.3390/rs6021007
Ryan JP, Davis CO, Tufillaro NB, Kudela RM, Gao B-C. Application of the Hyperspectral Imager for the Coastal Ocean to Phytoplankton Ecology Studies in Monterey Bay, CA, USA. Remote Sensing. 2014; 6(2):1007-1025. https://doi.org/10.3390/rs6021007
Chicago/Turabian StyleRyan, John P., Curtiss O. Davis, Nicholas B. Tufillaro, Raphael M. Kudela, and Bo-Cai Gao. 2014. "Application of the Hyperspectral Imager for the Coastal Ocean to Phytoplankton Ecology Studies in Monterey Bay, CA, USA" Remote Sensing 6, no. 2: 1007-1025. https://doi.org/10.3390/rs6021007





